Abstract
Conjugation with polyethylene glycol (PEG) is a strategy for improving the pharmaceutical properties of therapeutic proteins. In nonclinical studies of PEGylated compounds, microscopic tissue vacuolation is often observed, characterized ultrastructurally in this report by lysosomal distension. Although PEGylation-associated vacuolation appears to be of limited toxicologic concern when alternative therapies are limited, the risk-benefit considerations may be impacted by uncertainty about reversibility, lack of methods for monitoring PEG accumulation in vivo without biopsy, and the variability in tissues affected depending on species studied. We demonstrate the use of magnetic resonance spectroscopy (MRS) to measure PEG concentrations at multiple time points in vivo in the kidney with comparison to PEG concentrations ex vivo in body fluids and tissue extracts using nuclear magnetic resonance (NMR) spectroscopy. Furthermore, we demonstrate the use of these techniques to study distribution and elimination of PEG in a dog model of PEGylation-associated vacuolation. This report suggests that MRS could be further investigated as a feasible imaging-based method for monitoring PEG accumulation in a clinical setting in conjunction with NMR quantitation of PEG in plasma and urine.
Covalent attachment of polyethylene glycol (PEG) has been used to prolong plasma retention time, increase hydrodynamic size, decrease proteolysis, increase solubility, and/or shield immunomodulatory epitopes of more than a dozen approved pharmaceuticals (Davis et al. 1981; Pedder 2003; Turecek et al. 2016) as well as many experimental compounds (Abuchowski, McCoy et al. 1977; Abuchowski, van Es et al. 1977; Pyatak, Abuchowski, and Davis 1980; Ivens et al. 2015). Cytoplasmic vacuolation is commonly observed by light microscopy in nonclinical studies with PEGylated proteins (Conover, Lejeune, Linberg et al. 1996; Bendele et al. 1998; Rudmann et al. 2013; Ivens et al. 2015; Turecek et al. 2016) but has not been described in human tissue. In nonclinical studies, vacuolation is observed most often in phagocytic cells such as macrophages in the reticuloendothelial system (Conover, Lejeune, Linberg et al. 1996; Caliceti and Veronese 2003; Ivens et al. 2015) but also in diverse cell types such as synovial epithelium, adrenal cortical cells, choroid plexus, and renal tubular epithelial cells (Bendele et al. 1998; BLA #125160 2006; CHMP Safety Working Party of the European Medicinal Agency 2012; Gegg and Kinstler 2002; Rudmann et al. 2013; Ivens et al. 2015). The pattern of vacuolation observed for a given PEGylated therapeutic appears to depend on multiple factors including the route of administration, dose, duration of treatment, total molecular size, branching pattern of the PEG moiety, tissue receptor distribution and catabolic pathway of the non-PEG moiety, and the species to which the drug is administered (Yamaoka, Tabata, and Ikada 1994; Knauf et al. 1988; Lee et al. 1999; Edwards 1999; Caliceti and Veronese 2003; Bendele et al. 1998; Bauman 2014; Ivens et al. 2015). Empirically, use of low dose (µg/kg range) or of PEG substituents ≤20 kDa minimizes the likelihood of observing tissue vacuoles in nonclinical studies (Bauman 2014; Ivens et al. 2015) and also impacts cost of goods and marketing feasibility since the unit cost of pharmaceutical-grade PEG increases with molecular weight. PEG-associated tissue vacuolation in nonclinical studies is generally considered nonadverse when it occurs in characteristic cell types (such as phagocytes in the reticuloendothelial system), does not distort tissue architecture, and lacks evidence of pathological sequela (such as inflammation, degeneration, or hyperplasia) or impaired organ function (Ivens et al. 2015). The light microscopic appearance of PEG-associated vacuolation and the evident absence of eukaryotic enzymatic machinery capable of metabolizing PEG (Webster et al. 2007; Parton, King, Parker et al. 2009) combined with current understanding of intracellular catabolic pathways provides a strong weight of evidence that the vacuoles observed nonclinically are caused by accumulation of PEG in lysosomes (Baumann et al. 2014). This inference is confirmed in this report by ultrastructural evaluation of affected cells.
PEG-associated vacuolation is a predictable observation in nonclinical studies and, in many cases, is a nonadverse adaptive change of minimal toxicologic significance. However, in development programs with chronic dosing strategies, there can be interest in understanding the accumulation, distribution, and excretion of PEG in nonclinical models and in bridging that understanding to the clinical setting. Monitoring PEG levels in vivo or in biological samples by proton magnetic resonance (MR) can be advantageous due to the strong singlet peak at 3.7 ppm that is contributed by overlapping signals from ethylene oxide repeating units in PEG (Parton, King, van Asperen et al. 2008; Elliott et al. 2012; Baumann et al. 2014; Alvares et al. 2016, 2017). In addition, the nuclear magnetic resonance (NMR) signal intensity is linearly proportional to the amount of contributing nuclei in samples because all protons in an NMR sample experience the same electromagnetic excitation and NMR intensity is dependent upon the quantity of protons in samples (Xu, Klees et al. 2005; Xu, Sachs et al. 2006). Thus, the goal of this investigation was to explore the feasibility of using MR as a novel translational method of measuring PEG accumulation. This goal was accomplished by studying the use of NMR to measure PEG concentrations in biofluids and tissue extracts, and magnetic resonance spectroscopy (MRS) using a clinical strength magnetic resonance imaging (MRI) machine to measure the ratio of PEG to total choline in vivo in a dog kidney vacuolation model. PEGylation-associated vacuolation was produced by administering Compound X, a 38 amino acid protein conjugated at the C-terminus to a branched 40-kDa PEG moiety.
Methods
Animals, Drug Preparation and Administration, Clinical Pathology, and Histology
All animal experiments for this study were conducted in accordance with the Guide for the Care and Use of Laboratory Animals. All studies were conducted within a program accredited by the Association for Assessment and Accreditation of Laboratory Animal Care and approved by the research laboratories of Merck & Co., Inc. (West Point, PA), Institutional Animal Care and Use Committee.
Beagle dogs (Marshall BioResources, North Rose, NY) aged 30 to 31 weeks at study start were individually housed in stainless steel pens. Enrichment devices were provided to all dogs during the study. Dogs were provided approximately 350 g/day of PMI-Certified Canine Diet with additional food enrichment (biscuit) presented routinely to each dog. Food was withdrawn overnight prior to scheduled blood collections, during urine collections, and prior to MRI/MRS imaging sessions. Sprague-Dawley rats (Crl:CD(SD); Charles River Laboratories, Raleigh, NC) approximately 35 days old at study start were housed individually in suspended, stainless steel, wire bottom cages. Female and males rats received 16 g/day and 22 g/day, respectively, of PMI-Certified Rodent Diet. Food was withdrawn overnight prior to scheduled blood collections and necropsies. Dogs and rats were kept in environmentally controlled, high efficiency particulate air (HEPA)-filtered rooms with a 12-hr light/dark cycle. Water was available ad libitum.
Sterile dose formulations of Compound X diluted to 30 mg/ml in vehicle (3% mannitol, 75-mM NaCl, 6-mM sodium acetate, pH 5.0) were prepared and frozen until use. The formulation was thawed overnight prior to dosing. Samples of all dosing formulations were analyzed for concentration by high-performance liquid chromatography prior to the study start and after the end of the study and were within prespecified concentration limits demonstrating that the compound formulated in this vehicle was stable under the conditions of the studies.
Doses were calculated based on the total weight (protein + PEG) of Compound X and administered subcutaneously into the intrascapular area on a twice-weekly schedule. Dose groups described in this manuscript include a study of 2, 20, or 200 mg/kg of Compound X administered for 1 month to dogs (8 doses; n = 3/sex/dose) with toxicokinetics samples collected at 0- (predose), 2-, 4-, 8-, 24-, 48-, and 72-hr postdose on day 25. A second dog study was conducted with 120 mg/kg of Compound X administered on study days 4 and 7 to enable MRI–MRS imaging (n = 3), with blood concentrations determined from samples collected on days 1, 5, 8, 11, 13, and 15. A rat study was conducted with 10, 40, or 400 mg/kg administered for 1 month (8 doses; n = 12/sex/dose) with toxicokinetics samples collected at 0 (predose), 4, 8, 24, 48, 72, and 96 hr after the final dose. An exploratory study was conducted in female rats with 750 or 1,500 mg/kg of Compound X administered for 1 week (2 doses; n = 10/dose). Toxicokinetics in rats dosed with 750 mg/kg was measured in a separate study with samples collected at 2, 4, 8, 24, 48, 72, 96, 120, 144, 168, and 196 hr after dosing. In dogs, blood was collected from jugular vein; and in rats, blood was collected from the orbital sinus or the vena cava at necropsy.
Dogs and rats sacrificed at study termination were anesthetized and euthanized by exsanguination. Tissues collected for further analysis were fixed in 10% neutral-buffered formalin or flash frozen in liquid nitrogen in sterile polypropylene vials. Routine methods were used to prepare paraffin blocks of tissues and hematoxylin and eosin–stained 5-µm thick sections for light microscopic evaluation. A routine complete Good Laboratory Practice histomorphologic evaluation with an extensive tissue list suitable for enabling a clinical study was performed in the 1-month rat study and the 1-month dog study. In the 1-week exploratory rat study, the bone, bone marrow, heart, injection sites, kidney, liver, spleen, and uterus were evaluated. In the dog study conducted for imaging, histomorphologic evaluation was limited to kidney and spleen. In all cases, where imaging was performed or tissue levels of PEG were measured by NMR in the rat or dog, tissue from the same organ was evaluated histomorphologically.
Tissues prepared for transmission electron microscopy were transferred from 10% neutral-buffered formalin for postfixation in 4 parts paraformaldehyde to 1 part glutaraldehyde, embedded in LX-112 epoxy (Ladd Research, Williston, VT), sectioned at 90 nm, stained with 4% uranyl acetate and Reynold’s lead citrate, mounted on a copper mesh grid, evaluated with a Tecnai Spirit Biotwin electron microscope (FEI, Hillsboro, Or), and photographed with a digital camera system (Advanced Microscopy Techniques, Woburn, MA).
NMR
Frozen tissues from individual animals were weighed in the range of 30 to 100 mg per sample and homogenized in 1-ml ice-cold, 80% methanol with a Polytron PT 1200E (Kinematic AG, Switzerland). For rats, frozen kidney tissue samples were collected from the apex of the renal poles to maximize cortical sampling to best match tissue sample for analysis with the histologically vacuolated tissue compartment. The homogenized samples were centrifuged in an Eppendorf centrifuge 5415 R (Barkausenweg, Germany) at 12,000 rpm for 20 min at 10°C. The supernatants were lyophilized overnight. To the dried lysates, 700 µl of 80-mM potassium phosphate buffer (pH 7.0) with 2.02-mM deuterated 4,4-dimethyl-4-silapentane-1-sulfonic acid (DSS-d 6) in deuterium oxide (Isotec, Miamisburg, OH) was added. The solutions were mixed and centrifuged at 12,000 rpm for 20 min and 10°C. Aliquots of the supernatants (∼700 µl) were transferred to 4" × 5 mm NMR tubes (Wilmad Glass, NJ).
Urine samples (350 µl) were diluted with an equal volume of potassium phosphate buffer in 100% deuterium oxide at pH 7.0 (Isotec). The final diluted solutions contained 100-mM potassium phosphate with 2.5-mM DSS-d 6 and were transferred to 4" × 5 mm NMR tubes. Plasma samples (400 µl) were admixed with 2,000 µl of methanol, and the solution mixtures were centrifuged at 12,000 rpm for 20 min and 10°C. The supernatants were lyophilized overnight. To the dried lysates, 700 µl of 80-mM potassium phosphate buffer (pH 7.0) with 2.02-mM DSS-d 6 in deuterium oxide (Isotec) was added. The solutions were mixed and centrifuged at 12,000 rpm for 20 min and 10°C. Aliquots of the supernatants (∼700 µl) were transferred to NMR tubes. Deuterium oxide was used as the magnetic field lock, and DSS-d 6 was used as an internal chemical shift reference (0 ppm) and a quantification reference. The NMR tubes were loaded into 96-well NMR tube racks on a Varian768AS robot, and the samples were kept at 8°C.
One dimensional proton spectra were acquired on a Varian UnityInova 700 MHz NMR Spectrometer (Palo Alto, CA) equipped with a cold probe and an AS768 robotic system. Water suppression was achieved with a wet 1D pulse sequence. The probe temperature was set to 25°C. The 1D proton NMR spectral width was 10,000 Hz covering the range of −2.37 to 11.91 ppm. The acquisition time was 3 sec, corresponding to a digital resolution of 0.33 Hz. The water suppression with the wet 1D pulse sequence was based on the selective excitation of the water peak using SINC pulses and gradient dephasing. The selective excitation bandwidth (centered on the water peak) was 50 Hz for tissues and plasma and 100 Hz for urine. Transients of at least 128 scans were acquired with a relaxation delay of 15 sec. The time domain data were multiplied with an exponential decay function of 0.2-Hz line broadening. The data were zero filled to 64k data points before Fourier transformation. Chemical shifts were referenced to the internal DSS-d 6 resonance (0 ppm).
The quantification of PEG was based on the peak integration of the PEG signal (ca. 3.7 ppm) compared to that of the known amount of the internal reference DSS-d 6. The amount of PEG in tissues was calculated in mg/g tissue, in mg/ml in urine and plasma. Urine concentrations were multiplied by the total volume collected and expressed as mg/collection. The molecular weight of the repeating unit of PEG (44 Da) was used to convert the mole-based NMR quantification to the weight-based expression.
MRS
In rat, kidney poles were thawed at room temperature, and 1H-MRS measurements were performed at 37°C on a Bruker 7 T MR scanner (Bruker Biospin, Billerica, MA) using a vendor-supplied quadrature surface coil (RAPID Biomedical, Germany). After all first- and second-order shim currents were automatically adjusted on tissue water, 1H-MRS was acquired for PRESS-localized voxels placed in near the apex of each sample (presumed to be cortex). All acquisitions were <2 min. In dogs, on days 1, 5, 8, and 15, animals were induced with intravenous propofol, anesthetized with isoflurane, and scanned on a Siemens 3Y MR scanner (Siemens, Erlangen, Germany) using a vendor-supplied volume coil. The magnetic field homogeneity was optimized by automatic adjust of first- and second-order shim currents and manual optimization on tissue water. Water suppressed 1H-MRS was acquired based on point resolved spectroscopy (PRESS)-localized voxels placed in the upper right kidney pole of each dog. MR total study length ranged from 1 to 2 hr per animal. The free induction decay from rat and dog was processed with line broadening, and Fourier transformed to frequency spectra with phase correction. Peaks were fit with the jMRUI v3.0 software, and quantification of the PEG amount was obtained by making reference to the total choline peak in the spectra to create a ratio of PEG to choline. Total choline was selected as an internal spectral reference for normalizing the PEG peak because the choline peak is distinct in tissue spectra due to the presence of 9 chemically identical nonexchangeable (3 methyl groups) protons, because choline is a ubiquitous and abundant constituent of cell membranes, and because choline levels were expected to remain relatively constant with Compound X administration, which was confirmed by in vitro NMR measurements in the kidney extracts.
Results
Morphologic Characterization of PEGylation-Associated Vacuolation in Dogs
Vacuolation of the cytoplasm of renal tubular epithelial cells in the kidney of dogs administered 200 mg/kg of Compound X for 1 month was characterized by light and electron microscopy (Figure 1). Transmission electron microscopy demonstrated that the change was primarily in proximal tubular epithelium and associated with lysosomes. In affected cells, lysosomes were evident based on their cytoplasmic location; membrane delineating the organelle from the cytoplasm; and the typical size, shape, and characteristic electron-dense portion of their content. Affected lysosomes often contained scant to moderate amounts of relatively electron lucent flocculent material, which eccentrically marginated the electron-dense content. Similar ultrastructural changes in lysosomes have been described for PEGylation-associated vacuolation in the rat choroid plexus (Rasmussen et al. 2016). Membranes and other organelles typical of proximal renal tubular epithelium were not remarkable (Figure 1). The renal tubular epithelial vacuolation was not associated with pathological sequela such as inflammation, degeneration, or hyperplasia.
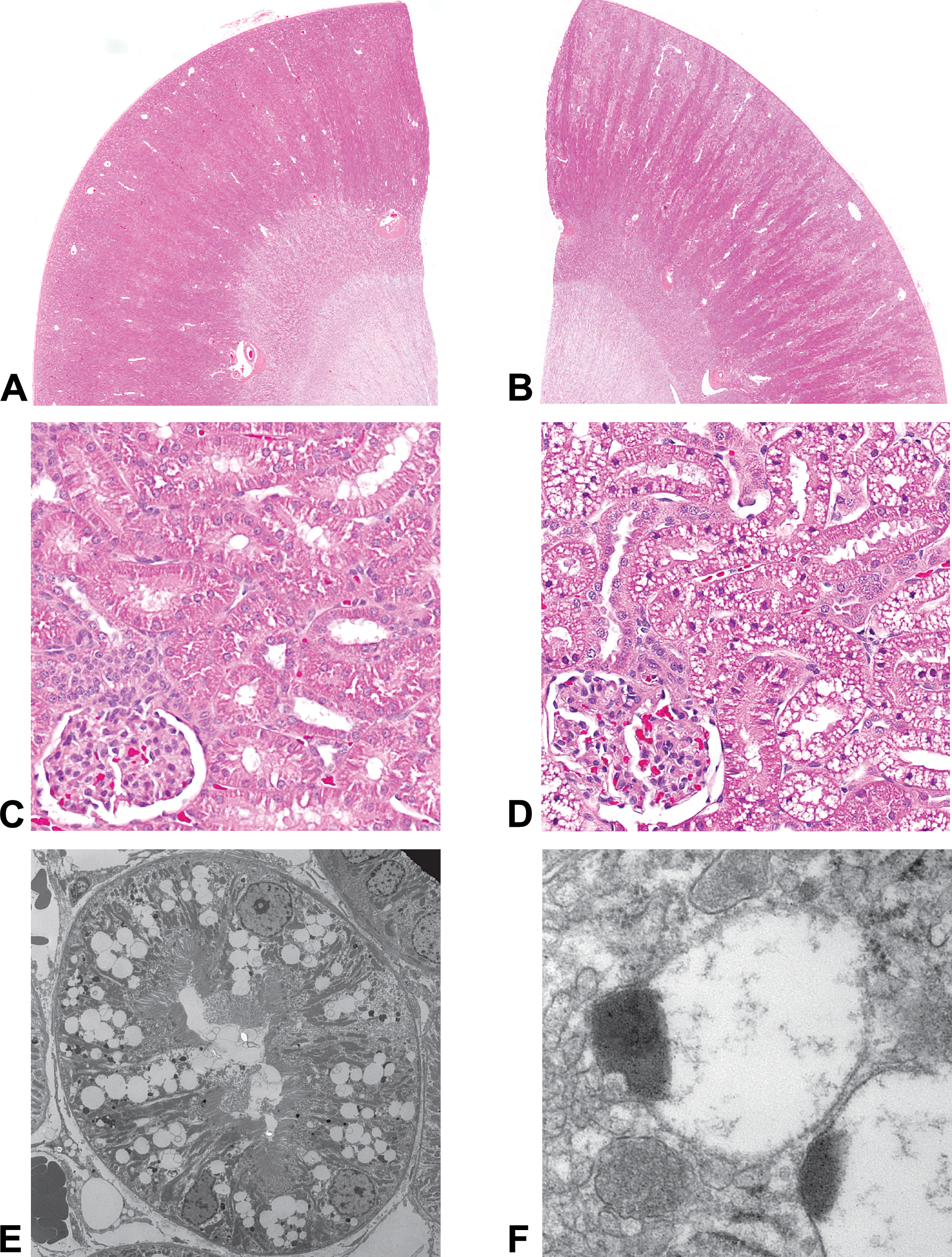
Panels A and C illustrate the histomorphology of control (vehicle) dog kidney using 0.5× and 20× objectives, respectively. Panels B and D illustrate representative renal cortical tubular vacuolation in a dog administered Compound X. In panel B, the renal cortex is tinctorially lighter compared to the cortex in panel A. Panel C illustrates the unstained cytoplasmic vacuoles in the cytoplasm of cortical tubular epithelial cells. Panel E illustrates the ultrastructure of the vacuolar change (direct magnification = 1,900×). Panel F illustrates the characteristic eccentrically placed electron-dense structures within most vacuoles, the electron lucency of the remaining portion of the vacuole, and the membrane delineating the vacuole from the cytoplasm (direct magnification = 68,000×).
Species Differences in Tissue Vacuolation Profile with Compound X
Cytoplasmic vacuolation of renal tubular epithelial cells occurred in dogs administered 20 mg/kg or greater amounts of Compound X for up to 1 month. In affected dogs, vacuolation was also present in histiocytic cells in the dermis at injection sites, but no other tissues were vacuolated (Table 1). In contrast to dogs, vacuolation did not occur in the kidney in rats administered up to 400 mg/kg for 1 month. At these doses, in rats, nonrenal vacuolation occurred in compartments such as the interstitium of many tissues and locations rich in reticuloendothelial cells such as the spleen (Table 1). The difference in vacuolation pattern between dogs and rats was also evident when considered on the basis of blood exposure. In dogs, the distribution pattern involving kidney was observed in an exposure range up to Area Under Curve (AUC)0–72 hr = 2,680 µM · hr. In contrast, in rats, a pattern of nonrenal vacuolation was observed with an exposure up to AUC0–96 hr = 1,440 µM · hr. Renal vacuolation with a morphologic appearance and distribution like that observed in dogs was observed in rats administered 750 mg/kg for 1 week (AUC0–196 hr = 5,820 µM hr). Based on mg/kg dosing and systemic exposure, dogs were more sensitive to renal vacuolation than rats, and the sensitivity of nonrenal tissues to vacuolation also differed between dogs and rats.
Species Differences in Polyethylene Glycolation–associated Vacuolation.
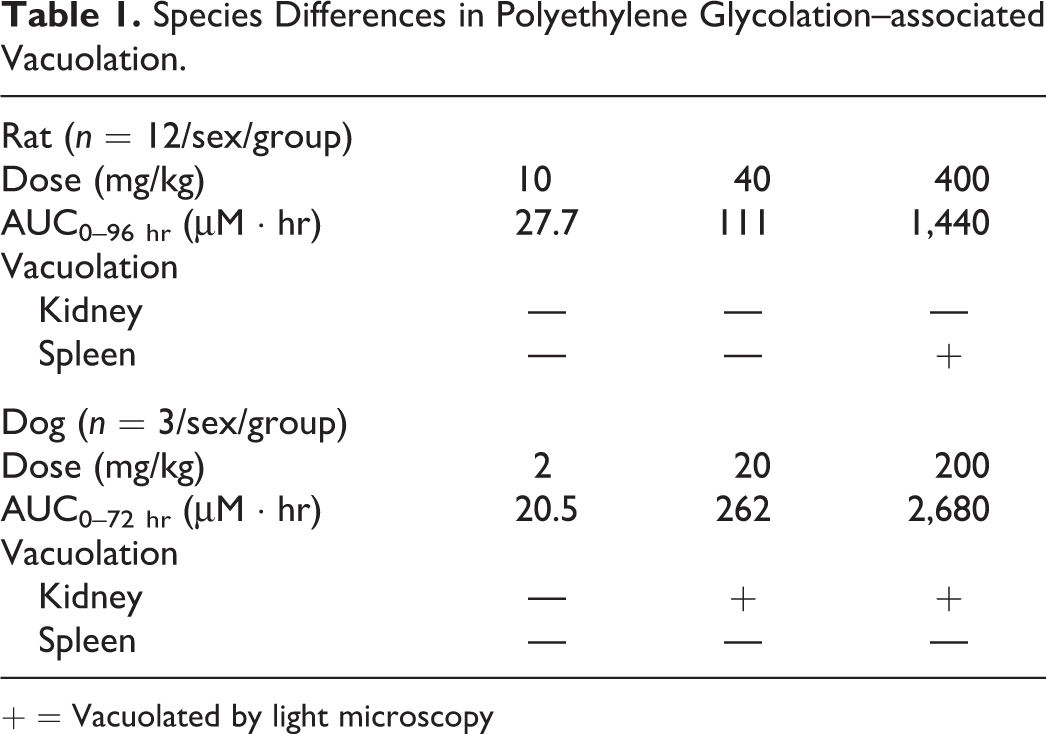
+ = Vacuolated by light microscopy
MR Properties of Compound X
Compound X is a 38 amino acid protein conjugated to a 40-kDa branched PEG polymer. The PEG portion of the molecule contains many magnetically equivalent protons (Figure 2, inset), which produce a distinct singlet MR peak at 3.7 ppm, with a signal amplitude much greater than any other protons on the molecule (Figure 2, main panel). The peak at 3.7 ppm can be used to identify and quantify Compound X to nM sensitivity. A mixture of different length PEG polymers in the range of sizes used for pharmaceutical conjugation could not be resolved by this resonance method due to the magnetic equivalence of the protons on polymers of different lengths.
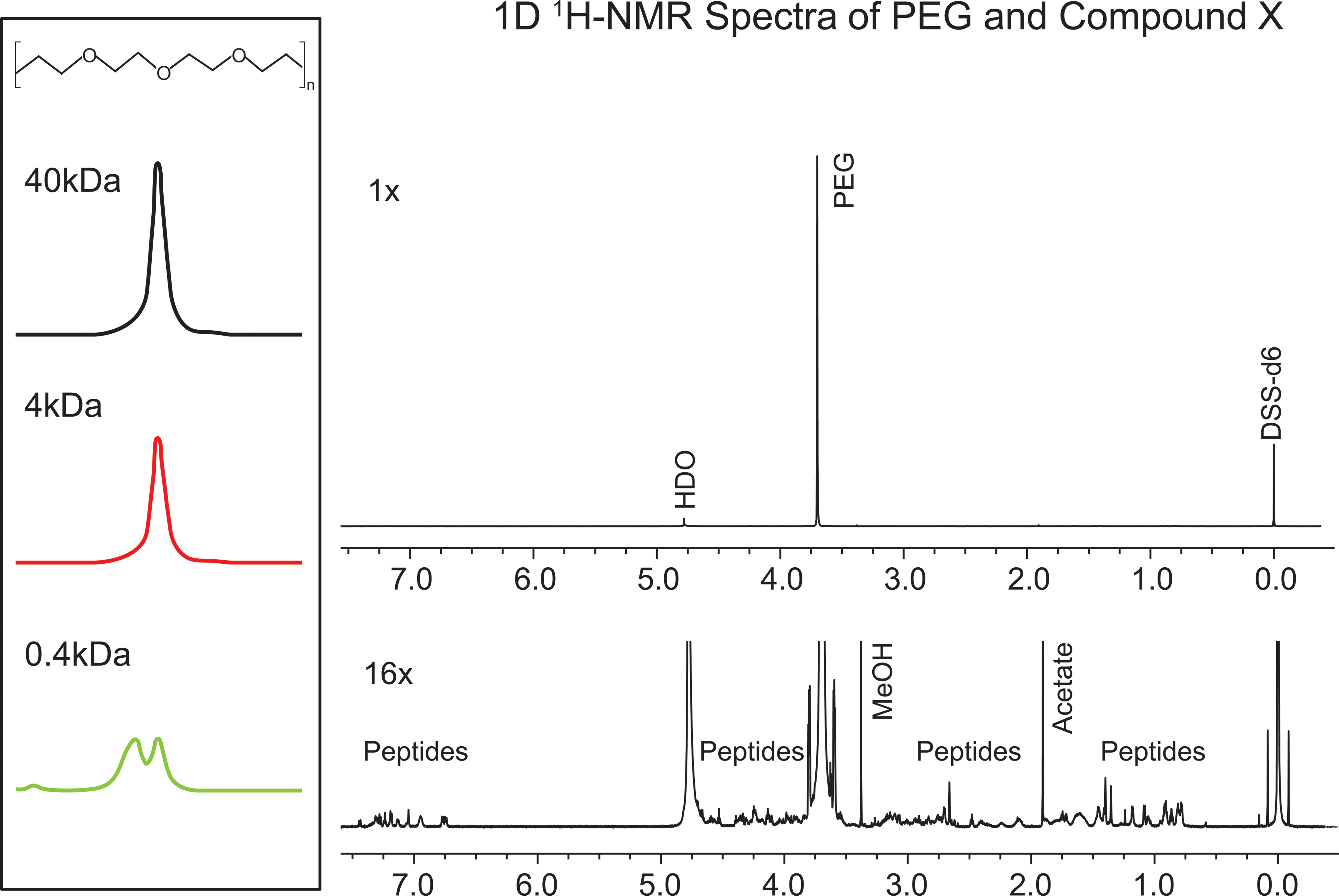
Insert (left): Structure of polyethylene glycol (PEG) polymer segment illustrating the arrangement of identical protons facilitating 1D 1H-nuclear magnetic resonance (NMR) observation and analysis, and 3 spectra illustrating the magnetic resonance frequency shift peak at 3.7 ppm produced by unconjugated PEG polymers of various lengths. Main panel: 1× and 16× NMR spectrum of Compound X illustrating the magnitude of the 40-kDa PEG polymer peak at 3.7 ppm compared to peaks contributed by the peptide portion.
Comparison of Microscopic Vacuolation, NMR, and MRS in Rat
Rats administered 750 or 1,500 mg/kg of Compound X for 1 week had light microscopic PEG-related renal cortical vacuolation, and as anticipated, the change was observed in the cytoplasm of renal cortical epithelial cells but not in the renal medulla in the same pattern of distribution observed in dogs. Associated with the light microscopic findings of tissue vacuolation in the renal cortex, there was trend toward an increased concentration of PEG in extracts of renal cortex by 1H-NMR (Figure 3, panel A). Levels of choline in the renal cortex of rats were not significantly affected by treatment (p = .12; Figure 3, panel B). There was a strong correlation (p < .0001) between PEG concentrations in tissue extracts measured by NMR and the ratio of PEG to total choline measured by ex vivo 1H-MRS (Figure 3, panel C). Based on these results, renal cortical choline levels are not significantly altered by treatment with Compound X and MR methods appear to be able to detect PEG ex vivo in tissues with no detectable histologic vacuolation.
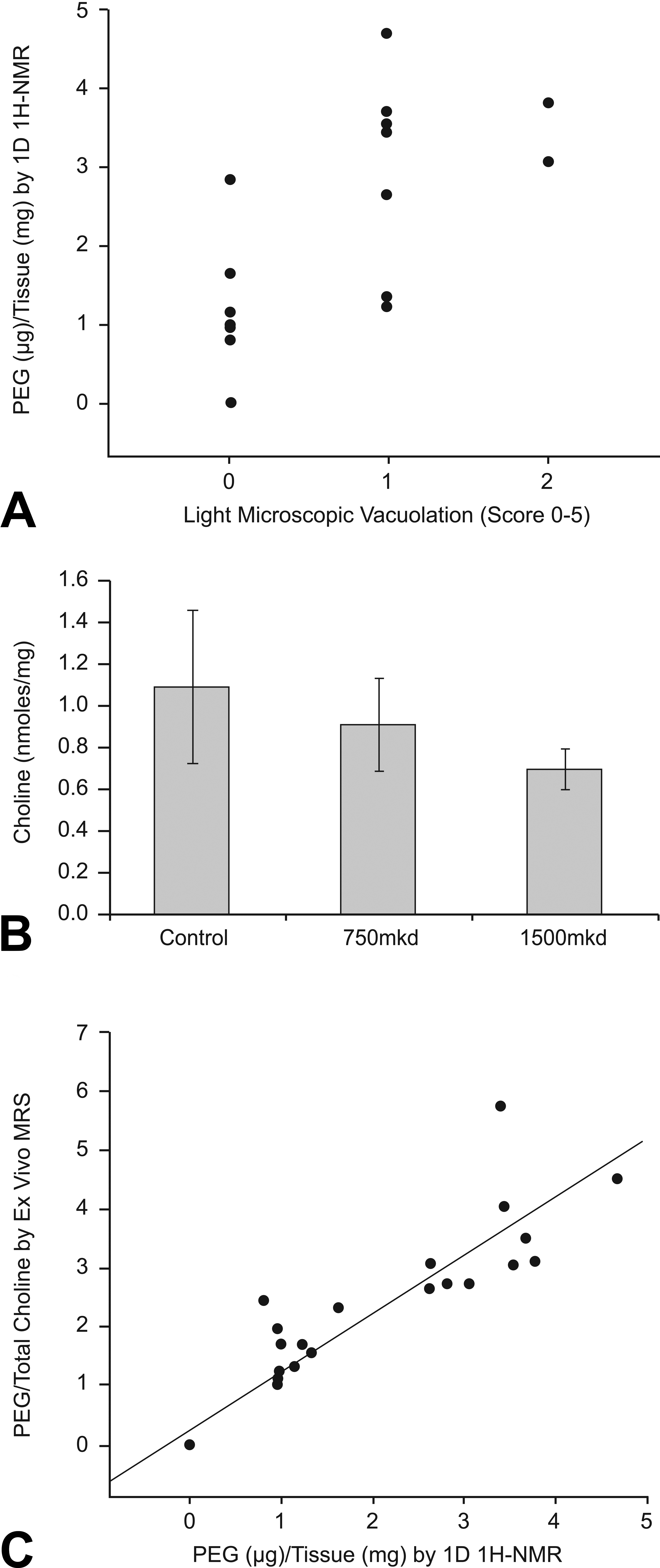
Panel A: Comparison of qualitative renal cortical vacuolation histology (score 0–5) against polyethylene glycol (PEG) concentration in kidney tissue (µg/mg) measured by 1D 1H-nuclear magnetic resonance (NMR) illustrating a trend toward higher histology scores in rats with greater tissue concentrations of PEG. Panel B: Choline per weight of tissue in rat renal cortex (nmoles/mg) was not statistically different between dose groups (p = .12). Panel C: Correlation plot of PEG concentration in rat kidney cortex measured by 1D 1H-NMR and the PEG/total choline ratio measured by 1H-MRS. Pearson’s correlation was significant for MRS and NMR (0.9247, p < .0001).
In Vivo PEG Accumulation and Elimination in Dog
To study changes in PEG concentration in vivo over time (longitudinally) with a 3-T clinical MR imager, dogs were administered 120 mg/kg of Compound X on study days 4 and 7. Measurement of PEG by 1H-NMR after necropsy demonstrated greater accumulation of PEG in the renal cortex than the renal medulla (∼20×) or spleen (∼120×) consistent with the anticipated distribution of renal cortical cytoplasmic vacuoles by light microscopy (Figure 4, panel A). Furthermore, the amount of PEG present in renal cortex, renal medulla, and spleen measured by 1H-NMR expressed as PEG concentration per tissue choline concentration correlated well with PEG concentration per gram of tissue (R 2 = .9903; Figure 4, panel B). By 1H-MRS, there was an increase in renal cortical PEG concentration with repeat dosing (Figure 4, panel C).
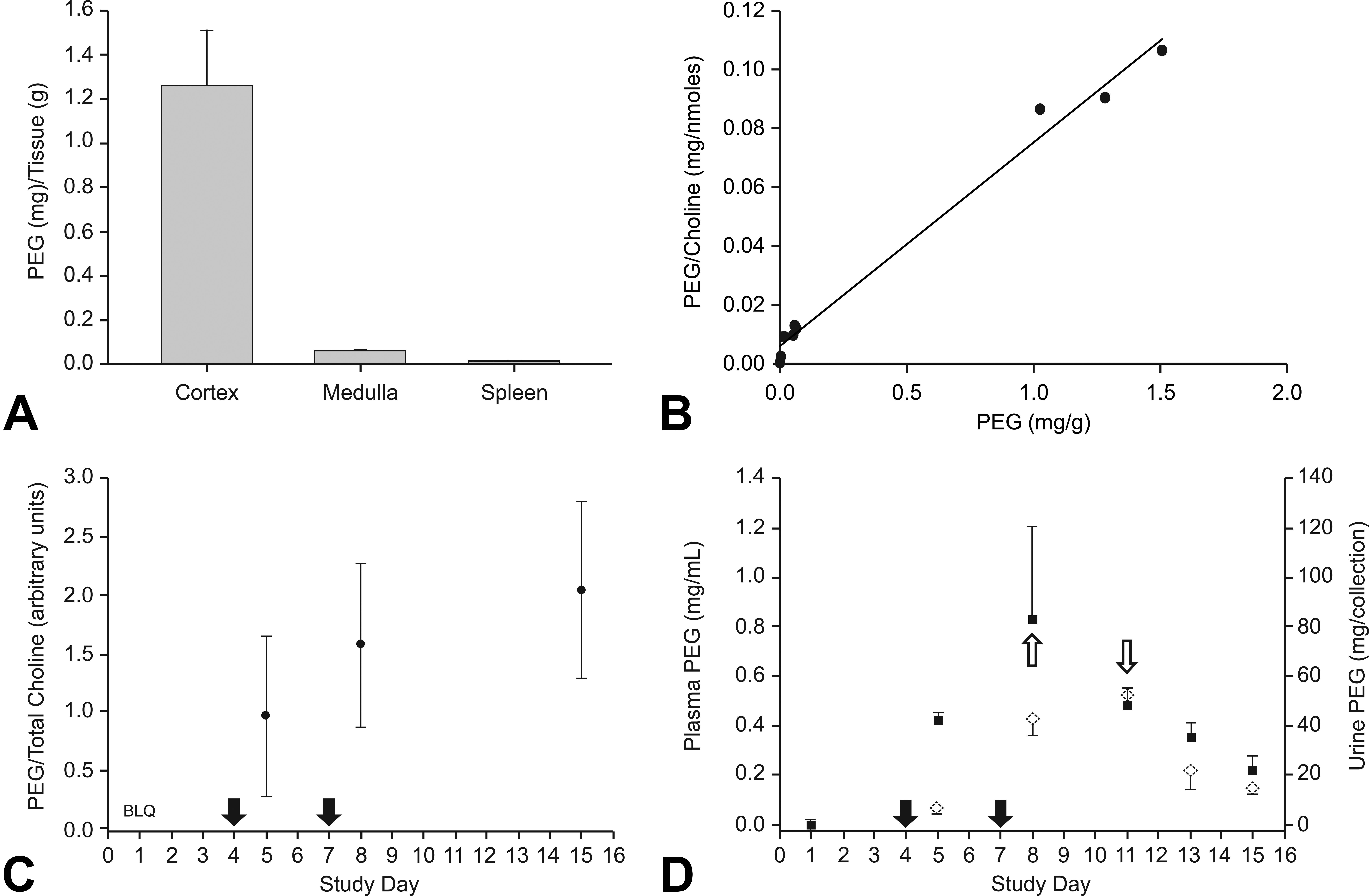
Panel A: Mean ± SD of polyethylene glycol (PEG; mg) per tissue sample (g) of renal cortex (cortex), renal medulla (medulla), and spleen in dogs measured by 1H-nuclear magnetic resonance (NMR) on study day 15 after administration of 2 doses of Compound X (black arrows in panel C and D), illustrating differences in PEG concentration between renal cortex, renal medulla, and spleen. Panel B: PEG concentration measured by 1H-NMR in dog renal cortex, renal medulla, and spleen was similar whether normalized to tissue choline or to tissue weight (R 2 = .9903). Panel C: Mean ± SD of PEG per total choline in serial measurements by 1H-MRS in vivo in dog kidney, illustrating an increase in renal PEG following repeat dosing. Panel D: Elimination profiles of PEG in plasma and urine by 1H-NMR. PEG concentration peaks in plasma (open arrow) on study day 8, before peaking in urine (open arrow) on study day 11.
In serial urine and plasma samples, PEG concentration by 1H-NMR peaked in plasma on study day 8 immediately following administration of the second dose of Compound X (study day 7) and peaked in urine on study day 11 lagged behind the peak plasma concentration (Figure 4, panel D). It is noteworthy that the mean renal cortical tissue concentration measured by 1H-MRS was greater on day 15 than on day 8; however, for practical reasons, no 1H-MRS data were collected on day 11; therefore, it was not possible to determine whether peak concentration in renal cortex coincided with peak urine concentration.
Discussion
Compound X, a 38 amino acid protein conjugated at the C-terminus to a branched 40-kDa PEG moiety, produced cytoplasmic vacuolation of minimal toxicology significance in the rat and dog in multiple tissues including the spleen and renal cortex. Tissue distribution of vacuoles was species and dose dependent. Ultrastructural characterization in proximal renal tubular epithelium was consistent with PEG accumulation in lysosomes consistent with the preexisting weight of evidence, regarding the affected subcellular compartment (Baumann et al. 2014; Ivens et al. 2015).
In addition to the species-specific and tissue-specific variability in sensitivity to vacuolation we observed with Compound X, other investigators describe compound-specific differences with regard to potential for vacuolation, although cytoplasm of renal tubular epithelial cells is a commonly encountered manifestation of the finding (Bendele et al. 1998; Caliceti and Veronese 2003; Baumann et al. 2014; Ivens et al. 2015). The specific distribution of PEGylation associated tissue vacuoles is hypothesized to depend on the molecular size of the molecule administered and the cellular binding target of the protein portion of the molecule (Yamaoka, Tabata, and Ikada 1994; Conover, Lejeune, Linberg et al. 1996; Nakaoka et al. 1997; Bendele et al. 1998; Caliceti and Veronese 2003; Baumann et al. 2014). In this model of tissue vacuolation, molecular size exclusion from glomerular filtration in the range of 30 kDa limits passage of PEG conjugates into the glomerular filtrate and thus limits contact of the drug with the apical surface of renal tubular epithelial cells. Therefore, urinary elimination of the conjugate is limited, and the renal tubular vacuolation phenotype is not observed (Baumann et al. 2014). For conjugates above the 30-kDa size exclusion threshold, vacuolation is more likely to be observed in phagocytic cells such as those present in the spleen or observed in the cytoplasm of cells where the protein portion of the molecule is internalized (Baumann et al. 2014).
Our data from 1-month studies demonstrate a difference in the light microscopic vacuolation phenotype between the rat and the dog at equivalent plasma exposures (observed in dog kidney from 262 to 2,680 µM · hr; observed in rat spleen at 1,440 µM · hr). If the renal filtration size exclusion model is a relevant explanation of renal and nonrenal tissue vacuolation associated with PEG-conjugates, then the difference we observed in between dog and rat with Compound X may reflect differences in renal physiology between species. Given that vacuolation was not observed in the tissue compartment with the highest known Compound X receptor binding (data not shown), it seems unlikely that for Compound X the differences observed between dogs and rats in tissue vacuolation were primarily driven by pharmacologic receptor interactions. Regardless of the reasons for these differences, the presence of a species difference in vacuolation phenotype with Compound X suggests that species selection could be relevant to clinical risk assessment of PEG conjugates and that there is potential use for translational methods of monitoring PEG levels in tissue.
Some of the physical properties of PEG that make it suitable for protein conjugation create bioanalytical challenges. For example, PEG is water soluble, has low chemical reactivity, and is poorly immunogenic; and therefore, the development of chemical- or antibody-based analysis techniques has been slow (Rudmann et al. 2013). In our laboratory, it has not been possible to convincingly stain PEG-associated tissue vacuoles immunohistochemically (IHC) using commercially available anti-PEG antibodies (data not shown). Difficulty with use of IHC approaches for study of PEG-associated tissue, vacuoles may relate to the polar solvents and detergents typically used in the preparation of IHC specimens. Bioanalysis of PEG conjugates has most often been investigated using radiolabels, which creates complexity in metabolic studies related to label retention after the protein portion of the conjugate is metabolized and due to the effects of radiolysis on the length of the PEG portion of the molecule (Baumann et al. 2014). Artifactual fragmentation of PEG is particularly relevant to urine evaluation since metabolism of high molecular weight PEG is negligible (Webster et al. 2007; Parton, King, Parker et al. 2009), and renal glomerular filtration of PEG is likely dependent on molecular size (Yamaoka, Tabata, and Ikada 1994). Use of MR to measure PEG concentrations in biological matrices addresses some of the challenges of PEG bioanalysis, and the use of 1H-NMR has been described for this purpose by other investigators in nonclinical and clinical applications (Parton, King, van Asperen et al. 2008; Parton, King, Parker et al. 2009; Elliott et al. 2012), where ELISA and 1H-NMR were described as providing comparable measures of drug concentration in clinical plasma samples (Parton, King, Parker et al. 2009).
We elected to use total choline as an internal reference to normalize 1H-MRS measurements of PEG content in tissue in part because it has been commonly used for this purpose by other investigators (Kreis 1997). Choline is a common choice for internal reference in 1H-MRS studies because it is a ubiquitous and abundant constituent of cell membranes with a distinct and relatively large peak. Creatinine or tissue water content is also commonly used to normalize 1H-MRS data because, in most tissues, creatinine and water concentrations are relatively constant (Kreis 1997; Blüml 2013). However, in this experiment, we were uncertain how the test article might affect renal cortical creatinine or water concentrations, and therefore, we elected to use tissue choline. We determined by 1H-NMR analysis of kidney tissue extracts that choline levels were not significantly affected by treatment with Compound X. Normalizing PEG signal intensity by using an internal spectral reference creates a unit-less ratio rather than an absolute tissue concentration, and therefore, the 1H-MRS-based tissue measurements are designated as having arbitrary units. While tissue choline was suitable for the analysis performed in these experiments, it may be less ideal as an internal reference in other applications such as the analysis of models of neoplasia where differences in tumor choline content have been described (García-Figueiras et al. 2016).
The increasing PEG concentration in blood observed in dogs over several days following repeat administration of Compound X is consistent with slow release from a subcutaneous deposit, and the further increase after a second dose is consistent with drug accumulation, which is typically observed when frequency of administration is less than plasma half-life (Swartz 1997). The presence of PEG in urine and in renal tubular epithelial cells is consistent with free filtration of PEG through the dog glomerulus, which would not be anticipated based on rat data generated with PEG molecules in the molecular weight range (40 kDa) of the PEG conjugate used to synthesize Compound X (Yamaoka, Tabata, and Ikada 1994; Conover, Lejeune, Linberg et al. 1996; Nakaoka et al. 1997; Bendele et al. 1998; Caliceti and Veronese 2003; Rudman 2013) but has been previously described in the dog with lower (5 kDa) molecular weight PEG conjugates (Conover, Lejeune, Shum et al.1997). Based on the hypothesis that PEG appears in urine due to plasma filtration by the glomerulus and the observation that urine PEG concentration peaked after serum concentration, we speculate that a tissue compartment other than serum made a net contribution to urinary PEG after the peak in serum concentration. The most likely source of increasing urine PEG concentration was renal cortical tubular epithelial cells since these cells are in physical contact with glomerular filtrate and the renal cortex contains large amounts of PEG compared to other tissues.
The increased mean renal cortical PEG by 1H-MRS observed on study day 15 compared to study 8 seems, at first glance, to conflict with the decrease observed on study day 15 in urinary PEG concentration; however, for practical reasons, no 1H-MRS data were collected on study day 11, when renal cortical PEG concentrations may have been greater than on study day 15. Future studies of this type would benefit from 1H-MRS data collection closer to the urine PEG concentration maximum. Given the effort needed to collect MRS/MRI data, a pilot study focused on plasma and urine measurements by 1H-NMR may be most efficient in such studies.
MR methods proved useful and complementary means of quantitating PEG concentrations in vivo and ex vivo in accessible biofluids or tissue extracts, and have potential as clinically useful markers for PEG accumulation and elimination, as shown in the dog model. PEG is a widely used excipient and food additive (European Food Safety Authority 2007), and low molecular weight PEG can be absorbed from the gastrointestinal tract (Donovan, Flynn, and Amidon 1990), and therefore, PEG is likely routinely present in human tissues. Toxicologic concern is therefore not related to the presence of PEG in human tissues but rather is focused on the histologic vacuolation observed following parenteral administration of high molecular weight PEG, particularly, when vacuoles are present in the central nervous system (CHMP Safety Working Party of the European Medicinal Agency 2012). Our results in the rat kidney show that MR methods are capable of detecting PEG below the threshold for light microscopically identifiable tissue vacuolation. Therefore, MR could have utility as a translational method for measuring PEG conjugates where a nonclinical plasma exposure is identified that is not associated with light microscopic tissue vacuolation and a corresponding nonclinical tissue concentration ratio has been established by 1H-MRS or in the special case of renal tubular epithelium possibly by 1H-NMR of urine. In such a scenario, clinical 1H-MRS could be used to confirm that tissue concentration ratios of PEG remain at a level below that associated with nonclinical light microscopic tissue vacuolation. For compounds with renal elimination of PEG, a combination of 1H-MRS of kidney tissue and 1H-NMR of urine could be used in an animal model of PEG metabolism to establish in vivo PEG accumulation limits at the nonclinical no adverse effect level.
In contrast to other analytical methods currently available, MR techniques offer a straightforward analytical method for measuring PEG levels in biological samples such as blood and urine and a noninvasive methodology for potentially measuring PEG levels in difficult to access tissue compartments. However, the relationship between detection of PEG by MR and histomorphologic identification of tissue vacuolation likely varies depending on the affected tissue and compound studied, and therefore, analytical relationships would need to be established for each potential use.
Footnotes
Authors’ Note
Rose-Ann Blenman-Abange conducted the MRI portions of these experiments but was not available to review and approve of the manuscript.
Authors’ Contribution
Authors contributed to conception or design (TF, QX, SX); data acquisition, analysis, or interpretation (TF, QX, SK, HV, KV, WG, CH, SX); drafted the manuscript (TF); and critically revised the manuscript (TF, QX, SK, HV, KV, WG, CH, SX). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
