Abstract
With the increasing use of animal-based biomaterials for regenerative medical applications, the need for their safety assessment is paramount. A porcine cholecyst-derived scaffold (CDS), intended as a muscle repair graft, prepared by a nondetergent/enzymatic method was engrafted in a rat abdominal wall defect model. Host tissue–scaffold interface samples were collected 2, 8, and 16 weeks postimplantation and evaluated by histopathology, immunohistochemistry, and electron microscopy. The nature of the tissue reaction was compared with those induced by a jejunum-derived scaffold (JDS) prepared by the same method and a commercial-grade small intestinal submucosa (CSIS) scaffold. A study of the immunopathological response in major lymphoid tissues and immunophenotyping for M1 and M2 macrophages was performed at the host tissue–scaffold interface. Further, “irritancy scores” for CDS and JDS were determined using CSIS as the reference material. Both CDS and JDS appeared to be potential biomaterials for muscle grafts, but the former stimulated a skeletal muscle tissue remodeling response predominated by M2 macrophages. The data support the notion that biomaterials with similar biocompatibility, based on local tissue response on implantation, may cause differential immunogenicity. Additionally, CDS compared to JDS and CSIS was found to be less immunotoxic.
The emerging field of regenerative medicine (RM) uses cells, scaffolds, and signaling molecules for repairing and replacing damaged organs/tissues (Langer and Vacanti 1993; Atala 2009). Scaffolds used in RM form substrates for cell proliferation, cell migration, and cell differentiation. Although synthetic biomaterials are widely preferred, bioscaffolds of mammalian origin are becoming increasingly popular, attributable to the richness of biomolecules congenial for cell proliferation/migration and differentiation (Swinehart and Badylak 2016; Dziki et al. 2016). Thus, biologic scaffold materials prepared from mammalian organs/tissues have been used for surgical repair or reconstruction of dura mater (Bejjani and Zabramski 2007), oesophagus (Badylak et al. 2011), nerve guidance conduits (Kehoe, Zhang, and Boyd 2012), tendon (Longo et al. 2012), ventral hernias (Alicuben and DeMeester 2014), skeletal muscle (Sicari, Dziki, and Badylak 2015), and many other organs/tissues (Badylak 2016). These scaffolds are essentially extracellular matrices (ECMs) of variable purity and biomaterial quality, depending on the method deployed for the scaffold preparation, the source organ, the donor species, and the intended use (Crapo, Gilbert, and Badylak 2011).
Porcine small intestinal (jejunal) submucosa is a well-studied animal-derived ECM, and data collected over the last 3 decades have indicated that it is a robust scaffold for biomedical applications (Badylak, Freytes, and Gilbert 2015; Badylak 2016). However, its use is often discredited because of the immunogenic potential leading to pain, discomfort, and graft rejection (Petter-Puchner 2007). The safety assessment of any biomedical product using animal-derived materials has a significant immunotoxicologic component (International Organization for Standardization [ISO] 10993-Part 20 2006; Schuh 2008; ISO 10993-Part 6 2007). It has been argued that many of the safety evaluation protocols listed in that standard have been designed for synthetic products of nonanimal origin, therefore not containing the diverse antigenic epitopes of biological molecules. Thus, the safety evaluation of scaffolds of animal origin needs different or expanded standards (Muhamed, Revi, Rajan et al. 2015). Indeed, supporting this notion, the authors have reported that biomaterials of animal origin while having a similar biocompatibility can have differential M1/M2 macrophage and Th1/Th2 lymphocyte reactions (Muhamed, Revi, Rajan, Geetha et al. 2015). Therefore, immunopathological observations form an essential component of safety data of biomaterials of animal origin.
Porcine cholecystic ECM is a relatively new decellularized scaffold derived from gall bladder that may have potential biomedical uses (Burugapalli et al. 2007). Its rich macromolecular content (Burugapalli et al. 2007) and mechanical properties (Coburn et al. 2007) are suitable for tissue engineering applications. Moreover, the extent of cross-linking and functionalization of the matrix can be modulated (Chan et al. 2008; Burugapalli et al. 2009, 2014) as required for specific tissue engineering uses. The scaffold has a potential application in skin-wound healing (Revi et al. 2013, 2016) and staple line reinforcement during gut surgery (Burugapalli et al. 2008). Kajbafzadeh et al. (2014) used homologous cholecystic ECM for urinary bladder reconstruction in the rabbit, and Shakya et al. (2016) found that bubaline (buffalo) cholecystic ECM can be used as a skin graft substitute. More recently, cholecyst-derived scaffold (CDS) prepared by a nondetergent/enzymatic method has been shown to cure corneal ulcers in dogs (Sainulabdeen et al. 2017). CDS is also a candidate scaffold for cardiac tissue engineering applications (Nair et al. 2017). Nowadays, volumetric skeletal muscle loss (VML) injuries present a complex and heterogeneous clinical problem that result from a massive loss of muscle tissue. Recent advances in RM suggest that immune-mediated tissue regeneration driven by a bioscaffold is an innovative strategy for repair of VML injuries (Dziki et al. 2016).
Like any other ECM-scaffold of animal tissue origin, CDS is largely an acellular scaffold (Anilkumar et al. 2014) with permissible cell/nuclear debris. This cell/tissue debris may be potentially immunogenic. In addition, bioscaffolds intended for repairing VML injuries must be evaluated before their introduction, so that the toxicological effects on the implanted host can be reduced. Therefore, the main objective of this study was to evaluate the immunopathological response induced by the porcine CDS in a male rat abdominal wall defect model.
Material and Method
Bioscaffold Preparation
Fresh porcine gall bladder and jejunum were obtained from a licensed slaughter house, and the CDS and jejunum-derived scaffold (JDS) were prepared by a nondetergent and nonenzymatic method as reported previously (Anilkumar et al. 2014) by delaminating the tissue layers and salvaging the fibromuscular layer beneath the mucosa and the submucosa, respectively, after ex situ cross-linking of the biomolecules with formaldehyde. A 1-layer tissue graft derived from small intestinal submucosa, marketed by Cook Surgisis®, was used as the reference scaffold (commercial-grade small intestinal submucosa [CSIS]) in the present study.
Animals and the Experimental Design
All animal procedures were approved by the Institutional Animal Ethical Committee of the College of Veterinary and Animal Sciences, Mannuthy, India, and all experimental procedures were performed in accordance with the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (Government of India). Naive 7 to 8 weeks old conventional male Sprague-Dawley rats (200 to 250 g) were procured from the Small Animal Breeding Station, College of Veterinary and Animal Sciences, Mannuthy, and acclimatized for at least 7 days before the study and had free access to food and water.
Fifty-four rats were randomized by body weight into 3 groups (n = 18 each) for implanting CDS, JDS, or CSIS. The rat abdominal wall defect was created as a modification from the rat model, previously described by Sicari, Turner, and Badylak (2013), except that the peritoneum was left intact to minimize postsurgical complications. Briefly, the rats were fasted for 12 hr prior to surgery and anaesthetized with ketamine hydrochloride (70 mg/kg) and xylazine hydrochloride (5 mg/kg) by intraperitoneal injection. A rectangular abdominal wall defect (2 cm × 2.5 cm) was created in each animal through a skin incision by removing the fascia and muscle, but leaving the underlying peritoneum intact. The defects were primarily repaired by grafting CDS, JDS, or CSIS with interrupted 4-0 prolene nonabsorbable sutures. The skin was closed with a nylon 3/0 suture in a horizontal mattress pattern over the implant.
Six rats in each group were humanely sacrificed at 2, 8, and 16 weeks postimplantation (WPI). The implant site was identified by the presence of nonabsorbable sutures. The abdominal wall including the implanted area was excised with adjacent host native tissue, lifted gently and examined macroscopically on both implant site and peritoneal surface for the extent of vascularity. Potential complications such as infection, abscess, hernia, fistula, adhesion, seroma, haemorrhage, encapsulation, graft shrinkage, graft loss, or failed graft incorporation if any were recorded.
A 3 cm long implant with the surrounding native muscle, at least 0.5 cm as margin (Supplemental Figure SF1), was retrieved en bloc, fixed in 10% neutral buffered formalin (NBF) for a morphological study including histological examination and immunohistochemistry or fixed in 2.5% glutaraldehyde for electron microscopy (Bancroft and Gamble 2008). Lymph nodes in the region of and distant to the implanted scaffolds (popliteal, submandibular, axillary, mandibular, and iliac), thymus, Peyer’s patches, spleen, and bone marrow were collected in 10% NBF for evaluating the toxicologic pathology of the immune system. Bone marrow smears were also made at the time of necropsy from the distal shaft of the femur by using a paint brush technique in 5% bovine serum albumin and 7.5% ethylene diamintetraacetic acid (2:1) and then stained with Wright–Giemsa stain (Reagan et al. 2011; Elmore 2012).
Histopathology and Immunohistochemistry
Histopathology on formalin-fixed and paraffin-embedded specimens was performed on 4- to 5-μm thick sections using haematoxylin and eosin (H&E) staining. For immunohistochemistry, epitope retrieval was performed by placing the slides in citrate buffer solution (pH 6.6) at 95°C for 20 min. Primary antibody incubation was performed for CD68 (pan-macrophage; 1:400 dilution, mouse monoclonal, Abcam, UK), CD80 (M1 phenotype; 1:400 dilution, rabbit monoclonal, clone EP1155Y, Abcam, UK), and CD163 (M2 phenotype; 1:400 dilution, mouse monoclonal, clone ED2, Santa Cruz, CA). After an hour, the sections were incubated with Super Enhancer reagent (BioGenex) for 20 min. The sections were then incubated in supersensitive polymeric horseradish peroxidase (HRP)-conjugated secondary antibody (BioGenex Laboratories, Fremont, CA) at room temperature for 30 min and incubated in a 4% diaminobenzidine substrate solution (BioGenex Laboratories, Fremont, CA). The sections were counterstained with Harris’s hematoxylin (Merck). Sections from spleen were stained as positive controls in parallel. Nonspecific isotype–matched immunoglobulins G at similar concentrations were used as primary antibodies for negative controls.
Determination of “Irritancy Score”
Irritancy score was calculated based on the procedure recommended in the ISO Standard 10993-Part 6 (2007). For this, the sections stained with H&E were evaluated for necrosis, infiltration by polymorphonuclear leucocytes, lymphocytes, plasma cells, macrophages, presence of giant cells, neovascularization, and fatty infiltration. Six microscopic fields within the host–scaffold interface for each section per implant under high power field (HPF) were observed and relevant cells counted. There were a total of 36 fields per implant per time point (n = 6 animals/time point). A score of 0 was given for the absence of inflammatory cells per HPF, score 1 for 1 to 5 cells/HPF, score 2 for 5 to 10 cells/HPF, score 3 for heavy infiltration, and score 4 for presence of packed cells (Supplemental Table ST1). An average semiquantitative score was then calculated as (Subtotal I × 2 + subtotal II), whereas subtotal I is the sum of scores for neutrophils, lymphocytes, plasma cells, macrophages, giant cells, and severity of necrosis and subtotal II is the sum of the scores for neovascularization, fibrosis, and fatty infiltration. The difference in the score for the test materials CDS and JDS from the reference material CSIS was denoted as the irritancy score for each of the grafts (Table 1). In this semiquantitative scoring scheme, due to the greater importance of inflammatory cell infiltrates and necrosis, these parameters were multiplied by a factor of 2 to provide a weighted value as compared to neovascularizaton, fibrosis, and fatty infiltration parameters. The values were totaled, and then, an average score for test and control treatments was calculated separately. Comparison of irritancy score was performed by subtracting the irritancy score of the reference material from the irritancy score of the test material. (Note: Since the completion of the work and preparation of the original manuscript, the ISO 10993-Part 6 (2007) has been replaced by ISO 10993-Part 6 (2016), in which the irritancy score has been substituted by the “reactivity score,” and several optional procedures have been suggested, but the method of calculation of both these scoring systems remain the same for both versions of the standard.)
Irritancy Scores (All Values Represent Mean of n = 6 Animals/Histology Slides/Implant/Time Point, Please See the Supplemental Figure SF1 and Figure 4 for the Sampling Plan and the Supplemental Table ST1 for Scores of Each Field), Determined (Based on ISO 10993-Part-6 2007) Following Semiquantitative Histomorphological Evaluation of the Implanted Cholecyst-derived Scaffold (CDS), Jejunum-derived Scaffold (JDS), and the Reference Material viz. Commercial-grade Small Intestinal Submucosa (CSIS).

Note: The criteria for comparison of irritancy scores with the reference material (a negative difference is recorded as zero): <2.9 = nonirritant; 3.0 to 8.9 = slight irritant; 9.0 to 15.0 = moderate irritant; and >15.1 = severe irritant). CDS = cholecyst-derived scaffold; JDS = jejunum-derived scaffold; CSIS = commercial-grade small intestinal submucosa; GC = giant cells; SN = severity of necrosis; NV = neovascularization; F = fibrosis; FI = fatty infiltrate.
aInflammatory cells: N = polymorphonuclear cells, L = lymphocytes, P = plasma cells, and M = macrophage. Gradings based on the number and distribution of cells (0 = 0 no cell, 1 = 1–5 cells, 2 = 6–15 cells, 3 = heavy infiltration, and 4 = packed cells represented as average of 6 fields at magnification, 400×).
bGiant cells (0 = 0 cells, 1 = 1–2 cells, 2 = 3–5 cells, 3 = heavy infiltration, and 4 = packed cells represented as average of 6 fields at magnification 400×).
cSeverity of necrosis: Grading determined by the presence of cell debris and inflammation (0 = not present, l = minimally present, 2 = mild degree, 3 = moderate degree, 4 = severe degree).
dNeovascularization: The extent measured as counts of the number of detectable vasculature under magnification of 400× (0 = no capillaries, l = l–3 capillaries, 2 = 4–7 capillaries, 3 = broad blood vessels, and 4 = extensive vascularization).
eFibrosis: Measured as thickness of fibrous tissue deposition, under magnification of 400×, around the implant (0 = absent, 1 = <5 μm, 2 = 6–15 μm, 3 = 16–30 μm, and 4 = >30 μm).
fFatty infiltrate: Grading determined by the amount of fatty tissue (0 = not present, 1 = minimally present, 2 = mild degree, 3 = moderate degree, and 4 = severe degree).
Electron Microscopy
The CDS explant samples were fixed in 2.5% cold glutaraldehyde in 0.1-M phosphate buffer (pH 7.2) for 24 hr for transmission electron microscopy (TEM). For TEM, the tissues were dehydrated in graded acetone solutions and embedded in Spurr resin (Sigma Aldrich, St. Louis, MO). Ultrathin sections were cut using an ultramicrotome (Leica ultracut UCT) with glass knives (Leica EMKMR2) at a thickness of 60 nm and were collected on 3-mm 200 mesh copper grids. Ultrathin sections were counterstained with uranyl acetate and lead citrate and examined by TEM (Phillips–CM 100) at an accelerating voltage of 100 kV.
Quantitative Histomorphology
Images captured under a 40× objective of an Olympus (BX51) microscope (Olympus Corporation, Japan) loaded with a DP70 camera and Image-Pro software (version 3DS6.1 software, Media Cybernetics, Silver Spring, MD) were used for the histomorphometric analysis. Quantitative analysis was performed by manually counting the number of immunopositive cells in 6 host–scaffold interface microscopic fields per implant. The percentage of M2 and M1 macrophages was determined by dividing the number of CD163+ and CD80+ cells by the number of CD68+ cells in each field. A ratio of the percentage of M2 cells to M1 cells was also scored for each field by dividing the percentage of M2 cells by the percentage of M1 cells (Badylak et al. 2008; Faulk et al. 2014).
Statistical Analysis
All experiments were performed with n = 6 for each experimental time point. The data are expressed as mean values ± standard error. All analyses were performed using Statistical Package for Social Sciences software (version 21.0, SPSS Inc. Chicago, IL). A 2-tailed p value of less than .05 was considered as a significant difference between groups. Because the cellular and tissue response was assessed using a semiquantitative score, Kruskal–Wallis 1-way analysis of variance (ANOVA) test was applied to compare between the 3 bioscaffold grafts. The statistical analysis of CD68+, CD80+, and CD163+ macrophages was performed by a 2-way ANOVA (p < .05) for every time point; data were further analyzed for significant differences between the 3 bioscaffold grafts using a 1-way ANOVA along with a post hoc Tukey’s honestly significant differences test.
Results
Gross Pathology and Histomorphology
There was no animal mortality or gross sign of infection/rejection of the implanted bioscaffold materials in any of the animals. The peritoneal surface of explanted CDS, JDS, and CSIS grafts is shown at different time points, respectively (Figures 1, 2, and 3). The peritoneal surface of explanted CDS and CSIS grafts had minimal to mild inflammatory swelling with moderate neovascularization at 2 WPI (Figures 1A and 3A), and the JDS graft site (Figure 2A) had mild swelling and minimal to mild neovascularization over the graft. At 8 WPI and 16 WPI, all the grafts appeared to have integrated well to the abdominal wall with signs of prominent neovascularization and muscle ingrowth (Figures 1C, E and 3C, E), but moderate thinning of the graft area was visible on the peritoneal surface of the JDS grafts (Figure 2C and E).
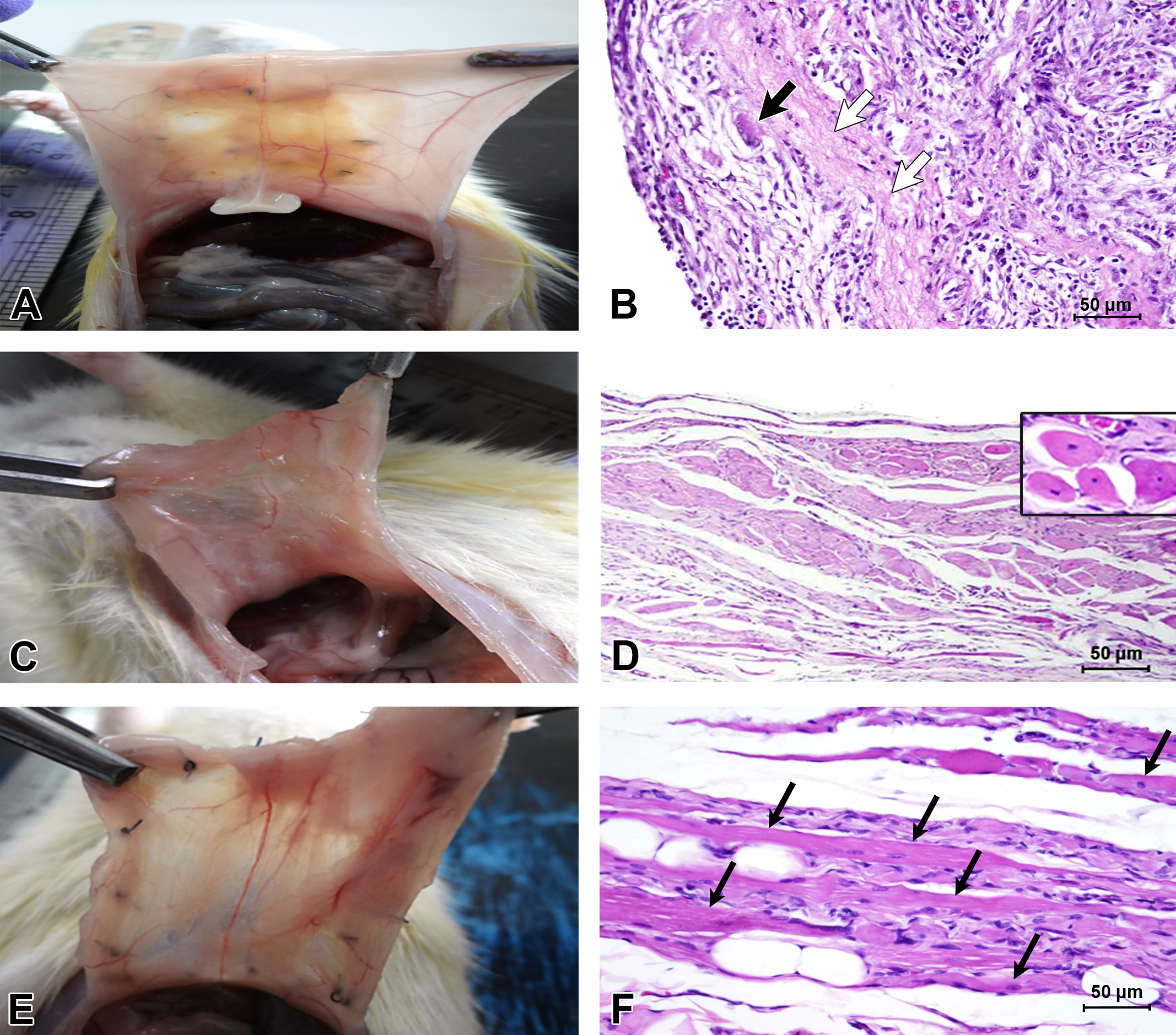
Gross (A, C, and E) and histomorphological (B, D, and F) appearances of the implants retrieved from the rat abdominal wall after 2 (A and B), 8 (C and D), and 16 (E and F) WPI from CDS-grafted abdominal wounds in rat. Grossly, the engraftment was characterized by neovascularization with complete integration as early as 2 WPI. Histomorphologically, there was moderate cellular infiltration by 2 WPI (B) which was reduced at later time points. The scaffold and giant cells are shown by thick white and thick black arrows, respectively. Regenerating myoblasts were evident from 8 WPI onward (D, inset). There were elongated muscle fibers (thin black arrows) with marked neovascularization, minimal adipocyte deposition between newly formed muscle bundles with organized minimal connective tissue deposition at 16 WPI (F). CDS = cholecyst-derived scaffold; H&E = hematoxylin and eosin; WPI = weeks postimplantation.
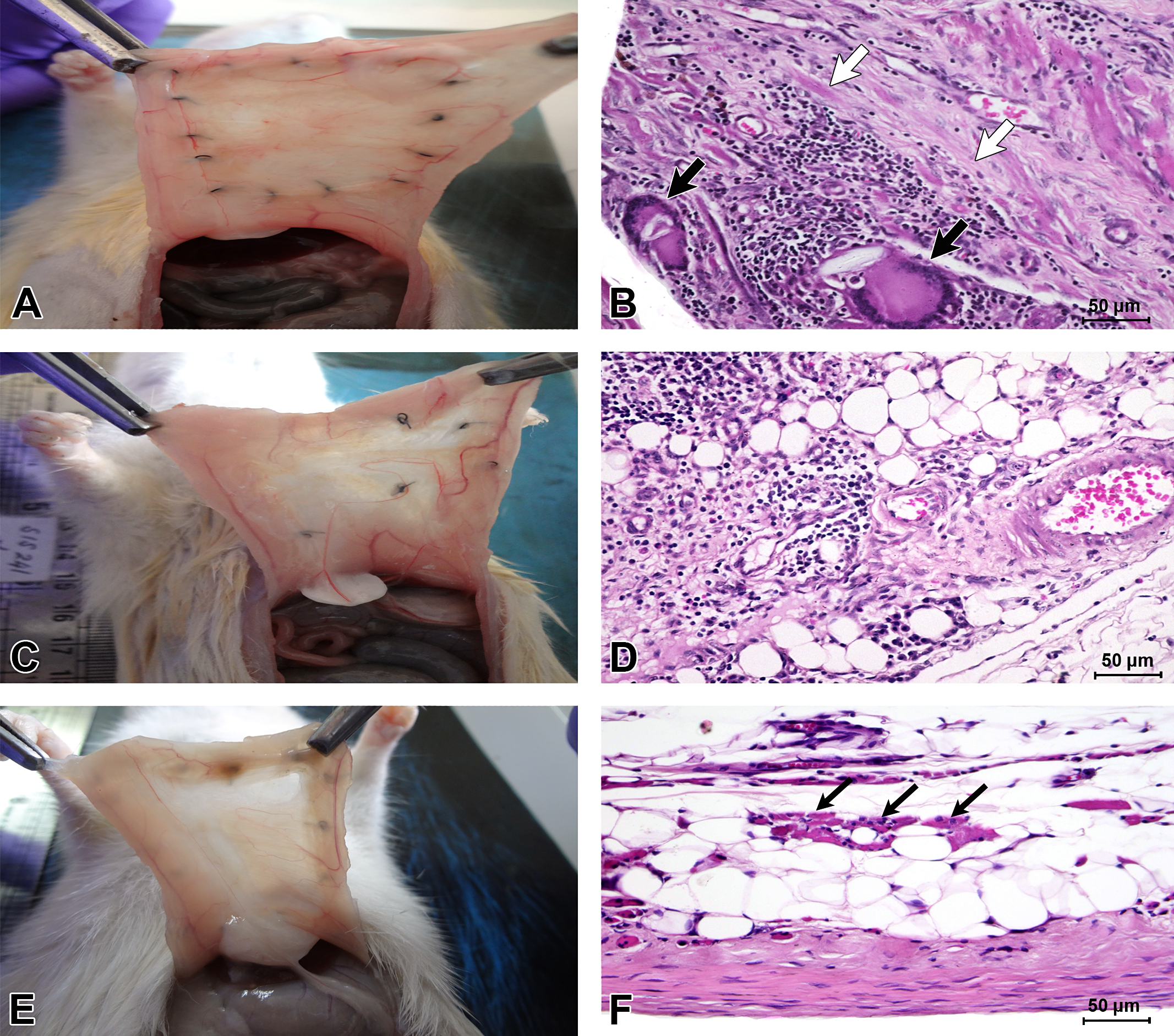
Gross (A, C, and E) and histomorphological (B, D, and F) appearances of the implants retrieved from the rat abdominal wall after 2 (A and B), 8 (C and D), and 16 (E and F) WPI from JDS-grafted abdominal wounds in the rat. Grossly, the peritoneal surface had mild neovascularization over the graft with mild swelling at 2 WPI (A). However, complete integration appeared to have happened by 8 WPI (C) and moderate thinning by 16 WPI (E). Host–JDS graft interface revealed mononuclear cell infiltration, giant cell formation, muscle necrosis, and neovascularization at 2 WPI. The scaffold and giant cells are shown by thick white and black arrows, respectively (B). A persistent chronic inflammatory response with mild adipocyte infiltration was seen by 8 WPI (D), and a group of regenerated muscle fibers (thin black arrows) were seen in the middle of the moderate adipocyte infiltration at 16 WPI (F). JDS = jejunum-derived scaffold; H&E = hematoxylin and eosin; WPI = weeks postimplantation.
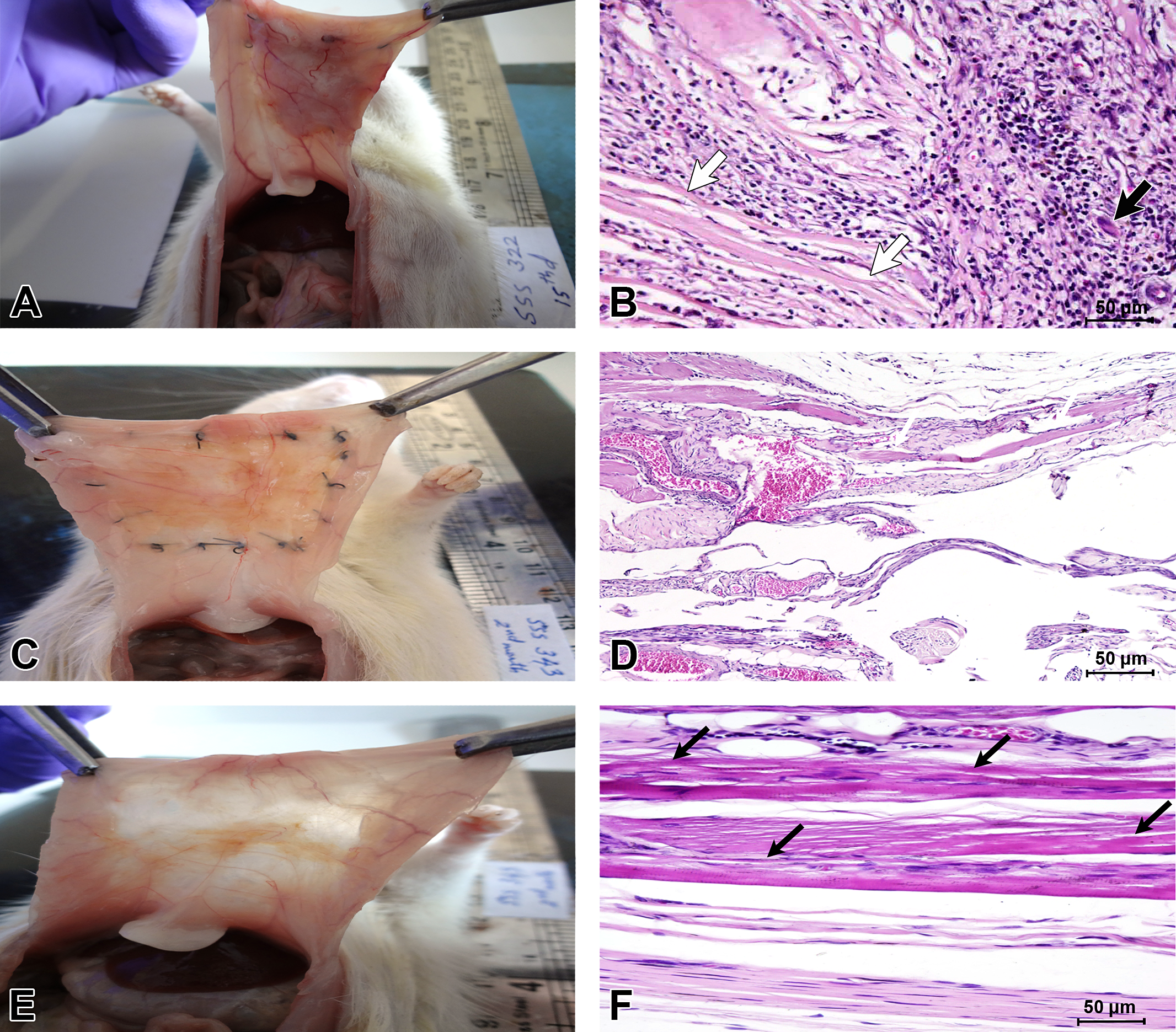
Gross (A, C, and E) and histomorphological (B, D, and F) appearances of the implants retrieved from the rat abdominal wall after 2 (A and B), 8 (C and D), and 16 (E and F) WPI from CSIS-grafted abdominal wounds in the rat. The graft showed complete integration with the host tissues with mild to moderate neovascularization at 2 WPI. Histologically, at the tissue–material interface, chronic inflammation was prominent with occasional muscle necrosis at 2 WPI. The scaffold and giant cells are shown by thick white and thick black arrows, respectively. Later, the extension of tissue in-growth toward the graft region (thin white arrows) and collagen deposition occurred by 8 WPI (D) and newly formed muscle bundles (thin black arrows) with minimal adipocyte deposition were seen at 16 WPI (F). CSIS = commercial-grade small intestinal submucosa; H&E = hematoxylin and eosin; WPI = weeks postimplantation.
Microscopically, at 2 WPI, H&E-stained host–graft interface sections revealed a mild to moderate inflammatory reaction characterized by the presence of polymorphonuclear cells, macrophages, lymphocytes, plasma cells, fibroblasts, and giant cells (Figures 1B, 2B, and 3B). There was tissue ingrowth between the graft folds characterized by moderate neovascularization, mild fibroblast proliferation, and occasional adipocyte infiltration in CDS and CSIS. The JDS appeared to have induced more giant cells (Figure 2B) than the CDS or CSIS. Degradation of the grafts was also noticed, which appeared as scattered graft debris in the reacting tissue in all the groups irrespective of the materials.
At 8 WPI, persistent mononuclear cell infiltration and fibroblast proliferation with ECM deposition were prominent. Many myoblasts with centrally placed nuclei, suggestive of muscle regeneration and extension of muscle from the native muscle tissues, were seen at the middle of CDS and at the interface of CSIS grafts, respectively (Figures 1D and 3D). On the other hand, JDS continued to have induced giant cell formation and mild adipocyte infiltration (Figure 2D). Later by 16 WPI, formation of elongated muscle bundles was evident with organized collagen deposition and stabilized sprouted vessels on the remodeled site in the CDS and CSIS grafts (Figures 1F and 3F). Occasional adipocytes were seen between the muscle bundles with a well organized but minimal connective tissue deposition on the CDS and CSIS grafts. At the host–JDS graft interface, moderate collagen deposition and mononuclear cell infiltration along with moderate fatty infiltration were observed (Figure 2F). Interestingly, a group of regenerated myoblasts were observed in the midgraft region of the JDS-induced tissue reaction (Figure 2F). The CDS (Figure 1F) appeared to have induced a better constructed muscle remodeling reaction in terms of cellular invasion toward the graft regions, collagen deposition, blood vessel formation and stabilization, and neomuscle formation compared to the other 2 types of graft (Figures 2F and 3F).
There was no gross abnormality in the popliteal, submandibular, inguinal, axillary, mandibular, or iliac lymph nodes, thymus, Peyer’s patches, or spleen in any rat. Histologically, no lesions were observed in any of these lymphoid tissues at any time point. The bone marrow/smears were also free of any induced lesions. There were no variations in bone marrow cellularity at any of the time points in all the groups.
Irritancy Score
A uniform sampling plan (Figure 4) was followed while determining the semiquantitative parameters at the tissue–material interface in all animal tissue samples. The score was 0 at 2 and 8 WPI, but at 16 WPI, the score for CDS was 0.46. In contrast, for JDS, the scores were 7.14 and 4.27 at 2 and 8 WPI, respectively, although the score was reduced to 2.94 at 16 WPI. Thus, compared to the reference material CSIS, CDS with a score of less than 3 was designated as a nonirritant material (Table 1). On the other hand, the JDS remained as a mild irritant until 8 WPI.
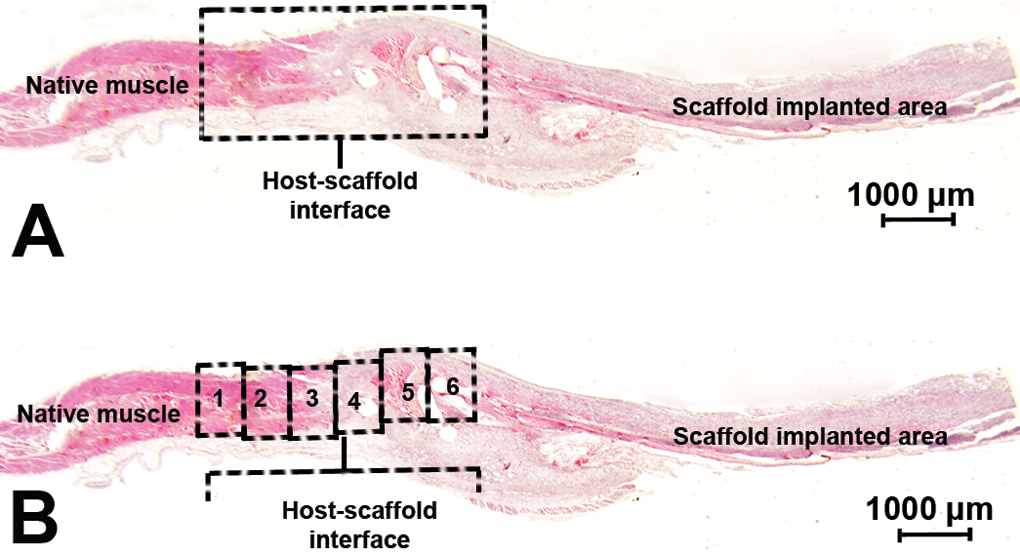
Light microscopic photograph showing the primary sampling area for histomorphometry (A) in which the cellular and tissue responses were graded semiquantitatively for each retrieved implant sample from 6 microscopic fields (B) at the host–scaffold interface. H&E = hematoxylin and eosin.
Immunohistochemistry
The immunoreactivity to antibodies against M0 macrophages (Figure 5A–C), M1 macrophages (Figure 5D–F), and M2 macrophages (Figure 6A–C) is indicated by brown cytoplasmic staining. The quantitative data indicated that M0 population was highest in JDS graft at all postimplantation study time points followed by CSIS and CDS grafts, respectively (Figure 7). Comparisons of the macrophage (M0) population, M1 phenotype percentage, M2 phenotype percentage, and M2/M1 ratio between these 3 grafts were performed for each time point separately, and the results are presented in Figure 8A–D.
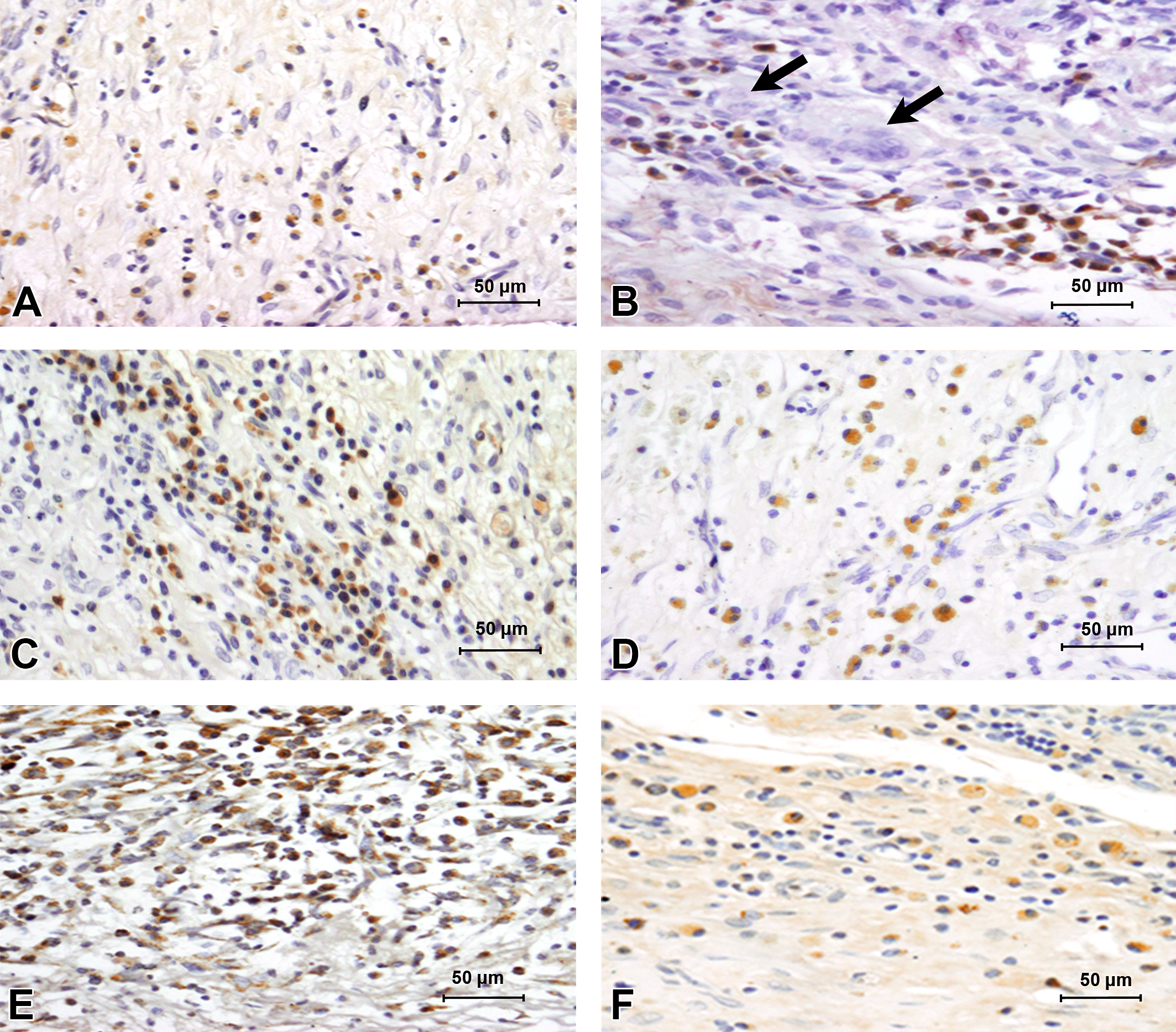
Immunohistochemical expression of CD68-positive M0 macrophages and CD80-positive M1 macrophages in the retrieved implants of rats at 2 WPI. Representative sections showing CD68-positive M0 macrophages in CDS (A), JDS (B), and CSIS(C) and CD80-positive M1 macrophages in CDS (D), JDS (E), and CSIS (F). JDS showed a greater M1 response compared to CSIS followed by CDS, which may indicate the order of biocompatibilities of the grafts. Note the specific negative staining of giant cells (thick black arrows in B). CDS = cholecyst-derived scaffold; JDS = jejunum-derived scaffold; CSIS = commercial-grade small intestinal submucosa; WPI = weeks postimplantation.
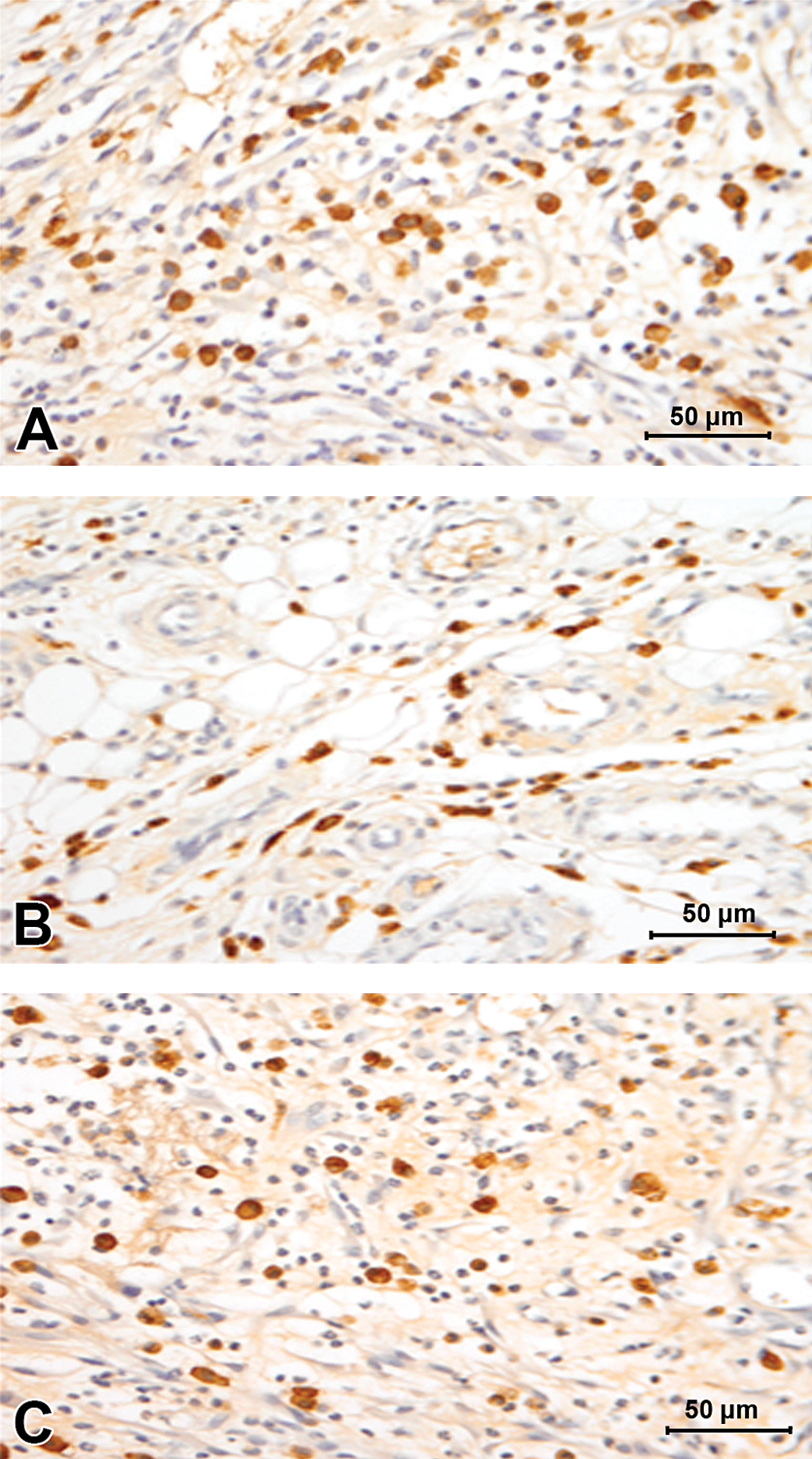
Immunohistochemical expression of CD163-positive M2 macrophages in retrieved implants of rats at 2 WPI. Representative sections showing CD163-positive M2 macrophages in CDS (A), JDS (B), and CSIS(C) retrieved implants in rats at 2 WPI. CDS showed a greater M2 response compared to CSIS followed by JDS, likely to reflect a better biocompatibility. CDS = cholecyst-derived scaffold; JDS = jejunum-derived scaffold; CSIS = commercial-grade small intestinal submucosa; WPI = weeks postimplantation.
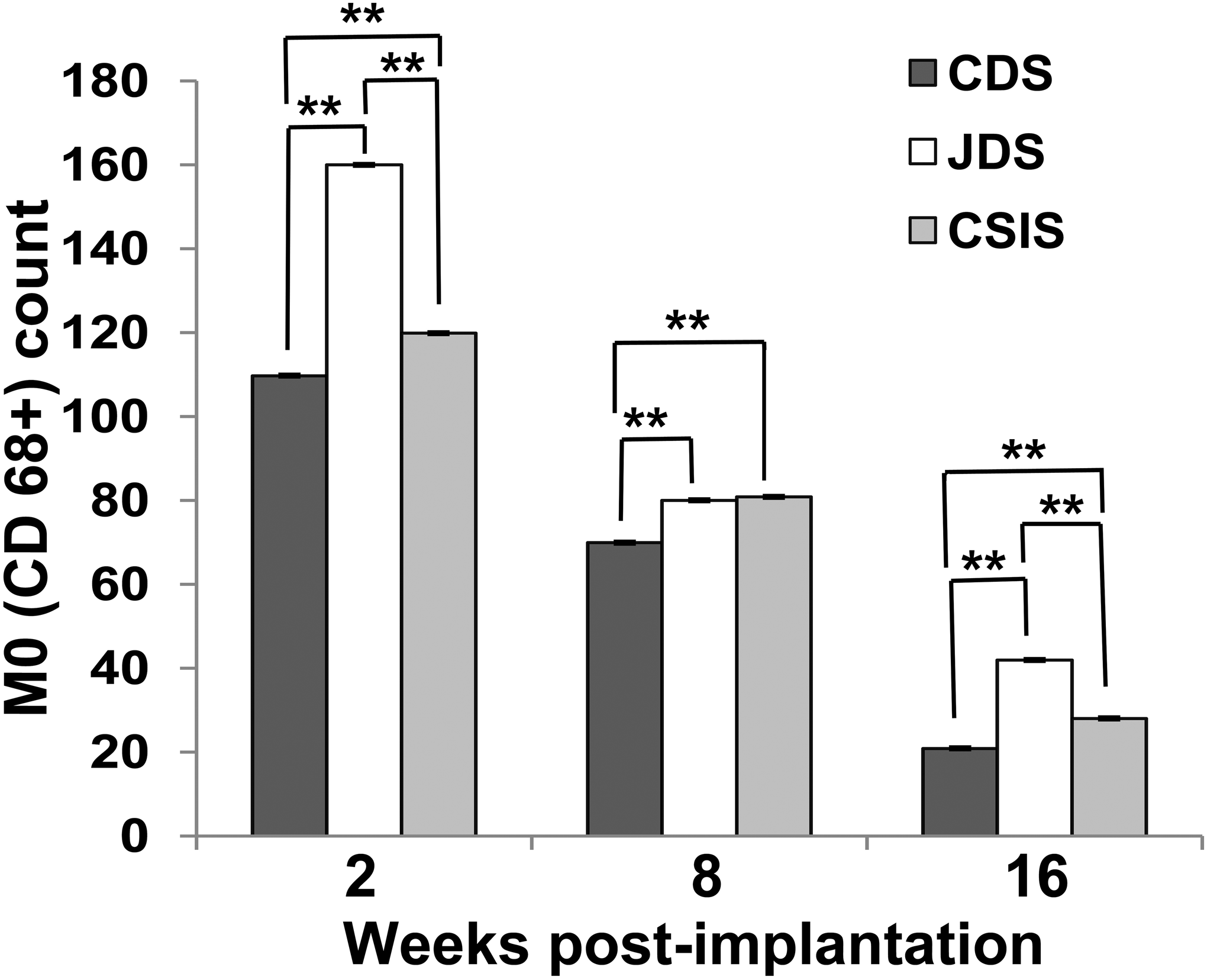
Immunohistochemical evaluation of CD68+ expression for CDS, JDS, and CSIS scaffolds at 2, 8, and 16 WPI (n = 6; mean ± SE; **p < .001). CDS = cholecyst-derived scaffold; JDS = jejunum-derived scaffold; CSIS = commercial-grade small intestinal submucosa; WPI = weeks postimplantation.
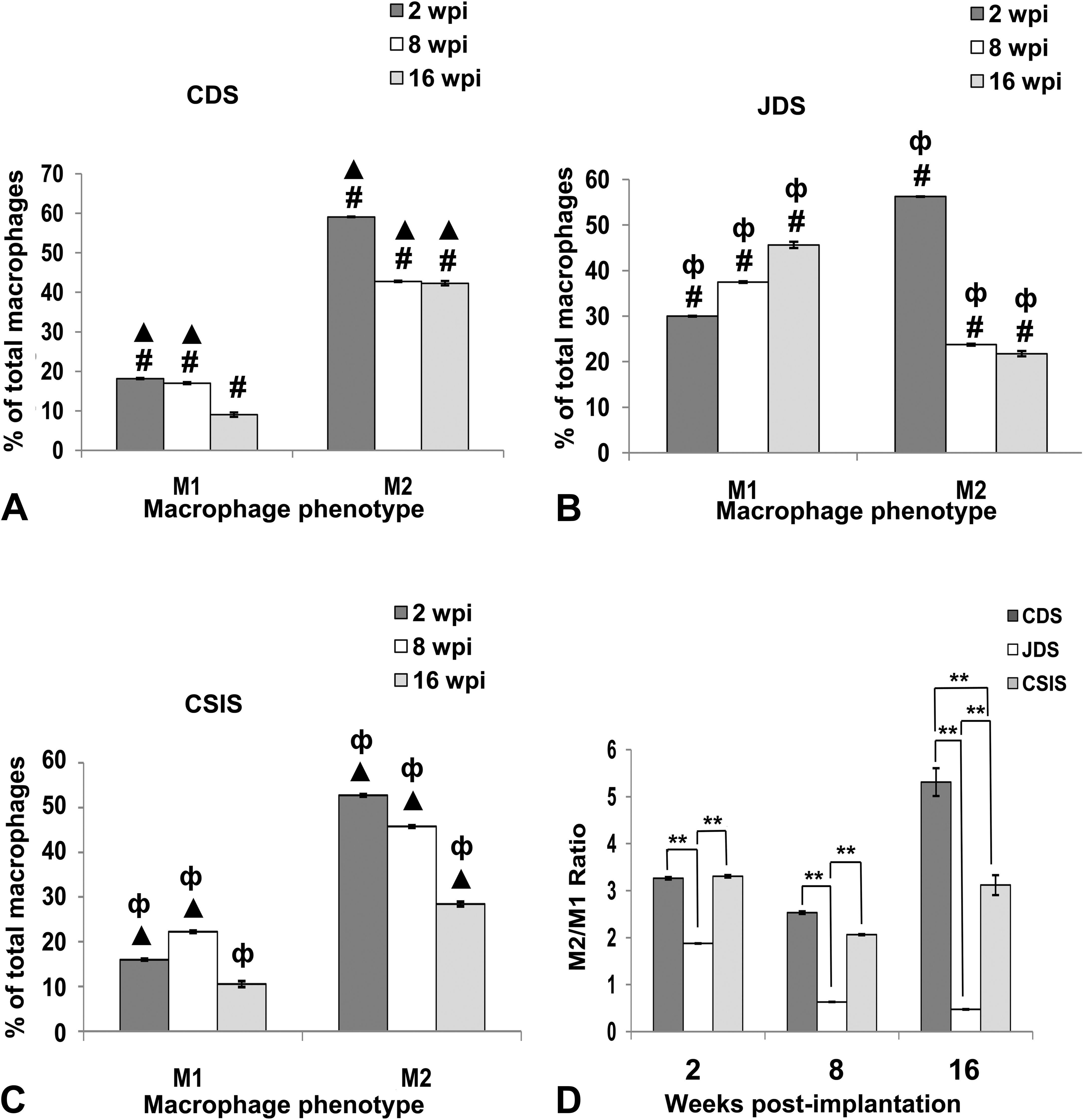
Immunohistochemical evaluation of the percentage of both M1 and M2 macrophages in relation to total macrophages for (A) CDS, (B) JDS, and (C) CSIS scaffolds at 2, 8, and 16 WPI, n = 6; mean ± SE; CDS vs. JDS (#p < .001), CDS vs. CSIS (▴p < .001), JDS vs. CSIS (φp < .001). (D) The ratio of number of CD163-positive M2 macrophages to the number of CD80 M1-positive macrophages (M2/M1 ratio) for CDS, JDS, and CSIS scaffolds at 2, 8, and 16 WPI (mean ± SE, n= 6). **Significant difference between the groups, p < .001. CDS = cholecyst-derived scaffold; JDS = jejunum-derived scaffold; CSIS = commercial-grade small intestinal submucosa; WPI = weeks postimplantation.
The proportion with an M1 phenotype was significantly higher in the JDS grafts throughout the study (Figure 8B). The CSIS had a significantly lower percentage with an M1 phenotype (Figure 8C) compared to the CDS and JDS grafts at 2 WPI (Figure 8D). There was no significant difference with M1 phenotypes between CDS and CSIS at 16 WPI. Further, at 2 WPI, a higher (p < .001) percentage of M2 phenotype was recorded in the CDS grafts compared to other grafts (Figure 8A). The CSIS graft showed a significantly smaller M2 phenotype proportion compared to JDS at 2 WPI (Figure 8D). Later at 16 WPI, the highest percentage of macrophages with an M2 phenotype was observed in the CDS followed by the CSIS graft (Figure 8A and C). JDS had a significantly reduced M2 phenotype percentage compared to the other grafts both at 8 WPI and 16 WPI (Figure 8B). In general, a dominant M2 response was induced in both CDS and CSIS grafts, but CDS induced a greater M2 response compared with the CSIS graft (Figure 8D).
TEM
The characteristics of chronic inflammation such as the presence of macrophages, lymphocytes, mast cells, fibroblasts, plasma cells, and giant cells with neovascularization were prominent in CDS retrieved implants at 2 WPI as seen by TEM. Neovascularization and significant fibroblastic activity with collagen deposition was also apparent in the grafts at this time (Figure 9A–D). Many myoblasts with central nuclei were apparent at 8 WPI (Figure 9E). Organized muscle bundles with good vascularization were observed at 16 WPI. Satellite cells on the surface of newly developed muscle bundles were also apparent in some of the electron micrographs (Figure 9F).
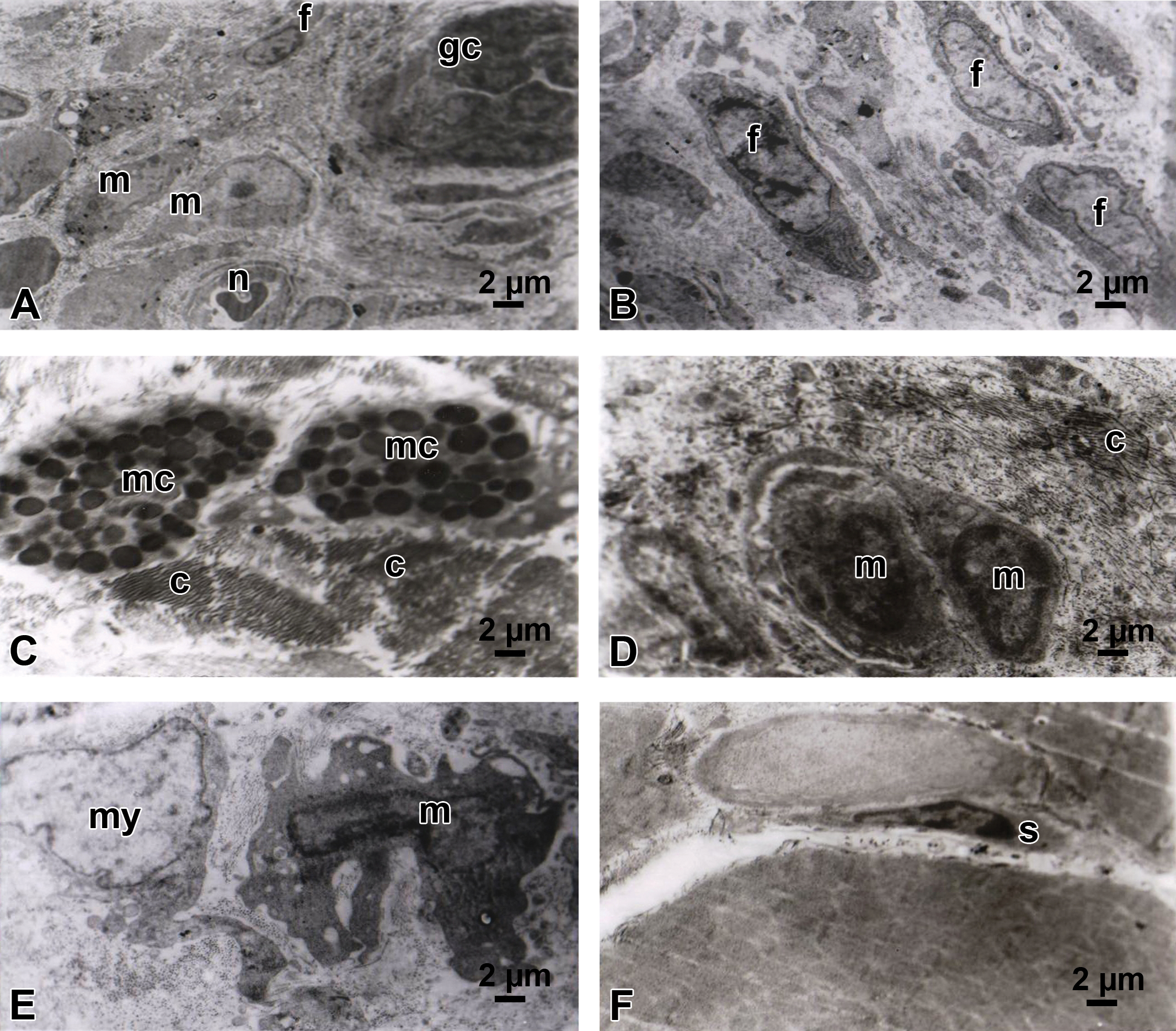
Representative TEMs of the host–graft interface showing the nature of the cellular infiltration following engraftment with cholecyst-derived scaffold. (A) The predominant cells were macrophages, fibroblasts, and occasional giant cells with neovascularization and collagen deposition at 2 WPI. (B) Active fibroblastic activity in the middle of the graft at 2 WPI. (C) Mast cells were visible in the middle of collagen deposition at 2 WPI. (D) Macrophages with numerous pseudopodia and many phagocytic vesicles enclosing degraded graft components in a collagen matrix environment at 2 WPI. (E) A myoblast with a pleomorphic macrophage in the middle of collagen deposition at 8 WPI. (F) Satellite cells were seen on the surface of newly developed muscle bundles at 16 WPI. m = macrophage; n = neovascularization; gc = giant cell; f = fibroblast; mc = mast cell; c = collagen deposition; my = myoblast; s = satellite cell; WPI = weeks postimplantation; TEM = transmission electron microscopy.
Discussion
Over the past few years, acellular biological scaffolds have become the leading choice as grafts for repairing volumetric muscle loss (Corona and Greising 2016). The rat abdominal wall defect model was utilized in this study for evaluating the nature of induced pathology, thereby assessing the potential uses of CDS as a skeletal muscle repair graft. The same model was used for evaluating the local effects of implantation at the intended site of use, required for its safety evaluation as a biomaterial. These safety evaluations were mainly conducted by histomorphological examination as recommended in a widely accepted standard, ISO 10993-Part 6 (2007). The data were supported by immunohistochemistry, investigating subpopulations of macrophages (e.g., M1/M2 macrophages) participating in the local tissue reaction and modulating the graft rejection/acceptance. Concurrent with these studies, organs/tissues modulating host defence were examined for routine toxicologic pathology (ISO 10993-Part 20 2006). In all these studies, considering that any commercial product fabricated out of cholecystic ECM of farm animals is not available, a popular commercially available similar tissue graft, the CSIS was used as the reference material (Rice et al. 2010). The details of the various components of the study, the observations, and the significance are described below. The CSIS being a graft made of small intestinal submucosa, the JDS prepared by the same method as that of the CDS was used as an additional biomaterial for comparison. This also facilitated the assessment of the potential use of JDS, prepared by a nondetergent/enzymatic method (Anilkumar et al. 2014), as a muscle repair graft.
Gross observations made on the retrieved tissue grafts indicated that reasonable integration of the grafts had happened as early as 2 WPI (Figures 1A, 2A, and 3A) during the graft-assisted healing, with significant neovascularization. Complete integration was apparent by 8 WPI (Figures 1C, 2C, and 3C), and the differentiation of grafted tissue from native tissue was difficult by this time and later on (Figures 1E, 2E, and 3E). There was no sign of any necrosis, seroma formation, or graft rejection. This gave a first line of evidence that both CDS and JDS are probable biomaterial candidates for fabricating muscle defects.
Histomorphological evaluation revealed a biomaterial-induced tissue reaction with varying degrees of chronic inflammation at all time points studied (Figures 1 to 3). The data collected through TEM (Figure 9) also suggested a chronic inflammatory reaction. The reaction was very prominent at 2 WPI (Figures 1B, 2B, and 3B) but which was reduced significantly by 8 WPI (Figures 1D, 2D, and 3D) and 16 WPI (Figures 1F, 2F, and 3F). The nature of the histomorphological reaction appeared similar for CDS and CSIS grafts throughout the study periods. On the other hand, despite the similarity in tissue of origin, the JDS showed higher numbers of lymphocytes, macrophages, plasma cells, giant cells, and mast cells compared to other grafts, likely attributable to the differential biocompatibility compared to CSIS (Soiderer et al. 2004; Zheng et al. 2004; Anderson and McNally 2011; Cohen, Joyce, and Kao 2013), probably due to the difference in the method of scaffold preparation. However, these variations in the semiquantitative data on the cellular responses noted here were similar to the nature of tissue reactions observed when the CDS and JDS were used as subcutaneous implants in a rodent model (Muhamed, Revi, Rajan et al. 2015).
Semiquantitative biocompatibility evaluation in experimental animals for local tissue responses at the intended site of application is the gold standard for assessing the potential safety of any biomaterial (Chapekar 1996; Williams 2008). In the present study, the calculated irritancy score as per ISO 10993-Part 6 for CDS and JDS at 16 WPI in comparison with CSIS indicated acceptable scores (Table 1). On the other hand, the JDS appeared to be a mild irritant at 2 WPI and 8 WPI compared to CDS and CSIS. However, considering that muscle grafts (e.g., ventral hernia repair grafts) are usually expected to be long-term implants, both CDS and JDS were deemed as safe biomaterials with respect to the nature of the local tissue reaction in skeletal muscle. These observations are in agreement with the data reported in a rabbit model (Anilkumar et al. 2014). However, in the present study, partial to complete degradation of the grafts occurred by 8 WPI based on histomorphology. Hence, data on mutagenicity and carcinogenicity are desirable before any clinical evaluation of CDS- and JDS-based biomedical devices as a long-term graft material (ISO 10993-Part 1 2009).
No fibrous tissue capsule (Thevenot et al. 2011) formed around the xenografts at any point and the material appeared to have degraded over a period of time. Some collagen deposition was observed in the implanted area, the extent of which was measured as a fibrosis score in this study. These appeared to have disappeared as the tissue remodeling progressed by 16 WPI. Remnants of the biomaterials were scanty by 8 WPI and thereafter. In the present study, the CDS and JDS were used as single sheets of scaffold, and the mechanical strength has already been determined previously for these sheets (Anilkumar et al. 2014). Although the strength was sufficient for repairing abdominal hernias in rats, it was not evident from this study whether the material had sufficient mechanical strength for repairing abdominal hernias in human beings or larger animals. Therefore, the preparation of multilaminated sheets as performed with small intestinal submucosa (Badylak et al. 2008) may be required for augmenting mechanical strength before using CDS/JDS as a hernia-repair graft in larger animals and human beings. Nevertheless, these materials appear to be candidate biomaterials for fabricating grafts intended for repairing VML.
In reality, commercial products fabricated out of animal tissue/organs are known to cause many complications associated with graft rejection; biomaterials of animal origin are potent immunogens (Kalota 2004; Ho, Witte, and Bird 2004; Zheng et al. 2005; Petter-Puchner et al. 2006; John et al. 2008). Therefore, it has been argued that a study of the nature of local immunological reactions is important when evaluating biomaterials of mammalian origin. Despite the similarity in the biocompatibility reaction of CDS and CSIS, differential immunogenic reactions have been revealed by phenotyping of the various immunocompetent cells participating in the tissue reaction (Muhamed, Revi, Rajan, Geetha et al. 2015). Hence, the histomorphological observations were supplemented with immunohistochemistry (Figures 5 and 6) and electron microscopy (Figure 9). Macrophages, the key players in the inflammatory response and immunoregulation, are considered to be crucial regulators of tissue remodeling and vascularization (Wynn and Vannella 2016; Olingy et al. 2017). Macrophage activation, releasing pro-inflammatory and anti-inflammatory cytokines, resulting from biomaterial implantation leads to the classification of macrophages into M1 (classically activated/ pro-inflammatory) and M2 (alternatively activated/reparative) macrophages that are required to support tissue regeneration (Martinez et al. 2008). Transition to an M2 phenotype is associated with constructive tissue remodeling. In the present study, the presence of the 2 immunophenotypes of macrophages was studied, as suggested in previous studies (Badylak et al. 2008; Brown et al. 2012). Generally, the macrophage response against the CDS was lower than those against CSIS or JDS grafts (Figure 7). The data (Table 1; Figures 7 and 8) support the belief that biomaterials with similar biocompatibility, based on local tissue responses on implantation, may cause differential macrophage responses (Muhamed, Revi, Rajan et al. 2015; Muhamed, Revi, Rajan, Geetha et al. 2015). Therefore, rather than as optional procedure as per ISO 10993-Part 6, it may be desirable to consider immunophenotyping for immunocompetent cells as a standard feature for biocompatibility evaluation of materials of animal origin or those materials suspected to have antigenic components.
The CDS is known to induce an M2 macrophage predominated tissue remodeling reaction in rat subcutaneous tissue (Muhamed, Revi, Rajan, Geetha et al. 2015), probably due to the differential protein content in the ECMs of gall bladder and jejunum (Muhamed et al. 2017). A prominent M2 macrophage response over M1 macrophages in tissue reactions is considered as a pro-regenerative response rather than a pro-inflammatory response (Brown et al. 2012). In this study, quantitative data were collected (Figures 7 and 8) to confirm the qualitative observations on the relative proportions of M1 and M2 macrophages. Considering the expertise of the laboratory in immunophenotyping, selection of antibodies from previous experience (Muhamed, Revi, Rajan et al. 2015; Muhamed, Revi, Rajan, Geetha et al. 2015), and maintenance of appropriate controls, the data certainly provide for further insights into the nature of the macrophage responses to the bioscaffold grafts. The CDS graft showed a higher M2/M1 ratio compared to the other 2 grafts. The JDS graft showed the lowest ratio, whereas the CSIS graft showed an intermediary ratio throughout the study. Moreover, the nature of the M2/M1 macrophage reaction varied over time. The initial M1-type response (2 WPI) observed around all the grafts may be related to a persistent acute pro-inflammatory reaction. From 8 WPI, CDS had a less pronounced inflammatory response compared to JDS and CSIS grafts. At 16 WPI, a lower macrophage response was observed in CDS grafts compared with the other grafts of which JDS group showed the highest response. Based on these observations, it can be surmized that the preferential M2 macrophage–mediated pro-regenerative response of CDS grafts has a crucial advantage as a candidate biomaterial for fabricating scaffolds for tissue engineering applications. However, in the present study, the sum of the percentage of M1 phenotype-positive cells and M2 phenotype-positive cells did not reach 100% for all treatment groups at any time point, which suggests the presence of a subset of CD68-positive macrophages that expressed neither M1 (CD80+) nor M2 (CD163+) surface markers. Such cells might not have been stimulated to undergo activation and polarization toward an M1 or M2 phenotype. Similar findings were reported by earlier workers (Badylak et al. 2008; Brown et al. 2012).
The CDS and CSIS grafts showed greater numbers of fibroblasts at the early phase that fell to a low level later on (Table 1). Fibroblast proliferation is responsible for new ECM deposition. The tissue necrosis observed in JDS grafts might be related to a higher percentage of M1 macrophages at the late time points. In reality, the CDS and CSIS grafts were comparable in their tissue fibrotic response (Figures 1B and 3B), which was negligible at 2 WPI and 8 WPI while the response was higher in JDS at all the time points including 16 WPI. Compared to the CSIS graft, CDS showed a significantly lower fibrotic response at 16 WPI (Figure 2F). Allman et al. (2001) and Gilbert, Freund, and Badylak (2009) demonstrated that the presence of DNA fragments within scaffolds following the decellularization process could produce fibrosis due to an increased M1 differentiation of recruited macrophages, resulting in an inflammatory host reaction followed by scar tissue deposition and fibrosis.
Fatty infiltration was noted in several of the grafts and is likely a connective tissue stem cell metaplasia (Schuh 2008) caused by the inflammatory milieu. The CDS and CSIS grafts had negligible fatty infiltration while the JDS graft showed the highest fatty infiltration in the later phases (Figure 2F). Badylak et al. (2008) reported similar findings with implantation of small intestinal submucosa. All of the 3 grafts showed a moderate to marked neovascularization: the greatest response was observed in CDS grafts followed by CSIS with the least response in the JDS grafts.
Considering xenografts of mammalian origin, a preliminary immunotoxicity study was conducted based on ISO 10993-Part 20 (2006; Schuh 2008). The weights of the lymphoid organs were not recorded, but there was no variation in histomorphology of these organs between groups. The lymphoid tissue of all the rats showed a normal structure which along with normal bone marrow/smear studies indicated that the degradation products of all the bioscaffolds had not adversely affected animal health, further indicating the biocompatibility of these implant materials. It can be concluded that the release of peptides, growth factors, and contaminants such as endotoxins, nucleic acids, extraction aids, and other proteins from the scaffold materials, if at all any, was not sufficient to cause any adverse effects in these immune organs (data not shown).
This study has a few limitations. Firstly, the study used only 1 marker each for identifying M0, M1, and M2 macrophages as has been practiced recently for evaluating the nature of the macrophage response to hybrid ECM polypropylene mesh (Faulk et al. 2014). However, the use of multiple markers would have facilitated a firmer conclusion on the biased M2 macrophage reaction in response to CDS in the muscle. Secondly, we did not evaluate cytokines or chemokines in determining the nature of the cytokine milieu in the host foreign body reaction. Moreover, further experiments are needed to determine the detailed molecular mechanisms through which bioscaffolds regulate an M2 polarization. Our findings suggest that an M2 modulation may define the early biocompatible safety evaluation that may be a strategy to develop novel biomaterial candidates for clinical purposes. The present study may also aid in the development of new strategies based on muscle regeneration to treat abdominal wall defects such as umbilical and incisional hernias in human and veterinary patients.
Conclusion
This study has shown that the CDS and JDS grafts prepared by nondetergent/enzymatic methods are candidate biomaterials for fabricating muscle repair grafts. The former induced a faster graft-assisted healing of skeletal muscle defects than the latter by promoting muscle regeneration, angiogenesis, and a tissue remodeling reaction mediated by M2 macrophages. In comparison with a commercial-grade bioscaffold prepared from porcine small intestinal submucosa, they were biocompatible with respect to a local tissue response. Moreover, there was no histomorphological evidence of toxicity in mandibular lymph nodes, popliteal lymph nodes, Peyer’s patches, spleen, thymus, or bone marrow.
Footnotes
Acknowledgments
The authors acknowledge financial support from the Indian Council of Agricultural Research, New Delhi, as senior research fellowship for the first author. The authors acknowledge the dean, College of Veterinary and Animal Sciences, Mannuthy, for his support. They are also grateful for the assistance of Dr. C. Sunanda, PhD, for the statistical analysis.
Author Contribution
Authors contributed to conception or design (DB, NN, TA); data acquisition, analysis, or interpretation (DB, NN, SV, NV, SG, MJA, SE, MRA, AS, TA); drafted the manuscript (DB, NN, MRA, TA); and critically revised the manuscript (DB, NN, SV, NV, SG, MJA, SE, MRA, AS, TA). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Part of the research work was supported by Sree Chitra Tirunal Institute for Medical Sciences and Technology, Thiruvananthapuram 695012, India, Kerala (TRC project No 8144) and Kerala State Council for Science Technology and Environment (No. 1275/2014/KSCSTE).
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
