Abstract
Cyclosporin A (CsA) is an immunosuppressive drug widely used in medicine to reduce the immune system activity and, therefore, the risk of organ rejection after transplantation. However, many side effects can be related to its use, such as, reduction in serum testosterone levels due to damage of the testis structure and, consequently, male infertility. The present study aims to evaluate the effects of chronic CsA administration on the ventral prostate tissue (15 mg/kg per d, for 56 days). Stereological, morphometrical, morphological and ultrastructural observations were employed. The plasmatic testosterone and glucose levels were measured. An androgen receptor (AR) immunohistochemical method was applied on ventral prostate sections. Apoptosis was detected with the terminal deoxynucleotidyl transferase dUTP nick end labeling technique. CsA treatment caused reduction in plasmatic testosterone levels and an increase in glycemia. The volume of all ventral prostate tissue components (lumen, epithelium and muscular and nonmuscular stroma) and ventral prostate weight were reduced in the CsA-treated group. Light and transmission electron microscopy confirmed epithelium atrophy of treated animals. There was no alteration of AR expression or apoptotic index. CsA chronic treatment in the therapeutic doses caused damage to prostate tissue of adult Wistar rats, probably due to increase in the glucose levels and reduction in the plasmatic testosterone levels.
Introduction
Cyclosporin A (CsA) is a hydrophobic, cyclic undecapeptide isolated from the fungi
CsA is selectively cytotoxic and a suppressor of the lymphocyte subpopulation, although it does not interfere with phagocytes’ functions or hematopoietic stem cells.4,5 Also, it may cause many side effects after chronic administration in organs like kidneys, liver, lymph nodes and pancreas as well as in the nervous and cardiovascular systems. 1 CsA-induced nephrotoxicity and hepatotoxicity are related to increased reactive oxygen species production.6,7 The CsA-induced damage to pancreas tissue, more specifically to the Langerhans islets, leads to disturbed glucose tolerance. 1
Moreover, male reproductive organ dysfunctions induced by sustained CsA intake are extensively reported in the literature.8–18 CsA intake causes dose-dependent reduction in the testis (10, 20 and 40 mg/kg administered daily, subcutaneously, for 14 days) as well as seminiferous tubule atrophy due to impairment of Sertoli cell function (in animals treated with 40 mg/kg per d of CsA),11,13 besides reducing testicular weight. The epididymis, seminal vesicle and prostate weights were also reduced, 11 besides epididymal sperm counts, sperm motility and fertility.11,12 Daily administration of CsA by gavage (15 mg/kg per d, for 56 days) causes structural and functional damages to the testis of Wistar rats. 18 CsA also impairs steroidogenesis,12,14–16 which is probably due to a suppression of the hypothalamic-pituitary axis rather than direct inhibition at the testicular level.9,10,14,15
Prostate weight reduction caused by CsA administration is commonly reported in the literature.3,8,9 The prostate provides the bulk of the seminal fluid, adding proteins and ions; therefore, any alterations of its optimal function could lead to impairment of fertility. 19
Prostate development and function are androgen dependent.20–23 Androgen receptors (ARs) are present in most cells of the prostatic epithelium and some fibroblasts, but not endothelial cells. 24 ATPases could be the possible sites for testosterone action in the prostate. They would affect water, ion and metabolite transport through blood vessels, to reach target cells. 22 Prostate response to the reduction in testosterone levels, in castrated animals, begins with a decline in blood flow followed by epithelial cell apoptosis.19,24
Despite many proven CsA side effects that could imply in deterioration of male reproductive tissue functions, no previous research has shown the effects of administration of this drug on the prostate tissue. Therefore, this study was designed to evaluate possible CsA side effects, using stereology, morphometry and image analysis of the ventral prostate of Wistar rats.
Methods
Animal protocol
A total of 12 adult male Wistar rats (90 days old) were obtained from the Center of Biological Investigation—CEMIB (State University of Campinas, Campinas, São Paulo, Brazil) and maintained under standard conditions with 12 h light and 12 h dark cycles and free access to food and water. Food consumption did not vary significantly for different groups.
Six animals received CsA (Sandimmun Neoral—Oral Solution; 100 mg/mL; Novartis Pharma Ag, Switzerland) daily, by gavage (15 mg/kg per d),7,18,25,26 dissolved in 0.5 mL distilled water. Control rats (
Before euthanasia, the animals were weighed, anesthetized with xylazine and ketamine (5 and 80 mg/kg body weight, respectively) and the abdomen and thoracic cavities were opened. The testis, epididymis, seminal vesicle, coagulating gland and ventral prostate were excised and weighed. To obtain the relative weight, the weight of each organ was multiplied by 100 and divided by the body weight.
Testosterone and glucose assay
Blood samples were collected by cardiac puncture (left ventricle) immediately before death and stored in Vacuette® tubes under refrigeration (4°C). Plasma was obtained by blood centrifugation (1400
Morphometrical and stereological analyses
Fragments of one lobe of the ventral prostate were immersion-fixed in Karnovsky’s fixative (4% paraformaldehyde, 4% glutaraldehyde in 0.1 M phosphate buffer, pH 7.2) for 24 h. After fixation, the tissue was routinely prepared for embedding in 2-hydroxyethyl methacrylate (Historesin®, Leica). Sections of 2 μm thickness were obtained, stained with hematoxylin–eosin and observed with an Olympus BX-40 light microscope.
All quantitative measurements were performed using Image Pro Plus software. The relative volume of the tissue compartments (lumen, epithelium and muscular and nonmuscular stroma) was determined employing a modified methodology according to Weibel 27 using a 130-point grid system. Ten microscopic fields were randomly chosen (200× magnification). Absolute volumes were estimated multiplying relative volume by prostate weight, 28 since the ventral prostate density could be considered approximately 1.0. 29 Morphometric analysis included the determination of epithelial cell and muscular stroma thickness (µm), nuclear area (µm2) and perimeter (µm), cytoplasm area (µm2) and form factor (4π × nuclear area/(nuclear perimeter) 2 ). All analyses cited above were employed on the intermediary region of the ventral prostate.
Ultrastructural analysis
Ventral prostate fragments were immersion-fixed in modified Karnovsky’s fixative (2.5% glutaraldehyde and 4% paraformaldehyde in phosphate buffer, pH 7.4) for 24 h. The materials were postfixed in 1% osmium tetroxide for 2 h, dehydrated in acetone and embedded in epoxy resin. Ultra thin sections (60–70 nm) of the intermediary region of the ventral prostate were stained with 3% uranyl acetate (30 min) and 2% lead citrate (5 min) and then observed with a transmission electron microscope (Leo 906, Zeiss).
AR immunohistochemistry and detection of apoptosis
The contralateral lobe of the ventral prostate was immersion-fixed in Methacarn fixative (methanol:chloroform:acetic acid of 6:3:1), routinely prepared for paraffin embedding and cut into 5 µm sections for AR immunohistochemistry and terminal deoxynucleotidyl transferase dUTP nick end labeling assays.
Antigen retrieval was applied to AR development. A high-temperature heating method was used in order to recover the antigenicity of tissue sections. It was carried out by incubating sections in citrate buffer (pH 6.0) and kept at high temperature (100°C) for 45 min. Endogenous peroxidase was blocked with hydrogen peroxide (H2O2; 0.3%) in methanol for 20 min. After rinsing with phosphate saline buffer for 5 min, the slides were incubated with the anti-AR primary antibody (AR; SC-816, Santa Cruz Biotechnology, Santa Cruz, California, USA) at 37°C for 1 h and incubated with horseradish peroxidase-conjugated biotinylated antibody (SC 2018, Santa Cruz Biotechnology) at 37°C for 45 min. The slides were then incubated with peroxidase-conjugated avidin–biotin complexes and diaminobenzidine (DAB) and counterstained with Harris’s hematoxylin for 10 s.
The detection of apoptotic nuclei was obtained using a kit for fragmented DNA detection (US1QIA33-1EA; TdT FragEL TM DNA Fragment. Detect, Merck). Briefly, the slides were incubated with proteinase K for permeabilization followed by endogenous peroxidase activity blocking with 3% H2O2 in methanol (1:1) for 5 min. Afterward, they were submitted to the equilibrium reaction. Detection was performed as indicated by the kit’s manufacturer. Positive reaction was developed by DAB/H2O2. Finally, the sections were counterstained with Harris hematoxylin for 10 s.
Apoptotic nuclei (brown colored) were quantified in 10 different high power fields (400×) for each animal and compared with the total number of epithelial cells in the field to obtain the apoptotic index. 24
Statistical analysis
Statistical tests were performed with Statistica® 5.1 software. Results were expressed as mean ± standard deviation. The means were compared using
Results
Biometric analysis
All biometrical data are shown in Table 1. The gain of body weight in CsA-treated animals was reduced 32.81% (
Biometric data obtained from Wistar rats treated with CsA (mean ± SD)

CsA: cyclosporin A.
aSignificant difference (
Plasmatic testosterone and glucose levels
The plasmatic testosterone levels reduced 60.76% in CsA-treated group when compared with the control (
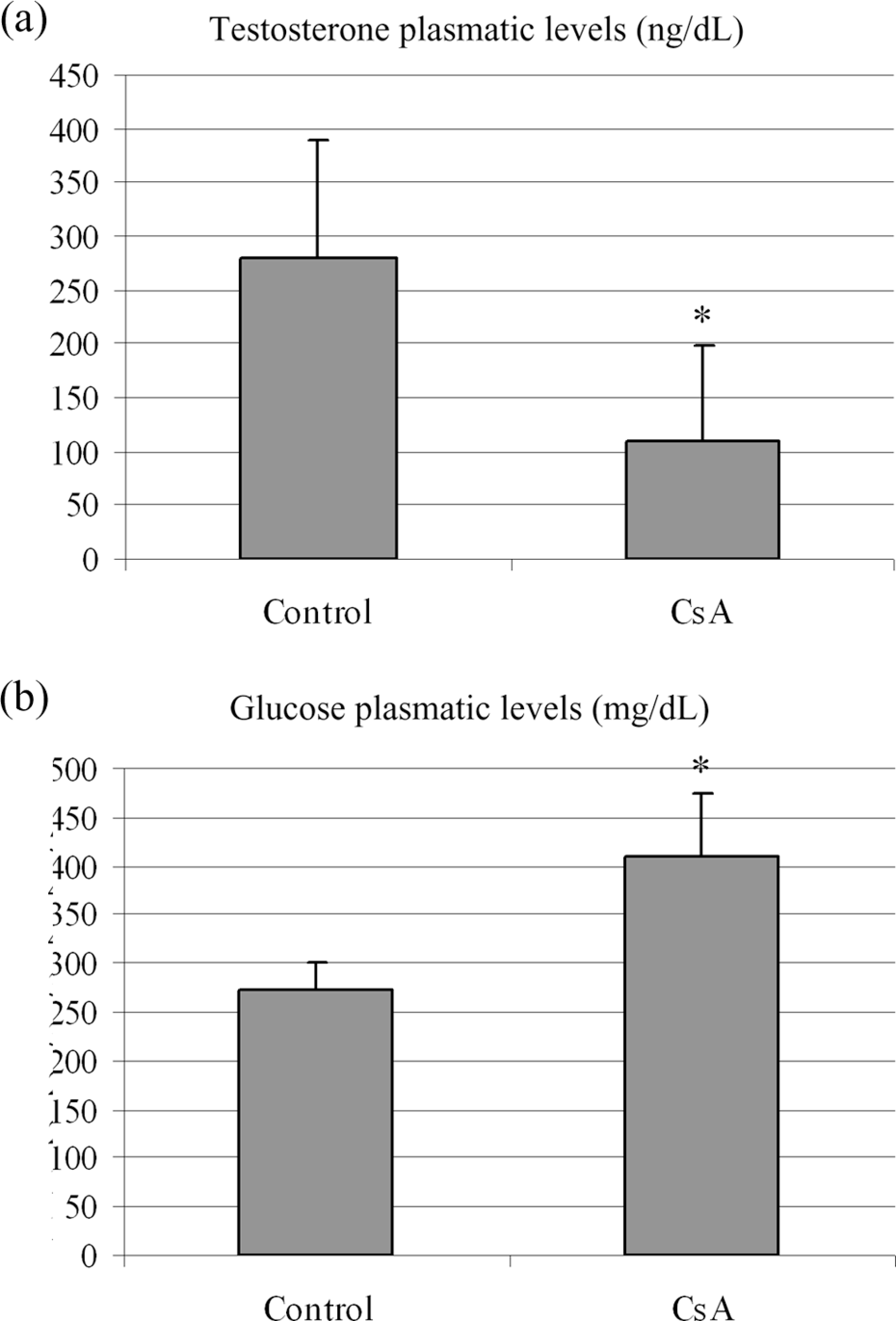
Plasmatic levels of testosterone (a) and glucose (b). *Significant difference (
Morphometrical and stereological parameters
The relative volume of prostate tissue elements did not differ between the experimental groups. The absolute volume of lumen, epithelium and muscular and nonmuscular stroma was significantly reduced in the CsA treated group (p= 0.03, 0.01, 0.005 and 0.001 respectively) (Table 2).
Stereological and morphometrical data after long-term treatment with CsA (mean ± SD)

CsA: cyclosporin A.
a
b
The nuclear, cytoplasmic and total areas of prostate epithelial cells, as well as epithelial and muscular thicknesses were reduced in CsA treated animals (Table 2).
Morphological and ultrastructural aspects
The ventral prostate of control animals showed a normal morphology. The epithelium had cubic/columnar secretory cells with basal nuclei and a clear supranuclear region, which corresponded to the Golgi complex region (Figure 2(a) and (b)). Most of the epithelial cell cytoplasm was occupied by enlarged rough endoplasmic reticulum in control animals (Figure 2(e)). Secretion vesicles could be observed near the Golgi complex (Figure 2(e)).

Optical micrographs of Wistar rats’ ventral prostate stained with hematoxylin and eosin ((a) to (d)), and transmission electron micrographs of the same tissue ((e) to (g)). (a) and (b) represents normal ventral prostate tissue structure (control), the epithelium is columnar and there is a large lumen (lu). The ventral prostate of CsA-treated animals ((c) and (d)) showed some atrophic regions with squamous epithelium (arrowhead). Ultrastructural micrographs of control animals (e) showed the normal epithelium with basal nuclei cytoplasm rich in rough endoplasmic reticulum and apical secretion vesicles (arrows). There are also blood vessels associated with the epithelium. (f) and (g) detailed the atrophy observed in CsA-treated animals. The cells did not show developed rough endoplasmic reticulum, some secretion vesicles were enlarged and empty (triangles). Lysosomal vesicles (white asterisks) were also found. Scale bars = 10 μm ((a) to (d)); 2 μm ((e) to (g)). e: epithelium; v: blood vessel; n: basal nuclei; rer: rough endoplasmic reticulum.
The atrophy observed in the prostatic epithelium was variable: some parts showed severe damage due to CsA treatment, while others were apparently normal or less damaged. The ventral prostate epithelium varied from normal with cubic/columnar cells to completely atrophied squamous epithelium (Figure 2(c), (d), (f) and (g)). Very few regions of the ventral prostate of control animals presented atrophy. Ultrastructural analysis confirmed epithelial atrophy with reduction in secretory organelles (rough endoplasmic reticulum and Golgi complex) or their total absence (Figure 2(f) and (g)) as well as enlarged secretion vesicles and lysosomal vesicles (Figure 2(f) and (g)).
AR immunohistochemistry and apoptosis detection
No change was observed in AR immunocytochemistry in CsA-treated ventral prostate animals: even atrophied epithelial cells nuclei were positive to AR assay (Figure 3).

Light microscope images of ventral prostate with AR immunohistochemistry of control ((a) and (b)) and CsA-treated animals ((c) and (d)). The arrowheads indicate the atrophied secretory epithelium with positive nuclei staining for AR expression. Scale bars = 50 µm ((a) and (c)); 10 µm ((b) and (d)). AR: androgen receptor; *: lumen; s: stroma; e: epithelium.
The apoptotic index was lower than 0.5 in both the experimental groups and did not differ statistically in the CsA-treated group when compared with the control.
Discussion
Many side effects are related to chronic treatment with CsA. There are extensive studies on the side effects of testicular structure, sperm and testosterone production.7–18 However, even after a wide search in specialized journals, no previous study determining the effects of chronic treatment with CsA on the ventral prostate tissue was found. Therefore, this is the first study that shows CsA effects on the structure of Wistar rat ventral prostate.
The dose of CsA administered during this study was according to that specified by the manufacturer (Novartis Pharma Ag, Switzerland) for treatment after organ transplantation and was the same used by other authors.7,18,25,26
Body weight is commonly evaluated in order to determine the effect of CsA treatment on the general health of the animals.11,30 Body weight gain was 32.64% lower in the CsA-treated group, although this was not statistically significant, probably due to large differences in the animals’ sensitivity to the treatment. This result was not due to changes in feed consumption since this was very similar in both experimental groups (28.22 ± 2.22 g/day per animal in the control group and 29.50 ± 2.77 g/day per animal in the CsA group). A similar result was observed by Monteiro et al. 18 in Wistar rats treated with the same CsA dose for the same period of time.
Some reproductive organs, such as the ventral prostate, coagulating gland and epididymis, showed significant weight reduction after CsA treatment, which probably occurred due to reduction in plasmatic testosterone levels, since they are testosterone dependent.22,31 On the other hand, testis and seminal vesicle gland weight were not altered in the treated group. In a previous study using animals treated with the same CsA dose, no alteration was observed in ventral prostate weight, coagulating gland, seminal vesicle, epididymis or testis. 18 However, Türk et al. 7 observed the maintenance of testis, epididymis and prostate weights, while the seminal vesicle weight was reduced in animals treated with 15 mg/kg per d of CsA. The differences found could be associated to the individual sensitivity to drug treatment.
CsA treatment caused significant reduction in the absolute volume of all the prostate components (epithelium, lumen, and muscular and nonmuscular stroma), leading to decreased prostate weight. Besides, prominent epithelial atrophy was observed in CsA-treated animals. Atrophy of the ventral prostate epithelium can be related to reduction in cellular secretory capacity, since there was significant reduction in nuclear and cytoplasmic volume. The organelles directly involved in cellular secretion, mainly the rough endoplasmic reticulum and Golgi complex, were also greatly reduced, as observed with transmission electron microscopy. Although atrophy is considered a common histological alteration in human prostate, it is apparently uncommon in wild-type mouse species. 32 Atrophy caused after CsA treatment could be due to the reduction in the plasmatic testosterone levels and/or the increase in plasmatic glucose levels.
Reduction in testosterone levels in animals treated with CsA was extensively reported by other studies.12,14–17 In castrated animals, the ventral prostate epithelium was reduced to one half within 2 days after castration. 29 According to Huttunen et al., 29 the ventral prostate epithelium is especially sensitive to androgen deprivation.
Neto et al. 33 observed that the Wistar rats treated with CsA for 14 or 21 days (15 mg/kg body weight per d) showed high insulin levels as well as hyperglycemia, causing peripheral resistance to insulin action. Also, animals that developed alloxan-induced diabetes had blood glucose levels higher than 400 mg/dL, 34 which could be related to drastic changes in ventral prostate structure, mainly in the intermediary region. In the present study, these combined results were also observed after CsA treatment, along with the reduction in the acinar lumen and epithelial height, transforming the columnar epithelium into a squamous epithelium.
In the ventral prostate of adult rats, AR are concentrated in the epithelial cell nuclei, while in stromal cell nuclei, they occur in moderate levels or are absent. 35 The pattern of AR expression suggests that the epithelial cells are the major target of androgen action on the ventral prostate. 35 There is a dramatic loss of nuclear AR 12 h after castration, and it appears to be an important (not the only) apoptosis trigger point within the prostate. 36 Atrophy of the prostatic epithelium is associated with a large increase in the apoptotic index, which reaches its maximum range 72 h after castration. Afterward, the apoptotic index decreases, going back to normal after 7 days.20,24 In the present study, the constancy of the apoptotic index can be due to the chronic treatment with CsA (56 days). Similar to the findings of the present study, Arcolino et al. 34 showed that long- and medium-term alloxan-induced diabetes did not alter androgen levels of the ventral prostate epithelium.
The similarity between our results and the study with alloxan-induced diabetes indicates that CsA damage to the ventral prostate is related to a link between lower plasmatic testosterone levels and higher plasmatic glucose levels, and could include other effects of possible CsA-induced diabetes. Therefore, 8 weeks administration of CsA showed that the damage caused to the prostate tissue could increase damage to male fertility, caused by this drug.
Footnotes
Acknowledgments
The authors thank the students of the Laboratory of Microscopy and Microanalyses of UNESP, São José do Rio Preto for the technical support and also thank Felipe Boschiero from “Espaço da Escrita” for correcting the formatting of this work.
Funding
This study received financial support from Fundação de Amparo à Pesquisa do Estado de São Paulo—FAPESP (grant number: 2009/07824-3), Fundo de Apoio ao Ensino, à Pesquisa e à Extensão—FAEPEX/UNICAMP (grant number: 92910) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—CAPES.
