Abstract
Delivery of therapy to the cochlea is a challenge and limits the efficacy of therapies meant to treat hearing loss, reverse tinnitus, and protect hearing from chemotherapy regimens. Magnetic injection is a technique that uses magnetic fields to inject nanoparticles from the middle ear into the cochlea, where they can then elute therapy to treat hearing disorders. To evaluate the safety of this treatment in the middle ear, 30 rats were subdivided into 6 groups and treated by single or multiple intratympanic injections of saline, prednisolone, nanoparticles, or nanoparticles loaded with prednisolone. A specially designed magnet array was used to magnetically inject the particles from the middle ear to the cochlea. Treatment began at study day 0, and animals were euthanized on study day 2, 30, or 90. Temporal bones were collected and prepared for histopathological examination. Intratympanic administration of magnetic nanoparticles and/or prednisolone resulted in minimal to mild inflammatory changes in all treated groups. The incidence and severity of the inflammatory changes observed appeared slightly increased in animals administered nanoparticles, with or without prednisolone, when compared to animals administered prednisolone alone. At study day 90, there was partial reversibility of the findings noted at study day 2 and 30. Repeat administration did not appear to cause greater inflammatory changes.
Keywords
Introduction
It is difficult to deliver drugs into the cochlea because it is located deep in the temporal bone and is protected and isolated by the blood–labyrinth barrier. The standard of care for treating sudden sensorineural hearing loss and other hearing problems has been to either inject steroids intratympanically into the middle ear and rely on free diffusion of drugs from the middle into the inner ear (a procedure that creates unreliable drug delivery and undesirable drug gradients along the cochlea) or to administer steroids orally. Both procedures result in widely varying patient outcomes (Spear and Schwartz 2011; Sharon et al. 2015; Meyer 2013; Rask-Andersen et al. 2012; Salt et al. 2012; Lee et al. 2017).
Magnetic injection is a technique in which drug-loaded magnetic nanoparticles are placed in the middle ear by an intratympanic procedure and then a magnetic injector is used to transport the particles from the middle ear, through the round and oval windows, into the cochlea (Figures 1 and 2). The effectiveness of magnetic delivery to the cochlea has already been reported (Shapiro et al. 2014; Sarwar et al. 2013; Ramaswamy et al. 2017). The current article focuses on the histopathological findings in the middle ear associated with intratympanic placement of prednisolone-loaded iron nanoparticles in the middle ear and their subsequent magnetic injection into the cochlea.

Magnetic delivery to the cochlea. The anticipated size and position of the magnetic injector for human patients. The magnet is colored in blue.
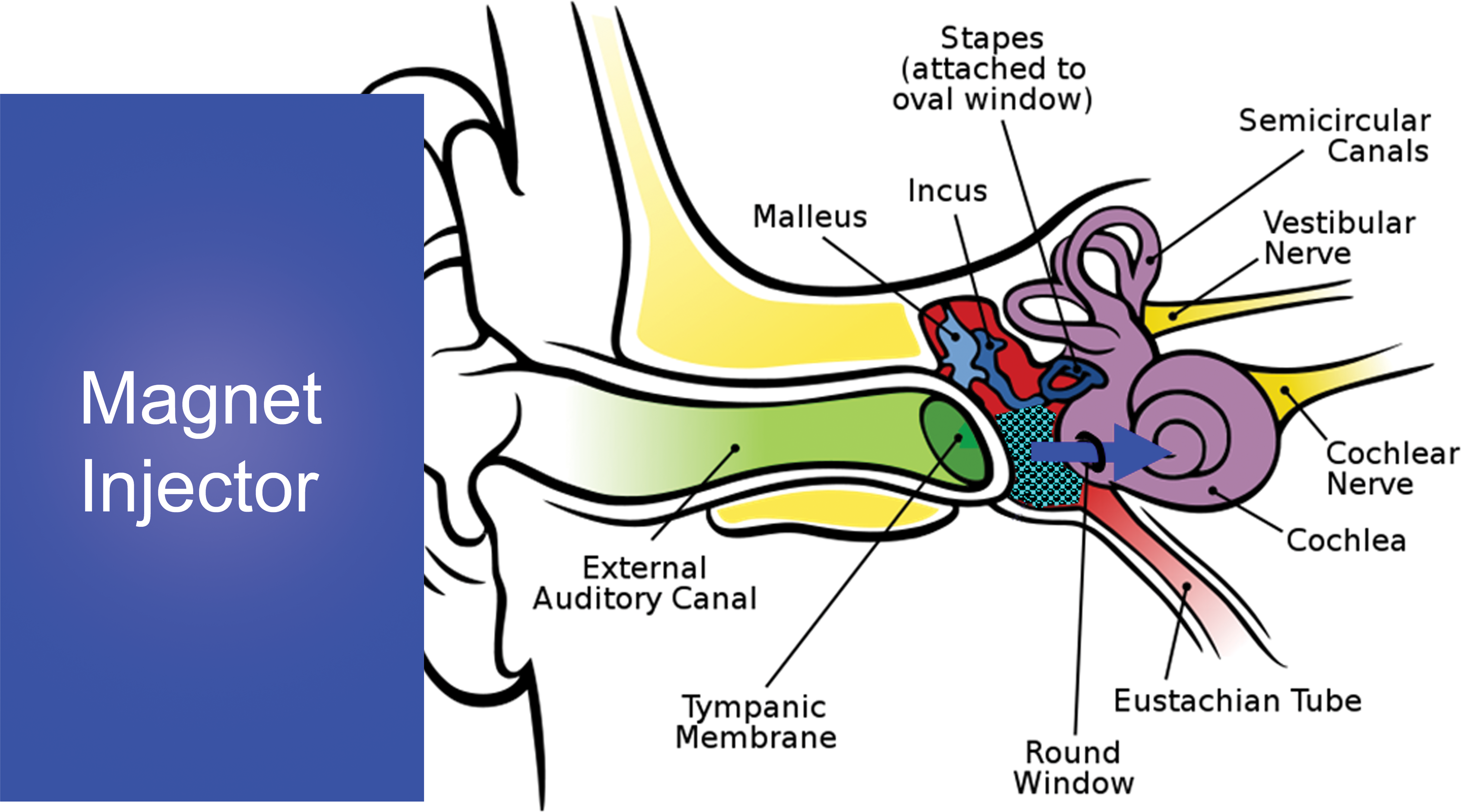
Schematic of magnetic delivery to the cochlea. The magnet applies a push force (blue arrow) on magnetic nanoparticles that have been placed in the middle ear by an intratympanic injection. This push force transports them through the round and oval windows and into the cochlea (Shapiro et al. 2014; Sarwar et al. 2013; Ramaswamy et al. 2017; illustration of ear anatomy is from “Auditory system,” Wikipedia, The Free Encyclopedia).
Materials and Methods
Adult male Long Evans rats (7–8 weeks old) were purchased from Charles River Laboratories (Wilmington, MA). Animals were housed in pairs in a climate-controlled facility with a 12-hr light/dark cycle. Food and water were provided ad libitum, except during hearing testing. All the rats were tattooed with their ID number on their tail for identification. Animals were randomly subdivided into 6 different groups, and all animals were treated by intratympanic injection in 1 ear only (left ear). There were 4 single administration groups: saline control (A), prednisolone (B), magnetic nanoparticles + magnet (C), and prednisolone-loaded nanoparticles + magnet (D); and 2 repeat administration groups: saline control (E) and prednisolone-loaded nanoparticles + magnet (F) as detailed in Table 1.
Experimental Design.
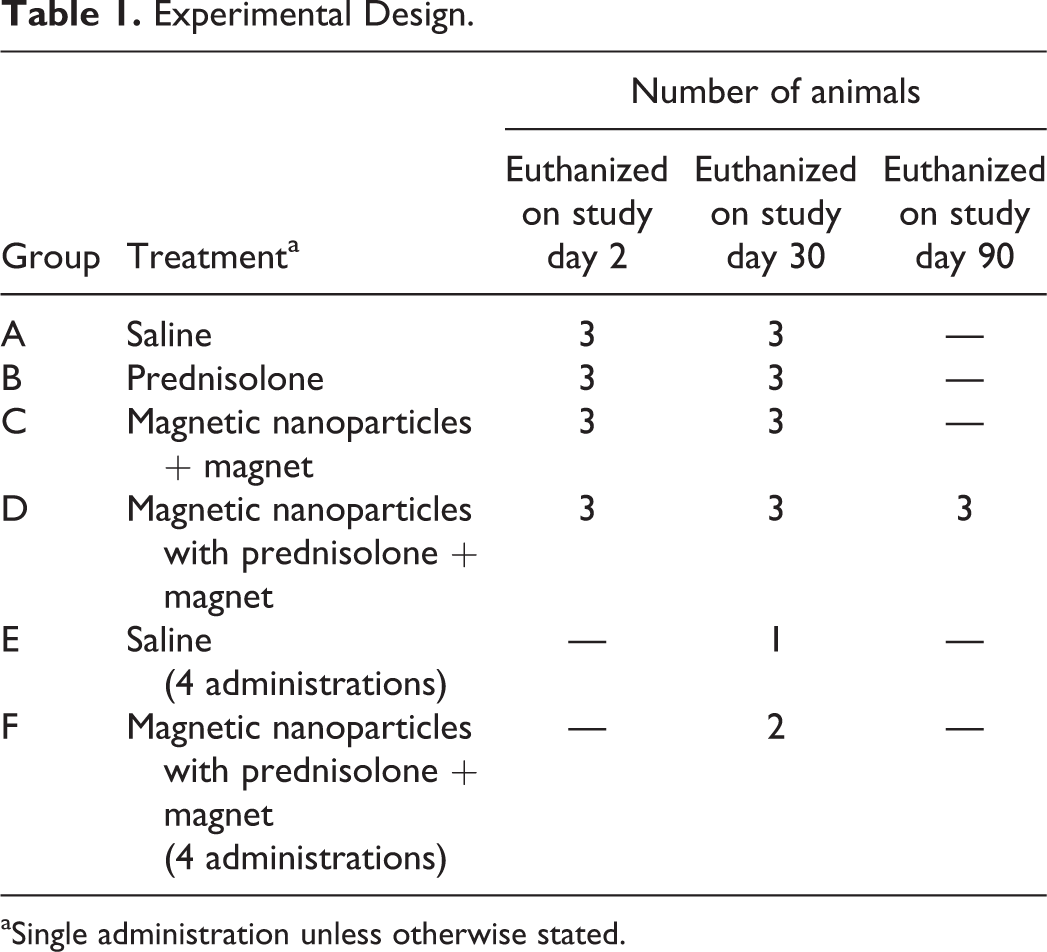
aSingle administration unless otherwise stated.
A 31G needle was directed through the pars tensa of the tympanic membrane. By facing the bevel caudally, the ejected solution was directed toward the round and oval windows. All solutions were injected with a total volume of 30 µl. When the injection was complete, the needle was carefully withdrawn. All administrations of saline or drug were into 1 ear via intratympanic injection. Group A animals received 30 µl of sterile saline (0.9% NaCl). Group B animals received 30 µl (40 mg/ml) of a prescription grade methyl-prednisolone (Pharmacia Upjohn, LLC, Kalamazoo, MI, USA). Group C rats received 300 nm diameter magnetic nanoparticles without any attached drug in an injection of 30 µl of nano-screenMAG/R-Chitosan particles from Chemicell GmbH, Berlin (25 mg particles/ml of solution; ∼6.56 × 1013 particles/g). Group D rats were treated the same as group C animals, except we used 300 nm diameter magnetic particles with ionically associated prednisolone sodium phosphate. As above, the rats received 30 µl of nano-screenMAG/R/Chitosan-Prednisolone particles (25 mg particles/ml solution; ∼6.56 × 1013 particles/g; 0.04 g prednisolone/g particles). Animals in the multidose test group (Group F) were administered 20 µl of nano-screenMAG/R/Chitosan-Prednisolone (25 mg particles/ml; ∼6.56 × 1013 particles/g; 0.04 g prednisolone/g particles) once a week for 4 weeks. Animals in the multidose saline control group (Group E) received an equal volume (20 µl) of saline once a week for 4 weeks. After particle administration, for groups C, D, and F, our magnetic injector was applied at a 4 cm distance from the rat’s middle ear to mimic the magnet to particle distance anticipated in adult human patients.
Treatments began at study day 0 and animals were euthanized on study day 2, 30, or 90. Temporal bones were collected for histological sectioning and histopathological examination of the middle ear. After decapitation and removal of the jaw, the head was hemisected and the right half placed in ice-cold saline. The left temporal bone was isolated, connective tissues and muscles were removed, and a small hole was made in the anterior aspect of the tympanic bulla to allow better penetration of the fixative solution. The temporal bone and bulla were then fixed in 4% paraformaldehyde and sectioned and stained with H&E and Perl’s Prussian Blue. After the left temporal bone was prepared, the same procedure was applied to the right temporal bone. Histopathological evaluation was performed to assess the integrity and possible signs of toxicity to the middle ear structures. The presence of hemorrhage and inflammation was semiquantitatively evaluated, and inflammatory responses were characterized and localized in detail. A 5-step semiquantitative scale was used (1 = minimal, 2 = slight, 3 = moderate, 4 = marked, 5 = severe). Histopathological evaluation was limited to the middle ear; the inner ear was not evaluated in this study.
All animal procedures were conducted in accordance with the policies and recommendations of the National Institute of Health Guide for the Care and Use of Laboratory Animals and under approval from the Institutional Animal Care and Use Committee of the University of Maryland.
Results
Treatment-related inflammatory changes were noted in all treated animals euthanized at study day 2 or 30. Minimal inflammation was observed in animals administered prednisolone alone, while minimal to slight inflammation was seen in animals administered magnet + nanoparticles with or without prednisolone. None of the animals given saline once had any inflammatory changes, while repeated saline administration resulted in minimal inflammation which was considered secondary to the experimental procedures (i.e., due to intratympanic injection).
The incidence and severity of the inflammatory changes seen in treated animals appeared mildly increased in animals administered magnet + nanoparticles (with or without prednisolone) when compared to animals administered prednisolone alone. Only 2 treated animals did not have any inflammatory changes, and both of these animals were in the prednisolone-only group. Also, all animals with inflammation in the prednisolone-only group had minimal inflammation while 4 of the 12 animals administered nanoparticles (with or without prednisolone) had more severe (slight) inflammation. Repeated administration of magnet + nanoparticles did not result in increased severity of the inflammatory changes observed.
The inflammatory changes noted in this study were limited to the middle ear and were characterized, in animals euthanized on study day 2, by the presence of relatively low numbers of neutrophils and lesser numbers of mostly foamy macrophages (Figure 3) in various locations of the middle ear (e.g., tympanic cavity, Eustachian tube, footplate of stapes, malleus, malleal ligament, oval window). In animals euthanized on study day 30, the inflammatory changes persisted and were of comparable incidence and severity. However, these changes showed a shift toward a more chronic reaction and were characterized by the presence of mainly macrophages (foamy to pigment-laden and often clumped), with lymphocytes in 2 animals and rare neutrophils in a single animal (Figure 4). In animals administered magnet + nanoparticles (with or without prednisolone), some macrophages contained golden to brownish pigment, consistent with iron-based pigment (Figure 5), which was confirmed with Prussian Blue stain (Figure 6). Of note, extracellular Prussian Blue–positive material was noted in the lumen of the middle ear in animals euthanized on study day 2 but not in animals euthanized on study day 30. In the latter subset of animals, all of the Prussian Blue–positive material observed was intracellular, in macrophages. No pigment was observed in animals administered prednisolone by itself, without nanoparticles. The 2 animals administered nanoparticles with prednisolone 4 times had inflammation that was not distinguishable from which was seen in animals administered the same combination only once.
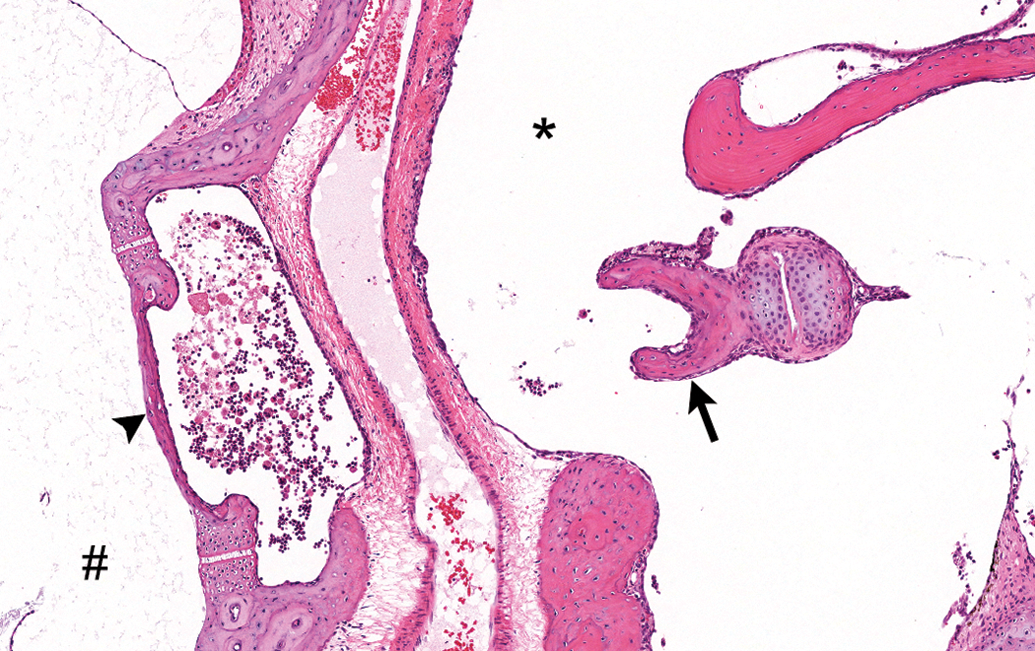
Exudate consisting of mainly neutrophils accompanied by a few foamy macrophages near the oval window and stapes. Middle ear of animal administered nanoparticles with prednisolone and euthanized on study day 2. # = vestibule; arrowhead = footplate of stapes (oval window); * = tympanic cavity; black arrow = head of stapes. Tissue stained with H&E.
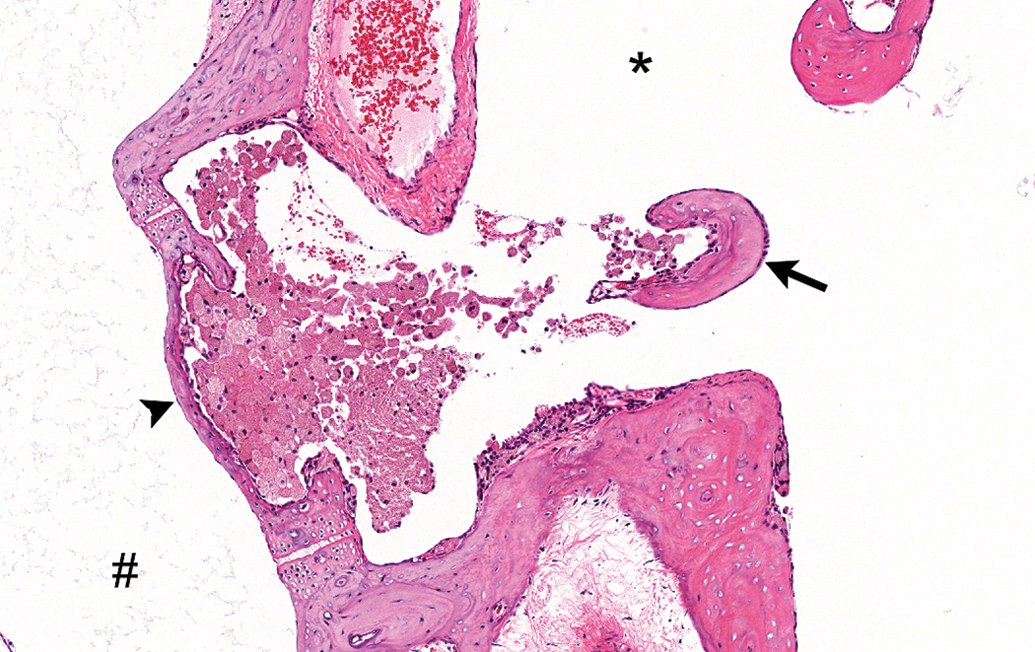
Inflammatory response consisting of almost only macrophages, with rare neutrophils, near the oval window and footplate of stapes. Middle ear of animal administered nanoparticles with prednisolone and euthanized on study day 30. # = vestibule; arrowhead = footplate of stapes (oval window); * = tympanic cavity; black arrow = head of stapes. Tissue stained with H&E.
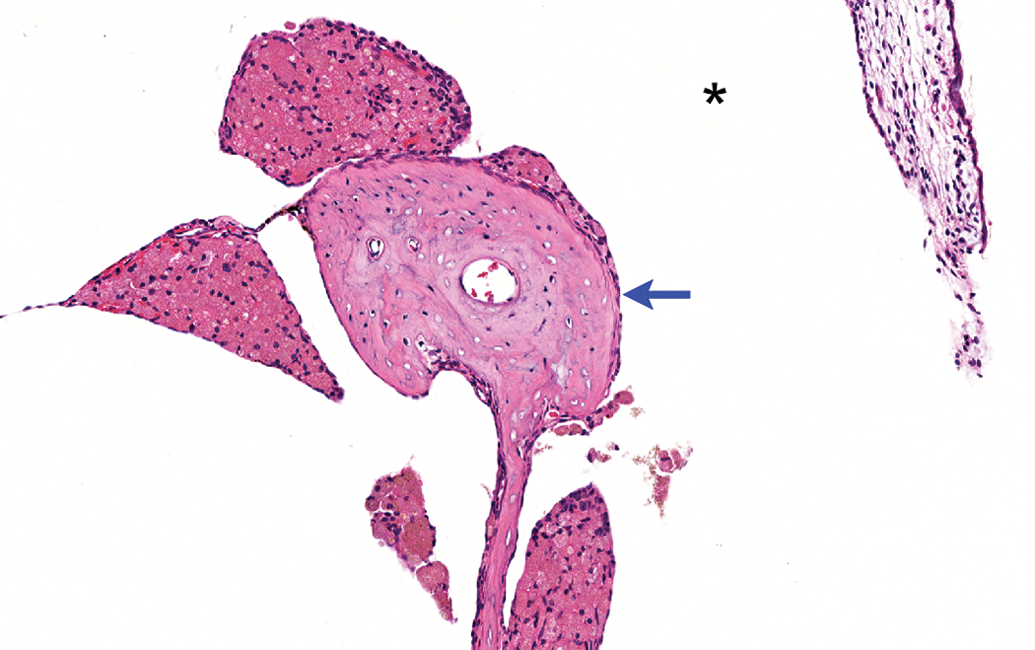
Foamy and finely vacuolated macrophages near the incus. Middle ear of animal administered magnet + nanoparticles with Prednisolone and euthanized on study day 30. Some macrophages contain brownish to golden pigment. * = tympanic cavity; blue arrow = incus. Tissue stained with H&E.
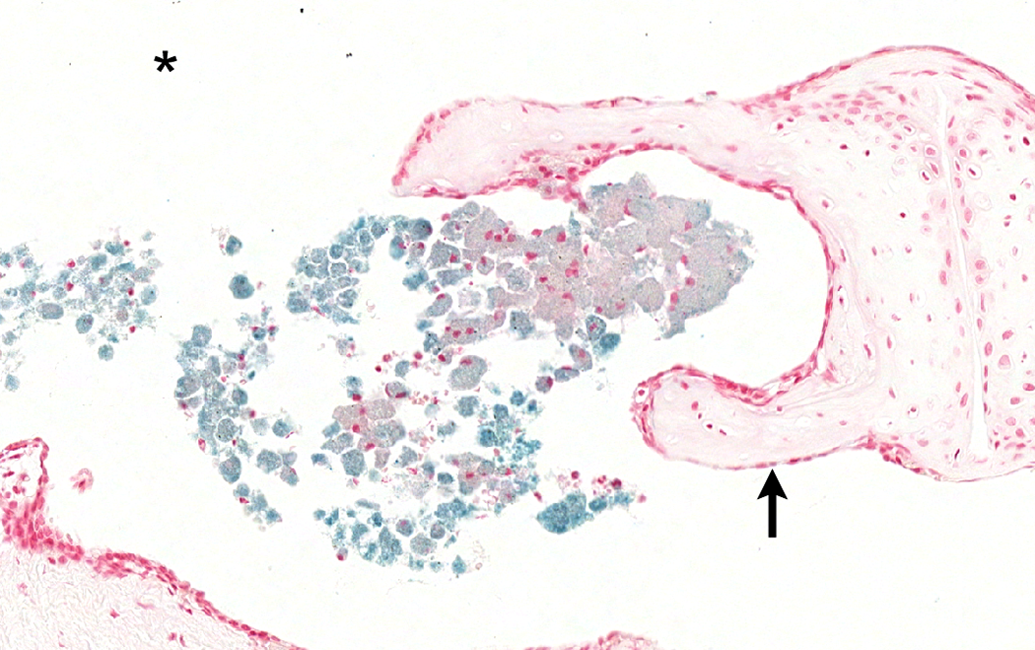
Middle ear macrophages containing abundant intracytoplasmic iron. Animal administered magnet + nanoparticles with prednisolone and euthanized on study day 30. * = tympanic cavity; black arrow = head of stapes. Tissue stained with Prussian Blue for iron in the nanoparticles.
In the animals euthanized on study day 90, minimal to slight numbers of macrophages were still present in the middle ear, mainly close to the head of the malleus and next to the round window. Macrophages were present in minimal numbers, were not foamy or vacuolated, and did not appear to contain any pigment on H&E (Figure 7). However, the macrophages seen in 1 of these 2 animals contained minimal amounts of small granules that stained positive on Prussian Blue stain (consistent with iron-based pigment likely from nanoparticles; Figure 8). Neutrophils and lymphocytes were not observed in the middle ear of any of the animals euthanized at study day 90.
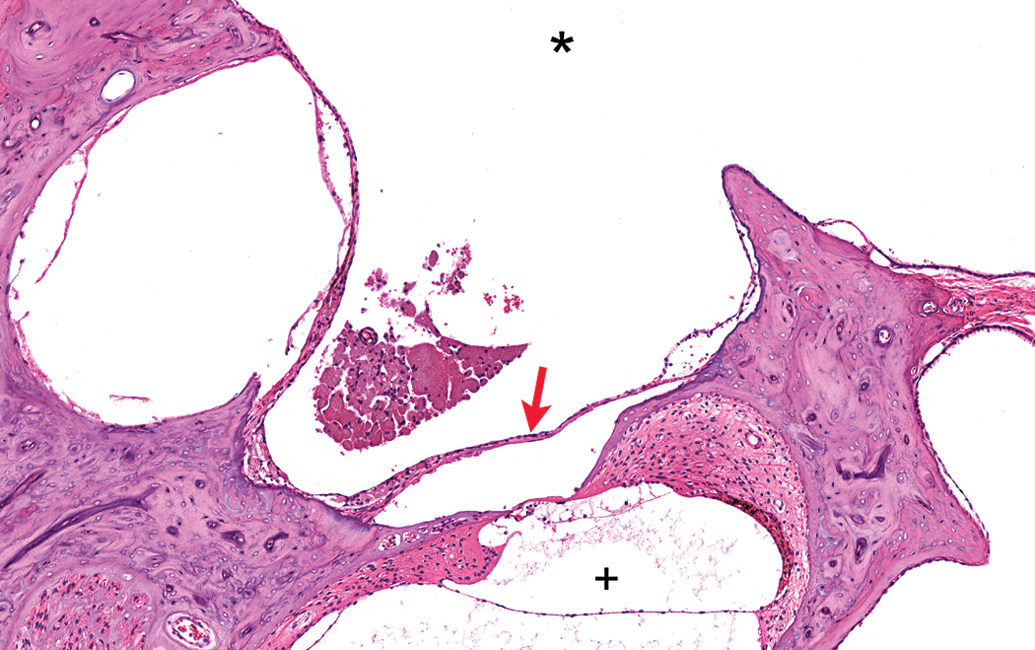
Small clump of apparently unpigmented macrophages near the round window. Middle ear of animal administered nanoparticles with prednisolone and euthanized on study day 90. * = tympanic cavity; red arrow = round window membrane; + = cochlea. Tissue stained with H&E.
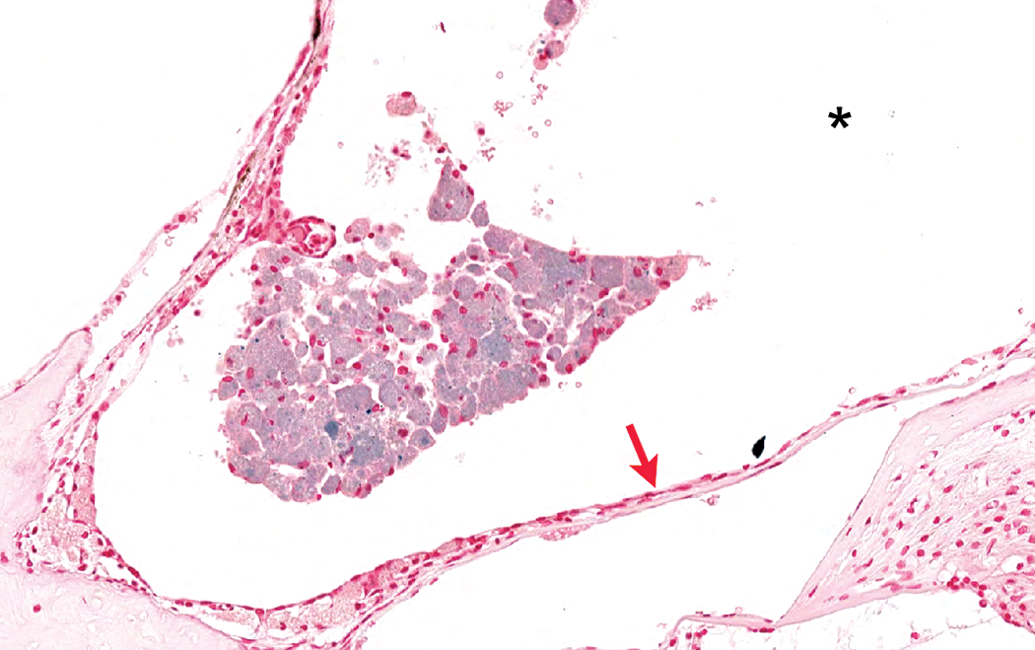
Middle ear macrophages containing intracytoplasmic iron in lesser quantities when compared to those observed on study day 30. Animal administered nanoparticles with prednisolone and euthanized on study day 90. * = tympanic cavity; red arrow = round window membrane. Tissue stained with Prussian Blue for iron in the nanoparticles.
Discussion
This study was focused on an evaluation of histopathology in the middle ear after intratympanic placement of magnetic nanoparticles in the middle ear, and their magnetic injection into the cochlea. Intratympanic administration of magnetic nanoparticles and/or prednisolone resulted in inflammatory changes in all treated groups. Single administration of saline did not result in inflammatory changes, while 4 administrations of saline resulted in minimal inflammation in a single animal. The incidence and severity of the inflammatory changes seen in treated animals appeared slightly increased in animals administered magnet + nanoparticles with or without prednisolone (minimal to slight; present in 12 of the 12 animals) when compared to animals administered prednisolone alone (minimal only; absent in 2 of the 6 animals). At study day 90, there was partial reversibility of the findings noted in animals euthanized on study day 2 and 30, as demonstrated by the absence of neutrophils and lymphocytes and by the decreased presence of iron in these animals. Repeated administration of nanoparticles (with prednisolone, 4 administrations in each of 2 animals) did not appear to cause greater inflammatory changes when compared with changes noted in animals administered the same combination once.
All the inflammatory changes noted in this study were very mild in severity and were present in the lumen of the middle ear. No structural damage, degeneration, necrosis, fibrosis, or otosclerosis was noted in any of the ears examined, and all the changes described were considered nonadverse.
We have demonstrated that iron nanoparticles can be placed intratympanically in the middle ear and then magnetically injected into the cochlea without any local detrimental effects to the middle ear. To complete the characterization of the ototoxicologic potential of our magnetic delivery treatment, we shall focus our future efforts on assessing hearing and on histopathological examination of the cochlea, most particularly the organ of Corti, the stria vascularis, and the spiral ganglia.
Footnotes
Acknowledgement
The authors would like to thank Hamid Boubekeur for his invaluable help with the images.
Author Contribution
Authors contributed to conception or design (JL, BS, MS, DD, MIS, BR, PM); data acquisition, analysis, or interpretation (JL, BS, MS, DD, MIS, BR, PM); drafting the manuscript (JL); and critically revising the manuscript (BS, MS, DD, MIS, BR, PM). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Benjamin Shapiro and Didier Depireux are co-founders of Otomagnetics and disclose an equity stake in the company.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: Funding is gratefully acknowledged from Action on Hearing Loss in the UK, from the Congressionally Directed Medical Research Program (CDMRP), from the National Institutes of Health (NIH), from the Maryland Innovation Initiative at the Maryland Technology Development Corporation (TEDCO) and the Maryland Industrial Partnerships (MIPS) program in the State of Maryland, as well as from the Sheikh Zayed Institute for Pediatric Surgical Innovation, Children’s National Medical Center, in Washington DC.
