Abstract
Infusion site reactions are common following subcutaneous infusion of drugs. Such reactions can lead to discontinuation of the treatment. Therefore, assessment of such reactions is essential during preclinical safety studies, and magnetic resonance imaging (MRI) can assist in evaluation. Here, in vivo and ex vivo MRI evaluations were used in addition to classical histopathology to assess the infusion site reaction to ND0701, a novel formulation of apomorphine base developed for the treatment of Parkinson’s disease, in comparison to the commercial apomorphine hydrochloride (HCl) formulation. Both formulations, each at two concentrations, were continuously administered subcutaneously for 20 hr to each of 3 male and 3 female domestic pigs. Based on MRI evaluations, there was a gradual decrease in the volume of the subcutaneous lesions over 4 weeks, with smaller lesions and quicker resolution with ND0701 at concentrations 2.5- to 5-fold higher when compared to the commercial apomorphine HCl formulation. Histopathological evaluation of ND0701 revealed only minimal inflammation at the sites of infusion, whereas the commercial apomorphine HCl caused persistent inflammatory reactions and necrosis. This study provides support to the use of MRI in preclinical testing of subcutaneous drugs when evaluating local site reactions.
Keywords
Injection site reactions are a common and bothersome response to subcutaneous or intramuscular parenteral administration of many drugs. The incidence of such reactions can be 70% for some of the drugs (e.g., interferon β-1a) or even as high as 90% (e.g., anakinra; Gold et al. 2005; Haibel et al. 2005). Although these effects are commonly minor and disappear spontaneously with a few hours or days, they can sometimes be severe and lead to discontinuation of the drug (Gold et al. 2005). Therefore, proper assessment of local site reactions in preclinical animal toxicity studies is of utmost importance. Indeed, severe infusion site reactions can sometimes lead to termination of the drug development program as in the case of protiramer (Ramot et al. 2012; Varkony et al. 2009).
Apomorphine is a nonselective dopamine D1 and D2 receptor agonist, which is used to treat Parkinson’s disease (PD), usually in its later stages, when levodopa, the gold-standard treatment for PD, is no longer effective (Subramony 2006; Stacy and Silver 2008; Trosch, Silver, and Bottini 2008). Apomorphine is also highly effective in reversing the so-called OFF phenomenon, a state of decreased mobility, that accompany PD treatment (Gancher, Nutt, and Woodward 1991). There have also been sporadic reports of its possible beneficial effect on nonmotor symptoms such as liquid swallowing, drug-related neuropsychotic complications, restless leg syndrome, “OFF”-period pain, urinary dysfunction, and insomnia, commonly appearing in PD patients and affecting their quality of life (Todorova and Ray Chaudhuri 2013). Due to its very low oral bioavailability, only parenteral formulations of apomorphine hydrochloride (HCl) are currently available. Apomorphine HCl can be administered by intermittent subcutaneous injections, leading to quick relief from OFF periods (Bhidayasiri et al. 2015), or it can be administered as a continuous infusion, providing continuous dopaminergic stimulation that mimics the physiological conditions, thereby reducing motor complications that can be observed with intermittent applications (Trenkwalder et al. 2015). The most common motor complications, which are associated with the disease progression, include dyskinesia and motor fluctuations (e.g., sudden “OFF,” morning akinesia).
The most common adverse reactions with continuous apomorphine infusion are local, which can be observed in up to 87% of the patients using this administration technique (Garcia Ruiz et al. 2008). Infusion site reactions usually include temporary flushing or itching; however, the most common reaction is nodule formation (Henriksen 2014; Bhidayasiri, Boonpang, et al. 2016). The nodules can sometimes be accompanied by scarring and discoloration, can be tender, and might become infected, leading to the formation of abscesses and rarely necrotic ulcers, necessitating antibiotic treatment or surgical debridement (Bhidayasiri, Garcia Ruiz, and Henriksen 2016; Henriksen 2014). The formation of skin nodules and the complexity of operating the infusion pump are among the main reasons for drug discontinuation by patients (Garcia Ruiz et al. 2008; Manson, Turner, and Lees 2002). Large nodules can also affect drug absorption, resulting in lower efficacy, and might have psychological effects on the patient, being painful and unsightly (Acland et al. 1998).
ND0701 is a newly developed, concentrated formulation of apomorphine to be continuously administered subcutaneously via a discrete patch pump for the treatment of PD (Titova and Chaudhuri 2016). This new formulation was developed to provide a more compatible, tolerable, and safer, yet an equally efficacious alternative to the commercially available apomorphine HCl solution. To evaluate the safety, tolerability, and relative efficacy of ND0701, we performed a preclinical toxicokinetics study in domestic pigs. In this preclinical study, we used both in vivo and ex vivo magnetic resonance imaging (MRI), in addition to histopathology analyses, to test the ability of the MRI technique to provide information on local site reactions. Plasma concentrations of apomorphine were used as a surrogate marker for efficacy.
Animals, Material, and Method
Formulation
Commercial Apo-Go® (Britannia Pharmaceuticals, Berkshire, United Kingdom) was purchased and used as a reference apomorphine HCl.
Animal Husbandry and Treatment
This study was performed at Biotech-Farm, Israel, after the approval from the Committee for Ethical Conduct in the Care and Use of Laboratory Animals. Domestic pigs, n = 6 (Swine/Landrace × large White), 3 males and 3 females, weighting 45 ± 5 kg, were acclimatized for 1 week before experimentation. The animals were individually housed in indoor enclosures of approximately 230 cm × 105 cm floor area. The enclosures were cleaned daily. Drinking water was supplied ad libitum to each enclosure via an automatic “lixit” valve system. Each pig was offered a weighed amount of diet (Amir Dagan 150, Israel) daily, divided into two rations.
Prior to study initiation, animals were subjected to intravenous jugular cannulation under general anesthesia (xylazine/ketamine), with an indwelling catheter subsequently exteriorized and affixed to the skin. The back and flanks were shaved with care to avoid abrading the skin.
For pump application, animals were anesthetized by intravenous administration of Ketamine: Midazolam (2.5:0.25 mg/kg, respectively). The subcutaneous infusion was performed using an infusion pump (Crono-ND®, Canè Medical Technologies) connected to an infusion set (Accu-chek®, FlexLink, Roche) with a percutaneous soft cannula of 10-mm length. On each day of treatment, before the application of the pump, the skin of each treatment site was cleaned, disinfected with SeptiScrub and 70% ethanol, and wiped with sterile gauze. The infusion pump was placed in a protective pocket held in place by an elastic belt placed around the thorax just behind the scapulae of the animal. The infusion set was held at the injection site by means of adhesive tape. The infusion set was protected and held at the injection site by means of a tubular elastic bandage. Following pump and infusion set removal, a gauze pad with iodine solution was applied to the site of administration and secured with an adhesive tape.
For the evaluation of local site reactions, the drug formulations, 1% ND0701, 2.5% ND0701, 0.5% apomorphine HCl, or 1% apomorphine HCl (equivalent to 8.8, 25, 4.4, and 8.8 mg/ml apomorphine, respectively), were administered to each of the animals by continuous subcutaneous infusion of 44 or 50 mg apomorphine for 20 hr (Table 1, Online Figure S1). The 2.5% concentration was selected as the highest concentration based on several criteria, including formulation stability, biocompatibility, and clinical need. For the determination of plasma concentration of apomorphine, blood samples were collected via the indwelling jugular vein catheter at predetermined time points.
Rate, Dose, and Volume of Administration of Apo-Go and ND0701.
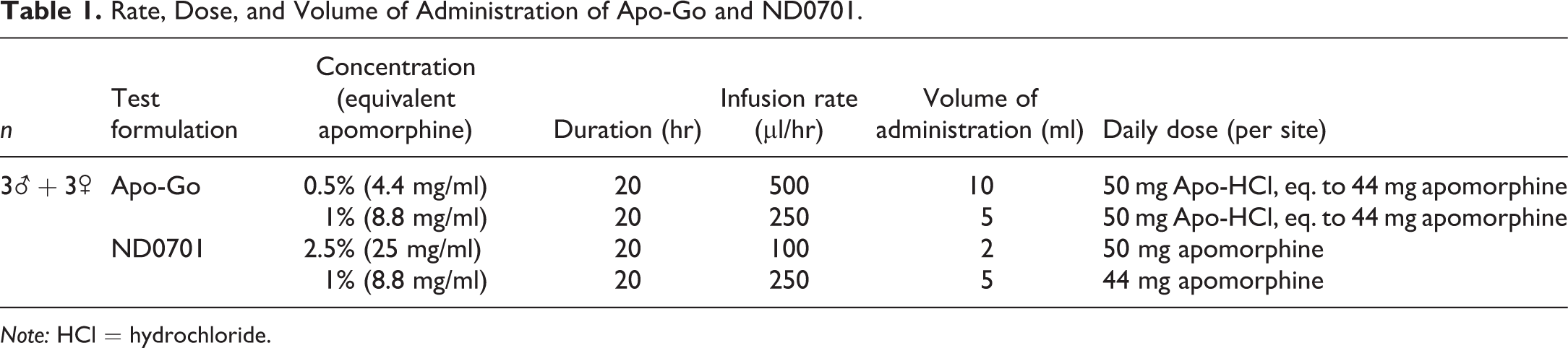
Note: HCl = hydrochloride.
Blood Sampling
Blood samples were collected into tubes containing K3EDTA via an indwelling jugular vein catheter. Samples were then transferred into 15-ml tubes containing 10 µl/ml of 0.5 M L-ascorbic acid. Plasma was transferred into Eppendorf tubes containing 10 µl/ml of 1% 2-mercaptoethanol and stored frozen at −20°C until analysis.
Quantification of Plasma Apomorphine
The analysis was performed by Analyst Research Laboratories, Israel. For sample preparation, 20 µl internal standard solution (250 ng/ml Boldine solution in 0.02% 2-mercaptoethanol (ME)/methanol), 200 µl of 1% ammonium hydroxide, and 3 ml methyl tert-butyl ether were added to 200 µl plasma. The supernatant organic layer was collected and evaporated to dryness. The residue was reconstituted with 200 µl of 0.2% formic acid and 0.1% 2-ME in water and then analyzed by LC-MS/MS, Waters 2795 using C18 100 × 2.1 mm. A 5-µl sample was injected at 0.2 ml/min.
Local Skin Reactions
Animals were examined for any local reactions at administration sites immediately following pump removal, once weekly thereafter and prior to collection of infusion sites (just before euthanasia). Reactions were scored according to a 5-point grading scale in ascending order of severity. Erythema and eschar formation were graded as follows: 0 = no erythema, 1 = very slight erythema (barely perceptible), 2 = well-defined erythema, 3 = moderate to marked erythema, and 4 = marked erythema (beet redness) to eschar formation preventing grading of erythema. Edema formation was graded as follows: 0 = no edema, 1 = very slight edema (barely perceptible), 2 = slight edema (edges of area well defined by definite raising), 3 = moderate edema (raised approximately 1 mm), and 4 = marked edema (raised more than 1 mm and extending beyond the area of exposure).
In Vivo MRI
In order to monitor lesion progression and/or recovery, MRI scans of the infusion sites were conducted on all 6 pigs, 2 and 4 weeks postdrug administration, using a 0.35-tesla Magnetom-C Siemens MRI machine (The Hebrew University Veterinary Hospital, Israel). MRI protocol sequence was transverse T2 (with fat saturation enabling the elimination of the fat tissue from the image): turbo spin echo sequence, repetition time (TR) = 5,850 ms, echo time (TE) = 68 ms, inversion time (TI) = 100 ms, resolution 0.63 × 0.63 × 5.5 mm, 4 averages. Analysis was performed by BioImage (Haifa, Israel). The local reaction to treatment was compared by quantifying the size and intensity of the abnormal tissue observed at the infusion sites. Volumetric analysis of each lesion was performed using an in-house software written by MATLAB 2011b. The signal intensity was demonstrated by 3-dimensional (3-D) surface plot. Statistical analysis was performed using a 2-tailed, 2 sample equal variance Student’s t test.
Collection of Infusion Sites
Four weeks postinfusion, animals were euthanized. Infusion sites were excised and fixed in 4% paraformaldehyde. After dehydration, as part of the routine tissue processing, tissue samples were embedded in paraffin wax; sections of the tissues were cut at 5 µm thickness and stained with hematoxylin and eosin by Patho-Labs diagnostics, Israel.
Ex Vivo MRI
MRI scans of fixed tissue samples (i.e., infusion sites) were performed by Harlan Biotech, Israel, from 2 pigs 4 weeks postdrug administration were obtained using a 1-tesla M2TM Aspect imaging MRI machine. MRI protocol sequence was TE/TR = 80/4,700 ms, field of view = 50 mm, slice thickness = 0.7 mm, matrix of 256 × 256, number of excitations = 16, in-plane resolution = 195 µm, total acquisition time = 33 min. Analysis was performed using Image Pro Plus 7 software, Media Cybernetics, Rockville, MD. The chosen area was presented in a 3-D representation according to its gray-level values distribution, ranging from 0 (black level) to 255 (white level) and pseudo color look up table given in Figure 3.
Histopathological Evaluation
Evaluation was based on grades given to tissue tolerance parameters (i.e., inflammation, necrosis, and fibrosis). The scoring of the lesions was done in a blinded manner, using a semiquantitative scoring system of 5 grades (0–4, grade 0 = no lesion, 1 = minimal change, 2 = mild change, 3 = moderate change, and 4 = marked change), taking into consideration the severity and extent of the changes, based on the criteria explained by Shackelford et al. (2002). The histopathology scoring of all slides was peer reviewed by a second senior pathologist.
Results
Local Skin Reactions
No external skin reactions were observed throughout the 4-week study period, except for small firm areas (<1 cm) that were palpated in the first 24 hr after pump removal in 15–30% of infusion sites from all 4 treatments (data not shown).
Plasma Concentration of Apomorphine
Blood samples were collected from the pigs (n = 6) during continuous subcutaneous administration of ND0701 and apomorphine HCl formulations, each at a total dose of 44 or 50 mg apomorphine (see Table 1) over a period of 20 hr. The pharmacokinetic (PK) profile of apomorphine following continuous subcutaneous administration was similar among all 4 tested formulations, exhibiting constant, steady state plasma concentrations (Figure 1).
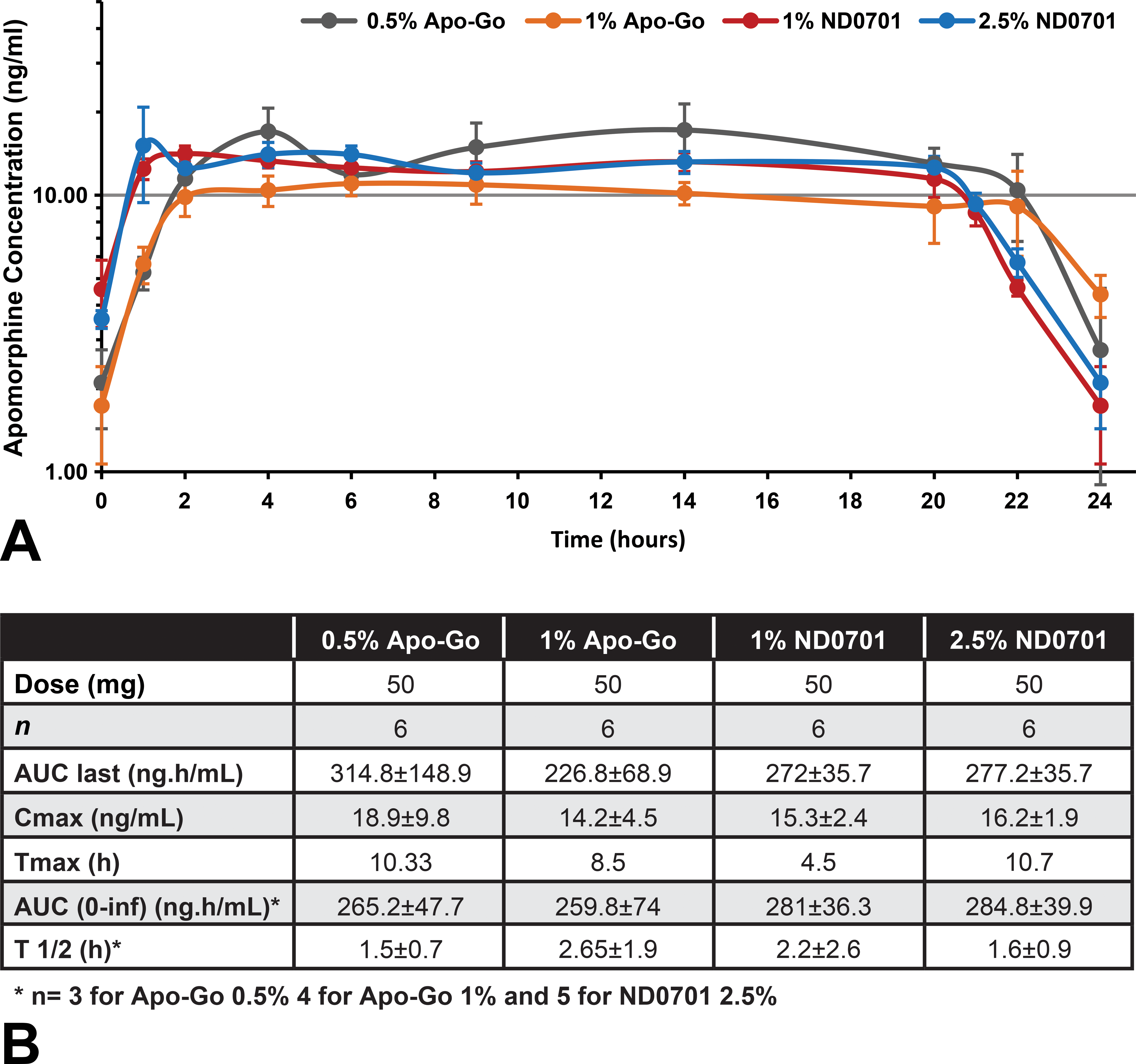
Plasma concentration of apomorphine following 20-hr subcutaneous infusion. On different occasions, pigs (n = 6) were continuously administered with 1% and 2.5% ND0701 and 0.5% and 1% apomorphine HCl formulations, for a period of 20 hr, corresponding to a total dose of 44 or 50 mg apomorphine (see Table 1). Plasma samples were collected for 24 hr from the start of infusion. (A) Mean apomorphine plasma concentration-time profiles, expressed in nanogram per milliliter. (B) Pharmacokinetic parameters described as mean (± standard deviation) values (median for T max).
Evaluation of Subcutaneous Lesions In Vivo Using MRI
The volume of the lesions was significantly smaller following administration of ND0701 2.5% and 1% as compared to apomorphine HCl 1% 2 weeks (789 and 492 vs. 1,672 mm3, p = .027 and p = .005, respectively) and 4 weeks (143 and 98 vs. 513 mm3, p = .004 and p = .0004, respectively) following a 20-hr infusion. When compared to 0.5% apomorphine HCl, a statistically significant difference was evident only 2 weeks following infusion when compared to 1% ND0701 (956 vs. 492 mm3, respectively, p = .03). The size of the lesions was not significantly different between the 2 concentrations of ND0701 or apomorphine HCl formulations. Notably, between week 2 and week 4, the size of the lesions was reduced by 5- to 5.5-fold and by 3- to 3.2-fold following administration of ND0701 formulations and apomorphine HCl formulations, respectively (Figure 2). Taken collectively, local site reactions following continuous subcutaneous administration of 25 mg/ml ND0701 were smaller and recovered faster than those administered 4.4 mg/ml apomorphine HCl formulations. These data suggest ND0701 is safe and well tolerated in the domestic pig.
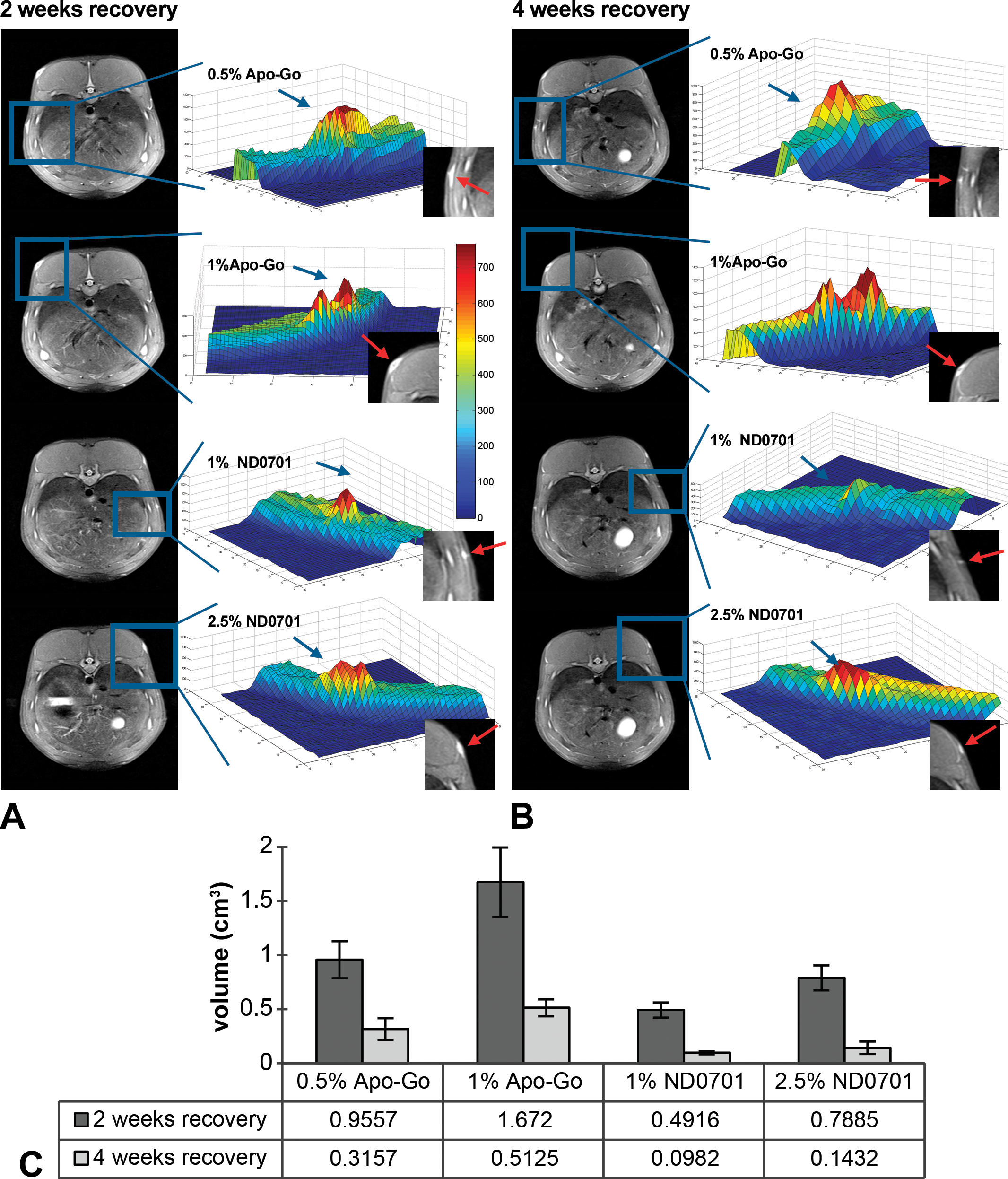
In vivo magnetic resonance imaging (MRI). Infusion sites were evaluated using a 0.35-tesla MRI machine. The intensity of the lesion was demonstrated in 3-dimensional representative images (A) 2 weeks and (B) 4 weeks following a single 20-hr infusion of apomorphine formulations: blue color indicates normal fat tissue and orange–red indicates affected tissue. (C) The mean volume (mm3) of subcutaneous lesions (n = 6) following a single 20-hr continuous SC infusion of the apomorphine formulations (corresponding to 44 or 50 mg apomorphine per infusion site; see Table 1).
Ex Vivo Evaluation of Subcutaneous Lesions Using MRI
The mean lesion volume at the injected site of 1% apomorphine HCl 4 weeks postinfusion was 0.314 cm3. In contrast, the mean lesion volume at the injected site of 1% ND0701 was 0.057 cm3, 5 times smaller (Figure 3). The volume of the lesion following administration of the most concentrated formulation, 2.5% ND0701, was 0.061 cm3, 2 times smaller than the volume of the lesion in the lowest concentration of apomorphine HCl, 0.5%, 0.133 cm3.
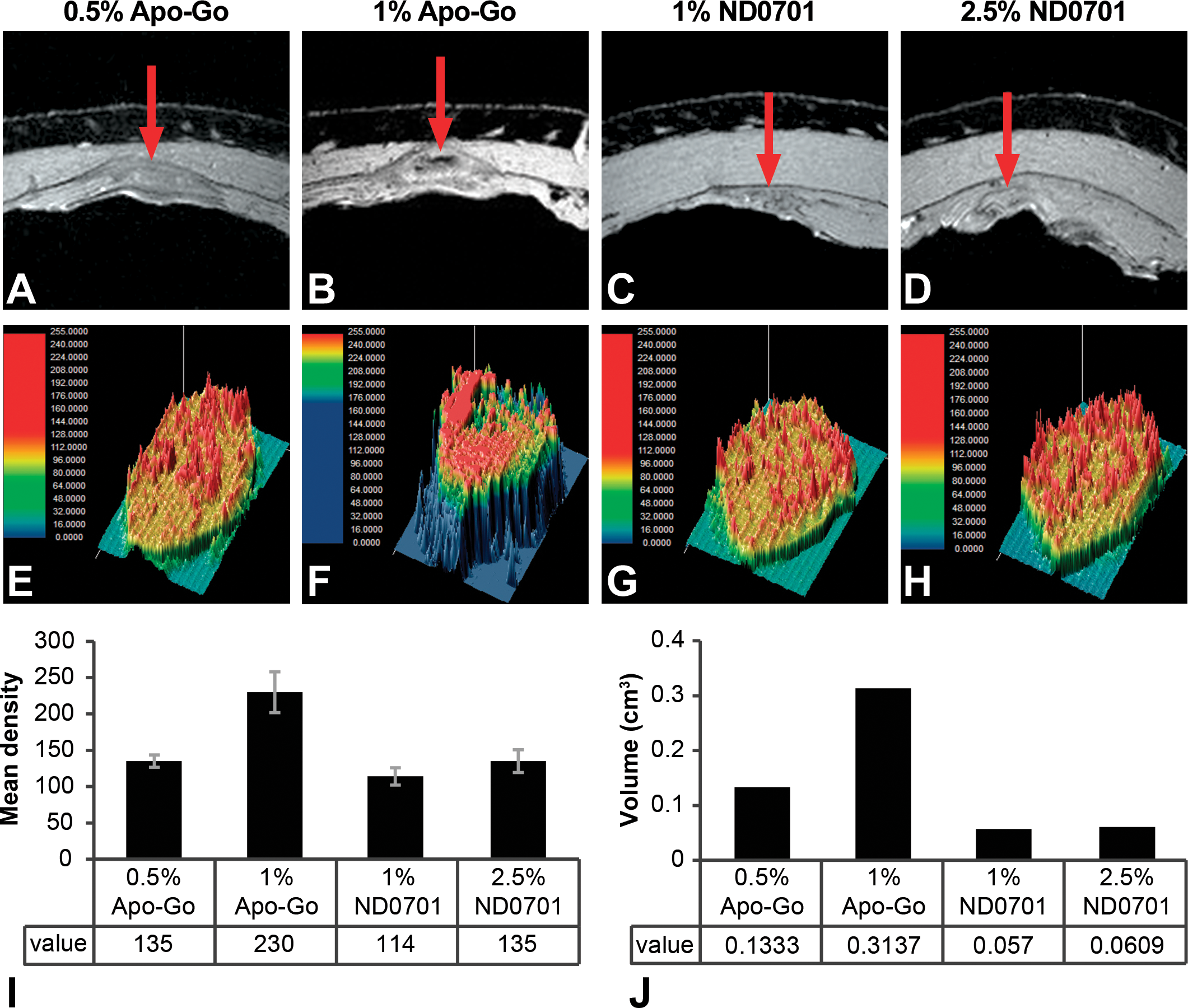
Ex vivo magnetic resonance imaging (MRI) evaluation. Tissue biopsies from infused sites were evaluated using M2TM Aspect imaging 1-tesla MRI 4 weeks following a single 20-hr continuous subcutaneous infusion of apomorphine formulations, corresponding to 44 or 50 mg apomorphine per infusion site (A–D, see Table 1). Morphometric evaluation of the MRI scans was performed (E–H) showing (I) inflammation and fibrosis (represented by the yellow and red colors) and (J) mean intensity and mean volume of the lesion (n = 2). Representative images. Red arrows indicate the areas of inflammation and fibrosis identified by MRI.
Histopathological Evaluation of the Injection Sites
Histopathological evaluation of administration sites was conducted 4 weeks following a single 20-hr infusion. Infusion sites administered apomorphine HCl, 0.5 and 1%, exhibited mild to moderate degrees (grade 2 and 3), respectively, chronic, granulomatous inflammation in the subcutis, characterized by the presence of macrophages, multinucleated giant cells, lymphocytes, fibroblasts, and collagen deposition (Figure 4). The tissue exposed to apomorphine HCl, 1%, also exhibited irregular areas of hemorrhage associated with necrosis and therefore was overall graded as moderate degree. Necrotic material was surrounded by a granulomatous inflammatory reaction (Figure 4). In contrast, the subcutaneous tissue in the ND0701-infused sites exhibited minimal, chronic inflammation characterized by macrophages, lymphocytes, fibroblasts, and collagen deposition, with no multinucleated giant cells. The inflammatory cell components were comparable among sites administered 1% or 2.5% ND0701. However, the grade of inflammation (reflecting the intensity and extension of the response) was somewhat higher following the administration of 2.5% ND0701, versus 1% ND0701, presenting minimal (grade 1) versus no inflammation to minimal (grade 0–1) quantity of lymphoctic cell component, respectively (Figure 4).
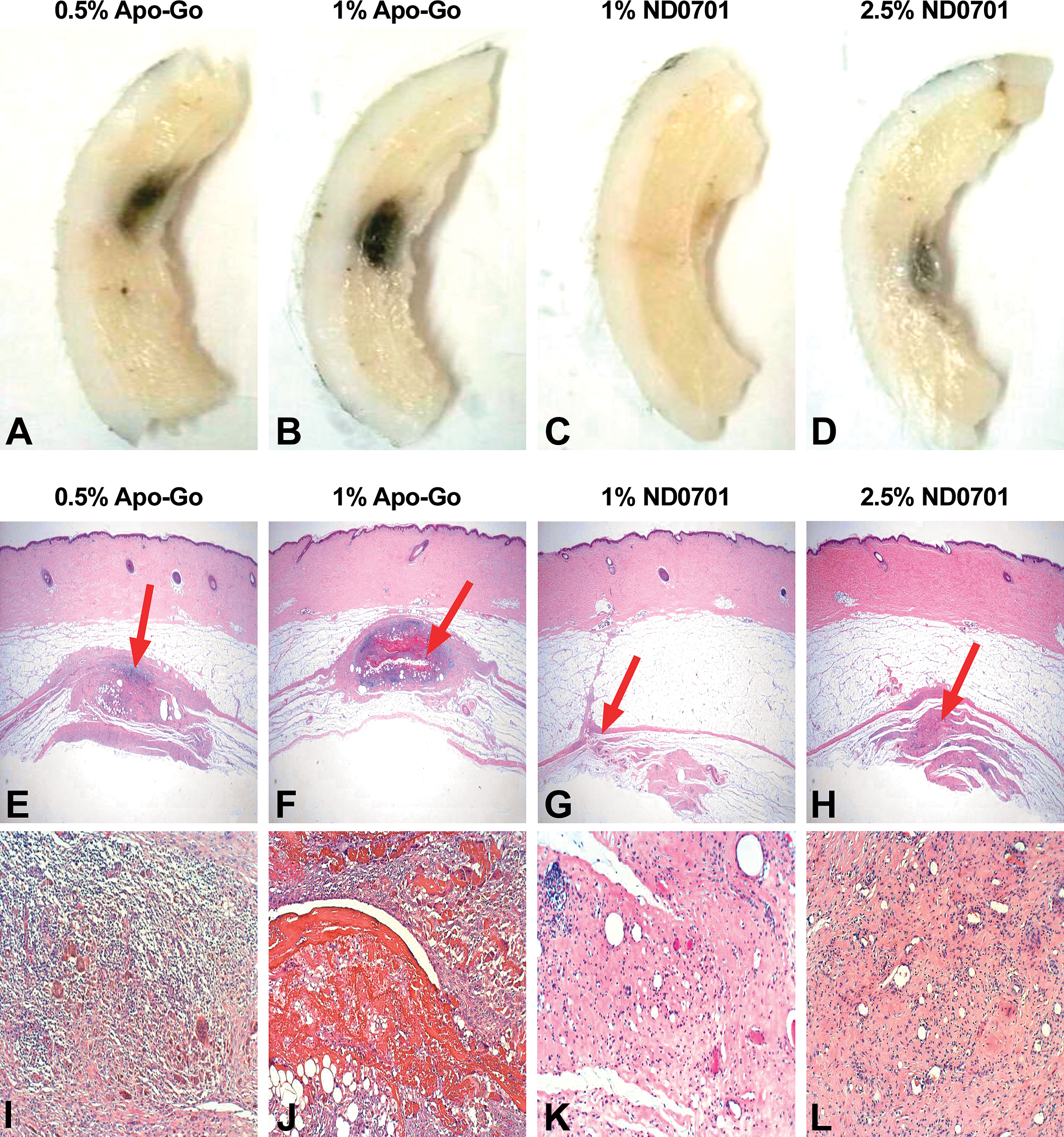
Macroscopic and microscopic evaluation of infusion sites (representative images). Macroscopic (A–D) and microscopic (E–L) evaluation of the subcutaneous tissue was performed 4 weeks post 20-hr continuous subcutaneous administration of the test formulations, corresponding to 44 or 50 mg apomorphine per site (see Table 1). In the macroscopic photos, infusion sites appear darker in color. In the microscopic photos, photos E–H present low magnification view, and photos I–L show high magnification view. Red arrows in photos E–F indicate the site of the corresponding higher magnification areas shown in photos I–L. Photos E and I demonstrate the prominent granulomatous reaction; photos F and J demonstrate the significant necrosis and cavity formation, which are surrounded by a granulomatous inflammatory reaction; in photo J, the red areas represent hemorrhage associated with necrosis; and photos G, K, H, and L demonstrate the relative smaller size of the infusion site reaction, consisting of minor inflammatory reaction and prominent fibrosis.
Discussion
The aim of the present study was to assess the local skin reactions, tolerability, and the PK of a single 20-hr continuous subcutaneous infusion of 1% and 2.5% ND0701 compared to the clinically available 0.5% and 1% apomorphine HCl formulations. Histopathological evaluation revealed that 4 weeks following administration of ND0701 formulations, minimal and transient inflammation was present at the sites of infusion. In contrast, apomorphine HCl caused persistent mild to moderate inflammation and necrosis 4 weeks after apomorphine HCl administration. According to in vivo and ex vivo MRI analyses of lesions size, recovery of local site reactions was nearly complete by 4 weeks following ND0701 infusion. More limited recovery was observed following apomorphine HCl administration. ND0701 provides a similar PK profile to apomorphine, suggesting that an equivalent efficacy can be achieved in patients. Overall, this study shows that continuous subcutaneous administration of ND0701, even at a concentration as high as 2.5%, is safe and tolerable in the domestic pig, a well-established animal model. Furthermore, based on MRI and histopathology data, the extent and severity of local reactions due to ND0701 are considerably lower as compared to reactions observed following administration of apomorphine HCl.
Currently, the commercially available drug product (Apo-Go®) is provided at 5 and 10 mg/ml apomorphine HCl, which corresponds to 4.4 and 8.8 mg/ml apomorphine. The daily recommended dose is not more than 100 mg, and the daily recommended duration of infusion is not more than 20 hr, up to 12-hr infusion to one site. ND0701 1% contains 8.8 mg/ml apomorphine and ND0701 2.5% contains 25 mg/ml apomorphine. The use of the concentrated 2.5% ND0701 formulation provided for a substantial decrease in the volume administered and therefore, only a single infusion site was necessary. The reduced volumes of injection were due to development of a novel formulation consisting of a more soluble form of apomorphine and glutamic acid, which is more stable in vitro and more compatible in vivo.
Although still not good laboratory practice–compliant, imaging techniques are gaining popularity during drug development processes. Such techniques include radiography and ultrasound and more sophisticated modalities such as microcomputed tomography, micropositron emission tomography, single-photon emission computed tomography, and in vivo optical imaging (Ettlin 2013; Ying and Monticello 2006). MRI is one of the modalities that is especially promising for toxicity studies, thanks to its ability to provide highly detailed information on pathologic changes in soft tissues in addition to quantitative 3-D data (Dixon et al. 1988). It also allows the performance of magnetic resonance histology, which is the use of MRI for high resolution characterization of tissue structure performed on formalin-fixed tissues (Johnson et al. 1993). The advent of compact MRI systems has significantly facilitated its use in toxicology and preclinical studies (Tempel-Brami et al. 2015). Indeed, MRI has been utilized for several toxicology studies, including neurotoxicity and renal toxicity, evaluation of the safety of stem cells, pulmonary fibrosis, and in cancer models (Ramot et al. 2017; Tempel-Brami et al. 2015).
MRI has been only sparsely used in the past to assess local infusion reactions in animals. It has been used to evaluate local reactions to vaccines in pigs and was also used to evaluate antigen clearance following vaccination in mice (Bernau et al. 2015, 2016; Brewer et al. 2014). MRI was also used for the assessment of biodegradation of polymers implanted subcutaneously in pigs (Nyska et al. 2014). However, to the best of our knowledge, this is the first noninvasive, in vivo study presenting quantitative measurements that correlate with qualitative histopathological findings and provide means for the monitoring of the evolution of subcutaneous lesions following subcutaneous administration of pharmaceuticals.
In our study, we were able to detect and quantify the local reaction to the infused drugs using MRI as well as to follow the resolution of the skin reaction individually for each animal. This methodology thus eliminates the need for interim sacrifice of animals and therefore decreases the number of animals needed for each study. This study also shows that by quantifying the tissue reaction in vivo, it is possible to objectively compare between different treatment modalities when assessing local site reactions.
MRI data correlated well with histopathological evaluation, which is still considered to be the gold standard. Accordingly, the volume of the lesions following administration of the most concentrated formulation of ND0701 was significantly smaller than the volume of the lesions seen in the lowest concentration of apomorphine HCl, in connection with the corresponding reduced severity of the histopathologic changes. It is obvious that imaging in general, and MRI in particular, cannot replace the classical pathological evaluation which provides important information on the pathomorphology of the lesions. Nevertheless, in addition to reducing the number of examined animals, it can assist in directing the pathologist to the relevant examination locations in the affected tissue.
In conclusion, based on both MRI and histopathological analyses, continuous subcutaneous administration of ND0701 is generally more advantageous in pigs, when compared to a commercially available apomorphine HCl formulation. We demonstrated the ease of use of compact MRI histology equipment to obtain diagnostic quality ex vivo MRI images of experimental tissue samples, thereby greatly enhancing conventional preclinical studies (Ramot et al. 2017).
We also show that there is a high level of correlation between MRI and histopathological findings, thereby giving further support to the use of MRI as a tool when screening and assessing the local safety and tolerability of subcutaneous investigational drugs in preclinical studies.
Footnotes
Authors’ Note
Ronit Shaltiel-Karyo, Yonit Tsarfati, Anna Rubinski, Eduardo Zawoznik, Irena Weinstock, Mara Nemas, and Oron Yacoby-Zeevi are employees of NeuroDerm Ltd.
Acknowledgment
We thank our colleagues at NeuroDerm, Ltd., for fruitful discussions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by NeuroDerm Ltd.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
