Abstract
Chronic prostatitis characterized on light microscopic examination by moderate, multifocal, predominantly lymphocytic inflammation associated with epithelial atypia and intranuclear and cytoplasmic inclusion-like material was identified in the prostate gland of 2 Wistar Han rats administered an immunomodulatory test article in a 6-month chronic toxicity study. Transmission electron microscopy of the prostate glands identified 45-nm, nonenveloped, icosahedral virions arranged in paracrystalline array within the cell nuclei in 1 of the 2 rats. The size, shape, location, and array pattern were most consistent with a polyomavirus. The light and electron microscopic findings after immunosuppression in our case have a resemblance to a polyomavirus recently reported to affect prostate gland epithelium in a colony of immunocompromised X-linked severe combined immune deficiency rats. To the best of our knowledge, this is the first report of light and electronic microscopic lesions in the reproductive tract associated with polyomavirus following chronic immunosuppression in a widely used, wild-type Wistar Han rat.
The Polyomaviridae comprises a family of nonenveloped, DNA-based, 40- to 45-nm icosahedral viruses that infect mammals, birds, fish, and insects (Buck et al. 2016). Subclinical infection is considered to be widespread in many mammalian species, including humans, and may be associated with disease in immunocompromised hosts. In stable colonies of laboratory mice, polyomavirus infection is considered to be rare and not associated with recrudescent disease in adult, immune competent animals (Barthold, Griffey, and Percy 2016). To the best of our knowledge, disease associated with polyomavirus has not been reported in adult rat strains commonly used in routine toxicity studies. The current report describes light and electron microscopic characteristics of prostatitis attributed to polyomavirus in a group of immunosuppressed Wistar Han rats administered an immunomodulatory drug for 6 months in a routine chronic toxicity study.
Material and Method
Male and female Wistar Crl: WI(Han) rats, 6 to 7 weeks old at the initiation of dosing, were assigned to 4 groups and administered vehicle control or 1 of 3 dose levels of an immunomodulatory test article via oral gavage once daily for 6 months. Male and female rats were housed individually in conventional open top polycarbonate cages for the duration of the study. Animals were checked twice daily for mortality, abnormalities, and signs of pain or distress. At the end of the 6 months, a full tissue list consistent with good laboratory practice was collected, and selected organ weights and macroscopic observations were recorded at necropsy. Collected tissues were formalin-fixed and embedded in paraffin; and hematoxylin and eosin–stained sections were evaluated by a board-certified veterinary pathologist. Routine clinical chemistry and hematology parameters were analyzed on blood samples collected from rats on days 92 and 183 of the study, and routine urinalysis was performed on urine collected at necropsy. The in-life portion of the study was conducted in a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care, and all procedures were conducted in compliance with applicable animal welfare acts and were approved by the local Institutional Animal Care and Use Committee.
For transmission electron microscopy (TEM), minced samples of formalin-fixed prostate gland and/or deparaffinized, rehydrated sections of formalin-fixed paraffin embedded (FFPE) prostate gland from 2 control males and 2 males exhibiting microscopic evidence of viral prostatitis were further fixed in phosphate buffered 2% formaldehyde and 2.5% glutaraldehyde, postfixed in phosphate buffered 1% osmium tetroxide, and processed using epoxy resin and embedded. Thick sections (0.6 μm) were prepared for light microscopic evaluation. Thin sections (90 nm) from select tissue blocks were then prepared for electron microscopic evaluation on a Hitachi H-7100 TEM.
Results
On light microscopic examination, 2 of the 15 male rats administered the high dose of an immunomodulatory test article in this 6-month chronic toxicity study exhibited moderate, multifocal, predominantly lymphocytic inflammation in the prostate gland that was associated with prominent atypia of epithelial cells lining involved glands (Figure 1). Affected epithelial cells were enlarged, abnormally shaped, and contained nuclei that were expanded by stippled or glassy amphophilic–basophilic inclusion material. Increased numbers of enlarged, degenerate epithelial cells containing similar nuclear and/or cytoplasmic material were present within glandular lumens. There were no associated histologic alterations noted in other tissues of the urogenital tracts in either animal, nor in the salivary glands or airways. There were no macroscopic findings in the prostate gland at necropsy, and prostate gland weights for these 2 animals were comparable to the other animals in the same dose group and to the concurrent control rats. The light microscopic findings were also not associated with alterations in urinalysis, nor were there related clinical signs. No comparable lesions were identified in males from lower dose groups, nor any observed in the reproductive tract in any of the females in this study. Specific fertility assessments were not performed in the course of this study, and thus potential effects on reproductive fecundity were not assessed.
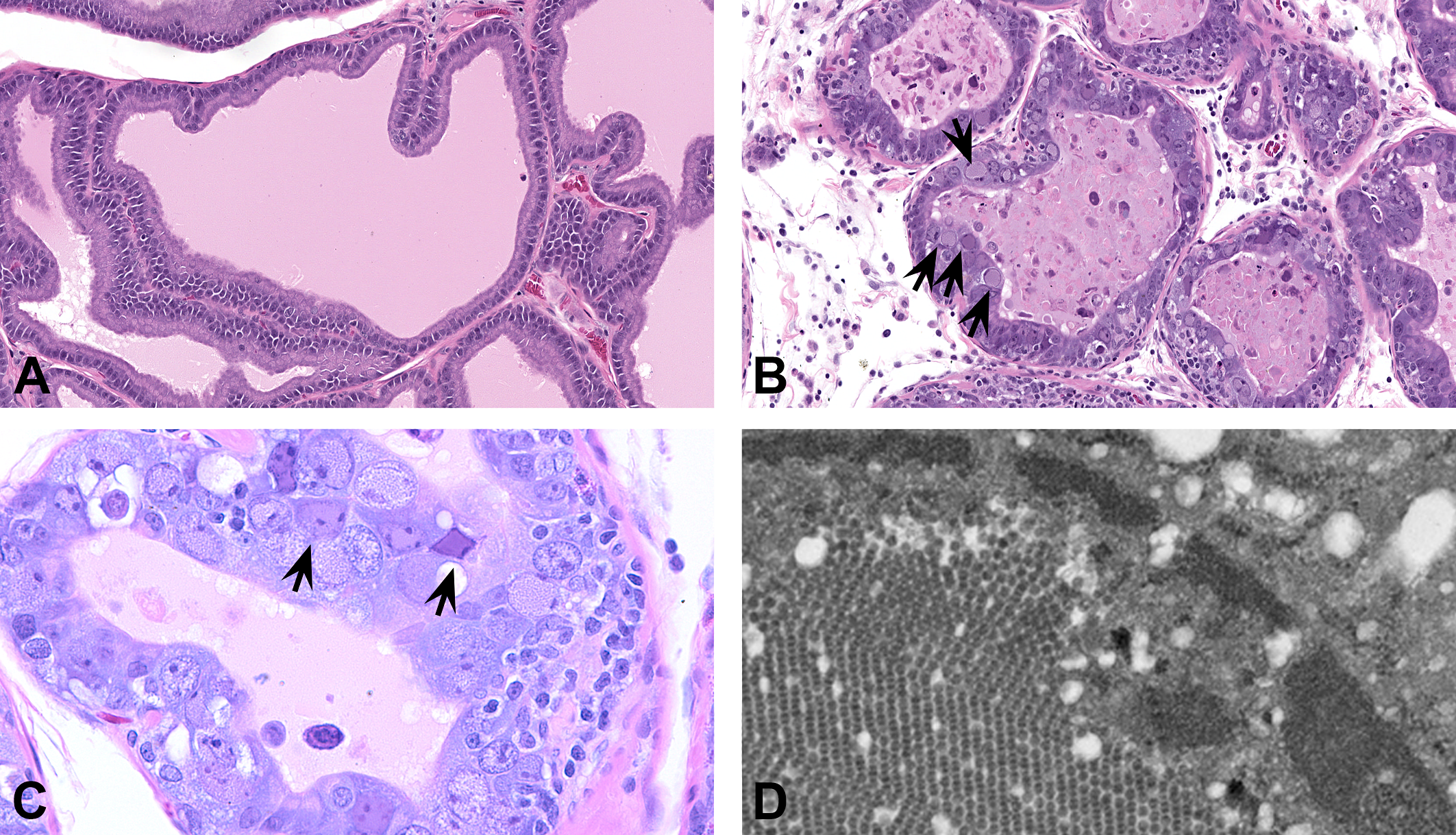
Representative hematoxylin and eosin (H&E) images of prostate gland from control (A) and affected (B and C) high-dose rats. Note cellular atypia, intranuclear inclusion bodies in prostatic epithelial cells (arrows), interstitial inflammation, and degenerate cellular debris within lumens (H&E; original magnification 20–40×). (D) Transmission electron microscopy image of prostatic epithelial cell from affected rat in (B) showing numerous 45-nm icosahedral virions arranged in paracrystalline arrays within the nucleus (original magnification 20,000×).
Relevant hematology findings indicative of immunosuppression in the 2 affected rats included lower white blood cell (WBC) counts relative to concurrent control males (0.28× and 0.31× control group mean on study day 92, 0.29× and 0.31× control group mean on study day 183), predominantly driven by decreased lymphocytes (0.15× and 0.18× control group mean on study day 92, 0.14× and 0.15× control group mean on study day 183) but also to lesser extent by lower neutrophils, eosinophils, and monocytes. Similar hematology findings were also present in the other males in the same dose group, although there was no microscopic evidence of viral infection in any tissues in the other animals.
TEM examination was performed on formalin-fixed prostate gland tissue from the 2 affected high-dose animals as well as on 2 control rats in which there was low-level mononuclear cell infiltrates in the prostate gland considered within the spectrum of background findings that can be observed in the prostate gland of Wistar Han rats (Blankenship and Skaggs 2013; Creasy et al. 2012). On TEM, numerous icosahedral virions consistently measuring 45 nm were observed in paracrystalline arrays in epithelial cell nuclei from 1 of the 2 high-dose rats. The particles were also present in small numbers scattered within the cytoplasm in involved cells. In this animal, viral inclusion bodies had been observed on light microscopy to be widespread and highly frequent. There were no viral particles identified in the small section that was available to examine from the other affected high-dose male in which inclusion bodies were less numerous, and additional tissue was not available for further analysis. Viral particles were not observed in the control animals. Based on the size, icosahedral shape, paracrystalline array pattern, and the subcellular localization, the virus was identified as most consistent with a polyomavirus (Gelderblom 1996; Ward et al. 1984). Polymerase chain reaction (PCR) was attempted on nucleic acid derived from FFPE samples from both affected animals and 2 control rats using 3 sets of primers designed from relatively conserved regions of the published sequences of the hamster and mouse polyomavirus VP1 viral capsid genes. However, this did not yield products of the expected size, and isolation and sequencing of the virus in these 2 rats was not possible with the material remaining from the study. Residual nucleic acid material generated for this work was also tested for other known viruses in a mouse/rat comprehensive virus PCR screening panel (Charles River Laboratories Wilmington, MA, USA) and was found to be negative. Thus, the diagnosis of polyomavirus-associated prostatitis was made based on the unusual microscopic findings and distinctive intranuclear inclusions observed on light and TEM.
Discussion
To the best of our knowledge, this is the first report of polyomavirus-associated lesions in the prostate gland of the widely utilized Wistar Han rat strain related to immunosuppression in a routine chronic toxicity study.
We hypothesize that test article–associated immunosuppression created a permissive environment for viral replication in the prostate gland of the 2 rats in our study; however, it is unclear whether these rats were previously infected and immune suppression led to viral recrudescence or whether the lesions resulted from new infection while on the study. In the latter scenario, the source of the virus in the current study would be unknown; all rats were housed individually in conventional open top cages under strict biocontainment at the laboratory where the study was performed. We speculate that the viral infection in the affected animals correlates with test article–associated decreased WBC counts, particularly T lymphocyte populations within the lymphocyte differential, which are thought to contribute significantly to maintenance of immunity to polyomavirus (Rigatti et al. 2016; Ward et al. 1984). However, it is interesting to note that similar suppression of WBC and lymphocytes occurred in other individual animals in which viral reactivation was not observed, indicating sporadic infection with the virus and/or requirement for additional individual host factors.
At the time of this study (2013 to early 2015), rat polyomavirus infection was not recognized to be widespread. A polyomavirus affecting athymic nude rats had been reported previously in a cohort of animals at the National Institutes of Health (Ward et al. 1984). In these animals, lymphocytic inflammation and intranuclear inclusions were present within ductal and glandular epithelium in the parotid salivary gland. TEM identified 45-nm icosahedral virions arranged in crystalline arrays in nuclei and scattered throughout the cytoplasm of affected cells, and these were further described as reactive for polyomavirus SV40 antigen by immunohistochemistry (IHC). IHC reactivity was also present in inclusion material observed in lung and Harderian gland epithelium in that report.
Very recently, several reports have emerged detailing polyomaviruses circulating in feral and laboratory rat colonies. Most consistent with the cases we report here, a novel rat polyomavirus 2 (RPyV2) was identified in a colony of immunosuppressed, X-linked severe combined immune deficiency rats (Rigatti et al. 2016). This was associated with inflammation, epithelial hyperplasia/dysplasia, and/or intranuclear inclusion bodies in multiple epithelial sites, including the prostate gland. Clinical observations were limited to respiratory signs, which were considered related to coinfection with Pneumocystis carinii, as well as chromodacryorrhea, and reduced fecundity. Rat polyomavirus 1 (RPyV1) has also been recently reported in feral rats in Europe (Ehlers et al. 2015; Heuser et al. 2017), although thus far this virus has not been associated with microscopic lesions. Based on the recently published descriptions, it is possible that the virus identified in our study is identical or related to the polyomavirus described in athymic nude rats and/or RPyV2, although the possibility of a novel polyomavirus in the Wistar Han strain should also be considered. A major diagnostic laboratory recently reported unpublished data in which antibodies to RPyV2 were found in 32% of surveyed immunocompetent rats used in biomedical research in North America (IDEXX BioResearch, 2016). However, screening for polyomavirus was not performed routinely in rats at the time of this study, and thus the serologic profile of animals in our study is unknown. Moreover, at the time of our study, neither RPyV1 nor RPyV2 had been described and sequence information had not been published, and attempts to confirm polyomavirus sequence from prostate gland tissue by PCR using primers based on mouse and hamster polyomavirus VP1 sequences were unsuccessful. Unfortunately, additional PCR using primers specific for the newly described rat polyomaviruses could not be pursued because the formalin-fixed prostate tissue and nucleic acid material from our study were consumed during the initial analyses.
Review of the literature and our internal histopathology historical database from control Wistar Han and Sprague-Dawley rats used in routine chronic toxicity studies indicates that mononuclear inflammation in the prostate gland occurs as a spontaneous finding in control adult rats and may be associated with epithelial atypia (Blankenship and Skaggs 2013; Creasy et al. 2012). The possibility that polyomavirus infection may be more widespread than currently recognized in colonies of laboratory rats used in toxicity studies should be considered when evaluating inflammation and epithelial atypia in prostate gland from control rats. Moreover, the possibility of rat polyomavirus infection should be considered as a differential diagnosis for inflammation, cellular atypia, and/or inclusion bodies involving epithelial sites in toxicity studies involving immunomodulatory agents in rats.
Footnotes
Acknowledgments
The authors would like to acknowledge the contributions of Kenneth Henderson and Cheryl Perkins at Charles River Laboratories for their work attempting to amplify polyomavirus sequences from nucleic acid isolated from control and affected prostate gland samples in this study. We would also like to thank W. Mark Vogel and Daniel Morton for scientific input and helpful discussion.
Author Contribution
Authors contributed to conception or design (KM, TB, ZR); data acquisition, analysis, or interpretation (KM, TB, WB, LT, MD, LW, ZR); drafting the manuscript (KM, TB); and critically revising the manuscript (KM, TB, WB, LT, MD, LW, ZR). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: This work was funded and supported by Pfizer Inc.
