Abstract
Two 6-month repeat-dose toxicity studies in cynomolgus monkeys illustrated immune complex–mediated adverse findings in individual monkeys and identified parameters that potentially signal the onset of immune complex–mediated reactions following administration of RN6G, a monoclonal antibody (mAb). In the first study, 3 monkeys exhibited nondose-dependent severe clinical signs accompanied by decreased erythrocytes with increased reticulocytes, neutrophilia, monocytosis, thrombocytopenia, coagulopathy, decreased albumin, azotemia, and increased serum levels of activated complement products, prompting unscheduled euthanasia. Histologically, immunohistochemical localization of RN6G was associated with monkey immunoglobulin and complement components in glomeruli and other tissues, attributable to immune complex disease (ICD). All 3 animals also had anti-RN6G antibodies and decreased plasma levels of RN6G. Subsequently, an investigational study was designed and conducted with regulatory agency input to detect early onset of ICD and assess reversibility to support further clinical development. Dosing of individual animals ceased when biomarkers of ICD indicated adverse findings. Of the 12 monkeys, 1 developed anti-RN6G antibodies and decreased RN6G exposure that preceded elevations in complement products, interleukin-6, and coagulation parameters and decreases in albumin and fibrinogen. All findings in this monkey, except for antidrug antibody (ADA), reversed after cessation of dosing without progressing to adverse sequelae typically associated with ICD.
Keywords
Introduction
In cases where a humanized monoclonal antibody (mAb) is infused into nonhuman primates for nonclinical safety testing, the humanized mAb can be sufficiently “foreign” to elicit immune-mediated adverse reactions, including IC–mediated pathologic changes. Often, monoclonal antibodies directed against soluble circulating targets can complex with those targets and lead to increased IC formation. However, increased IC formation can also occur when a therapeutic mAb is recognized as foreign by the host, resulting in formation of antidrug antibodies (ADA) that bind to the therapeutic agent. Depending on the size and charge of the complexes formed and saturation of the clearance mechanism, ICs may deposit in various tissues causing inflammation and subsequent tissue damage, facilitated by release of vasoactive amines, cytokines, and complement degradation products (Schreiber and Penney 1984; Hardy and deShazo 2001; Rojko et al. 2014). IC can also deposit in filtering organs such as liver, spleen, and kidney and have been implicated in the pathogenesis of many autoimmune diseases with systemic lupus erythematosus being the classical example (Wahren-Herlenius and Dorner 2013). One of the common findings in immune complex diseases (ICD) is the development of glomerulonephropathy (Herbert et al. 1991).
RN6G is a humanized mAb that binds to the major species of amyloid beta peptides and is currently in development for age-related macular degeneration (AMD). To support the dosing of RN6G to patients in clinical trials, repeat-dose toxicity studies were conducted in cynomolgus monkeys, a pharmacologically relevant species. In the 6-month repeat-dose toxicity study, 3 monkeys in the mid-dose group showed adverse clinical signs postinjection on days 57, 99, and 127 and were subsequently euthanized in a moribund state. Data collected from these monkeys (clinical observations, clinical pathology, toxicokinetics, immunogenicity, macroscopic observations, microscopic observations, electron microscopy, and immunohistochemistry) confirmed the pathogenesis associated with immunogenicity of RN6G. In general, immunogenicity observed in nonclinical studies is not predictive of immunogenicity in clinical settings. Furthermore, adverse findings in nonclinical species are the result of frank immunogenic response with doses and dosing regimen often overwhelming the clearance mechanism of ICs. However, in this case, the lack of dose-responsiveness, the severity of reaction which resulted in moribundity, and not picking up early signals for ICD in monkeys to this humanized mAb generated concern for human safety. A subsequent investigative study, therefore, was conducted in monkeys to identify early signals of IC-mediated reactions occurring prior to an onset of severe toxicity and clinical signs, and to assess reversibility of any adverse reaction markers upon cessation of dosing. Such investigation was deemed necessary by a regulatory agency and authors in order to ensure human safety of RN6G, prior to moving forward to multiple clinical doses.
Materials and Methods
Six-month Repeat-dose Toxicity Study in Cynomolgus Monkeys
Test substance
RN6G was formulated at a concentration of 11 mg/ml in an injectable histidine buffer (20-mM histidine buffer at pH 5.5 including the following components: 0.7856 g/L histidine, 3.1316 g/L, histidine·HCl·H2O, 0.05 g/L, 2NaEDTA·2H2O, 0.1 g/L methionine, 84 g/L trehalose·2H2O, 0.2 g/L Polysorbate-80] prepared in sterile water for injection). The control test substance consisted of the identical injectable histidine buffer without RN6G.
Test animals
Fifty-two drug naive cynomolgus monkeys (Covance Research Products, Alice, TX, Mauritius origin) were individually housed, maintained, and handled in compliance with all federal regulations and recommendations set forth in
Experimental design study (1)
Animals were randomized into groups with dose levels of 0 (vehicle control), 10, 30, and 100 mg/kg. Four males and females were randomized to each group and 2 additional males and female animals were included in each group and assigned to a treatment-free recovery period. Animals were dosed intravenously, once weekly for 6 months (total of 27 doses). Animals surviving to scheduled necropsy were euthanized on day 185 of the study, and recovery animals were euthanized on day 240 of the study. Clinical observations and qualitative food consumption evaluations were made daily.
In-life Data Generation
Urinalysis
Urine was obtained by cystocentesis during necropsy and analyzed (Bayer Multistix, Bayer Diagnostics, Tarrytown, NY, and Leica TS Meter, Reichart, Inc., Depew, NY). Specific urine biomarkers were not implemented.
Clinical pathology
Scheduled blood samples for hematology (ADVIA 120 hematology analyzer; Bayer Diagnostics, Tarrytown, NY), coagulation (STA Compact/Physical clot detection, Diagnostica Stago, Cedex, France), and serum chemistry analysis (Olympus AU 2700 chemistry analyzer, Tokyo, Japan) were collected from each animal during acclimation and on days 59, 71, 184, and 239. Blood samples for clinical pathology were also collected as necessary for unscheduled euthanasia.
Toxicokinetics
Blood samples were collected and processed to plasma from all animals at predose, 0.5, 2, 7, and 24 hr post dose on days 1, 64, and 183. Samples were also collected prior to dosing once weekly throughout the study and during the recovery period. Unscheduled collections were performed shortly before necropsy in animals euthanized prior to the end of the dosing phase. Plasma level of RN6G was measured using a quantitative enzyme linked immunosorbent assay (ELISA) method.
Serum complement
Serum samples collected from a subset of scheduled and all unscheduled euthanized RN6G-dosed animals and control animals were analyzed using Enzyme Immuno Assay Kit (Quidel, San Diego, CA) for complement (C) C3a, C4d, and SC5b-9.
Microscopic data generation
All animals (scheduled and unscheduled) underwent necropsy and protocol required tissues were collected for organ weights, and macroscopic and microscopic evaluation. Tissues for all animals were collected and immersion fixed in 10% neutral buffered formalin (NBF) or modified Davidson’s fluid dependent upon tissue. Tissues were trimmed, processed to paraffin block, sectioned, stained with hematoxylin and eosin (H&E), and examined microscopically.
Transmission electron microscopy
Kidney sections from the unscheduled sacrifice animals were evaluated with transmission electron microscopy. Animals surviving to scheduled necropsies were not evaluated with transmission electron microscopy. Kidney samples originally fixed in 10% NBF were minced to approximately 1 to 2 mm2, fixed in 2.5% glutaraldehyde + 2% formaldehyde in 0.1 M sodium phosphate buffer, postfixed in 0.1 M sodium phosphate buffered 1% osmium tetroxide, dehydrated through a graded ethanol series, and embedded in epoxy resin. Sections (∼0.6 µm thick) were prepared from 2 randomly selected epoxy resin tissue-bearing blocks of each animal, stained with Toluidine Blue O, and examined using light microscopy. Sections (∼70 nm) from selected blocks were prepared and stained with uranyl acetate and lead citrate, examined in a Hitachi H-7000 transmission electron microscope and digital images were acquired (AMT, Inc Woburn, MA).
Immunohistochemistry
For immunohistochemistry analysis, paraffin-embedded liver and kidney tissue sections from unscheduled death animals and donor paraffin embedded normal (negative control) and rheumatoid arthritis diseased (positive control) animals (Charles River Laboratory Pathology Associates, Frederick MD) were stained with monkey-adsorbed, goat antihuman immunoglobulin G (IgG; to detect exogenously administered humanized test article, RN6G); human-adsorbed, goat anti-monkey IgG (to detect endogenous cynomolgus monkey IgG); goat antihuman IgM (to detect cross-reacting endogenous cynomolgus monkey IgM); goat antihuman IgA (to detect cross-reacting endogenous cynomolgus monkey IgA); and goat antihuman C3 and mouse IgG2a antihuman SC5b-9 (to detect complement components in “immune complexes”). Negative control antibodies were matched for species and staining concentration. All staining were visualized by immunoperoxidase methods followed by examinations with light microscopy. A complete list of immunohistochemical reagents is located in Supplemental Table 1. No additional tissues from study 1 were examined with immunohistochemistry techniques.
Summary of key clinical pathology parameters and serum biomarkers.
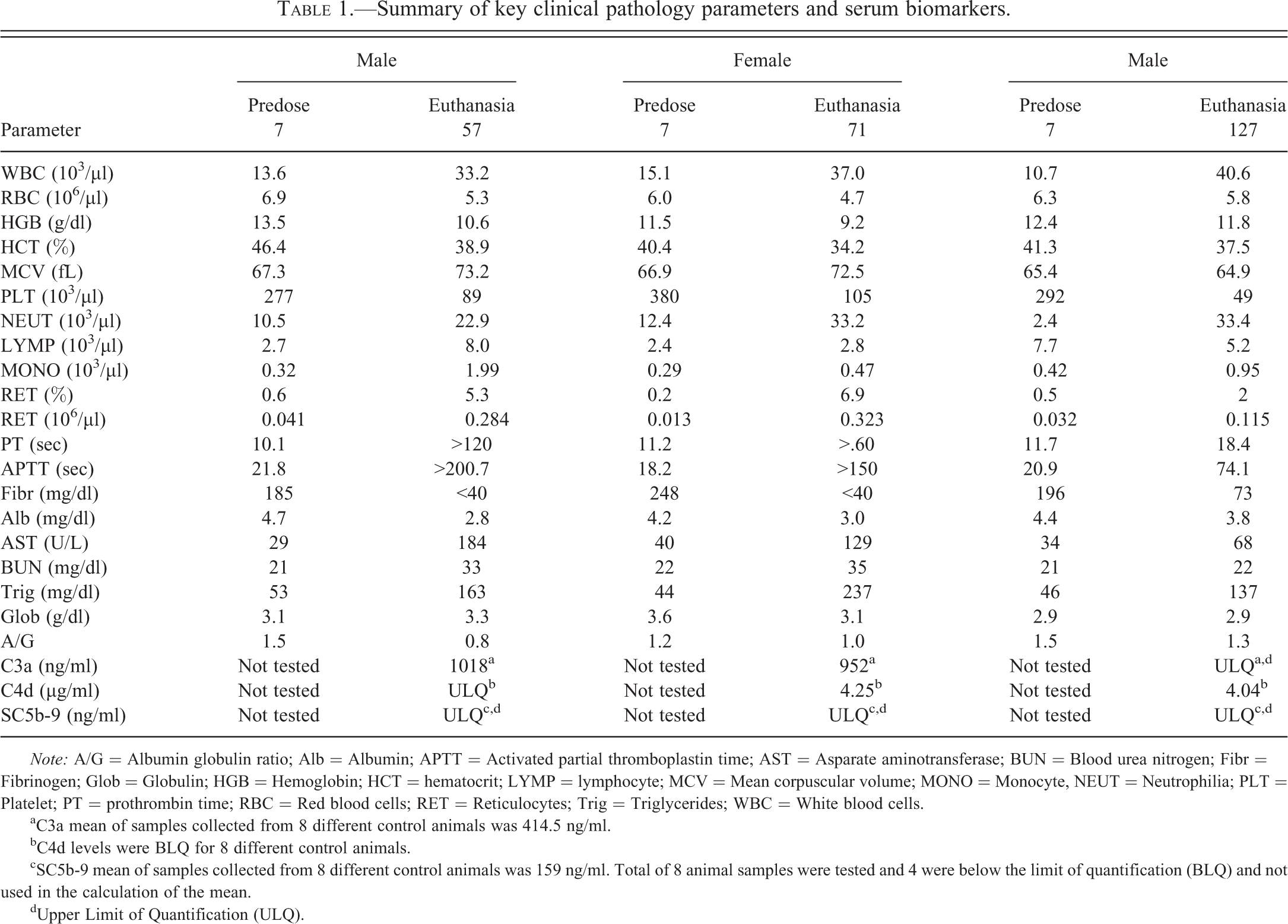
aC3a mean of samples collected from 8 different control animals was 414.5 ng/ml.
bC4d levels were BLQ for 8 different control animals.
cSC5b-9 mean of samples collected from 8 different control animals was 159 ng/ml. Total of 8 animal samples were tested and 4 were below the limit of quantification (BLQ) and not used in the calculation of the mean.
dUpper Limit of Quantification (ULQ).
Six-month Investigational Study in Cynomolgus Monkeys
The investigational monkey study was conducted with large number of animals (
Test substance
The lot and formulation techniques for RN6G and associated control used were identical to study 1.
Test animals
Twenty drug naive cynomolgus monkeys were obtained from Covance Research Products, Inc. (Alice, TX). Animals were individually housed, maintained, and handled in compliance with all federal regulations and recommendations set forth in
Experimental design study 2
Animals were randomized into groups with dose levels at 0 (vehicle control,
In-life evaluations
Blood samples for serum complement, cytokine (including C-reactive protein), hematology, coagulation, and clinical chemistry analyses were collected from fasted animals twice during the predose phase and once every 2 weeks for the entire study period.
Urinalysis
Urine was obtained by collection from cage pans on days 3, 17, 31, 45, 59, 73, 87, 101, 115, 129, 143, 157, and 171 and analyzed for color, clarity, pH, protein, glucose, ketones, bilirubin, occult blood nitrites, leukocytes, urobilinogen, and specific gravity, and by microscopic evaluation of urine sediment.
Clinical pathology, toxicokinetics, anti-drug antibodies, and complement
Blood samples for hematology, serum chemistry, toxicokinetics, antidrug antibodies, and serum complement were collected twice during the predose phase and approximately 4 hr post dose on days 1, 15, 29, 43, 57, 71, 85, 99, 113, 127, 141, 155, and 169. For coagulation, blood samples were collected twice during the predose phase and approximately 4 hr post dose on days 1, 29, 57, 85, 113, 141, and 169.
Serum cytokines and C-reactive protein (CRP)
Blood samples were collected and processed to serum from all animals, predose on days 1, 15, 29, 43, 57, 71, 85, 99, 113, 127, 141, 155, and 169. Serum interleukin (IL)-6, tumor necrosis factor (TNF)-α, IL-1β, and (interferon [IFN])-γ levels were measured using Millipore Milliplex Map Kit (Millipore, Billerica, MA) and CRP levels were determined on Siemens Advia 1200 instrument using Siemens CRP reagents (Bayer Healthcare, Terrytown, NY).
Microscopic data generation
All animals were necropsied as scheduled and protocol required tissues were collected for organ weights, macroscopic evaluation, and microscopic evaluation of sections stained with H&E as described previously.
Regulatory interaction
Results from the 6-month repeat-dose toxicity study were submitted to the regulatory agency as part of nonclinical safety data update in support of ongoing clinical trial. Based on observed adverse findings from the repeat-dose toxicity study, a 6-month investigational toxicity study was designed in consultation with the regulatory agency.
Results
Six-month Toxicity Study in Cynomolgus Monkeys
Clinical observations
Three animals administered 30 mg/kg/week of RN6G were euthanized in moribund condition prior to the end of the dosing phase: a male was euthanized on day 57, a female was euthanized on day 99, and another male was euthanized on day 127. Clinical observations in these 3 animals included hypoactivity, excessive salivation, hunched posture, emesis, fast respiration, and recumbency. All adverse clinical signs leading to early euthanasia occurred within 1 hr of dosing. No other animals in the study showed any RN6G-related adverse clinical signs.
Clinical pathology and urinalysis
The 3 animals euthanized prior to the end of the dosing phase exhibited similar hematology and serum chemistry changes that were attributed to IC-mediated findings (Table 1). At the time of euthanasia, these animals had minimal reductions in red blood cells (RBC) count, hemoglobin concentration, and hematocrit. They also had increases in absolute reticulocyte count, consistent with either blood loss or erythrocyte destruction and regeneration. Also noted in these 3 monkeys were generalized increases in leukocyte counts with increased neutrophil and monocyte counts, indicating an inflammatory process. Marked thrombocytopenia accompanied by increased mean platelet volume (MPV) was noted in all 3 animals at the time of euthanasia consistent with platelet consumption accompanied by regeneration. Coagulation profiles, performed in these animals at the time of euthanasia, showed prolonged prothrombin time (PT) and activated partial thromboplastin time (APTT), as well as decreased fibrinogen consistent with consumptive coagulopathy. These monkeys had decreased total serum protein, lower albumin, with a concomitant decrease in albumin globulin ratio (A/G) and increased triglycerides; azotemia with increase in blood urea nitrogen (BUN) and creatinine in 2 animals and increased creatinine in the 3rd animal. In 2 animals, azotemia was accompanied by an elevation in phosphorus. Taken together, these findings were thought to be most suggestive of glomerular damage with resultant nephrotic syndrome (Stockham and Scott 2008). AST elevations noted in the 2 animals were unaccompanied by histologic evidence of hepatic or muscle damage. Clinical pathology analysis of animals undergoing scheduled necropsy was unremarkable and within historical controls. No meaningful changes in urinalysis were observed in any animal in study 1.
Complement
In all 3 animals euthanized prior to scheduled necropsy, serum levels of complement split products, C3a, C4d, and SC5b-9 were higher compared to the subset of control animal samples (Table 1).
Plasma drug concentration and antidrug antibody
All animals treated with RN6G initially showed circulating drug levels generally proportional the dose administered. However, the animals euthanized prior to scheduled necropsy began exhibiting lower plasma levels compared to cohorts in the same dose group starting at day 22. By day 64, their plasma RN6G concentrations were reduced by >90% compared to other animals in the same dose group when assessed 24 hr post dose. All 3 animals were positive for anti-RN6G antibodies, while no other animals in the study were positive for anti-RN6G antibodies.
Microscopic findings
In all 3 animals euthanized in moribund condition, neutrophilic infiltrates were observed in the lungs and sinusoids of the liver. One animal had perivascular neutrophils in the synovium of the knee joint (data not shown). In addition, all 3 animals had minimal to mild increases in glomerular mesangial matrix morphologically consistent with immune complex deposition (Figure 1A). Similar microscopic findings were not present in animals examined during scheduled euthanasia.
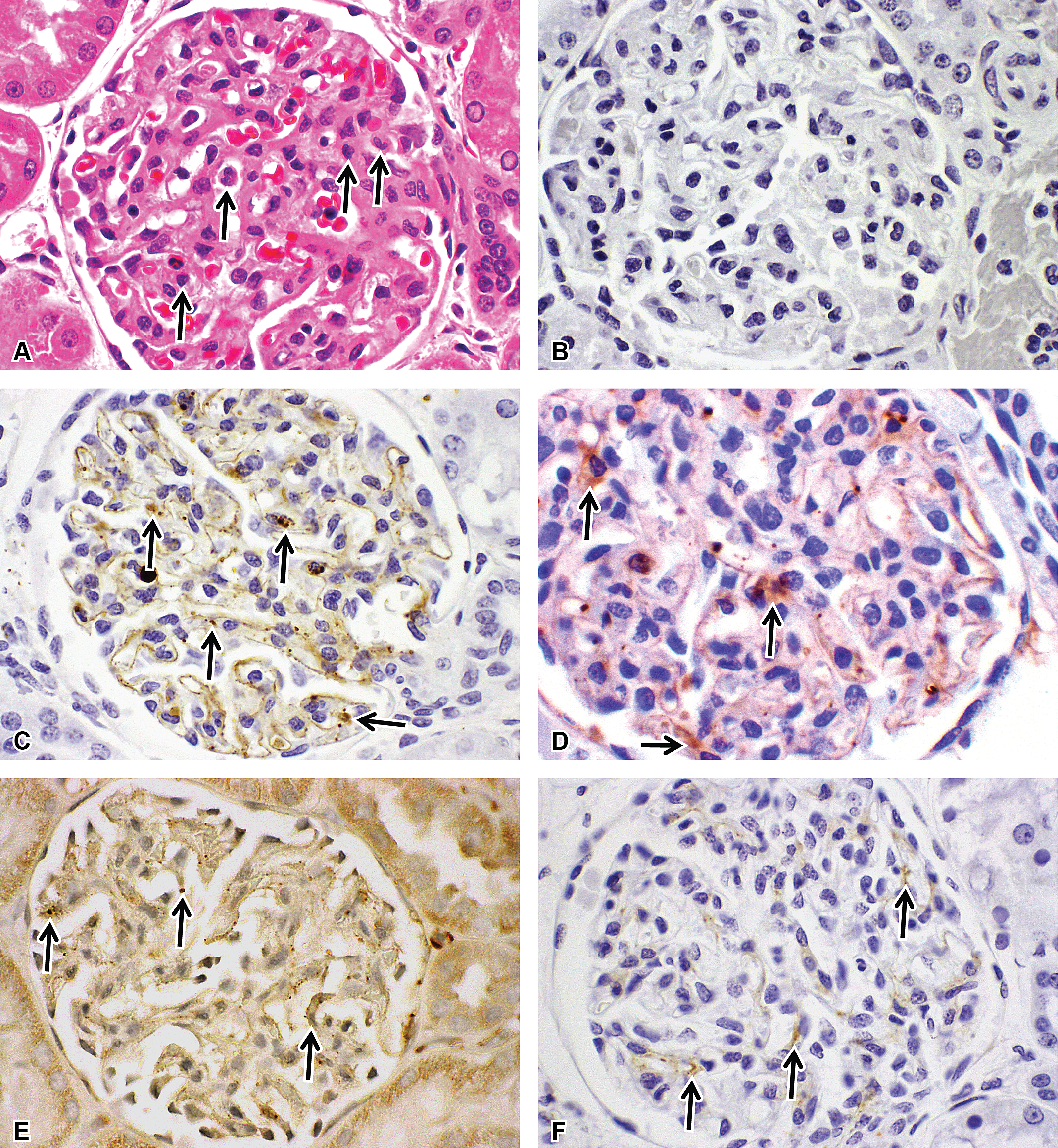
(A) Hematoxylin and eosin staining of the glomerular changes including moderate neutrophilic infiltrates (arrows), as well as mild to moderate thickening of the mesangium. Immunohistochemical staining of the glomerulus representing negative control IgG (B), arrows indicating areas of positive staining for human IgG (C), monkey IgG (D), and complement factors C3a (E) and SC5b-9 (F). All images are at 40× magnification. IgG = immunoglobulin G
Transmission electron microscopy
Multiple dome-shaped moderately electron dense deposits external to the lamina densa of the glomerular capillary basement membranes were present in the kidneys of all 3 animals euthanized early and were morphologically consistent with immune complex deposits (Figure 2).
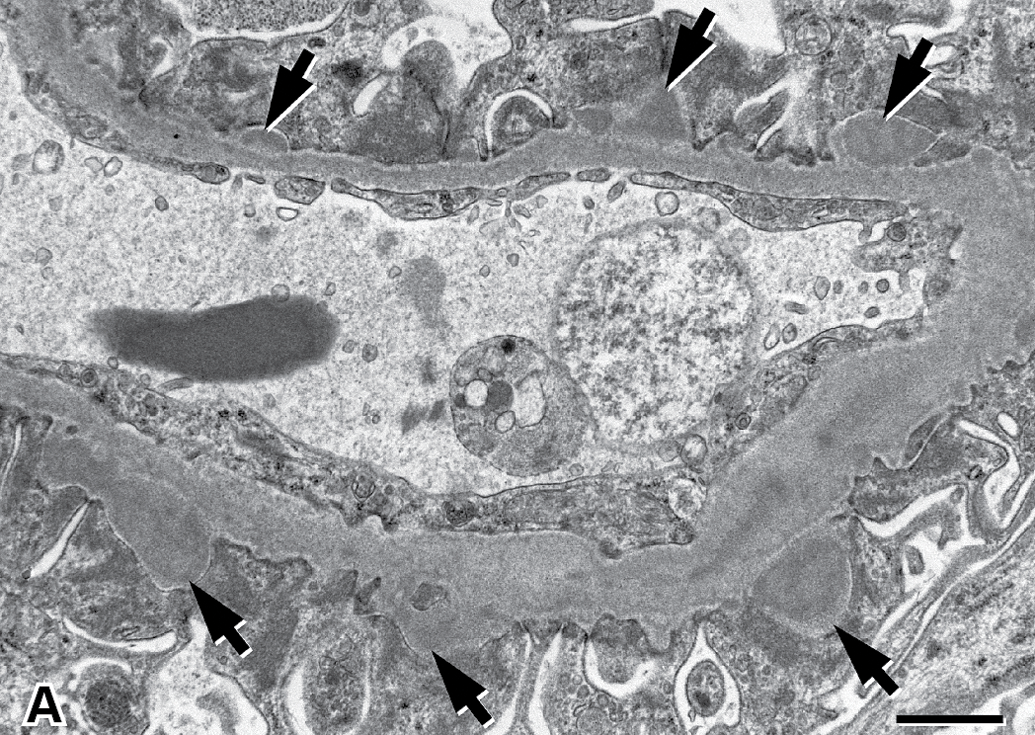
Multiple dome-shaped moderately electron dense deposits external to the lamina densa of the glomerular capillary basement membrane. Scale bar = 1 µm.
Immunohistochemistry
There were granular deposits of RN6G, monkey IgG, IgM, IgA, C3, and/or SC5b-9 in multiple tissue elements in the liver and kidney (Figure 1B–F; Supplemental Table 4) of the 3 animals euthanized prior to the end of dosing phase. These immunohistochemically positive materials were observed as granular intravascular proteinaceous material as well as granular deposits in tissue granulocytes, monocyte/macrophages, liver Kupffer cells, hepatocytes, and spindloid/dendritic cells. Other granular deposits were observed in the mesangial, subepithelial, and/or subendothelial locations within kidney glomerular tuft, glomerular parietal epithelium and or proximal tubular epithelium in kidney, and intima/media of arteries and or arterioles.
Six-month Investigational Study in Cynomolgus Monkeys
Clinical signs
No treatment-related clinical signs were observed during the study; however, dosing of 1 female was suspended after administration of the first 18 of the intended 27 doses due to findings in the parameters monitored below as the dose stopping criteria. Dosing of this animal was discontinued starting with the dose on day 127; therefore, the last administered dose was on day 120 with necropsy occurring as scheduled on day 185. All summaries given subsequently refer to this particular female, as the results for all other animals on the study were normal.
In this study, of the 12 animals administered RN6G, only 1 animal demonstrated IC-mediated findings, including presence of high concentration of anti-RN6G antibody (ADA) with concomitant decrease in systemic exposure, followed by an increase in IL-6 and complement split factors SC5b-9. In addition, decreased serum albumin, platelets, and fibrinogen were observed with prolongation of coagulation parameters (PT and APTT). After dosing of this animal was stopped on day 127 (after 18 doses), all monitored parameters, except ADA, returned to near baseline level within 7 days. This animal and others in its cohort group completed the study without any other adverse or test article–related findings, including terminal histopathological assessment.
Clinical pathology data
Observations include mildly decreased serum albumin on days 43 to 113, mild to moderately decreased platelet counts on days 99 and 113 with prolonged PT and APTT and decreased fibrinogen on day 113. Low albumin resulted in lower A/G ratios. No other clinical chemistry, urinalysis, or urine chemistry parameters suggesting renal damage/dysfunction were present. Dosing for this animal was discontinued after the dose administered on day 120 and abnormal clinical pathology findings returned to approximate predose levels by day 127.
Toxicokinetics
Plasma RN6G concentration in this animal was notably decreased compared to other animals starting on day 43. On days 43 and 120 (the last day of treatment), the RN6G plasma concentrations of this animal were 44% lower (938,000 ng/ml vs. 1,670,000 ng/ml) and >99% lower (46.5 ng/ml vs. 1,490,000 ng/ml) than the RN6G concentrations in other dosed animals, respectively.
ADA data
Anti-RN6G antibodies were detected in the serum of this female starting on day 43 through the end of the study. The ADA response persisted in this animal until the end of the study period, even though the last dose was administered on day 120.
Complement and cytokines
This female had changes in serum SC5b-9 and IL-6 over the time course of administered treatment. Although on days 1 and 15 small increases were observed in both IL-6 and SC5b-9, these changes were within the range of vehicle animals and returned to pretreatment values on day 29. Consequently, the relationship to test article was difficult to determine. Levels of SC5b-9 were increased on day 85 with peak concentrations detected on day 113. After cessation of dosing (day 120), SC5b-9 returned to pretreatment values from day 127 through study termination. IL-6 concentrations were elevated on day 57 with the highest concentrations measured occurring on day 85. From day 99 to day 127, IL-6 levels were higher than pretreatment; however, the trend was decreasing. After cessation of dosing (day 120, last dose administered) IL-6 levels returned to pretreatment below limit of quantification (BLQ) levels by day 127. All other complement and cytokine findings were considered normal fluctuations unrelated to ICD or RN6G administration (Figure 3).
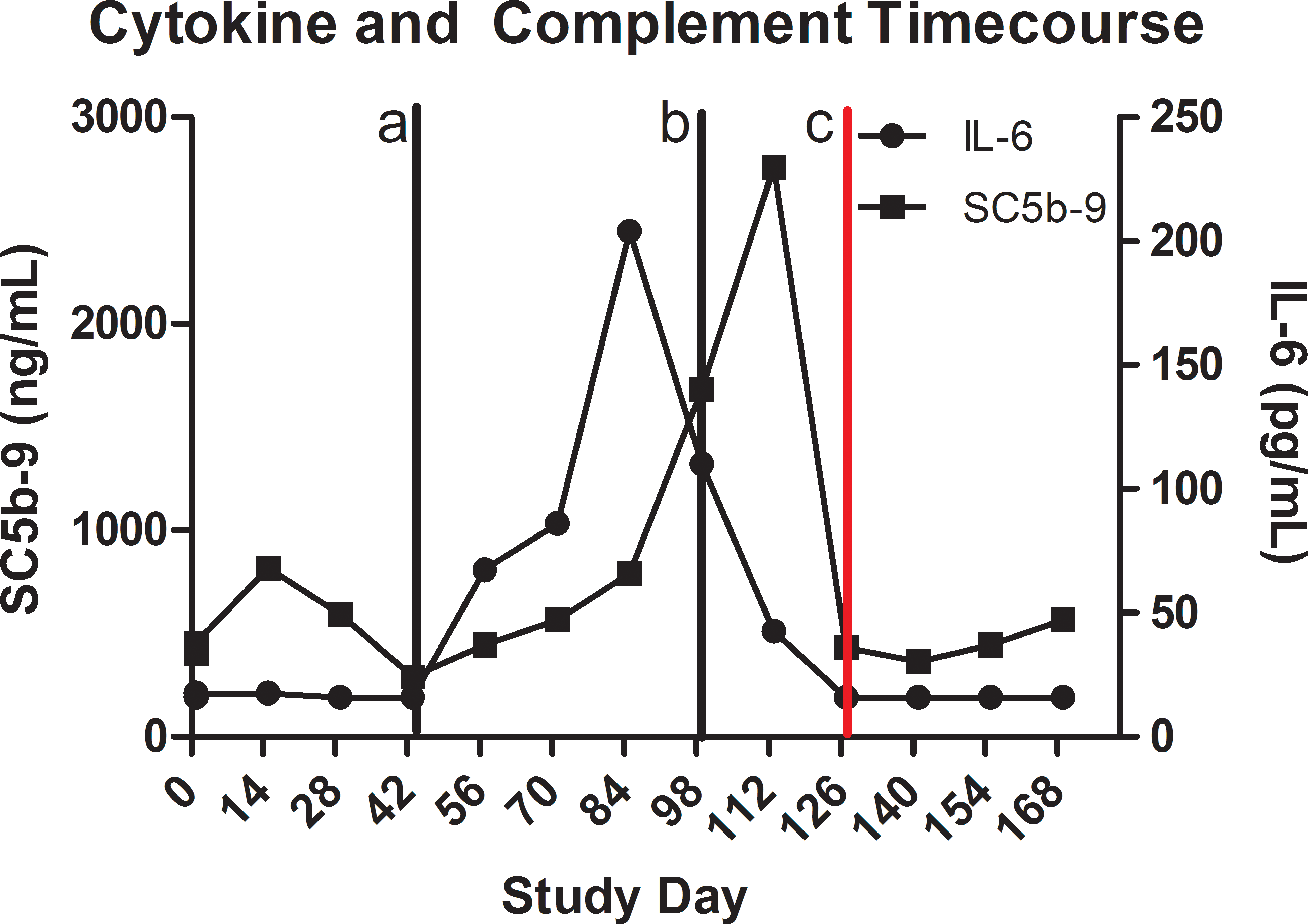
Time course changes of key clinical pathology biomarkers, complement (SC5b-9, square) and cytokines (IL-6, circle), from the female monkey in study 2 that developed immunogenicity in response to RN6G administration. Vertical lines represent onset of decreased plasma concentration of RN6G coupled with antidrug antibody formation on day 44 (a), decreased platelet counts on day 99 with prolonged PT and APTT (b), and the cessation of dosing on day 127 (c). PT = prothrombin time; APTT = activated partial thromboplastin time.
Anatomic pathology
There were no biologically significant or test article–related findings in organ weights, macroscopic observations, or microscopic observations. No microscopic findings to suggest ICD were observed in the animal with ADA at the end of the recovery period.
Discussion
The 2 studies summarized in this article describe analysis and interpretation of ADA and ICD in nonclinical toxicology studies. When the results of the first study were shared with regulatory authorities, regulatory scientists had concerns regarding the clinical safety of RN6G based on potential immunogenic responses leading to the potential development of ICD. The severe clinical signs in monkeys leading to euthanasia combined with the sporadic occurrence of such events combined with not having an early indicator to signal the moribundity and little knowledge of reversibility contributed to the regulatory agency’s concerns. A proactive investigational approach was employed based on discussion and collaboration with a regulatory agency to identify and track a set of biomarkers that may be useful in the early identification of ICD and the subsequent reversibility associated with any potential IC-mediated adverse findings. Although immunogenicity of a therapeutic antibody in nonclinical species is not predictive of immunogenicity in humans, the immune mechanisms and sequelae (if they occur) in response to a protein therapy are similar in humans and monkeys, and biomarkers that demonstrate these immune-mediated effects may translate well across species (Brinks, Jiskoot, and Schellekens 2011). The investigative study described in this article was designed to frequently monitor ADA, toxicokinetics, and biomarkers that would indicate an immune response to RN6G. Only 1 monkey in this investigative study developed ADA; however, all biomarkers that signaled ICD with the exception of ADA returned to near normal levels within 1 week of cessation of dosing in this animal. Upon submission of the data from the investigative study, the potential concerns from the regulatory agency were alleviated and the development of the program was continued.
IC are normally formed during an immune response; however, they are generally cleared by phagocytic cells without inciting tissue damage. IC disease, also known as type III hypersensitivity, results when antigen–antibody complexes either form directly within the tissues or form in circulation and deposit within tissues, eliciting an inflammatory response. The most important factor in determining whether a type III reaction will occur is the proportion of antibody to antigen, though other contributors to the development of this response also exist, including quantity, size, and charge of the complexes, and strength of binding between the antigen and antibody (McGavin and Zachary 2005).
The first study exemplifies a typical presentation of a drug-induced ICD in nonclinical safety studies where animals present with sudden, unexpected clinical signs in nondose responsive manner, resulting in unscheduled euthanasia. In this study, all findings support IC-mediated reaction with an alignment of the toxicokinetics (rapid drug clearance), ADA (presence of high titer anti-RN6G antibody), clinical pathology (thrombocytopenia, complement activation, and neutrophilia), microscopic findings (lack of any histological changes to indicate pharmacology-mediated toxicity but histological indication of IC deposition in predilected organs), transmission electron microscopy (TEM) (granular deposits in the glomeruli), and IHC (positive IC staining for test article, monkey IgG and IgM, and complement split products). The pathogenesis of ICD in this study is considered “classical” in that a humanized mAb (RN6G) served as a foreign protein in monkeys to elicit ADA formation, which complexed with RN6G resulting in sufficient size, valence, and charge to deposit in predilected vessels and tissues. The sudden and severe onset of the adverse clinical findings were likely due to activation of the complement system by ICs, resulting in the production of large amounts of C3a, C4d, and C5b-9 degradation products that can act as anaphylatoxins and induce release of chemical mediators such as histamine and leukotriene (Tawara et al. 2008; Rojko et al. 2014). Vasoactive amines can also increase glomerular permeability, resulting in enhanced deposition of immune complexes. In this study, the presence of decreased albumin, increased triglycerides, and azotemia (components of nephrotic syndrome) in affected animals provided strong antemortem evidence of glomerular disease. Platelet reductions may also have been in part due to activation by vasoactive amines. The data gathered from this study assisted in designing an investigational study to assess the early constellation of changes that may potentially signal IC-mediated reactions. Furthermore, determining the reversibility of these changes and the fate of eventual clinical status upon cessation of dosing was considered important in risk analysis.
In the requested follow-up investigational study, the 30-mg/kg dose previously associated with adverse findings resulted in RN6G ADA and ICD in only 1 animal suggesting that immunogenicity is difficult to predict. Furthermore, the outcome from the investigational study illustrated that, as expected, positive anti-RN6G antibody response and resultant reduction in exposure precede any clearly meaningful clinical pathology changes (such as reduction in platelets and coagulopathy, elevations in IL-6, and modulated complement split products) by several weeks. In the case of this particular animal, dosing was stopped and despite observed changes in ADA and biomarkers, there was a lack of any clinical signs. It is generally accepted that immunogenicity or ADA development does not always lead to an adverse finding, and the ultimate sequela of these adverse findings can be presented differently in individual animals and sometimes between individual doses for individual animals. However, these studies outline a few hallmark changes, such as coagulopathy and elevation of complement degradation factors, which occurred with IC-mediated reactions to this test article. Although recovery could be assessed in only 1 animal in the investigative study, cessation of dosing in that animal led to full reversal of these observed changes without tissue injury commonly associated with adverse immunogenic responses suggesting that immunogenicity was the key factor behind the observed pathological findings.
Immunogenicity in cynomolgus monkeys is not predictive for its incidence or severity of ADA or ICD in humans and may be overpredictive, however, because the clinical sequelae are similar to those in humans and can be utilized to monitor the occurrence of ICD in early clinical trials. As previously described in human reactions to therapeutic proteins or antibodies, the manifestations of ICD occur in a similar manner in monkeys and humans (Bugelski and Treacy 2004; Ponce et al. 2009; Brinks, Jiskoot, and Schellekens 2011). Consequently, serum biomarkers for complement components (C3a, SC-5b9), cytokines (IL-6), coagulation parameters (APTT, PT, and platelets), and serum albumin often provide acceptable markers for monitoring status of patients in early clinical trials. Antithrombin III, a protein that is close in size to albumin, may also be a useful biomarker to detect a developing glomerulopathy, as are more standard urinary biomarkers including urine protein, urine albumin, urine protein:creatinine ratio, and/or urine albumin:creatinine ratio. Based on the studies presented here, reduction in plasma concentrations (pharmacokinetics) of the therapeutic protein or antibody therapies, coupled with detection of ADA, is detected prior to ICD and should be considered as early markers for the potential development of a reaction. Clinical signs such as hypoactivity, excessive salivation, hunched posture, emesis, fast respiration, and recumbency during chronic monkey toxicology studies may provide the first indication of potential severe immunogenicity. Consequently, it may be advantageous to collect and analyze additional scheduled or unscheduled blood samples for TK/ADA and other biomarkers for early identification of the potential for ICD in chronic toxicology studies. Often all of these parameters are included as a clinical laboratory panel in early clinical trials with biological molecules, and if considered necessary, the evaluation would not be too onerous in clinical trials.
Footnotes
Acknowledgment
The authors would like to acknowledge Cris Kamperschroer for his thoughtful and critical review of the data and manuscript.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The work was supported and funded by Pfizer Inc.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
