Abstract
The use of minipigs in preclinical safety testing of pharmaceuticals is considered an alternative to the more traditional dog and nonhuman primate (NHP) nonrodent species. Substantial evidence exists to suggest that the anatomy, physiology, and biochemistry of minipigs are similar enough to humans to consider them as valid nonrodent models for pharmaceutical safety testing. Since the utilization of minipigs was last assessed over 5 years ago, the Preclinical Safety Leadership Group (DruSafe) of the International Consortium for Innovation and Quality in Pharmaceutical Development conducted this survey to provide an updated assessment of the utility, perceived value, and impediments to the use of minipigs in preclinical safety testing. Of the 32 participating members of DruSafe, 15 responded to the survey representing both large and small companies. Respondents indicated that the minipig has been utilized mostly for short-term safety assessment studies with dermal, oral, and parenteral routes of administration. Minipigs are widely accepted as appropriate models for cardiovascular assessments and have been used to a limited extent for reproductive toxicology testing. Overall responses indicated that safety testing for large molecules using this species is relatively low due to a lack of background data, reagents or biomarkers, concerns regarding immune system characterization and poor suitability for developmental toxicity assessments. Most companies utilized contract research organizations for definitive safety toxicity assessment studies. Conclusions of this survey indicate that minipig is an acceptable nonrodent species largely limited to studies using small molecules, primarily dermal products, and results are comparable to those reported 5 years ago.
Introduction
The International Consortium for Innovation and Quality in Pharmaceutical Development (IQ Consortium) is an international association of pharmaceutical and biotechnology companies aiming to advance innovation and quality in the development of pharmaceutical products through scientifically driven best practices and standards. The ultimate goal is to improve safety and efficacy of medical products for patient benefit. As part of the IQ Consortium, the Preclinical Safety Leadership Group (DruSafe) is composed of senior preclinical safety assessment leaders from IQ Consortium member companies (Table 1). A working group was formed by DruSafe to survey member companies on the current experiences with using minipigs in preclinical toxicology testing of pharmaceuticals (large and small molecules). The survey concentrated on factors considered in selecting the minipig as a nonrodent species for safety assessment, the extent to which the minipig is currently being utilized for preclinical safety assessment, and the requirements for an appropriate test facility.
Members of the IQ DruSafe Leadership Group.

aResponded to the survey.
Pigs have been used for biomedical research because of their similarity to human anatomy, physiology, genetics, and biochemical function (Forster et al. 2010a). A key milestone for the research community was the introduction of specific pathogen-free (SPF) domestic pigs in the late 1950s (Safron and Gonder 1997). The more recent interest in using minipigs for toxicology assessments is due to an increase in the historical database for normal clinical and anatomic pathology parameters and the smaller size and relative ease of handling and housing compared to domestic farm breeds. Because of these benefits, various strains of minipigs (Hanford, Yucatan, Yucatan micro, Sinclair, and Göttingen) have been developed and are available for use in preclinical safety assessment (Swindle et al. 2012). It should be noted that Göttingen minipig genomic sequence has recently been published (Vamathevan et al. 2013), which may lead to broader utilization of this specific strain in both medical research and drug development. Biomedical researchers have been following the acceptance and use of minipigs as the nonrodent species for toxicology testing for several years (Ganderup et al. 2012), and minipigs have been used as the nonrodent species for safety assessment of pharmaceuticals with scientific justification (McAnulty et al. 2012).
The November/December 2010 issue of the
In 2011, Ganderup of Ellegaard Göttingen Minipigs collected government registration data on the patterns of use and number of pigs/minipigs, dogs, and NHPs used by pharmaceutical company laboratories in the United States, Canada, the European Union (EU), and Japan from 2002 to 2008 (2011). Because data collection by each country varied in terms of frequency and specificity of questions, the data were fragmented and vague (i.e., pigs and minipigs were not always distinguished). Canada and the EU used more pigs/minipigs and fewer dogs during this time period, but the use of NHPs was relatively constant among countries.
The use of minipigs in safety pharmacology studies was surveyed by the Safety Pharmacology Society in 2012. The survey of 361 individuals, including participants from pharmaceutical companies and contract research organizations (CROs), was queried on the integration of functional central nervous system observations within toxicology studies, of which preliminary results were presented in a 2012 webinar (Authier 2012). They found that a functional observation battery (FOB) was included in 198 regulatory toxicology studies of new chemical entities and that 2 of these studies used minipigs, while none of 81 FOB-inclusive studies for biologics used minipigs.
The use of minipigs for toxicology testing as a substitute for the more traditional nonrodent species (dogs and NHPs) is an ongoing consideration in the pharmaceutical industry. For example, the minipig was utilized as the nonrodent species for the approved tyrosine kinase inhibitor, afatinib (Gilotrif; FDA pharmacology review 2013), and for juvenile toxicity studies of the GLP-2 agonist, teduglutide (Gattex; FDA pharmacology review 2012). While the decision to use minipigs may be driven by efforts to reduce or exclude the use of dogs and NHPs in biomedical research, substantial evidence exists to suggest that because the anatomy, physiology, and biochemistry of minipigs are comparatively representative of humans, the minipig may be a valid species for pharmaceutical safety testing. The minipig has not been considered a default nonrodent species for toxicology testing across the pharmaceutical industry because of gaps in understanding some of the nuances of management, development of universal standards, and regulatory acceptance. The objective of this DruSafe survey was to provide a current assessment of the utility, perceived value, and impediments for using the minipig in preclinical safety testing from a representative cross section of the pharmaceutical industry.
Methods
A survey on the current use of the minipig in preclinical safety testing and factors that influence the selection of this species was distributed to members of IQ DruSafe (Supplementary Appendix). The survey was designed in Excel format by a cross-functional team within Bristol-Myers Squibb with input from DruSafe members. There are 32 member companies within DruSafe, representing a broad cross section of the pharmaceutical industry including large, midsize, and small companies. The member companies are listed in Table 1. The survey was not distributed to CROs. Fifteen companies responded to the survey, and all responses were blinded by the IQ Secretariat before review by the authoring team. The survey covered a range of topics including general questions on whether minipigs are used in preclinical safety testing (why or why not), what factors influenced the decision, are the studies conducted internally or outsourced, what kind of studies are performed, and what logistical factors (e.g., personnel training and facilities) need to be considered when using minipigs. All survey responses were received by January 2015. In this document, the provided denominator (ratio of respondents) represents the number of companies that responded to a given question or set of questions, as not all companies responded to each question (Table 1).
Results
Respondent Company Demographics
Fifteen of 32 DruSafe member companies accepted the invitation to participate in this survey, an overall response rate of 47%. Of these 15 respondent companies, 12 identified themselves as a large pharmaceutical or biopharmaceutical company and 3 identified themselves as a small/specialty biopharmaceutical or biotechnology company.
Eight of the respondents (7 large companies and 1 small company) are currently using minipigs in their preclinical safety assessment programs for biologic and/or small molecules. All 8 have exclusively used the Göttingen minipig strain and have conducted studies primarily in the United States (4 companies) or Europe (4 companies). Three of the 8 companies conduct minipig studies in-house (2 in Europe and 1 in the United States). Two of these 3 companies conduct both non-GLP and GLP studies in-house, although 1 outsources studies longer than 13 weeks’ duration, while the third company outsources its GLP studies. These 3 companies indicated they utilized an in-house historical minipig database, although the size of the database was limited relative to that of the dog or NHP. A fourth company that conducts minipig efficacy studies in-house indicated that it had renovated its facility for low usage support of minipig safety studies. Otherwise, companies outsource minipig studies to CROs and rely on the experience and historical data available at the testing facilities.
Key Factors Influencing Minipig Use for Safety Assessment
Seven of the 15 respondents have not used the minipig for preclinical safety assessment. Reasons included (1) no scientific reason or other driver to replace the dog or NHP as standard nonrodent species, (2) do not develop dermal agents, (3) limited historical data availability for molecule classes of interest, and (4) no experience with this species. Two companies noted they either had explored the use of minipigs or were in the process of doing so and were investigating CRO capabilities.
Overall, the 8 respondents who have experience with using the minipig for preclinical safety assessment estimated that this species was employed in less than 15% of their safety studies (responses ranged from less than 2% to 15%). Two primary reasons were provided for selection of the minipig for safety testing: (1) when the dog was not a suitable species or (2) when the dermal route of administration was required. The survey specifically asked if the minipig was considered by any company to be their “default” nonrodent species. Only 1 of the 8 companies, a small company, responded “yes” to this question but clarified that this was for dermal programs only. A second company indicated that the minipig was given consideration for topical/transdermal programs. Two respondents stated that the minipig was considered secondary to the dog as the nonrodent species. One of the companies explicitly noted that they needed to determine the utility of the minipig when the dog was not suitable and would need to justify “deselection” of the minipig before considering the NHP. In relation to the 3Rs’ principles of animal use, 5 companies (4 of which use the minipig, all of which are large companies) stated that public perception has had an impact on their selection of a nonrodent species. One company specifically commented on the principles of 3Rs as a factor, indicating that in many cases the minipig may be considered a lower species and therefore a replacement for NHPs.
Companies were asked what factors they would consider before selecting the minipig as the appropriate nonrodent species. Pharmacodynamic response was the primary or secondary factor that would influence company decision making (11 responses) regarding the minipig, followed by biomarker availability (10 responses), the intended route of administration (7 responses), and pharmacokinetics (i.e., exposure and/or metabolism and 6 responses). Additional factors included body weight (in relation to limited availability of test article and/or estimated test article requirements) and the frequency and method of sample collection.
Minipig-specific concerns and impact on development plans
Company responses regarding key factors that influence their decision to use the minipig for safety assessment are summarized in Table 2. Nine of the 13 companies indicated that selection of the minipig as the nonrodent toxicology species influenced or would influence their development plan. Size of the animal and the consequent test article requirements was the most commonly cited reason (8/9 responses). Interestingly, however, companies were split about whether the increased test article requirements impacted their decision to utilize the minipig in the first place: 6 companies cited the cost of increased drug supplies as their key concern, whereas 7 companies base their decision to utilize the minipig on scientific, rather than drug requirement reasons (as noted above). The second most cited reason (2/9 responses) for why selection of the minipig influenced, or would influence, a development plan concerned the reproductive toxicology strategy for biologics because of the lack of large molecule placental transfer in the minipig and need for an alternative approach to assess embryo-fetal development. As noted by 1 company, when a monoclonal antibody does not cross-react with a rodent, this may necessitate development of a surrogate molecule in order to conduct reproductive toxicity studies or the use of NHPs. On this matter, another company stated that if it were to consider the minipig as the nonrodent species for a monoclonal antibody (presently it does not), then the ability to assess developmental toxicity in the rodent or rabbit would weigh into its decision. If neither the rat nor rabbit was a pharmacologically relevant species, the company stated it would most likely avoid the minipig in favor of the NHP for both general and developmental toxicity assessments.
Company Responses to Key Factors Influencing Minipig Use for Safety Assessment.
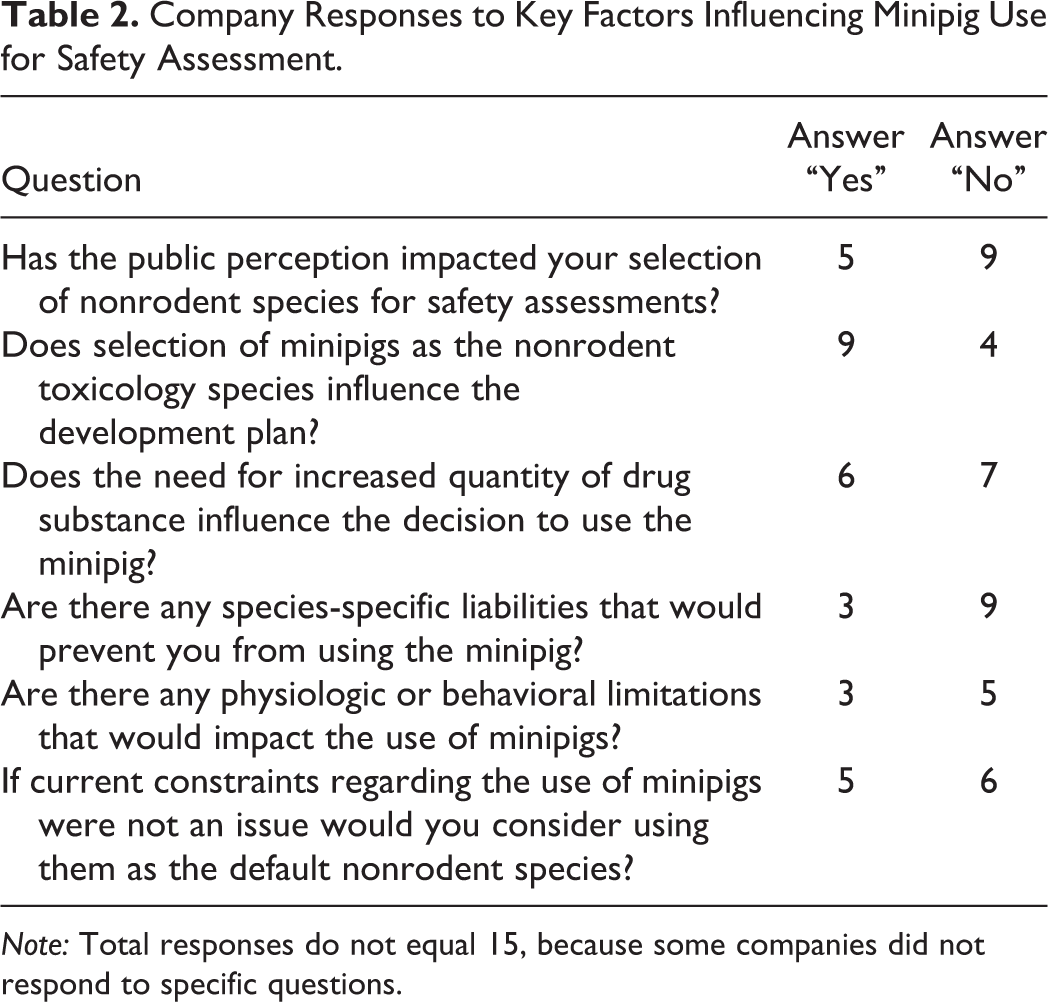
Nine of the 12 respondents did not identify any species-specific liability that would preclude the minipig as the nonrodent toxicology species. Of the 3 companies that had species-specific liability concerns, 2 cited concerns over their use for the assessment of biologics or macromolecules, including potential for immunogenicity and the lack of placental transfer of such molecules. One company acknowledged the immune system of the minipig to be fairly well characterized but noted that the sensitivity of this species to monoclonal antibody–mediated actions (e.g., cytokine release, complement activation, antibody-dependent cell-mediated toxicity) is not well understood and that some immune function assays to evaluate immune toxicity are not presently available. Three respondents stated that there are physiologic and/or behavioral limitations that impact minipig utility, the most commonly cited reason being that minipigs are prone to being highly stressed during study-related procedures especially during handling/restraint, with potential adverse consequences (1 company experienced a stress-related mortality).
Minipigs as a potential default nonrodent species
Companies were also asked if the minipig would become their default nonrodent toxicology species if the current perceived constraints regarding minipig use (cost, facilities, training, scientific/technical/regulatory issues, historical data, etc.) were not an issue. Responses were split, with 5 of 11 respondents stating yes and another stating it could happen but would require a policy change. There were no common reasons among those that responded “no,” but these generally reflected those noted earlier regarding minipig use, including no ethical or scientific reason to switch from standard nonrodent species or that the minipig would only be used as a default nonrodent species for dermal programs.
Facility Considerations
As noted above, 6 of the 8 companies that use minipigs for preclinical safety assessment indicated that they outsource their minipig studies to CROs. Key influences reported for the selection of CROs for minipig studies are housing/facility requirements, study duration and design, technical experience, and the availability of historical data that are critical to appropriately interpret results and to identify potential species-specific findings. Availability of CROs to conduct minipig studies was not an issue, though lack of experience at CROs offering the minipig for toxicological evaluation and a limited understanding of the background rates of spontaneous findings were noted as being key concerns. As for other nonrodent models, acclimation, general handling procedures, age of animals at study initiation, and timing of studies all factored into the selection of CROs.
No issues were reported in conducting minipig studies in compliance with GLP or Organization for Economic Cooperation and Development (OECD) requirements. A few respondents noted that companies and CROs needed to consider special requirements from regulatory agencies when using minipigs. These included animal welfare standards, importation licenses that apply specifically for pigs (which may vary by country), housing requirements, staff training, and use of special reagents or procedures for anesthesia, analgesia, or surgery.
Housing specifications
Five of the companies that use minipigs, including the 4 companies that have in-house minipig facilities, responded to questions concerning housing requirements for this species. Of these 5 companies, 2 use rooms that are dedicated to minipigs and 3 use multipurpose rooms. Three of the companies noted that the vivarium had been renovated to accommodate minipigs. One company that had used the minipig for preclinical efficacy studies and was expanding their use to include safety studies indicated that renovations were completed to provide a more appropriate environment for enhanced living conditions and to promote the ease of animal handling. The other 2 companies, both of which have in-house accommodations for minipigs, reported they had modified their existing caging systems; 1 specified changing flooring grates of dog cages. None of the 5 companies reported any special enrichment or socialization requirements for minipigs that would pose an issue for their use in toxicity studies. When asked if social housing was easily accomplished, 3 companies said yes. Two companies communicated difficulties with social housing, especially related to the aggressive nature of sexually mature males. Three companies indicated no specific health concerns for minipigs; however, 2 companies commented on the susceptibility of minipigs to the same pathogens as domestic farm pigs, the need for increased health monitoring, given that minipigs are usually procured unvaccinated and SPF, and the need for special housing considerations (e.g., feed, temperature, and humidity). No company reported disease-related occupational health concerns.
Technical and training concerns
Four companies responded that training their technical staff to handle minipigs was an obstacle, and 5 companies responded that the use of minipigs was more physically demanding on their staff because of the size and inherent behavior of the species, especially once minipigs reach 9 to 10 kg body weight. In addition, procedures such as dosing and blood collection were challenging because minipigs were not easily trained to adapt to manipulations necessary for these procedures. Companies that responded to questions about study technician numbers indicated that 2 to 3 technicians were required for minipig procedures. In general, it appears that 3 technicians are needed for dosing and blood collection and 2 are needed for simpler procedures such as electrocardiograms and eye exams. There was a wide range of answers regarding the amount of time it took companies to fully train their technical staff to conduct studies in minipigs, the range being 1 to 12 months and the duration depended on the task required and the complexity of the procedures.
Minipig Use in Small Molecule and Biologics Development
Four of the 8 companies that use minipigs for preclinical safety assessment have employed the minipig in early screening studies. Three of these 4 companies have generated early data on pharmacology, pharmacokinetics, or metabolic profiling of small molecules. Two of these companies also reported experience conducting genomics/metabolomics investigations in minipigs, though it was noted that the current annotation for the minipig is lagging compared to other species and could possibly complicate data analysis/interpretation. Three of the 4 companies also routinely include the minipig in target binding, target expression, or activity assays to serendipitously assess biologics for cross-reactivity and thus pharmacological relevance. Another company had evaluated target binding in the minipig “for cause” for one biologic program as there was no cross-reactivity in their standard toxicology species. No company reported that it had purposefully engineered a biologic to cross-react with a target in the minipig.
Five companies stated that when the minipig is used for preclinical safety assessments, the discovery work is also conducted in this species. One company that uses the minipig only for safety evaluation of dermal development programs noted that it had experience using the minipig as a preclinical efficacy model for nondermal programs though the dog or NHP was subsequently used for safety evaluation.
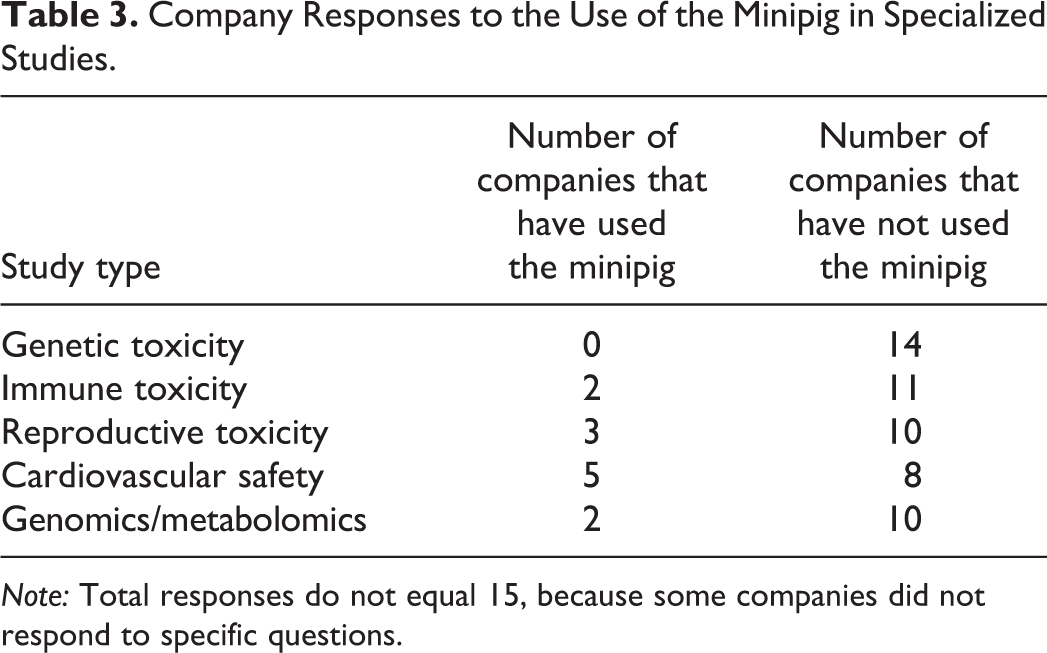
Respondents indicated that preclinical safety assessments using the minipig included safety pharmacology, general toxicology, and reproductive toxicity evaluations. None of the respondents had used the minipig to evaluate genetic toxicity, and 1 company even noted that it had never heard of this being considered. Company responses regarding minipig use for specialized study types are summarized in Table 3.
Company Responses to the Use of the Minipig in Specialized Studies.
General toxicity assessments
General toxicity studies using the minipig have included both short-term studies (5 responses) and long-term studies (2 responses). Studies have been conducted by the dermal, oral, or parenteral route of administration (5 responses for each route), although 2 companies reiterated they have only conducted dermal studies in the minipig. Based on responses from 6 companies, the typical age of minipigs has minimally been 3 months or at least 5 months, when requiring sexual maturity at study initiation.
Assessment of safety pharmacology
Survey questions regarding safety pharmacology focused on cardiovascular testing, specifically whether the minipig was used in cardiovascular screens or for telemetry studies. Five respondents answered yes to doing telemetry studies, and 1 company noted that this was only done when the minipig was selected as the nonrodent species, while another noted that this had been part of their package to support phase 1 clinical development for a couple of programs. One additional company noted that its safety pharmacology experience in the minipig was limited to a cardiovascular safety study for a topical agent. No concerns were raised regarding the incorporation of safety pharmacology end points within standard toxicity studies.
Assessment of immunotoxicity
Only 2 companies reported having assessed immune toxicity in the minipig. In each case, this was done in the context of a general toxicology study. One company simply evaluated peripheral cell counts and draining lymph nodes for a small molecule. The other company evaluated immunoglobulin response to a vaccine as well as cytokine production for a biologic; for this program, the minipig was the only species used.
Assessment of reproductive toxicity
Three respondents reported using the minipig for reproductive toxicity studies. In each case, the minipig was not the only species used for preclinical safety assessment and did not replace the rabbit. One of these companies noted that the only work they had done was an exploratory/pilot program for a biologic. This was an internal decision because the minipig was used for the general toxicology assessments. The other 2 companies had evaluated a small molecule in a minipig embryo-fetal development study. This was an internal decision by 1 company, based on pharmacological relevance and/or pharmacokinetic suitability of the species. The other company had conducted the minipig embryo-fetal development study in response to a regulatory agency request where the dog was the nonrodent species for general toxicology studies. In this case, the regulatory agency (U.S. Food and Drug Administration [U.S. FDA]) was concerned about inconclusive, low-incident skeletal findings in 2 species, the rabbit and rat, and wanted to better understand if the findings were test article related by conducting a study in the minipig as a third species.
Biologics development
Although there was not a specific question regarding the extent to which the minipig is being employed for safety assessment of biologics versus small molecules, the overall responses indicate that minipig use for biologics is relatively low. In fact, 5 companies stated they would currently avoid the use of minipigs for monoclonal antibodies and/or immune modulators. The key reasons cited (perceived or realized), were a lack of background data, lack of reagents or biomarkers to study toxicity and/or pharmacodynamic mechanism of action, concerns regarding immune system characterization and evaluation (as noted above), and poor suitability for developmental toxicity assessments due to the lack of placental transfer.
Regulatory Acceptance
The majority of respondents (9/14) did not consider a perceived lack of experience of regulatory agencies in evaluating minipig studies as a limiting factor for using this species for safety assessment. This was particularly the case for dermal drug development programs, for which minipig studies have at times been requested by the U.S. FDA. Of the 8 companies that have used minipigs for preclinical safety assessment, only 1 reported an occasion where a regulatory agency requested data in an additional nonrodent species to the minipig. The particular concern involved a finding in minipigs observed at a lower dose than expected and the difficulty in comparing this finding to published data available only in the NHP.
On the other hand, 5 respondents did consider perceived regulatory agency inexperience with minipig studies to be a concern. This was mostly in the context of biologics drug development (particularly monoclonal antibodies). Remarks from 2 companies indicated there is presently very little experience with biologics development in minipigs and there is no agency-approved monoclonal antibody or antibody-like molecule for which safety was evaluated using minipigs. One company contrasted this with a steady increase in minipig use for safety evaluation among agency-approved small molecules and for several peptide molecules.
Discussion
Thirty-two companies associated with the IQ Consortium were queried on their current practices regarding the use of the minipig in the safety testing of pharmaceuticals. Fifteen of the companies responded, providing answers on company demographics, facility considerations, and key factors that influence minipig use for safety assessment, the use of minipigs in small molecule and biologics development, and regulatory acceptance. Recognizing the limitations of making broad conclusions based on 15 company participants, there were several practices and trends that emerged from the survey which are largely consistent with the last industry survey that was conducted approximately 5 years ago.
Roughly half of the respondents stated that they use the minipig for nonclinical safety assessments but that the frequency of use is actually relatively small (2–15% of their studies). It appears that in the majority of cases, the minipig is used because the dog is not suitable and the company wishes to avoid the monkey, with 1 company explicitly stating that the minipig needs to be evaluated and negated prior to utilizing the monkey. Most respondents cite that a scientific rationale for the selection of the minipig must be provided, just as it is for the dog or monkey (e.g., pharmacodynamics response, biomarker availability, pharmacokinetics, metabolic profile, etc.) and most also state that there is no species-specific liability that would preclude the minipig as the nonrodent toxicology species. It is clear, however, both from the numbers presented and the responses received that the minipig has yet to reach the status of a default or widely accepted nonrodent species for toxicity testing of pharmaceuticals. This is despite half the respondents stating that they utilize the minipig. Thus, the adoption of the minipig for preclinical safety testing has been slow across the industry, even among companies where it is used. The reasons for these disparities are not entirely clear from the current survey. Several possibilities are (1) the perception that selection of the minipig will alter the development plan, with the size of the animal (and the subsequent drug amounts needed to support the safety studies in later phases of development), (2) lack of adoption for large molecules due to the scarcity of regulatory experience or approved drugs using that development path and challenges in conducting reproductive studies, (3) not normally needed/requested outside of dermal programs, and (4) limited historical data when compared to either the dog or NHP.
In response to the question of whether companies considered health authorities perceived lack of experience with minipig data as a limiting factor for the use in small molecule safety assessments, the majority of companies said no. This likely reflects the recognition that there have been several examples where minipigs have been used in the safety assessment of systemically administered pharmaceuticals, along with their more routine use for toxicology testing of dermal products (reviewed in Bode et al. 2010; van der Laan et al. 2010). Only 1 company reported a negative regulatory consequence, when safety studies were conducted in the minipig. In that particular case, a regulatory agency asked for data in another nonrodent species because of a finding in the minipig at a lower dose than expected. Although the specific details were not provided in this particular case, it seems to represent an example of a species difference in sensitivity which would be no different than what might be expected when comparing results between dogs and monkeys. As such, as long as the selection of the minipig is scientifically justified, there do not appear to be any concerns with using minipigs in general toxicity studies from a regulatory perspective. Of the 5 companies that expressed concerns with regulatory acceptance of minipigs as a toxicology species, most of the concerns were related to safety testing of biopharmaceuticals.
Only companies indicated experience screening or using the minipig for evaluating the safety of biopharmaceuticals. While this may represent an underreporting of use across the industry (given that only 15 of 32 companies responded to the survey), this response is consistent with the previous survey data from Bode et al. (2010) when only 2 of 22 companies reported screening biologics in the minipig. This suggests little progress over the last 5+ years in utilizing the minipig for biologics development. The present survey identified concerns regarding characterization and evaluation of the immune system of the minipig as a key factor regarding their use in safety testing of biopharmaceuticals. This is despite published assertions that the minipig immune system is well studied, has generally similar structure and functions to the immune system of humans, and is perhaps better characterized than the dog and Macaque monkey (Bode et al. 2010). In addition, 1 respondent evaluated immune end points in the minipig for a biologic, showing that characterization of immune system effects in this species does appear to be feasible. Other key concerns that companies cited were lack of historical data, lack of reagents and assays for this species, concerns about immunogenicity, and an understood lack of antibody transfer across the pig placenta. Clearly, there has been continued inability and/or reluctance to generate the necessary data to address such concerns, and possibly such evidence could enable safety testing of biopharmaceuticals in minipigs on a routine basis.
The use of minipigs for the safety evaluation of small molecules is more common, with reproductive toxicology, general toxicology, and safety pharmacology studies being described in the minipig. The minipig placenta is not a barrier for fetal exposure to small molecules, and the minipig is being used to a small extent for embryo-fetal development studies, though notably not as a replacement for the rabbit. That the FDA requested 1 sponsor to conduct an embryo-fetal development study in the minipig as a third species (in addition to rat and rabbit) would appear to indicate the agency has a level of confidence in the use of the minipig for evaluating teratogenic potential. As noted by Bode et al. (2010), a relatively short gestation period and large litter size provide advantages over the dog and NHP, and historical control data are available. Also described by Bode et al. (2010), similarities of the minipig cardiovascular system to humans make the minipig a suitable model for cardiovascular safety pharmacology studies and perhaps a preferable model over the dog. Consistent with this, the present survey indicates that the minipig, when selected as the nonrodent species, has generally been adopted to assess cardiovascular effects prior to human trials.
Consistent with conclusions in previous publications (Ellegaard et al. 2010; Forster et al. 2010b), there did not appear to be any major concerns with respect to animal welfare standards that would preclude or restrict the use of minipigs in toxicology studies. In addition, no company identified any challenges with minipig studies that would prevent compliance with GLPs or OECD. However, it is clear that special considerations must be taken into account before using minipigs, including their husbandry requirements (e.g., housing), importation licenses, and health status. This latter point is especially important since most minipigs bred for research purposes (e.g., Göttingen) are bred in barrier facilities and are unvaccinated; however, they are susceptible to the same pathogens as farm pigs (Ellegaard et al. 2010). Therefore, it is extremely important that the animal facility be monitored for external sources of pathogen contamination and that veterinary and technical staff are fully trained to identify early changes in minipig health status. While these principles are no different than for any other species, the lower frequency of minipig use may necessitate more rigorous and frequent training of staff to ensure full awareness and compliance with the unique health requirements of this species. While this survey did not ask about veterinary training specifically, Forster at al. (2010b) suggested that, from an animal welfare point of view, the use of the minipig is a plus since there is extensive background veterinary medicine experience with this species (similar to dogs) when compared to NHPs. In contrast, at least 2 companies in the previous survey reported by Bode et al. (2010) indicated that technical and/or veterinary staff may not have enough experience with this species to recognize abnormal clinical observations or common background infections. Training and hands-on experience would most certainly address this concern regarding “lack of familiarity” with this species as emphasized in the review by Ellegaard et al. (2010).
However, the current survey, as well as the previous publication of Bode et al. (2010), made it very clear that the minipig is not an easy animal to work with because of its size and aggressive behavior, especially in older/sexually mature males. This is particularly evident when it comes to social housing. In the current survey, 2 companies noted that this could not be accomplished due to the reasons just cited. In contrast, 3 companies reported that social housing of minipigs could be accomplished although one of these companies correspondingly acknowledged that it was not easy. Given the inherent behavioral characteristics of this particular species, it is likely that successful social housing was performed in relatively young animals since male Göttingen pigs reach sexual maturity in 3 to 4 months (Ellegaard et al. 2010). The challenge of working with minipigs from an animal handling perspective was also evident in the current survey. For example, dosing and blood collection were specifically mentioned as procedures that are more difficult to perform and, overall, more technician training seems to be required to conduct studies in minipigs. Whereas most companies acknowledged that performing studies in minipigs required more resources and was more time-consuming, they all reported that 2 to 3 technicians were needed to perform in-life procedures. Interestingly, while most companies with minipig experience also highlighted technician training as a major factor when considering the use of this species, the actual training periods reported by each company varied considerably from as short as 1 month to as long as 1 year. While we did not request additional details on the specific training programs, the variable responses most likely reflect different interpretations of what constitutes “sufficient training” as well as the specific procedure. For example, it may only take several weeks to train someone to perform relatively simple animal husbandry tasks, but it may take at least several months for a technician to become proficient in clinical observations, dosing, and/or blood collection. In the manuscripts by Ellegaard et al. (2010) and Forster et al. (2010b), the authors highly recommend positive-reinforced social interactions with humans to reduce stress and ensure easier animal handling. Therefore, it appears that the increased resources required for minipig studies was mostly attributed to up-front training and that once technicians are fully competent in minipig procedures, the number of technicians required for these studies is similar when compared to other nonrodent species, specifically NHP’s.
In regard to housing facilities for the minipig, several companies in this survey reported a requirement for redesigned accommodations. Two companies with an in-house facility had modified existing caging and another company reported renovations to animal rooms. Although limited information was provided, the intent of these redesigns was clearly to accept minipigs, improve the environment for minipigs, and/or aid technical staff by easing animal handling. Temperature, humidity, and feed were noted as special housing considerations for minipigs; however, details on how these environmental factors differed for minipigs compared to other nonrodents were not provided. Several literature references acknowledge that minipigs are sensitive to environmental conditions and should be maintained under controlled temperatures based on the size and age of the animals (Council of Europe 2006; Ellegaard et al. 2010; Swindle et al. 1994). However, the suggested temperature ranges for housing minipigs (22–30°C) seem to be well within the control range of most vivaria within large research facilities so it is not clear why this has been raised as an issue. Similarly, the recommended humidity range for housing minipigs (40–70%; Swindle et al. 1994) is not much different than other species. Nevertheless, the comments on facilities in the current survey are very consistent with those reported by Bode et al. (2010) in which housing, husbandry, and facilities were identified by several companies as logistical hurdles for the use of minipigs. We did not specifically ask for more detailed reasons so it could be that caging and floor space were the real drivers behind these concerns. If so, it is not hard to understand why the time and costs associated with new caging and/or facility renovations would be an added factor for the slow uptake of minipigs in safety studies since the last industry survey.
While the decision to use minipigs may be driven by efforts to reduce the use of other nonrodent models in biomedical research, substantial evidence exists to suggest that the anatomy, physiology, and biochemistry of minipigs are comparatively representative of humans, and therefore the minipig may be considered as a valid nonrodent species for pharmaceutical safety testing. The results of this survey, albeit a small representation of the pharmaceutical industry, indicate that questions of regulatory acceptance and housing are not perceived as barriers for the use of minipigs in safety assessment of small molecules; however, minipig use has not substantively increased more broadly in pharmaceutical companies since the last industry survey conducted over 5 years ago.
Footnotes
Acknowledgments
The authors will like to thank Jennifer Alldredge (survey formatting and design) and Sarah Vanbibber (technical and clerical coordination) and all of the members of the BMS Core Team (Randy White, Chandrashekhar Korgaonkar, Robert Schulingkamp, and Kristin Horn) for all of their assistance in making this manuscript a success.
Authors’ Contribution
All authors (CC, DB, AC, DC, PH, AO, and MG) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
