Abstract
Age, and in particular young age, can significantly impact the response to toxicants in animals and can greatly influence the interpretation of tissue changes by the toxicologic pathologist. Although this applies to multiple organ systems, the current review focuses on the male reproductive system. When performing microscopic evaluation of male reproductive organs, the toxicologic pathologist must be aware of the dynamic changes in histomorphology, predominantly driven by timed hormonal alterations, at various life stages. Specific challenges pathologists face are understanding the appearance of male reproductive tissues throughout the neonatal, infantile, and juvenile developmental periods, recognizing when normal looks abnormal during tissue development, defining sexual maturity, and working with high interanimal variability in maturation rate and histologic appearance in developing large laboratory animals, such as nonhuman primates, dogs, and pigs. This review describes postnatal development of the male reproductive system in the rat, demonstrates how assessing toxicity during a defined window of postnatal development in the rat may improve definition of toxicant timing and targets, and discusses challenges associated with the interpretation of toxicity in immature large animal species. The emphasis is on key age-related characteristics that influence the interpretation of tissue changes by the toxicologic pathologist.
Age impacts the histologic appearance of the male reproductive tract and its susceptibility to xenobiotics. But age, and in particular young age, also impacts the way in which toxicologic pathologists interpret histologic findings in the male reproductive tract. Evaluation of immature male reproductive organs is required by protocol in some preclinical safety studies, such as the pubertal and thyroid function assay and juvenile toxicity study (Environmental Protection Agency 2009; Food and Drug Administration [FDA] 2006). Immature reproductive tissues are also commonly encountered in routine 28-day studies involving large laboratory animals (e.g., nonhuman primates [NHPs], dogs, and pigs), since these animals are often sexually immature when placed on study. Juvenile toxicity studies are receiving particular attention because every New Drug Application in the United States or Marketing Authorization Application in Europe now requires a pediatric assessment if the drug is intended for therapeutic use in children (Barrow 2011; Tassinari et al. 2011). A juvenile toxicity study may be incorporated as part of the required pediatric assessment. While only general guidance exists on how to conduct juvenile toxicity studies, the male reproductive system has been identified as an organ system that should be examined since it undergoes significant changes during postnatal development (Bailey and Marien 2011; European Medicines Agency 2008; FDA 2006; Tassinari et al. 2011).
Immature testes and epididymides may present diagnostic challenges for the pathologist, yet this young age offers a unique window of opportunity to evaluate toxicity. While male reproductive organs include testes, epididymides, and the accessory sex glands (prostate and seminal vesicles), the current discussion is focused on the testes and epididymides. Microscopic evaluation of the testis can be difficult in any species at any age. In sexually mature rats, there is dynamic continuum of 14 spermatogenic stages that involves 4.5 cycles and 56 days for a basal spermatogonia to reach the final 19th step of spermatid development (Creasy 1997). In the cynomolgus monkey, for comparison purposes, there are 12 stages of tubules, 4 cycles, 56 days, and 14 steps of spermatid development (Creasy 1997; Dreef, Van Esch, and De Rijk 2007; Luetjens and Weinbauer 2012). In the dog, there are 8 stages of tubules, 4 cycles, 54 days, and 12 steps of spermiogenesis (Creasy and Foster 2002; Foote, Swierstra, and Hunt 1972; Greaves 2012; Vidal et al. 2013). In the minipig, spermatogenesis occurs rapidly and requires only 35 days (Greaves 2012; Jorgensen et al. 1998). Once a finding (or group of findings) is identified, the pathologist must back calculate chronologically in order to pinpoint the toxic event and target that would have led to those findings (Creasy 2001). In so doing, the pathologist often has to account for confounding endocrine imbalances, body weight changes, and effects of stress, any of which might result in nonspecific changes in the testes that accentuate or obscure the findings.
The daunting task of testis evaluation is made more difficult in the immature testis because of the superimposed changes associated with normal growth and development. When interpreting immature testes, the pathologist not only has to identify possible direct toxic effects but also must try to discern if development is precocious or delayed. As one further obstacle, and depending on the study design, pathologists who examine immature tissues often do not have the advantage of concurrent age-matched controls for comparison. Consequently, identifying test article–related effects in the testis and epididymis with certainty is not straightforward at any age, and special considerations are necessary when assessing juvenile animals.
Given these confounding issues and obstacles for the pathologist, it is not surprising that the conventional wisdom dictates that proper evaluation of the testis can only be performed in sexually mature animals (Halpern et al. 2016; Haruyama et al. 2012; Takakura et al. 2014), because all stages of spermatocyte development and all functions of the mature Sertoli cell can be assessed. Spermatocytes, spermatids, Sertoli cell-spermatid adhesion, and adult Leydig cells may not be present in the immature testis. While the use of sexually mature animals may be ideal for routine rodent studies, this is often not practical when studying larger laboratory animal species since such animals are generally not sexually mature when placed on study. The use of sexually mature animals is also not feasible for juvenile toxicity studies in which the underlying purpose is to identify either the test articles that can adversely impact organ development or the mode of action (FDA 2006).
Contrary to prevailing notions, the evaluation of immature testes can provide a wealth of information not otherwise obtained from sexually mature animals. This is because during the neonatal and infantile periods, the testis is uncomplicated by endocrine feedback loops, gonadotropins are less likely to be affected by confounding factors such as stress, tubules appear uniform prior to the formation of spermatogenic stages, and tubules contain highly visible, mitotically active, and toxicologically susceptible spermatogonia and Sertoli cell populations (Picut et al. 2015; van Haaster and De Rooij 1993; Whitney 2012). Therefore, the evaluation of testes during early reproductive development may be ideal for identifying toxic effects that are unique to the early postnatal time period and can also help determine the target cells that ultimately cause nonspecific lesions in the adult testis.
Postnatal Development of the Rat Testis and Detection of Toxicity
The pathologist should be aware of special considerations when evaluating immature testes and epididymides. Postnatal development of the rat testes can be divided into four recognized stages: neonatal (birth to postnatal day [PND] 7), infantile (PND 8–20), juvenile (PND 21–32), and peripubertal (PND 33–55). These correspond to similar stages recognized in humans (see Table 1). The following discussion will focus on a few important features of postnatal development that affect the pathologist’s interpretation, namely, unique cell populations, unique distribution and visibility of cell populations, and changes that could easily be misconstrued as toxic effects. The reader is referred to other publications that describe in detail the histologic development of the spermatogenic epithelium and Leydig cells in the rat testis (Picut and Remick 2016; Picut et al. 2015).
Age Comparison of Developmental Stages in Rat and Human Testis.

Note. Additional references: Hoover et al. (1989) and Paniagua and Nistal (1984). PND = postnatal day.
aModified from Ojeda, Advis, and Andrews (1980).
bModified from Barrow, Barbellion, and Stadler (2011).
From birth to PND 9, the seminiferous tubules contain a unique cell type, termed the gonocyte, which is the precursor to spermatogonia. Microscopically, a gonocyte is a large round cell with a proportionally large nucleus, prominent nucleolus, and finely dispersed chromatin that is positioned in the center of the spermatogenic tubule for the first 9 days of life (Figure 1). In addition to the central gonocytes, the neonatal tubules are lined by Sertoli cells that are soon joined by rapidly increasing numbers of spermatogonia. During the first 9 days of life, the gonocytes migrate to the basement membrane and transform into these spermatogonia.
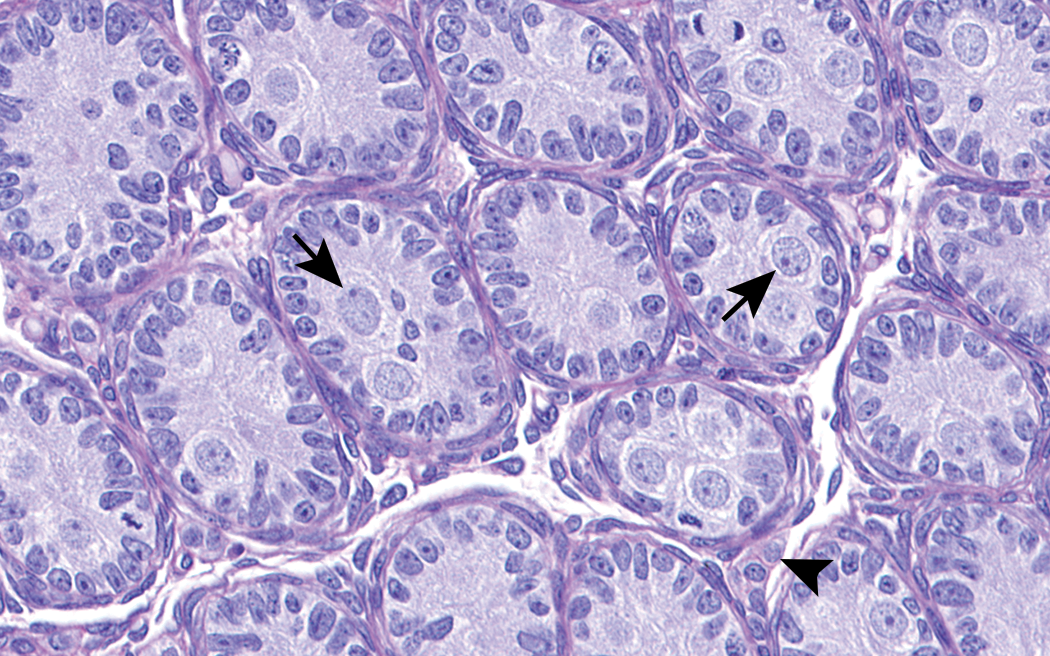
Testis from rat at birth. Immature tubules contain central pale staining gonocytes (arrows) and a peripheral rim of Sertoli cells. Cells in the interstitium represent a mixture of fetal Leydig cells (arrowhead) and undifferentiated mesenchymal cells. H&E. Original objective 53×.
The fetal Leydig cell is another cell type of interest in the first postnatal week. Fetal Leydig cells have been present since midgestation and are capable of producing testosterone as early as gestational day 15.5, after which they persist through the first postnatal week as testosterone-producing cells (Habert and Picon 1984). These fetal cells are required to virilize the embryo into a male, and more specifically to virilize the Wolffian duct into an epididymis, vas deferens, and seminal vesicle (Huhtaniemi and Pelliniemi 1992). Fetal Leydig cells can have a plump and “mature” appearance similar to those in the adult testis. However, after PND 6, the fetal Leydig cells revert to stem cells. Therefore, from PND 6–30, when postnatal Leydig cells are developing, the testis may appear to have a paucity of interstitial cells. This paucity should not be confused with Leydig cell hypoplasia.
Other unique attributes of immature tubules during the first postnatal week include a complete lack of spermatocytes or spermatids, no distinction between stages of tubules, very high mitotic rates within the Sertoli and spermatogonial cell populations, and high mitotic rates in the progenitor and immature Leydig cell populations. From approximately PND 5 until PND 18, spermatogonia and Sertoli cells proliferate robustly. This robust degree of proliferation ends at PND 18 when the blood–testis barrier (BTB) is formed and spermatocyte development is allowed to proceed (Orth 1982; Picut et al. 2015). At around PND 14–15, when the BTB first begins to form and tubular diameters are still small, the robust proliferation results in a crowded, pseudostratified population of spermatogonia and Sertoli cells within the tubules (Figure 2). Density-dependent apoptosis ensues, and this can be detected by routine light microscopy or by use of caspase-3 immunohistochemistry (de Rooij 2001; Picut et al. 2015). There are still no spermatocytes or spermatids at this point, no stage differentiation of tubules, and virtually no adult Leydig cells. The scarcity of adult Leydig cells partly explains the reason for the high density of spermatogonia and Sertoli cells that line these small tubules; this occurs because the proliferation of Sertoli cells and spermatogonia has outpaced the production of testosterone required for tubular expansion necessary to accommodate the proliferating cell populations. Apoptosis of excess cells is then a physiological response to overcrowding.
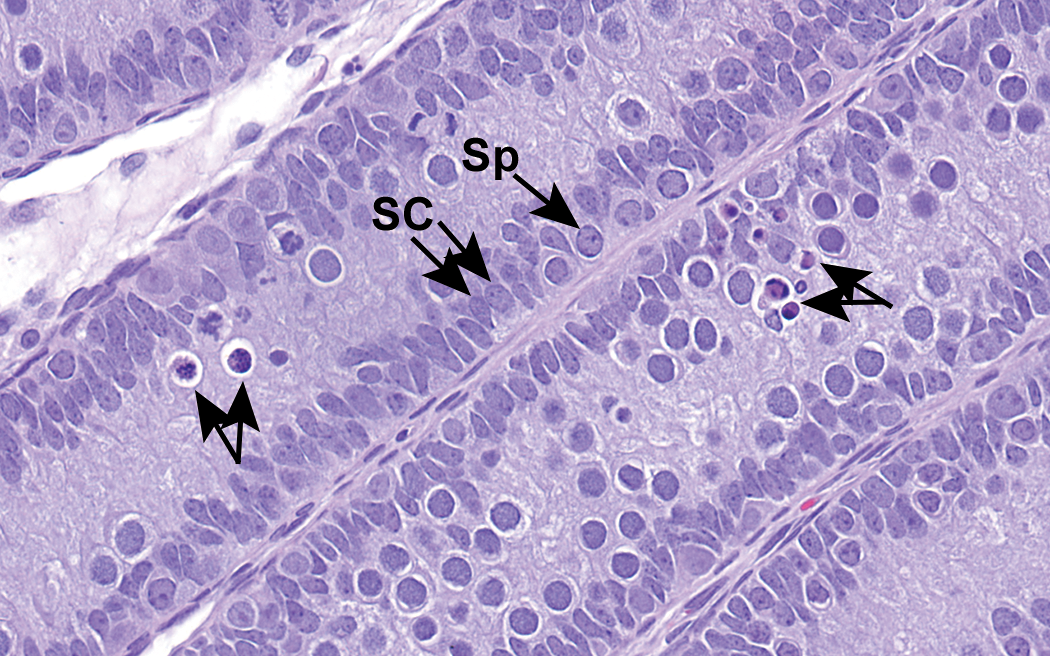
Testis from rat at postnatal day 14. There is a marked proliferation and crowding of the Sertoli cells (SC) and spermatogonia (Sp). Due to crowding, there is density-dependent apoptosis of these cell types (unlabeled arrows). Note the paucity of Leydig cells at this time. H&E. Original objective 56×.
Pathologists should easily recognize the normal process of apoptosis that occurs a few days prior to PND 18. However, pathologists should be wary not to misinterpret unusually positioned normal cells as apoptotic. During this time, as the BTB forms, there can be a flourish of development as late stage spermatogonia become preleptotene, leptotene, and zygotene spermatocytes (Figure 3). The preleptotene spermatocytes have a coarsely clumped irregular chromatin pattern that resembles karyrrohexis, and leptotene and zygotene spermatocytes have a nucleus with a condensed dark homogeneous chromatin pattern that resembles pyknosis. Because these early spermatocytes are highly visible near the center of the tubules in the immature testis, such chromatin patterns are unusually prominent. This is in contrast to the sexually mature testis, in which early spermatocytes are located peripherally in tubules admixed with Sertoli cells, pachytene spermatocytes, and spermatogonia, and thus their unusual chromatin patterns are not on obvious display.
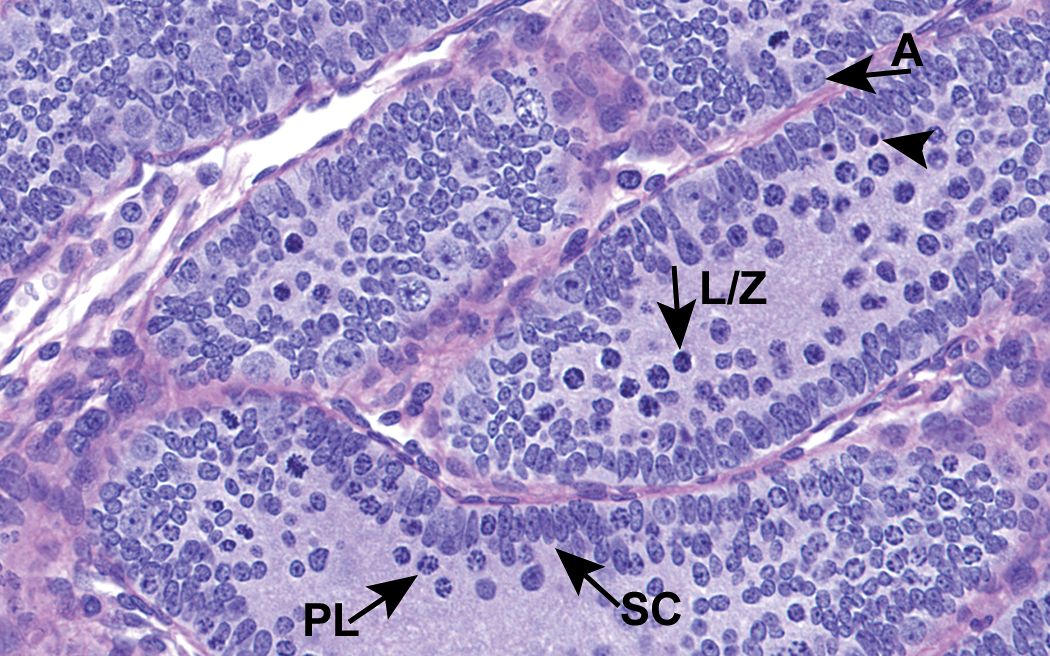
Testis from rat at postnatal day (PND) 15. The preleptotene (PL) spermatocytes have a chromatin pattern that resembles karyorrhexis, and the leptotene and zygotene spermatocytes (L/Z) have condensed chromatin patterns that appear similar to pyknotic nuclei. These early spermatocytes are centrally located unlike their peripheral position in adult seminiferous tubules, and such cell types should not be interpreted as apoptosis. A few apoptotic cells (arrowhead) with eosinophilic cytoplasm and pyknotic nuclei are present for comparison. A = spermatogonia A; SC = Sertoli cell. H&E. Original objective 60×.
Early testis development prior to until PND 15, before spermatocyte development and BTB formation occurs, is arguably an opportune time to evaluate the testis for potential effects of direct toxicity. As stated above, the tubules are uniform (without spermatogenic progression), the spermatogonia and Sertoli cells (two cell types commonly targeted by test articles) are visible in high numbers, and there is relatively little complication by endocrine disrupting events that involve fluctuating levels of gonadotropins (i.e., stress), because the hypothalamic–pituitary–testis axis is not yet mature (Figure 4). Hoover et al. (1989) has described this time (PND 5–15) as a critical window because of its “significant, uniform, and susceptible” cell populations and purports that short-term studies performed during this time could readily detect direct effects on either the spermatogonia or Sertoli cells. The GATA-4 (i.e., the transcription factor expressed in Sertoli cells) immunohistochemical stain is useful for distinguishing Sertoli cells from spermatogonia (Figure 5). GATA-4 is a zinc finger protein and transcription factor implicated in the regulation of gene expression and differentiation in a variety of tissues except, fortuitously, for germ cells (Li, Zheng, and Yan 2011). If designing a study to detect germ cell death, one should be advised that cell death progresses rapidly; apoptotic cells in the seminiferous epithelium are phagocytized within 24 hr (Lanning et al. 2002). Therefore, short-term studies are optimal for detecting the apoptotic process. If too much time ensues following the toxic insult, the testicular effects may merely involve the depletion of relevant cell types.
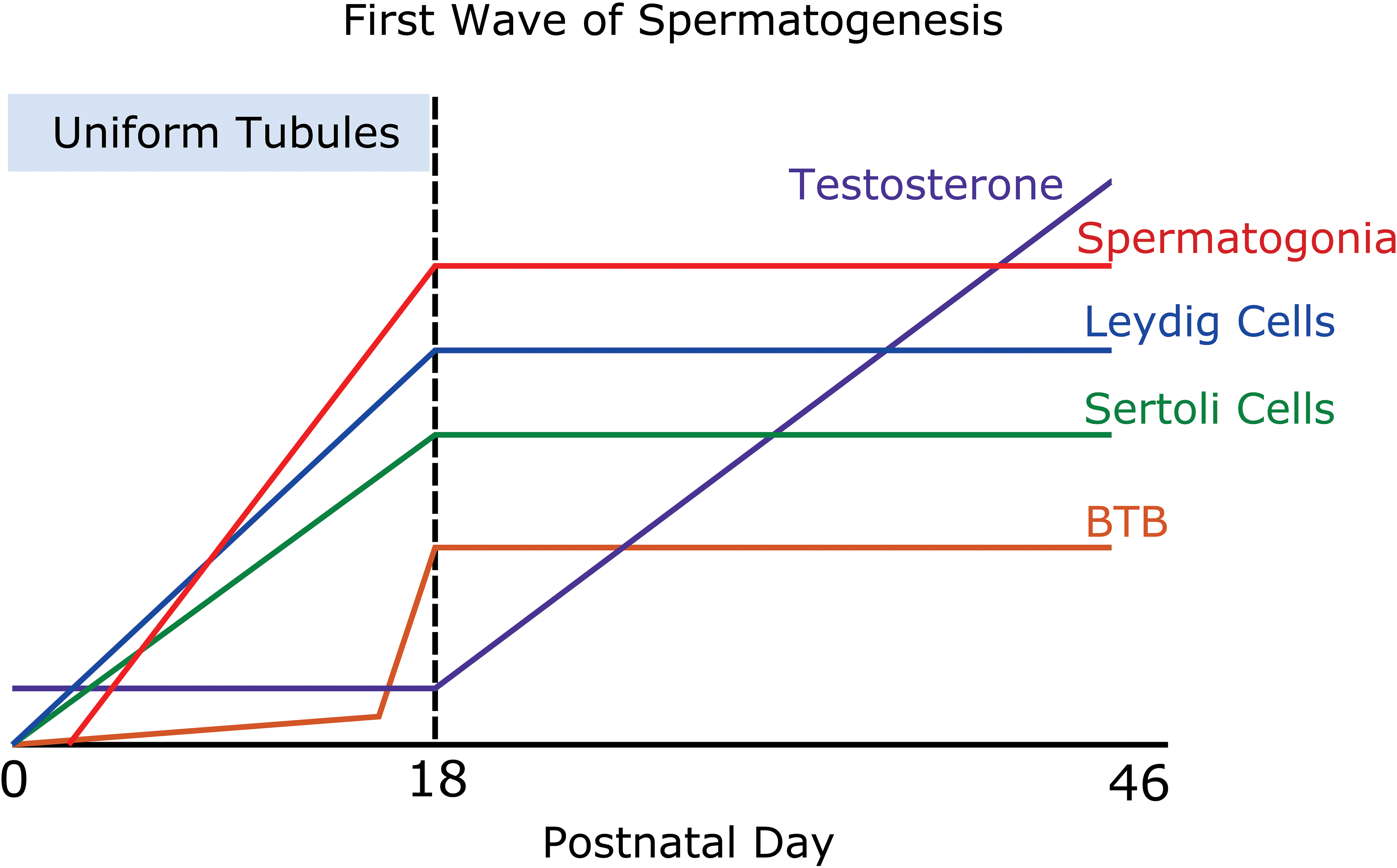
First wave of spermatogenesis. This schematic demonstrates the various parameters during postnatal development. It points out the critical window (postnatal day 5–15) when there are significant, uniform, and susceptible cell population due to proliferating spermatogonia and Sertoli cells. During this time, the blood–testis barrier (BTB) is not formed, the testosterone levels are low, and all the tubules appear uniform. Leydig cells are also proliferating during the critical window, but these cells are not as visible by light microscopy as are spermatogonia and Sertoli cells.
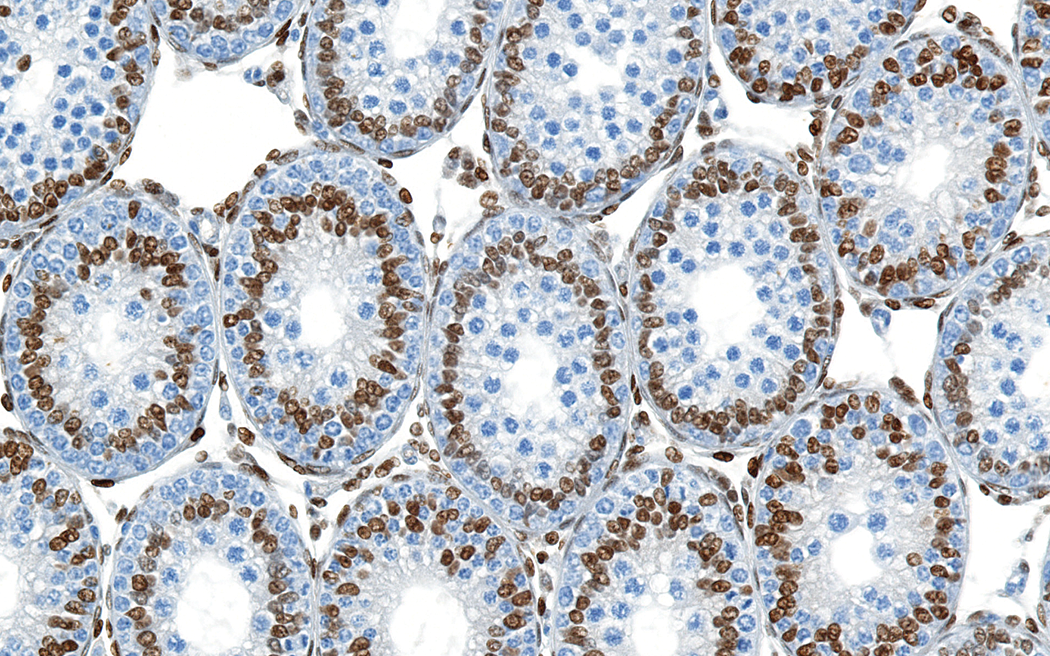
Testis from rat at postnatal day (PND) 18. The GATA-4 (i.e., the transcription factor expressed in Sertoli cells) immunostain clearly discerns between the nuclei of Sertoli cells (brown) and the unstained spermatogonia and spermatocytes. The Leydig cell nuclei in the interstitium also stain brown with GATA-4. This testis from an animal at PND 18 demonstrates the distinct rosette pattern with an inner layer of positive stained Sertoli cell nuclei and a peripheral rim of unstained spermatogonia. GATA-4 immunostain with hematoxylin counterstain. Original objective 40×.
Studies performed during this critical window have helped define testicular toxicants and their modes of action (Whitney 2012). N-methylthioltetrazole, a toxic agent in cefamandole (a β-lactam antibiotic), inhibits aldehyde dehydrogenase and interferes with proliferation of spermatogonia. This effect was readily observed by performing a targeted study from PND 4 through PND 13 and microscopically examining tissues shortly thereafter (Hoover et al. 1989, 1991; Whitney 2012). In that study, findings at the end of the critical window consisted of tubules lined by immature spermatogonia A, a deficiency in the numbers of smaller spermatogonia B, and a lack of any spermatocyte development when compared to age-matched controls (Hoover et al. 1989, 1991; Whitney 2012). When the same compound was administered over a wider time period (PND 6–36) and tissues were examined at a later time point, the findings were nonspecific and did not clearly reveal the mode of action. This example clearly supports the importance of targeted short-term studies in identifying specific target cell populations and windows of susceptibility to toxicants (Hoover et al. 1989).
Unique lesions can also be seen during this critical window. For example, test articles that interfere with tyrosine kinase receptor activity can inhibit migration of gonocytes to the basement membrane during the first week of postnatal development and prevent their transformation into spermatogonia (Whitney 2012). Imatinib mesylate (Gleevec®), a tyrosine kinase receptor inhibitor against c-kit and platelet-derived growth factor receptor, led to the increased persistence of central gonocytes in tubules, when given for 3 days during the first postnatal week (PND 5–PND 7; Nurmio et al. 2007; Whitney 2012). Understanding gonocyte migration and the use of age-matched controls enabled the pathologist to identify these unique lesions.
Steroid hormones given during this critical window also have effects that may not be apparent if the testis is allowed to mature. Estradiol administered to neonatal rats inhibited the maturation of Sertoli cells, which are normally mature by PND 18; this deficiency in Sertoli cell maturation resulted in a persistence of immature Sertoli cells that failed to support spermatocyte development (Gaytan et al. 1986). When estradiol was given daily to neonatal rats, histopathological lesions were first observed beginning at PND 22 and these were characterized by pseudostratified Sertoli cells (Sertoli cells with a nucleus that failed to marginate to the basement membrane), a paucity of spermatocytes, and no effect on spermatogonia (Gaytan et al. 1986). The immature tubule, with its uniform appearance and susceptible cell populations, gave unusual clarity to the fact that the Sertoli cell was the toxicological target.
At PND 18–20, the seminiferous tubule has an easily recognizable characteristic feature known as the infantile rosette (Picut et al. 2015). At PND 18, when the BTB is completely developed, the seminiferous epithelium forms two discrete peripheral layers: an outer layer of spermatogonia and an inner layer of Sertoli cell nuclei (Figure 5). Even though tubular dilatation is imminent in concert with increasing testosterone levels, the Sertoli cell nuclei are still crowded and immature and their nuclei are “pushed” into this inner row to form a characteristic rosette. This rosette is a convenient feature by which to age the testis and, in the case of one estradiol study, this structure provided a clue to the failure of Sertoli cell maturation (Gaytan et al. 1986). Seminiferous tubule lumen formation and descent of the testicle into the scrotal sac also take place around PND 18, and thus these additional features can also be useful indicators of age.
In the midjuvenile stage, the spermatogonia and Sertoli cells have retreated to an outer single layer of cells, the infantile rosettes disappear, and mitotic activity has subsided (Picut et al. 2015). The only mitotic figures that now exist are those associated with baseline mitotic and meiotic divisions that will sustain normal spermatogenesis. However, at this juvenile stage, the spermatogenic epithelium displays another unique pattern of cell distribution. Leptotene, zygotene, and pachytene spermatocytes are clearly visible in the centers of tubules as opposed to the more peripheral location seen in the adult testis (Picut et al. 2015). Pachytene apoptosis is common during the juvenile period, and this correlates with the relatively low level of testosterone production (Figure 6; Picut et al. 2015). Pachytene apoptosis associated with low testosterone is not unique to the juvenile stage, as it is also a feature of endocrine disruption in the adult testis (Lanning et al. 2002). Exfoliation of pachytene phases and other spermatocytes or spermatids is expected during this time, and clusters of exfoliated intact germ cells should be expected in the epididymis (Figure 7; Picut et al. 2015). Exfoliation and cell debris can be quite significant in the epididymis of immature animals, especially during the juvenile period, and is most likely a consequence of deficient Sertoli cell–spermatocyte adhesion due to Sertoli cell immaturity around PND 18 (Creasy et al. 2012; Haruyama et al. 2012).
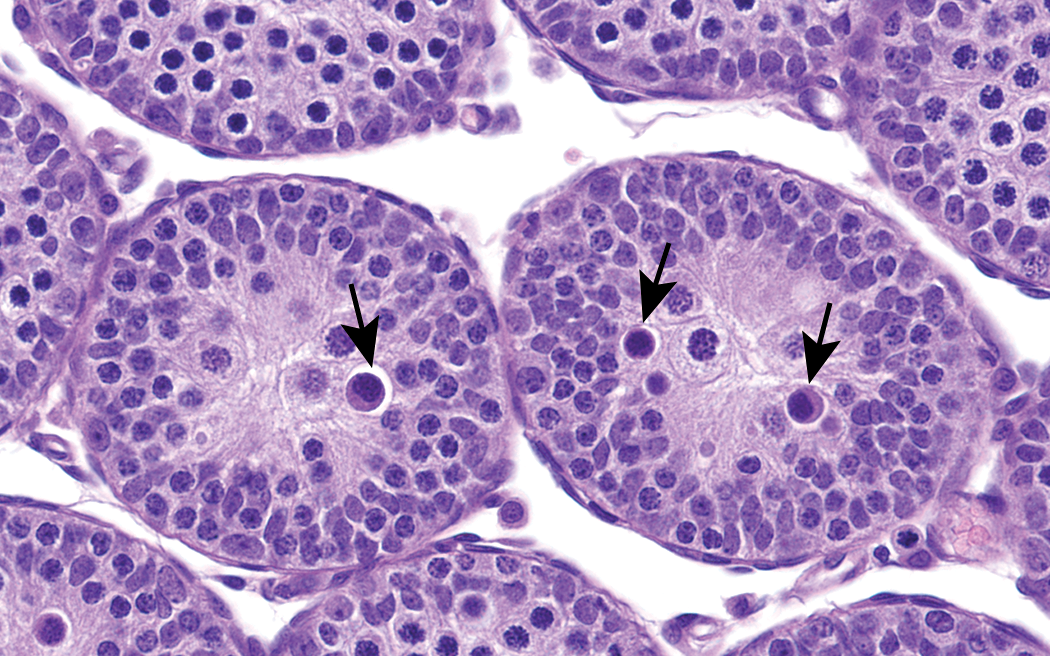
Testis from rat at postnatal day 21. Pachytene spermatocytes are located in the centers of the immature seminiferous tubules, which is not the normal placement of these cells in adult tubules. Apoptosis of pachytene spermatocytes (arrows) are typically seen in the early to midjuvenile stage. Apoptotic pachytene cells have a condensed chromatin pattern and eosinophilic cytoplasm. Periodic acid-Schiff (PAS) stain. Original objective 60×.
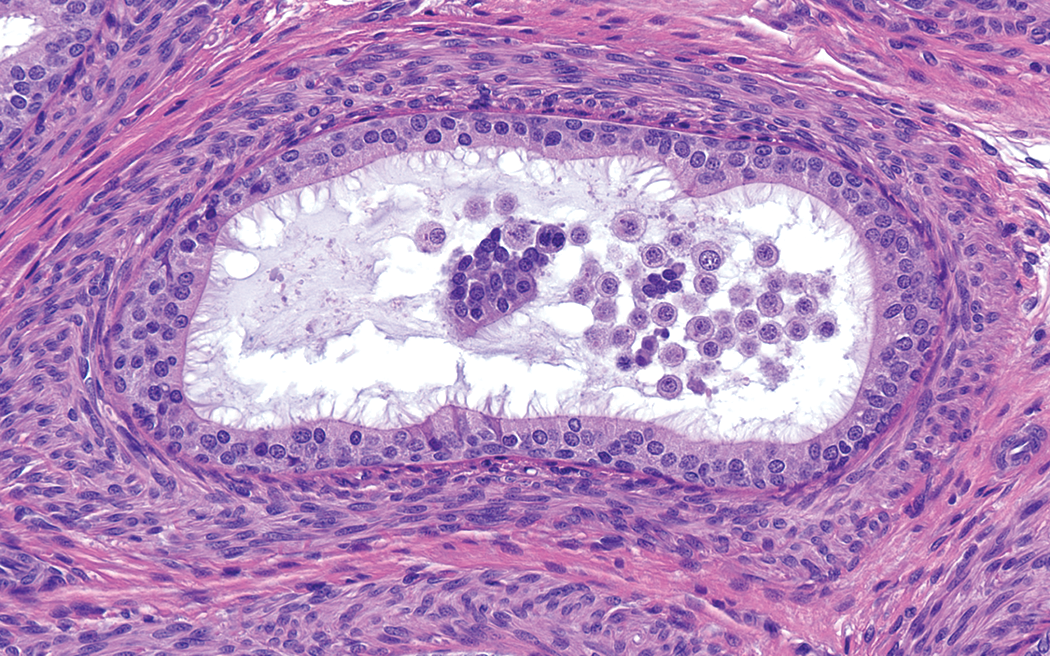
Epididymis from rat at postnatal day 28. Exfoliated spermatocytes in the cauda epididymal tubules are abundant in juvenile rats prior to puberty. PAS stain. Original objective 40×.
The peripubertal stage is from approximately PND 32–55, with puberty occurring at PND 43–46. The onset of puberty is defined macroscopically as the time of preputial separation (i.e., separation of the foreskin of the penis from the glans penis) and microscopically when step 19 spermatids are first seen in stage VII tubules accompanied by a lack of spermatozoa in the epididymis (Marty, Crissman, and Carney 2001; Picut et al. 2015). Sexual maturity, a term often equated with puberty, occurs when spermatozoa reach the epididymis around PND 52 in the rat (Picut et al. 2015). Therefore, sexual maturation is best determined by histologic examination of both the testis and epididymis in the rat. Even after sexual maturity is reached, there is a significant increase in the size of the testis and epididymis and significant increase in the density of spermatozoa in the epididymis (Campion et al. 2013). In the Charles River Laboratories’ (Ashland) historical control database for Sprague-Dawley rats, the weights of the testes and epididymides plateau after about 3 months of age and reach a maximum at about 8 months of age (weeks 32–34). This is consistent with published reports (Arroteia et al. 2012). After sexual maturity, there are also marked changes in the growth and development of accessory sex glands, which mature after the testis and epididymides (Dreef, Van Esch, and De Rijk 2007).
There has been some concern that any effect on the immature rat testis (during neonatal, infantile, and juvenile periods) has little relevance to human testis development. This concern is based on the fact that humans have a “quietly active” or quiescent period of testis development from 1 to 2 months of age until approximately 11 years of age (Table 1; Barrow 2011; Hoover et al. 1989; Ojeda, Advis, and Andrews 1980; Paniagua and Nistal 1984; Picut et al. 2015). During this quiescent period, the testis exhibits minimal proliferative activity. Then at approximately 11 years of age, a 3-year period of rapid spermatogonial proliferation occurs similar to the robust proliferation in rats that takes place from PND 5 to 15 (Table 1; Barrow 2011; Hoover et al. 1989; Ojeda, Advis, and Andrews 1980; Paniagua and Nistal 1984; Picut et al. 2015). The rapid proliferation in humans does not affect the testis in toto like it does in the rat, but in a lobule by lobule, nonuniform manner (Hoover et al. 1989). The presence of the rapid proliferation of a “significant, uniform, and susceptible” cell population in the rat testis on one hand, and the prolonged quiescent period and the “lobule by lobule” proliferation in the human testis on the other hand, calls into question the relevance of the immature rat to the immature human testis (Hoover et al. 1989). However, these differences do not render the immature rat testis unusable for evaluating safety. It merely means that the window of opportunity in the rat at PND 5–15 best correlates to the human testis at 11–13 years of age (Table 1; Barrow 2011; Hoover et al. 1989; Ojeda, Advis, and Andrews 1980; Paniagua and Nistal 1984; Picut et al. 2015). While rats may not have a defined quiescent period, NHPs certainly do. Marmosets have a quietly active period between 4 and 11 months of age during their infantile and juvenile periods when testosterone levels are low and there is a little proliferative activity in the Sertoli cell population (Chandolia et al. 2006; Kelnar et al. 2002; Lunn, Cowen, and Fraser 1997). Despite these differences, shifting our attention to the proliferative period following quiescence may be more relevant for assessing testicular toxicity in juvenile animals.
Postnatal Development of the Rat Epididymis
The development of the epididymis occurs in 3 stages. The first phase is the undifferentiated phase (PND 0–14) at which time there is no epithelial cell specialization and the caput tubules are larger in diameter than the cauda tubules (Arroteia et al. 2012). The next phase is called the differentiation phase (PND 15–44) at which time there is cell differentiation into principle, pencil (narrow), apical, basal, clear, and halo cells; this differentiation occurs in a head to tail gradient (Arroteia et al. 2012). During this phase, there is proliferation of fibromuscular tissue around the cauda tubules, and exfoliated germ cells and debris can be seen in the tubules as early as PND 20. The debris likely correlates to the density-dependent apoptosis and exfoliation in the testis at around PND 15–18. Exfoliated cells generally peak at around PND 25–32 prior to puberty. It is important to differentiate this normal exfoliation from the exfoliation of germ cells that can be seen with test articles that interfere with microtubule formation in the Sertoli cells, such as colchicine or carbendazim, or other Sertoli cell toxicants, such as phthalate esters (Creasy and Foster 2002; Lanning et al. 2002). The third expansion phase begins after PND 45 when the tubules enlarge and fill with spermatozoa (Arroteia et al. 2012). By PND 77, spermatid density in the epididymis reaches a maximum level in Sprague-Dawley rats; this occurs by PND 91 in Wistar Han rats (Campion et al. 2013). For additional information and images of the developing epididymis in the rat, the reader is referred to Picut and Remick (2016).
When interpreting the testis from animals of unknown age, the epididymis may offer clues to the age of the animal. The epididymis should be devoid of sperm until PND 52 when the step 19 spermatids that formed in the testis at PND 43 have had time to transport into the caput of the epididymis. Generally speaking, it can take about 2 weeks (11–13 days) for a released spermatozoon to transition the full length of the mature epididymal tubules (Bellentani et al. 2011; Jaakkola and Talo 1983).
Evaluating Testis in Immature Large Animals
Testis evaluation in large laboratory animals, such as the dog, pig, and cynomolgus monkey, presents special concerns for the toxicologic pathologist. In the rat, there is relatively minimal interanimal variability in the timing of development or in the histologic appearance of the testes or epididymis. However, in larger animals, there is considerable interanimal variability in these tissues. This variation, coupled with low group numbers, makes the identification of test article–related effects in the testis or epididymis especially difficult in large animals that may exhibit varying stages of maturity (Haruyama et al. 2012; Luetjens and Weinbauer 2012; Vogel 2000).
Different grading schemes and terminology have been proposed for recording the degree of sexual maturation in large laboratory animals (Haruyama et al. 2012; Navratil et al. 2014). Terminology such as prepuberty, puberty, and early adult do not correspond precisely with a determination of sexually immature and sexually mature, and this is because the transitions through these stages are gradual and difficult to pinpoint. Also, there is inconsistency in the literature concerning the use of terminology (Table 2; Bode et al. 2010; DeNoon 2012; Dreef, Van Esch, and De Rijk 2007; FDA 2006; Goedken, Kerlin, and Morton 2008; Haruyama et al. 2012; Helke et al. 2016a, 2016b; Hirsch et al. 1985; Howroyd 2015; Howroyd, Peter, and de Rijk 2016; Jorgensen et al. 1998; Lanning et al. 2002; Marty et al. 2003; Navratil et al. 2014; Ojeda and Skinner 2006; Parker 2011; Sengupta 2013; Smedley et al. 2002; Smidt 1970; Stoker et al. 2000; Svendsen 2006; Takakura et al. 2014; Thuilliez et al. 2014). A distinction must also be made between the onset of puberty and puberty itself. Some papers equate sexual maturation to the onset of puberty while others equate it to the adult stage (Haruyama et al. 2012; Navratil et al. 2014). Terminology aside, it is generally acceptable for the pathologist to simply record the male reproductive tract as being either sexually mature or sexually immature, with the former represented by the presence of all germ cell stages in the testis and spermatozoa in the epididymis (Halpern et al. 2016; Haruyama et al. 2012; Hukkanen, Halpern, and Vidal 2016; Luetjens and Weinbauer 2012). However, by this standard, the diagnosis of sexual maturity should depend on the examination of both the testis and epididymis.
Ages of Puberty and Sexual Maturity in Laboratory Animals.

Note. Publications refer to onset of puberty, puberty, or sexual maturation. Terminology is not necessarily consistent between publications; most publications either refer to sexual maturation or to puberty but not to both. Some publications refer to onset of puberty rather than puberty (noted as “onset” in table).
a Stoker et al. (2000). b Takakura et al. (2014). c Food and Drug Administration (2006). d Thuilliez et al. (2014). e Ojeda and Skinner (2006). f Sengupta (2013). g Marty et al. (2003). h Lanning et al. (2002). i Dreef, Van Esch, and De Rijk (2007). j Smidt (1970). k Navratil et al. (2014). l Bode et al. (2010). m Svendsen (2006). n Jorgensen et al. (1998). o Helke et al. (2016a, 2016b). p Howroyd (2015). q Goedken, Kerlin, and Morton (2008). r Parker (2011). s Haruyama et al. (2012). t Smedley et al. (2002). u DeNoon (2012). v Hirsch et al. (1985).
To underscore the high degree of interanimal variability in the timing of sexual maturation, Haruyama et al. (2012) demonstrated that the testes of 4-year-old or 4-kg cynomolgus monkeys can be at any stage from grade 1 (immature) through grade 6 (adult). However, Haruyama et al. (2012) identified age and body weight as useful guidelines for gauging puberty. Ninety-six percent of cynomolgus monkeys will be at least at grade 4 (puberty) if they are at least 5 years old or weigh at least 5 kg (Haruyama et al. 2012). Another guideline to help confirm puberty in cynomolgus monkeys is the testis weight; if an individual testis weighs more than 8 g, then the testis will assuredly (virtually 100%) be in puberty (grade 4) or greater (Haruyama et al. 2012). These age, body weight, and organ weight guidelines can be used during the study design and selection of animals and are useful to help confirm the pathologist’s determination of sexual maturity by light microscopy; however, they should not be used at the exclusion of histopathology. Unfortunately, such nonhistopathological parameters are not yet available for dogs or pigs. Even though external confirmatory parameters of sexual maturity are not necessary in rats in light of their low level of interanimal variability and precise knowledge of their age, balanopreputial separation is used as an external indicator of onset of puberty in the rat (Marty, Crissman, and Carney 2001).
Similar to cynomolgus monkeys, Gottingen minipigs exhibit a high degree of individual animal variability in terms of testicular development. Gottingen minipigs at 4 months of age may range from reproductively prepubertal to adult. Their epididymides may be devoid of sperm and contain abundant exfoliated germ cells in the prepubertal stage, or they may have tubules expanded with spermatozoa compatible with the adult stage. Sexual maturation varies with strain of minipig. Gottingen minipigs mature much earlier (i.e., approximately 2 months of age) than Yucatan minipigs (i.e., 4.4 months of age; Navratil et al. 2014).
Not only is there conflicting data and high individual animal variability in the timing of sexual maturation in large laboratory animals, the immature tubules are difficult to distinguish from tubular degeneration and/or atrophy caused by toxicity (Dreef, Van Esch, and De Rijk 2007). This is not the case in the maturing rat, in which, as long as apoptosis, germ cell exfoliation, and occasional Sertoli-only tubules are accounted for, the tubules appear healthy. Among dogs 6–7 months of age, during a time that corresponds to puberty, 75% of males may have spontaneous focal degeneration and/or atrophy of the testis (Figure 8; Goedken, Kerlin, and Morton 2008). This degeneration and/or atrophy of the immature testes has also been referred to as hypospermatogenesis, which is characterized by low numbers of spermatocytes and round spermatids that give the spermatogenic epithelium an irregular mottled appearance. The incidence of this finding decreases to 10% in dogs that are greater than 11 months of age (Goedken, Kerlin, and Morton 2008). In addition to hypospermatogenic tubules, immature dogs have a relatively high incidence of multinucleated giant cells composed of round spermatids (Figure 9) and swollen spermatids (Figure 10; Goedken, Kerlin, and Morton 2008; Rehm 2000). Hypoplastic tubules are another spontaneous finding in the testes of dogs, but such tubules are generally not mistaken for toxic effects. Hypoplastic tubules, also known as atrophic or Sertoli-only tubules, occur in 25–40% of dogs less than 12 months of age, and this incidence falls to 14–17% by 12 months of age (Goedken, Kerlin, and Morton 2008).
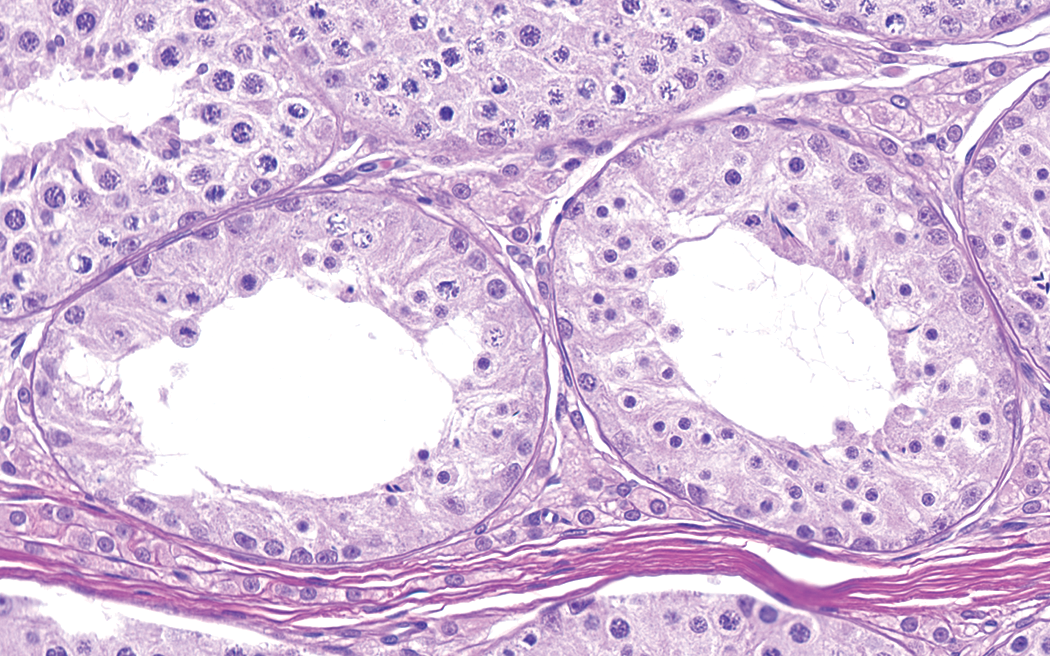
Testis from 10-month-old dog. Young dogs commonly have hypospermatogenic tubules, in which there is a paucity of spermatogenic epithelium. It is difficult to distinguish the low germ cell cellularity in these tubules from toxic lesions. H&E. Original objective 40×.
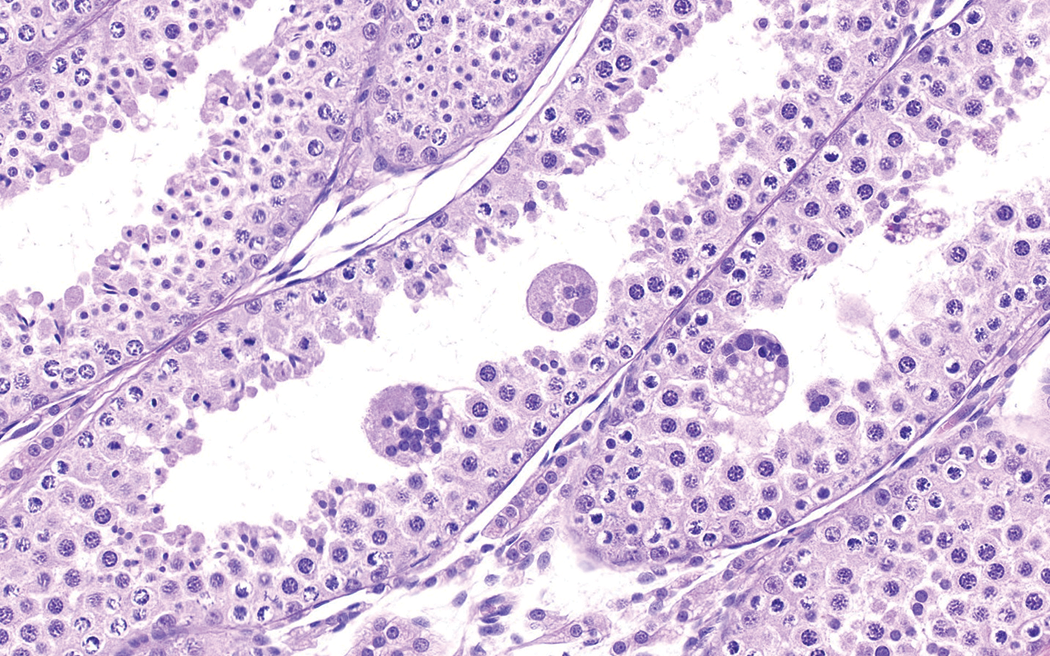
Testis from 8-month-old dog. Giant cells representing syncytial round spermatids are common in immature dog testes. This animal is 8 months old. H&E. Original objective 40×.
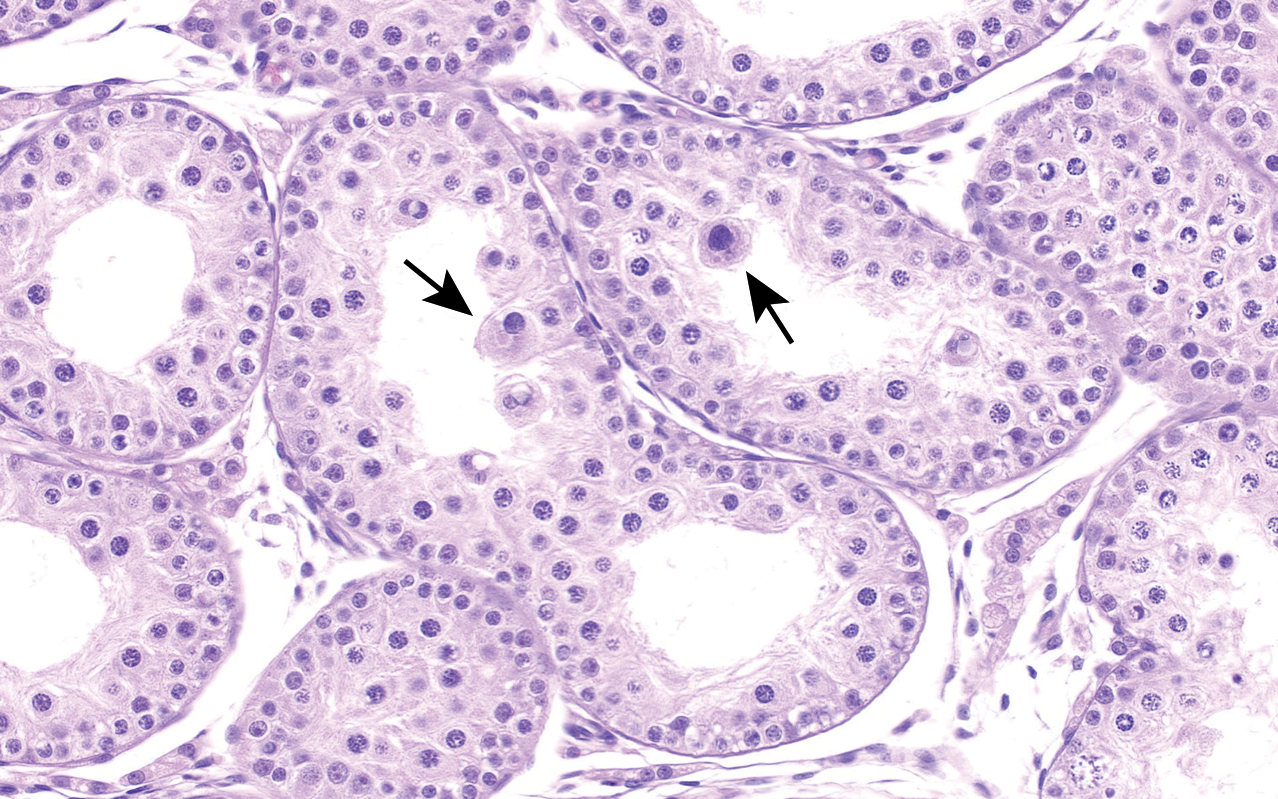
Testis from 8-month-old dog. Swollen spermatids (arrows) are a spontaneous finding in immature dogs and can also be mistaken for toxic changes. H&E. Objective magnification 40×.
In minipigs, just as in dogs, there is a baseline occurrence of degenerative-appearing tubules, but those degenerative-appearing tubules exist on a continuum with Sertoli-only tubules and are lumped under the more benign terms tubular hypoplasia and/or atrophy (Bode et al. 2010; Helke et al. 2016a; Thuilliez et al. 2014; Figure 11). Unlike dogs, there is currently no distinction in terminology between Sertoli only and hypospermatogenic tubules in pigs. However, terminology itself is not the primary concern; instead, it is important that pathologists recognize the high incidence of spontaneously occurring degenerative-appearing tubules in peripubertal dogs and pigs.
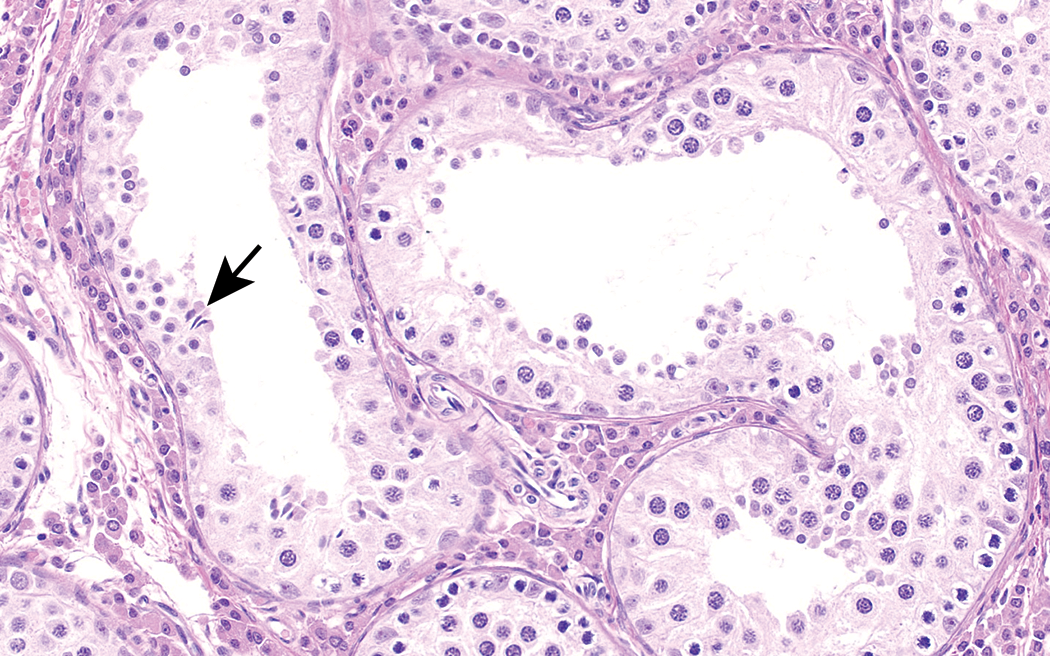
Testis from 4-month-old Gottingen minipig. Hypoplastic/degenerative tubules, characterized by a paucity of germ cells and a mottled irregular appearance, are a common spontaneous finding in the Gottingen minipig at puberty. These degenerative-appearing tubules should not be mistaken as toxic effects. The small size of the Leydig cells and rare elongated spermatids (arrow) provide clues that this animal is immature. H&E. Original objective 40×.
Haruyama et al. (2012) states that cynomolgus monkeys should be at least in puberty (grade 4) to be considered “appropriate” for evaluation of test article–related effects on the male reproductive system. However, pathologists should not automatically apply this principle to the dog or the pig. It could be argued that the pubertal period in dogs and pigs, riddled with degenerative-appearing tubules, is precisely the most inappropriate time to examine the testes.
Testis and Epididymides Weights
The age of animals (rats, dogs, pigs, and cynomolgus macaques) can impact the interpretation of testis and epididymides weights (Hukkanen, Halpern, and Vidal 2016). It is well known that the absolute testis and epididymides weights are conserved in adult animals even when there is a drop in body weight, in adult animals, and the testis and epididymis weight relative to body weight actually increases (Creasy 2003). A drop in absolute testis or epididymides weights in adult animals can be a signal for testicular toxicity and, in particular, germ cell loss (Lanning et al. 2002). This is not the case in immature animals where, in the authors’ experience, a drop in body weight will result in lower absolute testes and epididymides weights and lower or stable relative weights. One report stated that food restriction or loss of body weight did not affect the absolute weights of the testes or epididymides of maturing rats (Rehm et al. 2008). However, that report was based on a mild (20%) food restriction and did not use rats less than 6 weeks of age. In other words, the study did not address the effect of body weight loss or food restriction in prepubertal animals.
Conclusions
Even though the immature rat testis has unique histologic features (i.e., apoptosis, germ cell exfoliation, and absence of spermatocytes and spermatids), once the pathologist becomes familiar with these features, the neonatal through juvenile periods provide a convenient and opportune time to identify test article–related effects in short-term targeted studies. During this critical window, there is very little interanimal variability, and the immature rat testis can provide a significant uniform and susceptible cell population for identifying test article–related toxicity. In dogs, monkeys, and pigs, which are characterized by high interanimal variability in maturation rate and relatively high incidences of testicular degeneration and/or atrophy (dogs and pigs), early stages of sexual maturity (i.e., peripubertal stages) may be less appropriate times to evaluate test article–related effects.
Footnotes
Acknowledgments
The authors wish to thank the Society of Toxicologic Pathology’s (STP) Annual Symposium Committee, 2016 Scientific Program Committee, 2016 Continuing Education Subcommittee, and associated organizational support staff for their support and invitation to present this material at the 2016 STP Annual Symposium. The authors also thank Cynthia L. Swanson, MS, Charles River Laboratories (legacy WIL Research) in North Carolina, for her technical expertise in the preparation of the digital histopathology images used in this article.
Authors’ Contribution
Both the authors (CP, AR) contributed to the conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was fully funded by Charles River Laboratories (legacy WIL Research).
