Abstract
Whereas the nature of healing reaction in skeletal muscle following implantation of biomaterial has been extensively studied, the extent of variation in cell phenotypes is poorly known. Here, we studied the phenotypic alteration of cell types following injury in skeletal muscle of rabbits implanted with a commonly used biomaterial, polyethylene terephthalate (PET) fabric. Following implantation, histomorphological features were studied after 1, 4, and 12 weeks. Routine objective histomorphological evaluation was supplemented with histochemistry for collagen and immunohistochemistry for proliferating cell nuclear antigen (PCNA), CD34, vimentin, and alpha smooth muscle actin (α-SMA). The extent of reaction was quantified. The foreign body giant cells were found to comprise subpopulations, based on the variation in vimentin detectability or the presence of differentially capable proliferating nuclei (PCNA positive). Many rhabdomyocytes adjacent to the implant were PCNA-positive and some of them showed CD34 positivity. Often, the rhabdomyocytes very near to implanted PET fabric assumed a myofibroblast phenotype as evidenced by vimentin and/or α-SMA positivity at immunohistochemistry. Overall, the results suggested a phenotypic alteration of native cell types following implantation of PET fabric in rabbit skeletal muscle. Quantification of such cell types at the tissue–material interphase in relation to the deposition of collagen may be desirable during safety evaluation of biomaterials by histomorphology.
Introduction
The nature of tissue reaction induced by biomaterials implanted at various sites of the body of laboratory animals assessed by defined procedures is a screening test for identifying potential biomaterials for device fabrication. Widely accepted standard protocols and procedures, which provide insights into the safety and probable biomedical use of these biomaterials, are available (Schuh 2008; Turner, Lawrence, and Autian 1973; Helmus, Gibbons, and Cebon 2008). These standards/procedures largely evaluate the overall tissue reaction pattern of the extracellular matrix around the implant site, as assessed by the extent and nature of fibrosis, and the abundance of various kinds of cells participating in the healing response. The development of these standards is centered on the classical definition of biomaterials that it is the ability of a material to perform with an appropriate host response in a specific situation (Williams 1987). However, the definition of “biocompatibility” has been revisited repeatedly (Williams 1999, 2003, 2008) and necessitates an assessment based on a more mechanistic view of the healing response incorporating the current understanding of cellular and molecular pathology and clinical implications. Therefore, it is desirable to identify and include additional parameters in the safety assessment protocols, which have a bearing on cellular and molecular pathology and the expected clinical outcome (Helmus, Gibbons, and Cebon 2008).
Skeletal muscle continues to be the preferred test site for evaluating biocompatibility and an injury caused by an implanted biomaterial initiates and perpetuates a healing response (ISO 10993, Part-6). The sequence of events occurring in a healing reaction in muscle is predominated by repair mechanisms orchestrated by granulation tissue formation, angiogenesis, and fibrosis and substantially lacking a regenerative response as in epithelial tissue. Detailed investigations for identifying different cell types in reaction zones around the implant sites have indicated the presence of differentially altered cell types including stem-like cells (Swathy and Anilkumar 2007; Thevenot et al. 2010). Moreover, biomaterials are increasingly used for fabricating engineered tissue constructs (Jayo et al. 2008). Together with the biological molecules (Anilkumar et al. 2011) and the cells in the construct, the biomaterials are likely to stimulate cell types (e.g., stem cells and giant cells) that are not prominent in the tissue during physiological states. Hence, there is an increased quest for understanding the nature of cellular and molecular modulations occurring at the implant niche.
Various kinds of polymeric materials are used in the fabrication of biomedical devices and histopathology is used as a tool for assessing their biocompatibility (Velayudhan et al. 2005; Anilkumar et al. 2007). Polyethylene terephthalate (PET) is a widely used biocompatible polymeric material that has been tested on a number of occasions and found suitable for a variety of biomedical applications like vascular prostheses, hernia repair meshes, suture material, and patches for tissue repair (Cenni et al. 1996; Ratner et al. 2004; Seitz et al. 1998). When implanted in vivo, it causes chronic granulomatous inflammatory reaction in sites like muscle and subcutaneous tissues of animals (Hagerty et al. 2000; Tang and Eaton 1993; Van Bilsen et al. 2004). However, the nature of various cell types participating in the tissue reaction induced by implanted polymers has not been investigated in detail. In the present study, using PET as a model material we studied the nature of healing reaction caused by implanted polymeric biomaterial in rabbit muscle with special reference to the density, distribution, and function of mesenchymal cell types and giant cells occurring in the reaction zone.
In this article, we support the hypothesis that the traditional battery of histopathology test parameters listed in standard procedures designed for biocompatibility testing may have to be expanded by adding those parameters which might reflect the nature of cellular and molecular mechanisms regulating biocompatibility. Utilizing the advances in computer-assisted histomorphology-imaging techniques, efforts were taken to appreciate these additional parameters during biocompatibility evaluation.
Materials and Methods
Preparation of the Polymer Samples
PET fabric used for this study were cut from vascular graft samples provided by M/s. TTK Healthcare Ltd, Trivandrum. Vascular graft samples were prepared by weaving doubled 76 denier 34 filament texturized yarn. Weaving is carried out using modified conventional loom and the weaving parameters are adjusted to achieve a mean water porosity of 200 ml/min/cm2. The weaving generates a tubular structure without seams. These tubular fabrics are further crimped to ensure that the grafts do not kink during use. The crimped grafts are cleaned in a multiple stage cleaning/extraction process including ultrasonic cleaning, methanol extraction, and water extraction to remove all chemicals and oils that might have accumulated on the fibers during the fabrication process. For implantation, strips of size 10 mm × 6 mm were cut using clean scissors, washed with distilled water thrice and finally with acetone, and dried. Samples were sterilized with ethylene oxide prior to implantation.
Animal Implantation
The animal experiments were done with the approval and as per the requirements of the Institutional Animal Ethics Committee. Nine healthy adult rabbits weighing not less than 2.0 kg and whose paravertebral muscles are sufficiently large in size were used for the experiment. Prior to the experiment, fur on either side of spine was clipped off. Implantation procedure was carried out under clean and asceptic conditions. Rabbits were anesthetized using Ketamin hydrochloride (80 mg/kg) and Xylazin (5 mg/kg body weight). The skin of the anesthetized rabbits were lightly swabbed using 70% alcohol and air dried. The PET was inserted intramuscularly into paravertebral muscle through skin incision wound. Two implants were inserted, one each on either side of the vertebral column. The incision was then closed using sterile sutures. At the end of each experimental period (7 days, 14 days, and 12 weeks), the rabbits (3 rabbits per period) were euthanized by an overdose of the anesthetic agent. The implant material along with the surrounding tissue was collected and fixed in 10% neutral buffered formalin, as per the standard, so as to fix tissue specimens for subsequent studies.
Histotechnology
Formalin-fixed tissue samples were dehydrated through ascending grades of alcohol and impregnated with paraffin wax. Tissue sections (4 µm thick) were made from paraffinized tissue blocks using Leica RM2255 Microtome. Tissue sections were deparaffinized in three changes of xylene and brought to water through descending grades of alcohol. The sections were then stained with Harris's haematoxylin and eosin (HE). Additional histochemistry for evaluating the pattern of collagen, tissue sections were stained with picrosirius red and Masson’s trichrome (Bancroft and Gambling 2008).
Immunohistochemistry
Mouse monoclonal antibodies (Table 1) were used for detecting various cell types by immunohistochemistry . Essentially, deparaffinized tissue sections were first washed in water, treated in antigen retrieval buffer (citrate buffer, pH 6.8) at 92°C for 20 min and allowed to reach room temperature in about 30 min. Then, the endogenous peroxidase activity was prevented by treating the slides with 10% H2O2 for 15 min and the nonspecific binding of secondary antibody was blocked using 10% goat serum. Later, the sections were washed with trisodium citrate solution and treated with primary antibody (Table 1) for 1 hr at room temperature. The sections were then washed and treated with goat antimouse Fab’2-Biotin conjugate (Santa Cruz Biotechnologies, Inc.) for 30 min. Subsequently, sections were washed and treated with Streptavidin-horseradish peroxidase conjugate (Vector Laboratories) for 30 min. The sections were then washed thoroughly and treated with the 3,3′-diaminobenzedine-enhanced liquid substrate system (Sigma) for 5 min and the reaction was stopped by immersing the slides in excess distilled water. Finally, the sections were counterstained lightly with Harris’s hematoxylin, dehydrated in ascending grades of alcohol, cleared in xylene, and mounted in DPX.
List of mouse monoclonal antibodies used for immunohistochemistry.
Confocal Microscopy
Tissue sections stained with Alexa flour conjugated anti-CD34 mouse monoclonal antibody (ICO115, Santa Cruz Biotechnologies) were imaged under a Carl Zeiss LSM 510 META Laser Scanning Confocal Microscope equipped with differential interference contrast optics. For visualization, the stained sections were excited with 633 nm laser light, and emission wavelength was filtered by 650 nm long pass filter.
Histomorphology
Histomorphology of the tissue was evaluated routinely for studying the nature of tissue reaction as recommended in ISO 10993 for assessing biocompatibility. The parameters studied were the following. The number and distribution of neutrophils, lymphocytes, plasma cells, eosinophils, macrophages, and foreign body giant cells (FBGCs) around implant material were semiquantitatively graded and other parameters such as extent of fibrous capsule formation, inflammation, severity of degeneration, necrosis, fatty infiltration, granuloma formation, presence of material debris, and tissue in-growth were evaluated by a veterinary pathologist.
Histomorphometry
Images of histology slides were captured using an Olympus 51 BX microscope fitted with a DP71 camera using Image-Pro 3DS (version 6.1) software (Media Cybernetics, Silver Spring, MD). For all parameters, at least six histology slides were available. The parameters of interest were the following.
Extent of tissue reaction. The extent of tissue reaction caused by the muscle implant was determined on HE-stained tissue sections. The maximum distance from the border of implant material to the normal tissue having no inflammatory cells and fibrosis was measured and expressed as extent of tissue reaction (ISO 10993 Part-6).
Collagen quantification. Collagen deposited around implant materials was quantified morphometrically in picrosirius red–stained tissue. The collagen in the tissue is stained red by the Sirius red component of the picrosirius red stain. The percentage of red colored area was calculated and expressed as the percentage of collagenous area.
Number of proliferating cells. Number of proliferating cells in the implant tissue was counted using image-pro software. High-resolution photographs of proliferating cell nuclear antigen (PCNA)-stained sections were taken under 40× objective (n ≥ 10, per section) and proliferating cells were counted using manual tag option. The mean was calculated for each sample. Results were then expressed as mean number of PCNA positive nuclei per high power field (HPF) for each time point.
Number of SMA and vimentin-positive cells. The number of SMA and vimentin-positive cells with skeletal muscle morphology was counted in immunohistochemically stained sections at 40× objective using a Nikon (E200) microscope. The skeletal muscle morphology was confirmed by the presence of skeletal muscle striation. At least 10 HPFs were assessed for each of the six histology slides and the data as mean ± standard error mean [SEM] (n = 6).
Statistical Analysis
Student’s t-test was performed to calculate the level of significance between experimental groups. Those results with p value less than .05 were considered to be statistically significant.
Results
Implantation, Necropsy, and Gross Morphology
The general physical condition of all experimental animals was normal throughout the experiment. There was no gross evidence of encapsulation infection or necrosis around the implanted materials when retrieved.
Histomorphology
The implanted PET was obvious as a large material embedded in the muscle and the constituent fibers appeared as refractive bodies of 12 to 20 µm diameter fibers of varying size and shape. Tissue reaction was mainly constituted by the various types of inflammatory cells and fibroblasts around the implanted material with some extracellular matrix reaction (Figure 1). At the first week (Figure 1A and B) of implantation, the predominant cell types were polymorphonuclear leukocytes along with a few macrophages, plasma cells, and lymphocytes. FBGC reaction was minimal at that stage. Moderate degree of degeneration and necrosis of various cells were observed. At 4th week (Figure 1C and D) of implantation, macrophage infiltration predominated the infiltrate along with lymphocytes indicating a granulomatous type of tissue reaction. In addition, FBGCs (5–6 per HPF) were seen suggesting a foreign body granuloma. Necrosis and degeneration of cells were less compared to that of first week. At 12th week (Figure 1E and F) of implantation, the tissue response was characterized by chronic granulomatous reaction with a well-formed fibrous capsule limiting the reaction zone around the implant.
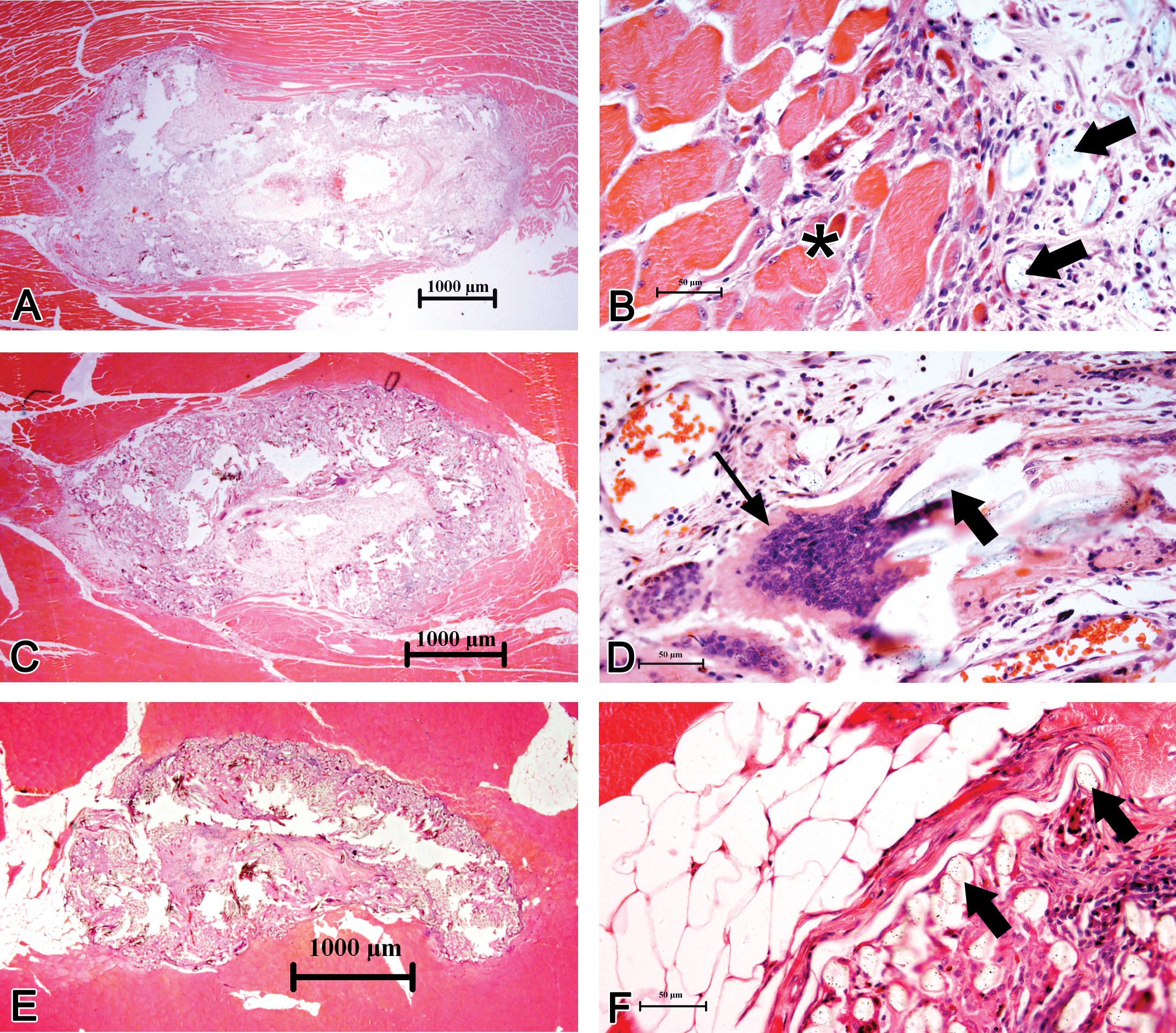
Photomicrographs of the progressive healing responses generated in the rabbit muscle initiated by the implanted biomaterial (polyethylene terephthalate [PET]), after 1 week (A and B), 4 weeks (C and D), and 12 weeks (E and F). At low magnification (A, C, and E), the material appeared as a mass of foreign body embedded in muscle surrounded by a narrow reaction zone but individual polymeric fibers were discernable as refractile bodies at higher magnification (a few examples are marked with thick black arrow). Extensive muscle injury and acute inflammatory response were evident with moderate degeneration and necrosis of rhabdomyocytes and proliferation of fibroblasts at the tissue material interface (B). Damaged muscle cells were seen even after 4 weeks, but chronic inflammation with foreign body giant cell (FBGC; thin black arrow) reaction and most of them were of conventional type with innumerable nuclei and without any obvious features of muscle-derived multinucleated cell (D). By the 12th week, moderate lipocytein filtration, formation of fibrous tissue capsule, and foreign-body granuloma were prominent (F). Please see Figure 2 and Table 2 for semiquantitative data on various parameters of tissue damage.
Quantification of Cell Types
The number of various kinds of cells at various stages is shown in Table 2.
Semiquantitative parameters studied for evaluating histopathology (parameters as per-ISO 10993, part-6).*

*All values are represented as Mean ± SEM (n = 6), please see the text for the data collection procedure.
aGrading based on skilled observation (MD, material debris; F, fatty infiltration; G, granuloma): 0 = not present, 0.5 = minimally present, 1 = mild degree, 2 = moderate degree, 3 = severe degree.
bNumber and distribution of cells expressed as scores based on skilled observation (N, neutrophils; L, lymphocytes; P, plasma cells; E, eosinophils; M, macrophages; GC, multinucleated giant cells): 0 = 0 cells, 0.5 = 1–5 cells, 1 = 6–15 cells, 2 = 16–25 cells, 3 = >26 cells.
Extent of Tissue Reaction
The extent of tissue reaction was found to be maximum (~135 mm) during the first week of intramuscular implantation (Figure 2). At 4th week, the reaction was significantly less (~90 µm) and by 12th week the extent of tissue reaction reached minimal (~70 µm).
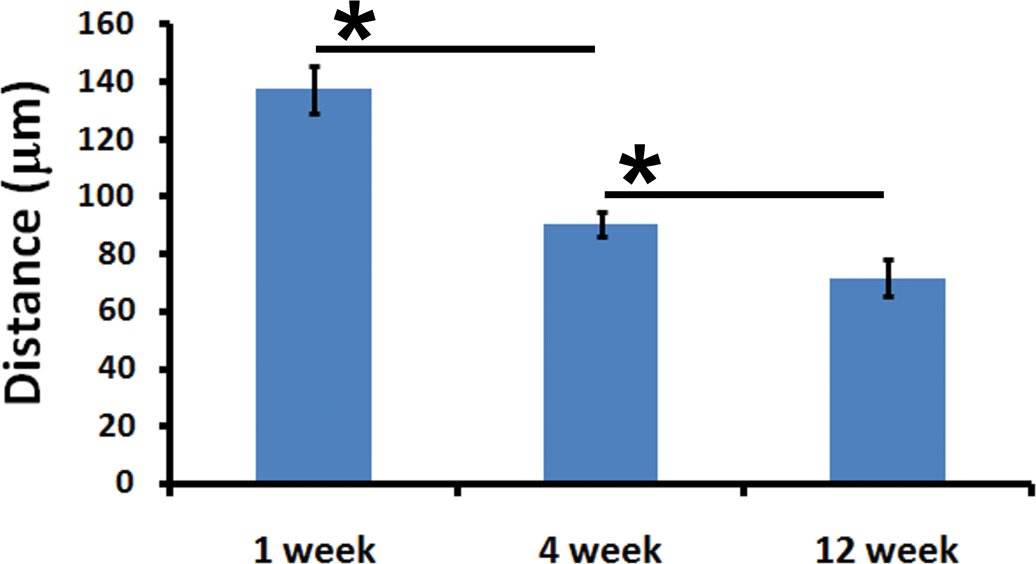
The extent of response, defined by the distance between normal tissue and the implant, gradually decreased over time indicating a favorable healing process (mean ± SD, n = 6).
Severity of Degeneration and Necrosis
The severity of degeneration and severity of necrosis of skeletal muscle cells (Figure 3) were least after 12 weeks, maximum at the 1st week and intermediate at 4 weeks.
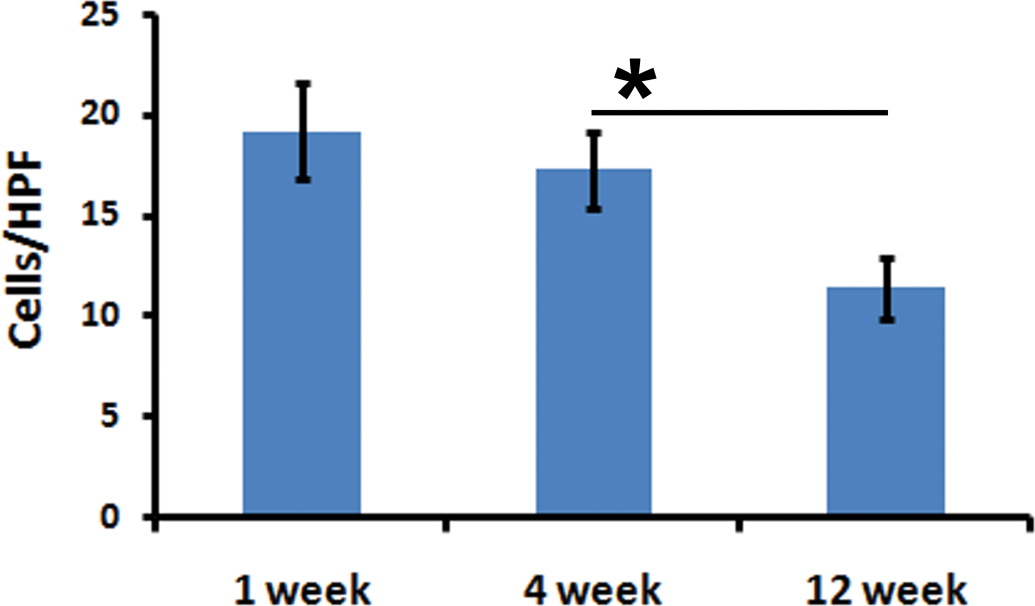
The number of rhabdomyocytes undergoing degeneration/necrosis decreased with advance in time (Mean ± SEM, n = 6).
Extent of Fibrosis (Collagen Content)
Picrosirius red staining showed that the extent of collagen deposited in the implant tissue increased as the period of implantation advanced. After the 1st week, collagen content in the implant tissue was 11.6% and at 4th week it increased to 21.1%. By the 12th week, almost all the PET fibers were surrounded by thick fibrous capsule with about 28.4% of the implant area deposited with collagen (Figures 4 and 5).
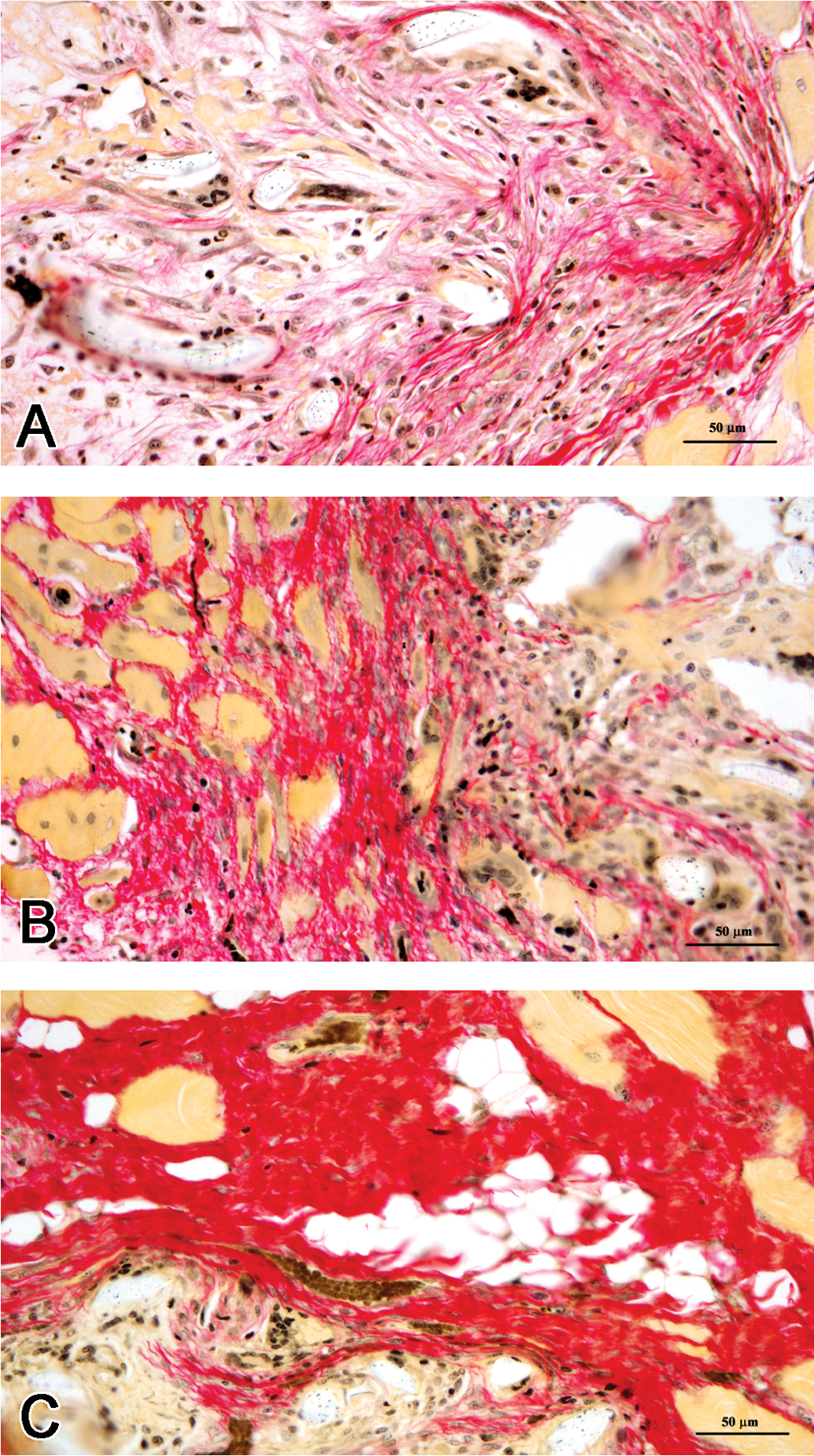
The extent of collagen deposition progressed with time (A, 1 week; B, 4 weeks; C, 12 weeks) as revealed by picrosirius red staining. The collagen deposition was largely restricted to the interphase between the material and the musculature.
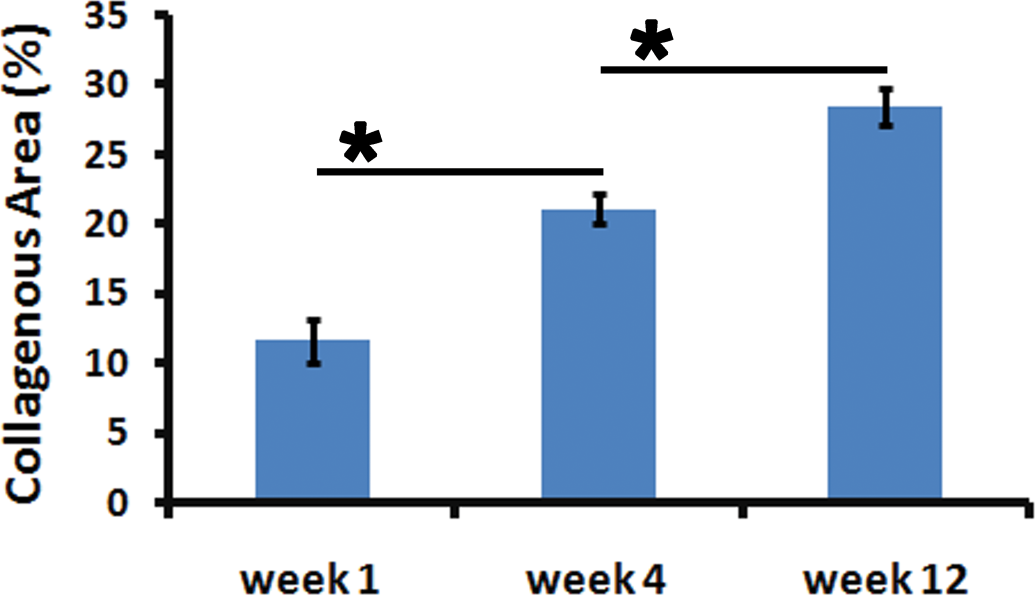
Data on the extent of collagen deposition as revealed by quantitative morphometry (Mean ± SD, n = 6).
Immunohistochemistry
PCNA
The cells positive for PCNA were primarily subpopulations of fibroblasts, macrophages, lymphocytes, endothelial cells, and FBGCs. In addition, a lot of cells at the reaction zone at the interphase between healthy muscle bundles and the implant were PCNA-immunopositive (Figure 6). The number of proliferating cells per HPF at 1st week was ~133 and at 4th week this was found to be maximum (~180). At the 12th week of implantation, the proliferating cells were least (~77; Figure 7).
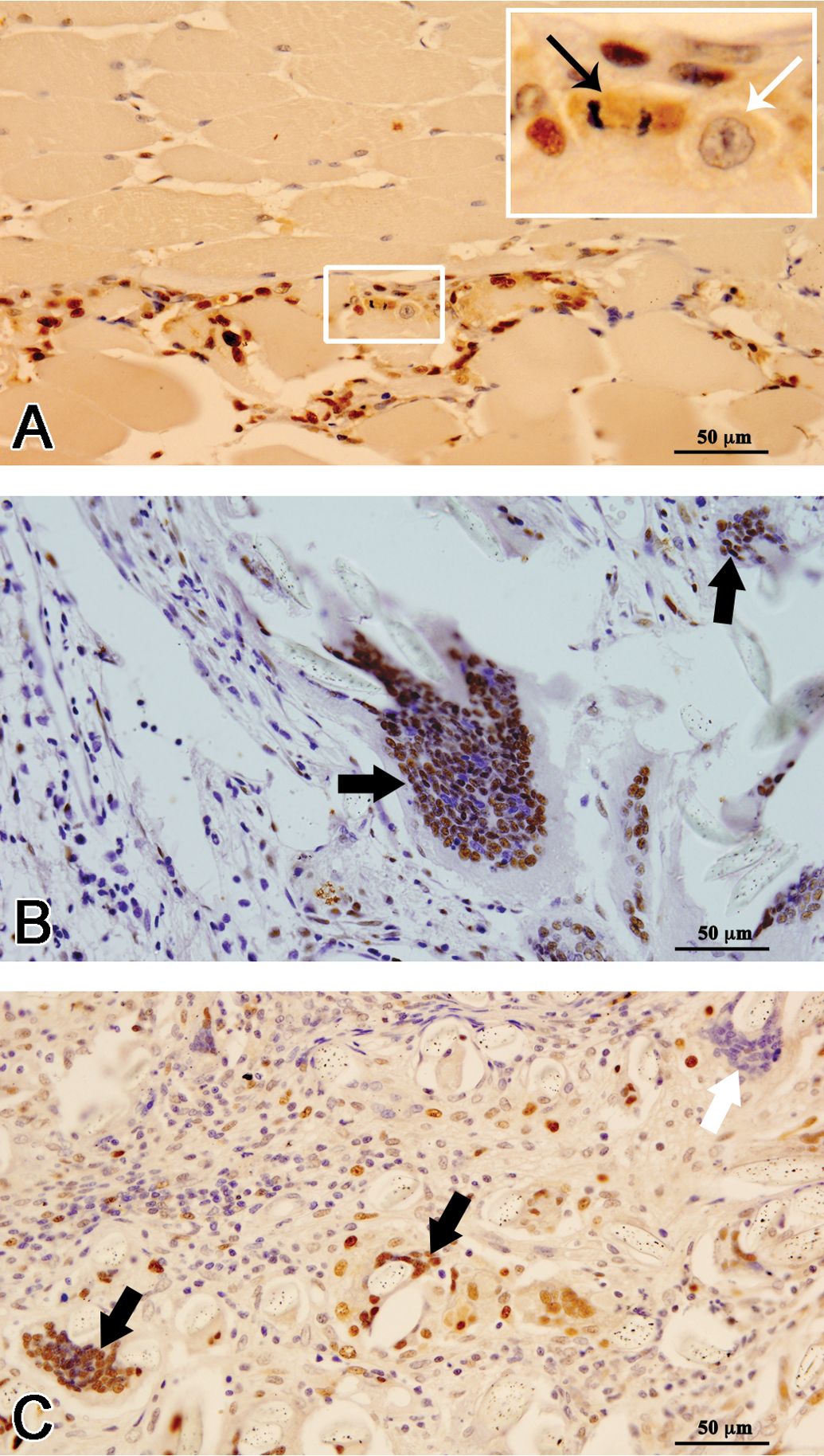
At early time point of the study (A, 1 week), many small cells around the rhabdomyocytes were positive for proliferating cell nuclear antigen (PCNA)-immunostaining. Occasional mitotic figures (thin black arrow) and blast-like cells (thin white arrow) were seen at the interface between healthy muscle (upper half) and injured muscle (lower half); presumably, these are satellite cells. Cells of inflammation and fibroblasts were also proliferatively active (B, 4 weeks and C, 12 weeks). Based on immunophenotyping for PCNA-antigen, there seems to be at least two subpopulations of phagocytically active (which have visibly engulfed foreign bodies) giant cells: those with PCNA-positive nuclei (thick black arrow) and those with PCNA-negative nuclei (thick white arrow).
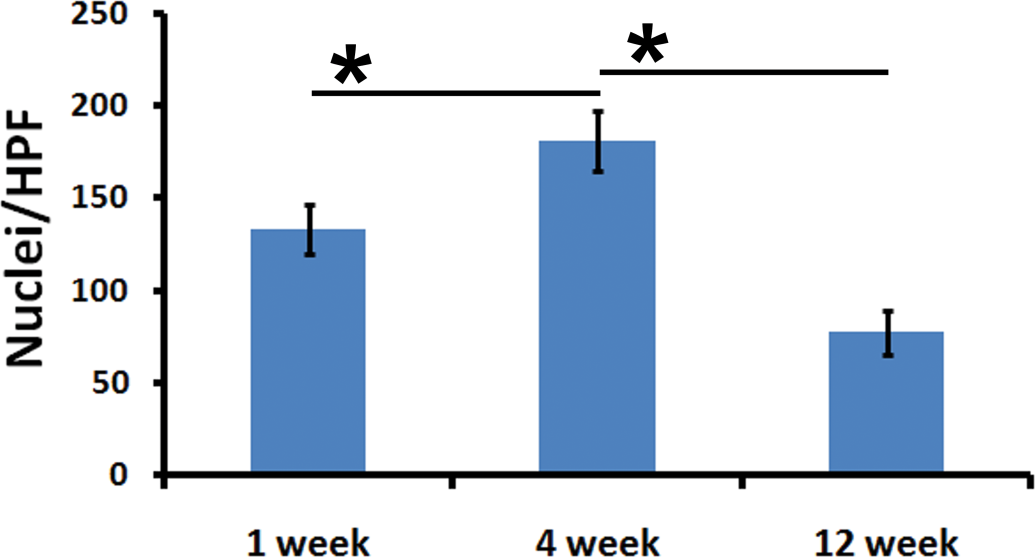
The number of PCNA-positive cells (Mean ± SEM, n = 6).
CD34
As expected, by routine immunohistochemistry, the CD34 antibody (clone, QBEnd/10) detected several satellite cells (data not shown) and the rhabdomyocytes were CD34 negative. However, occasional rhabdomyocytes were marked by the Alexa Fluor 647-conjugated CD34 antibodies (Clone, ICO115), when visualized by confocal microscopy coupled with differential interference contrast microscopy (Figure 8).
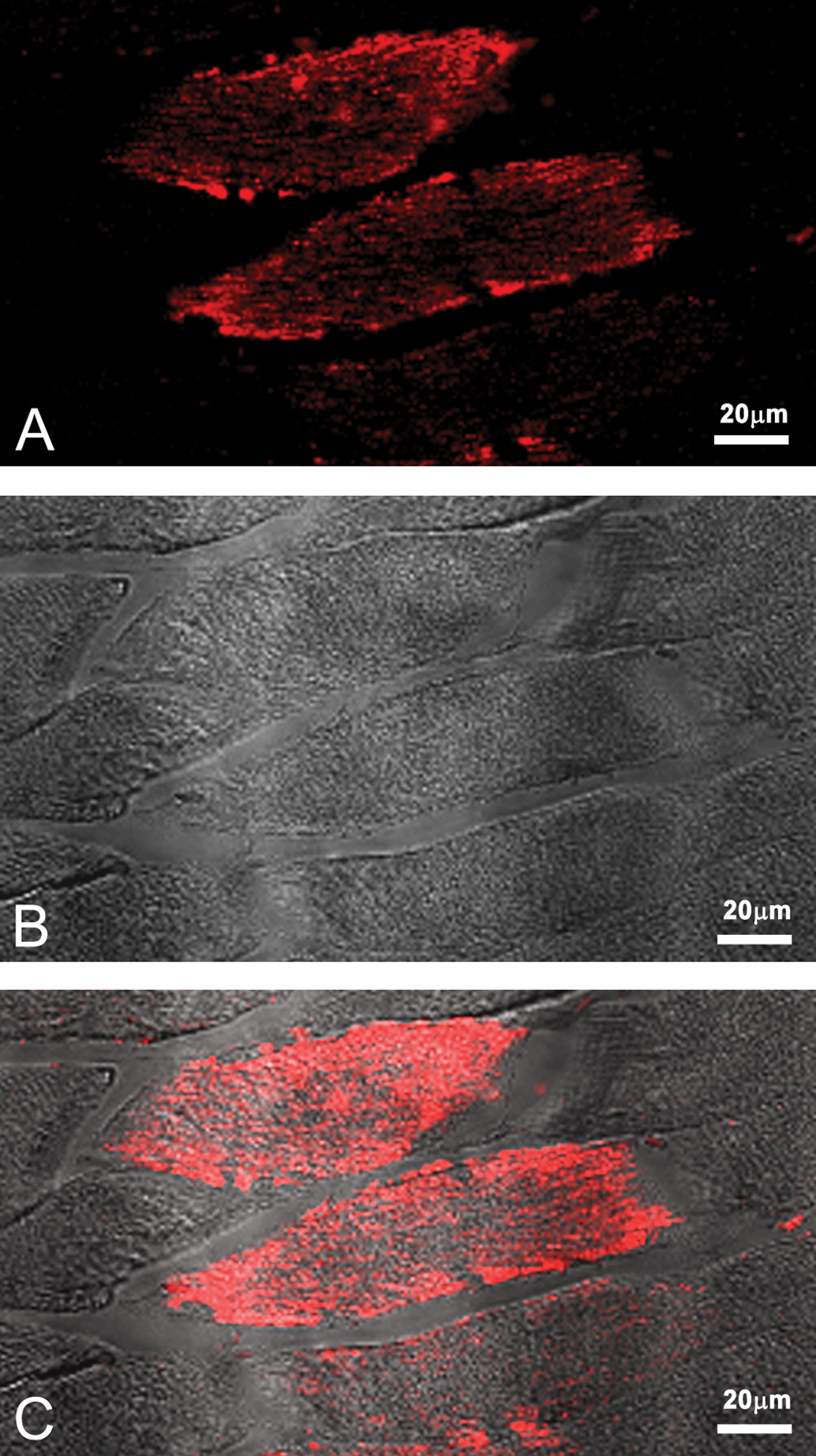
Laser scanning confocal microscopic images of cells containing CD34 antigen (A) suggested that some rhabdomyocytes with visible striations at differential interference contrast microscopy (B) assumed an immature phenotype. Image C is a merged image of A and B.
Vimentin
Fibroblasts were found to be the primary cells expressing vimentin. Neutrophils, macrophages, and lymphocytes in the reaction zone were also vimentin positive but they could be differentiated from the former based on cellular and nuclear morphology (Figure 9). Some of the skeletal muscles (~6.6 cells per HPF) immediately adjacent to the implant were found to be expressing vimentin especially at 1st week. Thereafter, these vimentin-positive cells with skeletal muscle morphology decreased in number (0.52 cells per HPF at 4th week and 0.2 cells per HPF at 12th week). Figure 10 shows the number of vimentin-positive cells with morphology of skeletal muscle.
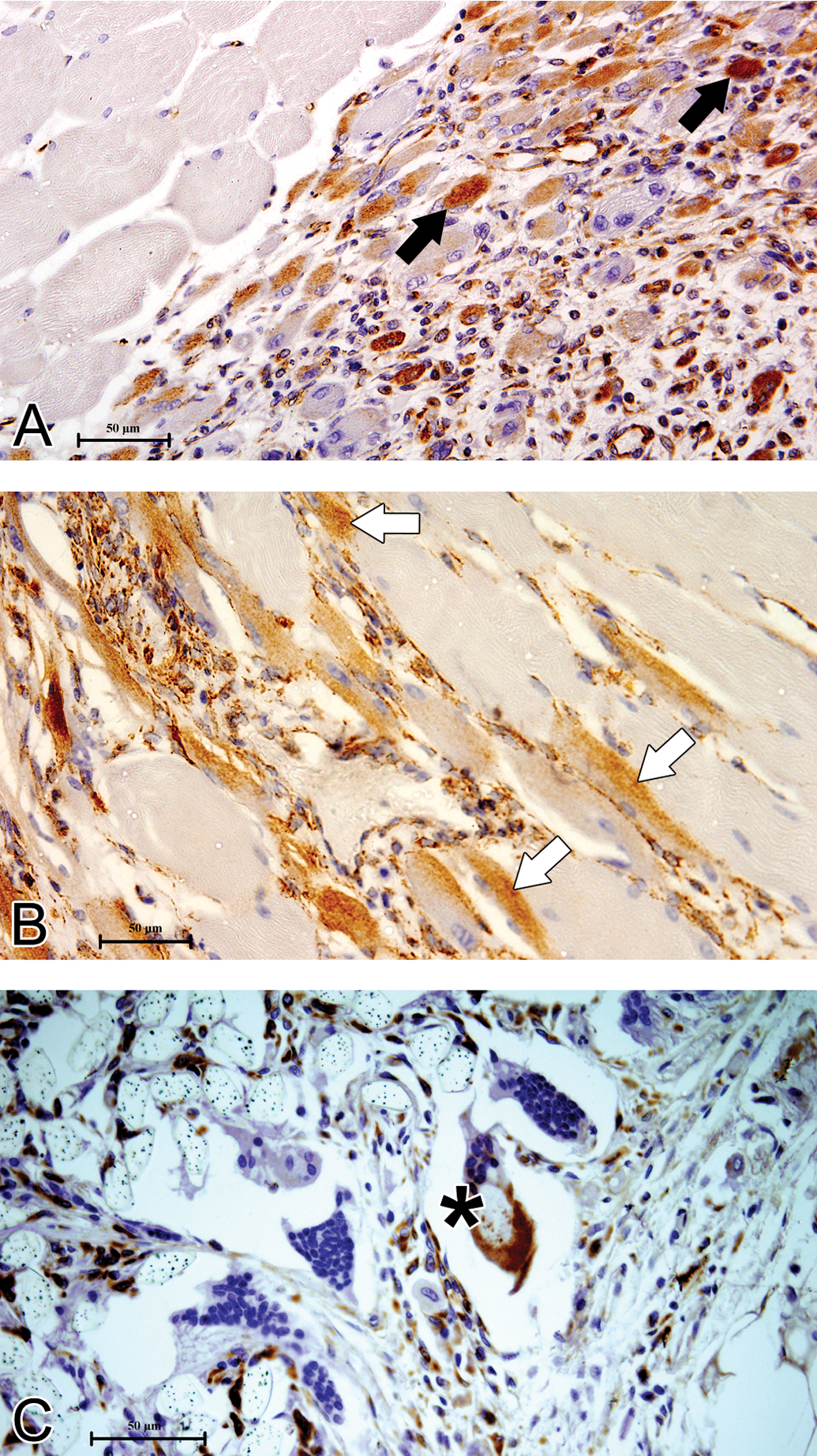
As expected, in a majority of tissue sections studied, the well-differentiated muscles (A, upper-left half) did not contain any vimentin, the immunohistochemical marker for nonmyocytic mesenchymal cells, but most of the cells in the zone of inflammation (A, lower-right half of the photomicrograph) were positive. Among these positive cells, many were injured rhabdomyocytes (black arrow), but at least some were apparently healthy striated muscle cells (B, white arrow) indicating a possible transient status of dedifferentiation. A subpopulation of foreign body giant cells (FBGC; C, black star) were also vimentin positive at immunohistochemistry.
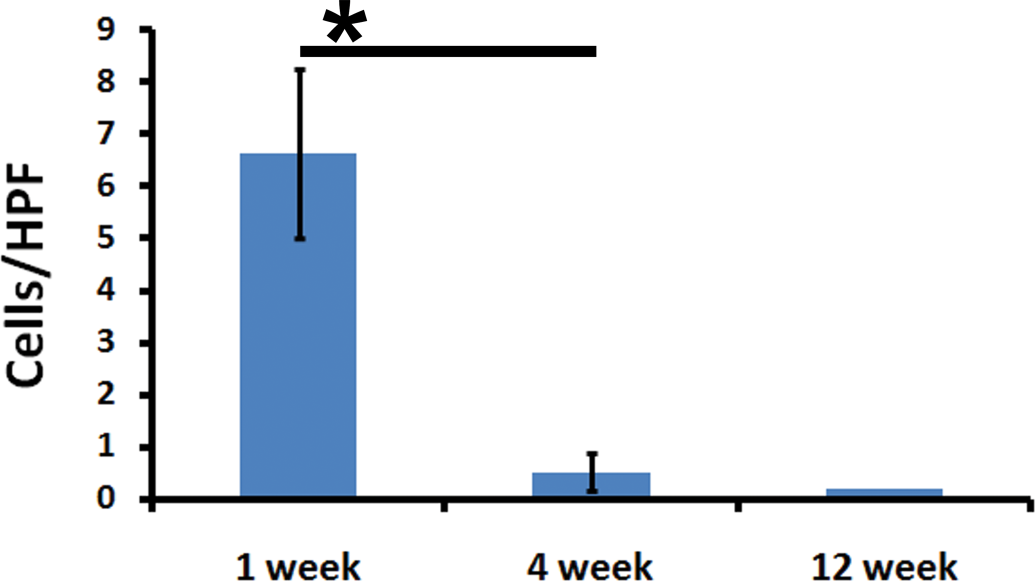
The number of vimentin-positive rhabdomyocytes reduced significantly over time.
Alpha smooth muscle actin (α-SMA)
α-SMA immunostaining detected smooth muscle cells as well as cells with myofibroblast phenotype. Several of the striated muscles immediately around the implant material were also found to be expressing α-SMA (Figure 11). The number of α-SMA positive skeletal muscles was more at early time point of the study (~7.4 cells per HPF) and thereafter decreased significantly (3.5 cells per HPF at 4th week and 2.5 cells per HPF at 12th week). Figure 12 shows the number of α-SMA positive cells with morphology of skeletal muscle. Smooth muscle cells present in the blood vessel walls of the same tissue sections served as internal positive control.
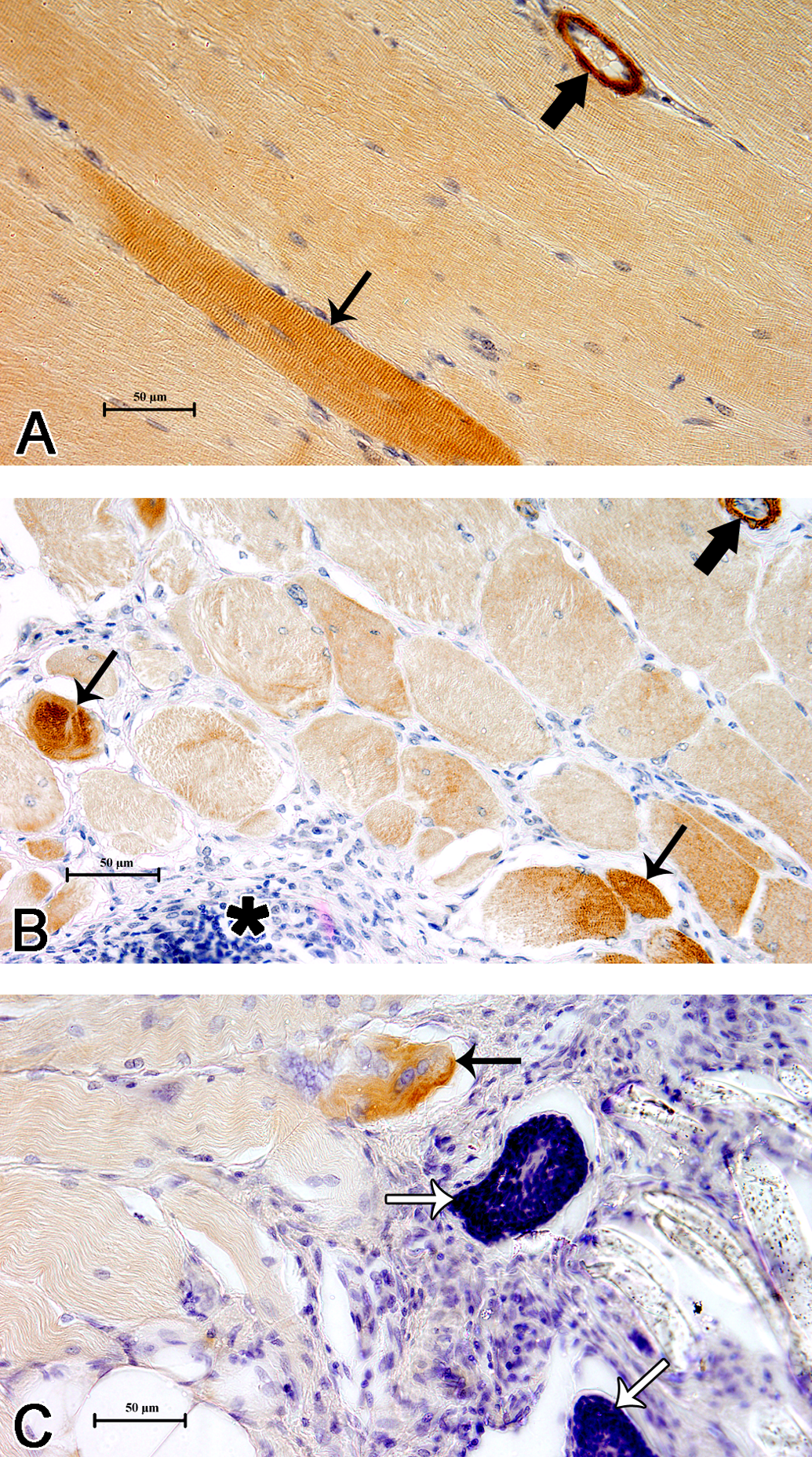
When studied by immunohistochemistry (A, B, and C) for α-SMA, an intermediate filament protein characteristically expected in cell types with contractile properties, smooth muscle cells of blood vessels (thick black arrow) and interestingly, a subpopulation of skeletal muscle cells (thin black arrow) were also positive. Cells of inflammation (black star) or foreign body giant cells (FBGC; thin white arrow) did not pick up any positive signal, as expected.
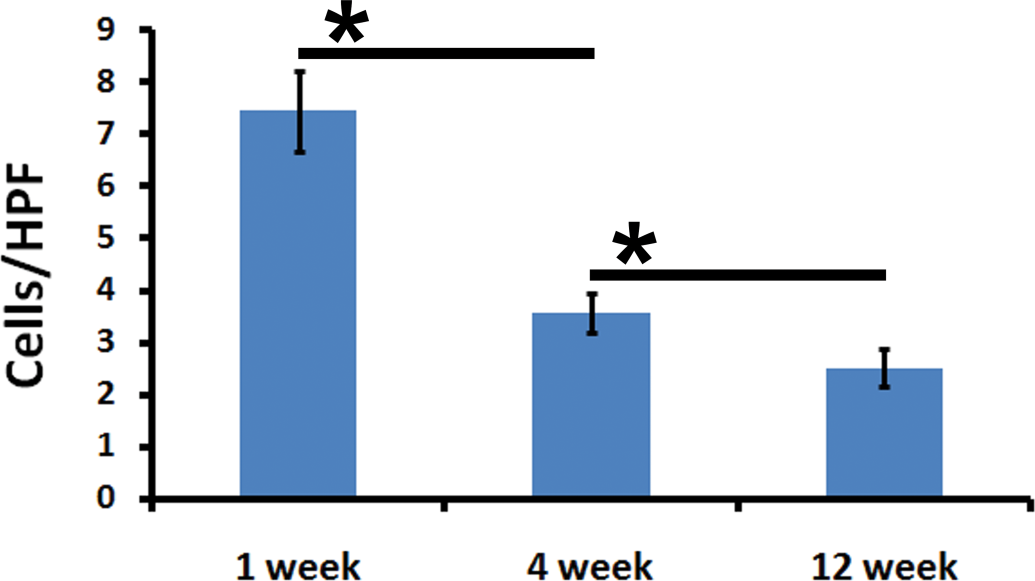
The density of alpha smooth muscle actin (α-SMA)-positive cells reduced significantly over time reflecting a pattern similar to that of vimentin-positive rhabdomyocytes demonstrated in Figure 10.
Discussion
In order to study the occurrence of novel cell types in biomaterial-induced healing response, we implanted a model polymeric material PET fabric intramuscularly into rabbits as stipulated in standard procedure (ISO 10993, Part-6) and histomorphologically evaluated the cellular reaction as recommended (Table 2). Although comparison with a biocompatible reference material was not performed, the data presented in Figure 1, Table 2, and previously published data (Anilkumar et al. 2007; Cenni et al. 1996; Ratner et al. 2004; Seitz et al. 1998) largely indicated that the biomaterial in consideration induced tissue reaction acceptable for a biocompatible material. We further studied the distribution and nature of various cell types generated at the reaction site. Emphasis was placed on quantification of the reaction and the histogenesis of the emerging cell types and phenotypically altered cell types in the reaction zone.
The number of proliferating cells at different time points of the study gave an indication of the severity of reaction around the implant. The cells undergoing proliferation around the implant site were classified as native cells in the skeletal muscle (fibroblasts in the supporting connective tissue, smooth muscle cells, or endothelium of the blood vessels) and those recruited from the general circulation as part of the inflammation. We studied the distribution of two populations of cells, one each from the above groups containing PCNA-positive nuclei: satellite cells and the giant cells.
Since rhabdomyocytes are terminally differentiated cells incapable of proliferation, the pool of proliferating cells is expected to contain endothelial cells, smooth muscle cells, fibroblasts, and cells of the reticuloendothelial system (lymphocytes and macrophages) rather than the former. The types and number of proliferating cells in the tissue reaction around the implant are shown in Figures 6 and 7. At the 7th day, some of the nuclei of the skeletal muscles adjacent to the reaction zone were found to be PCNA positive. The location and morphology of these cells suggested that they were satellite cells that have been activated upon injury associated with PET implantation. Mitotic activity and immature (blast-like) features, with scanty cytoplasm, large nuclei, and prominent nucleoli, of some of the cells in the reaction zone supports this claim (Figure 6A). Indeed, satellite cells are known to have stem cell properties (Zammit, Partridge, and Zipora Yablonka-Reuveni 2006) and activation of stem cells following tissue injury in muscles is possible in injured muscles (Zammit and Beauchamp 2001). Hence, the possibility of stem cell activation during biocompatibility evaluation cannot be ignored. Also, a previous study (Swathy and Anilkumar 2007) indicated the possibility of the occurrence of stem-like cells in the reaction zone following biomaterial implantation in muscle. With these in background, attempts were made to identify stem cells by phenotyping CD34 antigen using monoclonal antibodies from two different sources. One of these antibodies (clone, QBEnd/10) identified the conventional satellite cells. On the other hand, occasionally the skeletal muscle cells with muscle striations were positive when stained with Alexa Fluor–conjugated anti-CD34 monoclonlal antibodies (clone ICO115) and imaged by confocal microscopy coupled with differential interference contrast microscopy (Figure 8). The CD34-positivity of the striated muscle in the present study cannot be considered as a convincing proof for the reversion of rhabdomyocytes to stem cells. Nevertheless, it may be viewed as an attempt by well-differentiated phenotypes to revert back to more immature phenotype. Moreover, rhabdomyocytes, in this study, were found to assume myofibroblast phenotype (discussed later in this article) by revealing the presence of α-SMA intermediate filament proteins (Figure 11). These observations support the plasticity of skeletal muscle during muscle injury (Li and Huard 2002) and more specifically as a consequence to biomaterial implantation. Therefore, on a biological point of view, the occurrence of progressively differentiating stem cells (satellite cells) and dedifferentiating rhabdomyocytes in the healing response elaborated by PET-induced muscle injury cannot be ignored. However, more convincing data are needed for delineating their role in modulating the nature of biocompatibility reaction.
In this study, many cells of inflammation were found PCNA-positive especially the macrophages, but there was considerable variation in the density and distribution of PCNA immunopositivity around the implant (Figure 6). The PCNA positivity of nuclei in giant cells was monitored against the current concepts of FBGC histogenesis. The current belief about FBGC formation is that monocyte-derived macrophages in the implant tissue undergo frustrated type of phagocytosis and these macrophages experience phenotypic change to become fusogenic. Several such fusogenic macrophages combine together to form large FBGC (Brodbeck and Anderson 2009; Anderson and McNally 2011). Hagerty et al. (2000) have shown that macrophages very near to the biomaterial implants are proliferatively active. As shown in Figure 6B and C, interestingly, when proliferating, all nuclei in a FBGC did not seem to have potential for proliferation at a given point of time. This observation indicates that the nuclei within a FBGC are composed of differential population: either some nuclei have lost the proliferative potential vis-à-vis some have gained/retained proliferative efficiency or that differential population represents nuclei from different macrophages. All these nuclei with variable proliferative efficiency could not have originated from a single monocyte/macrophage, and thus this observation is consistent with the current belief that FBGCs are formed by the fusion of macrophages (Anderson and McNally 2011).
The nature of immunoreactivity of FBGCs to vimentin, which is a marker for cells of mesenchymal origin (Figure 9C), and α-SMA, which is a marker for contractile phenotype-like smooth muscle cells (Figure 11C), clearly indicated that there are different subpopulations of FBGS which could be phenotypically defined by the presence of conventional intermediate filament protein markers. The relationship and the functional significance of various subpopulations (including the differentially PCNA-positive cells described above, Figure 6B and C) in modulating the biocompatibility reactions need more clarification.
Detailed study of the vimentin and α-SMA identified differentially altered skeletal muscle cells with these intermediate filament proteins. First, a subpopulation of the skeletal muscles near to implants was found to be expressing vimentin especially at the 1st week (Figure 9). Though skeletal muscle is of mesenchymal origin, the differentiated mature skeletal muscle normally does not express vimentin (Duquette et al. 2005; Leader et al. 1987; Tokuyasu, Maher, and Singer 1984), vimentin-positive skeletal muscles have been reported in some conditions of skeletal muscle regeneration and muscle damage following neuromuscular disorders (Bornemann and Schmalbruch 1993; Gallanti et al. 1992; Misra, Menon, and Mishra 1992). Second, α-SMA were also detected in the tissue reaction zone (Figure 12). α-SMA is the most widely used marker for myofibroblasts, a cell type that can originate from different cell types and very high significance in wound healing (Hinz et al. 2007). Cells other than smooth muscle cells expressing α-SMA are also considered to be the myofibroblast precursor cells and such cells are shown to be involved in the development of fibrosis with variable pathology (Desmouliere, Darby, and Gabbiani 2003; Hao et al. 2006; Thannickal et al. 2004; Zalewski, Shi, and Johnson 2002). Li and Huard (2002) reported that the myogenic precursor cells (skeletal myoblasts) can differentiate into myofibroblasts upon skeletal muscle injury and consequently contribute to the development of fibrosis and to the possibility of triggering differentiated skeletal muscle cells in to fibrotic lineage. They attributed these cells as the cause of desmoplastic reaction in skeletal muscle fibrosis following injury. We also found skeletal muscle cells expressing α-SMA immediately around the biomaterial implant (Figure 11) even though we could not detect any α-SMA immunopositive fibroblasts in the reaction zone. This indicates that skeletal muscle injury caused by the biomaterial implantation possibly induced transdifferentiation of skeletal muscle cells to myofibroblasts.
Myofibroblasts are also responsible for modulating wound contraction and synthesis of extracellular matrix components like collagen (Hinz 2007). Therefore, we quantified the extent of collagen deposition against the number of myofibroblasts. Fibrosis, as indicated by collagen deposition (Figure 5), increased with progressive healing. However, the skeletal muscle–derived myofibroblasts, though abundant at early time point of the healing process, decreased substantially during the progression of the healing reaction (Figures 10 and 12). The data suggested the activation of myofibroblasts as a consequence of the implant-induced reaction. The lack of persistence of myofibroblast in the PET-induced reaction suggested a favorable healing reaction with minimal contracture.
Conclusion
In this article, we present evidence based on immunohistochemistry for CD34, vimentin, and α-SMA that rhabdomyocytes are phenotypically altered during the healing response elicited by implanted PET in skeletal muscle. In view of the current knowledge of the cellular and molecular mechanisms involved in regulating the healing response and the revised definition of biocompatibility, it is desirable to identify novel molecular markers for evaluating biocompatibility reaction. Although a wide range of markers are possible, the data presented here point out a significant role for stem cells, differentially altered native cell types, FBGCs with varied proliferation ability in determining biocompatibility. Quantification of these cell types in relation to the extent of deposition of extracellular matrix components (e.g., collagen) implicates these cells as potential parameters for evaluating biocompatibility of a candidate biomaterial.
Footnotes
Acknowledgments
The authors acknowledge the Director, SCTIMST and Head BMT wing for their support and encouragements. Mr. Jaseer is a JRF of the ICMR and Ms. Deepa is a JRF of the CSIR. The authors also acknowledge Mr. N. K. Thulaseedharan, Laboratory for Experimental Pathology, SCTIMST for his technical assistance.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
