Abstract
Within the substantially different time scales characterizing human and rodent brain development, key developmental processes are remarkably preserved. Shared processes include neurogenesis, myelination, synaptogenesis, and neuronal and synaptic pruning. In general, altricial rodents experience greater central nervous system (CNS) immaturity at birth and accelerated postnatal development compared to humans, in which protracted development of certain processes such as neocortical myelination and synaptic maturation extend into adulthood. Within this generalization, differences in developmental rates of various structures must be understood to accurately model human neurodevelopmental toxicity in rodents. Examples include greater postnatal neurogenesis in rodents, particularly within the dentate gyrus of rats, ongoing generation of neurons in the rodent olfactory bulb, differing time lines of neurotransmitter maturation, and differing time lines of cerebellar development. Comparisons are made to the precocial guinea pig and the long-lived naked mole rat, which, like primates, experiences more advanced CNS development at birth, with more protracted postnatal development. Methods to study various developmental processes are summarized using examples of comparative postnatal injury in humans and rodents.
“The well-known regional heterogeneity of the timing of developmental events may well result in each small region, or tract, or system, having its own “growth spurt,’ and hence its own vulnerability, in relation to each of its own component processes, hidden and largely uninvestigated beneath the major spurt in whole organ weight.”
The essential challenge when predicting the likely human impact of a developmental lesion identified in experimental species is encapsulated in this statement by Dobbing and Sands. 1 Although key processes are highly comparable across rodent and human brain development, these events occur within substantially different time scales. 1 –3 Further, developmental time scales across species are regionally heterogeneous across different brain structures, thus setting the stage for differential periods of vulnerability in a regionally specific manner. 1,4
For purposes of cross-species comparison, central nervous system (CNS) shared developmental processes of neurogenesis, neuronal death, myelination, synaptogenesis, and synaptic pruning are shown in Figure 1. In general, altricial rodents such as rats and mice experience greater CNS immaturity at birth and accelerated postnatal development compared to humans. 4 The fundamental processes of neuronal birth, migration, and death occur concordantly. These are largely complete at birth in humans 5 but may continue up to 18 months of age in human prefrontal cortex. 6 Proliferation of neuronal processes, synapse formation, and maturation of neurotransmitter systems is completed in early childhood 7 and results in enormous proliferation of synapses that require pruning to refine circuits. This is in part experience dependent and continues into young adulthood. 8 Similarly, myelination is also protracted; both processes are completed last in phylogenetically recent structures such as the prefrontal cortex. 9 Prefrontal cortex maturity with complete synaptogenesis and myelination occurs at approximately 17 to 25 years of age in the human 10 compared to postnatal day (PND) 90 in the rat.
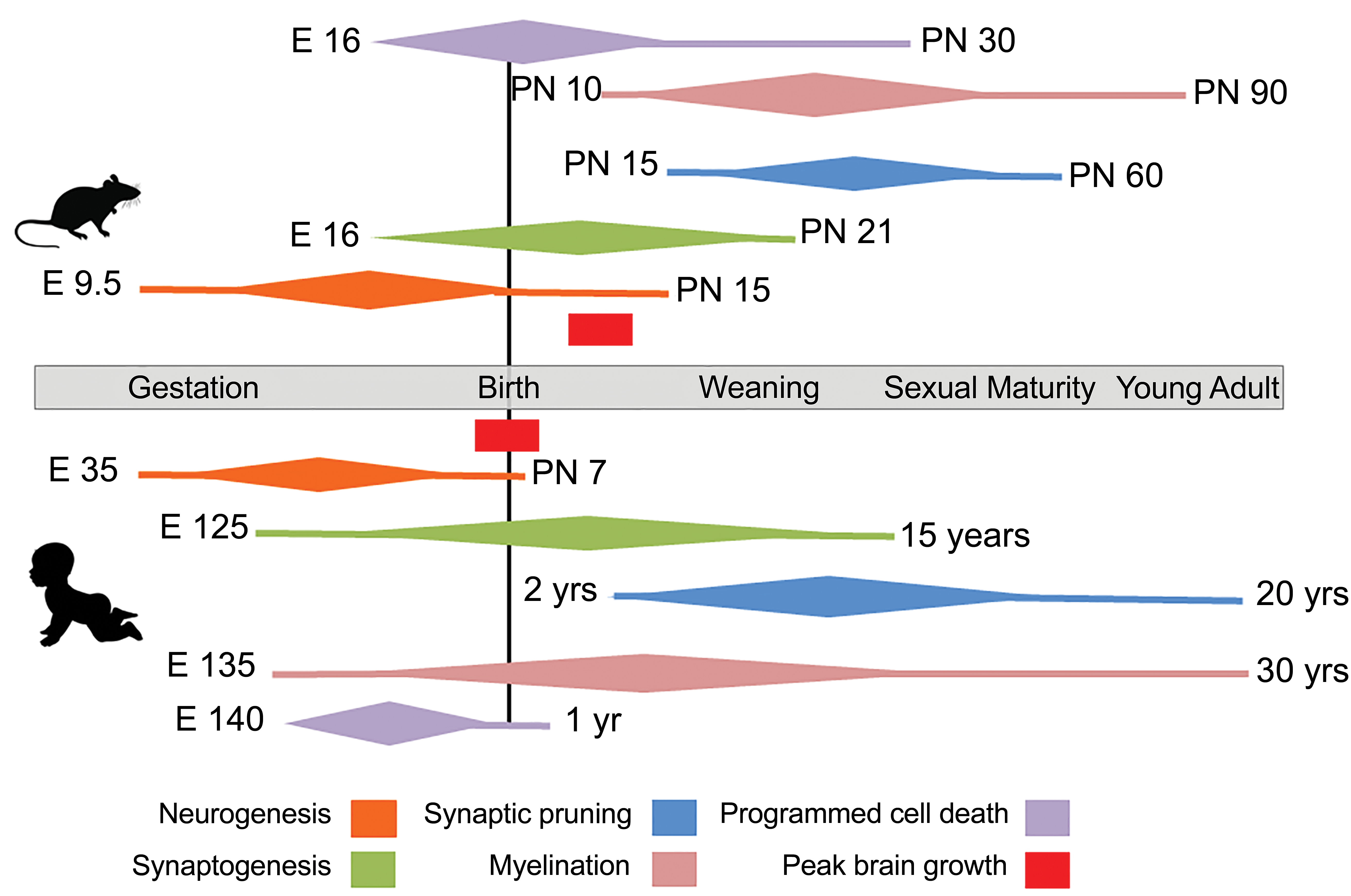
Major CNS developmental processes in rat and human. Approximate time lines of these processes are shown in relation to anchor events of birth, weaning, sexual maturity, and adulthood. Brain growth spurts are shown in red. Individual processes are color-coded, with peak activity indicated by the widest portion of the diamond. Adapted from Semple et al, 4 Lenroot and Giedd, 9 and Clancy et al. 11 CNS indicates central nervous system.
In altricial rodents such as rats and mice, many of these processes are contracted into the time period between birth, weaning, and sexual maturity, occurring at approximately 6 weeks of age. In these species, neurogenesis is still quite active in the first two postnatal weeks. 12 Myelination is rudimentary at birth but accumulates rapidly to near completion at sexual maturity. 13,14 Similarly, synapse formation and pruning are also near completion by young adulthood. These major developmental processes are compressed into a very short 3 to 4 week period after birth. 12 Because of this, neonatal altricial rodents subjected to an insult during this period are prone to more profound injury of multiple development processes. Postnatal CNS development of precocial rodents such as the guinea pig, or neotenic species such as the naked mole rat, 15 more closely approximates the developmental time scale seen in humans. The guinea pig is well developed at birth and has a correspondingly larger, more mature brain, a more developed hippocampus, 16 and some degree of cortical folding. 17 Like humans, the naked mole rat has a more developed brain at birth and a protracted postnatal development up to 3 years. 18 Further, it retains the 3R isoform of tau, which is lost in mice as they mature. This isoform is retained in humans, and altered ratios of tau 3R to 4R isoforms characterize several neurodegenerative diseases.
A general rule of thumb assumes that brain development of a 7- to 10-day old altricial rodent such as the rat is roughly equivalent to a baby at birth, with the last trimester of human gestation corresponding to PND 1 to 10 in rats. 11 This is based on the seminal work of Dobbing and Sands which characterized the timing of peak brain growth spurts across various species, with emphasis on rat–human comparisons. 1,19 In the rat, peak brain growth as a percentage of adult body weight occurs around PND 7 to 8, whereas this occurs at the time of birth in the human. 19 Adult brain weight is reached shortly before puberty in humans, 20 whereas this landmark is achieved well after puberty, on PND 60, in the rat. These guidelines are still a general basis for comparison but have been challenged and refined over time. 3
Postnatal Synaptic Development
An example of our evolving understanding of comparative development across species is illustrated in studies of postnatal synaptic growth in rats and humans. 21,22 The authors established a baseline for comparison (accumulation of 63% of the synaptic number of adult) in a defined brain region (visual cortex), then assessed the ages in human and rat at which this criterion was met. The authors concluded that synaptic development in the PND 15 rat was equivalent to that of a human at birth. Much of this seminal work was done using transmission electron microscopy 23 focusing predominately on development of type I and II synapses over time. Type I, or asymmetric, synapses occur between an axon and a dendritic spine, exhibit characteristic spherical synaptic vesicles, and constitute most abundant synapse in the CNS. Type II, or symmetric, synapses occur primarily on dendrite shafts and neuronal cell bodies and exhibit flattened synaptic vesicles within the axonal bouton. Synapses were quantified by taking multiple images at each developmental stage and assembling these into montages that encompassed a given area of neuropil. Presently, this type of analysis is done using a raft of synaptic markers, both pre- and postsynaptic. 24,25 The combination of a pre- and postsynaptic marker can be used to identify synaptic contacts. This can be achieved using immunohistochemistry 26 or in mice via transgenic approaches that allow large scale labeling of the entire brain. 27
Synaptic Pruning
In humans, there is massive synaptic proliferation during infancy, peaking at 1 to 2 years. Synaptic numbers at this age exceed the adult mean by approximately 50%, thus requiring pruning to refine circuits. 22,28 In the visual and auditory cortex, this is complete by about 6 years, whereas in the prefrontal cortex, this continues through adolescence. 7,29 In rodents, spine pruning occurs from weaning through early adulthood. 30 Abnormal synaptic pruning is a primary lesion underlying autism spectrum disorder 31 and is thought to underlie developmental psychiatric disorders. 32
Synaptic pruning occurs via innate immune mechanisms utilizing microglia through the process of trogocytosis. 33,34 Trogocytosis is the phenomenon whereby resident microglia are able to remove small portions of neuronal processes during postnatal development. Interference with this process in genetically altered mice results in impaired synaptic pruning 33 and social behavior. 35
Pruning is experience dependent 36 ; this has been modeled in macaques by assessing synaptic density in sensory territories after digit amputation. 37 Sensory deprivation using whisker trimming results in retention of dendritic spines in the associated region of the barrel cortex. 38 This process can be impacted by a variety of sublethal insults that create subtle defects. For example, while severe neonatal hypoxia causes obvious lesions of hemorrhage and necrosis, 39 mild hypoxia results in delayed dendritic development and reduced neuronal activity, without obvious effects on neuronal number, microglia, or gross alterations in brain growth. 40
Postnatal Development of Neurotransmitter Systems
An alternate means to examine development of neuronal circuits is to assess expression and function of various neurotransmitter systems. 2,41 By comparing developmental patterns of γ-aminobutyric acid (GABA) expression, Romijn et al identified rat PND 12 to 13 as the developmental stage comparable to human day 1 or birth and suggested that PND 2 to 7 in rat corresponds to the human third trimester. 3 The morphologic and behavioral consequences of agents impacting neurotransmitter function and development of associated circuits are illustrated by the effect of neonatal exposure to anesthetics. 42 Many anesthetics achieve neuronal suppression by acting as GABA agonists (eg, propofol and isoflurane) or N-methyl-D-aspartate antagonists (eg, ketamine). 43,44
Receptor hyperactivation or blockade results in neuronal death 45 exceeding programmed cell death characterizing normal development. 46 The primary means to assess neurotoxicity is to quantify regional neuronal cell death, using terminal deoxynucleotidyl transferase dUTP nick-end labeling, cleaved caspase-3 immunohistochemistry, or alternately, silver stains. 47 Overall, incidence of neuronal death declines in the first postnatal week 46 and but differs by region, thus requiring examination of the entire brain at several time points. 48
Because anesthetic use in children is thought to be associated with later learning difficulties, 45 the functional effects of toxicity in rodents are also assessed using behavioral tests that assess motor or cognitive function. 49 These reveal sex differences 50 in vulnerability to impairments in spatial and nonspatial memory, emergence of hyperactivity disorders, and anxiety-like behavior. Sexual differentiation of the brain is associated with testosterone surges in males. In rats, this occurs during embryonic day (E) 18 to 19 and again within a few hours of birth. 51
The deleterious effects of neonatal anesthetic exposure have been consistently described in rodents 52,53 ; however these are less clearly discerned in primates. Neuronal and oligodendroglial cell death in macaques is evident in subcortical regions following prenatal anesthetic exposure and in various laminae of sensory regions if exposure occurs after birth 54 but are less marked than in rodents. 55,56 In humans, the literature regarding the effect of neonatal anesthetic exposure on subsequent development is much less coherent. Although there is general agreement that repeated neonatal anesthesia is undesirable, 42 its impact on later learning and behavior is confounded by other variables (eg, the underlying condition that required anesthesia). This difference in outcome between rodents and humans is influenced by the greater developmental speed in the immediate neonatal period in rodents compared to humans.
Myelination
Regionally diverse oligodendrocyte progenitor cell populations differentiate in a spatially defined manner. 57 These oligodendrocyte populations are guided by a multitude of intrinsic and extrinsic cues to execute myelination in a tightly controlled spatial and temporal fashion. 58,59 This precise and time-limited process can be visualized using live cell imaging in zebrafish 60 and has been beautifully illustrated in rats and mice using myelin basic protein (MBP) immunohistochemistry or MBP-driven Lac Z expression. 61,62 Myelination proceeds in a caudal to rostral direction in the brain, beginning in cranial nerve roots, medulla, cerebellum, mesencephalon, and proceeding anteriorly through association fibers to anterior cortex. In the spinal cord, myelination starts in cervical regions, proceeding caudally, initially in ventral cord, then followed by dorsal cord.
In contrast to limited myelination at birth in rats, the peripheral nerves, pons, and cerebellar peduncles are fairly well myelinated at birth in humans. Myelination persists into young adulthood in humans and can exhibit significant remodeling in the adult human brain 63 in response to acquisition of new skills. 64 White matter is particularly sensitive to injury during 23 to 32 weeks of human gestation, and prenatal white matter injury can result in profound developmental consequences such as cerebral palsy. 65 Because myelination is so protracted after birth in humans, it can be influenced by a broad array of postnatal developmental or environmental cues to create subtle but pervasive phenotypes. 66 For example, early adversity in children is associated with callosal hypoplasia, altered myelination of multiple tracts, and reduced hippocampal, amygdalar, and cortical volume. 67,68 In some studies, these are lateralized. Maternal separation in neonatal mice 69 results in similar defects in myelination in left amygdala, cortex, and hippocampus. Tractography or diffusion tensor imaging is a powerful in vivo means to assess white matter development. 70 In the mouse study referred to above, 69 one can appreciate reduced white matter development in the amygdala. In humans, very subtle lateralizing defects in children exposed to early adversity can be appreciated using this technology. 67
Regional Differences in Postnatal Development
Apart from prefrontal and association cortices, commonly cited species differences in postnatal developmental trajectory occur in hippocampus, cerebellum, and olfactory bulb. 2 Although the bulk of neurogenesis across humans and most experimental species is complete at birth, these structures undergo a wave of secondary neurogenesis and are thus susceptible to postnatal insult. Hippocampal development is more advanced in humans and macaques at birth (approximately 80% of the granule cell population is formed by birth compared to approximately 20% in the rat). In the rat hippocampus, the majority of granule cell development in the dentate gyrus occurs between birth and PND 21 and continues throughout the animal’s life. This occurs to a much lesser extent in humans. 71 Hindbrain structures are among the earliest developing regions of the brain across species. 11 An exception to this is cerebellar development. The 50% adult size landmark is reached at 1 year in humans, at birth in the rhesus monkey, and at PND 15 in the rat. In the rodent cerebellum, extensive postnatal neurogenesis occurs until PND 10 and is largely complete by PND 21. 72,73 In contrast to humans, the olfactory bulb in rodents is far more developed, and neurogenesis continues throughout life.
Development of the visual system has been highly described and illustrates the extent of developmental heterogeneity across structures and species. 74,75 In both rodents and primates, most retinal cell types are formed in utero 76 with optic chiasm development occurring by birth. Synapse formation in the lateral geniculate nucleus (LGN) continues through the early postnatal period, with pruning continuing past weaning in rodents. 77 Synapse formation occurs in parallel in the superior colliculus and occurs along a more protracted time line in the visual cortex. Because visual acuity is lower and decussation is almost complete in rodents, lamination of the LGN is far less evident in mice than it is in primates. 75 Additionally, axons originating from the retina project largely to the superior colliculus in rodents, consistent with their reflexive lifestyle. 78 This contrasts markedly with primates that rely more heavily on visual acuity to guide decision-making. Their evolutionary adaptations include a foveate retina, approximately 50% of retinal axonal decussation, and a dominant geniculo-striate pathway.
Glial Development and the Blood–Brain Barrier
In the rat, astrogenesis and oligodendrogenesis are initiated in late gestation, peak in the early the neonatal period, and decline by PND 20. 79 In mice, astrogenesis starts in late gestation (E18) and lasts until approximately PND 7. Limited astrogenesis is retained through adulthood. Microglial invasion of the brain occurs earlier (approximately E9.5 in mice and slightly later in rats), and microglia accompany developing neurons from mid gestation until birth. 80 Microglial maturation and ramification take place after birth, when astrocytes are already present, suggesting that differentiated astrocytes might contribute to microglia maturation. Both cell types are thought to play a role in postnatal developmental angiogenesis through their close association with developing blood vessels. After the establishment of the primary brain vasculature, astrocytes contribute to the formation of the blood–brain barrier (BBB). The development of the BBB in the human is a gradual process, 81 beginning in utero and acquiring capabilities similar to that of an adult at approximately 6 months of age. 82 Similarly, in the rat, the permeability of the BBB is high at birth; this gradually decreases in the first few weeks after birth. 83
Conclusions
The substantially different developmental time scales of key CNS developmental processes across species underpin conduct and interpretation of experimental animal studies so as to accurately model human developmental injury. In general, altricial rodents experience greater CNS immaturity at birth and accelerated early postnatal development compared to humans. Consequently, major developmental processes are compressed into a short neonatal period, rendering neonatal rodents vulnerable to injury of multiple development processes during this period. Like humans, postnatal rodent CNS development is heterogeneous across different brain structures, thus setting the stage for differential periods of vulnerability in a regionally specific manner and requiring comprehensive evaluation of the entire brain at several time points. However, once these differences are appreciated, the highly conserved processes of neurogenesis, neuronal death, myelination, synaptogenesis, and synaptic pruning can be effectively studied across species.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
