Abstract
Frameworks have been developed to standardize the assessment of carcinogenic potential in the pharmaceutical and agrochemical industries, building upon decades of research. Carcinogenicity is also evaluated during the safety evaluation of food substances, using a comprehensive approach unique to each substance. To better understand these approaches, a retrospective assessment was conducted on the publicly available database of substances notified to the United States Food and Drug Administration (US FDA) as being Generally Recognized As Safe (GRAS). The data contained within these GRAS notifications (GRNs) were reviewed for the methods used to evaluate carcinogenic potential (genotoxicity studies, 2-year bioassays, other pre-clinical animal studies) to identify patterns that could provide an understanding of how this assessment has been conducted for different categories of food substances. While different approaches to the safety evaluation were required to adapt to the unique food substances, the data in all notifications supported the conclusion of safety. The evaluation of food substances for carcinogenic potential must consider all available data, including identifying the need for when more data must be generated to support an evaluation. Due to the complexity of substances used in food, ranging from defined chemical entities to minimally processed agricultural commodities to live microorganisms, the approach to conducting the safety evaluation of food substances must be able to adapt to the most relevant scientifically supported approach. This paper illustrates the data commonly used to support the safety of different types of food substances and proposes an approach familiar to other product sectors.
Introduction
The approach to evaluate carcinogenic potential of all substances, not just those found in food, has historically included rodent cancer bioassays. With the advent of newer technologies, better understanding of cancer biology, and interest to reduce animal experimentation through application of the 3Rs Principle (Replacement, Reduction, and Refinement of animal use), the pharmaceutical and agrochemical sectors have shifted their practices to adopting an integrated weight of evidence (WoE) approach, developing frameworks that include rationales to waive the rodent cancer bioassays.1–3 The food sector also recognizes the need to modernize the approach for the evaluation of carcinogenic potential and have questioned the relevance of animal bioassays to food substances.4,5 In regard to the evaluation of carcinogenic potential, there are unique challenges to food and food substances: foods are complex chemical mixtures that may raise or decrease overall cancer risk, 6 the majority of potential carcinogens found in food are not readily avoidable, overall diet and eating too much food can contribute to cancer risk, 7 and food substances are often derived from foods that have been consumed for decades or centuries. Collectively, these hurdles make the development and application of a one-size-fits-all, checklist safety assessment approach for all food substances challenging.
The US FDA has established two mechanisms for the pre-market review of food substances: the Food Additive Petition (FAP) process, 8 and the Generally Recognized as Safe (GRAS) process (21 CFR 170). Since the establishment of the GRAS Notification (GRN) process in 1998, GRAS has become the primary mechanism for the safety assessment of new food substances to be reviewed by the FDA. Under both the FAP and GRAS processes, the same safety standard is required to be met; all food substances must demonstrate a reasonable certainty of no harm under the conditions of their intended use prior to being added to food (21 CFR 170). Under both the FAP and GRN processes, the FDA conducts a comprehensive review of all available data relevant to the food substance, including (but not limited to) information provided by the submitter/notifier.
One of the differences between the FAP and GRAS processes is that while submission of data to the FDA through the FAP process is confidential, all pivotal data used to support safety of a substance through the GRAS process must be “generally available”. Thus, the FDA GRAS notification inventory represents a robust collection of publicly available information that can elucidate the data that has been used to support the safety of the food substances in question.
The US FDA provides guidance for the safety assessments of food additives, including data requirements, in the Guidance for Industry and Other Stakeholders Toxicological Principles for the Safety Assessment of Food Ingredients, commonly known as the “FDA Redbook”. 9 The FDA has made it clear that the guidance proposed in the Redbook is not always required to meet the standard of reasonable certainty of no harm that applies to food substances. 10 This is illustrated by the approval of food additives based on prior GRAS determinations, for example soybean leghemoglobin as a food color additive.11,12 It should be noted that he term food additive refers to all substances where “the intended use of which results or may reasonably be expected to result, directly or indirectly, either in their becoming a component of food or otherwise affecting the characteristics of food”. 13 While this definition of “food additive” includes a broad spectrum of intentionally added substances, the focus of this paper does not include “indirect food additives” used for contact applications such as in food packaging or “secondary direct food additives” used in the production of foods.
In the Redbook guidance, a food substance’s potential toxicity, as determined by its structural similarity to known toxicants, and its anticipated human exposure based on its intended use, forms the basis for the types of testing that FDA recommends. While the FDA Redbook allows for scientific discretion in toxicity testing, it does recommend a set of studies to establish the safety of a new or expanded use of a food substance. This guidance generally recommends a battery of genotoxicity assays and short-term toxicity testing along with long-term comprehensive pre-clinical toxicity studies, such as the 2-year rodent cancer bioassay, also known as the “2-year bioassay” for food substances with cumulative exposure levels above between 0.25 to 1 ppm, depending on the level of concern as indicated by the structure of the substance. 14
Since 1993, the FDA Redbook has not substantively changed, despite significant progress in the scientific understanding of carcinogenesis, with growing consensus that the 2-year rodent bioassay is limited in its value for assessment of cancer risk to humans. Alternative approaches that build upon accumulated knowledge of cancer processes that are both more human relevant and can help regulatory agencies meet their goal to reduce animal use are available.15–17 A series of three papers18–20 summarizes the history of carcinogenicity research and provides a unifying theory on chemical carcinogenesis. Cohen et al. (2019) propose a decision tree for carcinogenic categorization and risk assessment; a systematic approach to assessing the carcinogenic potential of any chemical substance that can be utilized by across all industry sectors. Through a series of questions, that decision-tree examines whether a substance exhibits signs of being a potential carcinogen, such as whether it’s mutagenic and/or produces effects that are known to precede carcinogenesis (e.g., promotes cell proliferation, immunosuppression, and/or estrogenic activity). In alignment with Cohen and colleagues’ work, pharmaceuticals and agrochemicals have established systematic approaches to evaluate the carcinogenic potential of substances more accurately.3,21
The chemical space that is represented by food substances is broad and encompasses a wide breadth of unique structures that require assessment, adding to the challenge of applying the Cohen 2019 decision tree to food substances. Structured approaches to assess the safety of food substances have been developed by certain niches within the food industry, including those designated solely for the evaluation of food enzymes, 22 proteins used in agricultural biotechnology, 23 microbial cultures, 24 and complex botanical mixtures of substances used as flavors. 25 Here, a retrospective analysis of publicly available FDA GRAS notifications (GRNs) was undertaken to identify trends in the use of data to support the carcinogenic assessment of food substances. This analysis was then compared to the approach presented in the Cohen 2019 decision tree to evaluate whether learnings from these new approaches to carcinogenic assessment can be leveraged in either the conduct and/or the description of data used to evaluate food substances.
Materials and methods
GRAS notice review process
187 GRAS notifications (GRNs) numbered between 680 to 900 which received a response stating “no questions at this time” from the FDA were reviewed.
26
These notifications serve as a representative subset of notifications submitted to the FDA after the finalization of the Final Rule on GRAS.
27
Data captured from each notified GRN were as follows (Supplemental Table 1): • Category of the food substance (further defined below). • Genotoxicity studies on the food substance itself or a similar substance. • Pre-clinical repeat dose toxicology studies on the food substance itself or a similar substance. o Studies of a duration shorter than 13-week in length were captured only if no other referenced studies of at least 13-week in duration were cited. • 2-year bioassays on the food substance itself or a similar substance
Food substance categorization
Food substances were assigned to one of five categories based on the following criteria: • Defined chemical entity: Food substances composed of >60% of a single, unique chemical. o Examples include: Ergothionine – GRN 734, 2’-fucosyllactose – GRN 735, and Orange Extract (85% hesperidin) – GRN 796 • Concentrated mixture: Food substance composed of multiple defined chemical entities where the total of the defined chemical entities together make up > 60% of the food substance. o Examples include: Stevia leaf extract – GRN 768, Fructooligosaccharides – GRN 797, and Monoacylglycerides from grape seed – GRN 886 • Minimally processed substance: Food substance that is a food or derived from a food that has been processed but still represents the chemical composition of the source of the substance. o Examples include: Hemp seed oil – GRN 778, Pea proteins – GRN 803, and Corn oil – GRN 900 • Isolated Protein: Food substance that is a specific isolated protein. o Examples include: Multiple enzymes including GRN 743, 751, and 825, Alpha-lactalbumin – GRN 763, and beta-lactoglobulin – GRN 863. • Microorganism: Food substance that consists of an intact (live or dead) microorganism. o Examples include: Multiple live organisms including GRN 685, 760, and 814, Preparations of bacterial phages including GRN 724 and 827, and inactivated Bacillus coagulans – GRN 725.
The 187 GRNs consisted of 43 defined chemical entities, 33 concentrated mixtures, 47 minimally processed substances, 35 isolated proteins, and 29 microorganisms (Figure 1). The subject of GRN 706, Siraitia grosvenorii (Swingle fruit extract), was considered as both a concentrated mixture and minimally processed substance, since its notification included multiple ingredients that belonged in different categories. Distribution of food substances contained within 187 GRNs across five categories.
Distinction between studies on the substance itself versus on a different but relevant substance
Studies that served as the basis for the safety determination for the 187 GRAS substances were captured with distinction of whether the study was conducted on the substance itself and/or on a relevant substance (Supplemental Table 1).
Studies on the substance itself refer to studies performed on the notified substance itself or on substances that were essentially the same with the same or a similar specification. To illustrate, the notification for galactooligosaccharide (GRN 729) did not include any genotoxicity or pre-clinical studies conducted on the specific galactooligosaccharide ingredient produced by the manufacturer. However, the notification concluded that the galactooligosaccharide produced with this manufacturing process was substantially similar (aligned with the specification of the finished ingredient) to the galactooligosaccharide evaluated in genotoxicity or pre-clinical studies from prior GRNs. Therefore, these studies were captured as being conducted on the substances itself. This approach was also applied to other GRNs in similar circumstances.
Studies on a relevant substance refer to studies that were conducted on a structurally related substance which may have a different composition but remains relevant for the safety of that notified substance. For example, in the notification for hemp seed oil (GRN 778), the notifier referenced studies conducted on tetrahydrocannabinol (THC), which is derived from the same food source from which the oil is extracted. The THC safety studies were considered relevant for the safety evaluation of hemp seed oil. In another example, notifications for concentrated mixtures of steviol glycosides (e.g., GRNs 702, 733, 744, 745) referenced studies on a single steviol glycoside.
Results
Data supporting carcinogenic risk assessments of food substances
Scientific studies to evaluate the carcinogenicity potential for each notified food substance were noted, including genotoxicity studies (e.g., Ames, micronucleus, chromosomal aberration, unscheduled DNA synthesis, sperm malformation, thymidine kinase assay), pre-clinical repeat-dose studies (e.g., 13-week rat studies, development and reproductive toxicity (DART) studies), and the 2-year rodent bioassay. These endpoints were selected based on their relevance to carcinogenic risk assessments 18 and frequency of use as a core battery of testing. For each referenced study, it was noted whether the subject of the study was the substance itself or a related substance (Supplemental Table 1). Additional details regarding how it was determined whether data was for the substance itself or a related substance are described in the Materials and Methods section. Twenty-five percent (47/187) of FDA GRAS Notifications (GRNs) only referenced toxicity studies on a relevant substance but not the substance itself; this read-across approach was spread across each of the five food categories, with frequencies of read-across of data within each category ranging from 9–36%.
Data supporting carcinogenic risk assessments. Values represent % of GRNs that contained study types indicated for each food substance category.

GRAS Notifications lacking both genotoxicity and in vivo toxicity studies.

Genetic toxicity assays based on food category.
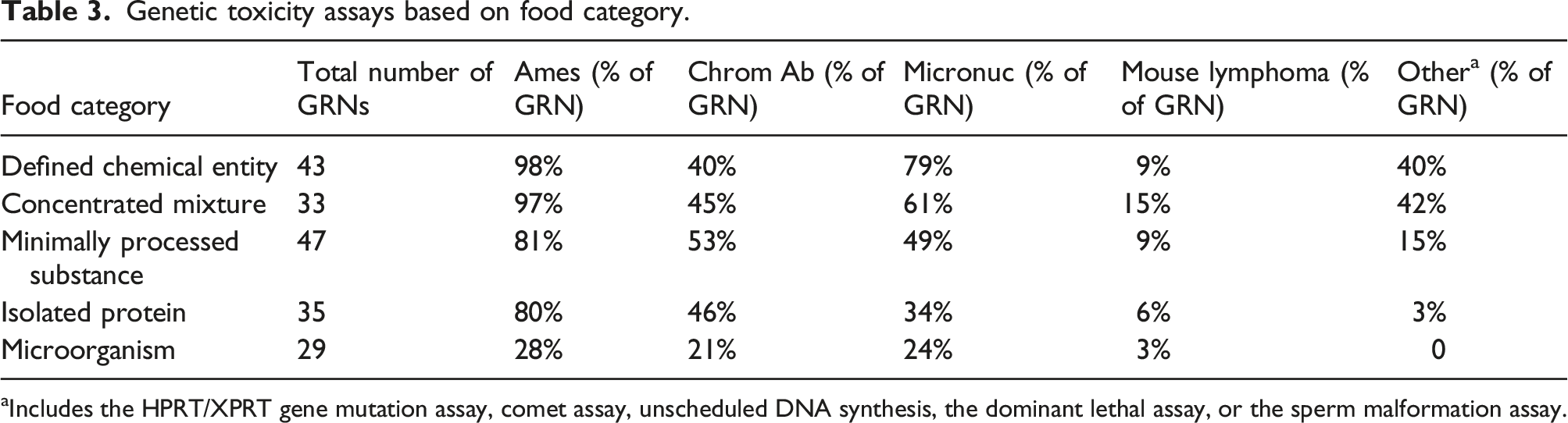
aIncludes the HPRT/XPRT gene mutation assay, comet assay, unscheduled DNA synthesis, the dominant lethal assay, or the sperm malformation assay.
Pre-clinical repeat dose studies based on food category. Values represent % of GRNs that have the study types indicated.

Roughly a quarter (46/187) of GRNs referenced a 2-year bioassay on the notified substance itself or a relevant substance (Table 4). Several of the 2-year bioassays were referenced in multiple GRNs for the same or relevant food substance.
Discussion
Evaluation of data supporting the carcinogenic evaluation of food substances in GRNs
A retrospective analysis of publicly available FDA-notified GRAS determinations (GRNs) was undertaken to identify the studies used to support the carcinogenic evaluation of these food substances. Data were analyzed by establishing five categories of food substances (concentrated mixture, defined chemical entity, isolated protein, microorganisms, and minimally processed substances) based on their composition and purity. It should be noted that this retrospective analysis only included a review of information in the FDA GRAS notification inventory. A separate comprehensive review of each food substance to identify additional studies was not conducted, although both the notifier and FDA conduct such an analysis at the time of review. This also means that studies published after the notification would not have been identified as part of this retrospective analysis.
An overall assessment on the frequency of both genotoxicity and pre-clinical repeat dose studies was conducted for each of the five categories of food substances. These studies represent the foundation for the safety evaluation of food substances as recommended by many food risk assessment agencies9,28–30 and therefore it is not surprising that these were the most common studies referenced in these notifications. While the majority of GRNs for the concentrated mixture, defined chemical entity, and isolated protein categories referenced genotoxicity or pre-clinical repeat dose studies (>80% and >70% for genotoxicity and pre-clinical repeat dose studies, respectively), GRNs for the microorganism category only referenced a genotoxicity or pre-clinical repeat dose studies 28% and 52% of the time, respectively (Table 1). A small percentage (15%) of GRNs categorized under concentrated mixture, minimally processed substance, isolated protein, and/or microorganism did not reference any toxicity study (Table 2). Instead, the safety narrative for these notifications relied on a long, well documented dietary history, an analysis of whether the substance was in a chemical class known to have carcinogenic potential, as well as a carcinogenicity assessment of relevant constituents.
The FDA Redbook recommends evaluation of two potential genotoxicity mechanisms: gene mutations and chromosomal effects (structural chromosome aberrations and numerical chromosome aberrations). Furthermore, the Redbook provides guidance that when the cumulative estimated dietary intake exceeds 1.5 µg per person per day (0.5 ppb) from all dietary sources the following assays should be conducted: (1) an Ames assay, (2) in vitro chromosomal aberration assay or an in vitro mouse lymphoma tk+/− assay, and (3) an in vivo micronucleus assay. Alternatively, the European Food Safety Authority (EFSA) 28 provides slightly different guidance to cover these two potential genotoxicity mechanisms, with a first step being a ‘basic battery’ of (1) an Ames assay, and (2) an in vitro micronucleus assay, followed in case of any positive or equivocal result by a confirmatory assay, which is usually an in vivo micronucleus assay. Aligned with both of these guidances, the Ames assay was the most frequently referenced genotoxicity assay, being cited in 80-98% of GRNs from all categories except the microorganism category, where it was only referenced in 28% of the GRNs. In line with FDA Redbook recommendations, 88% of notifications that referenced an Ames assay also cited chromosomal aberration test or micronucleus assays. Of the genotoxicity studies recommended by the FDA Redbook guidance, the mouse lymphoma tk+/− assay was the least referenced (Table 3).
The 2-year bioassay including in utero exposure is currently recommended by the FDA Redbook for food substances that have a concentration in food >0.25 ppm, dependent on the structural class of the substance. 14 Many of the notified food substances included in this analysis have cumulative daily exposure that exceed 1 ppm based on intended use, however, the 2-year bioassay was cited in only 47% of defined chemical entity notifications, 48% of concentrated mixture notifications, and 21% of minimally processed substance notifications (Table 4). The 2-year bioassay was not referenced in any of the isolated protein or microorganism notifications included in this review.
Interestingly, while in the FDA Redbook higher levels of exposure serve as a factor in testing recommendations, our analysis suggests that cumulative exposure is not the only determinative factor in toxicity testing. This is exemplified most clearly in the minimally processed substance category where several notifications of substances that are intended to be used as a macronutrient component (e.g., protein), did not reference a genotoxicity or pre-clinical repeat dose study (Table 2). Macronutrient substitutions would undoubtably result in high levels of cumulative exposure, yet other prevailing factors indicated that extensive toxicity testing was not required to establish safety for these substances. The commonality among these notifications is that the substances are compositionally similar to established components of the diet that have a long history of safe consumption (e.g., pea protein, apple peel powder, anhydrous milk fat), and belong to foods that have a long history within the food chain without being associated with carcinogenic potential.
Two-year bioassay studies referenced in GRAS determinations.

aSubstance the study was conducted on is considered a relevant substance, and not the substance itself.
bStudy is not published/generally available.
This retrospective analysis of GRNs identified a shift from the reliance on the 2-year bioassay to a combination of the 13-week rodent study and a standard genotoxicity battery for substances of relatively low concern based on other evidence. Data analysis reveals that the 13-week rat study was routinely referenced in more than 74% of the GRNs from the defined chemical entity, concentrated mixture, and minimally processed substance categories (Table 4), whereas the 2-year bioassay was cited in less than half of the GRNs for these categories. Even more strikingly, 69% and 41% of notifications for isolated proteins and microorganisms referenced a 13-week rat study, respectively, while none of these notifications referenced a 2-year bioassay.
The decreased reliance on the 2-year bioassay is supported by the Cohen et al. (2019) decision tree for assessing carcinogenic potential. The Cohen et al. (2019) decision tree relies on indications of genotoxicity through testing and structural analysis, cumulative exposure, and assessing for specific modes of action that are known to increase cell proliferation, endocrine activity, and/or immunosuppression and are relevant for development of cancer in humans. For substances that are not mutagenic, the absence of increased cell proliferation as well as effects that may precede or occur alongside cell proliferation such as hypertrophy or metaplasia in a 13-week rat study31,32 is predictive of the substance not producing cancer in a 2-year bioassay. Other industries have begun to adopt formal systemic approaches for the evaluation of carcinogenic potential based on this work,3,21 which have many benefits including reducing the need for animal testing and increasing the human-relevance of the assessment. To determine if a formalized systemic approach to assessing carcinogenic potential could be developed for food substances, the retrospective analysis of GRNs was further reviewed in the context of the decision tree proposed by Cohen et al. (2019).21,31,32
Applicability of the Cohen et al., 2019 decision tree to food substances
One aspect of the Cohen et al. (2019) decision tree is to assess mutagenicity potential through structure activity relationship analysis, genotoxicity testing, and Cramer classifications linked to the relevant Threshold of Toxicological Concern (TTC). One consideration unique to food substances is that many substances (especially within the minimally processed substances and concentrated mixture categories) are not discrete chemicals substances that can be easily evaluated using traditional toxicological classifications such as Cramer Classifications. Additionally, substances such as microorganisms (GRN 685, 758, 760, and 798), and bacterial colicin and bacteriophage preparations (GRN 676, 724, 757, and 775), cannot be evaluated with standard genotoxicity studies such as the Ames assay. This in itself does not preclude their inclusion in a systematic approach for carcinogenic assessment of food substances, as a systematic approach for the safety assessment of one of these groups of substances, microorganisms, 24 is in regular use today.
Following a genotoxicity assessment which is negative, the Cohen et al. (2019) decision tree considers non-DNA reactive mechanisms of carcinogenesis, which is most frequently through mitogenesis or cytotoxicity-induced cell proliferation. 33 For chemicals where exposure exceeds the relevant TTC for the structural class, Cohen et al. (2019) suggests the use of rodent study up to 13 weeks in duration to detect key events that are indicative of proliferation. This raises another complexity for food substances, in that exposures described in GRNs tend to be much higher for consumers than estimated exposures to other substances from other sectors such as pharmaceutical or agrochemical. Therefore, while the TTC has been leveraged for carcinogenicity and general toxicity assessments for some food substances (e.g., flavors, food contact substances) this part of the framework may not be as useful for food substances reviewed through the GRAS process.
Strict adherence to the Cohen et al. (2019) decision tree would lead to a frequent recommendation for conducting 13-week animal studies for food substances. However, food substances often have the advantages of both belonging to chemical classes for which extensive read across is possible, including in many cases a long-established dietary history for the comparator material, and often have a low potential for biological activity. It is our recommendation therefore that a WoE approach is more appropriate for determining the need for animal testing for food substances.
Simplified elements to standardize communication of carcinogenic risk assessment for food substances
Reflecting upon the Cohen et al. (2019) decision tree in the context of aspects that do not fit well with the types of substances and exposures from food, there remain aspects that have utility for food substances. The proposed approach focuses on evaluating and documenting the weight of scientific evidence that supports reasonable certainty that the food substance would
E1: Does the weight of the scientific evidence indicate potential for mutagenicity?
E2: Does the weight of the scientific evidence indicate a potential for increased cell proliferation?
It is important to note that these elements could be used to evaluate/document existing data as part of a safety assessment but could also be used earlier in the process as a guide to identify data gaps and additional studies that may be warranted. The following section discusses in greater detail how a user could use this approach to document that a food substance is unlikely to present a carcinogenic risk.
Additionally, this approach is specific to the food substance itself, and is not meant to address unintentionally present contaminants and residues (e.g., heavy metals, mycotoxins, pesticides). Developing specifications for food substances that control these compounds is a critical step in establishing the safety of a food substance, and a critical element of any GRAS determination. However, the development of a specification for unintentionally present compounds is a separate exercise than evaluating the safety of the food substance itself.
E1: Does the weight of the scientific evidence indicate potential for mutagenicity?
The assessment of potential mutagenicity should be a holistic evaluation of all available data, including from similar substances, acknowledging that a positive result in one assay should not lead to a conclusion that a substance is either mutagenic or not without considering all available data. This element demonstrates how this approach could be used to both evaluate existing data and aid in identification of gaps in data. When evaluating the existing data for a food substance, if the data show a clear lack of mutagenicity (e.g., a negative result in both an Ames assay and an in vitro micronucleus assay, with no conflicting studies) the notifier could document this data as evidence that a food substance is not mutagenic. However, if there is no data regarding the food substance (or similar substances) or conflicting results from genotoxicity studies, this would flag to the notifier that additional information would need to be generated to establish reasonable certainty. The assessment of DNA reactivity (E1) and chromosomal effects with non-animal methods, which are often included in the definition of New Approach Methodologies (NAMs), are already broadly accepted as an appropriate approach to evaluating this endpoint.
Notifications for minimally processed substances closely related to traditionally consumed foods are a good example of food substances that both belong to chemical classes with a low potential for adverse effects and a long-established dietary history. In many of the notifications for minimally processed substances, neither genotoxicity studies nor pre-clinical repeat dose studies were referenced and the safety narrative for these substances was based on reference to previously available evidence that the class of substances do not activate mechanisms that produce cancer (e.g., lack of mutagenicity and increase of cell proliferation) along with a history of safe use.
The notification for beta-lactoglobulin produced via precision fermentation (GRN 863) represents such an example. The notification for this food substance provides a detailed characterization of this protein and a comparison with traditional whey protein. The notifiers describe the substantial similarities between this beta-lactoglobulin with the milk proteins that have been consumed historically, and reference that other than milk-allergic individuals, there are no reported adverse effects in the literature tied to consumption of milk or milk proteins. The notifiers also indicate that presence of this beta-lactoglobulin in foods would not result in an increase in the estimated intake of milk proteins, as the intended uses were as substitutions for traditional whey protein. If the beta-lactoglobulin had significant compositional differences (e.g., different amino acid composition) than that found in cows’ milk, it may be determined that there was not sufficient evidence to answer this question, because there would not be an ability to rely on the existing data for intact milk proteins.
Desugared orange juice (GRN 679) highlights another aspect that should be considered when evaluating the weight of the evidence for a food substance in the context of E1 (and E2). In this case, the notifier provided a detailed characterization of the food substance, listing the most prevalent components: sugars, vitamin C, carotenoids, flavonoids, and folates, none of which would be different in this food substance in comparison with standard orange juice. Thus, in some cases (especially minimally processed substances) it would be important to document the characterization of the food substance to ensure that there are no components that themselves would raise concern.
E2: Does the weight of the scientific evidence indicate a potential for increased cell proliferation?
The majority of notifications reviewed provided evidence from a 13-week rat study which would provide support to the conclusion that a food substance does not act through increased cell proliferation. Also, like evaluation of mutagenicity, the broad chemical space represented by food substances also requires a flexible approach to evaluation, which in turn allows for the potential to reduce animal experimentation.
The assessment for E2 would encompass results from specific safety studies and a review of all available evidence, including whether the substance belongs to a class of chemicals that have a plausible mechanism through which effects that may lead to cell proliferation may act. As cytotoxicity is a pro-proliferative effect, established in vitro cytotoxicity assays can be combined with knowledge or prediction of systemic and target site concentrations to understand if such a mechanism is relevant. Other proliferative mechanisms are estrogenic and immunosuppressive activity. 18 Structure activity relationships and screening tests for estrogenic activity are well established 34 and may be recommended if information on the food substance itself, or a suitable comparator, are not available. Similarly, there are in vitro schemes for evaluating immune effects including immunosuppression available. 35 Overall, a substance-specific evaluation of whether these proliferative effects are likely could recommend additional assessment such as through methods that have been proposed elsewhere. 36
It is important to be pragmatic when considering the potential for proliferative effects, just as it is for other endpoints. Using live microorganisms as an example, there have been no reports of consumption of these microorganisms inducing cell proliferation nor is there any plausible mechanism through which they would produce this effect. Alternatively, as part of the weight of evidence approach for these food substances, GRAS determinations of microorganisms invariably consider finished product purity and whether the selected strain may produce toxigenic substances. 24 Therefore, while data from classic safety studies like the 13-week rat study would be informative for many food substances in answering this question, this should not be interpreted as this type of data being mandatory for every food substance.
In our review of the GRNs, 41-95% of substances referenced a 13-week rat study across the five categories of substances. It should be noted that none of the 13-week rat studies reported results that indicated increased cell proliferation via the mechanisms described above. However, recent developments in NAMs have resulted in the use of studies other than traditional pre-clinical repeat dose studies (e.g., 13-week rat study) becoming more common, especially in other sectors.35,37–40 As these methods gain further acceptance it is likely that they will be more frequently referenced in GRNs.
Is the mode of action relevant to humans via exposure through food?
Another consideration highlighted in the Cohen et al. (2019) decision tree is whether the mode of action of substance is relevant to human health. In the approach proposed here, an evaluation of whether the mode of action is relevant to humans via exposure to food would be included in the weight of the evidence approach to E1 and E2.
In the 187 notifications reviewed, there were no examples where the weight of the evidence would suggest the food substance would either be mutagenic or produce uncontrolled cellular proliferation. Thus, while having to consider whether a result is relevant to human exposure through food is expected to exceedingly rare, this approach recognizes that there are substances where consideration of human relevance of data is scientifically valid. In other industries, consideration of human relevance has been critical for the risk assessment of substances. In one of the most well-known examples, carcinogenesis can be produced in male rats through a mechanism involving alpha-2u globulin nephropathy. 41 However, it has also been conclusively demonstrated that this mechanism does not occur in human (nor female rats), and this is taken into consideration for the risk assessment of substances that produce this specific tumor type in rats. As the risk assessment of food substances advances in parallel with risk assessment of substances in other industries, the human relevance of effects should also be considered.
Summary of the proposed approach
The US FDA GRAS process requires that all substances intentionally added to food can demonstrate reasonable certainty of no harm under the proposed conditions of use. Demonstrating reasonable certainty of no harm includes consideration of whether the food substance has the potential to cause cancer due to specific mechanisms: (1) Mutagenicity, and (2) Increasing cellular proliferation. In this review of 187 GRNs, all food substances documented evidence that would rule out activity via these mechanisms. It is important to note that food substances are very diverse and flexibility in an approach is critical. The retrospective review of GRNs established that these elements are being addressed, leveraging data that is appropriate for the specific food substance. However, in comparison with other sectors and the Cohen et al. (2019) decision tree, there could be a benefit in adopting a standardized approach to addressing these elements in the narrative of a GRAS determination.
Having flexibility in the approach to demonstrate lack of carcinogenic potential is also crucial to ensure that as NAMs and other alternative methods are developed, which may have greater sensitivity and specificity than current models, that the approach can be leveraged to address these questions. Therefore, while consideration of carcinogenic potential is already part of the process for determining the safety of food substances, establishing a standardized approach in which carcinogenic mechanisms are directly addressed as part of a GRAS determination would provide consistency, clarity, and transparency to the process.
Supplemental Material
Supplemental Material - Retrospective analysis of carcinogenicity assessments within FDA-notified GRAS determinations
Supplemental Material for Retrospective analysis of carcinogenicity assessments within FDA-notified GRAS determinations by Khatera Rahmani, Yen-Ching Wu, Neil R. Buck, Alexandria Lau and Paul R. Hanlon in Human & Experimental Toxicology.
Data Availability Statement
The information analyzed as part of this work about the GRAS notifications is available on the United States Food and Drug Administration Inventory of GRAS Notices: https://www.cfsanappsexternal.fda.gov/scripts/fdcc/index.cfm?set=GRASNotices
Footnotes
Acknowledgments
We would like to thank our colleagues in the food toxicology community for discussions that inspired the production of this paper.
Author contributions
KR, YW, and PH authored the manuscript. All authors reviewed GRAS notifications, reviewed and edited the manuscript, and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Khatera Rahmani is a full-time employee of Mars, Incorporated. Yen-Ching Wu is a former employee of a food manufacturing company. Neil Buck is a full-time employee of General Mills. Alexandria Lau is a full-time employee of ToxStrategies. Paul Hanlon is a full-time employee of Abbott Nutrition.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
