Abstract
The medium-term multiorgan initiation–promotion chemical bioassay (diethylnitrosamine, methyl-nitrosourea, butyl-hydroxybutylnitrosamine, dihydroxypropylnitrosamine, dimethylhydrazine [DMBDD]) with the Fischer 344 rat was proposed as an alternative to the conventional 2-year carcinogenesis bioassay for regulatory purposes. The acronym DMBDD stands for the names of five genotoxic agents used for initiation of multiorgan carcinogenesis. The Brazilian Agency for the Environment officially recognized a variation of this assay (DMBDDb) as a valid method to assess the carcinogenic potential of agrochemicals. Different from the original protocol, this DMBDDb is 30-week long, uses Wistar rats and two positive control groups exposed to carcinogenesis promoters sodium phenobarbital (PB) or 2-acetylaminofluorene (2-AAF). This report presents the experience of an academic laboratory with the DMBDDb assay and contributes to the establishment of this alternative DMBDD bioassay in a different rat strain. Frequent lesions observed in positive groups to evaluate the promoting potential of pesticides and the immunohistochemical expressions of liver cytochrome P450 (CYP) 2B1/2B2 and CYP1A2 enzymes were assessed. Commonly affected organs were liver, kidney, intestines, urinary bladder, and thyroid. PB promoting activity was less evident than that of 2-AAF, especially in males. This study provides a repository of characteristic lesions occurring in positive control animals submitted to a modified alternative 2-stage multiorgan protocol for carcinogenesis in Wistar rat.
Keywords
For decades, the conventional rodent 2-year long in vivo carcinogenicity assay has been considered to be a reliable assay to identify chemical carcinogens (International Agency for Research on Cancer [IARC] 1986; Cohen 2001; Tsuda et al. 2010). However, this long-term test has several disadvantages, for example, extended time for performance, complexity, use of large numbers of animals, expensive operational procedures, and questionable relevance of findings to humans. Therefore, more convenient, inexpensive, faster, and relevant to humans’ alternative protocols are considered necessary (de Camargo 1991, 2007; Slamenová 2001; Gori 2013).
Several alternative assays as surrogate for the conventional two-year long carcinogenicity assay have been proposed, including animal, in vitro and computational modeling. The rodent assays have been envisaged as a useful cancer screening test and primary role in the identification of agents considered hazardous to humans. Despite high sensitivity, the specificity of the tumor response in animals is a critical point for human cancer prediction (Bucher and Portier 2004; Cohen 2004; MacDonald 2004; Woutersen et al. 2016; Knight, Bailey, and Balcombe 2006). Genetically modified mouse models have been proposed, resulting in transgenic bioassays with minimized influence of strain and/or species-specific genetic effects by using oncogenes and tumor suppressor genes as targets (Tennant 1997). However, interpretation of these assays should be cautious since strain issues such as different metabolism, anatomy, and toxicokinetics remain (Cohen 2004).
Since the 1970s, when the 2-year bioassay was first adopted as a routine assay for rodent carcinogen identification, progressive scientific knowledge about the mechanisms of carcinogenesis has changed from the traditional toxicology paradigm based on apical outcomes to the toxicologic pathway-based approaches (Martin et al. 2010; Schwetz and Gaylor 1998). The U.S. Environmental Protection Agency with the ToxCast program, the National Institutes of Health, and the European Commission with the Registration, Evaluation, Authorization & restriction of Chemicals (REACH) program have responded to this challenge with major new computational toxicology testing initiatives used to derive biological and chemical profiles predictive of in vivo end points and to serve as modes of action basis for new toxicity pathway biomarkers on screening and prioritization approaches (Martin et al. 2010). However, these models are not very strong for predicting carcinogenicity, as they are mostly restricted to DNA reactivity, mutagenesis, and receptor interaction, for example (Cohen 2004).
A multiorgan 28-weeklong system based on the mechanistic initiation–promotion concept of chemical carcinogenesis was proposed by a research group led by Nobuyuki Ito at the Nagoya City University Medical School, Japan, as an alternative/complementary approach to the conventional long-term bioassay system (Ito et al. 1996, 1988; Shirai, Hirose, and Ito 1999; Ito and Imaida 1992; Ito et al. 1998). It consists of sequential treatment of rats with five genotoxic carcinogens to establish multiorgan initiation of carcinogenesis followed by dietary exposure to a test substance in order to evaluate its promoting potential. This 28-week-long bioassay has been designated diethylnitrosamine, methyl-nitrosourea, butyl-hydroxybutylnitrosamine, dihydroxypropylnitrosamine, dimethylhydrazine (DMBDD) assay after the initials of the five chemicals used to initiate carcinogenesis: diethylnitrosamine (DEN), methyl-nitrosourea (MNU), butyl-hydroxybutylnitrosamine (BBN), dihydroxypropylnitrosamine (DHPN), and dimethylhydrazine (DMH). The assay was established using the inbred male Fischer 344 rat and has the advantages of shorter duration, the use of a single rodent species and gender, faster results at lower cost, and the possibility of mechanistic approaches. Besides being cost-effective, the results of the DMBDD assay have been shown to be in line with those provided by the long-term conventional bioassay (Hagiwara, Tanaka, et al. 1993a; Ito et al. 2000; Shirai et al. 1999; Tsuda et al. 2010; Doi et al. 2011). Among 44 compounds tested, this assay was able to identify as carcinogens 22/23 mutagenic and 13/15 nonmutagenic recognized chemical carcinogens (Tsuda et al. 2010). This medium-term multiorgan carcinogenesis bioassay has also been used to evaluate the carcinogenic potential of synthetic and natural substances (Hagiwara et al. 2011; Imai et al. 2012).
A task group gathered by the IARC recognized that assay systems based on the initiation–promotion concept of chemical carcinogenesis could be considered appropriate for identifying carcinogens in rodents (McGregor, Rice, and Venitt 1999). The International Conference on Harmonization (ICH) recommended these alternative bioassays as part of safety evaluation of pharmaceuticals, as surrogate for the second long-term rodent assay with mice (ICH 2010).
As many developing countries do not have enough technical expertise, animal breeders, and/or suitable facilities to routinely run the conventional long-term bioassay, the adoption of less complex, shorter, and less costly assays was deemed necessary (de Camargo et al. 1999b). An academic laboratory at the Department of Pathology, Botucatu Medical School, UNESP, developed a series of studies in order to establish a variation in the DMBDD bioassay that could attend those needs (Barbisan et al. 2003; Bononi et al. 1999; da Silva Franchi 2003; de Camargo 1991; de Camargo et al. 1999a, b; Moreira et al. 2000; Rodrigues et al. 2002; Spinardi et al. 1999). Eventually, this variation in the DMBDD bioassay (indicated from now on as DMBDDb) was officially adopted by the Brazilian Agency for the Environment as a valid source of evidence of the carcinogenic potential of agrochemicals (Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis [IBAMA] 1996). Upon this official acceptance, five different DMBDDb bioassays sponsored by agrochemical companies were conducted by this laboratory in order to evaluate the carcinogenic potential of pesticides. However, since a worldwide use and acceptance of the 2-step multiorgan alternative bioassays for regulatory purposes were not accomplished, in order to reduce costs, agrochemical companies in this country progressively decided not to conduct the DMBDDb assay anymore, despite it still be accepted by the local environmental agency. As a result, those five studies consist of a unique repository of lesions induced in Wistar rats submitted to the modified DMBDD assay.
Different from the original DMBDD assay with the Fischer 344 male rats, the DMBDDb is characterized by a 30-week instead of a 28-week experimental period, both sexes of the outbred Wistar strain of rats and two positive control groups for promotion of carcinogenesis, the nongenotoxic sodium phenobarbital (PB), or the genotoxic 2-acetylaminofluorene (2-AAF). The outbred Wistar strain of rats was chosen due to the easy availability of these animals in Brazil, and its relative resistance to spontaneous neoplasms (de Camargo 1991; de Camargo et al. 1999b). The most frequent lesions were observed in the liver, kidney, intestines, urinary bladder, and thyroid of the positive control animals. Since both positive controls PB or 2-AAF have the liver as main target organ, the expressions of the cytochrome P450 (CYP) 2B1/2B2 and CYP1A2 isoforms were also immunohistochemically analyzed as surrogate markers of rat liver enzymatic induction (Fukushima et al. 2005; Hagiwara et al. 1996; Martignoni et al. 2004; Tateishi et al. 1999) in the Wistar strain of rats. The present report on those positive control animals may contribute to the establishment of the alternative DMBDD bioassay in a rat strain different from the one originally adopted.
Materials and Methods
Animals
Five separate and successive studies used a total of 450 Wistar rats (225 males and 225 females) aged 4 weeks at the beginning of treatment. They were obtained from Biotecnologia Planalto (Bioplan, Brasília, Brazil) and were submitted to an acclimation period of 3 weeks. The animals were randomly allocated 5 per cage/sex in polypropylene cages, supplied with filtered water and fed NUVILAB-CR1 (NUVITAL, Curitiba, Brazil) ad libitum. They were maintained in a controlled room targeted at 22 ± 2°C, relative humidity of 55 ± 10%, and a 12/12-hr light/dark cycle. The experiment protocol was approved by the Ethics Committee on Care and Use of Laboratory Animals.
Chemical Agents
N-diethylnitrosamine (DEN), N-methyl-N-nitrosourea (MNU), N, N-dimethylhydrazine (DMH), and 2-AAF were purchased from Sigma Chemical Co. (St. Louis, MO); N-butyl-N-4(hydroxybutyl)nitrosamine (BBN) and sodium PB were purchased from Tokyo Kasei Industries Co. (Tokyo, Japan); and dihydroxy-di-N-propylnitrosamine (DHPN) was purchased from Nacalai Tesque, Inc. (Kyoto, Japan).
Experimental Design
Each study consisted of four groups as follows (Figure 1). One group was used as untreated negative control and received only basal chow diet. Three other groups were treated sequentially with initiating agents (DMBDD initiation; Shibata et al. 1990; Ito et al. 1998; Hagiwara, Tanaka, et al. 1993a): DEN (100 mg/kg body weight [bw.], i.p., single dose at week 0), MNU (20 mg/kg bw, ip, 2 doses per week for 2 weeks), and BBN (0.05% in drinking water for 2 weeks) administered during the first and second weeks. During the 3rd and 4th weeks, these groups were treated with DMH (40 mg/kg b.w., s.c., 2 doses per week, for 2 weeks) and DHPN (0.1% in drinking water for 2 weeks). One of these DMBDD-initiated groups was maintained without any further treatment until the 30th week (initiation control group); the other two initiated groups were, respectively, exposed through diet to 2-AAF 0.01% or PB 0.05% from the 6th until the 30th week. Beginning with the second study, the animals were fed PB 0.075% or 0.10% instead of PB 0.05%. Therefore, three levels of PB concentrations were used in successive studies because the originally proposed PB 0.05% did not exert a promoting influence on the development of liver (pre)neoplastic lesions as expected based on the studies in F344 rats (Ito, Shirai, and Fukushima 1991). Also, because four of the five studies were conducted with PB, the only study using the carcinogenesis promoter 2-AAF had fewer animals per group in the total. In brief, the number of animals per each group was of 50 males and 50 females in the negative control group, 80 males and 80 females in the only initiated group, and 30 animals (15 males and 15 females) initiated and promoted with 2-AAF, and 160 animals (80 males and 80 females) initiated and promoted with PB.
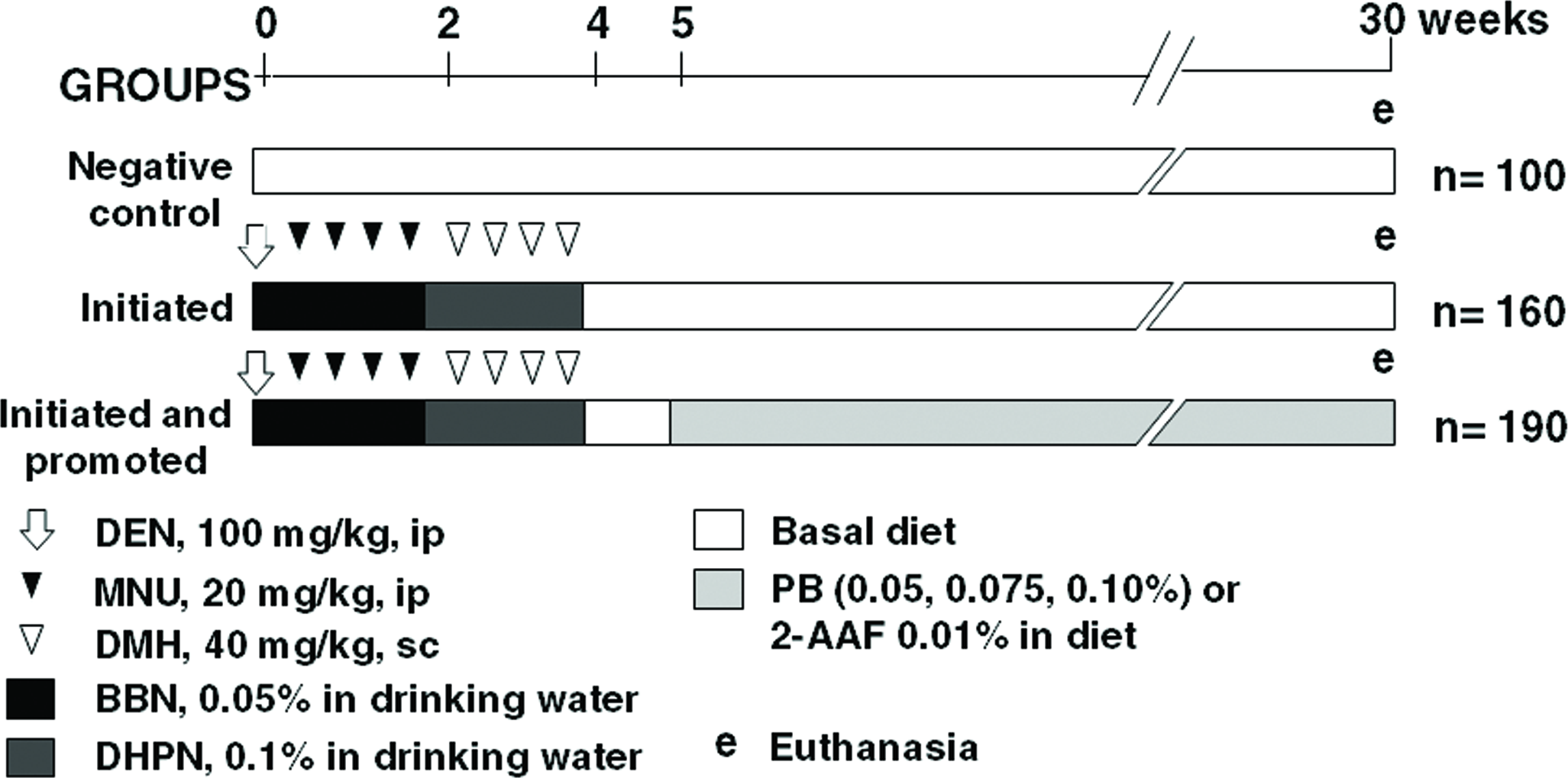
Experimental design of the DMBDDb bioassay, using N-diethylnitrosamine (DEN), N-methyl-N-nitrosourea (MNU), N, N-dimethylhydrazine (DMH), N-butyl-N-4(hydroxybutyl)nitrosamine (BBN), dihydroxy-di-N-propylnitrosamine (DHPN), sodium phenobarbital (PB), and 2-acetylaminofluorene (2-AAF).
Animal body weights were assessed weekly during the first 4 weeks and then every 4 weeks until the end of the experiment. Water and food consumption were measured weekly during the initiation phase and every 2 weeks from the 6th to the 30th week.
Euthanasia was performed at the 30th week by sectioning the inferior vena cava under ether anesthesia. Complete necropsies and histological examination were performed for all organs. After the necropsies, the liver and kidneys were weighed. Tissue samples were fixed in 10% phosphate-buffered formalin until processed and stained with hematoxylin and eosin (H&E) for histological examination. All gross lesions were collected for histological examination. Analyses of lesion incidences were made after histological diagnoses.
Histologic Analysis of H&E-Stained Sections
The DMBDD target organs in previous studies with the local Wistar rats were the liver, kidneys, intestines, urinary bladder, and thyroid (Barbisan et al. 2003; Bononi et al. 1999; da Silva Franchi 2003; de Camargo 1991; de Camargo et al. 1999a, b; Moreira et al. 2000; Rodrigues et al. 2002; Spinardi et al. 1999; Rocha 1998). In these organs, the preneoplastic and neoplastic lesions observed under H&E staining were classified according to the Standardized System of Nomenclature and Diagnostic Criteria (Society of Toxicologic Pathologists 2001). The number of neoplasms in each animal was individually quantified in the respective primary organ. Putative preneoplastic lesions (altered foci and hyperplasia) had only their incidences tabulated. In DMBDD-exposed groups, 4 different types of foci of altered hepatocytes (FAH; Bannasch, Zerban, and Hacker 1997) were observed: clear, eosinophilic, basophilic, and amphophilic cell foci.
Quantitative Evaluation of Immunohistochemically Identified Liver Glutathione S-transferase Placental (GST-P) Foci
Samples from the right, left lateral, and median liver lobes were also prepared for quantitative assessment of glutathione S-transferase placental form positive (GST-P+) foci demonstrated immunohistochemically by the avidin–biotin–peroxidase method (Hsu, Raine, and Fanger 1981). Briefly, 5-μm thick paraffin-embedded liver sections were placed on poly-D-lysine-coated slides, deparaffinized in xylene, and rehydrated. Endogenous peroxidase activity was blocked by 3% hydrogen peroxide, and nonspecific protein binding was minimized by the use of 1% nonfat dried milk. Slides were incubated with rabbit antirat GST-P primary antibody (Medical & Biological Laboratories Co., Tokyo, Japan) diluted at 1:1,000 in 1% bovine serum albumin (BSA, Sigma Chemical Co., MO) and maintained overnight at 4°C. Then the slides were successively incubated with a biotinylated goat antirabbit IgG secondary antibody (Vector Laboratories Inc., CA) diluted at 1:200 in 1% BSA for 60 min, followed by an avidin–biotin–horseradish peroxidase complex kit (Vector Laboratories Inc., CA) diluted at 1:100 in phosphate-buffered saline (PBS) for 45 min. Chromogen color development was made with 0.038% 3-3’-diaminobenzidine tetrahydrochloride (Sigma Chemical Co., MO) and 0.025% hydrogen peroxide. The sections were counterstained with Harris’s hematoxylin. The number and area of GST-P+ foci larger than 0.15 mm in diameter were measured using a color video image processor (Luzex-F-Nireco, Japan). Data were expressed as the number and area (mm2) of putative preneoplastic GST-P+ foci per cm2 of liver section.
Immunohistochemical analyses of CYP2B1/2B2 and CYP1A2 expression using liver tissue microarrays
Six random liver samples of each animal were collected for the evaluation of CYP2B1/2B2 and CYP1A2 expression in paraffin section tissue cores 2.0 cm in diameter. The streptavidin–biotin amplification method was used to determine the expression of CYP2B1/2B2 and CYP1A2 isoforms. The EnVision verification kit (Dako, CA) was used for the CYP2B1/2B2, and the labeled streptavidin–biotin (LSAB) kit (Dako) for CYP1A2. Briefly, 5-μm thick paraffin-embedded liver sections were deparaffinized and rehydrated. To retrieve the antigens, the sections were heated at 120°C for 3 min in citric acid buffer. The endogenous peroxidase activity was blocked with 3% hydrogen peroxide in PBS for 5 min. The primary antibodies, a polyclonal antibody for CYP2B1/2B2 (Fitzgerald, Acton, MA) diluted at 1:2,000 or a monoclonal antibody for CYP1A2 (Abcam Inc., Cambridge, US) at 1:1,000 dilution, were applied and the slides incubated at 4°C overnight. The sections were rinsed in PBS and incubated with EnVision antirabbit polyclonal antibody or antimouse LSAB, respectively, for 1 hr at room temperature. The antigen–antibody complex was visualized with 3-3’-diaminobenzidine solution and counterstained with Harris’s hematoxylin.
As rat CYP2B1 is highly homologous to CYP2B2, the antibody cross-reacts with both forms and thus the staining is indicative of CYP2B1, CYP2B2, or both (Parkinson 2001). Histologic analyses revealed three patterns of immunostained hepatocyte distribution within the lobule: strictly perivenular (centrilobular vein), broadly perivenous (including midzonal area), and diffuse throughout the lobule. No expressions of CYP2B1/2B2 or CYP1A2 were observed exclusively around the periportal area in any group. The intensities of CYP expression were graded as negative, mild, moderate, and intense. Among the three PB-treated groups, only the DMBDD + PB 0.075%-treated group was analyzed.
Statistical Analyses
For group comparisons, DMBDD-initiated animals were compared to the respective negative control group. DMBDD-initiated and promoted with PB or 2-AAF groups were compared to DMBDD-only initiated animals. The number of tumors per group and the number and area of GST-P-positive foci were evaluated by the Kruskal–Wallis test followed by Dunn’s test. The number of tumor-bearing rats and the number of rats with preneoplastic lesions were evaluated by the χ2 or Fisher tests (Hagiwara, Tiwawech, et al. 1993b). Significant differences were assumed when p < 0.05.
Results
Survival, Final Body Weights, and Organ Relative Weights
At the end of the experiments, a significant reduction in the mean final body weights occurred in the DMBDD-initiated male and female control groups and in the 2-AAF-promoted males (Supplemental Material, Table S1).
Animal deaths occurred among the DMBDD-initiated groups beginning at the 27th week. At the end of the experiment, at the 30th week, the average mortality in the 5 studies was 9.1% (8.9% males and 9.3% females), occurring at random and irrespective of treatment. The liver relative weights (referred to final body weights) were significantly increased in DMBDD-only initiated and in PB- or 2-AAF-promoted male and female rats. The mean relative kidney weights did not differ significantly from the respective controls, except the mean weight of DMBDD-only initiated females was increased compared to the negative control group (Table S1).
Liver—Immunoexpression of CYP2B1/2B2 and CYP1A2
In both genders of negative control animals, the intensity of CYP2B1/2B2 immunoexpression was mostly mild/moderate in the perivenular hepatocytes (Supplemental Material—Table S2; Figure 2). In DMBDD-only initiated animals of both genders, the expression of this enzyme was mild and occurred mostly in the perivenular area, but in some rats, it also showed a midzonal or diffuse pattern. PB treatment induced a clearly perivenular expression at high intensity and significantly different from the DMBDD-only initiated groups in male and female animals. Besides the predominant perivenular distribution, some PB-promoted animals also expressed CYP2B1/2B2 with midzonal and diffuse patterns. Both genders of animals exposed to 2-AAF presented significantly increased intensity of protein expression in all hepatic zones.
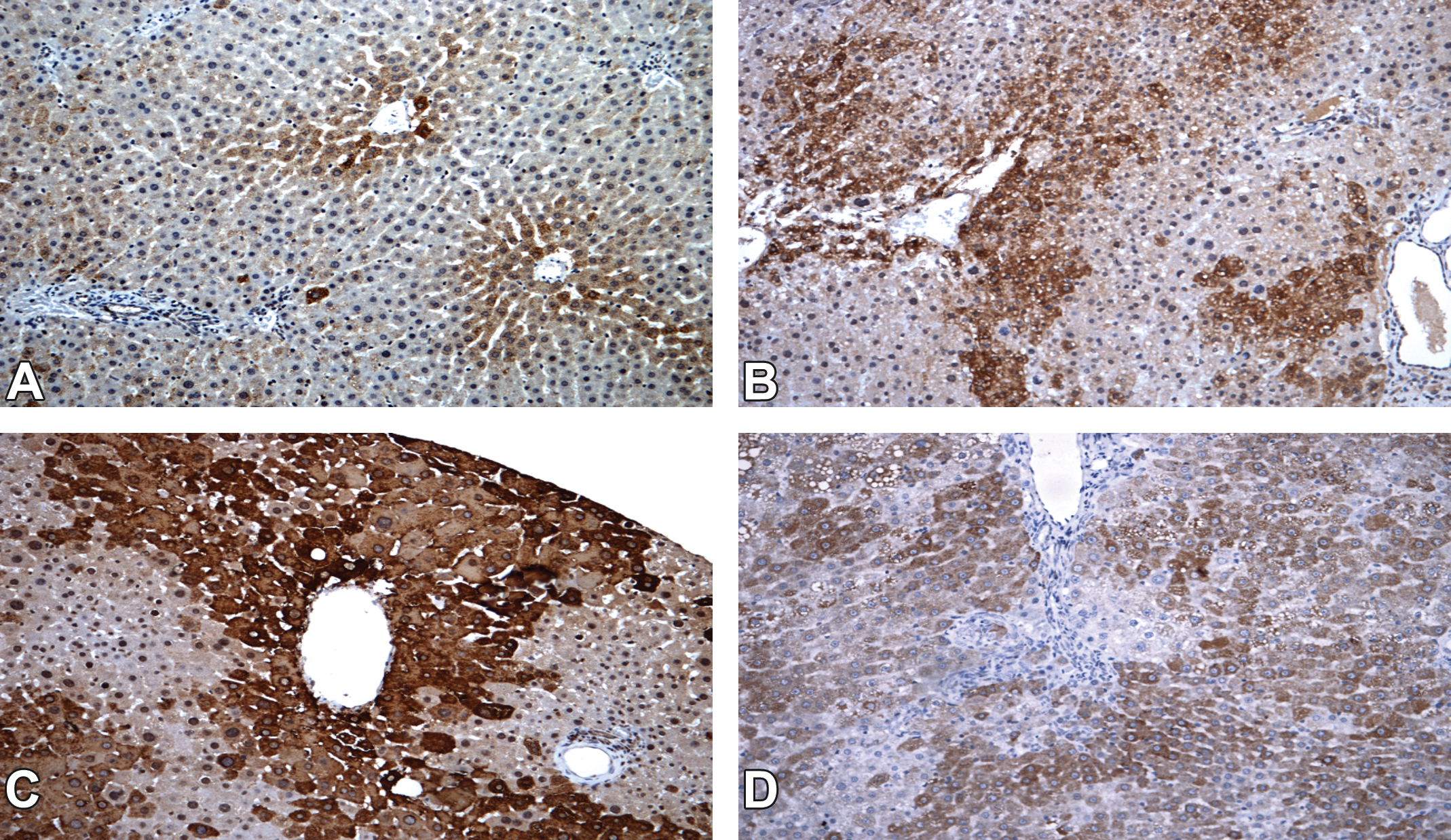
Cytochrome P450 (CYP) 2B1/2B2 immunoexpression in the rat liver (20×). (A) CYP2B1/2B2, negative untreated control group, (B) CYP2B1/2B2, DMBDD-initiated control group, (C) CYP2B1/2B2, DMBDD+phenobarbital, and (D) CYP2B1/2B2, DMBDD+2-acetylaminofluorene.
The immunoexpression of CYP1A2 (Supplemental Material—Table S3; Figure 3) in the negative control group was restricted to the perivenular zone with mild/moderate intensity. In DMBDD-only initiated animals, the same area was mildly stained, but with more animals expressing the protein diffusely (30% males and 40% females). In PB-exposed animals, the expression of this CYP isoform did not differ significantly from the respective DMBDD-initiated control, presenting basically the same intensity in the same areas in both genders. The 2-AAF promotion induced a diffuse and significantly increased intensity of CYP1A2 expression in male and female animals.
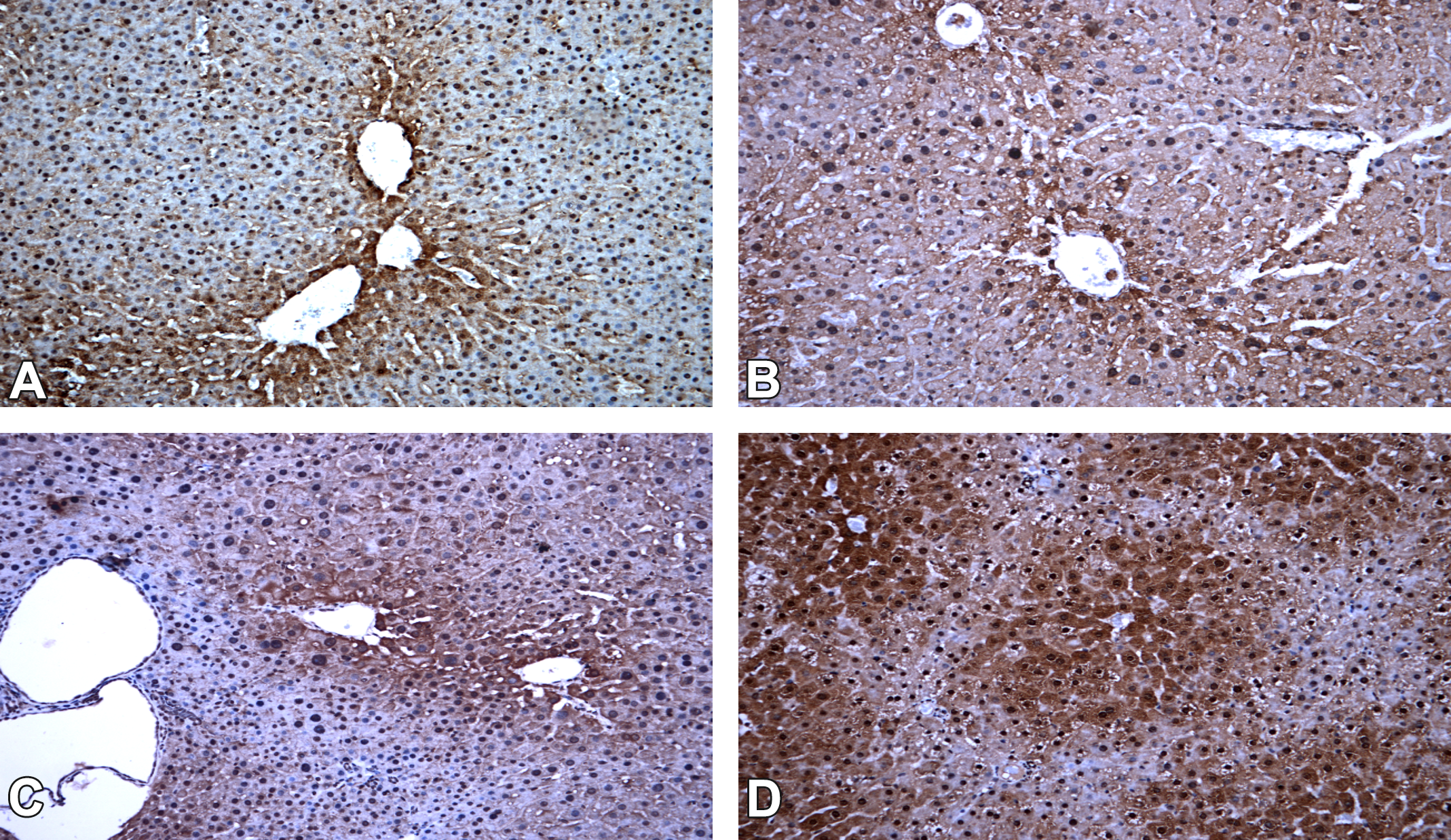
Cytochrome P450 (CYP) 1A2 immunoexpression in the rat liver (20×). (A) CYP1A2, negative untreated control group, (B) CYP1A2, DMBDD-initiated control group, (C) CYP1A2, DMBDD + phenobarbital, and (D) CYP1A2, DMBDD+2-acetylaminofluorene.
Preneoplasia and Neoplasia
Using a pooled occurrence of preneoplasia and neoplasia in different organs, the DMBDD treatment significantly increased their incidences in both male and female rats compared to the negative untreated control group (Tables 1 and 2). These initiated animals also had significantly increased numbers of benign and malignant tumors, attaining an average of 1.87 tumors per animal in each gender. Irrespective of its level in the diet (0.05%, 0.075%, and 0.10%), PB increased the incidences of preneoplasia and neoplasia in both male and female rats and also increased the amount of benign and malignant tumors compared to the levels observed in the DMBDD-only treated groups. Although fewer DMBDD-initiated animals were treated with 2-AAF in the second stage of this protocol, significantly increased incidences of preneoplasia and neoplasia were also registered in both genders, particularly in males, when compared to the DMBDD-only treated groups. The DMBDD + 2-AAF-treated animals presented the highest number of tumors per animal. Overall, these observations document the effectiveness of carcinogenesis initiation by the DMBDD treatment and the promotion by PB- or 2-AAF in both male and female Wistar rats.
Male Wistar Rats Undergoing the DMBDDb Protocol.a
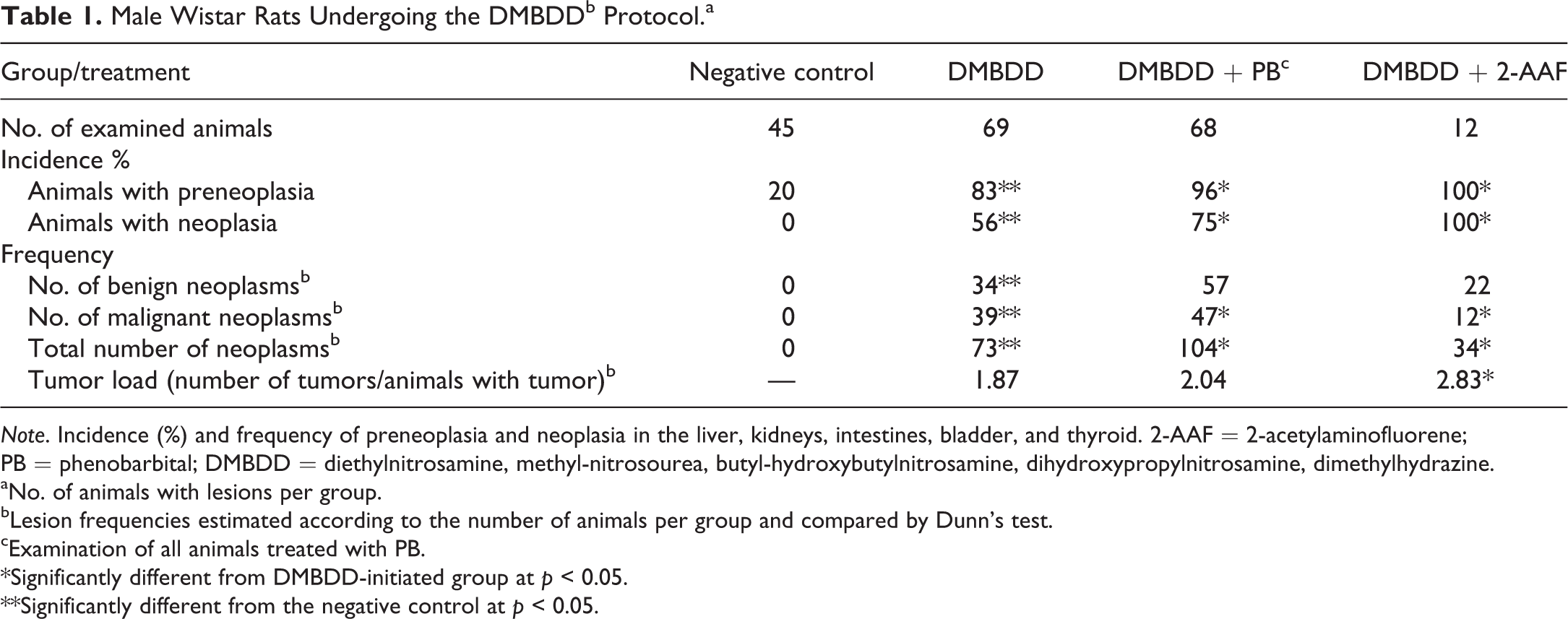
Note. Incidence (%) and frequency of preneoplasia and neoplasia in the liver, kidneys, intestines, bladder, and thyroid. 2-AAF = 2-acetylaminofluorene; PB = phenobarbital; DMBDD = diethylnitrosamine, methyl-nitrosourea, butyl-hydroxybutylnitrosamine, dihydroxypropylnitrosamine, dimethylhydrazine.
aNo. of animals with lesions per group.
bLesion frequencies estimated according to the number of animals per group and compared by Dunn’s test.
cExamination of all animals treated with PB.
*Significantly different from DMBDD-initiated group at p < 0.05.
**Significantly different from the negative control at p < 0.05.
Female Wistar Rats Undergoing the DMBDDb Protocol.a
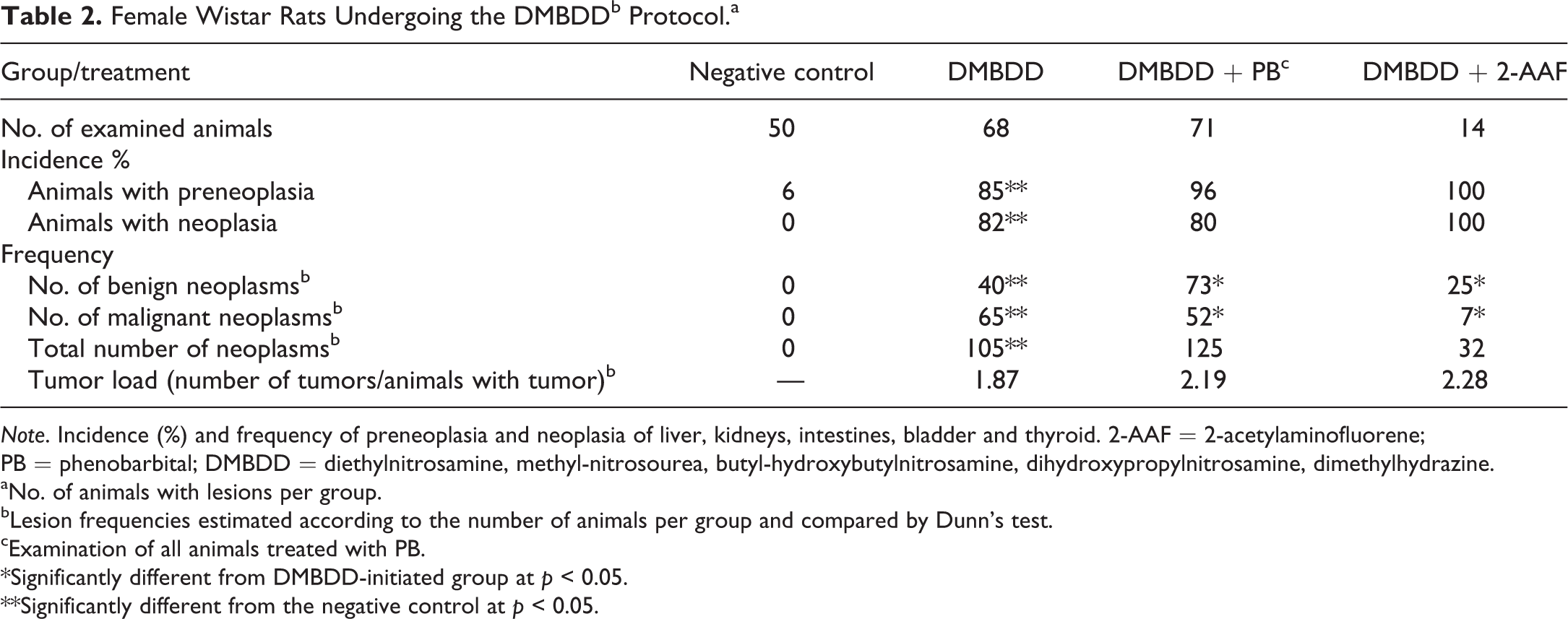
Note. Incidence (%) and frequency of preneoplasia and neoplasia of liver, kidneys, intestines, bladder and thyroid. 2-AAF = 2-acetylaminofluorene; PB = phenobarbital; DMBDD = diethylnitrosamine, methyl-nitrosourea, butyl-hydroxybutylnitrosamine, dihydroxypropylnitrosamine, dimethylhydrazine.
aNo. of animals with lesions per group.
bLesion frequencies estimated according to the number of animals per group and compared by Dunn’s test.
cExamination of all animals treated with PB.
*Significantly different from DMBDD-initiated group at p < 0.05.
**Significantly different from the negative control at p < 0.05.
Putative preneoplastic hyperplastic lesions and benign and malignant neoplasms occurred in the liver, large intestine, kidney, urinary bladder, and thyroid in both genders (Tables 3 and 4), as presented in the following section.
Incidences (%) of Preneoplastic and Neoplastic Lesions at the End of the 30th Week of Experiment in Male Wistar Rats Undergoing the DMBDDb Protocol.
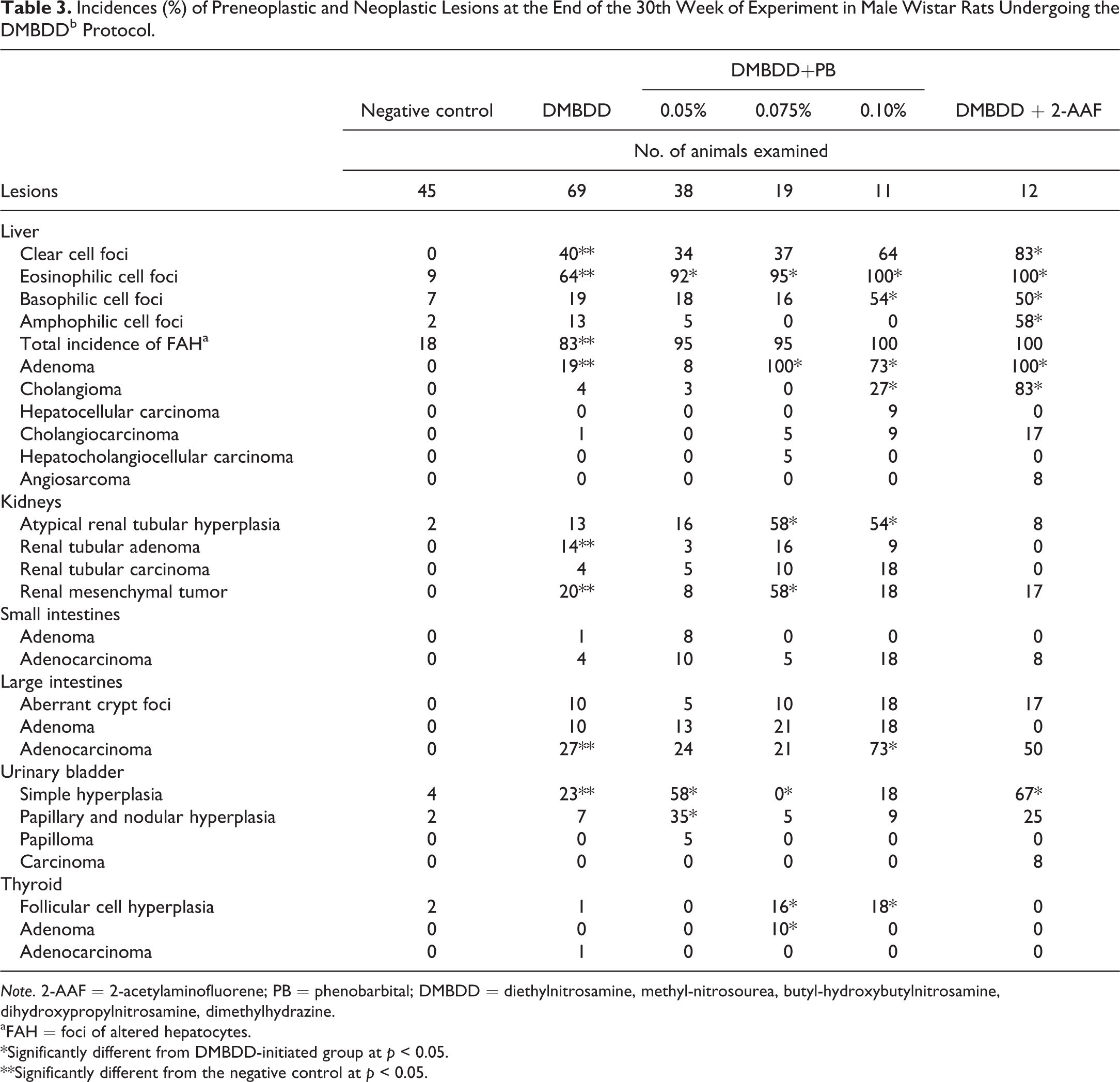
Note. 2-AAF = 2-acetylaminofluorene; PB = phenobarbital; DMBDD = diethylnitrosamine, methyl-nitrosourea, butyl-hydroxybutylnitrosamine, dihydroxypropylnitrosamine, dimethylhydrazine.
aFAH = foci of altered hepatocytes.
*Significantly different from DMBDD-initiated group at p < 0.05.
**Significantly different from the negative control at p < 0.05.
Incidences (%) of Preneoplastic and Neoplastic Lesions at the End of the 30th Week of Experiment in Female Wistar Rats Undergoing DMBDDb Protocol.
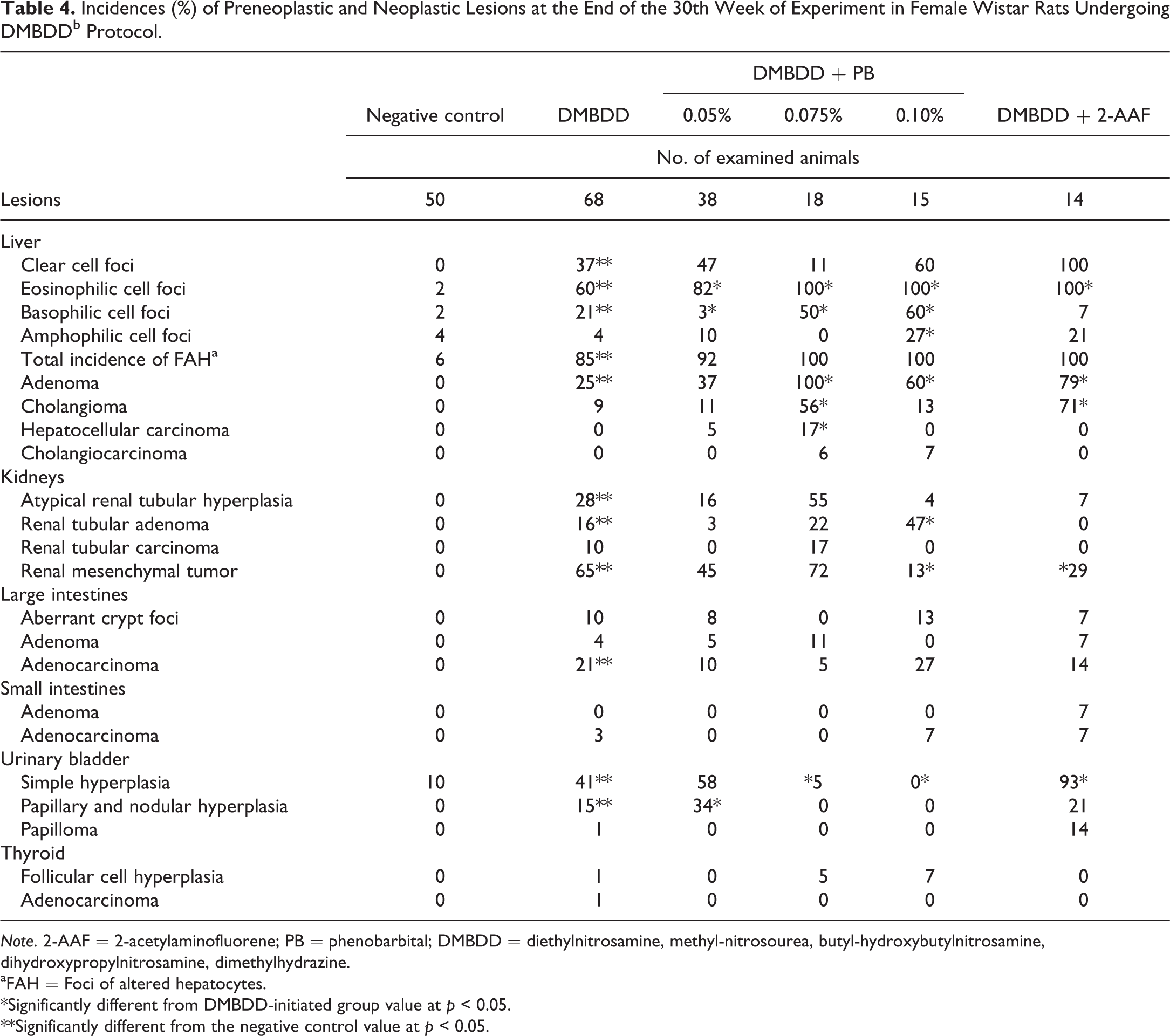
Note. 2-AAF = 2-acetylaminofluorene; PB = phenobarbital; DMBDD = diethylnitrosamine, methyl-nitrosourea, butyl-hydroxybutylnitrosamine, dihydroxypropylnitrosamine, dimethylhydrazine.
aFAH = Foci of altered hepatocytes.
*Significantly different from DMBDD-initiated group value at p < 0.05.
**Significantly different from the negative control value at p < 0.05.
Liver
DMBDD-only initiated animals developed significantly increased incidences of FAH in both genders (males, 83%; females, 85%) compared to the negative control groups (males, 18%; females, 6%; Tables 3 and 4). The predominant types of FAH were the clear (males, 40%; females, 37%) and the eosinophilic cell foci (males, 64%; females, 60%), and in lower incidences, the basophilic and amphophilic types. In both genders of the DMBDD-initiated and PB- or 2-AAF-promoted groups, the incidences of all FAH were numerically increased (92% to 100%) but not significantly different from the DMBDD-initiated group. Only when specific types of foci were considered, for example, eosinophilic or basophilic foci in PB- or in 2-AAF promoted animals showed significantly increased differences from the DMBDD-initiated animals identified.
Rare (less than 2 foci/cm2) and smaller than 0.15 mm GST-P+ foci were observed in the negative control group, and then only >0.15 mm GST-P+ foci were counted in each treated group. In contrast, DMBDD-initiated groups of each gender presented around 5 to 6 foci/cm2. The promoters PB or 2-AAF induced significant increases in the number of GST-P+ foci in both genders, 8 and 11 foci/cm2 in males, respectively, and 10 and 11 foci/cm2 in females. PB also increased significantly the aggregated area of foci in both genders, mainly in females at dietary concentrations equal to or higher than 0.075%. In 2-AAF-treated groups, this parameter did not significantly differ from the respective DMBDD-initiated controls. Overall, the increased incidences and variety of these liver preneoplastic lesions indicate that the DMBDD-initiation and DMBDD-promoting procedures were effective and allowed discrimination among groups, particularly when specific types of FAH were considered.
The promoting agents PB and 2-AAF increased the incidences of liver adenomas and cholangiomas in male and female rats, PB being more effective in concentrations equal to or higher than 0.075% (Tables 3 and 4). Liver adenomas were observed in treated animals of both genders: in the DMBDD-only initiated animals, there was a significant increase of 19% in males and 25% in females; in rats promoted with 0.075% or 0.10% PB, or 2-AAF, the incidences increased significantly and varied between 73% to 100% in males and 60% to 100% in females. Similarly, cholangiomas promoted by 2-AAF were significantly increased in males (83%) and females (71%). Also, cholangioma incidences were significantly increased in males (27%) exposed to 0.10% PB and females (56%) exposed to 0.075% PB compared to DMBDD-only initiated group.
As expected, the incidences of liver malignant neoplasms were relatively lower than the incidences of benign neoplasm in this organ (Tables 1 and 2). In the DMBDD-only initiated group, no more than 1 cholangiocarcinoma occurred in a male rat. In the groups promoted with each of the 3 concentrations of PB or of 2-AAF, these lesions did not surpass 17% in either gender.
Other organs—Preneoplasia and neoplasia
Atypical renal tubular hyperplasia (ARTH) occurred in DMBDD-only initiated male and female rats, although significantly increased incidences were detected in females (Tables 1 and 2). In PB-promoted groups, there were increased incidences of ARTH in both genders but significant only in males. In animals promoted with 2-AAF, only 1 animal of each gender (males, 8%; females, 7%) presented with ARTH.
Benign and malignant renal tubular neoplasms were induced by DMBDD initiation without PB or 2-AAF influence. Most renal adenomas occurred in DMBDD-only initiated males and females, with significant incidences of 14% and 16%, respectively. With PB treatment, the incidences of adenomas differed significantly from the DMBDD-only initiated animals only in female rats promoted with 0.10% PB. This lesion did not occur in the 2-AAF group. Tubular carcinomas occurred in relatively low incidences in DMBDD-initiated male and female rats not different from the negative control group. Both genders in the PB-promoted groups presented low incidences of carcinomas, not different from the respective controls. The 2-AAF group did not develop any renal tubular carcinoma.
The renal mesenchymal tumor (RMT) occurred in significantly increased incidences in DMBDD-only initiated males (20%) and females (65%). In the PB-promoted animals, the incidence increased in the medium dose (0.075%) of both sexes (58% males and 72% females), being significant in males. In the 2-AAF group, the incidence of RMT (17% males and 29% females) was lower than that in the DMBDD-only initiated animals.
Intestinal preneoplasia (aberrant crypt foci) and adenomas were not observed in the negative control groups and occurred at relatively low and variable incidences in all groups irrespective of the treatments (Tables 3 and 4). Adenocarcinoma did not develop in the small intestine but occurred in higher incidences in the large intestine of both genders of rats promoted by 0.10% PB (73% males and 27% females), being significant in males. This malignant neoplasm also occurred in high incidence in the large intestines of 2-AAF-treated animals (50% males and 14% females).
In the urinary bladder, higher incidences of simple hyperplasia (SH) and papillary and nodular hyperplasia (PNH) occurred in both genders of DMBDD-only initiated and in DMBDD+0.05% PB promoted animals (Tables 3 and 4). The incidences of these preneoplastic lesions were significantly increased in males (58% and 35% for SH and PHN, respectively). In females, the incidence was only significant for the PNH (34%) but was also high for SH (58%). Promotion by 2′-AAF also led to increased SH incidences in male and females rats (67% and 93%, respectively) compared to the DMBDD-only treated animals. PNH incidences were also increased in both genders of this group but not significantly. Urinary bladder neoplasia occurred in rats promoted by 2-AAF, with a 14% incidence of papilloma in females and 8% carcinoma in males.
Thyroid follicular cell hyperplasia occurred in both genders of DMBDD + PB animals exposed to the 2 highest doses of PB (0.075% and 0.10%; 16% and 18%, respectively; Tables 3 and 4). Adenoma incidence was also significantly increased in males treated with the PB middle dose (10%). Thyroid adenocarcinoma occurred only in a single male and female rat of the DMBDD-only initiated group.
The noninitiated males and females presented low incidences of preneoplasms (20% males and 6% females) and did not develop neoplasms in this study. The initiation protocol produced significantly increased incidences of preneoplasia (83% males and 85% females). In the DMBDD-only group, there were predominantly malignant neoplasms in both genders.
The treatment with the promoters PB or 2-AAF increased expressively the preneoplasia incidences of both genders but was not always significantly different from the respective initiated controls. In males, PB treatment increased significantly the number of animals with neoplasia and the total number of neoplasms, particularly the malignant ones. In females, PB treatment had a similar influence but producing mainly benign neoplasms. The 2-AAF treatment caused significantly increased incidences of neoplasm in males, occurring in all animals of this group, especially benign tumors. Although the total amount of neoplasia in this male group was lower than the only DMBDD-initiated group, the tumor load (number of tumors/animal with tumor) was significantly higher than the initiated or even higher than the PB groups. Comparable phenomena occurred with the 2-AAF treatment in female rats.
Discussion
The 2-stage carcinogenesis hypothesis implies a previous exposure to genotoxic initiating agents followed by exposure to the so-called carcinogenesis promoting agents. Herein, both genders of Wistar rats exposed to the DMBDD protocol demonstrated a lower weight gain in relation to the negative control group, suggesting a treatment effect. Water and food consumption (data not shown) and organ relative weights also indicated the influence of the agents administered. Despite the early mortality rates of 8.9% and 9.3% among males and females, respectively, probably due to the initiation protocol, the Wistar rat seemed to have a good resistance against mortality with the DMBDD protocol across the period of 30 weeks of the experiment.
Alterations indicating liver injury were the high and significant incidences of steatosis and centrilobular and/or diffuse hepatocyte hypertrophy in all DMBDD-treated animals of both genders (data not shown). Oval cell and bile duct hyperplasia and cystic degeneration also documented liver toxicity. This scenario may have contributed or was associated with the increased incidence of preneoplastic and neoplastic lesions in this organ.
In long-term rodent studies, hepatic foci of cellular alteration are commonly observed as both spontaneous and chemically induced lesions characterized by H&E staining as tigroid, basophilic, eosinophilic, and clear cell foci (Goodman et al. 1994). In fact, herein this potential first visible stage of cancer formation, which occurs spontaneously in variable proportions in aging rats of different strains (Mohr 1992), occurred in the negative control animals of both genders. All groups initiated by the DMBDD treatment developed high incidences of liver FAH, especially eosinophilic foci. Numerous enzyme alterations have been observed in these foci, the overexpression of glutathione-S-transferase form (GST) being a highly sensitive biomarker which allows quantitative evaluation of the efficiency of the initiation protocol (Goodman et al. 1994; Moore et al. 1987). Significant increases in the number and area of GST-P+ foci were detected in male and female rats undergoing the two highest concentrations of PB; but in the 2-AAF-treated animals, the increase was significant only in the number but not in the area of positive foci.
In addition, the incidences of cholangiomas and cholangiocarcinomas demonstrated that, beyond hepatocyte injury, the bile ductal cells responded to the treatments. In general, the incidences of liver benign neoplasia was slightly higher than the malignant, but the incidences of neoplastic lesions in this organ were low in both genders. In addition, the liver lesion incidences across the different doses (0.05%, 0.075%, and 0.10%) of PB did not follow a dose–response relationship, possibly representing a saturation of metabolic processes (Hoel, Kaplan, and Anderson 1983). Otherwise, our observations indicated more consistent results with a concentration of at least 0.075% PB.
In the rodent liver, many chemical tumor promoters transiently induce the drug metabolizing enzyme systems leading successively to endoplasmic reticulum proliferation, hepatocelular hypertrophy and/or hyperplasia, and eventually tumors (Parkinson 2001). The Cyp2B1/2B2 and 1A2 enzymes evaluated in this study were previously related to induction by PB and 2-AAF, respectively (Burt and Thorgeirsson 1988; Fukushima et al. 2005; Hagiwara et al. 1996; Martignoni et al. 2004; Nims et al. 1987; Parkinson 2001). The induction of these proteins was initially restricted to some regions of the liver acinus (Buhler et al. 1992; Chen and Eaton 1991; Watanabe et al. 2000), depending on the presence of specific nuclear receptors like the constitutive androstane receptor, usually dimerized with the retinoid X receptor, and the aryl hydrocarbon receptor (AhR). When animals receive repetitive injections of an enzyme-inducing chemical, like PB, receptors originally occurring in the perivenular region may also be expressed in the midzonal and periportal hepatocytes as a consequence of enzymatic induction of hepatocytes in all three zones (Watanabe et al. 2000). Our results indicated a zonation for both Cyp isoforms studied, which in the untreated control group were expressed in the acinus perivenular region. Although the DMBDD-initiated animals had already slightly increased expression of the isoforms analyzed in midzonal hepatocytes or through the acinus in general, the treatment with PB increased the number of animals and the intensity of the enzyme signaling for the same region, particularly for Cyp2B1/2B2. However, the same did not occur for the Cyp1A2, which showed only quantitative alterations of the zones already induced by the initiation process. The zonation of Cyp isozyme expression was better visualized with Cyp2B1/2B2 induction, probably because this isoform distribution is well characterized and predominant in the perivenular area (Watanabe et al. 2000). Apparently, Cyps1A1/1A2 are the subject of another type of region-specific expression control, suggesting a distribution of the Ah (aryl-hydrocarbon) receptor throughout the acinus (Buhler et al. 1992). In our study, the isoform 1A2 showed a diffuse distribution and also a heterogeneous precipitation of the protein within the hepatocyte cytoplasm. The group that received 2-AAF demonstrated induction of the isoform 1A2 better than PB in either gender, an activity previously described and expected for this genotoxic agent (Burt and Thorgeirsson 1988; Tateishi et al. 1999). The present results demonstrate that the Cyps2B1/2B2 and 1A2 were induced in all treated groups of both sexes, except in the animals promoted with PB, where the isoform 1A2 effects were minimal.
In the kidneys, there were spontaneous proliferative and nonproliferative lesions in the nontreated negative control group, probably related to the initial phases of chronic progressive nephropathy (CPN; Hard et al. 1995). It was characterized by cystic tubules, tubule dilatation, tubular cell pigmentation, simple tubular hyperplasia, hyaline casts, interstitial mineralization, hemosiderin deposition, and infiltration of mononuclear inflammatory cells (data not shown). These lesions should not be scored as separate entities but incorporated into the overall diagnosis of CPN, a spontaneous, age-related renal disease of rats (Hard et al. 1995). Although CPN can cause illness and death in older rats, its pathogenesis starts at an early stage (2 months to 3 months of age) and progresses continually through the life span of the animal (Anver and Cohen 1979; Seely and Hard 2008). The DMBDD-submitted groups presented a more severe CPN, with increased incidence of the CPN components (especially the simple tubular hyperplasia) but also atypical tubular hyperplasia (Tables 1 and 2). These lesions have been described in several long-term carcinogenic studies, sometimes associated with renal tubule adenomas and adenocarcinomas (Hard 2002; Hard et al. 2007, 1997, 1995), as also observed in the present study. The underlying factors that associate the development of these different lesions are not known but seem to be multifactorial and complex (Seely and Hard 2008). In addition, some nongenotoxic substances, like PB, are nephrotoxic and, therefore, may influence tumorigenesis (Khan and Alden 2002).
The renal mesenchymal tumor (RMT), a highly incident chemically induced neoplasia of the renal connective tissue in the Wistar strain (Hard 1990), was found in both genders of the DMBDD-treated groups, particularly in females. The hallmark of the RMT infiltrative neoplasm is the heterogeneity of connective tissue cell types that represent the multipotentiality of the stem cell, originally occurring within a single tumor (Hard 1990). The RMT is usually rapid growing and predominantly a neoplasm of the young rat, without any gender preference (Hard 1990; Mohr 1992). RMT was more easily inducted by dimethylnitrosamine in immature Wistar rats of both genders than in the rats dosed at the age of 4 months or older (Hard 1979).
In this study, the intestines demonstrated the sequential events of colon carcinogenesis from mucosal atypia (e.g., aberrant crypt foci [ACF]) to malignant tumors (Bird 1995; Takayama et al. 1998). These ACF are found in rats exposed to carcinogens that target the large intestine (Whiteley et al. 1996) but also the mid- and distal small intestine of Wistar rats treated with dimethylhydrazine (Rodrigues et al. 2002). In the DMBDD-treated groups, there was a low incidence of intestinal neoplastic lesions, but a relative increased incidence in the mid- and distal intestine of male and female animals (data not shown). In male Fischer 344 rats, Hasegawa et al. (1994) also found adenocarcinomas in small and large intestines induced by the DMBDD protocol alone. Our results also suggest a low interaction and inconsistent PB and 2-AAF effects on the intestines of these groups, indicating that the neoplasia incidences in this organ were mostly related to the initiation process only.
In the urinary system, the urinary bladder presented two types of spontaneous urinary bladder preneoplasia, simple and papillary hyperplasia, including in our negative control group of both genders. However, the hyperplasia may also be secondarily associated with acute and chronic inflammation induced by cystitis or calculus formation (Shirai et al. 1989). Papillary hyperplasia occurs much more frequently in rats, both as a spontaneous lesion and as an induced lesion (Frith et al. 1995). When induced, the urothelial hyperplasia is the primary response of the rat urinary bladder to urothelial carcinogens, such as BBN and intravesical MNU (Frith et al. 1995; Mohr 1992). In the present study, simple and papillary hyperplasia were clearly induced by the initiation protocol and also promoted by 2-AAF in both genders but were variable in the PB treatment. We also observed that the DMBDD protocol did not induce urinary bladder neoplasms efficiently in either gender.
The thyroid showed a low incidence of follicular cell hyperplasia in the animals exposed to the DMBDD protocol or not treated. It’s well known that diffuse thyroid hyperplasia in animals returns to normal if the endocrine status normalizes, or, in the case of an induced lesion, if the stimulus is withdrawn (Mohr 1994). Chemicals like PB may increase the development of thyroid hyperplasia and neoplasia indirectly, as a result of induction of liver phase 2 enzymes involved in thyroid hormone excretion. The enhanced catabolism and excretion of thyroid hormones leads to feedback to the pituitary to increase thyroid-stimulating hormone release and thyroid stimulation (Capen and Martin 1989). In this study, DMBDD-initiated males treated with 0.075% PB demonstrated a 10% adenoma incidence, probably due to the influence of DHPN, a genotoxic chemical that has the thyroid as a target. However, in general, the DMBDD protocol was not efficient in inducing thyroid lesions, especially in female Wistar rats.
In summary, the DMBDD protocol evaluated in this study established the initiation of carcinogenesis in multiple organs in male and female Wistar rats. Promotion was also effective with the administration of PB but less evident than the promotion exerted by 2-AAF, particularly in males. The initial steps of carcinogenesis were better characterized quantitatively with the incidences of tumors than with the incidences of preneoplasia.
The group of animals submitted only to the DMBDD treatment showed severe injury, showing several tumors by the end of the experiment, likely as a result of initial chemical cytotoxicity, compensatory hepatocellular proliferation, mutations, and reduced opportunity for repair (Tong, Fazio, and Williams 1980; Williams and Iatropoulos 2002) during the following 30 weeks. It’s also known that the rate of spontaneous tumors in the Wistar strain is low (2 to 6, 9%) in either sex (Poteracki and Walsh 1998), and in our study their incidences were relatively scarce, occurring only in 39-week-old animals.
In general, the performance of the Wistar rat strain to the DMBDD protocol was somewhat inconsistent and variable. Actually, the characteristic genetic variability of this rat strain appears to be a limiting factor for preneoplasia and neoplasia induction in the DMBDD assay (Asamoto, Tsuda, Kagawa, et al. 1989; Asamoto, Tsuda, Kato, et al. 1989). The Fisher 344/N rat used for more than 30 years in rodent toxicity and carcinogenicity bioassays developed certain undesirable characteristics over time related to specific colony reproduction (e.g., decreased fecundity, sporadic seizures, idiopathic chylothorax, increased spontaneous mononuclear cell leukemia, and testicular interstitial cells tumor incidences; Dinse et al. 2010). Consequently, these and other results have been constantly discussed by many international organizations such as the National Toxicology Program (NTP-US), which has decided to switch from the F344 rat strain to the Harlan Sprague-Dawley rat for most of its bioassays (King-Herbert and Thayer 2006; King-Herbert, Sills, and Bucher 2009). The NTP indicated that flexibility is important in protocol development (Dinse et al. 2010), the most important goal being to choose the strain with the best profile for the specific chemical to be studied. The Wistar rat strain was chosen to conduct the DMBDDb protocol due to its easy availability in Brazil and the relative resistance of this strain to spontaneous neoplasms (de Camargo 1991; de Camargo et al. 1999b).
Conclusions
The alternative multiorgan chemical carcinogenesis bioassay based on the initiation–promotion paradigm of carcinogenesis can be conducted with the Wistar rat strain. The carcinogenesis process was adequately initiated in the liver, intestine, and kidneys of adult male and female Wistar rats. Preneoplastic and benign and malignant proliferative lesions were found in those organs. Although less evident than with 2-AAF, PB also exerted promoting activity in the liver, particularly in males. The present study allowed the construction of a repository of histological lesions induced in the Wistar rat by a modified 2-step multiorgan chemical carcinogenesis alternative protocol.
Footnotes
Acknowledgments
The authors would like to thank Paulo Roberto Cardoso, Paulo César Georgette, Maria Luísa Ardanaz, and Marcos Roberto Franchi (UNESP, Botucatu/SP Brazil) for technical support. The authors also thank Dr. Samuel Cohen for the English text revision.
Author Contribution
Authors contributed to conception or design (MS, NR, LB, CF, AS, MD, DS, LR, JD); data acquisition, analysis, or interpretation (MS, NR, LB, CF, AS, MD, DS, LR, JD); drafting the manuscript (MS, JD); and critically revising the manuscript (MS, NR, LB, CF, AS, MD, DS, LR, JD). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Sao Paulo Agency for Support of Research (FAPESP).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
