Abstract
Intranasal administration of drugs is gaining popularity in medicine, and several animal models have been used to test the safety and efficacy of this delivery route. Nevertheless, the nasal anatomy of animals is different from humans, which can lead to pathological changes that stem from the delivery device and not the drug itself. Here, we report on nasal inflammation and ulceration in rabbits, secondary to the repeated trauma caused by the intranasal device. Similar changes were noted in the animals treated with the vehicle and with the tested drug, and therefore, these changes were not attributed to the drug itself. In some animals, superficial ulcer and stromal inflammation were noted in the eyes, secondary to nasal duct obstruction from the nasal inflammation. These observations emphasize the importance of proper interpretation of histopathological changes, attributed to trauma-induced pathological changes related to the handling of the animal and not to the tested product, which is the drug itself and the device that is optimized for clinical (human) use.
In recent years, there has been growing interest in intranasal drug administration, which can be utilized as a noninvasive route of delivery for local, systemic, or central nervous system distribution of drugs. 1 To study the safety and efficacy of intranasally administered drugs, animal models have been used. 1 Mice and rats are frequently used for drug absorption studies, 2 –4 while other animals, such as dogs and sheep, 5,6 are used mainly for pharmacokinetic evaluations. Rabbits, being a commonly used species for toxicological studies in general, have also been used for intranasal drug administration studies. 7,8 Interpretation of safety and toxicity studies in animals can be challenging because the anatomic structures and physiological properties of the tested animals are very different from their human counterparts. 9 Such anatomical differences can also lead to trauma-induced pathological changes related to the handling of the animal, and it is important that the toxicologic pathologist will not wrongly attribute such changes to the test drug or combination product. 10 –12
Here, we report on nasal inflammation and ulceration in rabbits, secondary to the mechanical damage induced by the repeated insertion and removal of the intranasal device, which were not related to the tested drug.
Forty-eight male and female New Zealand White rabbits were used in this study and were allocated to 4- and 8-week test drug and control arms. The vehicle groups contained 3 male and female rabbits for each sacrifice point, and the drug group contained 4 male and female rabbits for each sacrifice point. In addition, 2 animals per gender were allocated for respective recovery phases. The animals in the 4-week treatment arms were treated once daily, and the animals in the 8-week treatment arms were treated twice daily. There was one group of naive animals, which included 2 animals per gender, which did not receive any treatment (vehicle or the test drug) and were not exposed to the intranasal delivery system. This study was performed in compliance with “The Israel Animal Welfare Act” and following “The Israel Board for Animal Experiments” Ethics Committee approval #IL-19-10-414.
In all animals from the vehicle and test drug groups treated for 4 weeks, comparable histopathological changes were noted in part or all sections of the nose. The changes consisted of focal mild-to-moderate ulcer (usually unilateral); focal mild-to-moderate acute inflammation in the mucosa (mostly associated with the ulcer); and presence of acute exudate in the lumen of the nasal cavity. In a single case, the acute inflammation was associated with presence of bacterial colonies.
Comparable histopathological changes were also noted in part or all sections of the nose in the animals treated for 8 weeks (Figures 1 –4). The changes consisted of focal-to-multifocal mild-to-moderate ulcer, focal mild-to-moderate chronic inflammation in the mucosa and submucosa, septal abscess formation, and presence of free blood or acute exudate in the lumen of the nasal cavity. The presence of submucosal inflammation and abscess formation caused deformation of the turbinates (Figures 1, 2, and 4). Abnormal histopathological findings were also noted in the eyes of some of the animals either treated with the vehicle and/or with the test drug for 8 weeks. These changes consisted of superficial ulcer, stromal inflammation, and edema and/or congestion (Figure 5). The changes were mostly of minimal severity. Clinical ophthalmological abnormalities were noted in these animals and included lacrimation, redness of eyes, conjunctivitis, and discharge. No pathological changes were seen in the animals from the naive group.
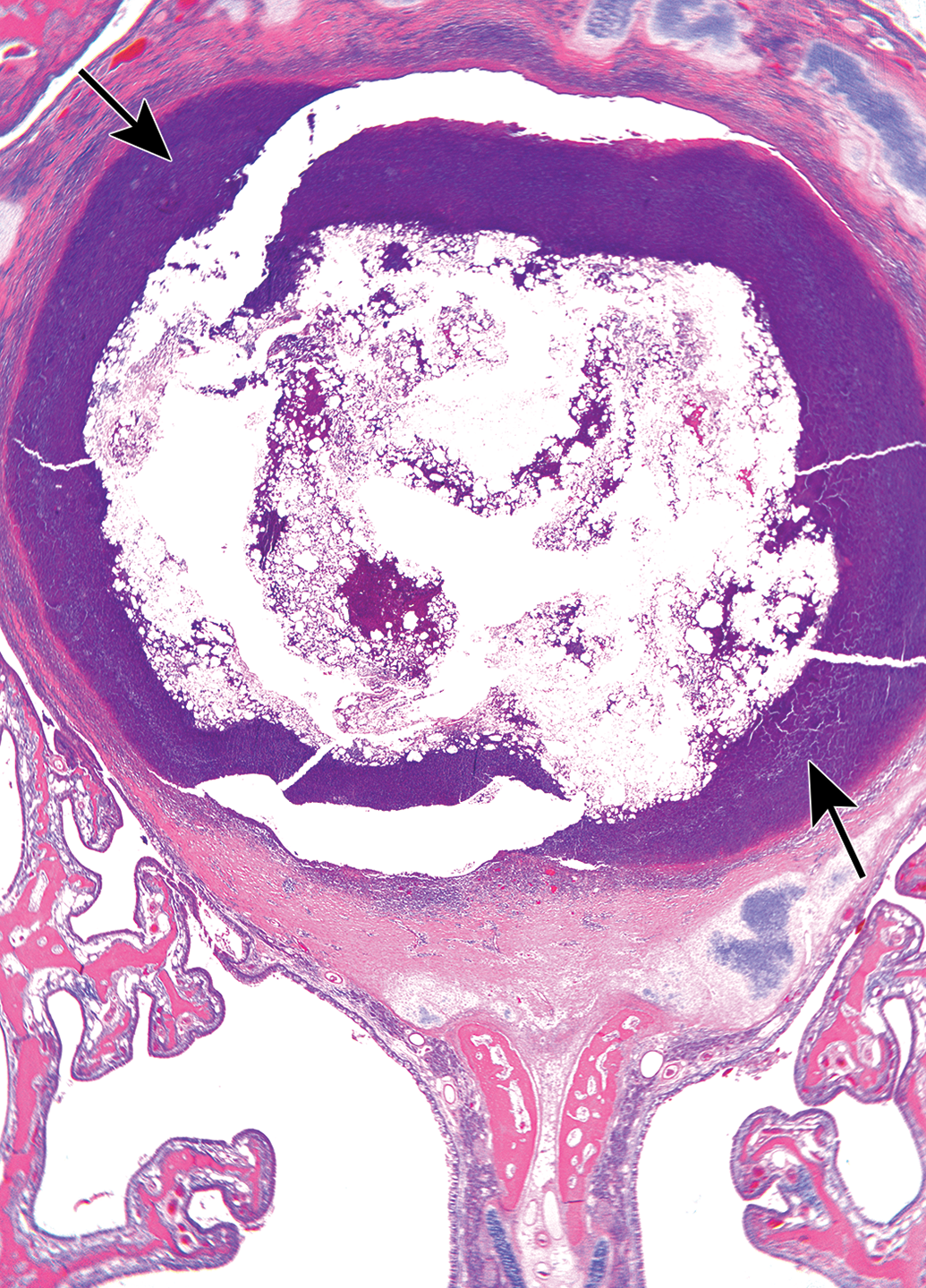
Photomicrograph of a level 2 section of the nose, taken from a female rabbit treated with the drug for 8 weeks. Arrows indicate the presence of a large abscess located in the upper part of the septum, associated with lysis of the cartilaginous septum and compression of the adjacent maxilloturbinates. Hematoxylin and eosin Original objective ×20.
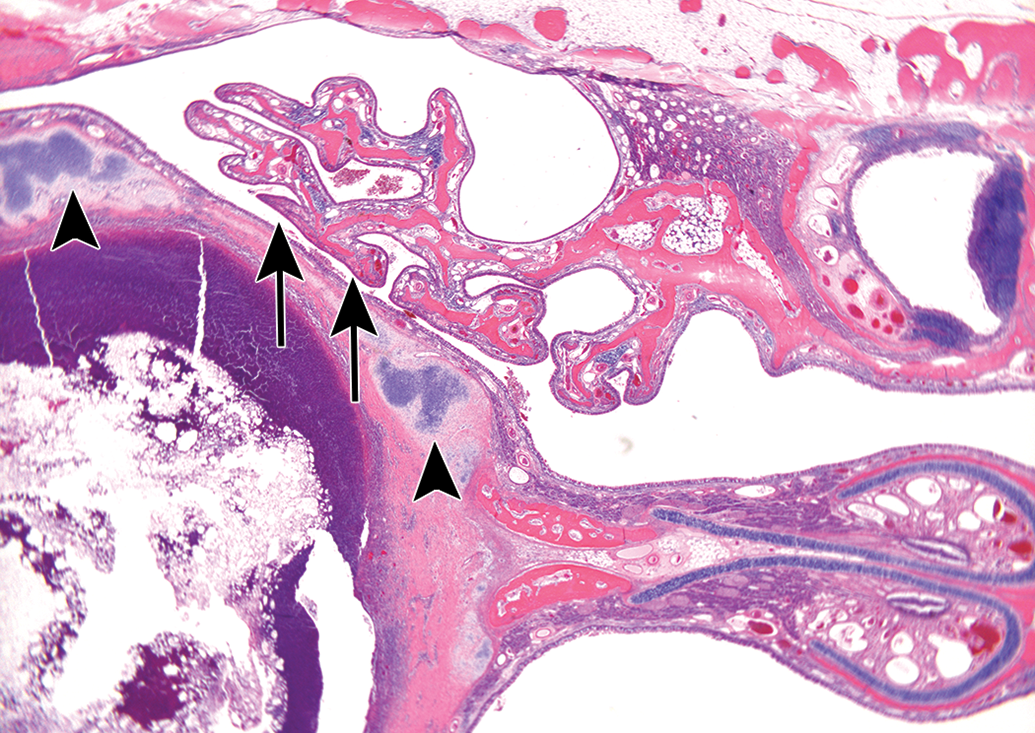
Photomicrograph of a level 2 section of the nose, taken from a female rabbit treated with the tested drug for 8 weeks. Same case as shown in Figure 1. Arrowheads indicate the displaced and lysed cartilaginous septum. Arrows indicate the compressed maxilloturbinates. Hematoxylin and eosin; Original Objective ×20.
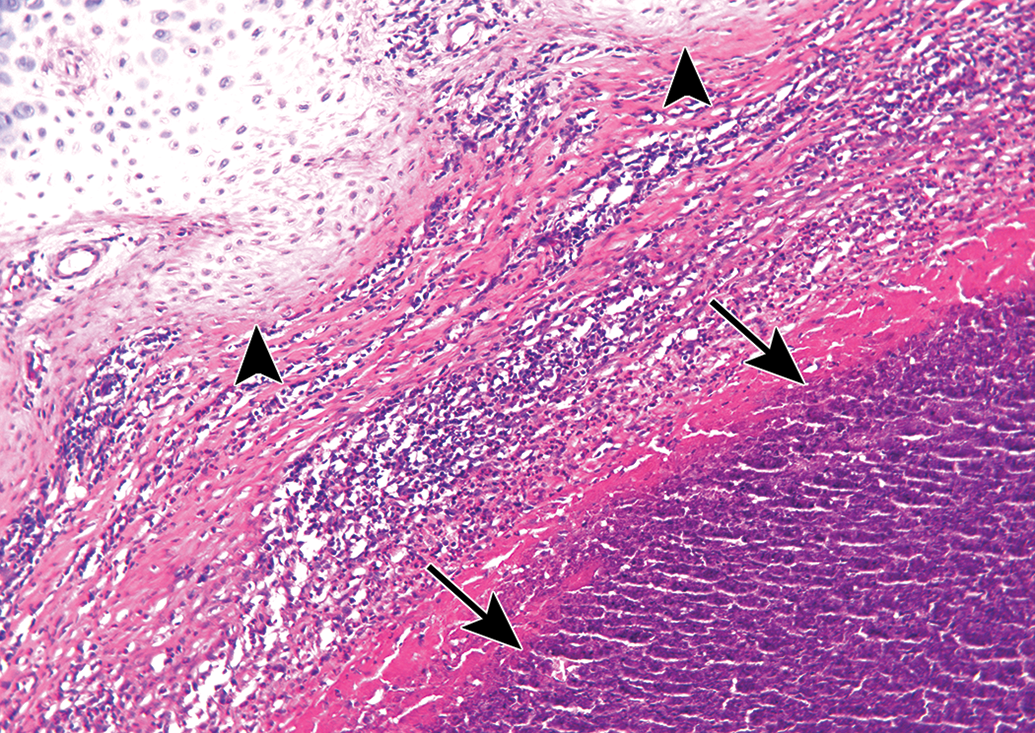
Photomicrograph of a level 2 section of the nose, taken from a female rabbit treated with the tested drug for 8 weeks. Higher magnification of the case shown in Figures 1 and 2. Arrowheads indicate the displaced and lysed cartilaginous septum. Arrows indicate the abscess located in the upper part of the septum. Hematoxylin and eosin; Original objective ×20.
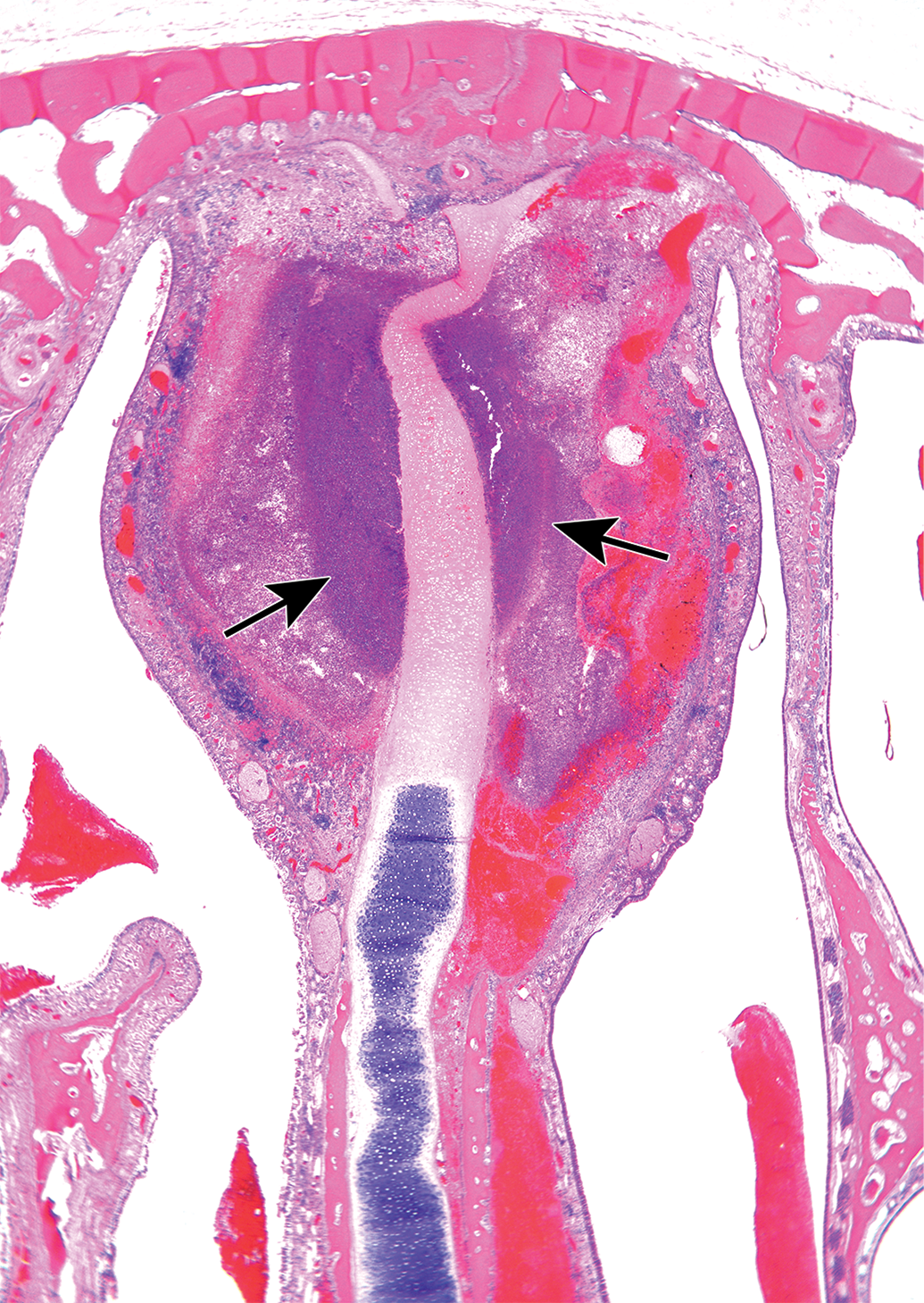
Photomicrograph of a level 3 section of the nose, taken from a female rabbit treated with the vehicle for 8 weeks. Arrows indicate the presence of a large abscess, associated with submucosal hemorrhage, located in the upper part of the septum, associated with lysis of the cartilaginous septum and compression of the adjacent naso- and maxilloturbinates. Hematoxylin and eosin; Original Objective ×20.
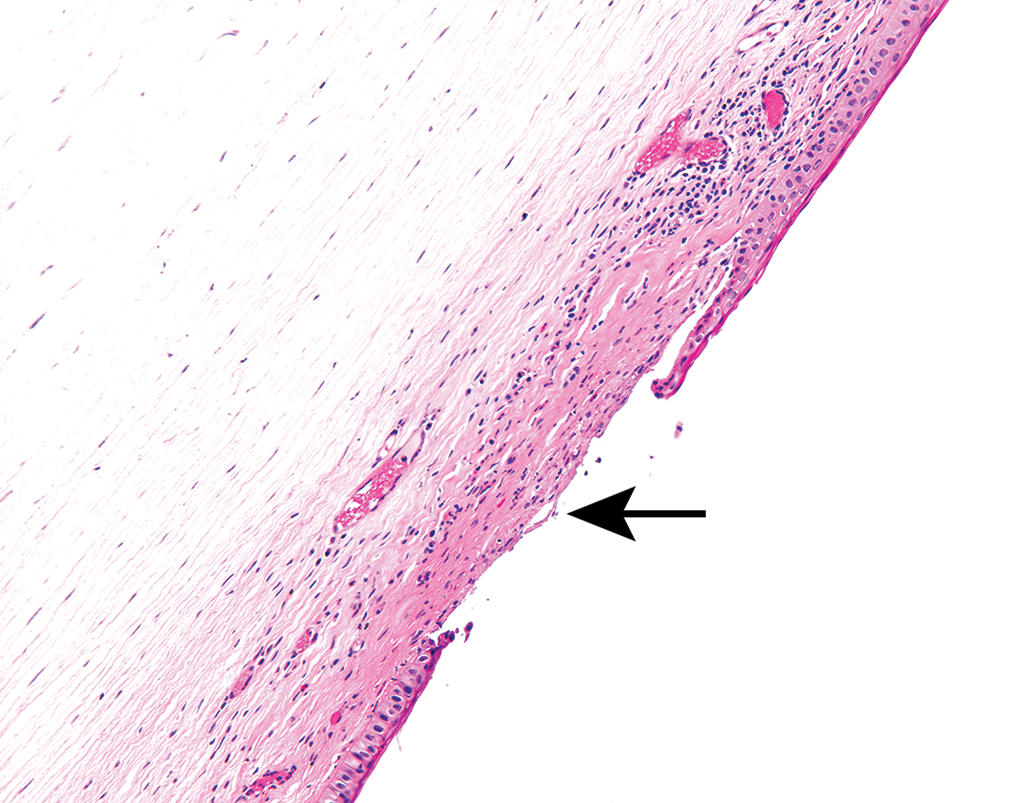
Photomicrograph of the eye from the same animal shown in Figure 1. Arrow indicates the presence of focal corneal epithelial ulceration associated with stromal inflammation. Hematoxylin and eosin; Original Objective ×20.
Records of nose bleeding were performed after every dosing, and occasional bleeding was observed in animals from all the study groups (except the naive group).
The rabbit’s nose is anatomically more complex than mice and rats and different from humans. 13 It is characterized by multiple branching of the concha to provide better protection of the lower respiratory tract. 1,13 –15 Detailed information on the anatomy of the nasal cavity of the rabbit can be found in dedicated reviews by Pereira et al 14 and Harkema et al, 13 which also provide comparison between the rabbit and other commonly used laboratory animals. This complex structure, however, can be a hurdle for the performance of intranasal administration safety studies in rabbits. The delivery device that was used in the current study, which was originally designed for use in humans, was modified to be more suitable for the rabbit’s nose. The modifications were made to the nosepiece, which is the part that is inserted into the nasal cavity during the activation of the device to deliver the drug. The nosepiece was narrowed in order to enable its insertion into the animal’s narrow nose. However, this adaptation was not optimal, partially because a narrower part has a much higher potential to scratch the tissue, and because the modification was done in a less controlled manner than the highly controlled production of human nosepiece parts. Therefore, the adapted nosepiece was less smooth than the one intended for use in humans. In addition, drug administration was performed on unanesthetized animals that were handheld and not kept in a restrainer to reduce stress. Therefore, the animal’s head movements led occasionally to scratches and other traumatic damage that are not relevant to their use in humans, for whom a much wider and rounded nosepieces are used. Thus, despite the optimizations that were made, the device’s use still resulted in repeated trauma, leading to the inflammatory reaction observed in the treated rabbits. This is also supported by the fact that longer use of the nasal device, with more frequent administration, led to more severe histopathological findings.
Interestingly, in the eyes of some of the animals either treated with the vehicle and/or with the test drug for 8 weeks, the inflammatory reaction in the nose was not limited to the nose of the rabbits but also extended to the eyes. Inflammation of the nasal structures can result in nasolacrimal duct obstruction and inflammation, especially in rabbits, which are more prone to nasolacrimal duct disorders. 16 Obstruction and stasis of the lacrimal duct can result in secondary infection of the duct that can eventually spread to the ocular surface, and in addition can cause overflow of tears to the ocular surface, which contributes to the occurrence of infection. 17,18
The nasal inflammation was seen in both the vehicle- and test drug-treated animals, and therefore, these changes were not attributed to the test drug itself or even the drug–device combination product, but rather to the repeated traumatic procedure. The nature of the changes in the nose, being focal, unilateral, hemorrhagic, ulcerative, and localized to the mucosa and underlying submucosa, suggests a mechanical puncture/scratch trauma, as was previously shown by our group in the case of subcutaneous injection of saline. 10 Knowledge of these changes that are induced by the traumatic application of the nasal device is important for the correct interpretation of future safety studies. This is especially true for studies performed in rabbits, where the nasal anatomy is more complex making rabbits more susceptible to nasolacrimal obstruction and infection, which may result in ocular infection. 16 –18 This case emphasizes the need to properly explore the cause of each of the findings and not to exclude the possibility of safe use in human because of tissue damage seen in rabbits.
The drug–device combination product reported in this communication was already used in clinical studies (phase 1) and no adverse events or safety issues were seen following its use.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
