Abstract
Non-glandular squamous cell carcinoma (NGSCC) is an extremely rare tumor in Tg.raH2 mice. There have been 5 NGSCC in 1615 control male mice (0.31%) and 2 NGSCC in 1560 control female mice (0.13%) on 26-week carcinogenicity studies, with a range of 0 to 1 of per group per sex in each study without statistical significance in 52 male and 51 female studies conducted in Tg.rasH2 mice. Every case of NGSCC was accompanied by profound granulocytosis.
Keywords
All studies presented in this article were conducted at BioReliance in Rockville, Maryland, now known as Millipore-Sigma. Historical control data were collected from 52 studies conducted in males (1615 mice) and 51 studies conducted in females (1560 mice). The percentage incidence non-glandular squamous cell carcinoma (NGSCC) in each sex was 0.32% and 0.13%, respectively, with a historical control range of 0 to 1 in each sex. This tumor never achieved statistical significance, and for statistical purposes this tumor is considered rare. 1 Similarly, very low incidence of NGSCC is noted by others. 1 –8
CByB6F1-Tg(HRAS)2Jic (+/- hemizygous c-Ha-ras) mice obtained from Taconic Biosciences were used in all studies. Each mouse was genotyped by Taconic to verify the presence of the transgene before being placed on study. Animals were assigned to groups using a computer-generated randomization program based upon each individual study design with a minimum of 25 animals per sex in each control group. Animals were single housed in polycarbonate cages with hardwood bedding chips in environmentally controlled rooms. Animals were verified to be free of illness prior to being placed on a study. All animals had ad libitum access to water and powdered feed (TEKLAD Global Diet, Envigo). The numbers of animals, procedures, and experimental design for each study were reviewed and approved by the BioReliance Institutional Animal Care and Use Committee. All procedures followed the specifications recommended in The Guide for the Care and Use of Laboratory Animals and were conducted in an Association for Assessment and Accreditation of Laboratory Animal Care-accredited facility. All procedures involving but not limited to quarantine and acclimation, randomization, application of unique identification system, housing, provision of food and water, administration of test article, recording of clinical signs, necropsy, and tissue processing were followed in strict accordance with the good laboratory practice regulations, standard operating procedures, and protocol for each study. At necropsy, protocol required tissues were saved in 10% neutral buffered formalin, processed routinely, stained with hematoxylin and eosin (H&E), and evaluated microscopically by a board-certified veterinary pathologist. A pathology peer review by another board-certified veterinary pathologist was also performed on each study.
There were a total of 5 NGSCC observed in males and 2 NGSCC observed in females. Diagnosis of SCC 9 was straightforward (Figures 1 and 2). There was extensive proliferation of neoplastic squamous cells that invaded the submucosa. The invading cells were highly pleomorphic, dyskeratotic, and dysplastic. There were numerous mitotic figures in the proliferating cells. Often the packets of neoplastic cells invaded the gastric serosa and spread into the peritoneal cavity attaching to the visceral organs such as liver, adrenals, peritoneal walls exhibiting metastatic implantation of the tumor. All tumors were very aggressive in nature and they caused invasion and implantation in the abdominal cavity but never distant metastasis to organs such as lungs and to the regional lymph nodes via the lymphovascular system. In every mouse diagnosed with a NGSCC, there was concurrent extensive granulocytosis noted in the routinely examined bone marrows of femur and sternum (Figures 3 and 4). Granulocytosis was also noted in other protocol required bony tissues examined such as nasal cavities (Figure 5) and Zymbal’s glands. Necrosis and associated inflammatory changes that could induce granulocytosis were not present in any of the tumors examined. We considered presence of extensive granulocytosis associated with NGSCC to be a paraneoplastic syndrome. Although NGSCC has been recognized by many researchers as a rare tumor in Tg.rasH2, none have documented granulocytosis as a paraneoplastic syndrome in these tumors. 1 –8
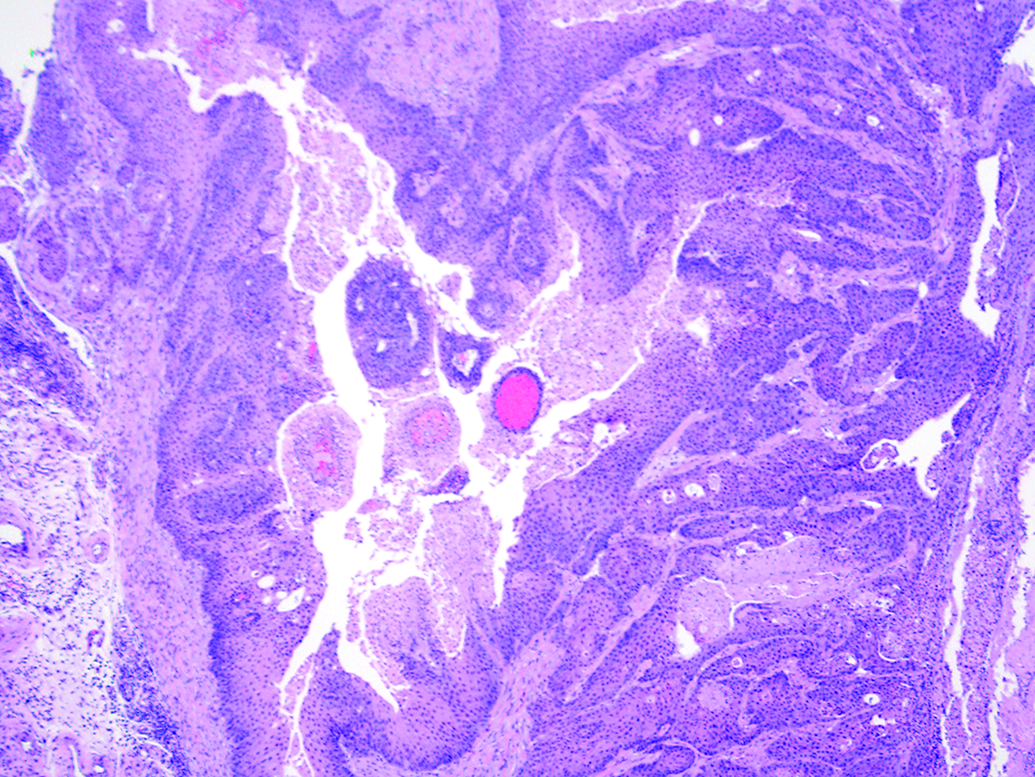
Non-glandular squamous cell carcinoma, ×4.
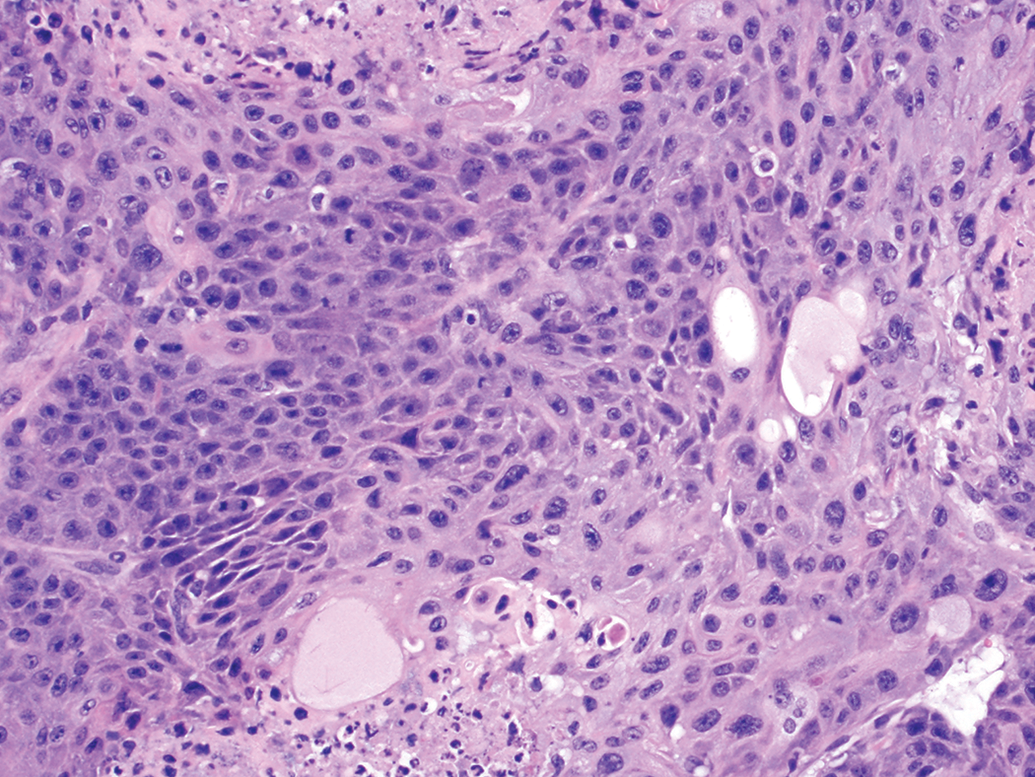
Non-glandular squamous cell carcinoma, ×40.
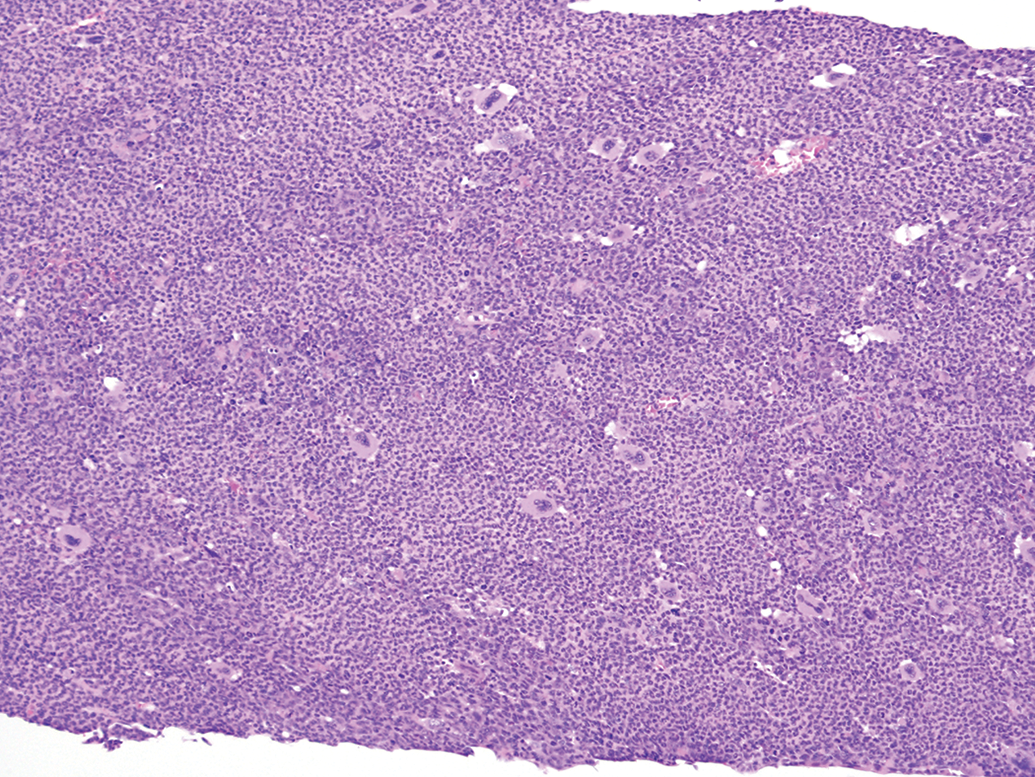
Sternum, granulocytosis, secondary to non-glandular squamous cell carcinoma, ×10.
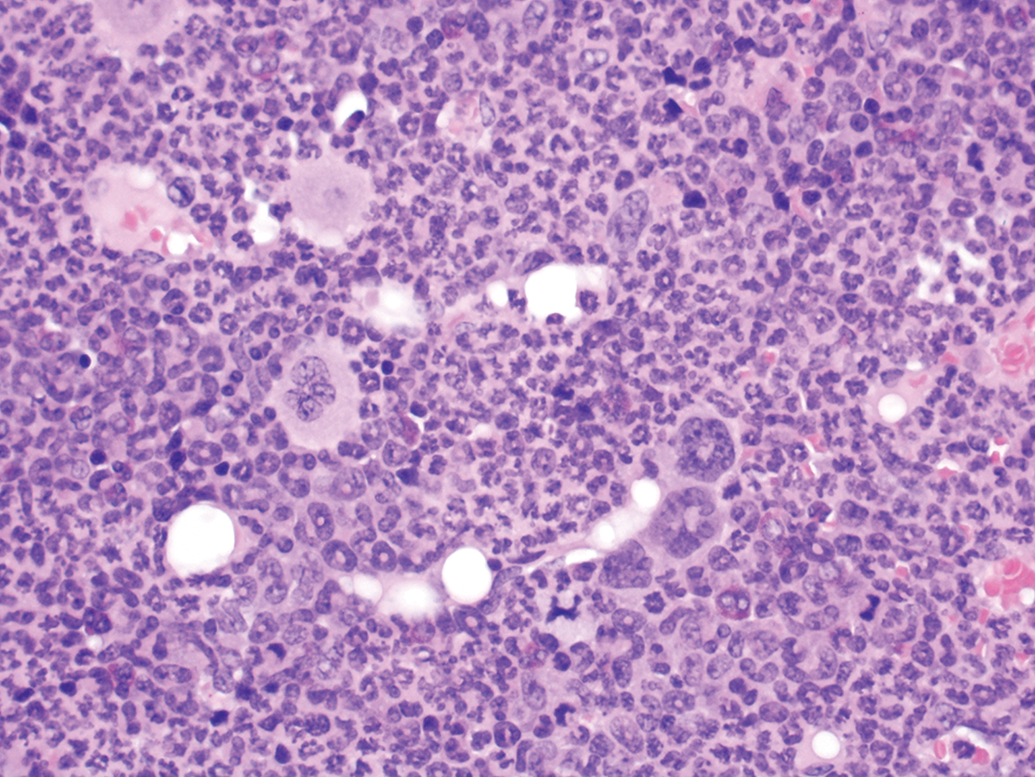
Sternum, granulocytosis, secondary to non-glandular squamous cell carcinoma, ×40.
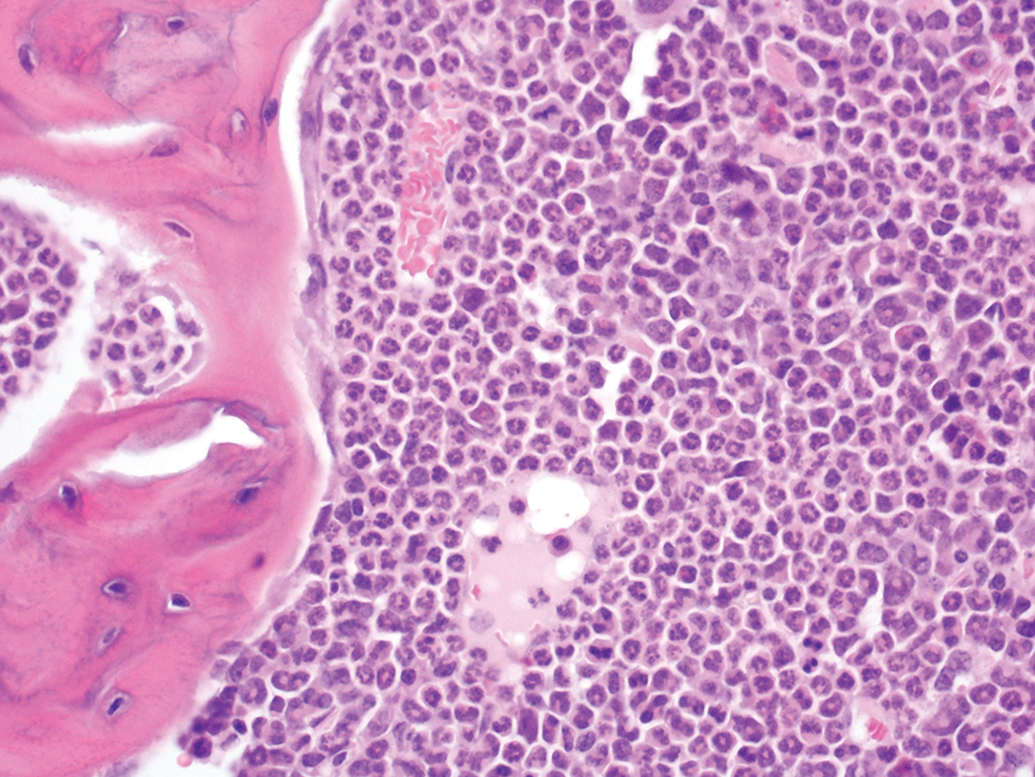
Nasal turbinates, granulocytosis in the marrow secondary to non-glandular squamous cell carcinoma, ×20.
The entire human stomach is glandular and glandular squamous cell carcinomas (GSCC) are extremely rare. Gastric adenocarcinoma (GAC) on the other hand is the most common tumor of humans arising in the glandular portion of the stomach and accounts for 98% of all human stomach cancers. There have been numerous cases of granulocytosis associated with GAC in humans. It has been suggested that the tumor causes the production of granulocyte colony stimulating factor (CSF), which is responsible for massive neutrophilia. Mechanisms of tumor-associated neutrophilia and production of granulocyte CSF and their synergistic actions have been studied, but the exact mechanism is still unknown. 10 Some human GACs also exhibit hypercalcemia of malignancy. 10 However, this has not been confirmed in NGSCC in Tg.rasH2 mice.
Although it is beyond the scope of a 26-week rasH2 mouse study to perform CBCs, clinical chemistry, or any other laboratory tests, the severe degree of granulocytosis noted in H&E sections of these rare NGSCCs strongly suggests neutrophilia, a paraneoplastic change described in human GAC. However, humoral hypercalcemia of malignancy associated with GAC in humans cannot be confirmed in Tg.rasH2 mice due to lack of laboratory tests.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
