Abstract
Routine husbandry procedures during animal toxicity studies can result in incidental pathological changes. We report on trauma-induced hepatopathy in newborn Göttingen minipigs. Sixty-four neonatal minipigs were allocated to 13- and 26-week treatment arms. There was a 4-week recovery period for both arms. The animals were divided into 2 treatment groups and a vehicle group and were dosed 3 times daily by direct oral administration using a syringe. During the feeding procedure in the first weeks, the animals had to be handled firmly. After 13 weeks, randomly distributed foci of degeneration/necrosis and focal congestion and/or hemorrhage were found in the livers of several animals from all groups. Reduced incidence and severity were evident in the recovery phase, and the lesions were absent after 26 weeks. These changes were considered as related to the manual handling of the animals. Knowledge of these findings is crucial for interpretation of studies utilizing newborn minipigs.
The use of minipigs in toxicity studies is gaining popularity as a potential substitute to dogs as a nonrodent species in toxicity studies (Manno et al. 2016; Ramot et al. 2016; Ramot et al. 2015; Schaefer et al. 2016; Vezzali et al. 2011). This is based on the better profile of minipigs in terms of similarity to humans, both anatomically and physiologically (Bode et al. 2010; van der Laan et al. 2010), and their decreased size, making them easier to handle (McInnes and McKeag 2016). The most used strain is the Göttingen minipig, which was developed in the University of Göttingen, Germany, by triple crossing of Minnesota minipig (60%), Vietnamese pot-bellied pig (33%), and German Landrace (7%; Bollen and Ellegaard 1997). Therefore, knowledge on possible factors that might affect the interpretation of pathological findings in this strain is crucial. Such factors may include the routine husbandry procedures, such as handling and orogastric gavage (Balcombe, Barnard, and Sandusky 2004). Here, we report on liver lesions that were observed in a toxicity study involving newborn Göttingen minipigs. This injury was attributed to the manual handling of the animals during oral administration with a syringe and was not related to the tested drug.
Thirty-two male and 32 female neonatal Göttingen minipigs were used in this study and were allocated to 13- and 26-week treatment arms. The vehicle group contained 3 male and female minipigs for each sacrifice point, and in addition, 1 animal per sex for the respective recovery phases. There were 2 treatment groups (low and high dose), which included 4 male and female minipigs for each sacrifice point and 2 male and female minipigs for the respective recovery periods.
Starting from the third day of life, the animals were dosed 3 times daily by direct oral administration with a syringe. During the first weeks of administration, dosing of the animals was not voluntary, and the caretaker had to pick the animals up and hold them firmly in the middle of the body part, close to the animal technician’s body (Figure 1). When the animals resisted, a second caretaker assisted by opening the animal’s mouth and administering the syringe deep enough to ensure administration. The dosing time could take up to 2 hr during the first weeks. The animals went through daily training, and after approximately 3 weeks were voluntarily dosed without restraint. During the study period, animals were observed for clinical signs of toxicity, body weight was measured, and blood samples were collected for hematology and clinical chemistry on days 1, 30, 60, and sacrifice for the 13-week study, and days 1, 30, 60, 90, and 140 for the 26-week study. Macroscopic and microscopic evaluation of the gastrointestinal system, including the esophagus, stomach, duodenum, jejunum, ileum, cecum, colon, liver, and gallbladder, was performed.
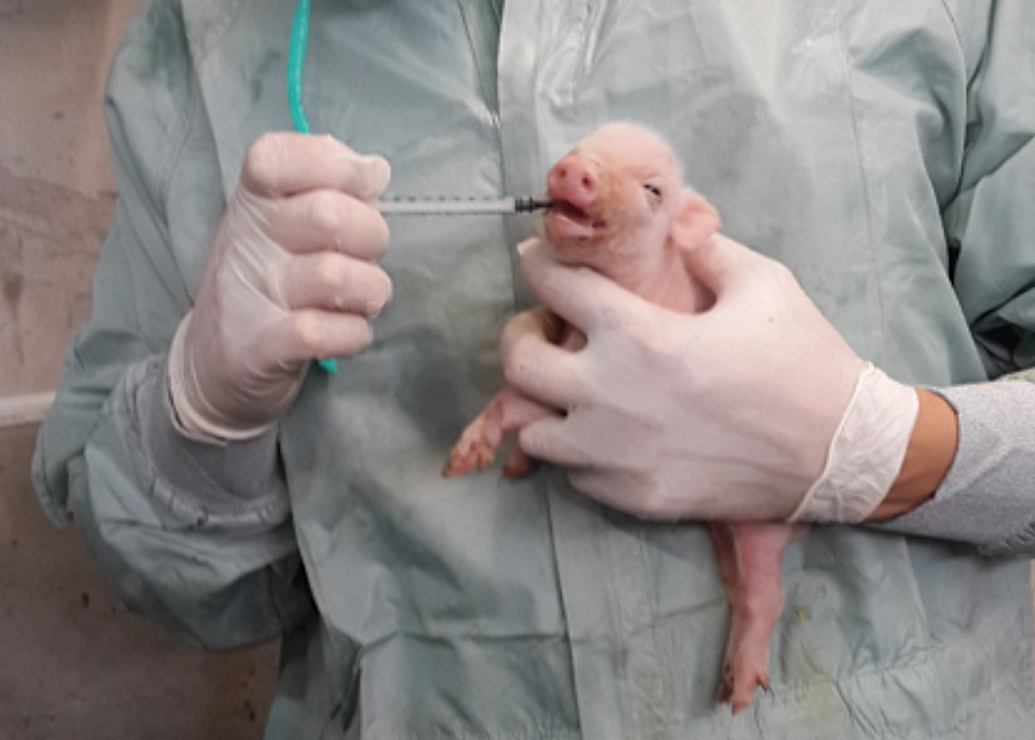
Method of animal holding during the initial phase of the study. The animal was held in its midsection close to the handler’s body. When the animal resisted, a second handler would assist by opening the mouth and administering the syringe deep enough to ensure administration.
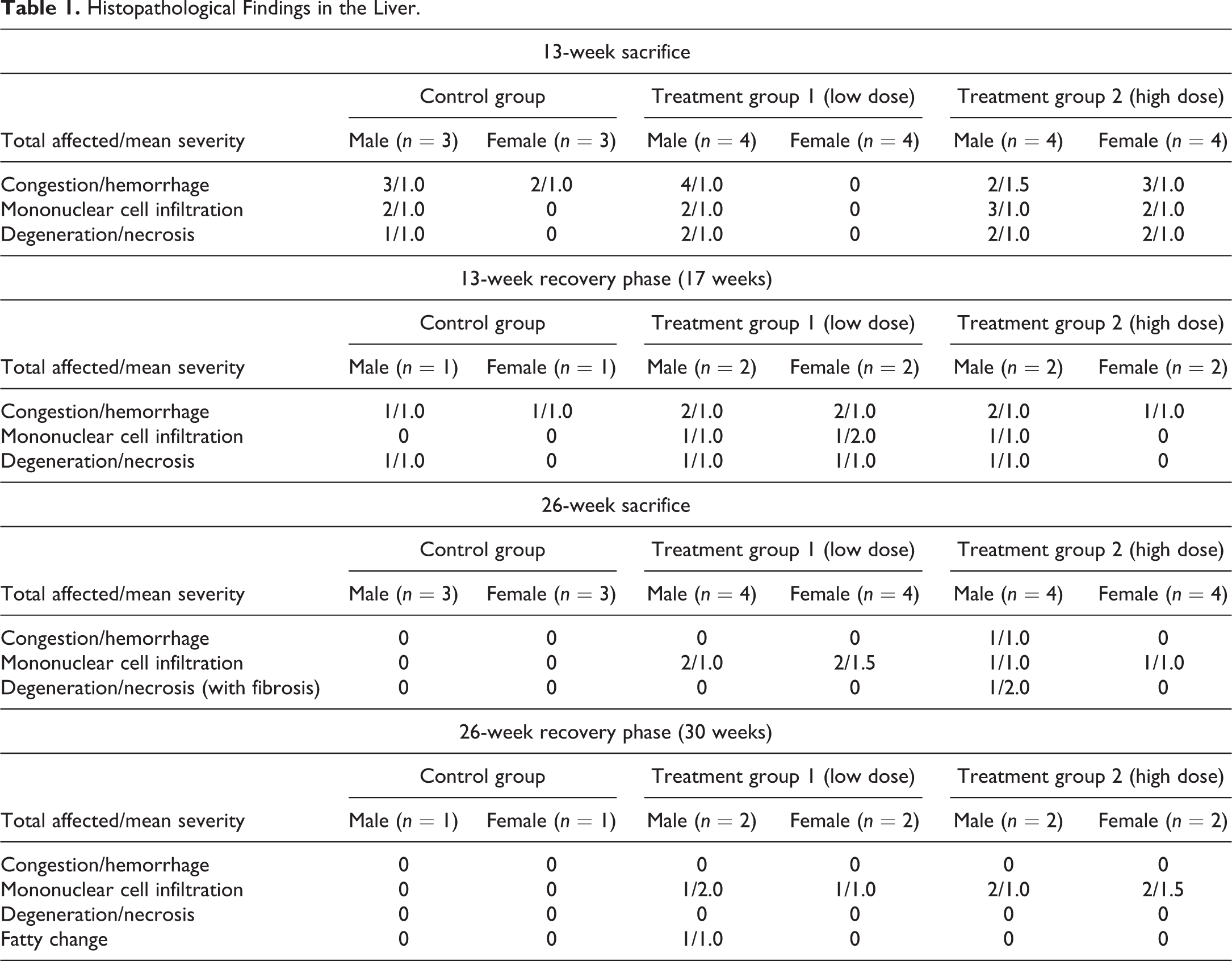
No mortality was noted throughout the study period, and no major clinical signs were observed in all groups. Hematology and chemical chemistry analyses did not reveal any significant changes between groups or when compared to reference values (Supplemental Table 1). The gross lesions appeared to be nonspecific, and most lesions were devoid of remarkable histological correlation. Several discolored foci in the liver, corresponding to petechial lesions, were noted in several control and treated animals from the 13-week sacrifice point, without any dose relation. These changes correlated with the histology findings and are considered as trauma related. Histopathology evaluation of the animals from the 13-week study revealed foci of degeneration/necrosis in the liver in several animals from all groups (control and test item treated). These lesions were limited to single lobules or sharply demarcated small groups (mainly subscapular) of a few liver lobules. They were characterized by single-cell apoptosis/necrosis, inflammatory cell infiltration, hepatocellular vacuolation (with features of hydropic degeneration and/or focal fatty change), as well as focal congestion and/or hemorrhage (Figures 2 –6; Table 1). These lesions were also found in the recovery phase animals (17 weeks). Nevertheless, none of these lesions could be found in the 26-week study animals nor in the recovery phase, except for slight focal hepatocellular degeneration in 1 animal from the 26-week treatment group. The histopathological picture had more chronic characteristics, revealing multifocal minimal fibrosis (Figures 7 and 8; Table 1). Since the incidence and severity of these lesions were equally and randomly distributed between the groups (both control and treatment groups), these lesions were not regarded as related to treatment.
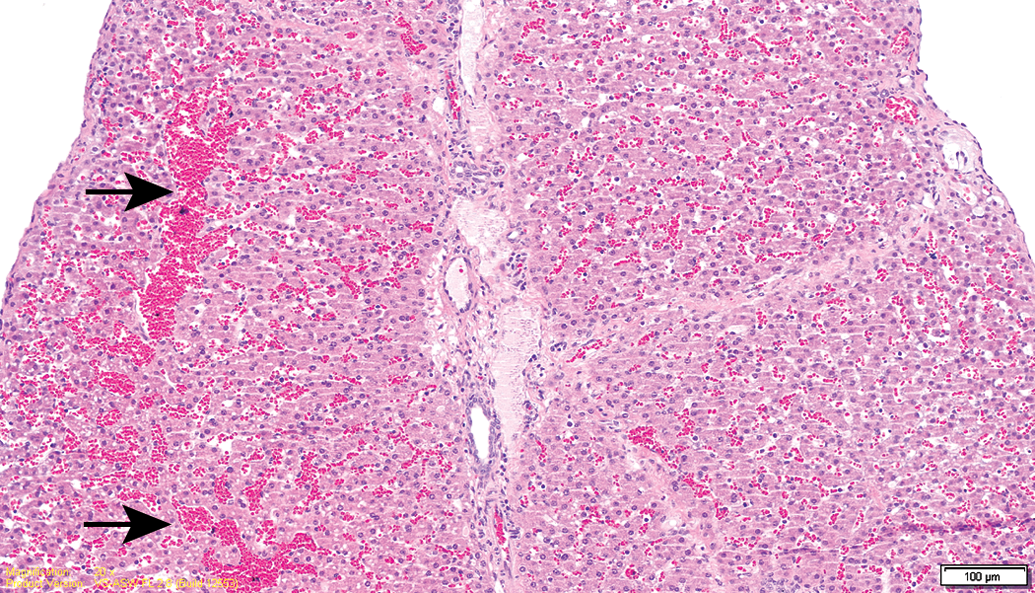
Test item–treated animal (13-week interim sacrifice). Focal subcapsular congestion in the liver (arrows).
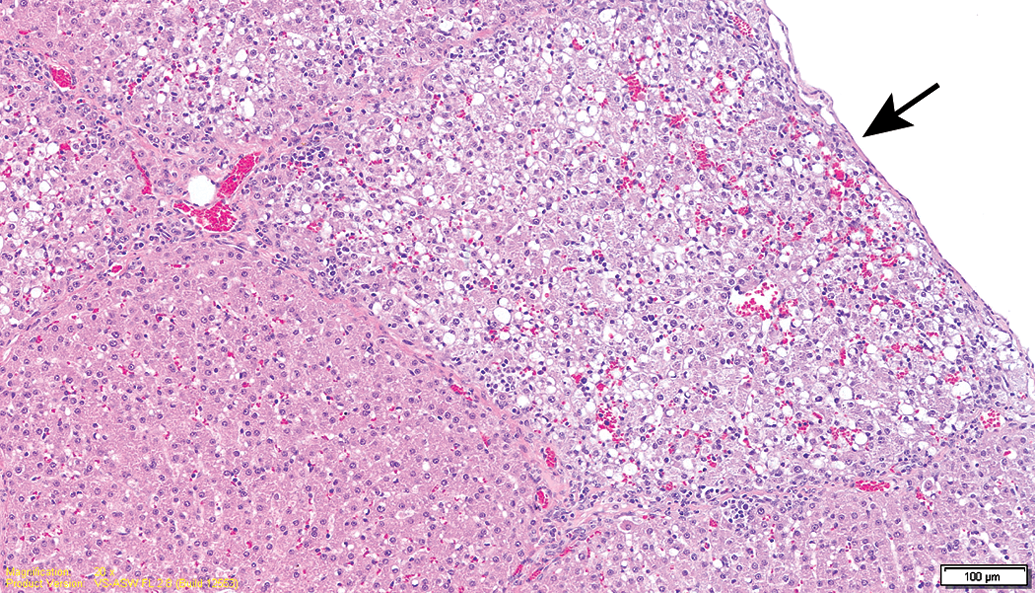
Control animal (13-week interim sacrifice). Focal subcapsular hepatocellular degeneration (arrow).
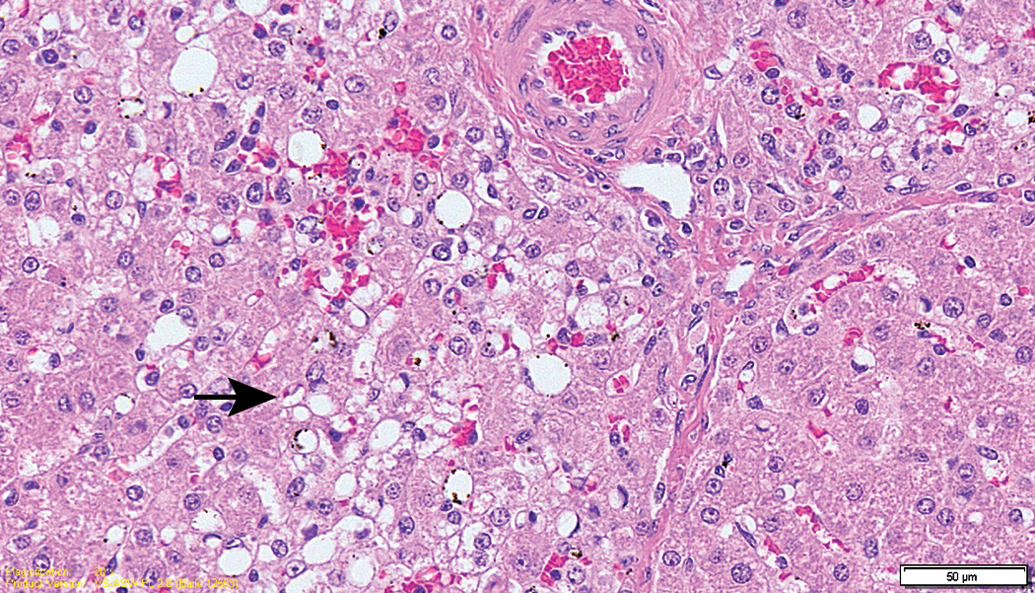
Control animal (13-week interim sacrifice). Higher magnification of Figure 3. Focus of hepatocellular degeneration characterized by congestion/hemorrhage, hepatocellular vacuolation (arrow), mononuclear cell infiltration, and single necrotic hepatocytes.
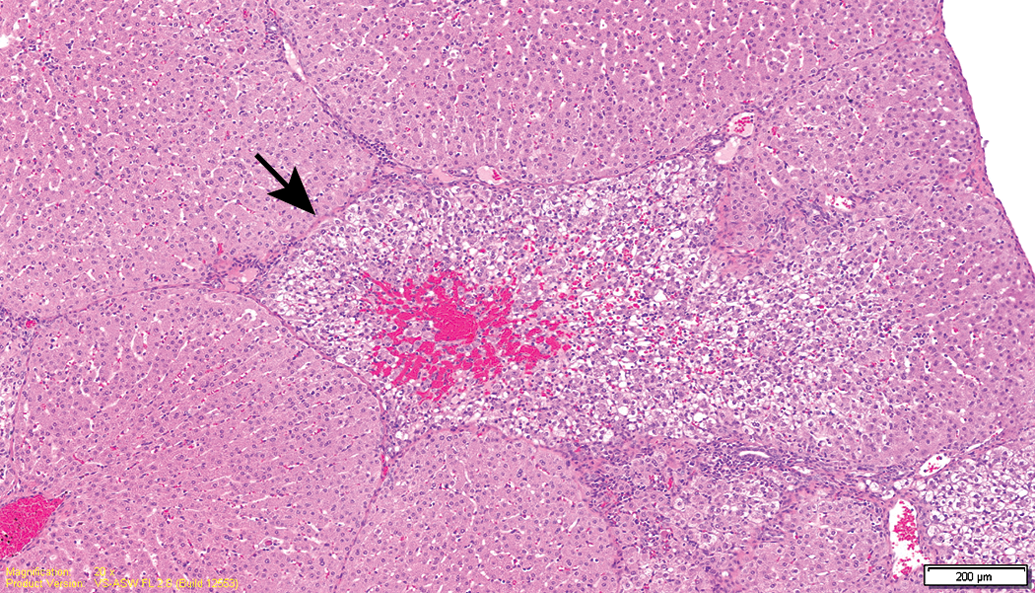
Test item–treated animal (13-week interim sacrifice). Overview of an area affected by degeneration (arrow).
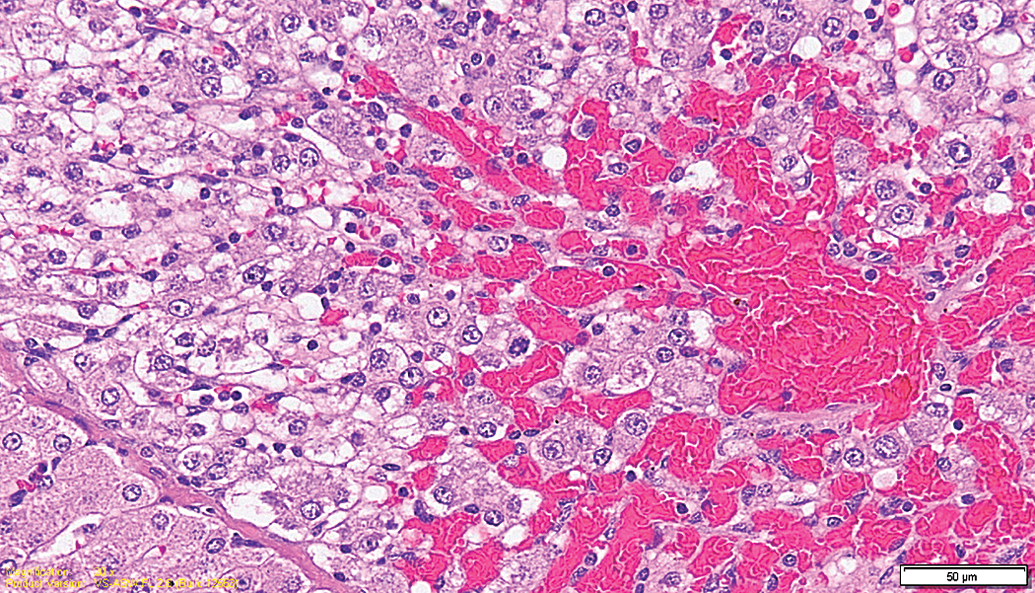
Test item–treated animal (13-week interim sacrifice). Higher magnification of Figure 5. Focal degeneration characterized by congestion/hemorrhage, hepatocellular vacuolation, and infiltration by mononuclear cells.
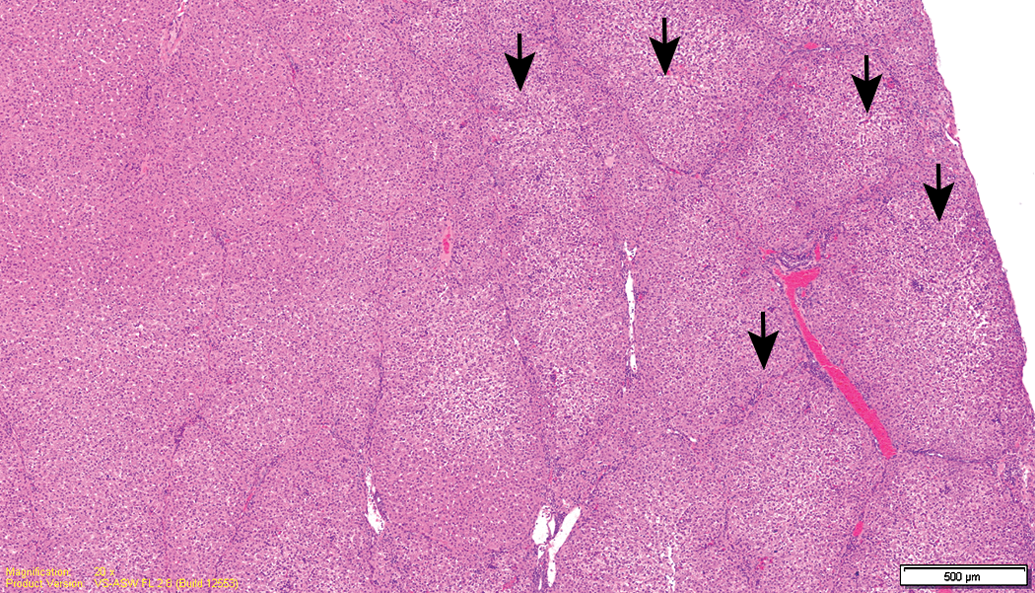
Test item–treated animal (26-week sacrifice). Subcapsular foci of hepatocellular degeneration (arrows).
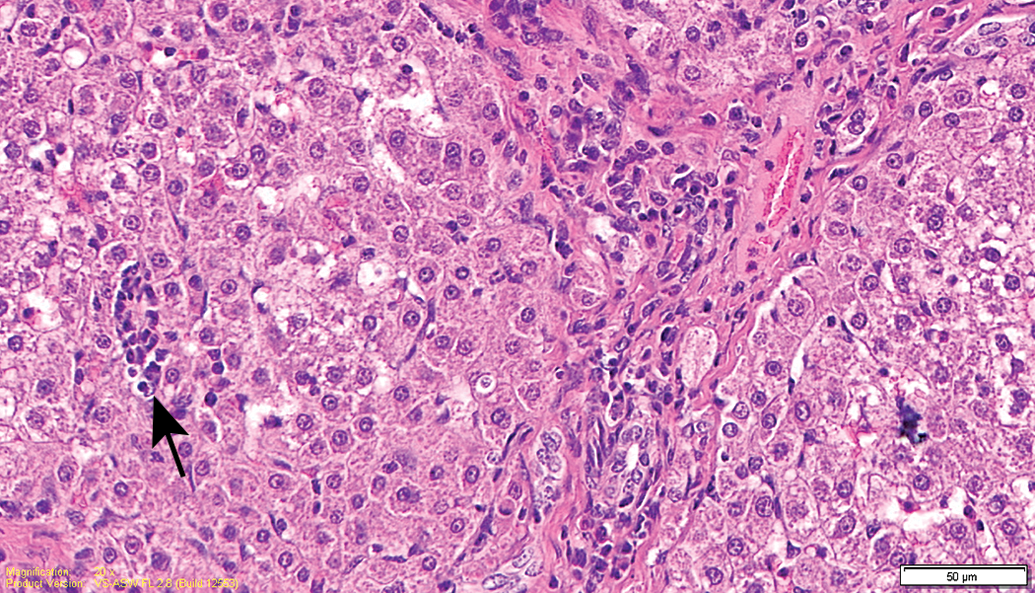
Test item–treated animal (26-week sacrifice). Higher magnification of Figure 7. Minimal mononuclear cell infiltration (arrow) and focal fibrosis.
Histopathological Findings in the Liver.
The routine handling of animals during toxicity studies may often lead to animal stress, which may affect the interpretation of the clinical and histopathological findings (Balcombe, Barnard, and Sandusky 2004). This is especially true when the animals that are tested are newborn minipigs, which are attached to their mothers and separation leads to much agitation early in life. In the current study, animals had to be orally fed using a syringe, necessitating the exertion of manual restraint on the animals. Since newborn minipigs do not have well-developed connective tissue framework to protect their internal organs, similar to children (Deluca et al. 2007), these might be affected by trauma, either direct or secondary to increased intra-abdominal pressure (Bloomfield et al. 1997). Indeed, the liver of newborn minipigs seems to be especially vulnerable to trauma, and liver rupture is a very common cause for mortality in preweaning pigs (Bille, Nielsen, and Svendsen 1974; Glastonbury 1977).
Incidental liver damage due to handling and dosing procedures has been reported in the past in several toxicity studies. This has been observed mainly in dermal toxicity studies, where the torso of the rodent is wrapped to guarantee close contact with the skin and to avoid ingestion and/or self-mutilation of the animal, leading to the so-called wrapping necrosis (Chandra et al. 2015). This is characterized histopathologically as hepatic centrilobular degeneration and necrosis leading to fibrosis (Nyska et al. 1992; Parker and Gibson 1995; Ramot et al. 2012; Nelson et al. 2016).
In the present case, most of the lesions in the liver were distributed in the subcapsular area, were small in size, and were combined with congestion/hemorrhage. These lesions, which were noted in animals from all groups (including control), were not seen in the 26-week study period, after the animals were already accustomed to the feeding procedure, and were not restrained. Taken together, we believe that the liver lesions observed in the current study are the result of incidental trauma. Of note is the observation of focal fatty changes in the livers of the minipigs, which can be seen commonly as a response to liver injury, sometimes referred to as “tension lipoidosis” (Sato et al. 2012).
While the minipig is emerging as a new animal model for toxicity studies, and data on background lesions are accumulating, not much is known on incidental lesions in newborn minipigs, which are used less frequently in toxicity studies. Therefore, this report is an important contribution for the toxicology community, and based on this report, it can be concluded that if the manual handling of newborn minipigs may cause intra-abdominal trauma, the presence of foci of degeneration/necrosis in the liver, associated with hemorrhage, may be considered as a trauma-related consequence. These findings, therefore, should not be interpreted as drug related.
Footnotes
Authors’ Contribution
Authors contributed to conception or design (AN); data acquisition, analysis, or interpretation (YR, KW, FL, JC, LG, RS, AN); drafting the manuscript (YR, KW, AN); and critically revising the manuscript (YR, KW, FL, JC, LG, RS, AN). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by NUTRINIA, Ramat Gan, Israel.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
