Abstract
Absorbable metallic implants have been under investigation for more than a century. Animal and human studies have shown that magnesium (Mg) alloys can be safely used in bioresorbable scaffolds. Several cardiovascular and orthopedic biodegradable metallic devices have recently been approved for use in humans. Bioresorbable Mg implants present many advantages when compared to bioabsorbable polymer or nonabsorbable metallic implants, including similar strength and mechanical properties as existing implant-grade metals without the drawbacks of permanence or need for implant removal. Imaging visibility is also improved compared to polymeric devices. Additionally, with Mg-based cardiovascular stents, the risk of late stent thrombosis and need for long-term anti-platelet therapy may be reduced as the host tissue absorbs the Mg degradation products and the morphology of the vessel returns to a near-normal state. Absorbable Mg implants present challenges in the conduct of preclinical animal studies and interpretation of pathology data due to their particular degradation process associated with gas production and release of by-products. This article will review the different uses of Mg implants, the Mg alloys, the distinctive degradation features of Mg, and the challenges confronting pathologists at tissue collection, fixation, imaging, slide preparation, evaluation, and interpretation of Mg implants.
The investigation of magnesium (Mg) alloys as cardiovascular and orthopedic implants is not a new concept and the quest has been ongoing for more than a century (Witte 2010). The implantation of biodegradable Mg was described for the first time in 1878 by the physician Edward C. Huse, who used Mg wire ligatures successfully to stop bleeding vessels (Witte 2010). Bioabsorbable implants, including both polymers and metals, have attracted increasing interest in the past years. The main advantage is a complete disappearance of the device once it becomes no longer useful without the need for a follow-up surgery to remove the implant after the tissue has healed sufficiently (James 2016). This could mean a reduction in lifelong problems caused by permanent implants, including long-term endothelial dysfunction, permanent physical irritation, chronic local inflammatory reactions (Moravej and Mantovani 2011), and stress shielding by permanent bone plates. Polymers are most prevalent in the current medical market, though metallic Mg-based (Zhang et al. 2010), Fe-based (Peuster et al. 2001), and Zn-based (Vojtěch et al. 2011) alloy implants have been proposed as better biodegradable materials for load-bearing applications due to their superior combination of strength and ductility over polymers. Research in metallic implants focuses on biodegradable implants that can dissolve in the biological environment after a predetermined length of time of functional use. However, the main concern for degradable metals is the degradation products that might induce local or systemic toxicity (Purnama et al. 2010).
Definitions
Magnesium-based Implant Applications
Cardiovascular (Stents)
Heublein et al. (2003) were the first to investigate the idea of using Mg alloys for cardiovascular stents. Mg stents have been developed to overcome the limitations of permanent metallic stents, which include late-term restenosis, delayed re-endothelialization, thrombogenicity, permanent physical irritation, inhibition of vasomotion, chronic inflammatory local reactions, long-term stent crushing and fractures, inability to adapt to growth in young patients, and complications associated with the aggressive anticoagulant regimen (Erne, Schier, and Resink 2006; Hofma et al. 2006). The ideal Mg biodegradable stent should possess sufficient mechanical properties, appropriate degradation rate, excellent hemocompatibility and biocompatibility, and drug delivery capacity. The stents should degrade at a slow rate for the first 6–12 months to maintain optimal mechanical integrity during arterial vessel remodeling. Afterward, the degradation should progress at a sufficient rate without causing an intolerable accumulation of degradation products around the implantation site, potentially leading to neointimal formation (Hermawan, Dubé, and Mantovani 2010; Moravej and Mantovani 2011). Pure Mg stents do not suit these requirements as they show rapid degradation rates, release and accumulation of hydrogen gas, and local alkalosis due to the corrosion of the Mg that can lead to vascular damage. Additionally, inferior mechanical properties or inadequate stent designs have contributed to early stent fatigue fractures, resulting in negative mechanical interference with the vessel wall (Waksman et al. 2006; Drynda et al. 2008).
Orthopedic Devices (Plates, Screws)
Currently, permanent and inert metals like titanium (Ti) alloys and stainless steel remain the gold standard for internal fixation devices; however, these materials are associated with various long-term complications such as interference with skeletal growth (particularly for pediatrics), tissue irritation, infection, interference with radiological imaging, stress shielding, and unfavorable esthetics (primarily for craniofacial implants). These complications may necessitate invasive removal surgeries, thereby increasing patient burden and risk and draining valuable hospital resources (Chou et al. 2013; Staiger et al. 2006; Hanson, van der Werken, and Stengel 2008; Busam, Esther, and Obremskey 2006). To mitigate these concerns, bioabsorbable polymer devices have been developed; however, their poor mechanical properties (Zheng, Gu, and Witte 2014) and long-term foreign body reactions during polymer degradation, likely due to their acidic degradation products (Chou et al. 2013), often limit them as viable options for orthopedic applications. The ideal orthopedic implants should have desirable biocompatibility and nontoxicity of degradation products, should also provide sufficient mechanical strength to withstand various biomechanical forces (yield strength, elastic modulus, and, ultimately, tensile strength for load-bearing applications), low weight, good wear resistance, a moderate degradation rate that is adapted to the fracture healing process, and excellent osseointegration (Waizy et al. 2013; Radha and Sreekanth 2017). Because Mg has mechanical properties similar to that of natural bone (density, elastic modulus, and compressive yield strength), it is potentially a better orthopedic implant for the fixation of bone fractures and pseudoarthrosis (Witte 2010) than currently used (Ti) alloys and ceramic biomaterials (Staiger et al. 2006; Witte et al. 2005). Pure Mg implants were first used in 1906 by Lambotte; however, the pure Mg plates needed to be removed due to extensive gas cavities, local swelling, and heavy pain as the pure Mg implant degraded rapidly
Dental Reconstruction (Dental Membranes)
Resorbable dental membranes are used in guided bone regeneration to enhance new bone formation in conjunction with bone graft substitutes. Biodegradable barrier membranes made of pure Mg or Mg alloys are currently under investigation for clinical indications for dental applications of guided bone regeneration. It is hypothesized that an Mg membrane may procure a better barrier against the ingrowth of soft tissue in the regenerating bone than the collagenous materials currently used (Lin, Lee, and Yeh 2017).
Safety Concerns
Although corrosion is generally considered as a failure in metallurgy, the corrodibility of certain metals including Mg and iron can be an advantage in their application as degradable implants. Essentially, the biodegradable metals maintain their original structure while the tissue heals and then degrade once the metal scaffold is no longer needed (Moravej and Mantovani 2011).
Degradation of Mg produces corrosion products on the surface of the implant (Zheng, Gu, and Witte 2014; Bowen, Drelich, and Goldman 2014). In the corrosion of Mg, initially Mg reacts with water, eliciting Mg2+, which then undergoes hydroxylation to form magnesium hydroxide (Mg(OH)2) along with magnesium chloride (MgCl2) and hydrogen gas (Perkins et al. 2016). The corrosive reaction of Mg in an aqueous environment produces Mg(OH)2, which is harmlessly excreted via the release of hydrogen gas, and ultimately through urine. The hydrogen is exchanged with the local environment, diffuses through the tissues, and is eliminated by the blood flow and renal system (Salahshoor and Guo 2012).
Depending on their composition, Mg implants could have significant disadvantages of rapid degradation rate with release of by-products, production of hydrogen gas and particulate debris, and the possibility of increased inflammation during the resorption process, which could ultimately result in implant failure. The production of high amounts of hydrogen gas in a short period of time is undesirable, especially when the gas is produced at a rate that is too fast for the host tissue to handle; this could result in accumulation of gas in tissue cavities or formation of subcutaneous gas bubbles (Witte et al. 2005; Staiger et al. 2006; Waizy et al. 2013; Song 2007; X. Gu et al. 2009). Hydrogen gas is a major concern in orthopedic applications because the excessive hydrogen gas is poorly eliminated from bone, thus resulting in the formation of potentially harmful gas pockets that could inhibit implant-bone apposition (Persaud-Sharma and McGoron 2012). Notable amounts of hydrogen gas production are most commonly found with orthopedic implants as the hydrogen evolution in lattice or mesh-like implants (i.e. stents) seems to be of minor importance; however, gas production can also be found with Mg stents (Erne, Schier, and Resink 2006; Heublein et al. 2003; Erbel et al. 2007).
Hydrogen gas production happens in the early stages of Mg alloy degradation, which is dependent on the corrosion rate (Waizy et al. 2013), and correlates with the decrease in implant volume (Berglund et al. 2016). The corrosion rates are not similar in all the anatomical locations and depends on the local blood flow prior to implantation (Willbold et al. 2013). In the bone marrow, fast Mg alloy degradation could be attributed to greater vascularization of the cancellous bone (Berglund et al. 2016; Xu et al. 2007).
Although pure Mg corrodes very quickly, the corrosion rate of Mg can be significantly reduced by alloying, heat treatment, processing, surface modifications, and coatings (Gray-Munro, Seguin, and Strong 2009; Hanzi et al. 2009; Staiger et al. 2006; Salahshoor and Guo 2012; X. Gu et al. 2009; Witte et al. 2005). Mg alloys composed of rare earth elements (such as yttrium and gadolinium), zirconium, manganese, zinc, calcium, lithium, and strontium have been developed (Luthringer, Feyerabend, and Willumeit-Römer 2014). Mg alloys exhibit different degradation rates and different degradation products depending on the alloying element they contain (Salahshoor and Guo 2012; James 2016; Waizy et al. 2013; Ben Hamu, Eliezer, and Wagner 2009; Wang et al. 2007; Ambat, Aung, and Zhou 2000). Alloys composed of Mg, calcium, zinc, and strontium have good corrosion resistance, and thus presumably minor hydrogen production, after implantation into the bone (X. N. Gu et al. 2012; Kuhlmann et al. 2013). Some degradation products may be poorly biocompatible and/or may impair the osteoconductive properties observed with Mg.
Surface treatments, such as protective films and coatings, could prevent the Mg from having direct contact with surrounding fluids (electrolytes) and will thus delay corrosion; therefore, surface modification can be an effective strategy to improve the corrosion resistance of Mg-based implants. Some examples of protective films and coatings are absorbable polymers, absorbable hydroxyapatites, or epoxy resins; these surface treatments prevent the exposure of the substrate Mg to electrolytes in the surrounding fluids (Chiu et al. 2007; Hiromoto and Yamamoto 2009; Gray and Luan 2002; Tamar and Mendler 2008). However, once the coating has fragmented or bioabsorbed, the problem of excessive corrosion remains (Zhang et al. 2010). Figures 5–7 show two different coated Mg alloy stents; the histological appearance of the stent struts in Figure 5 indicates that the Mg degradation/corrosion process is more advanced than in the Figure 7.
It has been shown that the initial degradation of the Mg implants, with formation of degradation and conversion products on the implant surface, appears to block further degradation and limit gas formation. This was attributed to the restricted contact of aqueous solutions with the implant material due to the barrier function of the surface layer containing the degradation and conversion products (Hanzi et al. 2010). In orthopedic implants, corrosion could potentially cause loosening of the implant with loss of mechanical integrity before the tissue can sufficiently heal (Staiger et al. 2006; Song 2007; X. Gu et al. 2009).
Biological Activity: Osteoconduction?
Mg is an essential element that is crucial for bone health (de Baaij, Hoenderop, and Bindels 2015; F. Y. Li et al. 2011) and ∼60% of Mg in the human body is stored in the bone matrix (Staiger et al. 2006). Mg deficiency leads to osteoporosis, and supplementation with Mg is known to be beneficial in patients with osteoporosis (Castiglioni et al. 2013). Some reports have shown that Mg devices have the potential to facilitate fracture healing and enhance bone formation, with increased mineral apposition, bone mass, and bone mineral density around Mg implants in bone; this phenomenon is not typically seen with bioabsorbable polymer or permanent metal devices (Witte et al. 2006; Z. Li et al. 2008; X. N. Gu et al. 2012; Witte et al. 2005; Janning et al. 2010; Yang et al. 2012; Witte et al. 2007, part II; Chaya et al. 2015). New bone formation can be seen adjacent to the Mg implants and/or degradation products without significant tissue reaction (Figure 1). Possible causes for this enhanced bone growth are the production of Mg(OH)2 as a corrosion product and neuronal calcitonin gene-related polypeptide-α (CGRP). The findings of Janning et al. support the hypothesis that the major corrosion product, Mg(OH)2, from any Mg alloy is the major origin of the observed enhanced bone growth
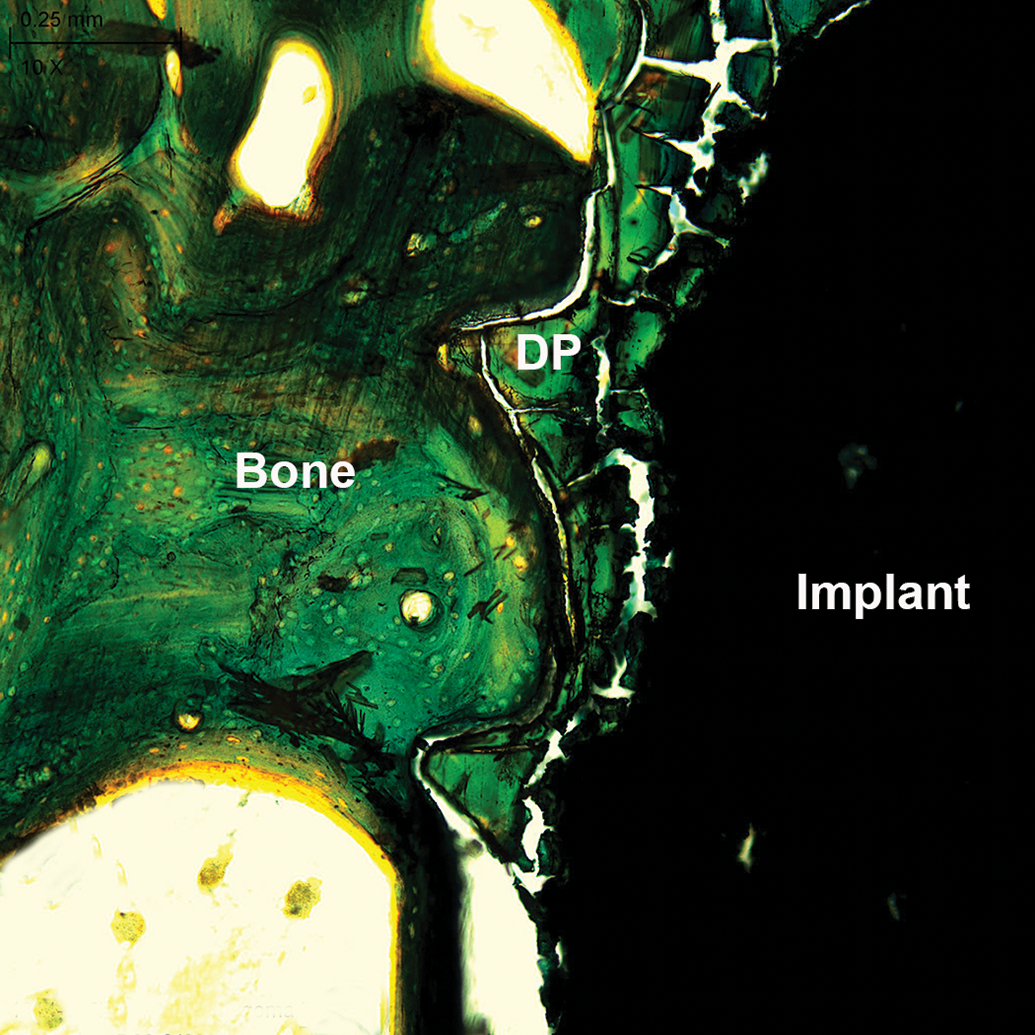
Osteotomy model in pigs implanted with magnesium (Mg) alloy plates and screws, zygomatic bone embedded in MMA, microground with the EXAKT Micro Grinding System, Goldner’s Trichrome stain, original objective 20×. New bone formation adjacent to the implanted Mg screw and degradation products (DP).
Challenges in the Evaluation of Magnesium Devices
Tissue Collection of Mg Stents and Orthopedic Implants
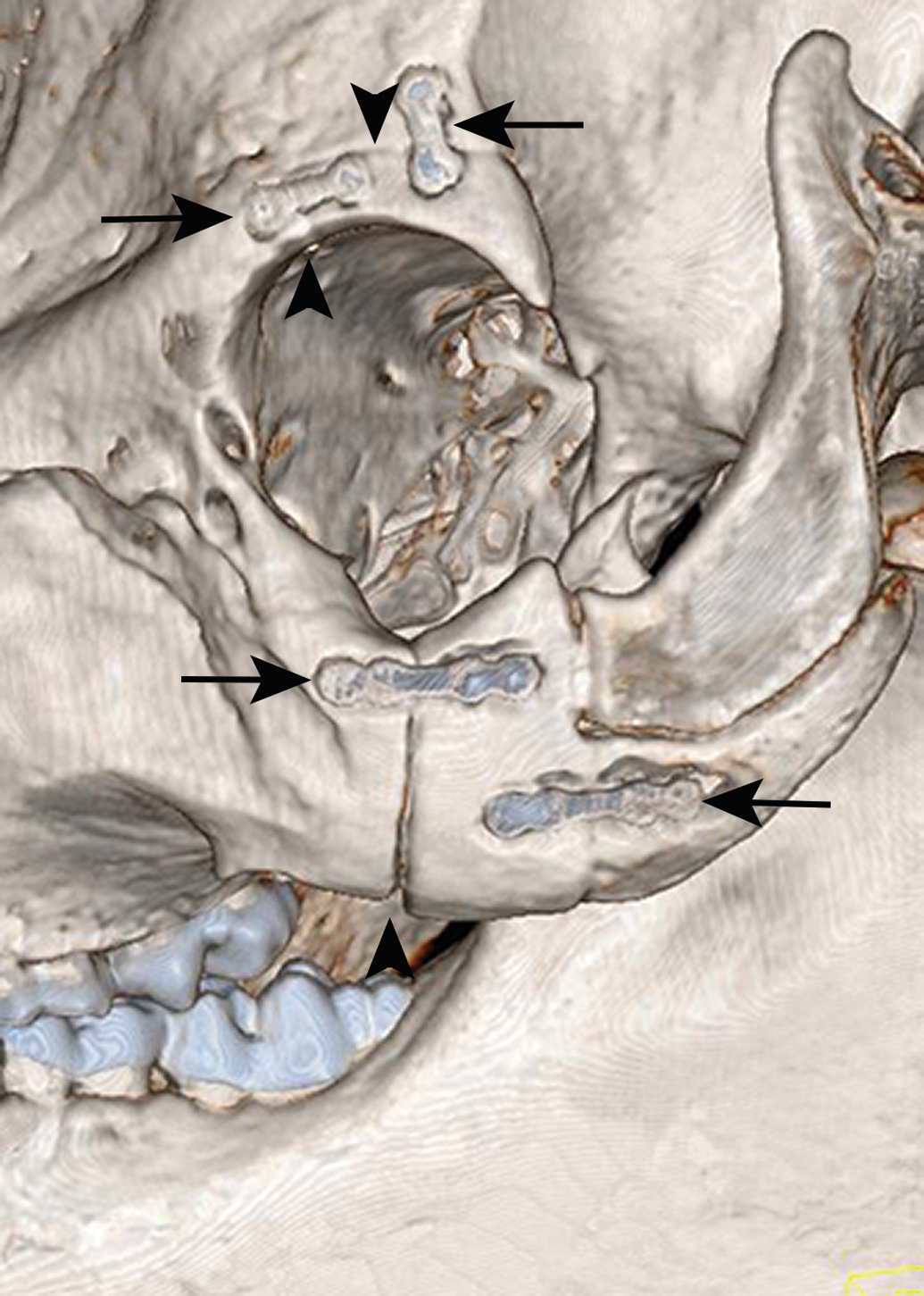
Because the Mg stents are more fragile than non-resorbable metallic stents, implantation of Mg stents in thoracic or femoral arteries of pigs should be avoided as their regular activity (i.e., lying down and walking) could potentially deform, fracture, or otherwise damage the Mg-based stent. Harvesting of Mg-based cardiovascular and orthopedic implants can present challenges, as the exact location of the implant sites can be difficult to visualize at late time points. In these cases, in-life or terminal radiographs or CT scans can be used to determine the precise location of the implant sites at necropsy (Figure 2), and the explanted tissue should include several millimeters of normal tissue on all sides of the expected implantation site. During macroscopic examination of the implant and implantation site, details about the implant location and implant appearance are described and photographed.
At tissue collection of Mg stents implanted in coronary, iliac/femoral, or thoracic arteries, the treated vessel sections should be handled with extra care to avoid fractures or deformation of the stents; this is particularly important at later time points with partially degraded stents. Perfusion fixation is the preferred fixation method for the stented arteries. For the coronary arteries, the heart is excised, pressure perfused with saline or Lactated Ringer’s Solution (LRS) through the aortic root with cross-clamping of external vessels, and subsequently pressure perfused with 10% neutral buffered formalin (NBF) for fixation. The heart is then immersed in NBF and the stented coronary arteries are removed from the heart after overnight fixation. For the iliac/femoral arteries, a lower body perfusion (swine) or full body perfusion (rabbits) is performed using saline or LRS first, followed by NBF for fixation. The treated arteries are excised, including a margin of proximal and distal nontreated tissue, and immersed individually in NBF. Stented internal thoracic arteries, including a margin of proximal and distal non-treated tissue, are carefully excised, rinsed with saline or LRS, and immersed in NBF. Stented vessels should be exposed to NBF at least overnight before further processing.
The bone implanted with an orthopedic device can either be harvested and immersion fixed or, alternatively, perfusion-fixed
Implant Imaging: Radiography, microCT
Advanced imaging is often needed for visualization and evaluation of implanted devices. Additional identification and visualization of the implant placement before further processing can be achieved with high-resolution radiographs of the whole fixed heart (Figure 3), the stented artery, or implanted bone. Due to their greater radiopacity, Mg implants are usually easier to visualize than polymeric implants, although, at late time points, radiopacity of the Mg-based implant is low due to biodegradation, and the appearance of the device may be similar to the surrounding tissue. High-resolution radiographs and microCT scans can also be used for identification and documentation of stent or implant fracture or other abnormalities.
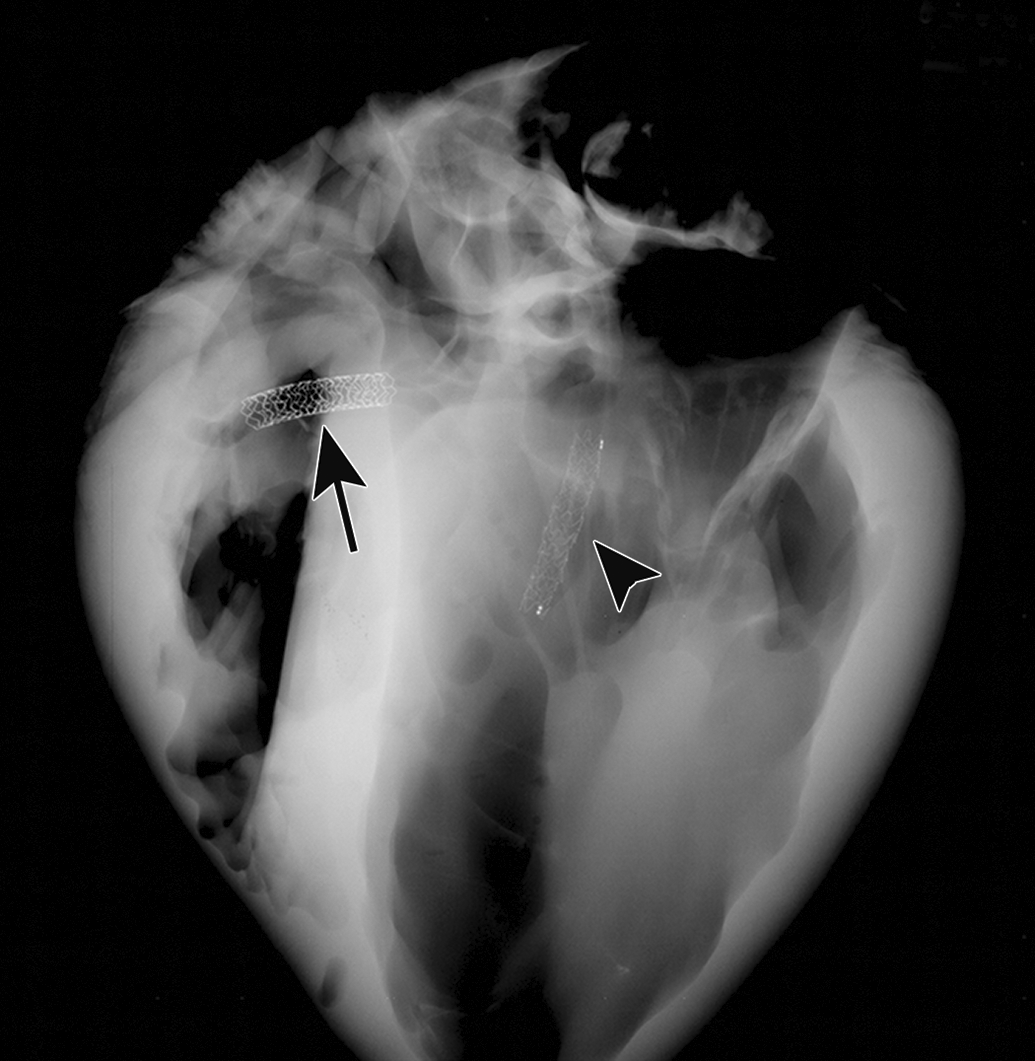
High-resolution radiograph of a porcine heart implanted with a magnesium (Mg) alloy stent in the left anterior descending artery (LAD, arrowhead) and a cobalt chromium stent in the right coronary artery (RCA, arrow); coronary implantation model in healthy swine. Note that radiopaque markers are located at the peripheral edges of the Mg alloy stent.
Postfixation Trimming of Implanted Tissues
Since the corrosion reaction of Mg occurs in an aqueous environment, postmortem corrosion and gas formation can potentially occur with Mg implants that are immersed in NBF. Because the implants remain in an aqueous environment, they continue to degrade and generate some hydrogen gas after the death of the animal. Therefore, the stented arteries and implanted bones with Mg implants should not remain in formalin for an extended period of time. Only 24 hr should elapse between the immersion of the heart/stented arteries in NBF and initiation of the tissue processing procedures. When it is not possible to start the processing promptly after the 24 hr of fixation time, the explanted treated vessel segments should be transferred into a nonaqueous fluid, such as 100% glycerol, for temporary storage until processing and embedding in the appropriate medium is possible.
Although it is known that corrosion of the Mg implant will occur, the bone specimens for orthopedic implants need to be fixed for a long period of time as NBF infiltrates mineralized tissue more slowly than nonmineralized tissue. Therefore, a fixation period of 24 hr is generally insufficient for orthopedic samples. The penetration of formalin fixative is approximately 2–3 mm in a 24-hr period; therefore, it is important to have either very small bone samples or trim and therefore reduce the size of large bone specimens as soon as possible after immersion in the fixative, generally after 24 hr of fixation, to allow for better fixation (Jenkins and Burg 2010). The implanted bone should be fixed at room temperature for approximately one week and then processed with the appropriate resin.
Histology preparation for pathology assessment
Bone decalcification should be avoided as complete degradation of the Mg implant may occur during the decalcification process. For new Mg-based implants, pilot studies or cadaveric trials with a low number of practice devices inserted within the appropriate tissue followed by different histological processing techniques and embedding media should be performed. The sectioning technique for plastic-embedded specimens should also be investigated ahead of time; whether to generate microtomed, thin sections versus ground sections via micro-grinding and micro-polishing needs to be determined. The resulting slides are stained and examined, and depending on the results, the best procedures are chosen for future processing of this particular device. The goal is to match the hardness of the embedding medium to the hardness of the implant and surrounding tissue in order to produce quality sections without artifacts. Due to the hardness of the Mg implant, the tissues/implant should be embedded in plastic resin (methyl methacrylate [MMA], Technovit, or Spurr/Epon) and, depending on the implanted tissue, will then be either cut with a conventional microtome (in the case of stented arteries (Figure 5) or microground with the EXAKT system (for orthopedic implants). Determination of the embedding medium is based on the type of implant and the presence/type of coating. Figure 6 shows zygomatic bone implanted with Mg alloy plate and screws; the bone blocks were embedded in MMA, microground with the EXAKT Micro Grinding System and stained with Goldner’s Trichrome. In order to choose the best embedding resin, preliminary tests should be done to ensure that the device and its coating, when present, are compatible with the processing and/or embedding medium as some surface treatments/coatings can be dissolved and removed from the surface of the device during processing and/or embedding with a specific medium. Removal of implant materials before histological evaluation should not happen, as it may remove most of the tissue reaction and distort the implant space, particularly when there is an important inflammatory reaction or presence of tissue ingrowth into the implant (Schuh 2008).
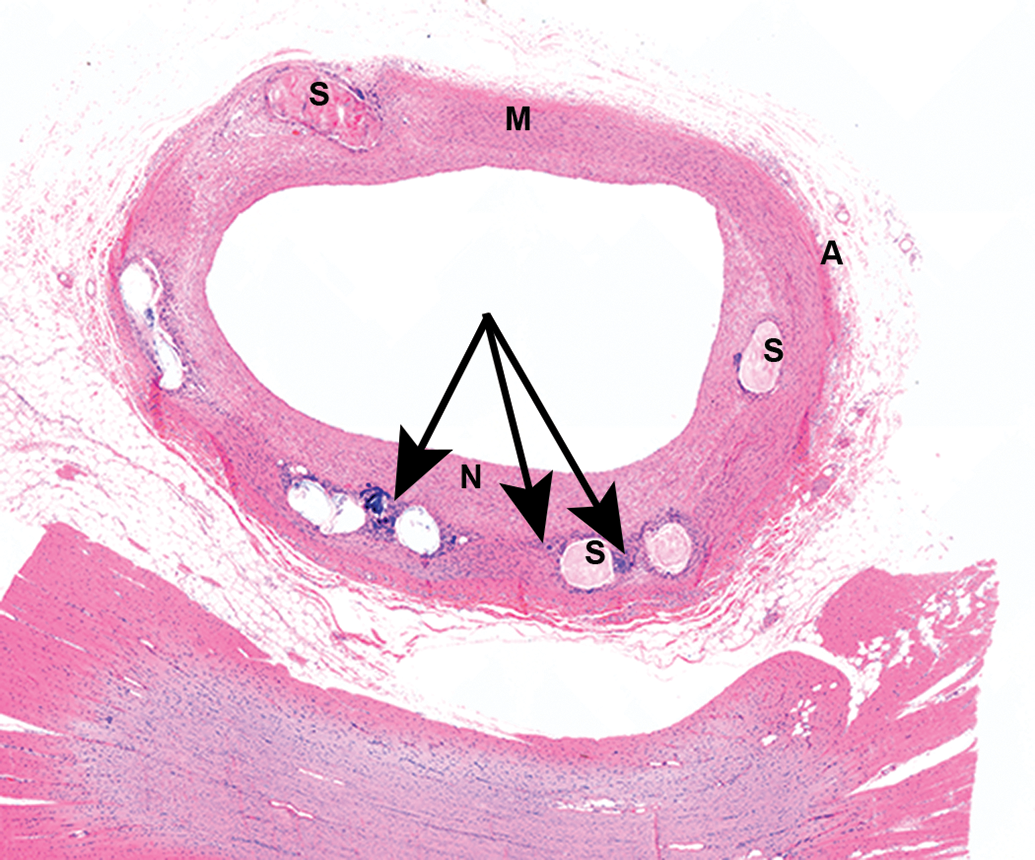
Coronary artery from a healthy swine implanted with a coated magnesium (Mg) alloy stent, embedded in methyl methacrylate (MMA), cut with microtome, H&E stain, low magnification; 12 weeks following implantation. Chronic inflammatory reaction (arrows) is seen within the neointima (N) around the struts (S); media (M), adventitia (A).
Degradation assessment
During degradation/corrosion of the Mg implants, there will be gas formation, release of by-products, and most likely, an increase in inflammation. Complete degradation of the Mg implant needs to be assessed and will require multiple time points up to and beyond the estimated degradation time of the implant; the degradation time will be highly variable depending on the alloy and the presence or absence of a surface coating. To evaluate the degradation, a baseline at time 0 or at a very early time point (i.e., week 1 or 2) when degradation has not occurred is required to allow for histopathological evaluation and scoring of the degradation. Degradation can be assessed histologically either semi-quantitatively during histopathological evaluation using a scoring scale from 0–4 or quantitatively with histomorphometric (2D) or microCT (3-D) measurements (Huehnerschulte et al. 2011).
Histopathological evaluation
Local safety assessment
Local tissue response to the Mg orthopedic implants and their degradation products is assessed semi-quantitatively using the ISO 10993-6 standard for Biological Evaluation of Medical Devices irritancy/reactivity scoring guidance (ISO 10993-6, 2016). The biocompatibility of Mg can vary depending on the tissue in which it is implanted and its rate of corrosion. Several authors have reported positive results for the
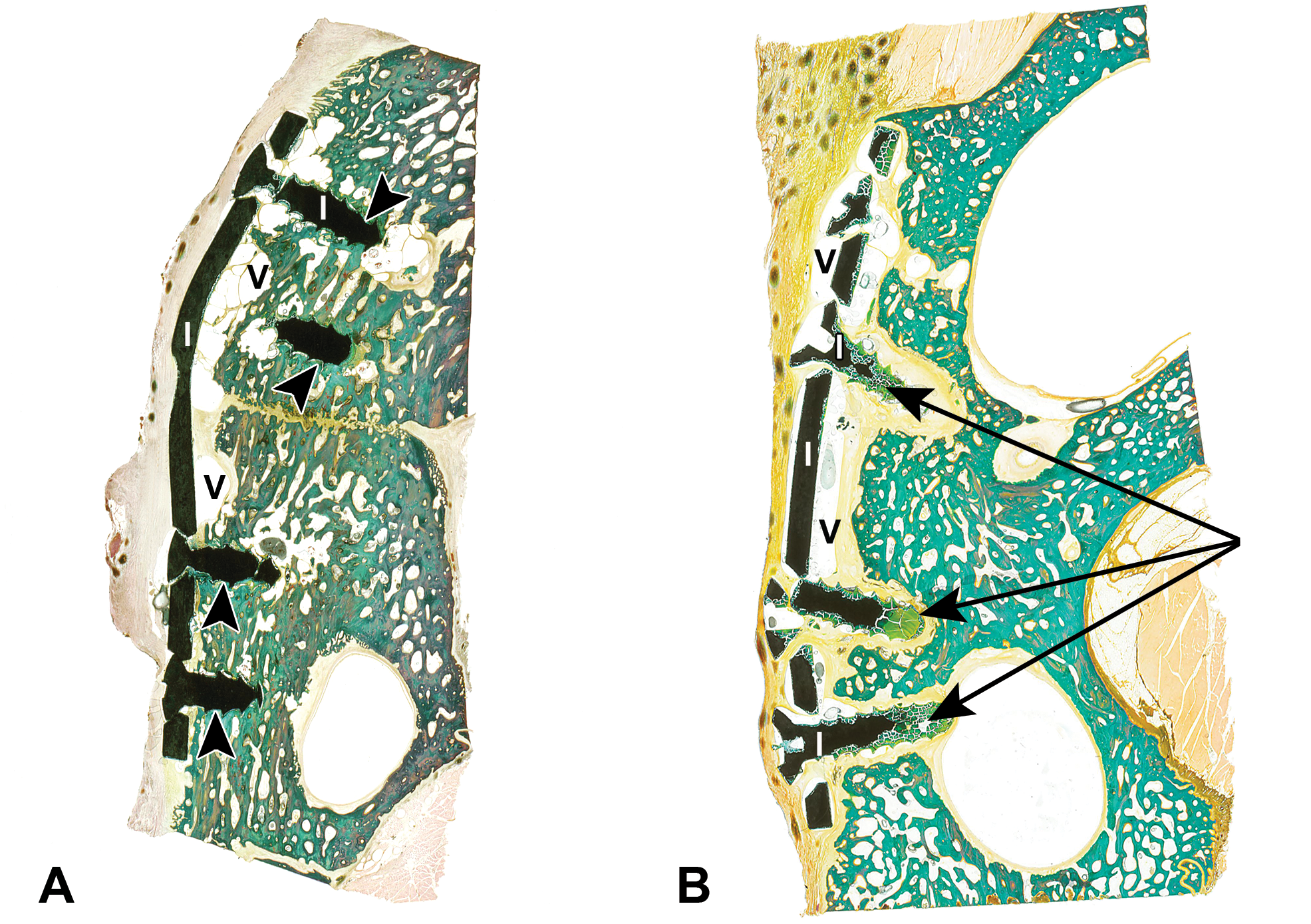
Osteotomy model in pigs implanted with magnesium (Mg) alloy plate and screws, zygomatic bone embedded in methyl methacrylate (MMA), microground with the EXAKT Micro Grinding System, Goldner’s Trichrome stain. (Reproduced and modified with permission from
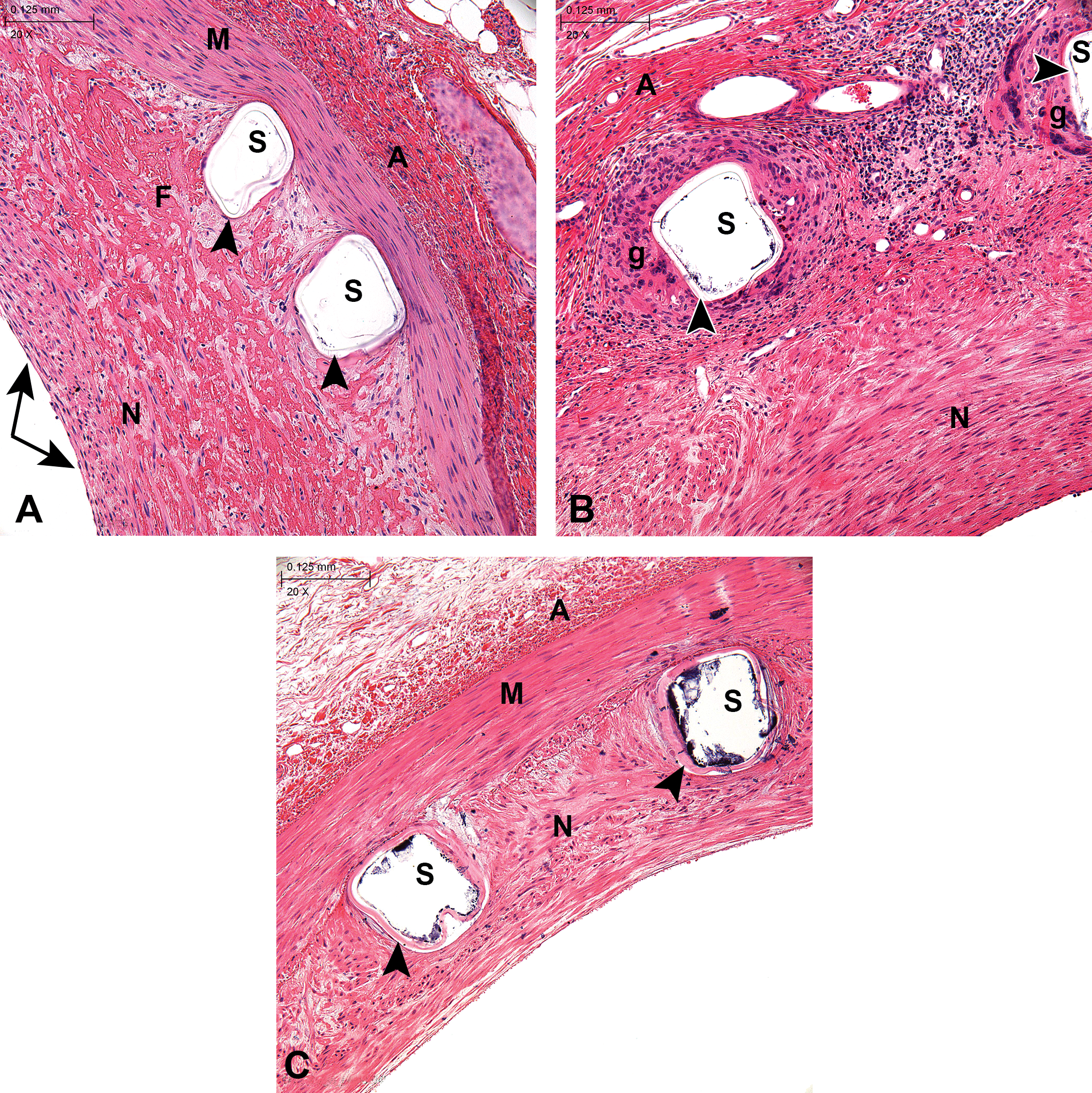
Coronary arteries from healthy swine implanted with a coated magnesium (Mg) alloy stent at 28 days (A), 90 days (B), and 180 days (C) following implantation. methyl methacrylate (MMA) embedding, hematoxylin & eosin stain, 20×. Endothelium (arrows), neointima (N), media (M), adventitia (A), struts (S), coating (arrowheads), and granulomatous inflammation (g) are shown. Note that the coating, translucent at 28 and 90 days, appears eosinophilic at 180 days due to the polymer’s degradation.
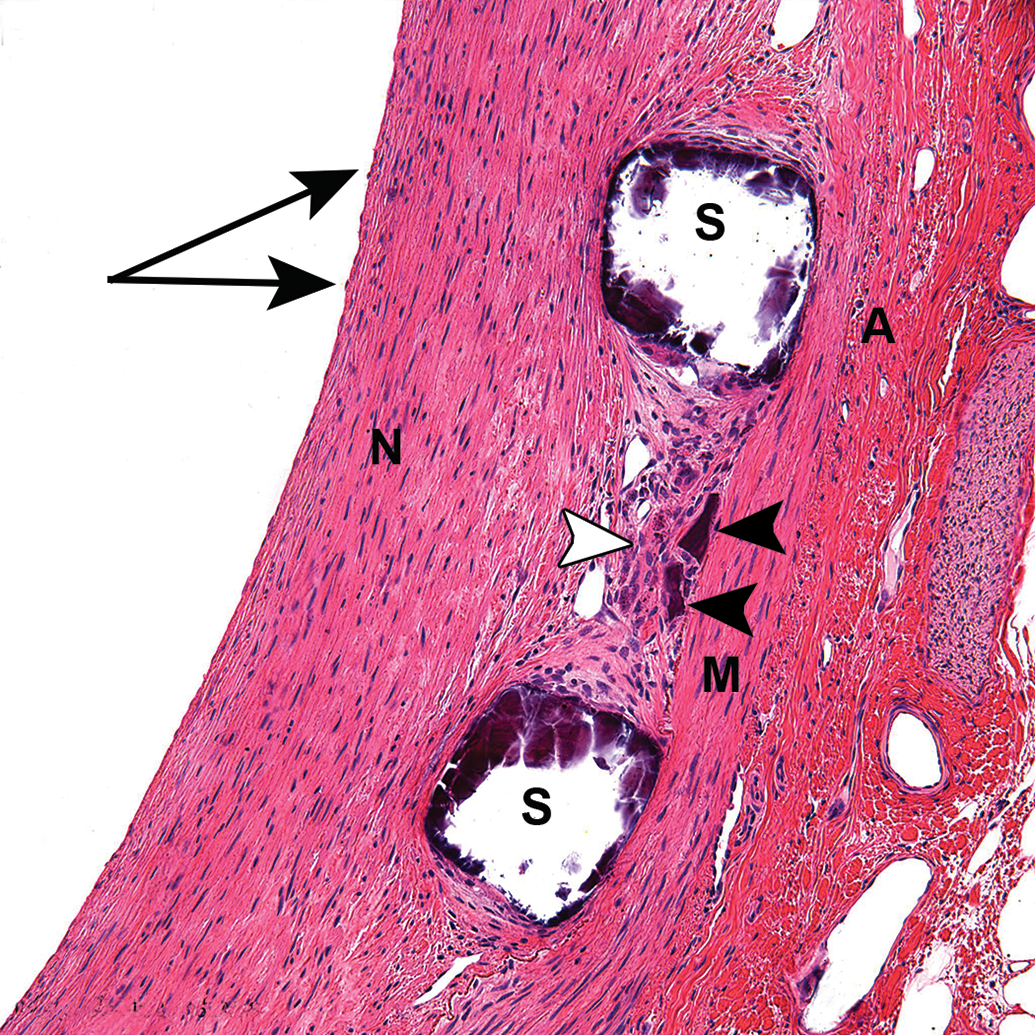
Coronary artery from a healthy swine implanted with a non-coated magnesium (Mg) alloy stent at 180 days following implantation. methyl methacrylate (MMA) embedding, hematoxylin & eosin stain, 20×. Endothelium (arrows), neointima (N), media (M), adventitia (A), struts (S), debris (black arrowheads). Struts are fragmented and a foreign-body reaction (white arrowhead) is seen around the strut debris present in the neointima.
Systemic safety evaluation
Although the presence of Mg particles has not been reported in tissues downstream to the implantation sites, the draining lymph nodes and downstream organs/tissues need to be carefully examined at necropsy, harvested, and evaluated for possible migration of Mg particles that may have detached from the implant during degradation/resorption of the implant (ISO 10993-6, 2016). The applicable downstream organs/tissues will vary depending on the implantation site. For endovascular stents, the downstream tissues will either be the myocardium (in the case of cardiovascular stents) or hind limb muscles and coronary bands (in the case of iliofemoral stents); these downstream tissues should be serially sectioned at necropsy, evaluated for gross abnormalities, and evaluated histologically for vessel patency and presence of thromboembolism, emboli, hemorrhages, or other vascular injury. For orthopedic implants, the adjacent organs and surrounding soft tissues will be examined, as well as draining lymph nodes. Any relevant gross abnormality and selected samples from the downstream or adjacent organs/tissues should be sampled and evaluated microscopically.
Histomorphometry evaluation
Quantitative 2D histomorphometry results for Mg cardiovascular stents need to be correlated with terminal angiography or intravascular measurement results, for example, intravascular ultrasound (IVUS) or optical coherence tomography (OCT), as the Mg stents lose their radial strength during the degradation process and may show artifactual lumen loss via histomorphometry even though perfusion fixation was performed. These results should always be interpreted with caution. The amount of residual implant material, along with numerous other parameters, may be quantified using histomorphometry techniques. For Mg-based devices, the degradation assessment, including measurements of the degradation by-products and void spaces, could also be performed.
Conclusion
In conclusion, Mg-based biodegradable implants have been under investigation for many years; they have been developed to overcome the limitations of permanent metallic implants. Mg implants have shown positive biocompatibility results and potential osteogenic effects; however, depending on the composition of the Mg (i.e. alloying, surface treatment/coating), the Mg-based implant may exhibit a rapid degradation rate with release of by-products, production of hydrogen gas, and particulate debris. Due to their degradability and degradation features, Mg-based devices can present challenges for histological processing and typical procedures cannot always be followed. Due to their fragility and complete degradation over time, perfusion fixation followed by careful handling of the implant site/device should be performed. At later time points, when visualization of the implant sites is difficult, a larger area of normal surrounding tissue should be included with the implant site and radiographs or CT scans can be used to precisely locate the implant sites. Fixation and preservation of the tissue implanted with an Mg device is problematic as Mg degrades/corrodes in an aqueous environment and corrosion and hydrogen gas formation can potentially occur postmortem. Due to the hardness of the Mg implant, the tissues with device should be embedded in plastic resin and, depending on the implanted tissue, will either be cut with a conventional microtome or microground with the EXAKT system. Preliminary tests should be done to ensure that the device and its coating, when present, are compatible with the processing and/or embedding medium as some surface treatments/coatings can be dissolved and removed from the surface of the device during processing and/or embedding with a specific medium. Due to their radiopacity, Mg implants are usually easier to visualize as opposed to polymeric implants; however, after significant bioresorption has occurred at late time points, the radiopacity of the residual implant material is greatly decreased and it may not be possible to differentiate the device from the surrounding tissue. At histopathological evaluation, Mg-based devices may exhibit void spaces, composed of hydrogen gas, surrounded by a thin fibrous capsule, as well as particulate debris, bone resorption, and/or degradation products surrounding the Mg device.
Footnotes
Acknowledgments
The authors would like to thank the AccelLAB’s Histology and MicroCT personnel for their help with image acquisition.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
