Abstract
Glial cell line-derived neurotrophic factor (GDNF) is a potent neuroprotective biologic in Parkinson's disease models. Adeno-associated viral vector serotype 2 (AAV2)-human GDNF safety was assessed in rats treated with a single intracerebral dose of vehicle, 6.8 × 108, 6.8 × 109, or 5.2 × 1010 vector genomes (vg)/dose followed by interim sacrifices on day 7, 31, 90, and 376. There were no treatment-related effects observed on food consumption, body weight, hematology, clinical chemistry, coagulation parameters, neurobehavioral parameters, organ weights, or serum GDNF and anti-GDNF antibody levels. Increased serum anti-AAV2 neutralizing antibody titers were observed in the 5.2 × 1010 vg/dose group. Histopathological lesions were observed at the injection site in the 6.8 × 109 vg/dose (day 7) and 5.2 × 1010 vg/dose groups (days 7 and 31) and consisted of gliosis, mononuclear perivascular cuffing, intranuclear inclusion bodies, and/or apoptosis on day 7 and mononuclear perivascular cuffing on day 31. GDNF immunostaining was observed in the injection site in all dose groups through day 376 indicating no detectable impacts of anti-AAV2 neutralizing antibody. There was no evidence of increased expression of calcitonin gene-related peptide or Swann cell hyperplasia in the cervical and lumbar spinal cord or medulla oblongata at the 5.2 × 1010 vg/dose level indicating lack of hyperplastic effects. In conclusion, no systemic toxicity was observed, and the local toxicity observed at the injection site appeared to be reversible demonstrating a promising safety profile of intracerebral AAV2-GDNF delivery. Furthermore, an intracerebral dose of 6.8 × 108 AAV2-GDNF vg/dose was considered to be a no observed adverse effect level in rats.
Introduction
Parkinson's disease (PD) is a progressive nervous system disorder that involves degeneration of dopaminergic neurons of the substantia nigra (SN) pars compacta in the midbrain. Progressive loss of nigral dopaminergic neurons (the pathological hallmark of PD) results in progressive neurologic dysfunction and death. There is an unmet need for better treatments for the disease since current therapies can temporarily alleviate the symptoms of PD, but they do not improve the degenerative process. It has been demonstrated that neurotrophic factors can promote the growth, survival, maintenance, and differentiation of both developing and mature neurons in the central nervous system (CNS). Glial cell line-derived neurotrophic factor (GDNF) is a potent trophic factor that plays an important role in the postnatal survival of mesencephalic dopamine neurons. 1 -3 Rodent and nonhuman primate (NHP) models have demonstrated that direct infusion of recombinant GDNF protein or virus-mediated GDNF gene elevation in the SN or striatum results in the protection of dopaminergic neurons in neurotoxicant models. 4 -8 However, clinical studies of putaminal infusion of recombinant GDNF for the treatment of PD have failed to demonstrate clinical benefit in randomized controlled studies. 9 -11 Poor or inappropriate distribution of the infused neurotrophic factor within the target region could have been a possible explanation for discordant results within patient cohorts and across clinical trials. Nevertheless, enthusiasm for the development of GDNF-based therapies for PD is still high because of its demonstrated ability to restore dopaminergic activity and reverse motor deficits in animal models and anecdotal evidence from clinical study participants. 12 Recent histological data from patients with PD who previously participated in gene therapy studies of neurturin (a less potent homolog of GDNF) further demonstrated the ability of trophic factors to induce regrowth of dopamine neurons. However, restored tyrosine hydroxylase expression was associated with the region of neurturin expression, which represented only 3% to 18% of the targeted putamen. 13 Recent clinical optimization of magnetic resonance imaging (MRI)-monitored convection enhanced delivery (CED) in patients with PD now allow for safe delivery of larger infusion volumes which are required to reliably target large brain structures like the putamen. 14
Gene therapy is used to introduce genetic material into target cells using viral or nonviral vectors to intervene in disease progression by correcting or supplementing defective genes. Therapeutic effects of gene therapy generally last longer without a need for repeated interventions. The adeno-associated virus (AAV) vector system is a valuable gene delivery tool for treating a variety of diseases with little, if any, pathogenic effect in humans. The AAV vectors are well-established vectors in clinical trials for in-vivo gene therapy due to their unique attributes including broad tropism, low immunogenicity, ease of production, rare integration into the host chromosome, and result in long-term expression of transgene. The adeno-associated virus has been used to treat cardiovascular, muscular, neurological, hematological, ophthalmological diseases, and infectious disorders as well as cancer. 15 The AAV2 serotype’s origin of isolation is human with tissues of tropism in liver, CNS, and muscle. 15 When administered directly into the brain, AAV2 selectively transduces neurons present at the site of injection without any detectable transduction of cells in adjacent areas of the brain. 16 This localized biodistribution, and the selective transduction of nonantigen-presenting neurons, make AAV2 vectors ideally suited for CNS gene therapy applications where restricted distribution of the transgene is required. One exception to the localized CNS transduction is anterograde neuronal transport of AAV2 vectors with subsequent trans-synaptic transduction of secondary neurons. 17,18 For example, the transduction of cortical neurons after AAV2 delivery to the thalamus or SN pars reticulata neurons after striatal injection.
AAV2-GDNF is a nonreplicating recombinant adeno-associated viral vector serotype 2 (AAV2) containing a DNA construct encoding human GDNF and is under development for the treatment of PD. The GDNF gene therapy may alter the disease process directly by inducing the expression of a specific protein that slows dopaminergic neuronal loss. AAV2-GDNF mediated gene transfer has been used in non-clinical studies to deliver GDNF to the nigrostriatal system as a means by which constitutive GDNF production can be achieved. 19 These studies in rodent 20 and NHP models of PD 19,21 have demonstrated functional recovery of motor deficits and enhanced dopamine production as a result of the elevated GDNF expression levels in the basal ganglia. The current Investigational New Drug (IND)-enabling Good Laboratory Practice (GLP) toxicology study was designed to provide key support for the clinical development of AAV2-GDNF. Hovland and colleagues previously reported a study of recombinant GDNF delivery in NHPs showed cerebellar lesions characterized by Purkinje cell loss, molecular layer atrophy, and granule cell loss, 22 potentially due to reflux of the infused GDNF solution resulting in elevated levels of GDNF protein within the cerebrospinal fluid (CSF). The objective of the current study was to determine the target organ toxicity of AAV2-GDNF in rats administered a single dose of AAV2-GDNF via bilateral CED to the striatum followed by a 12-month observation period. Adult, naive, Sprague Dawley rats were selected for this study. Prior non-clinical studies have demonstrated that the human GDNF protein is biologically active across multiple species, and adverse immune responses following expression of the human GDNF protein in rats have not been observed. 20,23 An extensive series of assessments was conducted of in-life behavioral and clinical parameters, serum and CSF levels of GDNF protein, GDNF antibody and AAV2 neutralizing antibody (nAb), organ histopathology, and peripheral and CNS immune/inflammatory reaction to AAV2 and/or GDNF protein. The results of this study support the clinical potential of this gene therapy approach with a safe path forward to clinical studies that are currently being conducted with MRI-monitored CED of AAV2-GDNF in patients with PD. 24
Materials and Methods
Test System
Rats (Sprague Dawley, 80 male and 80 female) were received from Charles River Laboratories and were quarantined for 7 days to observe for general health and acceptability for use in this study. On day 1, the rats were 9 to 12 weeks and weighed between 262 and 380 g (males) and 206 and 273 g (females). Rats were individually housed in solid-bottom plastic cages on stainless steel racks in an environmentally monitored, well-ventilated room that was maintained at a temperature of 66 °F to 79 °F and a relative humidity of 24% to 71% during the prestudy and study periods. Room lights were controlled by an automatic timer set to provide illumination approximately 12 hours per day.
Certified rodent diet 2016C (Harlan) was provided ad libitum. Tap water (public water supply) was available ad libitum via automatic watering system. Animal housing and care conformed to the guidelines of the Guide for the Care and Use of Laboratory Animals, 25 the US Department of Agriculture through the Animal Welfare Act (Public Law 99-198), and to the applicable standard operating procedures. The study was conducted in compliance with the US Food and Drug Administration GLP Regulations (Code of Federal Regulations Title 21 Part 58).
Test Article and Vehicle/Control Article
Test article
The AAV2-GDNF vector consists of serotype 2 adenoassociated viral particles, containing single stranded DNA encoding human GDNF (hGDNF) complementary DNA, human cytomegalovirus promoter, and 3′-untranslated region sequences. The DNA is flanked by AAV2-derived internal repeat sequences. The AAV2-hGDNF vector (AAV2-GDNF, NSC-D751136) was manufactured by the Children’s Hospital of Philadelphia (CHOP) clinical gene therapy core facility. The vector was generated by transient transfection of HEK293 cells in roller bottles, purified by combined column chromatography and cesium chloride gradient ultracentrifugation, resulting in empty capsid-free material, and tested for safety, purity, and concentration (titer) measured by quantitative polymerase chain reaction (qPCR) that was determined as 5.2 × 1012 vector genomes (vg)/mL. The vector was stored at < −60 °C. During this study, the stability of the stock vector was assessed by qPCR titer determination of samples of the vector that were obtained during the period of use of the vector on this study.
Vehicle/control article
Vehicle vials containing 4.5 mL each of 180 mM sodium chloride, 10 mM sodium phosphate, and 0.001% Pluronic F68, pH 7.3, were supplied by CHOP and were stored at approximately −10 °C.
Dose formulation preparation
On each day of dosing, a fresh vial of stock AAV2-GDNF (5.2 × 1012 vg/mL) was thawed and used for preparation of the dose formulations. Dilutions of the stock were made in vehicle to the required vector concentrations for groups 2 and 3. Undiluted stock AAV2-GDNF (5.2 × 1012 vg/mL) was administered to animals in group 4.
Dose formulation concentration analysis
On each day of dose formulation preparation, 1 sample (50 µL) from the vial of stock vector and a sample (50 µL) from the top, middle, and bottom of each vector dilution were collected for vector genome concentration analysis.
Experimental Design
Randomization and group assignment
After randomization, 160 rats (equal number of males and females) were assigned to 3 test article dose groups, or to a vehicle control group, as described in Table 1.
The Study Design.

Abbreviations: AAV2, adenoassociated viral vector serotype 2; F, female; GDNF, glial cell line-derived neurotrophic factor; M, male.
a Split between 2 hemispheres with 5 µL dosing volume per hemisphere.
b Calculated from the actual dilution scheme of the vector and a stock AAV2-GDNF titer of 5.2 × 1012 vg/mL.
Dose procedure
Prior to dosing, each animal was anesthetized through isoflurane inhalation. The appropriate area of the skull was clipped free of hair and surgically prepared. Artificial tears ointment was placed on each animal’s eyes prior to surgery. Each animal was mounted in a stereotaxic frame.
A longitudinal incision was made in the skin on top of the skull, and blunt dissection was used to remove connective tissue overlying the skull. A small dental drill was used to make 2 burr holes in the skull 0.5 mm anterior and 3 mm left and right from bregma for the infusions in the left and right striatum. For the bilateral infusion, 2 cannulae were connected to 100-µL Nanofil-100 syringes (World Precision Instruments) connected to an automated pump and lowered to the appropriate coordinates relative to bregma (AP, anteroposterior = +0.5 mm; ML, mediolateral = ±3 mm; DV, dorsoventral = 5 mm with tooth bar at −3.3 mm). The infusion rate was set at 0.5 μL/min with a total infusion of 5 μL/hemisphere. Following infusion, the cannulae were left in place for approximately 2 minutes to minimize back diffusion and then slowly withdrawn. The burr holes were filled with bone wax and the incision closed using wound clips and/or tissue glue.
Buprenorphine was administered to each animal subcutaneously (∼0.05 mg/kg/dose) twice a day for 3 days, except as noted, with the first dose administered prior to surgery.
The total doses (vehicle, 6.8 × 108, 6.8 × 109, or 5.2 × 1010 vg) were split between 2 hemispheres with 5 µL dosing volume per hemisphere. Intracerebral route of administration was chosen to be consistent with intended clinical route of administration. It has been shown in the rat study that striatal delivery of AAV2-GDNF is the preferred method for trophic rescue of dopaminergic neuronal function. AAV2 is transported throughout the nigrostriatum by anterograde transport that directs GDNF expression throughout the basal ganglia in regions that are adversely affected in Parkinson's disease in addition to SN compacta. 18
Clinical observations
All rats were observed twice daily throughout the prestudy and study periods for signs of moribundity and mortality. Cageside examinations of each rat were performed twice daily through day 30 and then once daily through the end of the study or more often as clinical signs warranted. Detailed observations of each rat (cage removal) were performed once weekly or more often as clinical signs warranted.
Food consumption
Individual food consumption was recorded weekly and prior to unscheduled euthanasia.
Body weights
All rats were weighed during the prestudy period for randomization, day 1 (prior to dosing), and then weekly and prior to euthanasia.
Functional observational battery
A standardized battery (including hypersensitivity to pain and handling) of neurobehavioral evaluations to assess central/peripheral nervous system status was conducted on all rats at baseline and on the day prior to scheduled necropsy. For all animals euthanized moribund, functional observational battery (FOB) evaluations were conducted on the day of euthanasia. The FOB evaluations included the following: home cage measurements, handling measurements, open-field measurements, and stimulus reactivity measurements.
Clinical pathology
Blood for clinical pathology, GDNF levels, anti-AAV2 antibody, and anti-GDNF antibody determinations were collected from the retro-orbital plexus in each dose group at baseline and on the day of scheduled or moribund euthanasia. Blood was collected into a tube containing EDTA (approximately 0.5 mL; hematology), no anticoagulant (approximately 0.5 mL at baseline collections and 1 mL on other days; clinical chemistry), sodium citrate (approximately 1.8 mL; coagulation evaluations), or no anticoagulant (approximately 0.5 mL at baseline collections and 1 mL on other days; GDNF levels and antibody determinations).
Serum obtained after centrifugation of blood samples collected on days other than baseline for subsequent GDNF and antibody determinations were aliquoted into 3 samples. All samples collected on each day of necropsy for GDNF and antibody determinations were stored at or below −70 °C prior to analysis. All samples were processed and analyzed for white blood cells, red blood cells, hemoglobin, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, platelets, reticulocytes, neutrophil, lymphocyte, monocyte, eosinophil, basophil, and nucleated RBC counts.
Clinical chemistry parameters included urea nitrogen, serum aspartate aminotransferase, serum alanine aminotransferase, alkaline phosphatase, γ-glutamyl transferase, glucose, creatinine, total protein, albumin, globulin, cholesterol, sodium, potassium, chloride, prothrombin time, activated partial thromboplastin time, and fibrinogen.
Serum GDNF Levels and Immunogenicity
Serum GDNF levels were measured using a commercial ELISA kit (Promega). Antibodies against GDNF in serum were determined using inhibition enzyme-linked immunosorbent assay (ELISA) assay based on the DuoSet ELISA Development kit (DY212) from R&D system. Amount of anti-AAV2 nAb in rat serum samples was assayed for their ability to neutralize recombinant AAV serotype 2 vector expressing eGFP transfection of 293A cells, genetically modified 293 cells.
Macroscopic and microscopic pathology
Scheduled necropsies included 5 rats/sex from groups 1 to 4 on day 7, 31, and 90; 2 female rats from group 4 on day 144; and the remaining surviving rats on day 376. Two female rats showing clinical signs of head tilt from group 4 were subjected to early necropsy on day 144 instead of their scheduled necropsy on day 376 to investigate plausible cause of the head tilt. Unscheduled necropsies occurred on day 175 (1 male rat in the 5.2 × 1010 vg/dose group; moribund euthanasia); day 209 (1 male rat in the 5.2 × 1010 vg/dose group; moribund euthanasia); day 276 (1 male rat in the 5.2 × 1010 vg/dose group; moribund euthanasia); day 313 (1 male rat in the vehicle control group; moribund euthanasia); and day 364 (1 male rat in the 6.8 × 109 vg/dose group; found dead).
Macroscopic observations
All rats were subjected to a complete gross necropsy examination. The postmortem examination included, but was not limited to, examination of the external surfaces of the body, all orifices of the body, and the cranial, thoracic, abdominal, and pelvic cavities and their contents. All of the tissues/organs in the following list were collected from each rat. Additionally, the skull with the inner ear was collected from the 2 female rats euthanized on day 144. All tissues/organs collected at necropsy were fixed in 10% neutral buffered formalin, with the following exceptions: eyes were fixed in Davidson’s solution; testes/epididymides were fixed in modified Davidson’s solution; and brain and spinal cord were fixed in 4% paraformaldehyde. The tissues collected included all gross lesions, adrenal gland, aorta, bone, bone marrow, femur, brain, eye, heart, kidney, intestine, liver, lung, lymph node, pancreas, parathyroid/thyroid, pituitary, salivary gland, sciatic nerve, skeletal muscle, spleen, stomach, testis, thymus, trachea, uterus, urinary bladder, and vagina.
Organ weights
The following organs, gender appropriate, were weighed at scheduled necropsy. Paired organs were weighed together. Organ-to-body weight ratios and organ-to-brain weight ratios were calculated for adrenal glands (postfixation), brain, heart, ovaries, spleen, and testes/epididymis.
Histology
All tissues listed above (in macroscopic observation section) from rats in groups 1 (vehicle control) and 4 (high dose) that were euthanized during the study, and target tissue (brain) from animals in groups 2 and 3, were processed for histopathological examination. Sections of each tissue were trimmed and subsequently processed to slides. For hematoxylin and eosin (H&E) staining, the tissues were microtomed as approximately 5-µm thick sections and for immunohistochemistry, the tissues were microtomed as approximately 3-µm thick sections.
For brain, the right half was sectioned in a sagittal plane from the midline to the lateral edge of the brain; the left half of the brain was sectioned in a coronal plane. Coronal sections of the left half of the brain were placed in 2 blocks. Block one included the following sites: site 1—cerebral cortex and ganglia; site 2—cerebral cortex, hippocampus, and mesencephalon; and site 3—medulla oblongata and cerebellum. The remainder of the coronal sections of the brain was placed in block 2. In addition, the skull collected from the 2 female rats in group 4 euthanized on day 144 to investigate plausible cause of head tilt was decalcified. Sections (approximately 3-µm for immunohistochemical staining and 5-µm for H&E staining) of the skull in the region of the inner ear were prepared.
Microscopic observations
Slides of all tissues that were stained with H&E or a specialized diagnostic stain(s) from rats in groups 1 (vehicle control) and 4 (high dose) were evaluated. In addition, slides of target tissue (brain) from animals in groups 2 and 3 euthanized on day 7 or 31 were also evaluated.
Immunohistochemical staining and analysis for GDNF and calcitonin-gene-related peptide (CGRP) expression were performed on sections of the brain and/or spinal cord collected from animals in all dose groups. Immunostaining was performed on the cerebrum, cerebellum, pons, medulla oblongata, and brain injection site of animals in the vehicle control, 6.8 × 108, 6.8 × 109, or 5.2 × 1010 vg/dose group euthanized between day 7 and 376. The CGRP immunostaining was performed on the cerebrum, cerebellum, pons, medulla oblongata, brain injection site, cervical spinal cord, and lumbar spinal cord of animals in the vehicle control or 5.2 × 1010 vg/dose group euthanized between day 7 and day 376.
Statistical analysis
Group means and standard deviations were calculated when appropriate for body weights, food consumption, clinical pathology parameters, absolute, and relative organ weight data. Evaluation of body weight, clinical pathology parameters, absolute organ weights, and relative organ weight data for the differences between groups utilized analysis of variance and Dunnett test for multiple comparisons. All statistical analyses were performed using the Provantis automated data collection system (Instem), and the lower limit for statistical significance was defined as P ≤ 0.05.
Results
Dose Formulation Analyses
The measured concentration of vector genomes indicated the animals received the intended dose of the test article.
Mortality
Mortality or unscheduled euthanasia occurred for 5 male animals; all other animals on study survived until scheduled euthanasia. One male rat in the vehicle control group was euthanized as moribund on day 313 due to decreased food consumption and marked body weight loss over an approximate 2-week period. One male rat in the 6.8 × 109 vg/dose group was found dead on day 364; this animal had a head tilt that was observed beginning on day 57 through the day of death. One male rat in the 5.2 × 1010 vg/dose group underwent moribund euthanasia on day 175 due to limping and extensive swelling of the limbs. In addition, 2 other male rats in the 5.2 × 1010 vg/dose group were euthanized moribund (day 209 or 276) due to markedly decreased food consumption and significant body weight loss within an approximate 2-week period.
Clinical Observations
The most notable adverse clinical signs observed during the study included head tilts that were noted for 1 male rat in the 6.8 × 109 vg/dose group (day 57 through day 363, the day prior to the animal being found dead) and 2 female rats in the 5.2 × 1010 vg/dose group (day 16 or 107 to scheduled euthanasia on day 144); limping and excessive swelling of the limbs, with muscular atrophy of left hindlimb observed on veterinary evaluation of the animal the week prior to necropsy, that were noted for 1 male rat in the 5.2 × 1010 vg/dose group (day 117 to unscheduled euthanasia on day 175); and a hooked tail that was noted for 1 female rat in the 5.2 × 1010 vg/dose group (days 194-288 and then on day 295 through scheduled euthanasia on day 376).
Other adverse clinical signs were observed for male and/or female rats in the vehicle control and vector-treated dose groups, with no correlation to dose level, hence considered incidental findings. These incidental signs included, but were not limited to, alopecia of the limbs, feet, and/or abdomen; sores/ulcers on the back, hindfeet, and/or abdomen; piloerection; eye discharge; masses at the surgical site or on the back, abdomen, or chest; and scabs at various anatomical sites.
Food Consumption
There were no changes in food consumption that appeared to be directly related to treatment with the vector.
Increases in food consumption were observed over time for individual animals in the vehicle control or vector-treated dose groups; these increases in food consumption generally were correlated with increases in animal body weight. Exceptions to this included the marked decrease in food consumption was observed for 2 males in the 5.2 × 1010 vg/dose group; and 1 male in the vehicle control group that necessitated the moribund euthanasia of the animals.
Body Weights
There were no changes in body weight that appeared to be directly related to treatment with the vector. Throughout the study, the group mean body weights of animals in each of the 3 vector-treated dose groups were generally comparable to the group mean body weight of animals in the respective vehicle control group.
For individual animals in the vehicle control or vector-treated dose groups, progressive increases in body weight generally were observed over time. Exceptions to this included the marked body weight loss (8% to >10%) observed for 2 male rats and for 1 male rat in the vehicle control group that necessitated the moribund euthanasia of the animals.
Functional Observational Battery
Due to the intracerebral delivery system and neurological therapeutic indication, FOB assessment was included in the study to evaluate central/peripheral nervous system function. There were no treatment-related effects on any of the neurobehavioral parameters evaluated for any animals on the study on any day of evaluation.
Clinical Pathology
There were no vector-related changes in hematology, clinical chemistry, or coagulation parameters for animals that underwent euthanasia on day 7, 31, 90, 144, between days 91 and 375, or day 376.
Serum GDNF Levels and Immunological Evaluations
Serum samples collected prior to necropsy were assayed for GDNF levels, anti-GDNF antibody, and anti-AAV2 nAb.
Serum GDNF levels
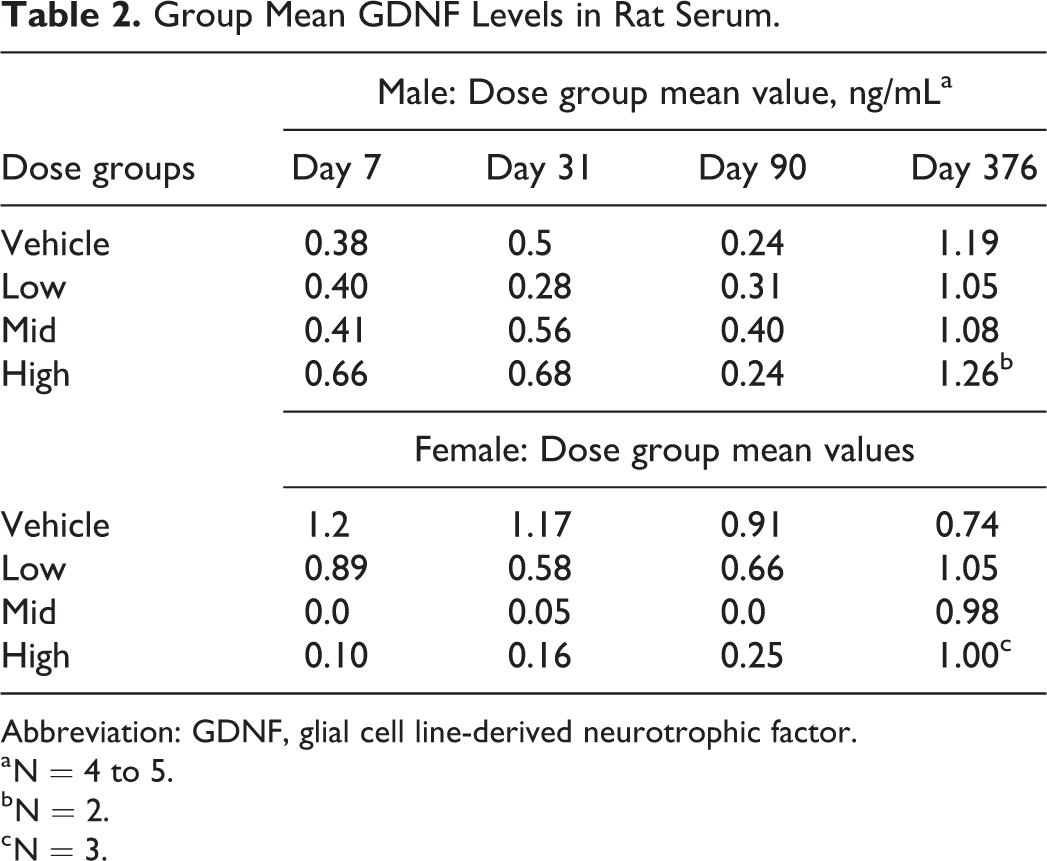
Detectable levels of GDNF were observed in the rats, however, the levels were not appreciably higher than the GDNF values at baseline in samples from days 7, 31, 90, and 376, suggesting no increases in GDNF levels occurred as a result of dosing (Table 2). The observed base line levels may have been due to endogenous GDNF in the circulation. The standard curve of the assay ranged from 0 to 1,000 pg/mL.
Group Mean GDNF Levels in Rat Serum.
Abbreviation: GDNF, glial cell line-derived neurotrophic factor.
a N = 4 to 5.
b N = 2.
c N = 3.
Anti-GDNF Ab Levels
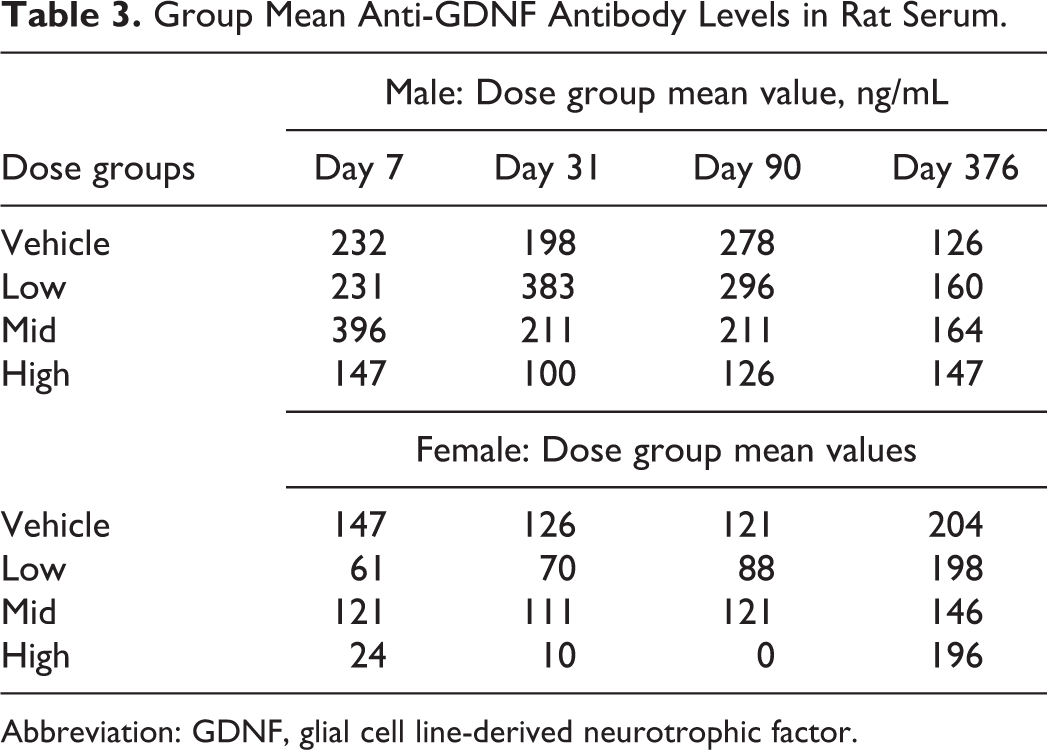
The standard curve for the positive control ranged from 0 to 250 ng/mL. Baseline serum levels of anti-GDNF antibody found in rats prior to treatment ranged from 0 to 424 ng/mL in males (mean ± SD of 125 ± 122 ng/mL) and from 0 to 650 ng/mL in females (mean ± SD of 259 ± 163 ng/mL). Anti-GDNF levels measured on samples from days 7, 31, 90, and 376 were all generally within the range observed in the prestudy animals suggesting no anti-GDNF antibodies were formed as a result of treatment during the study (Table 3).
Group Mean Anti-GDNF Antibody Levels in Rat Serum.
Abbreviation: GDNF, glial cell line-derived neurotrophic factor.
Anti-AAV2 nAb Levels
Mean values from the Anti-AAV2 nAb levels analyses are presented in Table 4. The lowest titer that could be measured was 40 (the inverse of the lowest dilution where the relative fluorescence unit of the diluted sample remained above 50% of the assay maximum signal), which in this assay represented background (group 1). The titers on day 7 were 40 in nearly all animals (with 1 animal in group 1 showing a titer of 80). By day 31, there was no nAb above background in the rats in groups 1 to 3, while there was nAb activity in most (4 of 5) of the female and male rats in group 4 (the high dose group); positive titers ranged from 160 to 2,560. By day 90, the animals in groups 1 to 3 remained all negative with one exception of 1 animal in group 3 which had a titer of 640. By day 90, not only all animals in group 4 had positive titers (ranging from 320 to >5,120) but also appeared to have increased from day 31. By day 376, the titers in groups 1 and 3 were still 40 (with 1 exception in group 3 with a titer of 320), whereas 4 of the 5 group 4 rats had positive titers (ranging from 160 to 1,280) but were lower than they were on day 90.
Anti-AAV2 Neutralizing Antibody Titers in Rat Serum.

Abbreviation: AAV2, adenoassociated viral vector serotype 2.
Macroscopic Observations
There were no vector-related macroscopic changes observed on any day of scheduled (day 7, 31, 90, 144, or 376) or unscheduled (between days 91 and 375) necropsy for male or female rats in the 6.8 × 108, 6.8 × 109, or 5.2 × 1010 vg/dose group.
Organ Weights
There were no vector-related effects on organ weights observed for animals necropsied on day 7, 31, 90, or 376.
Microscopic Observations
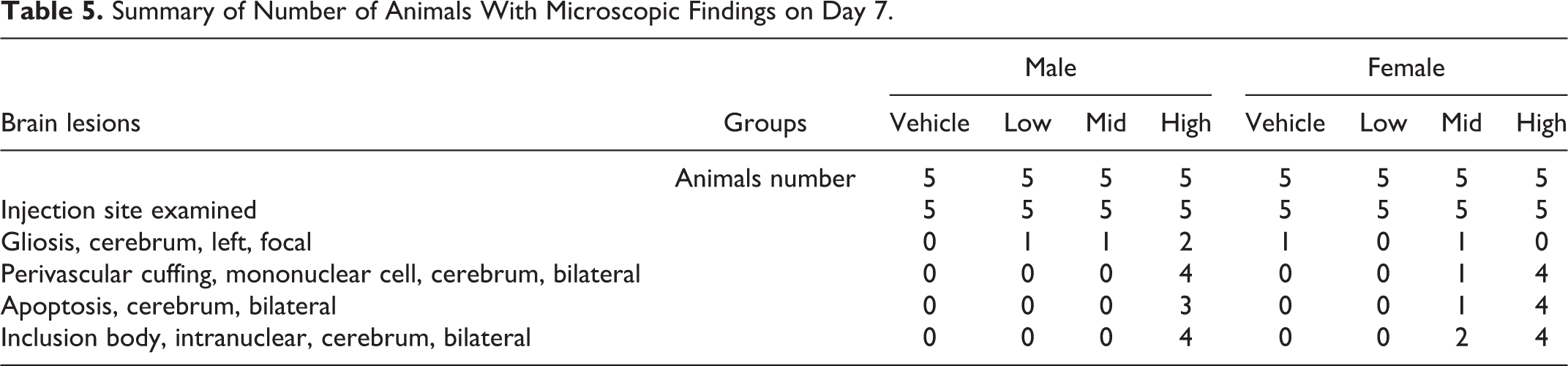
Vector- and dose-related microscopic lesions were observed only in the brain injection site on day 7 for animals in the 6.8 × 109 vg/dose and 5.2 × 1010 vg/dose groups, and on day 31 for animals in the 5.2 × 1010 vg/dose group (Table 5). The microscopic lesions observed on day 7 consisted of gliosis, mononuclear perivascular cuffing, intranuclear inclusion bodies, and/or apoptosis. By day 31, the lesions were limited to mononuclear perivascular cuffing. No vector-related histopathological lesions were observed in the brain on day 90, 144, and 376, or unscheduled necropsy between days 91 and 375.
Summary of Number of Animals With Microscopic Findings on Day 7.
The GDNF immunostaining was observed in the brain, injection site (cerebral striatum) and, less frequently, in the cerebrum (the base of the brain [hippocampal section] or cortex) in the 6.8 × 108, 6.8 × 109, or 5.2 × 1010 vg/dose group, but not for animals in the vehicle control group, that were euthanized on day 7, 31, 90, 144, 376, or intervening days between days 91 and 375 (Figure 1). No GDNF immunostaining was observed in the cerebellum, pons, or medulla oblongata of the brain. Where present, GDNF immunostaining was most pronounced for animals in the 5.2 × 1010 vg/dose group indicating increased levels of anti-AAV2 nAb resulted in no detectable impacts on tissue transgene expression. A dose–response was observed in association with GDNF immunostaining in the cerebral striatum of the brain injection site. The incidence and intensity of GDNF immunostaining in male and female rats were similar within dose groups.
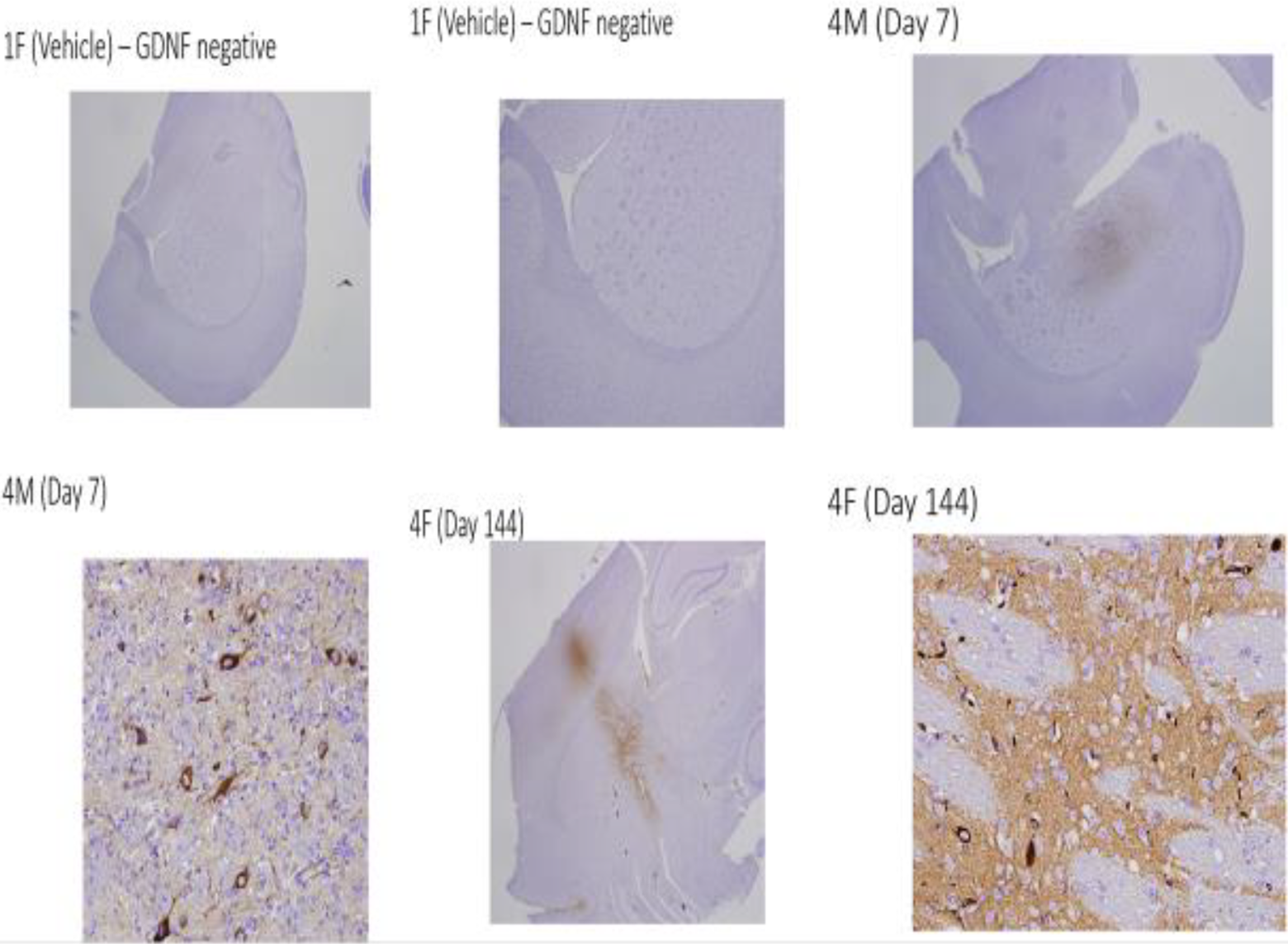
Representative images of anti-GDNF immunohistochemistry staining in control and high dose animals on day 7 and 144. Note GDNF immunostaining was limited to the injection site. GDNF indicates glial cell line-derived neurotrophic factor.
Since GDNF is a neurotrophic factor that can promote the growth, survival, maintenance, and differentiation of neurons in the CNS, potential axonal sprouting (CGRP immunostaining) and Swann cell hyperplasia (cresyl violet staining) were assessed in brain and spinal cord for animals in the vehicle control and high dose (5.2 × 1010 vg/dose) groups on day 7 and 376 and, with the exception of 1 male rat in the high dose group, for animals in the 6.8 × 109 or 5.2 × 1010 vg/dose group that died or were euthanized moribund between days 91 and 375. The incidence and intensity of CGRP immunostaining in the cervical spinal cord, lumbar spinal cord, and medulla oblongata in the 5.2 × 1010 vg/dose group euthanized on day 7, 31, 90, or 376 were similar to those observed for animals in the corresponding vehicle control groups. Minimal CGRP immunostaining was also observed in the cervical spinal cord, lumbar spinal cord, and/or medulla oblongata for animals in the 6.8 × 109 or 5.2 × 1010 vg/dose group that died or were euthanized between days 91 and 375. There was no evidence of Schwann cell hyperplasia in the cervical or lumbar spinal cord or in the medulla oblongata on any day of necropsy for male and female rats in the 5.2 × 1010 vg/dose group.
For the 2 female rats in the 5.2 × 1010 dose group that had head tilts and that were euthanized early on day 144 to determine plausible cause of the head tilt, no abnormal areas of tyrosine hydroxylase immunostaining (marker for dopaminergic neuron) were observed in any sections of brain evaluated.
Discussion
The study was conducted to determine the target organ toxicity of AAV2-GDNF (NSC-D751136) in rats treated with a single dose followed by a 12-month observation period. AAV2-GDNF was administered intracerebrally (left and right striatum) to male and female rats at a dose of 0, 6.8 × 108 vg/dose, 6.8 × 109 vg/dose, or 5.2 × 1010 vg/dose. The doses were selected to provide adequate safety margin for the proposed clinical starting dose of 9 × 1010 vg considering a 600-fold difference in brain weight between human and rat (1,200 g human brain vs 2 g rat brain weight). The study provided a safety margin of 4.5-fold when the no observed adverse effect level (NOAEL) dose (6.8 × 108 vg) compared with the proposed clinical starting dose (9 × 1010 vg) per gram of brain tissue between rat and humans.
During the study, although mortality or unscheduled euthanasia occurred only for animals given the mid (1 male rat) or high (3 male rats) dose level of AAV2-GDNF, the mortality/morbidity observed for these animals did not appear to be directly related to treatment with the vector. This was indicated by the observations that the clinical manifestations of ill health, most notably decreased food consumption and body weight loss, that lead to the euthanasia of 2 male animals in the 5.2 × 1010 vg/dose group were similar to those observed for the animal in the vehicle control group that also underwent unscheduled euthanasia; for one of these AAV2-GDNF-treated animals euthanized on day 209, the cause of death was attributed to a neoplasm in the gastrointestinal tract. For the third male rat in the 5.2 × 1010 vg/dose group that was euthanized as moribund, the nature of the clinical signs that led to the euthanasia of the animal suggested that the injuries may have been related to prior trauma or mechanical injury. In addition, the morbidity of the male rat in the 6.8 × 109 vg/dose group that died was determined to be related to a pituitary gland adenoma. For other animals in the AAV2-GDNF-treated dose groups that survived to scheduled necropsy, no signs of CNS toxicity, as assessed by clinical observations and FOB evaluations, no treatment-related effects on body weight or food consumption, and no vector-related changes in clinical pathology parameters were observed. These incidental findings are less extensive than results previously reported for direct GDNF protein administration. Hoveland et al conducted a 6-month study in monkey with continuous unilateral intraputaminal infusion of recombinant methionyl human GDNF (rmetHuGDNF) that produced drug-related effects including reductions in food consumption, body weight decrements (at the high dose of 100 μg/d), inflammatory changes in the brain parenchyma along the catheter tract and at the infusion site (≥15 μg/d, i.e. all doses), and meningeal thickening along dorsal aspects of the spinal cord and underlying the medulla oblongata (≥30 μg/d). Cerebellar lesions characterized by Purkinje cell loss, molecular layer atrophy, and granule cell loss were also reported. 22
It has been reported that nigrostriatal GDNF overexpression decreases body weight and food consumption in rodents and nonhuman primates, 23,26 however, we didn’t see significant changes in these endpoints which would speak for the intrastriatal delivery of AAV2-GDNF. Excluding the rats that died or underwent unscheduled euthanasia, the most notable adverse clinical sign noted during the study was a head tilt that was observed for 1 male rat in the 6.8 × 109 vg/dose group and 2 female rats in the 5.2 × 1010 vg/dose group. No histopathological tumors were present in the brain of the 2 female animals in the 5.2 × 1010 vg/dose group and no abnormalities were observed in the inner ears of either animal. A pituitary gland adenoma that was found during the necropsy of the male rat in the 6.8 × 109 vg/dose group may have been responsible for the head tilt observed for this animal. However, for all 3 rats that exhibited head tilts, no evidence of increased GDNF, CGRP, or, where evaluated, tyrosine hydroxylase expression in the brain and/or spinal cord was observed. In addition, no apparent vector-related changes in clinical pathology parameters were evident and no neurobehavioral signs indicative of CNS toxicity were observed for these animals. Thus, a direct correlation between treatment with the vector and the occurrence of a head tilt was not established for these 3 animals.
The results of ELISAs established that, for male and female animals in the 6.8 × 108, 6.8 × 109, or 5.2 × 1010 vg/dose group, serum levels of GDNF and serum levels of anti-GDNF antibody observed on day 7, 31, 90, or 376 were lower than or comparable to the values determined prior to treatment of the animals with AAV2-GDNF. Thus, there was no evidence for the presence of GDNF in blood (serum) following intracerebral administration of AAV2-GDNF to rats. Similar findings have been reported for Rhesus monkeys administered AAV2-GDNF by convection-enhanced delivery into the putamen and SN. 19 In contrast, increased anti-AAV2 nAb titers were observed on days 31, 90, and 376 for both male and female rats in the 5.2 × 1010 vg/dose group but not for rats in the 6.8 × 108 or 6.8 × 109 vg/dose group, with highest titers occurring on day 90. These data indicated that, for animals given the highest dose level, some AAV2-GDNF appeared to have entered the peripheral circulation; however, this did not result in the presence of detectable levels of circulating GDNF protein or in the development of an antibody response against expressed GDNF. In addition, increased levels of anti-AAV2 nAb did not appear to influence tissue transgene expression.
The histopathological evaluations of tissues indicated that no histopathological lesions indicative of systemic toxicity were present for any animals in the AAV2-GDNF-treated dose groups on any day of necropsy. The injection site in the brain was determined to be the only target organ of AAV2-GDNF-related toxicity. Vector- and dose-related microscopic lesions observed at the injection site were most extensive and severe on day 7 for animals in the 5.2 × 1010 vg/dose group; however, similar lesions were observed on day 7 for animals in the 6.8 × 109 vg/dose group. Lesions observed on day 7 included mild to moderate gliosis, mononuclear perivascular cuffing, intranuclear inclusion bodies, and apoptosis. By day 31, the vector-related microscopic lesions were limited to perivascular cuffing that was observed only for animals in the 5.2 × 1010 vg/dose AAV2-GDNF dose group. It is quite possible that some of these observed histopathological lesions in rat could be due to immunogenic reaction to foreign transgene (human GDNF). No vector-related histopathological lesions were observed in the brain of any animals in the AAV2-GDNF-treated dose groups on day 90 and subsequent days of necropsy, indicating that the lesions had resolved over time. It appears that these histopathological changes did not affect transgene expression since a dose-dependent transgene expression was observed at the injection site throughout the duration of study (376 days).
Immunohistochemical staining techniques were utilized to visualize GDNF expression in the brain and CGRP expression in the brain and upper portions of the spinal cord. The results indicated that GDNF expression in brain was persistent. The GDNF immunostaining was observed in the injection site (cerebral striatum) on the left and/or right side of the brain for animals in all 3 GDNF-treated dose groups (6.8 × 108, 6.8 × 109, or 5.2 × 1010 vg/dose), but not for animals in the vehicle control group, through the last day of necropsy (day 376). Among animals in individual dose groups, the intensity of staining was similar for male and female rats. A dose–response was observed for GDNF immunostaining in the cerebral striatum (site of injection), with the most robust expression observed for animals in the 5.2 × 1010 vg/dose group. It was observed that GDNF immunostaining was not confined to the injection site in the cerebral striatum but, for some animals, GDNF immunostaining was also observed in the base of the cerebrum on days 7, 31, and 90 and occasionally in the cerebral cortex on days 90 and 376. The distribution of GDNF to regions in the brain outside of the injection site was consistent with results obtained by Hadaczek and coworkers for rats given intracerebral doses of GDNF, using the same dosing technique and same site of injection as used during the current study. 3 These investigators found that, in addition to the targeted striatum, GDNF was present in the septum and base of the brain of rats dosed with GDNF and proposed that active perivascular transport may result in the spread of GDNF and other therapeutic agents beyond the infused structure.
Calcitonin-gene-related peptide is expressed at high levels following injury to spinal sensory neurons, and altered CGRP expression has been reported for rats treated with GDNF. 27,28 CGRP immunostaining was used during the current study as a marker of potential axonal sprouting. No differences in the extent of CGRP immunostaining of the brain or cervical or lumbar spinal cord were observed between rats in the 5.2 × 1010 vg/dose group and rats in the vehicle control group. Similarly, a quantitative assessment of Schwann cell hyperplasia based upon thickening of the pia was obtained with cresyl violet staining of the medulla oblongata, cervical spinal cord, and lumbar spinal cord. For male and female animals euthanized on day 7, 31, 90, 144, or 376, no differences in cresyl violet staining of these regions of the brain and spinal cord were observed between animals in the 5.2 × 1010 vg/dose AAV2-GDNF group and animals in the vehicle control group. These results indicated that intracerebral administration of AAV2-GDNF to rats did not stimulate neuronal hyperplasia indicating very little risk, if any, for tumorigenicity.
Conclusions
There was no vector-related systemic toxicity elicited following intracerebral injection of a single doses of 6.8 × 108, 6.8 × 109, or 5.2 × 1010 vg/dose AAV2-GDNF to rats. The local toxicity of AAV2-GDNF was characterized by dose-related histopathological lesions at the site of injection in the brain that were observed at a dose level of 6.8 × 109 or 5.2 × 1010 vg/dose; these lesions appeared to be reversible. No sex-related differences in the response of rats to intracerebral administration of AAV2-GDNF were observed. AAV2-GDNF appeared to enter the peripheral circulation at 5.2 × 1010 vg/dose; however, this did not result in the presence of circulating GDNF protein or in the initiation of a GDNF-antibody response. The increased levels of anti-AAV2 nAbs did not appear to influence tissue transgene expression. The NOAEL for a single intracerebral dose of AAV2-GDNF was considered to be 6.8 × 108 vg/dose. Thus, this study delineates a dosing level in which generally safe and effective AAV2-GDNF intracerebral gene delivery can be used to initiate clinical evaluation of AAV2-GDNF gene therapy for the treatment of PD.
Footnotes
Acknowledgments
The authors thank the staff at Southern Research Institute, Birmingham, Alabama, for conducting the study, and Olga Zelenai and Bernd Hauck at the Children’s Hospital of Philadelphia, Pennsylvania, for production and characterization of the recombinant AAV vector used in the study.
Author Contributions
Pramod Terse contributed to conception and design, contributed to acquisition, drafted the manuscript, and critically revised the manuscript; Adrian Kells contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript; Fraser Wright contributed to acquisition, analysis, and interpretation and critically revised the manuscript; Patricia Noker contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. Krystof S. Bankiewicz contributed to conception and design, contributed to interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: APK is employee of and has an equity interest in Brain Neurotherapy Bio, Inc, a company developing AAV2-GDNF gene therapy for Parkinson’s disease; JFW is a cofounder of Spark Therapeutics and Kriya Therapeutics, serves as an advisor to companies developing gene therapy products, and is an inventor of patents relating to recombinant AAV and lentivirus technologies; KSB is the founder and equity holder in Voyager Therapeutics and Brain Neurotherapy Bio. PN and PST declare no potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NCI Toxicology contract N01-CM-42201, NHLBI GTRP vector synthesis contract (CHOP, PA) and NINDS under BrIDGs (former NIH-RAID)/NCATS Program.
