Abstract
Lymph nodes and associated lymphatics filter extracellular fluid and lymph to maintain tissue–fluid balance and detect distant tissue injury. Examination of regional draining lymph nodes (RDLs; lymph nodes that drain the route of article dosing) is an important step in detecting immunotoxicity and other associated changes during general toxicology studies. Similarly, evaluation of RDLs is often a key component of evaluating medical devices. Nonclinical medical device studies can present challenges for RDL evaluation, due to the wide variety of tissues and organs that are implanted with devices, the potential for wear debris/degradation products, and the likely disruption of normal lymphatic drainage by surgical procedures. This article discusses concepts for consideration when designing a nonclinical medical device study that includes the macroscopic evaluation, collection, histologic processing, microscopic assessment, and documentation of findings within RDLs. References describing RDLs for common implantation sites are reported, as are considerations for specific tissues and species commonly used in medical device biocompatibility and functional testing.
Lymph nodes and associated lymphatic capillaries/ducts are the primary pathway for returning extracellular fluid to systemic circulation and serve as an early detection system for tissue injury. Regional draining lymph nodes (RDLs) are the specific lymph nodes/lymphoid aggregates that drain a particular anatomic location or organ. In some organ systems, lymphoid tissue located outside of lymph nodes may also respond to dosing, including mucosal-associated lymphoid tissue (MALT), tertiary lymphoid structures, and serosa-associated lymphoid clusters (located in the serosa and adipose tissue of the peritoneal, pleural, and pericardial cavities). Evaluation of RDLs can aid in detecting immunotoxicity, and it is considered a best practice by the Society of Toxicologic Pathology to examine RDLs associated with a dosing route (Haley et al. 2005).
RDLs in Medical Device Studies
As with general toxicology studies, examination of RDLs is an important part of the overall evaluation of medical device safety and efficacy (for the purpose of this article, “medical device” refers generally to the wide variety of articles, combination products, and tissue engineering constructs that can be classified as medical devices). Several factors can make evaluating RDL responses to medical devices more challenging than for general toxicology studies, including a wider array of delivery routes/implantation sites and the potential for physical degradation of device components. Medical devices can be implanted in virtually every organ/tissue and may cause changes in different RDLs than those that drain standard dosing routes. Additionally, portions of a device may travel directly to RDLs. This can include wear debris (chemically unaltered fragments that broke off of the main article after mechanical wear), degradation fragments (often transported to RDLs within phagocytic cells; Bondarenko et al. 2011), or unaltered small articles (e.g., magnetic beads or nanoparticle aggregates). Ultimately, these additional factors have contributed to an increased emphasis on RDL examination by current medical device guidances (International Organization for Standardization 2016, 2017).
This article outlines concepts to consider when evaluating RDLs in medical device studies. Concepts are organized into different stages of a study, including (1) study design, (2) macroscopic assessment and tissue collection, (3) tissue processing and histologic staining, (4) microscopic evaluation, and (5) report documentation. These concepts are applicable to both early developmental studies and for studies intended for regulatory submissions. Finally, this article summarizes references that can aid in identifying RDLs for different anatomic sites in nonclinical species commonly used in medical device studies.
Concepts to Consider for RDLs in Medical Device Studies
Stage 1—Study Design
Whenever possible, a pathologist and/or veterinary surgeon should evaluate the study design or protocol prior to study initiation. In addition to providing general guidance, individuals with extensive anatomy training can help ensure that standardized terminology for lymph node identification and collection is established. The use of standardized terms for certain anatomic regions is necessary to avoid ambiguities. For example, approximately 300 of the over 800 lymph nodes described in humans are located in the neck (e.g., “cervical lymph nodes”, Harisinghani 2013). While each of these lymph nodes has a standardized human anatomy term and harmonized nomenclature systems have been proposed for rodents (Tilney 1971; Van den Broeck, Derore, and Simoens 2006), lymph node names are less standardized for many larger nonclinical species used in medical device studies. The use of clinical lymph node names like cervical lymph node often appear in published reports (Dunne et al. 2003; Soto-Miranda, Suami, and Chang 2013), and it can be challenging to correlate those reports with the standardized Latin anatomic terms used in anatomy atlases (e.g., rabbit in Popesko, Rajtova, and Horak 2002). Therefore, to ensure consistency across a study, it is important that all members of the study team agree on the names and locations of all RDLs to be collected and evaluated prior to study initiation. The use of diagrams and appropriate references can be instrumental in eliminating ambiguity and ensuring consistent evaluation and tissue collection at necropsy.
Additionally, surgery procedures may complicate RDL evaluation in implantable medical device studies. If an implantation procedure disrupts normal lymphatic drainage, different RDLs may become relevant to evaluate. This is recognized both in the clinical (Harisinghani 2013) and nonclinical literature. For example, while mesenteric lymph nodes are a major RDL for the peritoneal space prior to surgery, intestinal resection in rats can shift primary drainage of the peritoneum to lymphatic capillaries on the peritoneal surface of the diaphragm, which is rapidly drained into mediastinal nodes/thoracic duct (Parungo et al. 2007).
Surgery itself can also produce profound macroscopic and microscopic changes in RDLs (e.g., hemorrhage and increased immune cells). Therefore, in some studies, it may be necessary to include control RDLs (lymph nodes draining sham or control article-implant site(s)) in order to differentiate surgical effects from potential device-specific effects.
Medical device studies may include implantation of multiple articles within the same individual. This is done commonly for biocompatibility studies in muscle and subcutaneous tissue to help control for individual animal variation (International Organization for Standardization 2016). The use of multiple articles in the same individual can confound interpretation of RDL changes if there is overlap in drainage between the two implant sites. Additionally, in subcutaneous tissue, several lymph nodes clustered close together anatomically may drain different anatomic regions (illustrated in Tilney [1971] for each of the four axillary lymph nodes).
Finally, it is important to consider age/physiological changes that may take place over the length of a study. This is especially important for studies that assess medical device degradation over several years in tissues such as bone or if a device is implanted in an organ that undergoes significant physiological changes (e.g., lymph node changes during pregnancy; Allen and McLean 1971).
Stage 2—Macroscopic Assessment and Tissue Collection
The presence of a pathologist and/or other appropriately trained individual at necropsy can be vital when evaluating complex devices or devices implanted in areas with intricate lymphatic drainage patterns (Nikula and Funk 2016). Such individuals can also aid in detecting macroscopic changes in lymph nodes other than the RDLs listed in the protocol and ensuring those other potential RDLs are appropriately collected. Depending on which tissues are being collected, it may be useful to remove lymph nodes early in the necropsy before anatomic landmarks are removed or disrupted (e.g., collecting axillary lymph nodes before the forelimb is removed).
Certain factors may make it challenging to identify and collect lymph nodes at necropsy. Lymph nodes that are smaller than normal (e.g., cytotoxic effect from device) or are typically small may be hard to distinguish from other tissues. Lymph nodes can be particularly hard to identify in older animal with abundant white adipose tissue or in animals that typically form large subcutaneous fat depots (e.g., rabbits). If it is anticipated that a specific RDL may be hard to visualize at necropsy, it may be advisable to perform specific training studies using dyes or other markers (examples in Gopee et al. 2007) and take photographs to use as a reference during necropsy. Study animals could also be administered dye shortly before necropsy to aid in identifying RDLs; however, the benefit of macroscopic lymph node identification must be weighed against the potential that the dye may obscure microscopic lymph node changes. If (despite all efforts) a lymph node cannot be clearly distinguished from surrounding adipose tissue at necropsy, it may be necessary to collect a larger piece of fat from the correct anatomic site and then use secondary methods to dissolve the surrounding fat prior to histologic processing. This can be done using specialized fixative at the time of necropsy or subsequent treatment of formalin-fixed tissues with acetone or acetic acid (Lavy et al. 2014). Finally, if an animal was implanted with multiple devices, separate collection and labeling of RDLs is important to maintain orientation for histological processing.
Macroscopic color changes can be present in RDLs and should be distinguished from article-related effects from the device. These include melanosis in individuals with heavy pigmentation (e.g., black-faced sheep) and the discoloration of RDLs that drain tattoos (in these cases, the color of the lymph node will match the tattoo ink). Macroscopic findings should be described using standard terminology. In specific cases, macroscopic photographs may aid in documentation.
Stage 3—Tissue Processing and Histologic Staining
The procedures routinely used for trimming and processing lymph nodes for general toxicology studies are typically appropriate for RDLs from medical devices studies (Haley et al. 2005). Morawietz et al. (2004) describe trimming specific to the lymphoreticular system. Altered trimming procedures for other standard tissues (e.g., nasal cavity and intestine) may be needed if MALT or other lymphoid aggregates are being evaluated. If multiple different articles were implanted in an individual animal, RDLs from each site should be clearly identified and processed separately as needed to maintain lymph node identity. Additionally, if it is known in advance that stains other than hematoxylin and eosin are needed to visualize article/article fragments that may be present in RDLs, those stains can be prepared in parallel with the standard sections.
Stage 4—Microscopic Evaluation
It is beyond the scope of this article to provide an in-depth review of the microscopic evaluation of lymph nodes. Instead, the reader is directed to the wealth of resources in this area (Elmore 2006a, 2006b; Willard-Mack 2006; Frith, Ward, Brown, et al. 1996; Frith, Ward, Chandra, et al. 2000; www.goreni.org). The specific compartments and cellular components with microscopic changes should be described (Haley et al. 2005).
A variety of microscopic changes may be seen in RDLs that drain medical device implant sites. Surgical related findings are common, especially if the time between surgery and the tissue collection was short; changes can include extracellular fluid, blood (components and whole cells), and increased numbers of innate immune cells. Article may be present either as phagocytosed material or free within the RDL; polarized light and special stains may be useful for detecting specific articles/article degradation products. Finally, in studies using animals with an unknown or nonspecific pathogen-free status (e.g., conventional swine and small ruminants), RDLs may show responses to pathogens unrelated to the device being tested.
If lymph nodes are being assessed from multiple time points, it may be necessary to recognize and distinguish age-related changes (e.g., fat infiltration in older animals) from test article–related effects. Additionally, RDLs that drain sites implanted with degradable articles may have widely variable responses over time as article is removed from the implantation site. These confounding factors can make the inclusion of appropriate control lymph nodes essential for medical device studies. Controls may be RDLs from control animals (untreated/sham/negative control/sponsor provided controls) or RDLs from the same animal that were collected from an area distant from the test article implantation site (e.g., left hind limb implanted with test article and right hind limb underwent a sham procedure or was implanted with a control article).
Stage 5—Documentation of Findings
Nomenclature used for general toxicology studies is generally appropriate for describing findings in RDLs in medical device studies. Standard International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) nomenclature for lymph nodes is under review at the time of writing (found at www.goreni.org). Additional terms may be needed for device-specific changes (e.g., article or degradation products are visible microscopically within RDLs). Macroscopic or microscopic findings in RDLs should be correlated with other study findings (clinical pathology, reaction in implant sites, etc.). Whenever possible, findings in RDLs should also be differentiated into article or nonarticle caused (e.g., procedural, underlying infection). If changes appear to be article related, the findings should be put in context for the overall safety of the medical device.
Macroscopic and microscopic images can be relatively common in reports for medical device studies. When microscopic changes can be adequately described using standard nomenclature, microscopic images are unlikely to add significant additional value to a report. In specific cases where article/article degradation products are present in RDLs or there are other unusual findings that are not easily captured with standard terminology, it may be appropriate to include representative images.
Tissue/Organ System and Species-specific Considerations
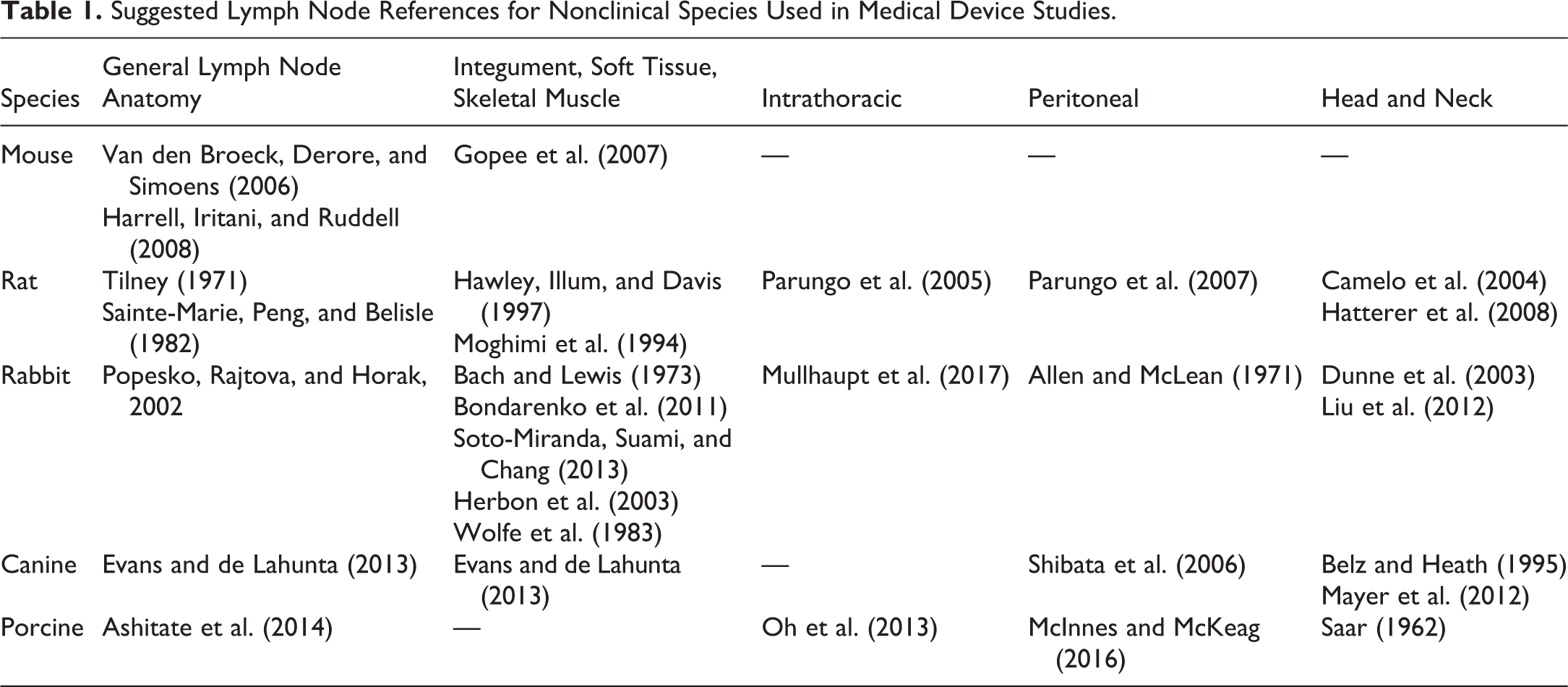
Table 1 lists references for general lymph node anatomy and specific tissues/organ systems in nonclinical species often used in medical device studies. The majority of references include images/diagrams of normal lymph node anatomy and/or techniques for dye enhancement of lymph nodes to aid in macroscopic identification of RDLs. General lymph node differences between species have been reviewed previously (Haley 2017), and the forthcoming nonrodent INHAND publications (rabbit, dog, minipig, and nonhuman primate) will note lymph node anatomy differences where appropriate. Additionally, references documenting lymph node anatomy and lymphatic drainage patterns may be found in the literature of related fields including human and veterinary surgical oncology/reconstructive surgery (Munn and Padera 2014; Beer et al. 2018). Finally, since bone and central nervous system tissue are common sites for medical device placement, unique considerations for these tissue are highlighted below.
Suggested Lymph Node References for Nonclinical Species Used in Medical Device Studies.
Bone
Bone (and associated bone marrow) is generally considered to lack lymphatic vessels. Consequentially, cellular phagocytic responses can have a relatively prominent role in responding to medical devices implanted in bone. Reports have shown that removal of wear debris from the bone-implant interface may be primarily through phagocytosis of particles and not through lymphatic drainage (Edwards et al. 2008). For bone implants, it may be useful to combine examination of RDLs with evaluation of the overlying soft tissue and organs to which phagocytic cells often traffic (e.g., spleen and liver).
Central Nervous System
Drainage of brain and spinal cord tissue by the glymphatic system (reviewed in Aspelund et al. 2015; Louveau et al. 2017) is a rapidly expanding area of study and specific details are outside the scope of this article. Recent reports have produced evidence that material from the brain may be transferred via glymphatic drainage to cervical lymph nodes in humans (Eide et al. 2018), and similar mechanisms may exist in nonclinical species. Since details on glymphatic drainage to peripheral lymph nodes remains limited at this time, it may be useful to collect multiple lymph nodes in the head, cervical, and peri-axial regions for intracranial and spinal cord devices. Additionally, since many devices implanted in central nervous tissue require surgical removal/disruption of bone, considerations discussed above for bone may also apply (Rhalmi et al. 2007).
Concluding Thoughts
Medical device studies present unique challenges in identifying, collecting, and evaluating RDLs. Future studies that better characterize drainage patterns for certain organs (e.g., glymphatic drainage of the central nervous system), and large animal nonclinical species will hopefully aid in overcoming these challenges. In the interim, careful planning, communication, knowledge of species-specific anatomy, and recognition of common findings in RDLs can help ensure successful completion of a medical device study.
Footnotes
Author Contributions
All authors (LW) contributed to conception or design, data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
