Abstract
Since their development in the 1960s, Göttingen minipigs have become a popular nonrodent animal model in biomedical research, especially for ocular studies, because of their ease of handling, size, well-monitored genetics, and ocular anatomy that is similar to humans. The purpose of this mini-review is to introduce the reader to the various ways in which this animal model is currently being utilized in ocular research and to provide an overview of the diagnostic modalities that aid in this research. To date, the Göttingen minipig has been used for a variety of ocular research endeavors, including studies evaluating the safety and efficacy of ocular therapeutics, glaucoma etiopathogenesis and treatment, novel biomaterials, surgical procedures, and implantable materials/devices. In addition to histopathologic evaluation of enucleated globes, the majority of these studies make use of advanced in vivo diagnostic techniques, including electroretinography, optical coherence tomography, fundoscopic imaging, and fluorescein angiography.
Swine have become an important animal species for translational research and toxicology testing, and, in many instances, have begun to replace dogs and nonhuman primates as the large animal model of choice in preclinical studies. There are multiple miniature pig breeds utilized for research (e.g., Yucatan, Hanford, Göttingen, and Sinclair) and excellent review articles that describe their overall utility in various research settings (Stricker-Krongrade, Shoemake, and Bouchard 2016; Swindle et al. 2012). The smallest of these minipig models is the Göttingen, and it has rapidly gained popularity among researchers for ocular studies, including those investigating ocular diseases, novel therapeutics, surgical techniques, and implantable materials/devices. Because of its growing popularity in such studies, the aim of this mini-review is to expound upon the various ways in which this animal model is currently being utilized in ocular research and to provide an overview of the diagnostic modalities that aid in this research.
The Göttingen minipig was developed in the 1960s at the University of Göttingen in Germany by Fritz Haring and his colleagues. To develop this novel minipig, they used three founder breeds: Minnesota minipigs, Vietnamese potbelly pigs, and German Landrace pigs. There are now five Göttingen minipig populations maintained across the globe. In North America, Marshall BioResources remains the exclusive supplier; however, for all of the populations, the University of Göttingen continues to provide genetic management in order to avoid genetic drift, limit inbreeding, maintain colony uniformity, and manage certain selection traits (including fecundity; Simianer and Kohn 2010). Over recent years, Göttingen minipigs have become popular as an alternative animal model because of their ease of handling, well-characterized and controlled genotype, small size compared to the domestic pig and other minipigs (adults weighing 35–55 kg), and ability to reach sexual maturity by 4 to 5 months of age.
In addition to the above-mentioned attributes, pigs, in general, have become increasingly utilized for ocular studies because their ocular anatomy and intraocular physiology are similar to that which is described in human eyes. Although this mini-review is not intended to be an exhaustive primer on comparative ocular anatomy, a basic understanding of the similarities and differences between pigs and humans is necessary for appropriate study design and therefore warrants a brief discussion. As shown in Figure 1, pigs and humans have similarly sized globes, making the porcine globe useful for the study of dosing volumes, implants, and surgical techniques. Unlike humans, however, pigs have a slightly thicker cornea that is horizontally ovoid (not depicted), corresponding to a horizontally ovoid pupillary aperture.
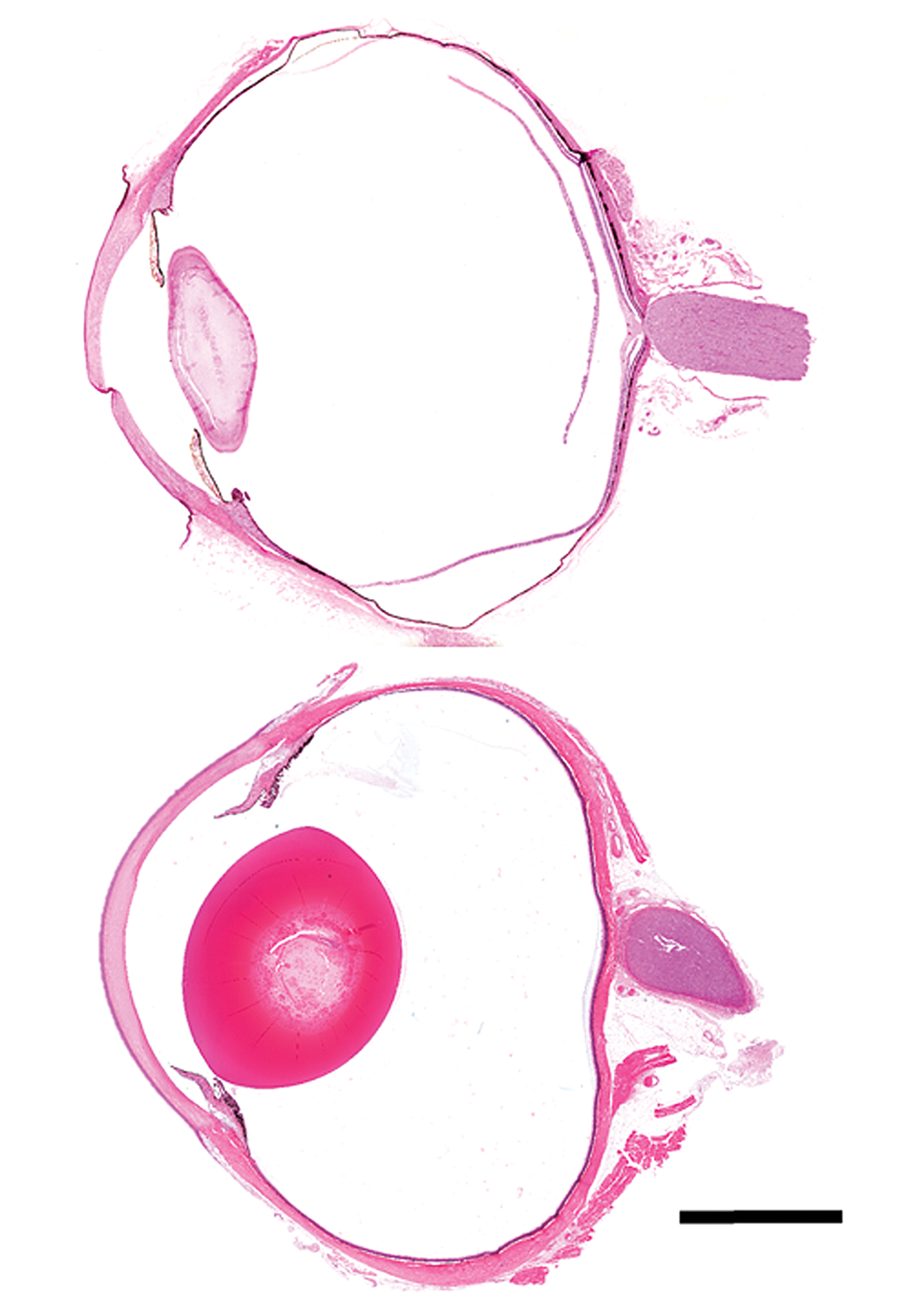
Subgross images of bisected human (top) and Göttingen minipig (bottom) eyes. Hematoxylin and Eosin. Bar = 5 mm.
Humans and pigs also share similar vitreal volumes (see Table 1) and viscoelastic vitreous humor properties. In all species, the vitreous is a transparent gel composed predominantly of water, collagen, and hyaluronic acid. There are, however, species variations in the concentrations of these components. In both pigs and humans, hyaluronan (HA) is the predominant glycosaminoglycan, and chondroitin sulfate (CS) is the minor glycosaminoglycan. The relative proportions of large vitreal HA molecules and the length of the CS chains are also similar. However, disaccharide analysis has shown that the CS chains in the human vitreous consist of 6-sulphated disaccharides, whereas in the pig, 4-sulphated disaccharides predominate (Noulas et al. 2004). In addition to variations in individual component concentrations, there are species differences in the proportions of these components with age, resulting in variations in the rheologic (gel–liquid) state. For instance, in many animal models (including the rabbit and guinea pig), the vitreous remains in a gel state throughout life. In humans and nonhuman primates (NHPs), however, the vitreous becomes less gelatinous with age (Berman 1991). To the authors’ knowledge, comparative age-related changes in the viscoelastic properties of the porcine vitreous have not been reported.
Anatomical Comparison of Pig and Human Eyes.
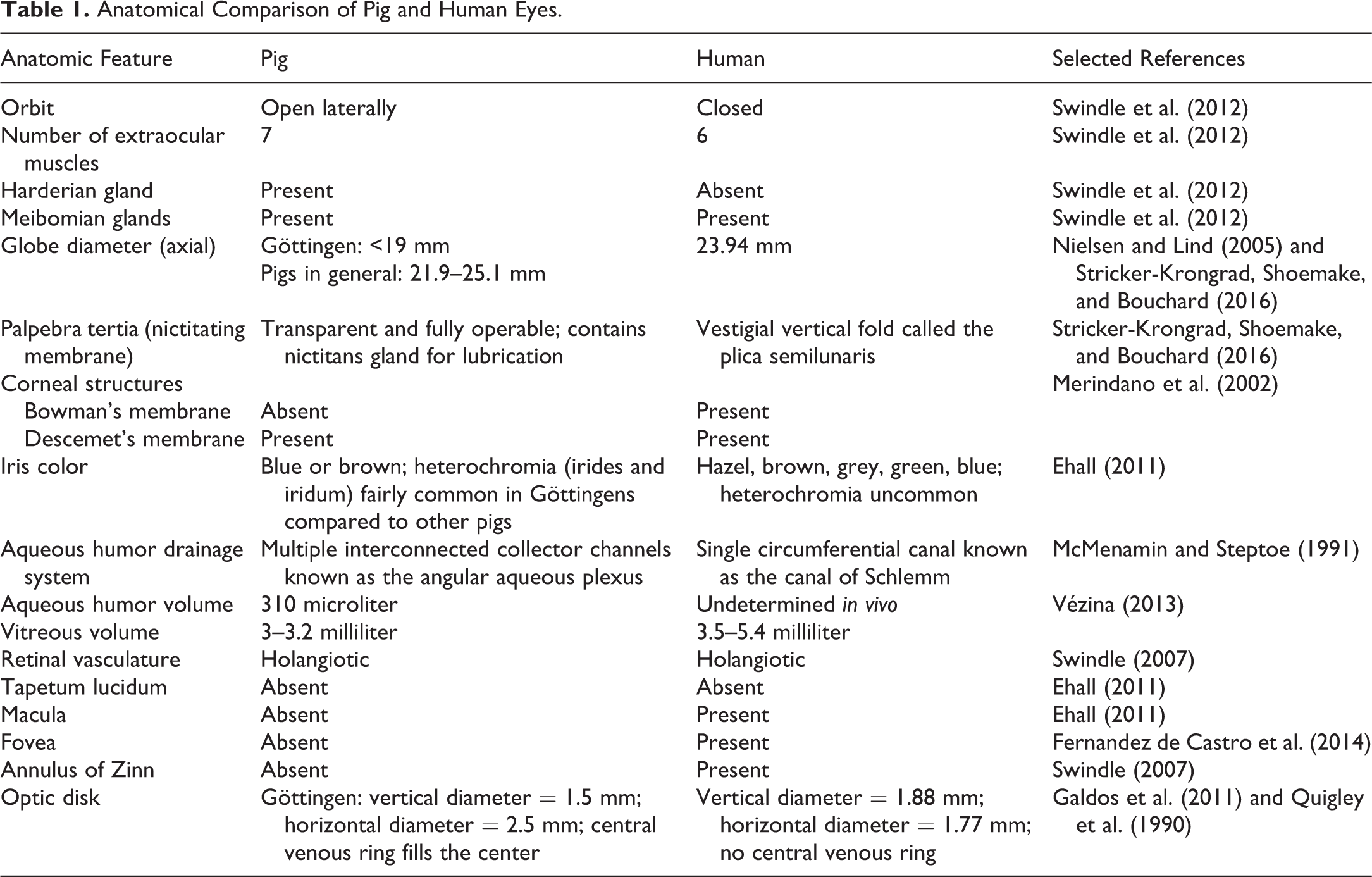
In addition to understanding the anatomic differences between pigs and humans, it is important to understand differences in vision and associated biometrics. Pigs have an open visual field, binocular vision, and are believed to have some degree of color vision. They also have ocular magnifying capabilities similar to humans, with an ocular power of 78 diopters (D). Göttingen minipigs specifically have been noted to have an uncorrected mean refractive error of +1.3 D (Nielsen and Lind 2005), consistent with a mild myopia (near-sightedness), a common condition in people. Myopia results from an inability to properly focus parallel rays of light on the retina; in people, these refractive errors are commonly corrected with eyeglasses, contact lenses, or surgery. Similar to what is described in humans, the mean refractive corneal power in Göttingen minipigs has been noted to be 44.1 D (Nielsen and Lind 2005). Unlike humans and NHPs, pigs lack a macula, an area of the retina located at the temporal side of the optic nerve head in humans. The central portion of the macula is the foveal pit, a 1- to 2-mm diameter area of highly concentrated cone photoreceptors. The fovea functions to provide high-resolution daytime vision. Although pigs lacks a macula, they do have highly concentrated regions of cone photoreceptors known as the area centralis and visual streak; like the macula, these are regions of higher visual acuity (Vézina 2013) and provide researchers with the opportunity to study human retinal diseases (such as macular degeneration) in pigs. An overall anatomical comparison of pig and human eyes is shown in Table 1.
One of the uses of Göttingen minipigs in ocular preclinical studies is to assess the safety and efficacy of topically, intravitreally, and subretinally administered pharmaceuticals. Topical therapeutics have traditionally been effective in delivering drugs to the ocular surface and anterior segment of the eye; however, various novel topical compounds are currently being formulated to deliver therapeutically relevant drug concentrations to the back of the eye. The most common method of delivering a pharmaceutical agent to posterior ocular structures is via intravitreal injection. Subretinal dosing, which is a more recently developed drug delivery method, has been utilized for experimental injection of gene therapy vectors and stem cells and for the placement of subretinal drug delivery implants. Because intravitreal and subretinal dosing are of interest and use in human medicine, both drug delivery techniques are utilized in Göttingen minipig research. In people, intravitreally administered drugs have been used with increasing frequency over the last few decades for the treatment of posterior segment diseases, especially diabetic macular edema and age-related macular degeneration. The most commonly administered medications are antivascular endothelial growth factor preparations and corticosteroids. In addition to these pharmacotherapies, agents that continue to be studied are intravitreal steroid implants, adenovirus vector-mediated gene transfer techniques, stem cell therapy injections, antimicrobials, antivirals, tissue plasminogen activator, gas, and air (Shikari and Samant 2016).
In order to evaluate the eye following intravitreal injection of a compound in a preclinical setting, various diagnostic tools are now being utilized. One of these diagnostic tools is the electroretinogram (ERG), used to measure the electrical activity generated by neural and nonneuronal cells in the retina in response to a stimulus (light). Varying ERG stimulus conditions (color, dark or light adaption, flashes, and patterns) can elicit response from different retinal cellular components and therefore allow the clinician to evaluate the functionality of these components. ERG can therefore be used to evaluate the effects of administered therapeutic agents, conditions that cause elevated intraocular pressures (IOPs), and retinal diseases. Recently, ERG recordings were performed on 162 Göttingen minipigs (aged 4–6 months) using an adapted Société Française d’Etudes et de Recherches en Ophtalmologie Vétérinaire (SFEROV) dog ERG procedure in an effort to establish a standardized ERG protocol for this animal model. During this study, photopic and scotopic ERGs were simultaneously recorded in both eyes from anesthetized minipigs using sclerocorneal clip electrodes. The authors concluded that the modified SFEROV procedure was appropriate for use in Göttingen minipig preclinical studies and resulted in no significant gender differences (except for higher b-wave amplitude for the photopic ERG responses in the female pigs compared to males; Forster, Augsburger, and Haag 2013).
Additional diagnostic techniques used to evaluate intravitreally administered agents in Göttingen minipigs include optical coherence tomography (OCT) and histopathology. OCT is a noncontact, noninvasive imaging technique used to generate high-resolution images of the retina and optic nerve following intravitreal drug administration. It is important to note that OCT can also be used to image the cornea, drainage angle, and anterior chamber. In human medicine, anterior segment OCT is considered to be a valuable tool for monitoring and managing glaucoma in some patients and for pre- and post-op LASIK surgery evaluations. Histopathologic evaluation of the enucleated globe following intravitreal drug administration is another important diagnostic step in the drug development process. When performing such assessments, it is critical to differentiate between artifactual changes (amphophilic vitreal material, lenticular vacuolation, and retinal detachment), injection-related findings (focal hemorrhage, inflammation, and intraocular structural damage), and test article–related effects (vitreal cellular infiltrates, retinal atrophy, and retinal inflammation). Primary test article–related effects can be difficult to differentiate from immunotoxicity (an adverse effect on the immune system resulting in impaired humoral or cellular immunity); in such instances, additional diagnostic tests, such as immunoassays, immunophenotyping, cytokine assessment, and evaluation of complement, are often utilized to aid in the distinction.
Although the domestic pig was utilized as an early experimental model to better define the etiopathogenesis of glaucoma, the size of the adults makes general handling and ocular examination difficult. Göttingen minipigs, therefore, began to be utilized as an experimental glaucoma model. In both domestic pigs and minipigs, episcleral vein cauterization has been successfully utilized as a means to experimentally induce glaucoma. IOP is then measured via tonometry. Recent studies have shown that Göttingen minipigs with experimentally induced glaucoma have a more asymmetric retinal vascular pattern than in humans (assessed via fluorescein angiography) and that the caudally displaced vessels associated with loss of the neuroretinal ring and exposure of the lamina cribosa (known as central excavation) is filled by a central venous ring (absent in humans with glaucoma). Glaucomatous optic disk changes (evaluated via fundoscopic and angiographic photography) include arteriolar incurvation and occasional displacement of the arterioles arising between ganglion cell axons (Galdos et al. 2011). These findings indicate that unlike humans with primary open angle glaucoma (in which both arterioles and venules are displaced) arteriolar involvement predominates in Göttingen minipigs. The same study documented that the mean IOP in control Göttingen eyes was 22.43 ± 4.46 mm Hg (normal IOP in humans is 12−22 mm Hg) and that the mean IOP in glaucomatous Göttingen eyes was 26.04 ± 5.01 mm Hg (P < .01; Galdos et al. 2011). Although glaucoma-associated retinal vascular changes differ between humans and Göttingen minipigs, the minipig model still remains useful for the study of therapeutics and medical devices designed to treat glaucoma and lower IOP.
Göttingen minipigs have also been utilized for the development of biomaterials aimed at treating various injuries resulting in corneal blindness, such as corneal perforations, corneal chemical burns, and autoimmune-associated corneal damage. One of these novel biomaterials is the BioCornea, a tilapia scale-derived collagen matrix that is being developed to seal corneal perforations. BioCornea is a premarket prototype developed by Aeon Astron Europe B.V. (Leiden, The Netherlands) and manufactured by Body Organ Biomedical Corp. (Taipei, Taiwan). To test the BioCornea’s effectiveness at sealing corneal perforations in the Göttingen minipig model, a 2-mm diameter full-thickness corneal perforation was made at the center of the eye using a trephine. The BioCornea was then sutured into place to seal the corneal perforation. In order to assess healing, the treated eyes were evaluated via a handheld slit lamp, OCT, and histopathologic evaluation. OCT imaging and slit lamp evaluations indicated that the BioCornea formed a stable seal of the perforation and that there was only mild swelling of the wounded cornea. Microscopically, there was corneal stromal edema (most prominent adjacent to the perforation site), thinning of the corneal epithelium beneath the BioCornea, a lack of epithelial ingrowth to the perforated wounds, and mild leukocytic inflammation (Chen et al. 2015). Other corneal biomaterial studies have utilized the Göttingen minipig to evaluate not only the biomaterial implant’s performance but also corneal vascularization, nerve ingrowth, surgical techniques, and suturing patterns.
The Göttingen minipig has also played an integral role in studies designed to investigate surgical techniques associated with the implantation of intraocular devices. One such study evaluated the surgical procedures required to implant a wireless intraocular epiretinal retina implant (EPI RET3). The intent of this device is to stimulate retinal neurons via electrical impulses in order to produce vision in people with blindness (often resulting from disorders such as retinitis pigmentosa). Earlier conceived retinal prostheses were controlled from an external ocular device; this novel epiretinal stimulation system, however, is a remotely controlled wireless device housed entirely within the globe. Surgical placement techniques for EPI RET3 were recently evaluated in 16 Göttingen minipigs. The procedure included phacoemulsification and vitrectomy followed by device implantation through a corneoscleral incision. The electrode array was tacked onto the retina, just temporal to the optic disc. The receiver module was placed into the sulcus ciliaris. Average surgical time was 2 hr and implantation was successful in all of the minipigs. Clinically, intraocular findings included fibrin exudation and iris synechiae with the receiver module (Laube et al. 2012). This study successfully demonstrated the surgical feasibility of implanting an epiretinal retina implant in the Göttingen minipig and underscores the importance of this animal model in translational ocular research.
Because of the growing popularity of the Göttingen minipig in ocular research and the current paucity of aggregated information available addressing this research niche, the intent of this mini-review is to introduce the reader to the various forms of ocular research that utilize the Göttingen minipig and to briefly introduce diagnostic modalities that are important in these research endeavors. As more ocular diseases, therapeutic agents, surgical techniques, and implantable materials/devices are studied in the Göttingen minipig, it becomes imperative for researchers (including pathologists) to have knowledge of this animal model’s ocular anatomy, utility in specific study settings, and available adjunctive diagnostic techniques so that informed decisions can be made regarding study design and evaluation.
Footnotes
Acknowledgment
The authors would like to thank Dr. Peter G. Anderson from the University of Alabama at Birmingham for providing the subgross photomicrograph of the human eye.
Author Contribution
Authors contributed to conception or design (SS); data acquisition, analysis, or interpretation (SS, WG); drafting the manuscript (SS); and critically revising the manuscript (SS, WG). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
