Abstract
A cutaneous response (localized swelling and/or erythema of the skin) has been noted in dog toxicology studies in which multiple, unrelated compounds were administered orally with copovidone as a vehicle. The response has been noted in studies with 6 different test items that are structurally unrelated and span several different therapeutic indications spanning an approximate 6-year period (2009–2015). A factor common among the studies is the formulation—a copovidone amorphous solid dispersion (ASD). Cutaneous responses have not been observed in dogs administered non-ASD formulations of the same test items but have occasionally been noted in placebo (copovidone control) dogs. Polyvinylpyrrolidone (a polymer of one of the primary components of copovidone) has been reported to result in similar findings in dogs when administered by the intravenous route. Considerations for the role of copovidone and the potential role of histamine in the cutaneous changes are outlined.
Keywords
Introduction
A cutaneous response (localized swelling and/or erythema of the skin) has been noted in dog toxicology with 6 different test items that are structurally unrelated and span several different therapeutic indications over an approximate 6-year period (2009–2015). A factor common among the studies was the formulation—an amorphous solid dispersion (ASD) containing copovidone as one of the principal inactive ingredients. Copovidone is a copolymer of equimolar vinylpyrrolidone (VP) and vinyl acetate (VA) and is commonly utilized as a pharmaceutical excipient. Cutaneous responses have not been observed in dogs administered non-ASD formulations of the same test items but have occasionally been noted in placebo (copovidone control) dogs. Polyvinylpyrrolidone (PVP; a polymer of one of the components of copovidone) has been reported to result in similar findings in dogs when administered intravenously (Halpern, Musso, and Neveu 1955; Marshall and Hanna 1957, 1958). Considerations for the role of copovidone in the cutaneous changes in the toxicology studies are discussed.
Cutaneous Responses in Studies with Copovidone-based ASDs
Timing and Characteristics of the Response
A cutaneous response has been identified in several dog toxicology studies in which a copovidone-based ASD was utilized (Tables 1 –3). The cutaneous findings were typically evident on the first day of dosing and became less frequent and less severe as dosing was continued. In one study (study “C1” in Table 1), cutaneous changes similar to those noted in dogs administered the copovidone-based ASD formulations were present in a placebo control dog that had received copovidone vehicle control. Since the cutaneous changes in the placebo control dog occurred on the first day of dosing and plasma samples from that day revealed no quantifiable levels of test item in any of the control dogs, the cutaneous changes were considered to be due to copovidone rather than an inadvertent administration of the test item.
Cutaneous Changes with Gavage but Not Capsule Dosing.
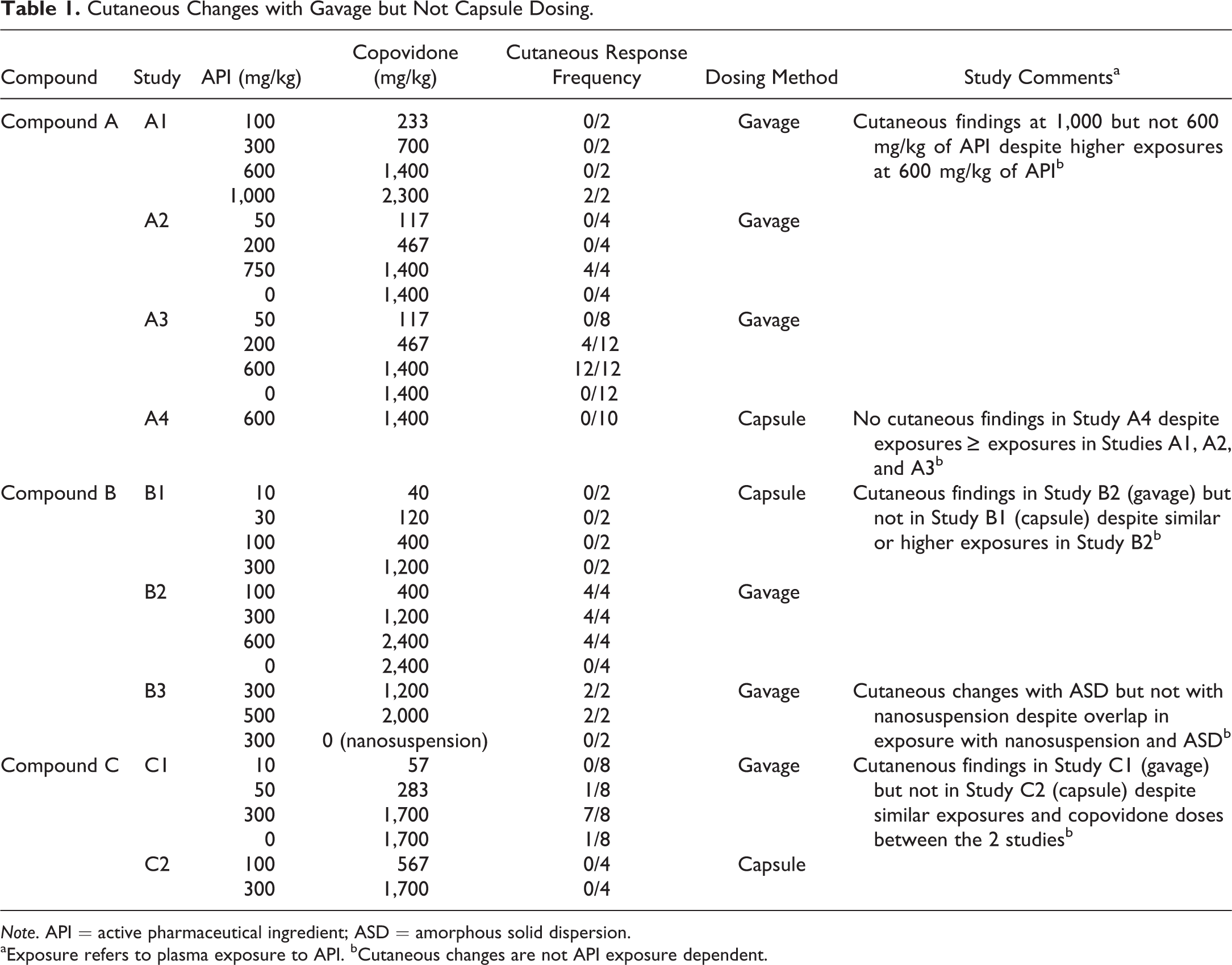
Note. API = active pharmaceutical ingredient; ASD = amorphous solid dispersion.
aExposure refers to plasma exposure to API. bCutaneous changes are not API exposure dependent.
Cutaneous Changes with Gavage Dosing (No Capsule Studies).
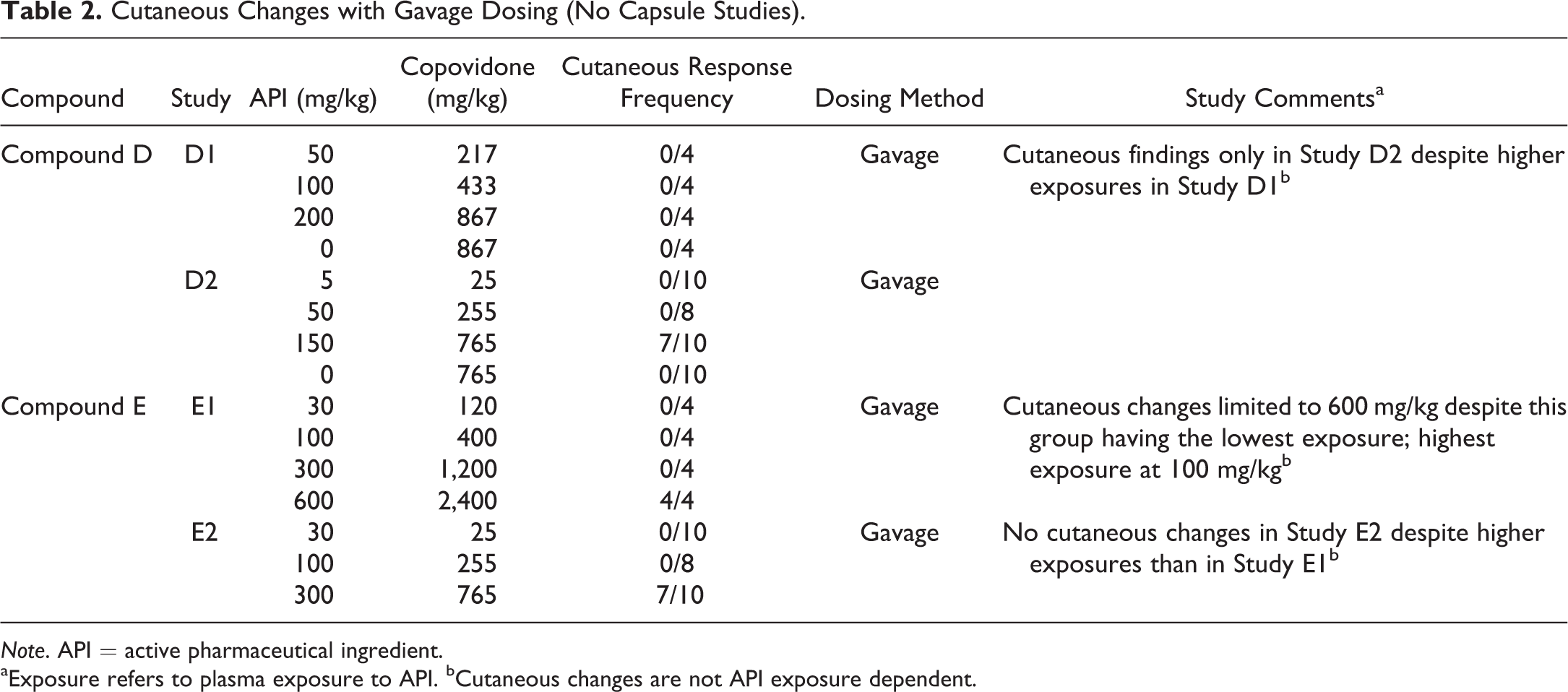
Note. API = active pharmaceutical ingredient.
aExposure refers to plasma exposure to API. bCutaneous changes are not API exposure dependent.
Cutaneous Changes Rarely Occur with Capsule Dosing.
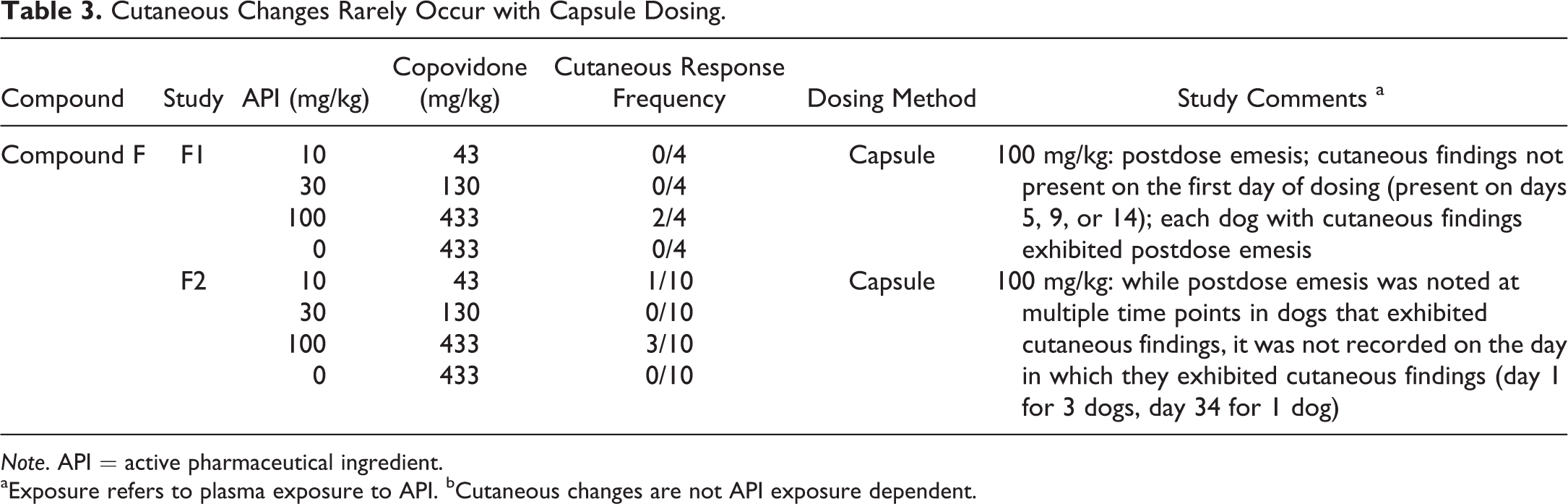
Note. API = active pharmaceutical ingredient.
aExposure refers to plasma exposure to API. bCutaneous changes are not API exposure dependent.
The cutaneous response was characterized by swelling and/or erythema of the ears and/or muzzle or, less frequently, the paws. Some dogs exhibited urticaria involving the neck, chest, and paws. The response was typically observed on the first day of dosing, being evident within a short time after dosing (within 15–30 min). Some dogs exhibited an increase in activity, characterized by shaking and rolling, which was considered to be a manifestation of pruritus. In some instances, diphenhydramine was administered to help ameliorate the clinical signs, resulting in rapid resolution of the cutaneous changes. Dogs that did not receive diphenhydramine exhibited resolution of the cutaneous changes over the course of the day. There was no evidence of a systemic response (e.g., no evidence of weakness suggestive of a decrease in systemic blood pressure, no respiratory distress suggestive of pulmonary edema).
Cutaneous Response Considered Unrelated to Test Item
The cutaneous response has been noted with 6 different test items, each formulated as a copovidone-based ASD formulation (compounds A–F; Tables 1 –3). The compounds all had different mechanisms of action and spanned a variety of therapeutic areas. Furthermore, the test items were structurally unrelated and lacked structural alerts (Deductive Estimation of Risk from Existing Knowledge [DEREK]) for hypersensitivity, which is consistent with a lack of relationship to test item. In both within and between studies of the 6 test items, there was a lack of test item/active pharmaceutical ingredient exposure dependency of the cutaneous response (Tables 1 –3). Furthermore, cutaneous responses have not been observed in dogs administered non-ASD formulations of the same test items (e.g., compounds A and B).
All compounds that were administered to dogs were also administered to rodents (rat and/or mouse) via gavage administration. For the majority of the compounds, an identical copovidone-based ASD formulation was utilized. Cutaneous findings were not apparent in the rodent.
Cutaneous Response Is Dose Dependent with Respect to Copovidone and Predominantly Noted with Gavage Dosing
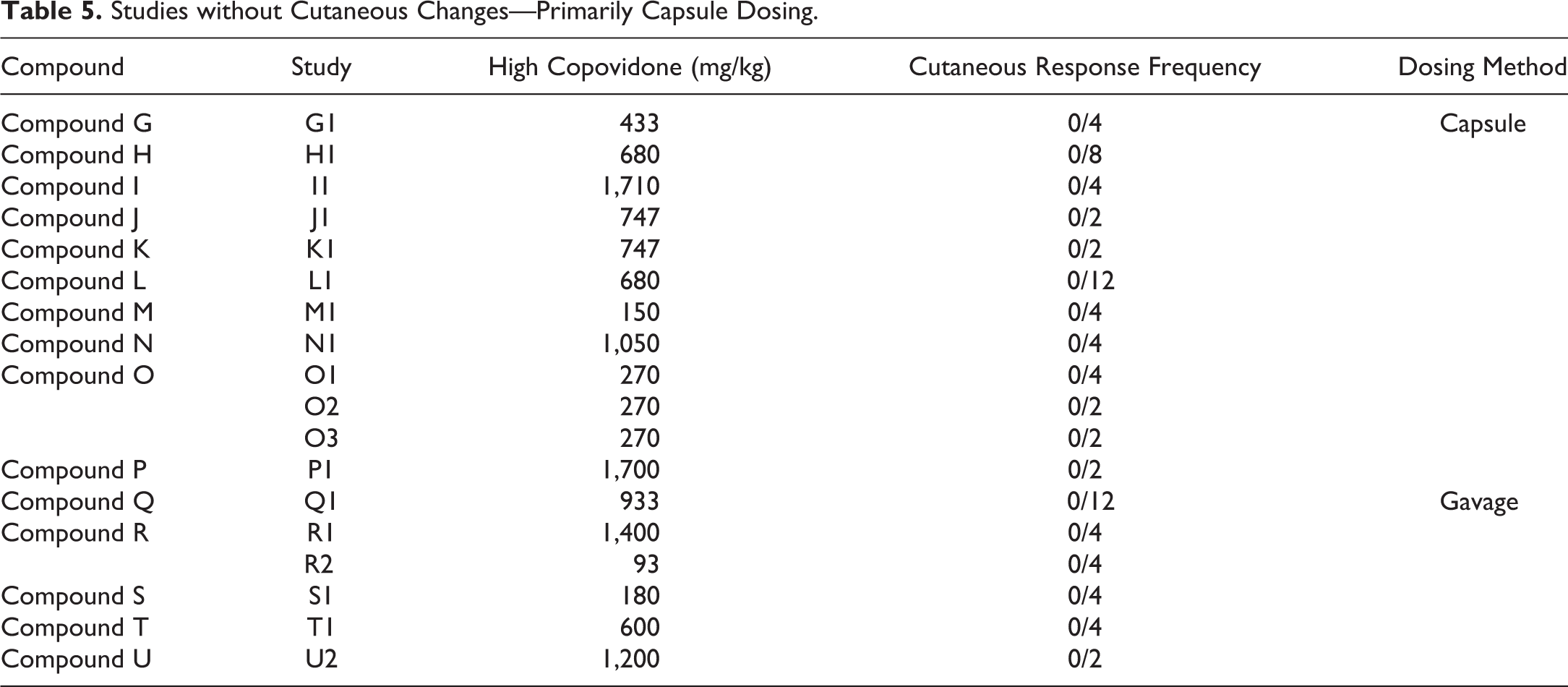
There was a clear dose dependency in respect to the dose of copovidone (Table 4). Furthermore, the method of oral administration of the copovidone-based ASD appears to be of importance in the cutaneous response, as it was frequently encountered in studies with gavage dosing and only rarely with dosing by gelatin capsule (Tables 1 –5), despite typically achieving similar or higher test-item plasma exposures with capsule versus gavage dosing (Table 1). Cutaneous responses with capsule administration have been limited to 2 studies with 1 test item (compound “F” in Table 3); and in those studies, there was considerable postdose emesis.
Considerably Higher Incidence of Cutaneous Responses with Gavage versus Capsule Dosing.
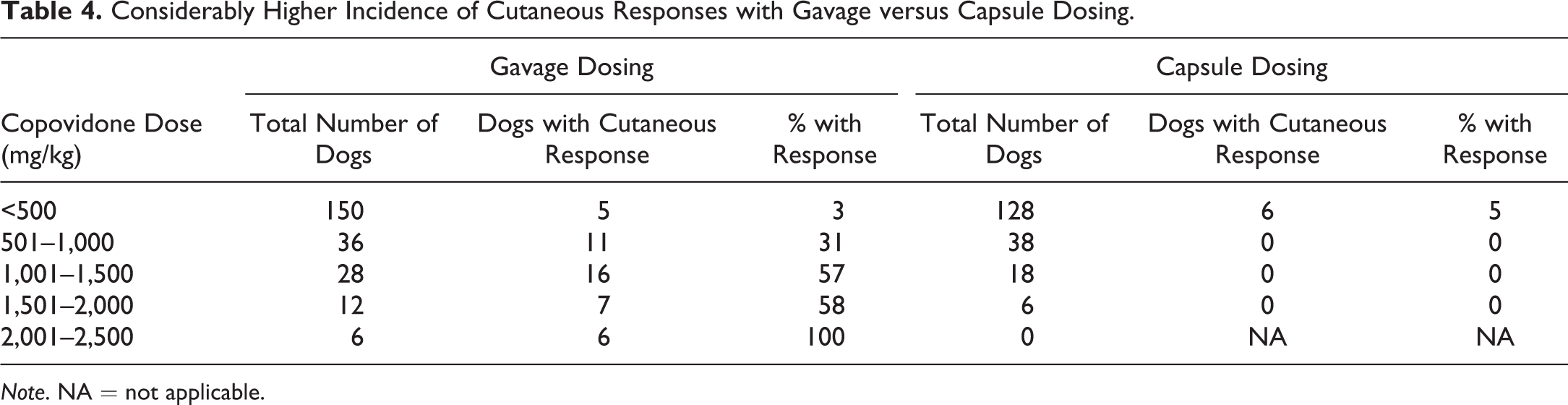
Note. NA = not applicable.
Studies without Cutaneous Changes—Primarily Capsule Dosing.
Copovidone Toxicology—Nonclinical Safety
Copovidone
Copovidone is a random copolymer of equimolar VP and VA (CAS Registry Number 25086-89-9) and is commonly utilized as a pharmaceutical excipient. The discussion of dedicated copovidone toxicology studies will focus on the effect of copovidone in the dog.
No adverse findings were noted in a copovidone 52-week dog study at dosages up to 2,500 mg/kg/day when administered in the diet. Findings were limited to minimal to moderate histiocytosis of the mesenteric lymph node (Mellert et al. 2004).
PVP
PVP is a linear polymer of 1-vinyl-2-pyrrolidone monomers (CAS Registry Number 9003-39-8) and is used as a binder, emulsification stabilizer, film former, hair fixative, and suspending agent nonsurfactant. As per investigations with copovidone, no adverse findings have been noted in chronic (up to 2 years) studies at dosages up to 2,000 mg/kg/day when administered in the diet (Nair 1998).
Copovidone Toxicology—Investigative Considerations
PVP
PVP, a polymer of vinyl-2-pyrrolidone (CAS Registry Number 9003-39-8), administered intravenously to dogs has been shown to result in clinical signs that are similar to those observed in dogs administered copovidone-based ASD formulations as outlined above (swelling, erythema, and pruritus) upon initial exposure. Effects have been noted at PVP doses ranging from 35 to 350 mg/kg (Halpern, Musso, and Neveu 1955; Marshall and Hanna 1957, 1958; Walton, Richardson, and Thompson 1959). PVP-related changes are considered to be the result of an alteration in capillary permeability secondary to histamine release (Halpern, Musso, and Neveu 1955; Jones et al. 1989; Thompson and Walton 1964). The findings in the copovidone-based ASD oral-dosed studies described above are consistent with a histamine response based on the rapid amelioration of clinical signs after administration of diphenhydramine as well as the known cutaneous distribution of mast cells in the dog (Emerson and Cross 1965; Noviana et al. 2004).
Intravenous administration of PVP can result in histamine-related clinical signs affecting tissues other than the skin. Intravenous administration at dosages as low as 20 to 35 mg/kg to conscious dogs has been associated with systemic hypotension and marked elevations of plasma histamine levels compared to baseline values (Jones et al. 1989; Marshall and Hanna 1957). Administration of PVP at 100 mg/kg intravenously to dogs anesthetized with sodium pentobarbital has resulted in increases in plasma histamine increases and profound hypotension (Thompson and Watson 1964). Profound hypotension, and in some instances death, has been associated with intravenous administration of PVP to anesthetized dogs (sodium pentobarbital) at concentrations ranging from 50 to 350 mg/kg (Walton, Richardson, and Thompson 1959).
Correlation between PVP-related Changes and Copovidone-based ASD Formulations
Cutaneous changes in dogs administered PVP via the intravenous route are similar to those noted in toxicology studies with high dosages of copovidone administered via the oral route (Tables 1–3). Other similarities to intravenously administered PVP and the responses in the oral studies with copovidone include the potential for tolerance. Short-term tachyphylaxis may result after administration of PVP (Marshall and Hanna 1957, 1958; Walton, Richardson, and Thompson 1959). The cutaneous responses in dogs in the above-described toxicology studies (Tables 1–3) tended to become less frequent and less severe with continued dosing of the copovidone-based ASD formulations.
In contrast to dogs, intravenous PVP administration in rats and humans has not been associated with histamine release. In fact, PVP has been used clinically in humans as a plasma expander for the treatment of shock (Ravin, Seligman, and Fine 1952). Therefore, the histamine-releasing effects of this chemical constituent of copovidone appear to be unique to the dog. The lack of cutaneous findings in the rodent with copovidone-based ASDs as compared to the dog in the above described oral toxicology studies is in agreement with the apparent species specificity.
Whereas the investigative studies with PVP in the dog have involved intravenous administration, our copovidone-based formulations were administered orally. It is of note that cutaneous findings in the studies utilizing copovidone-based ASDs were frequently encountered with gavage dosing and rarely encountered with capsule dosing (Tables 1 –5), which suggests that some pulmonary aspiration of the formulation may have occurred with the dosing procedure. Aspiration of the copovidone-containing ASDs may have resulted in relatively rapid transport of copovidone across the pulmonary capillary barrier with systemic exposure to PVP. The only capsule studies associated with cutaneous findings were 2 studies with 1 compound, in which there was considerable postdose emesis, with the potential for some aspiration of vomitus. The higher incidence of cutaneous findings in dogs administered the copovidone-based ASD formulations compared to the copovidone placebo control may be related to the high viscosity of the ASD formulations (suspension) compared to the copovidone placebo control (solution). The high viscosity would be expected to result in adherence to the gavage tube and potential for introducing the material into the pharyngeal area with potential for aspiration.
Cutaneous findings have not been noted in safety pharmacology studies with intravenous administration of test item (non-ASD) devoid of copovidone, despite achieving C max values similar or higher than those achieved in oral toxicology studies with the copovidone-based ASD formulations. It is evident that the achieved exposures in the intravenous study are typically similar to or higher than those associated with cutaneous changes in the oral ASD studies.
Correlation of Canine and Human Cutaneous Responses
At one time, PVP was utilized as a plasma expander in humans (Ravin, Seligman, and Fine 1952) and was not associated with adverse systemic effects. Intravenous administration of the PVP-based plasma expander was well tolerated in rats (Ravin, Seligman, and Fine 1952), but in dogs, it was associated with a reddening of the skin, hypotension, and increased plasma histamine levels (Walton, Richardson, and Thompson 1959).
Summary and Conclusion
A cutaneous response (swelling and/or erythema of the skin) has been noted in dogs receiving an oral copovidone-based ASD with 6 different compounds that are structurally unrelated and span several different therapeutic indications. In evaluating each of the 6 test-item ASDs separately, the following conclusions about the cutaneous reaction can be drawn: (1) they are dose dependent with respect to total copovidone administered; (2) between studies, they are typically not exposure dependent with respect to the test item; (3) they are not related to therapeutic indication or structure; (4) they are observed more frequently with gavage versus capsule dosing (and only studies with notable postdose emesis in the capsule dosing events); (5) they have not been present in the dog administered non-ASD formulations involving the same test items, despite achieving similar or higher plasma exposure to the test items; and (6) they tend to become less frequent and less severe with continued dosing.
Review of the literature indicates that PVP has been associated with a histamine-related effect unique to the dog, which results in similar cutaneous responses and/or systemic hypotension. The cutaneous changes noted in the above-described dog toxicology studies with copovidone-based ASD formulations are considered to be secondary to copovidone administration rather than test item. The timing of the appearance of the cutaneous response (within 15–20 min) is consistent with pulmonary aspiration and/or nasopharyngeal exposure rather than absorption from the gastrointestinal tract. The viscosity of the copovidone-based ASD formulations increased with increasing dose of copovidone and test item, with an increased likelihood for adherence of residual ASD on the tip of the gavage tube. The copovidone-related cutaneous responses noted in the dog are not considered likely to translate into an effect in humans.
Footnotes
Authors’ Contribution
Authors contributed to the conception or design (SM, LB, WB, HL, WR, YY); data acquisition, analysis, or interpretation (SM, LB, WB, HL, WR, YY); drafting the manuscript (SM); and critically revising the manuscript (SM, LB, WB, HL, WR, YY). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
