Abstract
Two similar benign, nonneoplastic vascular lesions have been described in the lymph nodes of humans and animals: angiomyomatous hamartoma (AMH), which is characterized by the replacement of lymphoid tissue by blood vessels, smooth muscle, and fibrous tissue, and vascular transformation of sinuses (VTS), which is considered a reactive transformation of lymph node sinuses into capillary-like vascular channels. We hereby report a lesion with features common to both lesions in the mediastinal lymph nodes of a 1-year-old beagle dog in a 1-month toxicity study. Grossly, enlargement and red discoloration were observed, while microscopically, the lesion was characterized by effacement of the lymph node parenchyma with replacement by mature blood vessels, smooth muscle, and fibrous tissue, associated with lymphoid atrophy, which is consistent with AMH. However, multifocal areas of anastomosing or plexiform capillary-like channels lined by normal to slightly plump endothelium, similar to those described for VTS, were also present. Immunohistochemistry analysis revealed abundant positive staining for smooth muscle actin and endothelial cells (von Willebrand factor/factor VIII) and the absence of proliferation (Ki67). In conclusion, these lesions most likely represent a mixture of both AMH and VTS.
Keywords
Two proliferative, nonneoplastic vascular lesions of lymph nodes, angiomyomatous hamartoma (AMH) and vascular transformation of sinuses (VTS), have been described, but uncommonly, in humans 1 –4 and a cynomolgus monkey. 5 Angiomyomatous hamartoma of the lymph node, sometimes referred to as angiomatous hamartoma, is a benign vascular lesion of unknown etiology, which was first described in the human by Chan et al in 1992. 2 It is a locally extensive lesion that is characterized by replacement of the lymph node parenchyma by blood vessels, smooth muscle, and fibrous tissue, occupying approximately 60% of the parenchyma on average. Starting from the hilus, the lesion frequently occupies the center of lymph nodes, extending toward the cortex and subcapsular sinus. In humans, where over 31 cases have been reported, it occurs preferentially in the inguinal or femoral lymph nodes, 3 although other locations, such as lymph nodes of the neck, 6 –9 have also been reported. Until recently, it had not been described in animal species other than humans to the best of our knowledge, but a recent report documented a similar finding in the axillary lymph node of a cynomolgus monkey. 5
Vascular transformation of sinuses, which is sometimes used interchangeably with nodal angiomatosis, 10,11 is another vascular lesion of the lymph nodes believed to be an acquired and reactive conversion of lymph node sinuses into capillary-like vascular channels that arise secondary to occlusion of efferent lymphatics and/or venous blood to the lymph nodes and start in the subcapsular and intermediate sinuses. 4,11 –13 It is characterized by the combination of focal lymphoid atrophy and the replacement of subcapsular and medullary sinuses by a complex network of capillary-like channels, typically with an anastomosing or plexiform pattern (although other patterns have been described), as well as fibrosis. 6,8,11,13 The term VTS was first used by Haferkamp et al 12 to describe a distinctive benign vasoproliferative lesion of lymph nodes in humans that needed to be considered as an important differential diagnosis for Kaposi sarcoma. Since then, several other cases of VTS have been described in humans 4,6,12 and in other animals such as dogs, 11 , cats, 8,9 and rabbits, 13 where the lesion was experimentally induced by the occlusion of efferent lymphatics. 13 Although described in humans as mainly affecting the intra-abdominal lymph nodes, 4,6,14 lesions in other lymph nodes such as the cervical, 6 –9 mediastinal, 10,13,14 or inguinal 14 lymph nodes have been described in both humans and animals.
Besides these 2 lesions, other nonneoplastic vascular proliferative lesions of lymph nodes that are considered to represent similar or variants of VTS include (lymph)angiomatosis or nodal angiomatosis in humans, 6,10 plexiform vasculopathy of lymph nodes in cats, 8,9,15 and angiomatous hyperplasia in mainly the mesenteric lymph nodes of Wistar rats. 16 In addition, lesions with features consistent with both AMH and VTS have been reported in humans, 7,17 and a causal association between the 2 has been suggested, whereby a lymphovenous congestion and distention caused by the hamartoma was described as the cause of VTS. 3,7,17
In humans where the majority of these 2 benign lesions have been reported, they commonly occur as incidental findings in lymph nodes excised during surgical procedures for various reasons such as cancer staging or removal and rarely present as lymphadenopathy. 4,6,18
We hereby present a case of a nonneoplastic vascular proliferative lesion in the mediastinal lymph node of a 1-year-old female beagle dog, with an unusual morphological presentation of both AMH and some features consistent with VTS in humans and dogs and plexiform vasculopathy in the cat. To the best of our knowledge, this is the first report of an AMH in the dog, with or without accompanying features of VTS. In addition, apart from reports in older domestic animals, to the best of our knowledge, there are no published reports on proliferative nonneoplastic vascular changes in the lymph nodes of experimental dogs used in toxicity studies. Therefore, the objective of this article is to report, document, and discuss a case of AMH with features compatible with VTS or plexiform vasculopathy in a beagle dog, with a focus on the differential diagnosis for a localized mass of vascular origin in the lymph nodes of dogs used on toxicity studies and the possible implications of the diagnosis on study interpretation. The comparative histomorphological presentation and toxicological relevance to similar lesions in other laboratory animals such as rodents, as well as in humans, are also discussed.
The Animal
The animal was a clinically normal 1-year-old female beagle dog in a standard 1-month Good Laboratory Practice (GLP) oral gavage toxicity study. It belonged to a low dose group comprised of 4 males and 3 females that did not exhibit any test article–related effects. The animal received all routine vaccinations, and a veterinary inspection was performed shortly after arrival and made available throughout the study. Animals in the study were group-housed in environmentally controlled conditions in groups of 3 or 4 animals in anodized aluminum caging with appropriate/standard sterilized bedding material, environmental enrichment, food, and water.
The study was conducted at Charles River Laboratories, Den Bosch (the Netherlands) in accordance with the European (European Commission directive 2010/63/EU) guidelines for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes.
Pathology Procedures
At terminal necropsy, the animal was deeply anesthetized using intravenous tiletamine/zolazepam combination, euthanized by intravenous injection of pentobarbital and subsequently exsanguinated and subjected to a full postmortem examination. After full routine necropsy, standard full tissue samples were collected, fixed in 10% buffered formalin, embedded in paraffin, sectioned, mounted, and stained with hematoxylin–eosin (H&E), and then examined histopathologically by a board-certified pathologist. After the initial evaluation of H&E-stained slides, Masson’s trichrome (MT) stain was applied to the mediastinal lymph nodes sections to highlight collagen. In addition, complimentary immunohistochemistry (IHC) was performed with a Discovery Ultra staining module (Ventana Medical Systems, Inc) using the following antibodies: anti-Ki67 antibody as a marker of proliferation (Abcam, rabbit polyclonal antibody, ab15580, dilution 1/2500), anti-alpha smooth muscle actin (SMA) antibody as a marker of smooth muscle component (Abcam, rabbit polyclonal antibody, ab5694, dilution 1/12,500), and anti-von Willebrand factor (vWF) antibody (Abcam, rabbit polyclonal antibody, ab6994, dilution 1/7000 or 1/6000) to highlight endothelial components.
Macroscopic Findings
Necropsy examination revealed no macroscopic findings apart from enlargement and red discoloration of 3 lymph nodes comprising the cranial mediastinal lymph nodes. No other lymph nodes were affected, including the adjacent contralateral lymph nodes (left or right not specified), and no other study subject exhibited the same alteration in any of their lymph nodes.
Histopathology
On all 3 sections of the cranial mediastinal lymph nodes, a lesion characterized by moderate to marked effacement of the lymph node parenchyma with replacement by fully formed, mature blood vessels, vascular channels, smooth muscle, spindle-shaped cells of varying cellular density and fibrous tissue, as well as lymphoid atrophy, was present (Figure 1A-D). The lesion affected approximately 60% to 75% of the lymph nodes and consisted of 2 main recognizable morphologic patterns (Figure 1A and B). The first, which was mainly located near the hilus or central lymph node, consisted of a proliferation of variably sized and haphazardly arranged mature blood vessels (Figures 1C and 2A and B), surrounded by or interspersed with abundant mature collagen (Figure 2A-C), which was confirmed with MT stain (Figure 2C), equally large amounts of spindle-shaped smooth muscle cells (confirmed with SMA immunostaining) arranged without a discernible pattern (Figures 1D and 2D), and smaller amounts of focally distributed adipocytes, which were considered to be in normal amounts and distribution for lymph nodes (Figures 1C and 2A, B, and D). In this lesion pattern, the vascular structures ranged from well-formed, muscular blood vessels lined by plump endothelium and surrounded by abundant stroma and smooth muscle cells to thinner-walled vessels containing vascular valves or thick-walled, dilated, blood-filled vascular channels lined by normal to plump endothelium (Figure 2A-F). There was no evidence of erythrophagocytosis or hemosiderosis. This first lesion pattern consisting of haphazardly arranged, fully formed blood vessels, abundant stroma, and spindle-shaped cells mostly representing smooth muscle cells (confirmed by SMA IHC) was considered more consistent with a diagnosis of AMH.
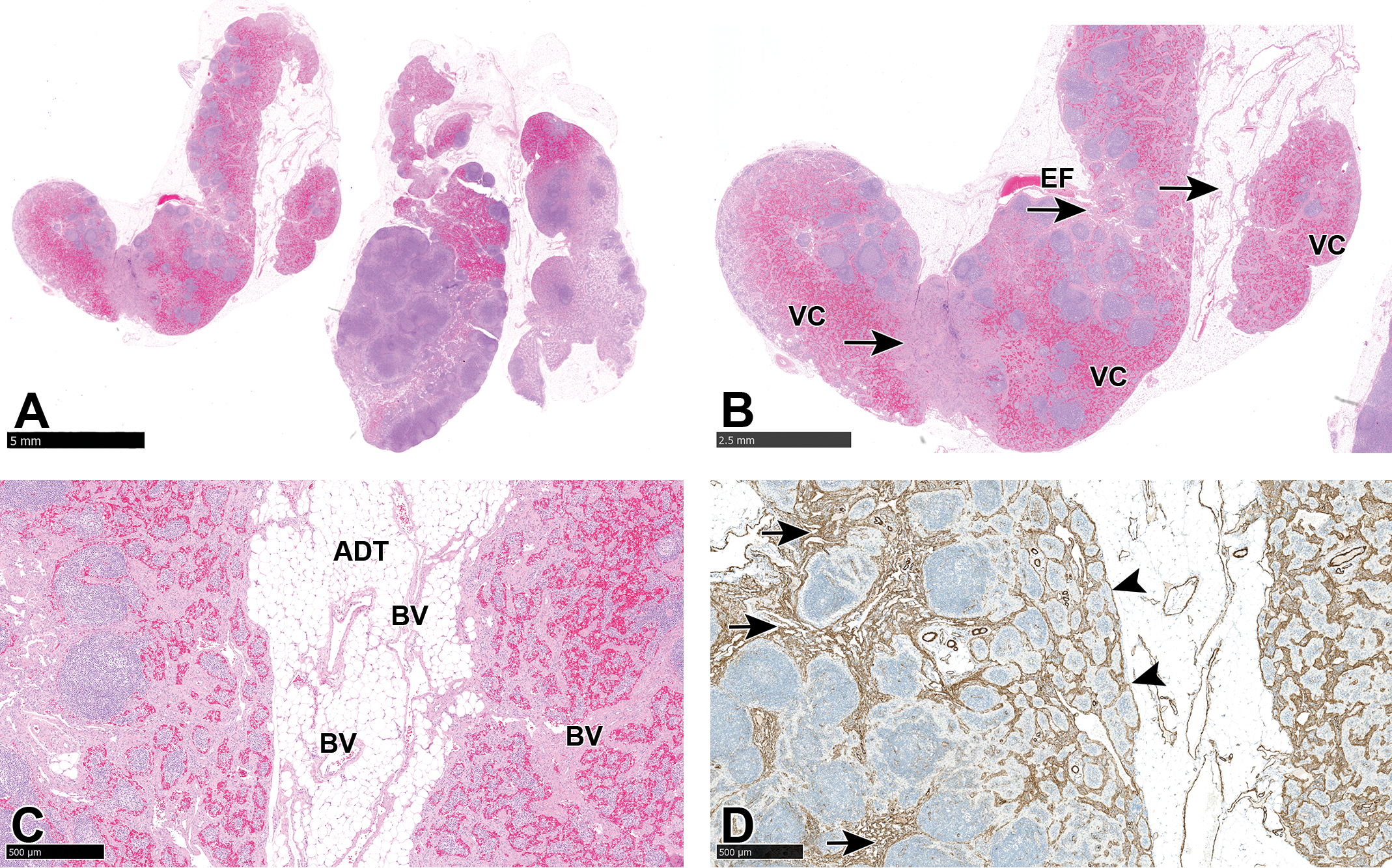
A-C, Cranial mediastinal lymph nodes showing replacement of lymphoid parenchyma by a proliferation of well-formed blood vessels (BV), capillary-like vascular channels (VC), and a mixture of smooth muscle cells and stromal tissue associated with lymphoid atrophy. Note that the adipose tissue (ADT) adjacent to the lymph node was considered normal and not part of the lesion. Mature vascular structures and more solid or abundant stroma are distributed near the hilus and efferent vessels (EF) or center of the lymph node (arrows), while blood-filled vascular channels (VCs) are distributed within the subcapsular sinuses. Hematoxylin and eosin. D, Alpha-smooth muscle actin immunostaining showing more dense distribution of smooth muscle near the hilus and center of the lymph node (arrows) compared to the medullary and subcapsular sinuses (arrowheads).
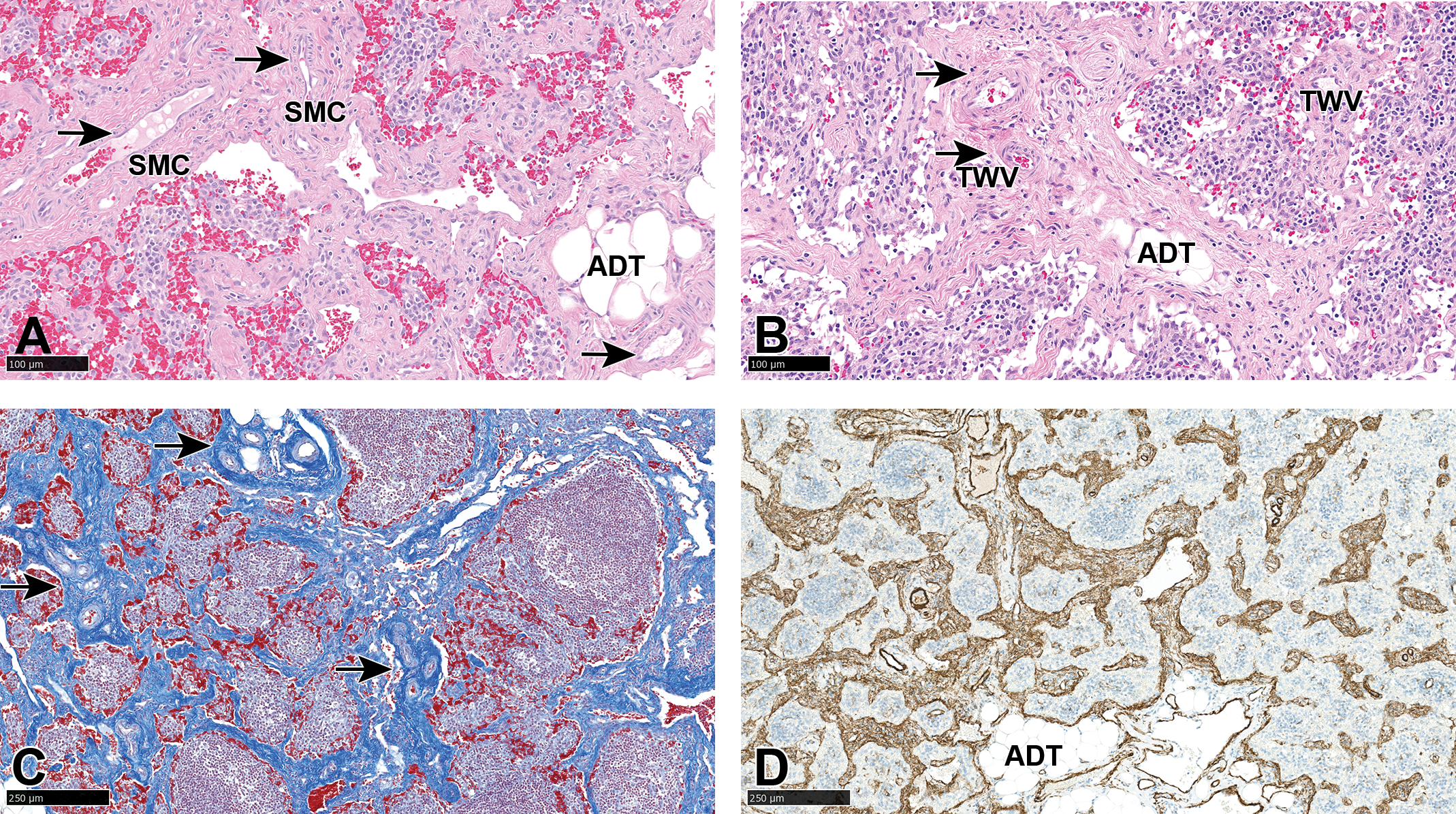
A-D, Cranial mesenteric lymph node: angiomyomatous hamartoma-like areas: haphazardly arranged fully formed, thick-walled, muscular blood vessels (arrows) or thin-walled vessels (TWV) surrounded by smooth muscle cells (SMC) and fibrous stroma, occasional focal normal adipose tissue (ADT), and remnants of atrophic lymphoid tissue (A and B). Hematoxylin and eosin. C, Masson trichrome stain showing dense fibrous stroma around thick-walled blood vessels (arrows). D, Smooth muscle actin immunostaining highlighting abundant smooth muscle interspaced between blood vessels and normal adipose tissue (ADT).
The second lesion pattern, which was located mainly in the subcapsular and medullary sinuses (Figures 1B and 3A-F), was largely comprised of a complex network of anastomosing, dilated, cavernous, blood-filled vascular channels supported by a dense collagenous stroma (Figure 3B) and lined by slightly plump endothelial cells (Figure 3C), which were confirmed by vWF immunostaining (Figure 3D). However, other morphologic patterns characterized by thin-walled, cleft-like vascular spaces or interconnected plexiform vascular channels lined by normal-sized to plump endothelial cells, interspaced by a thin fibrous stroma (Figure 3E insert), or “pericytomatous” concentric arrangement of spindle-shaped cells (most likely smooth muscle cells or pericytes) around small capillaries (Figure 3E), but lacking atypia and mitotic figures (Figure 3F), were present. This second spectrum of morphologic patterns composed of thin or thick-walled, blood-filled, interconnected vascular spaces, confined mostly to the subcapsular sinuses, was considered to represent VTS. Islands of remaining lymphoid follicles were often present within this area of blood-filled vascular channels and often contained hemosiderin-laden macrophages. No evidence of vascular obstruction, thrombosis, or lymph node edema was present in this lesion. Furthermore, no angiogenic effect of the test article and no vascular hamartomas or other similar lymph node changes were noted in the subsequent 9-month chronic toxicity study in Beagle dogs.
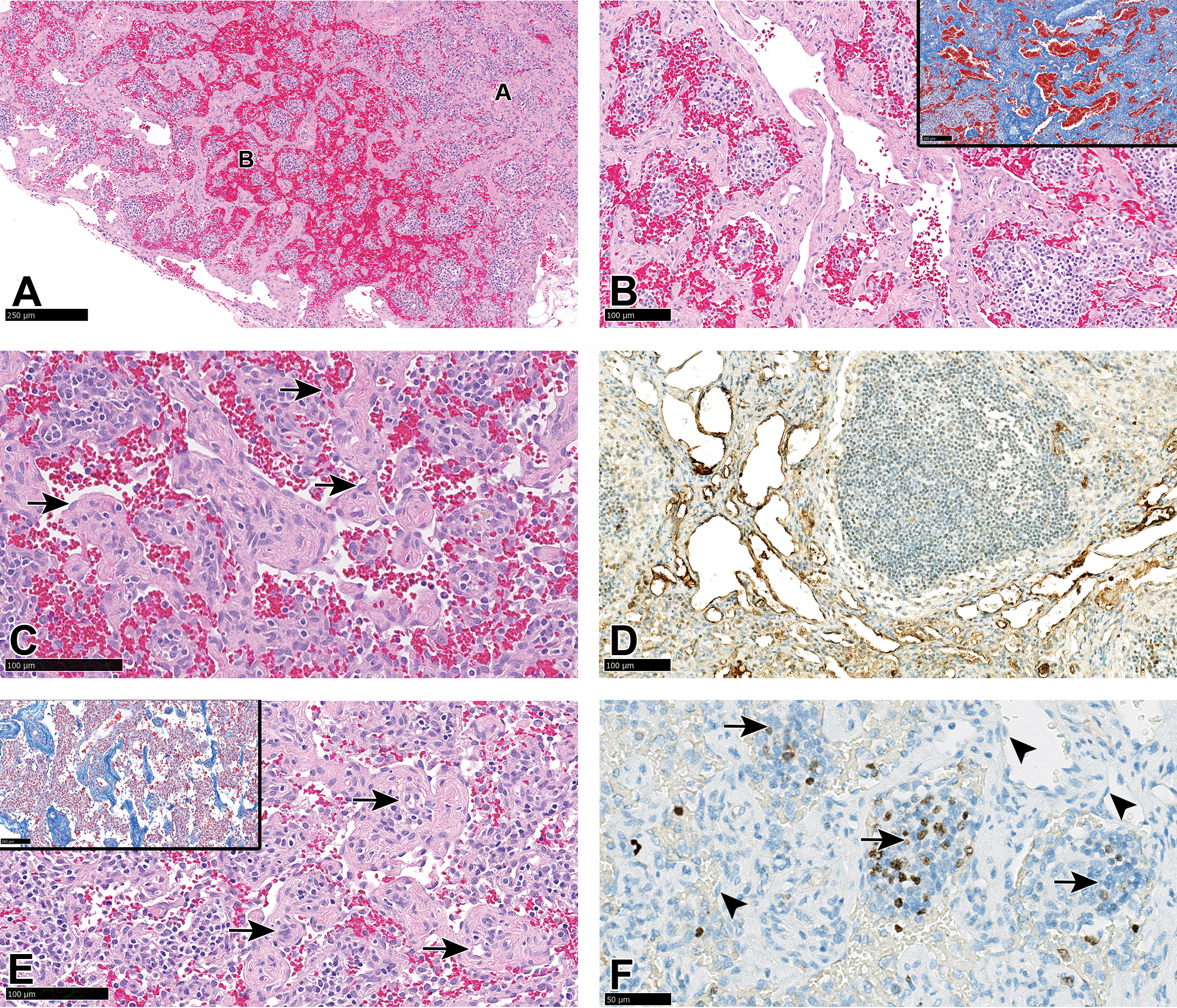
A-F, Cranial mesenteric lymph node: vascular transformation of sinuses-like lesion. A, Dilated, blood-filled, and thick-walled anastomosing vascular channels distributed mainly in the subcapsular region (letter B), lying adjacent to a region of dense stroma near the center of the lymph node (letter A). B, Higher magnification of the area depicted by letter B. (B insert) Masson trichrome staining of the dense stroma around the wide vascular channels. C, Slightly plump endothelial cells lining the thick-walled vascular channels (arrows). D, Positive von Willebrand factor immunostaining of the endothelium lining the vascular channels. E and insert, “Pericytoma-like” areas showing small-lumen capillaries surrounded by concentrically arranged spindle cells (arrows) and interspaced by sparse collagen demonstrated by Masson trichrome stain (insert). F, Ki 67 stain for proliferating cells showing negligible staining in endothelial, smooth muscle, or stromal cells (arrowheads) and positive staining (proliferation) only in the remnants of lymphoid tissue (arrows).
Immunohistochemistry
Immunohistochemistry analysis for SMA revealed abundant smooth muscle in the stroma as well as in the wall of well-formed vessels around the hilus, that is, in the AMH-like area, and less so in the periphery of the lymph nodes (Figure 1D). Staining for vWF allowed a better visualization of the endothelial lining of both the dilated and cleft-like vascular channels (Figure 3D), while Ki67 confirmed absent or negligible proliferation of endothelial, stromal cells, or smooth muscle cells in the lesion, showing positivity only in the remaining lymphoid tissue or sparse white blood cells in the vascular lumen (Figure 3F).
Angiomyomatous Hamartoma and VTS
On the basis of the characteristic microscopic features for the diagnosis of AMH in human and other animals (lesions concentrated in the hilus and center of lymph node and composed of mature well-differentiated blood vessels, fibrous tissue, and smooth muscle), 1,2,17 the first lesion pattern was considered to be more consistent with an AMH (Table 1). The main differential diagnoses were VTS, plexiform vasculopathy, angiomatosis, or lymphangiomatosis (described exclusively in women), which are considered by others to be the same lesion or morphological variants of the same lesion. 8,10,11 The main difference between AMH and VTS is that, as a hamartomatous lesion, AMH is mainly composed of a proliferation of haphazardly arranged well-formed, muscular blood vessels (rather than capillaries as in VTS), abundant smooth muscle, and variably present adipose tissue in a fibrous stroma, while VTS consists mostly of a proliferation of blood-filled, capillary-like vascular channels, organized in a complex network of branching/anastomosing channels. 8,12,18 In addition, VTS is believed to originate in the subcapsular and intermediate sinuses, as demonstrated experimentally in the rabbit, 13 unlike AMH which is thought to develop from the hilus, sparing the capsule. 1,4 Based on this morphological description and progression of VTS, the lesion observed in the subcapsular sinus and extending into the medullary sinus was considered to be consistent with VTS. Therefore, lesions consistent with both AMH and VTS were considered to be present in this lymph node.
Abbreviation: LN, lymph node.
Regarding the pathogenesis, although the cause and pathogenesis of AMH is unknown, others consider it to represent congenital or developmental localized malformation of hilar blood vessels or lymphatics resulting from a disordered angiogenic process, associated with hamartomatous proliferation of smooth muscle and adipose tissue. 3,17 Vascular transformation of sinuses on the other hand is believed to be a reactive lesion caused by partial or total obstruction of lymph vessels or efferent veins, although other processes such as disorganized angiogenesis are also thought to play a role. 4,6 In most reported cases of VTS in both veterinary and human pathology, the cause of the lymphoid obstruction is usually identified, and secondary lesions associated with lymph vessel obstruction such as edema, erythrophagocytosis, and lymphoid follicular atrophy are observed. 4
Besides the aforementioned lesions, our differential diagnoses included angiomatous hyperplasia, which is frequently observed in aged rodents, hemangioma, hemangiosarcoma, and lymphangioma. The absence of cellular atypia or mitotic figures, growth pattern, and the presence of well-formed blood vessels ruled out hemangiosarcoma 21 and hemangioendothelioma, 22 although the presence of hemangiopericytoma-like areas in the VTS lesions and plumper endothelial cells made the diagnosis more challenging, while the diagnosis of hemangioma was ruled out based on the growth pattern and lack of a well-circumscribed lesion or compression of adjacent tissue. 19 However, although vascular tumors of lymph nodes are relatively common in the dog, 19 the median age of dogs with angiomas (hemangioma or lymphangioma) is 14.5 years old, and they are found almost exclusively in the popliteal lymph node. 19
Considering these facts, the pathogenesis of the lesion in our present case was considered to be most likely a primary AMH with secondary VTS, with the latter developing due to a possible functional obstruction caused by the localized malformation of the lymphoid and vascular system of the affected lymph node. This pathogenesis has been proposed in previous cases where both AMH and VTS were present in the same lymph node, 7,17 and due to the absence of any other apparent obstructive cause of the VTS, this is considered the most likely mechanism in the present study. First, in the context of a well-controlled noninvasive (oral gavage) study in which relatively young barrier-reared animals were subjected to a thorough necropsy and histopathological examination, an obstructive cause such as a tumor or thrombus would be unlikely to occur or to remain undetected. In addition, besides being clinically normal, no other pathology findings were present in this animal other than the lymph node findings, and there were no test article–related effects on the lymphoid or vascular system in any other animal in the study. Therefore, considering the young age of the animal, and the absence of other local or systemic cause of obstruction, a preexisting or incidental lymph node vascular anomaly (AMH) was considered the most likely primary cause of VTS and determined to be unrelated to treatment with the test article.
Comparative Pathology and Human Risk of Proliferative Vascular Lesions of Lymph Nodes
In rodents, proliferative vascular lesions of lymph nodes are well-documented, and both the hematolymphoid 16 and cardiovascular 23 system International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) publications describe hemangioendothelial hyperplasia, angiomatous hyperplasia (angiomatosis), hemangioma/lymphangioma, and hemangiosarcoma as commonly encountered lesions in the lymph nodes of aging rats and mice, particularly in the mesenteric lymph nodes of Han Wistar rats (males more affected than females) and CD1 or B6C3F1 mice. What is interesting to note is that morphologically, angiomatous hyperplasia of the mesenteric lymph node in Han Wistar rats represents the closest comparative lesion to VTS. In an excellent review of lymph node angiomas in rats, Radi and Morton suggested that angiomatous hyperplasia of the mesenteric lymph nodes in Han Wistar rats might represent either VTS with similar pathogenesis and anatomic origin in the lymph node or a vascular developmental anomaly. 24 In addition, the hematolymphoid system’s INHAND describes angiomatous hyperplasia in rats and mice as being synonymous with VTS, having some morphological resemblance to angiomatosis in humans, and most likely caused by the occlusion of efferent lymphatics. 16 In mice, angiomatous hyperplasia can also occur in the mesenteric lymph nodes of aged animals and is considered to be the same lesion as the “vascular anomaly” described in rasH2 mice. 23 Finally, the progression of these lesions to hemangiosarcoma is considered doubtful, 16,24 and their relevance to human risk is considered minimal, since hemangiosarcomas are extremely rare in humans, and there are species-specific differences, such as the rate of endothelial cell proliferation, that account for the species differences in the occurrence of vascular tumors between humans and animals. 25
In toxicologic pathology, the description and characterization of preexisting, spontaneous findings observed in experimental animals is critical for distinguishing them from test article–related findings. Previously unreported background findings in particular should be documented, especially in the context of the current efforts to establish consistent terminology for nonrodents similar to those established for rodents. We hope this article will encourage the publication of more reports regarding vascular changes in the lymph nodes of dogs and other nonclinical species.
Footnotes
Authors’ Note
Sophie Nelissen and Ronnie Chamanza shared first authorship, contributed equally, and are co-first authors.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.