Abstract
Swine, especially the miniature swine or minipigs, are increasingly being used in preclinical safety assessment of small molecules, biopharmaceutical agents, and medical devices as an alternate nonrodent species. Although swine have been used extensively in biomedical research, there is a paucity of information in the current literature detailing the incidence of background lesions and differences in incidence between commonly used breeds. This article is a collaborative effort between multiple organizations to define and document lesions found in the common breeds of minipigs used for toxicological risk assessment in North America (NA) and the European Union (EU). We retrospectively assessed 10 years of historical control data from several institutions located in NA and EU, covering the period of 2004–2015. Here we report the background lesions with consideration of breed and geographical location. To our knowledge, this is the first report documenting spontaneous background lesions in commonly used breeds of swine in both NA and EU. This report serves as a resource to pathologists and will aid in interpretation of findings and differentiation of background from test article–related changes.
Introduction
Miniature pigs, also referred to as minipigs, are increasingly being used in research settings. There are several breeds of minipigs being used in North America (NA) and the European Union (EU), the most common of them being Göttingen, Hanford, Sinclair, and Yucatan. Among these breeds, the Göttingen has emerged as one of the most frequently used in both NA and EU. From the available literature and based on our experience with stocks of other laboratory animals including mice, genetic drift occurs and differences develop, which may lead to background lesions being different in populations over time. For this reason, we reported both NA and EU populations of Göttingen minipigs separately.
While pigs are increasingly being used in nonclinical studies, there is a paucity of literature on the background findings of minipigs, and mention of differences between the breeds is also lacking. This study was undertaken to determine if there are breed or population (geographic location)-based differences in the incidence and nature of commonly occurring background pathology. There have been a few reports out of the EU discussing background changes (Dincer and Skydsgaard 2012; Jeppesen and Skydsgaard 2015; Svendsen et al. 1998); however, these studies were undertaken primarily at one geographical site. Presented here is a multisite study that includes data from 1,390 Göttingen, 118 Hanford, and 88 Yucatan subjects.
Materials and Methods
In NA, 6 contract research organizations (CROs) and 2 vendors were contacted for input, in addition to 3 CROs in the EU, for comparison of the populations. A spreadsheet based on the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND), rodent nomenclature outline was formatted for minipig data entry that separated males and females, age-groups of pigs (<2 months, <2 months but <6 months, and > 6 months), and severity of lesion. The INHAND rodent nomenclature outline was used for classification of lesions since minipig-specific INHAND nomenclature was not yet available. The ages chosen were based on commonly used ages to differentiate infant, juvenile, and adult. Data were collected separately for Göttingen minipigs in NA and EU, Hanford, and Yucatan breeds. No data were available for the Sinclair breed. Tables were completed with available data from the previous 10 years of studies and submitted for collation. If the organization used a diagnostic term not provided on the original spreadsheet, the specific terminology was added to the spreadsheet and documentation proceeded with the number of animals in each group/subgroup that displayed a particular diagnosis, the severity of the diagnosis, along with the total number of animals in which that particular organ was examined.
Since there is currently no standardized nomenclature available for the minipig, each organization has its own protocol for describing lesions, which resulted in the nomenclature used to describe lesions being inconsistent between facilities. To overcome this, different terminologies representing the same pathological change were combined for clarity as shown subsequently; acute inflammation includes neutrophilic inflammation; subacute/chronic inflammation includes subacute, subacute/chronic, chronic, mixed, mononuclear, lymphoid, lymphocytic, and lymphohistiocytic; granuloma and granulomatous inflammation were combined; inflammation, not otherwise specified (NOS), included all remaining or unspecified inflammation including “-itis”; and infiltrate, inflammatory cell included infiltrates of mononuclear cells, lymphoid, lymphocytes, mixed cell, and inflammatory cell foci.
Abscess and chronic–active inflammation were kept separate, as they are distinct processes. Eosinophilic cellular infiltrates were also kept separate. In the liver, all diagnoses of macrophages, pigmented were combined with pigment, increased (Kupffer cell).
Animals
There were no primary animal subjects of this study. All data were collected retrospectively as part of other experiments, which were approved by respective IACUCs. Göttingen minipigs herein described as EU cohort indicate animals originating from the Ellegaard breeding facility in Dalmose, DK, housed in UK facilities. Animals in the NA cohort were obtained from the Marshall breeding facility in North Rose, NY, and housed in either United States (US) or Canadian facilities. Hanford and Yucatan minipigs originated from the Sinclair Bio Resources breeding facility in Missouri and were housed in the United States
All background changes reported were from control, placebo animals, or were noncompound related in nonclinical toxicity studies. Routes of administration of test article included oral gavage, topical application, and parenteral routes (dermal, subcutaneous, intravenous, intramuscular, and others).
Animal care and housing
Housing differences between the breeders exist. For Göttingen minipigs in NA and EU, young pigs, breeding or early gestation sows, and retired breeder sows were housed in groups of 4 animals or more. Hanford and Yucatan breeds of miniature swine were individually housed.
As numerous sites were surveyed for this report, there are variations in housing. Animals were either single housed or group housed (2 or more animals/group) depending on the facility. One facility used Aspen wood chip bedding in some cages over a raised floor and remaining cages and facilities had stainless steel or plastic-coated flooring. Most animals on study were single housed. Animals were on a 12-hr light–dark cycle. The diet differed between sites, but all animals were provided weighed amount of diet based on the sex and age of the animals, and were provided ad lib access to water. At NA sites, temperatures, air changes, and humidity were all within acceptable ranges according to the guide for the care of laboratory animal standards for pigs (National Research Council 1996, 2011). At the UK site, all animals were housed according to the guidelines set forth in the Code of practice for the housing and care of animals used in scientific procedures (Home Office 2014).
Figures
Copies of some original photomicrographs were cropped to required dimensions and globally adjusted for white balance and color saturation to match the original stained histologic slide using Adobe Photoshop CS5.5.
Results
Five CROs from NA, one CRO from the EU, and two U.S. minipig suppliers participated. Data provided here represent 1,596 total animals (1,390 Göttingen, 118 Hanford, and 88 Yucatan) from all sites, covering the period of 2004 to 2015. Not all organs were examined with equal frequency, and due to lack of standardized diagnostic nomenclature, inconsistencies were noted in terminology used to describe lesions between facilities. The most common background lesions for all organ systems were inflammatory cell infiltrates, foci of inflammation, and hemorrhage. The severity of most findings was recorded and is available in the Supplemental Tables 1–13. Severity of most changes were limited to minimal or mild. There were few instances of hemorrhage, congestion, or inflammation, where the severity was noted as moderate or marked. Lymph nodes and male reproductive organs most commonly had increased severity scores (see corresponding Supplemental Tables). Occasionally findings were not given a grade (e.g., thrombosis, atrium). Distribution was not separately defined but may have been considered when severity score was assigned.
No data were available for Göttingen or Hanford minipigs from studies at <2 months of age, although we do report previously unpublished data regarding congenital malformations in Göttingens.
In the following sections, the results from the table are briefly reviewed for each organ system. Background observations are only mentioned if they are found in >1% of the NA or EU Göttingen cohorts, in which case the values for the prevalence for each location will be noted. The threshold for reporting background findings in the Hanford and Yucatans is higher since there are fewer total animals. In these groups, the diagnosis had to be made on more than 1 animal in order for it to be mentioned.
All data from which this narrative is derived are available in Supplementary Tables for each organ system. These tables include total number of animals examined as well as severity scores. When stated that prevalence was increased in 1 group versus another (2–6 months vs. >6 months, or male vs. female), this indicates that the prevalence was greater than twice the value as the compared group, and percentages provided are calculated based on all animals of particular breed examined regardless of geographic location.
Inflammatory cell infiltrates are accumulations of inflammatory cells without corresponding tissue damage. In contrast, inflammation was interpreted to denote that there is tissue damage associated with the inflammatory cells present within the organ.
Congenital
Gross lesions present at birth in Göttingen minipigs from the NA colony included cataracts (0.49%), entropion (0.29–0.31%), cleft palate (0.18%), syndactyly (0.11%), atresia (0.07%), cryptorchid (0.21%), monorchid (1.48%), and hernias. Hernias included scrotal (0.17% right and 1.51% left), inguinal (0.41% right and 1.90% left), and umbilical (0.01%). In the EU colony, previously reported lesions include cleft palate (0.28%), syndactyly (0.76%), muscular–joint contracture (0.32%), diaphragmatic hernia (0.24%), cryptorchidism (2.8%), ventricular septal defect (0.24%), and aortic arch bifurcation are noted congenital lesions (Berggren and Jensen 2008). No congenital abnormalities were reported from either Hanford or Yucatan breeds.
Systemic
In this report, systemic diseases of thrombocytopenic purpura and porcine stress syndrome (1 animal each) were reported via clinical signs and histological diagnosis only in the EU cohort. Thrombocytopenic purpura has previously been reported as a finding in Göttingens in both NA and the EU (Carrasco et al. 2003; Dincer and Skydsgaard 2012; Maratea, Snyder, and Stevenson 2006), and subsequent breeding strategies have attempted to eliminate it. In this study, thrombocytopenic purpura was only reported in 1 male >6 months of age from the EU cohort. Gross necropsy of the animal revealed hemorrhages of the gastrointestinal (GI) tract, urinary bladder, ureter, heart, lung, thymus, lymph nodes, meninges, muscle, kidneys, and larynx. There was diffuse pallor in all tissues. Histologically, there was hemorrhage which correlated with the gross findings in addition to increased hematopoiesis in bone marrow and spleen, hemorrhagic casts within renal tubules, glomerulonephritis, and glomerulosclerosis. Porcine stress syndrome or malignant hyperthermia has not yet been reported in Göttingens in the literature. Here, we report a single case, again in the EU cohort, in a female between the ages of 2–6 months. Macroscopically, musculature was pale and wet with a rapid onset of rigor mortis (within 5 min). There was visceral congestion and pulmonary edema and hydrothorax. Microscopically, there was multifocal myofiber degeneration, swelling, and edema within the quadriceps and longissimus dorsi. Cardiomyocyte degeneration and necrosis with hemorrhage were also observed. Hepatic and pulmonary findings were consistent with gross observations. No systemic abnormalities were reported from either Hanford or Yucatan breeds.
Respiratory: (Supplemental Table 1)
Göttingen minipigs
In NA, the nasal cavity was rarely examined (8/1,018), larynx and trachea were more often examined (544 and 954/1,018, respectively), whereas lung, bronchi, and pleura were examined in all animals (1,018/1,018). In the EU, larynx, trachea, and lung/pleura were examined in 332 animals (100%). In the questionnaire table, lung was divided into bronchioles, terminal bronchioles, and alveoli. Responses indicated that the lung was scored as a single entity, as described in this report.
Although varying by geographic cohort, in the larynx the most common background lesions were inflammatory cell infiltrate (4.4% NA and 4.8% EU), acute inflammation (3.5% NA and 0% EU), subacute/chronic inflammation (3.5% NA and 0% EU), foreign bodies (0% NA and 1.2% EU), and hemorrhage (2.5% NA and 0% EU). Inflammatory cell infiltrate in EU animals was much more prevalent in males and in animals >6 months of age. In the NA cohort, acute inflammation was much more prevalent in females.
Inflammatory cell infiltrate was the most common finding in the trachea in EU (0.4% US and 3.0% EU). Hemorrhage was the most common finding in the trachea in NA (1.8% NA and 0% EU).
In the lungs, the most common background finding was the presence of macrophages within alveoli (9.4% NA and 9.6% EU) and was more prominent in 2- to 6-month-old animals relative to those >6 months of age (14% 2–6 months, 5.3% >6 months, NA and EU combined; Figure 1). Pulmonary hemorrhage (4.4%) and congestion (1.6%) were both commonly reported in NA but not at all in the EU. Infiltration of inflammatory cells into the lung tissue was seen in 28% of the EU animals but in only 1.3% of NA animals. Inflammation (NOS) was noted in both NA (1.3%) and EU (1.5%), and granulomatous inflammation/granuloma were also noted in NA (2.6%) but not the EU. Another common finding was mineralization (1.6% NA and 6.6% EU). The pleura displayed adhesions and associated fibrosis and inflammation (4.7% NA and 3.3% EU; Figure 2).
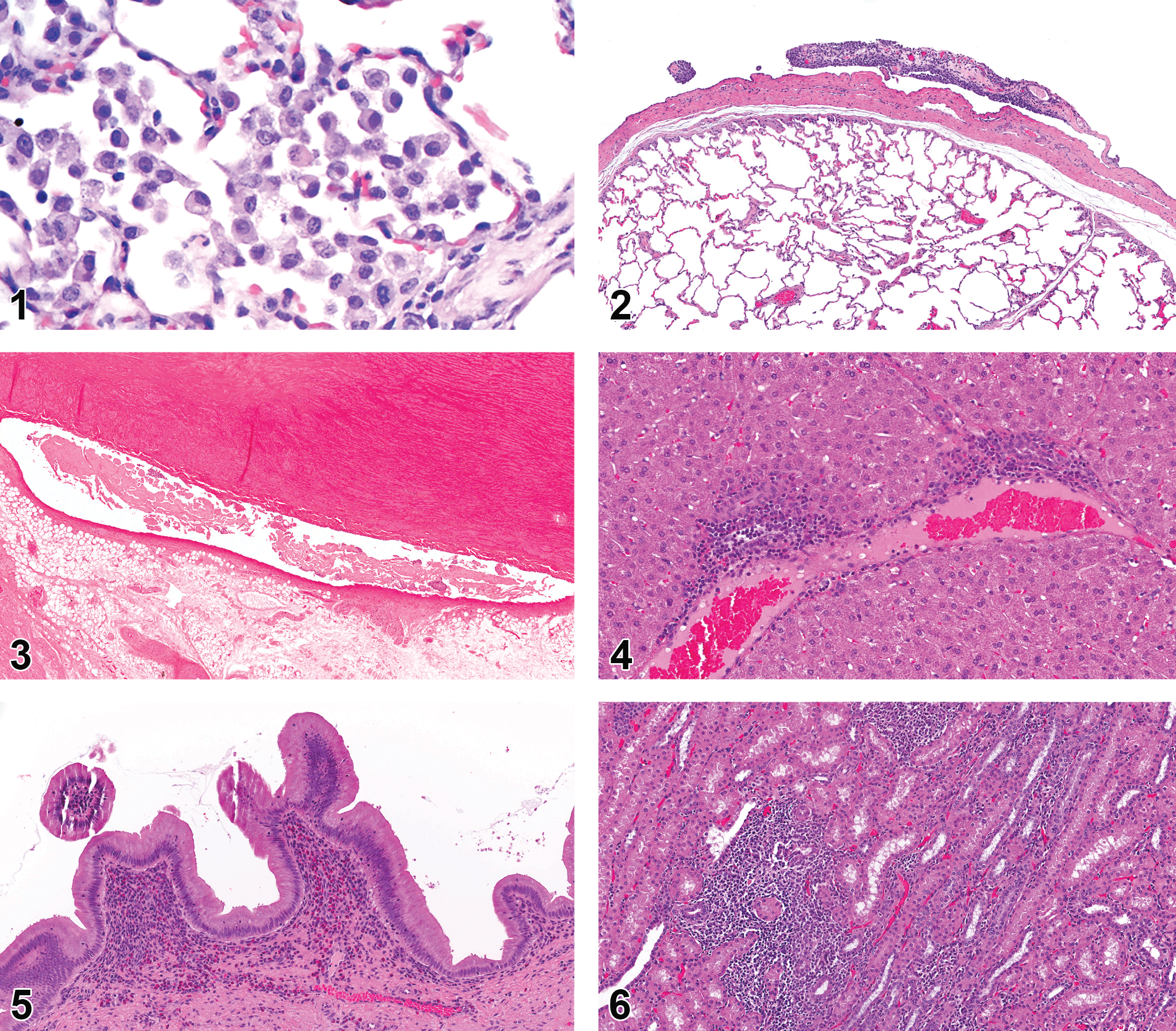
Hanford minipigs
No findings were noted in the larynx or trachea of Hanford minipigs. The most common findings in the lungs were microgranulomas/granulomatous inflammation (5.1%), which were only found in animals between 2–6 months old and inflammatory cell infiltrate (3.4%).
Yucatan minipigs
Inflammatory cell infiltrates were commonly seen in the trachea of young Yucatan minipigs (10% of animals <2 months) and were not present in animals of any other age-group. Many of the findings in the lungs of Yucatan minipigs were associated with age. Syncytial alveolar cells were only seen in animals <2 months old (17%). Pigmented macrophages (4.5%) were primarily in the <2 months cohort as well, although 1 animal between 2 and 6 months old also displayed this finding. The infiltration of inflammatory cells was also related to age and increased as the animals aged (0% <2 months, 3.3% 2–6 months, and 18% >6 months). Inflammation of many characters including granulomatous/granulomas (3.4%), subacute/chronic (8%), acute (5.7%), and uncharacterized (NOS; 11%) was often present. Pulmonary hemorrhage was reported in 8% of the animals.
Cardiovascular: (Supplemental Table 2)
Göttingen minipigs
The most common cardiac finding was inflammatory cell infiltrate (3.6% NA and 5.4% EU). Hemorrhage (1.9% NA and 0% EU) and myofiber degeneration were also reported (1.1% NA and 0% EU) in NA. Hypertrophy and hyperplasia of the mesothelial cells of the heart were seen in animals over 6 months of age (0.7% NA and 1.8% EU; 1.0% in animals >6 months old, 0% in animals <6 months of age; Figure 3).
Hanford minipigs
In the Hanford breed, cardiac changes were only noted in animals 2–6 months old. No changes or abnormalities were found in animals >6 months old. The only findings of note are subacute to chronic inflammation (14% 2–6 months) and endocardial inflammation (7% 2–6 months). No changes were reported in the blood vessels.
Yucatan minipigs
Yucatan minipigs displayed inflammatory cell infiltrate in both myocardium (6.8%) and perivascular (2.3%) locations. Epicardial changes noted were inflammation (2.3%) and cartilaginous metaplasia (2.3%). No changes were reported in the blood vessels.
Gastrointestinal: (Supplemental Table S3)
Göttingen minipigs
The GI tract includes the tongue, salivary glands, esophagus, stomach, and intestines. Background findings in the tongue of Göttingen minipigs included myocyte degeneration and necrosis (1.4% NA and 0.3% EU), which was more common in males (1.7% males and 0.5% females). Other findings included chronic inflammation (1.6% NA and 0% EU), granulomatous inflammation, (0.3% NA and 1.5% EU), and unspecified inflammation (0% NA and 1.8% EU). Infiltration of inflammatory cells without inflammatory changes was also observed (0% NA and 5.2% EU).
The salivary glands were not uniformly examined by all facilities. Sialoliths were routinely found in salivary glands examined by the EU organization but were not observed in any NA studies (0% NA and 1.8% EU). Inflammatory cell infiltrate was the most common finding in the mandibular (2.7% NA and 10.5% EU), parotid (0.7% NA and4.2% EU), and sublingual (1.3% NA and 1.2% EU) salivary glands. Mineralization (2.5% NA and 0% EU) was also detected in the sublingual gland.
In Göttingen minipigs, chronic inflammation of the esophagus (1.7% NA and 0.3% EU) was more often seen in animals that were between 2 and 6 months old compared to minipigs older than 6 months of age. Granulomatous inflammation of the esophagus was also prevalent (0.33% NA and 1.2% EU), with no age predilection.
The regions of the stomach were assessed separately as nonglandular (narrow strip near the cardia) and glandular portions. Erosions and ulcers were the most common changes found in the nonglandular portion (1.6% NA and 5.1% EU). Inflammatory cell infiltrates (0.7% NA and 16.26% EU), chronic inflammation (1.3% NA and 0% EU), and inflammation NOS (0.5% NA and 1.5% EU) were the most common gastric findings of the glandular portion. Inflammatory cell infiltrates were more commonly found in males (6.1% vs. 3.2% in females) and were more common in animals 2–6 months old (6.8%, 2.1% in >6 months). Inflammation NOS was also more common in the younger animals (1.4% 2–6 months and 0% >6 months).
In the intestine, few background findings were reported. The duodenum had inflammatory cell infiltrates (0.5% NA and 6.6% EU) with a predilection for the 2–6 months group (1.8% 2–6 months and 0.3% >6 month). Congestion in the duodenum (2.1% NA and 0% EU) was more often reported in NA. The jejunum had no background lesions with a prevalence >1.0%. In the ileum, the most common background change was an infiltrate of eosinophils (1.5% NA and 0% EU). No findings with a prevalence of >1% were described in either the cecum or the colon of Göttingen minipigs. Inflammation NOS (0% NA and 1.5% EU) and inflammation of the circumanal glands (0% NA and 1.2% EU) were seen in the rectum in the EU cohort.
Hanford minipigs
The tongue displayed no histological abnormalities in Hanford minipigs. The mandibular salivary gland (8.5%) and esophagus (2.5%) had inflammatory cell infiltrates that were only present in animals 2–6 months old. In the glandular stomach, both erosions/ulcers (5.7%) and mucosal hyperplasia (3.4%) were present only in animals >6 months of age. Perivascular inflammatory cell infiltrates (1.7%) were also observed in the glandular stomach.
Yucatan minipigs
Hemorrhage was reported (3.4%) in the tongue of Yucatan minipigs. Few animals had inflammatory cell infiltrate (2.3%) in the tongue. The salivary glands displayed few changes. The mandibular salivary gland had inflammatory cell infiltrates (3.4%), the parotid gland had vacuolation of cells (3.4%), and the sublingual gland had mineralization (4.5%). In the esophagus, 30% of animals <2 months old displayed acute inflammation. Erosions or ulcers (5.7%) and acute inflammation (3.4%) were found in the nonglandular portion of the stomach. The glandular portion displayed both chronic inflammation (2.3%) and inflammatory cell infiltrate (2.3%). Acute inflammation (3.4%) was also present in the ileum of animals >6 months old. The cecum (4.5%), colon (4.5%), and rectum (2.3%) all had acute inflammation reported. Inflammation of unspecified character (NOS; 2.3%) was also noted in the colon.
Liver, Gallbladder, and Pancreas (exocrine): (Supplemental Table 4)
Göttingen minipigs
The most common background finding in the liver of Göttingen minipigs was infiltration of inflammatory cells (13.8% NA and 34.2% EU; Figure 4). The only other remarkable finding in the liver was the increase in pigmented macrophages or Kupffer cells (1.0% NA and 0% EU).
Gallbladder observations included infiltration of inflammatory cells (3.5% NA and 3.6% EU), which was increased in animals >6 months of age (0.2% 2–6 months and 1.4% >6 months; Figure 5). Unspecified (0.5% NA and 1.8% EU) and subacute/chronic (1.3% NA and 0% EU) inflammation were both seen in the gallbladder. Aplasia of the gallbladder was present in 2.1% of animals examined in the EU. Hypoplasia of the gallbladder was reported in NA (0.1%) with a higher frequency in EU (0.9%). Inflammatory cell infiltrates in the exocrine pancreas were reported more frequently in the EU cohort (0% NA and 12.0% EU).
Hanford minipigs
The most common finding in the liver was adhesions or fibrosis of the hepatic capsule (5.9%). Inflammatory cell infiltrates were reported in only 1 animal (0.9%). No abnormalities were reported in the gallbladder or exocrine pancreas.
Yucatan minipigs
Hepatic inflammatory cell infiltrates were present in 41% of Yucatan minipigs. Vacuolation of hepatocellular cytoplasm was only reported in animals <2 months old (97%). No abnormal findings were present in the gallbladder or exocrine pancreas.
Urinary: (Supplemental Table 5)
Göttingen minipigs
Among all organs, the kidney has the highest prevalence of infiltration of inflammatory cells (19.2% NA and 50.7% EU; Figure 6), which are more commonly observed in females (2.2% and 0.9% males). Inflammation NOS (1.6% NA and 1.4% EU) is also observed in the kidney. Mineralization is commonly seen (2.1% NA and 23.4% EU; Figure 7). Spontaneous glomerulonephritis has been reported in the Göttingen kidneys (Vezzali et al. 2011). In this report, glomerulonephritis was found primarily in EU (2.3%) but also in NA (0.2%). Nephropathy was not common in EU Göttingens (0%) but was reported more in NA (2.5%). Renal background findings also consisted of many tubular changes including degeneration and regeneration of renal tubules (6.8% NA and 0% EU; Figure 8), tubular dilation (1.7% NA and 3.7% EU), tubular basophilia (3.9% NA and 1.4% EU), and hyaline casts (1% NA and 0% EU). Tubular dilation was more commonly seen in females (3.4% and 0.9% males). Other renal changes reported more frequently among females included renal cysts (2.1% NA and 1.1% EU; 2.3% females and 1.4% males) and fibrosis (1.3% NA and 0.6% EU; 1.8% females and 0.4% males). Cytoplasmic vacuolation of the urothelium in the kidney was also reported (1.0% NA and 5.9% EU). The most prevalent urinary bladder change was infiltration of inflammatory cells (0.6% NA and 4.2% EU).
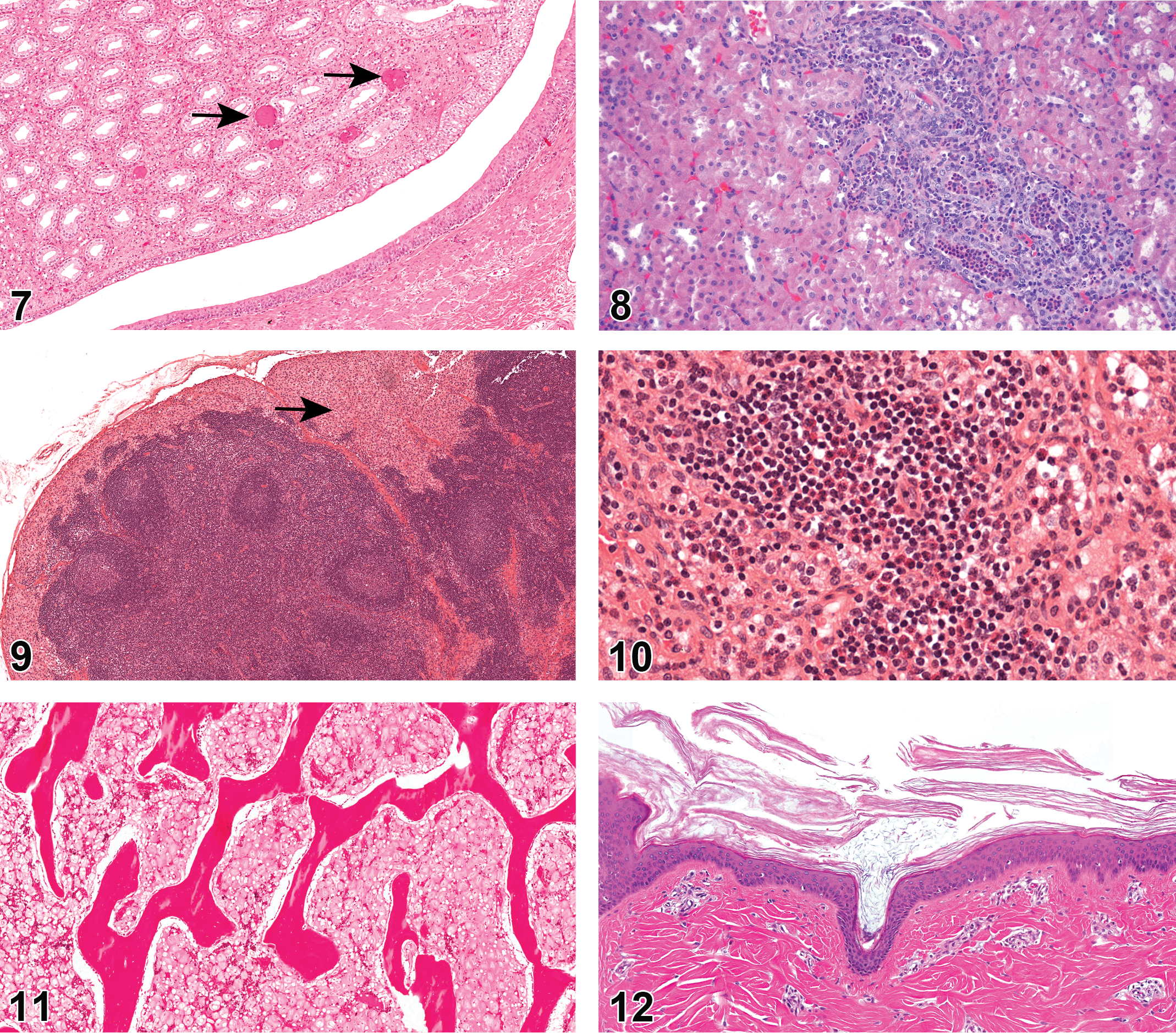
Hanford minipigs
Inflammatory cell infiltrates of the kidney were present in 20% of the Hanford minipigs.
Yucatan minipigs
Inflammatory cell infiltrate (36%) was the most common change seen in the kidney of Yucatan minipigs. Other renal changes in the Yucatan breed included hemorrhage (3.4%) and fibrosis (2.3%). Vacuolation of the renal tubular epithelial cells (3.4%) was present only in animals <2 months old. The urinary bladder also had inflammatory cell infiltrates (2.3%).
Lymphoid/Hematopoietic: (Supplemental Table 6)
Göttingen minipigs
Hemorrhage (7.6% NA and 0% EU), lymphoid depletion (3.8% NA and 0% EU), and mineral deposition (0.7% NA and 1.5% EU) were the most common background observations in the Göttingen minipig thymus. Due to the anatomical location within the cervical region, the thymus may be injured during blood collection (Dincer and Skydsgaard 2012; Jeppesen and Skydsgaard 2015).
The spleen did not have any background lesions with prevalence >1%. Lymph nodes regularly sampled included the mesenteric (885 animals) and mandibular (825 animals). Other lymph nodes that were examined from a smaller number of animals included mediastinal (69 animals) along with axillary, pancreatic, prescapular, and inguinal from an unspecified number of animals. Results were reported for lymph nodes from 320 animals that did not specify location of the lymph node. Sinus histiocytosis was prevalent (0–4.2%; Figure 9), as were intrasinusoidal erythrocytes and erythrophagocytosis especially in the mediastinal lymph node. Chronic inflammation was prevalent in both mandibular (1.6% NA and 0% EU) and mesenteric lymph nodes (1.6% NA and 0% EU). Lymphoid hyperplasia was occasionally present. Pigmented macrophages were present in both mediastinal (7.2% NA and 0% EU) and mandibular (5.8% NA and 0% EU) lymph nodes in NA. The mesenteric lymph node was most likely to have an infiltrate of eosinophils (7.5%; Figure 10). Lymphoid atrophy in lymph nodes was more commonly seen in males than females (4.7% males and 0% females; LN NOS).
Both mucosa- and gastric-associated lymphoid tissues were examined, and no lesions were noted for either tissue in any Göttingen minipigs.
Hanford minipigs
The only findings reported in more than 1 animal in the lymphoid system of Hanford minipigs were hemorrhage of the mandibular (1.7%) and mesenteric (2.5%) lymph nodes.
Yucatan minipigs
In the thymus of Yucatan minipigs, hemorrhage (2.3%) and lymphoid depletion (7.1%) were noted in animals over 6 months old, and pigment was present in animals 2 to 6 months old (20%). Lymphoid hyperplasia was noted in animals younger than 2 months in the spleen (6.7%) and in the mesenteric (83.3%) and mandibular (93.3%) lymph nodes. The mesenteric lymph node also displayed hemorrhage (2.3%), infiltration of inflammatory cells (2.3%), and eosinophils (3.4%). The mandibular lymph node displayed hemorrhage (4.5%) and pigment (2.3%). Increased intrasinusoidal erythrocytes were present in the axillary lymph node (3.4%).
Bone Marrow: (Supplemental Table 6)
Göttingen minipigs
Serous atrophy of fat in the bone marrow has been reported in many Göttingen minipigs, primarily males and has been hypothesized to be associated with restricted feeding (Bollen and Skydsgaard 2006). The change is represented by homogenous eosinophilic material with decreased hematopoietic tissue in the marrow cavity along with diminished or degenerated adipocytes (Dincer and Skydsgaard 2012). Serous atrophy of the bone marrow fat was a common finding in minipigs in EU (11.75%; Figure 11), whereas it was not reported at all in NA animals. Males were overrepresented (19.3% male, 4% female), and the finding was more prevalent in older animals (23.5% in animals >6-month-old and 0% in animals <6-month-old). In this report, serous atrophy of bone marrow fat was noted in females, whereas previously it had only been reported in males.
Hanford minipigs–Yucatan minipigs
No changes were noted in the bone marrow of Hanford or Yucatan minipigs.
Musculoskeletal: (Supplemental Table 7)
Göttingen minipigs
The most common findings in skeletal muscle of Göttingen minipigs were degeneration (1.7% NA and 0% EU), degeneration and regeneration of myofibers (0.1% NA and 5.4% EU), and inflammatory cell infiltrate (1.9% NA and 0.3% EU). Degenerate and regenerating myofibers have previously been reported (Dincer and Skydsgaard 2012).
The bones most commonly examined included the sternum, femur, and occasionally tibia. There were no lesions reported or incidence was below 1% in bones and joints.
Hanford minipigs
No changes were identified in the musculoskeletal system of Hanford minipigs in this study.
Yucatan minipigs
Only one Yucatan minipig had a change reported in the musculoskeletal system, which was fibrosis of the sternum. No other changes were observed.
Integument: (Supplemental Table 8)
Göttingen minipigs
Göttingen minipigs used for studies in NA often displayed minor epidermal changes such as hyperkeratosis (4.5% NA and 0.3% EU; Figure 12) and hyperplasia (2.5% NA and 0% EU; Figure 13), and sometimes had exudate present on the epidermal surface (3.1% NA and 0% EU; Figure 14) or a serocellular crust/scab (1.4% NA and 0.3% EU). Abscesses or pustules were present in the skin (1.5% NA and 0.3% EU) along with foci of inflammation, NOS (0.2% NA and 8.7% EU), and chronic inflammation (1.7% NA and 0% EU). Inflammatory cell infiltrates were found in the epidermis (4.51% NA and 0.3% EU) and in the dermis with a perivascular distribution (1.8% NA and 0% EU). Mammary tissue was examined in both males and females. Glandular dilation was observed in both sexes (2.9% NA and 0% EU; Figure 15).
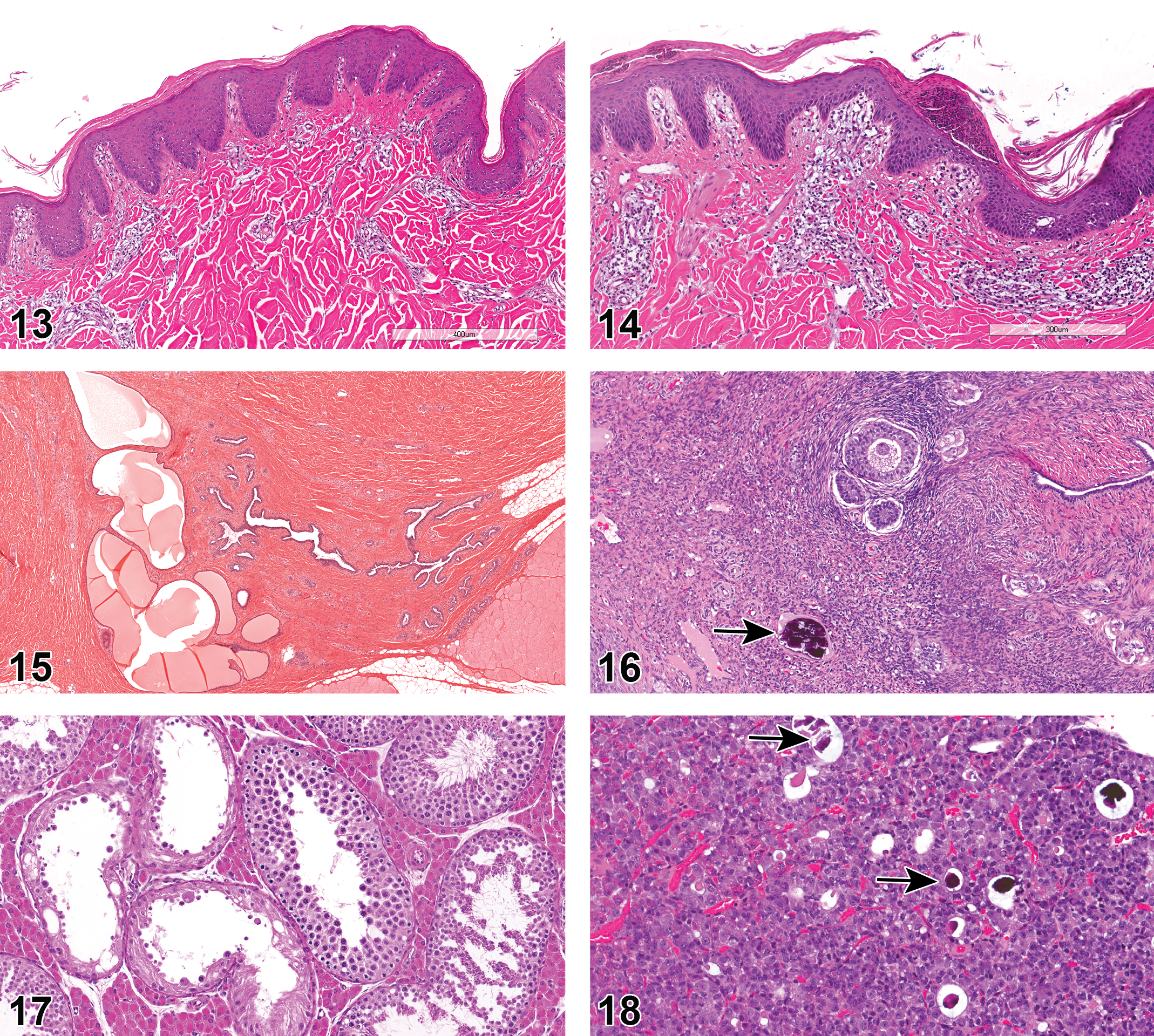
Hanford minipigs
The epidermis of Hanford minipigs displayed inflammatory cell infiltrates (3.4%).
Yucatan minipigs
The only changes reported in the Yucatan breed were in animals that were >6 months of age. Changes included exudate present on the epidermal surface (10.7%), epidermal hyperplasia (10.7%), and subacute to chronic inflammation (7.1%).
In the Yucatans, mammary tissue displayed cellular pigmentation in 8.5% of females examined and was not observed in males. The pigmentation in the mammary tissue was only present in animals 2 to 6 months old, not in younger or older animals.
Female Reproductive: (Supplemental Table 9)
Göttingen minipigs
Mineral deposition was a common finding in the ovaries of Göttingen minipigs (31.0% NA and 30.7% EU; Figure 16). Squamous metaplasia of the oviduct has been recorded in EU animals (0% NA and 3.0% EU). Immature or juvenile development of the uterus, based on the lack of or incomplete uterine glandular development, was seen in some females (1.3% NA and 0% EU). Endometrial hyperplasia (2.8% NA and 0% EU), inflammatory cell infiltrates (1.0% NA and 3.0% EU), and luminal exudate (1% NA and 0% EU) were also occasionally seen in the uterus. Epithelial hyperplasia (1.3% NA and 0% EU), hyperkeratinization (1.3% NA and 0% EU), and suppurative exudate (1.5% NA and 0% EU) were found in the cervix. No vaginal changes were reported in EU studies, however in NA, inflammatory infiltrates (3.4%), exudate (2%), edema (1%), and atrophy of the epithelium (1.5%) were all noted.
Hanford minipigs
No abnormal findings in the female reproductive tract of Hanford minipigs were identified.
Yucatan minipigs
Seventy-three percent of the Yucatan minipigs younger than 2 months old had immature ovaries. Mineralization (4.3%) was another common finding in the ovary.
Male Reproductive: (Supplemental Table 10)
Göttingen minipigs
Testicular hypoplasia, degeneration, and atrophy were combined as one call in this report. Hypoplasia and atrophy are difficult to separate (Thuilliez et al. 2014), and degeneration and atrophy may be a continuum (Foster 2007). This combination was also facilitated as some organizations used degeneration/atrophy, whereas others use hypoplasia/atrophy. We understand that they are separate processes, but the vast majority of calls fell into degeneration/atrophy, tubular. This was the most common finding in the male reproductive tract (24.1% NA and 37.3% EU; Figure 17). While the majority of degeneration/atrophy lesions were of minimal or mild severity, moderate and marked or severe grades were not uncommon. Leydig cell hyperplasia (2.2% NA and 0% EU), vacuolation of tubular cells (1.6% NA and 0.6% EU), and immature or juvenile development of the testes with decreased spermatocytes and decreased spermatocyte maturation (1.2% NA and 0% EU) were also reported.
Within the epididymis, aspermia or oligospermia (5% NA and 4.2% EU), luminal cellular debris (5% NA and 7.8% EU), epididymal cysts (5% NA and 3% EU), and hypoplasia (1.2% NA and 0% EU) were observed and were likely related to the testicular hypoplasia, degeneration, and atrophy.
The prostate commonly displayed mineralization (14.8% NA and 4.2% EU). Concretions (2.4% NA and 0% EU), acinar dilation or vesicles (1.3% NA and 0% EU), inflammatory cell infiltrate (1.1% NA and 1.8% EU), and inflammation NOS (0.2% NA and 3.6% EU) were also reported in Göttingens.
The most common findings in the seminal vesicle were inflammatory cell infiltrate (0.8% NA and 4.8% EU) and inflammation NOS (0% NA and 1.2% EU). The bulbourethral glands did not have any findings with prevalence >1%.
Hanford minipigs
In the testis of 2- to 6-month-old Hanford minipigs, 11.6% were found to be immature. Similarly, 7% of epididymides of the same age-group were immature. All male reproductive organs examined from animals >6 months of age were consistent with sexual maturity.
Yucatan minipigs
In the Yucatan, animals <2 months old were examined, and 100% of the testes from these animals were immature. The testis in these young animals also displayed hematopoiesis in 46.7% of the animals. This age-group also displayed inflammatory cell infiltrate in the epididymis (100%), seminal vesicle (93.3%), and immature prostate glands (93.3%). Aspermia or oligospermia was found in 10.8% of all male Yucatans examined.
Nervous system, Central, and Peripheral: (Supplemental Table 11)
Göttingen minipigs
Nonspecified inflammatory cell infiltrates were common in the cerebrum (2.2% NA and 8.4% EU) of Göttingen minipigs. Perivascular inflammation (1.5% NA and 0% EU) and gliosis (3% NA and 0.6% EU) were also reported. Mineralization of the meninges was present in NA (5.8%) but was not reported in EU animals in this study. Jeppesen and Skydsgaard also reported that mineralization is often seen in the meninges but not reported in their experience (2015).
The sciatic nerve was used most commonly to assess the peripheral nerve health, and no findings exceeded the prevalence of 1%. Some groups assessed the optic nerve separately, but again no findings exceeded 1%.
Hanford minipigs
Infiltration of inflammatory cells (4.2%) was present in the brain of Hanford minipigs.
Yucatan minipigs
Inflammatory cell infiltrate (6.8%) was observed in the brain of Yucatan minipigs.
Endocrine: (Supplemental Table 12)
Göttingen minipigs
In NA animals, hemorrhage of the thyroid gland was reported in 7.2% of Göttingen minipigs, whereas it was present but not reported from the EU group since it is a known finding in animals with venipuncture (Rinke 1997). There was inconsistency among NA CROs regarding whether it was recorded or not. It was not stated in the data submitted whether animals had repeated blood sampling. Venipuncture has been reported to be associated with thyroid inflammation, hemorrhage, and fibrosis in minipigs.
Mineralization of the pituitary was common in Göttingen minipigs (5.7% NA and 6.9% EU; Figure 18) and was more prevalent in animals >6 months old. Animals between 2 and 6 months of age displayed the lesion rarely (0.7%), whereas animals >6 months of age were 10 times more likely to have mineral deposition within the pituitary (10.7%).
The adrenal gland commonly displayed inflammatory cell infiltrates (5.1% NA and 15.1% EU). Vacuolation of the adrenal cortical cytoplasm was present occasionally (0.2% NA and 1.8% EU).
There were no lesions in the parathyroid gland, which exceeded the 1% prevalence, and no lesions were described in the endocrine pancreas.
Hanford minipigs
The Hanford adrenal gland displayed inflammatory cell infiltrates (1.7%).
Yucatan minipigs
Mineralization of the pituitary was only present in Yucatan minipigs older than 6 months (25%).
The thyroid gland commonly displayed hemorrhage (9.1%) and occasionally contained pigmented macrophages (2.3%).
Inflammatory cell infiltrates (4.5%) have been observed in the adrenal gland.
Special Sense Organs: (Supplemental Table 13)
Göttingen minipigs
Ocular lesions of Göttingen minipigs consisted of minimal inflammatory cell infiltrate (0.11% NA and 2.7% EU). Inflammatory cell infiltrate in the lacrimal gland was present in NA (4.1%) animals. The lacrimal gland was not assessed in the EU cohort.
Hanford minipigs
The lacrimal glands of Hanford minipigs had inflammatory cell infiltrates in 1.7% of animals.
Yucatan minipigs
Chronic inflammation of the eye was present in 3.4% of Yucatan minipigs.
Discussion
Comparison of background pathology data obtained from sources in NA and EU revealed several instances of differences in the incidence of these changes between Göttingen minipigs sourced from the US versus EU vendors. There was only one CRO from the EU that participated, which may have skewed the results, such as interpretation of inflammatory cell infiltration versus inflammation. However, there are also many differences which are unlikely to be attributed to the differences in interpretation between sites (such as serous atrophy of fat in the bone marrow, gallbladder aplasia, etc.). Some of this could also be potentially attributed to the lack of a common internationally accepted terminology (INHAND) guideline for minipigs, resulting in inconsistent terminology usage and inconsistent recording of common background changes among pathologists across various sites. Another point that arose was the use of breed versus strain when discussing different minipigs. In mice, strain implies inbred (brother × sister mating), and stock implies that the animals are outbred. Breed was used in lieu of strain in this report to further synchronize nomenclature used between species.
This study reported common findings, most of which were minimal to mild in nature and rarely of moderate or marked severity. Varying incidence and severity of common background pathology can often confound interpretation of the toxicological significance of these changes as they occur in the context of preclinical safety assessment. In the Göttingen minipig, the most common background findings in a toxicity or device study include those in the kidney, lymph nodes, testes, and cardiovascular systems as well as the integument lesions. Concurrent controls, historical control data, and published information regarding the prevalence and severity of background pathology are helpful in making a sound judgment on the toxicological relevance of a particular finding. Increased incidence and/or severity of common background lesions may indicate test article–related effects (Crissman et al. 2004). The Hanford minipigs had the fewest background pathological findings.
Inconsistent recording of common background findings between various test sites was also observed following the analysis of the data. For example, some CROs do not report findings known to be physiological or background changes (mineralization; Jeppesen and Skydsgaard 2015). Hemorrhage and congestion may not have been reported by all participating sites as it can be an artifact of euthanasia (Grieves et al. 2008). Eosinophilic infiltrates were not separated from other types of inflammatory cell infiltrates by the EU group, and all inflammatory cell infiltrates were lumped into the generic “inflammatory cell foci,” which may explain why it appears that eosinophilic reactions or infiltrates are more common in NA. Once these combinations occurred, and the data were analyzed, Göttingen and Yucatan minipigs frequently (14–40.9%) had inflammatory cell foci in the liver whereas in the Hanford minipigs, this change was reported in only 1 animal.
In EU Göttingen minipigs, the most common findings reported previously were arteritis, tubular atrophy/hypoplasia of testes, serous atrophy of bone marrow fat, and thrombocytopenic purpura (Svendsen 2006). As seen here, arteritis is a rare call in NA, and serous atrophy of bone marrow fat is not reported in any NA animals. Arteritis was still present in EU animals (see Supplemental Tables), but the prevalence did not exceed 1%. While some of the perivascular inflammation or inflammatory cell infiltrates reported in NA and EU may be the equivalent of arteritis, they may also be vascular-associated lymphoid tissue which has been reported in swine at vascular branching points which are susceptible to atherosclerotic changes (Langohr et al. 2008).
In this study, 6 months were chosen as the cutoff between juvenile and adult animals, because that is often the cutoff used based on previous reports (Damm Joergensen 1998). Tortereau et al. showed that as many as 50% of Göttingen females are not mature at 6 months of age, and that it takes female Göttingens nearly 7.5 months to reach sexual maturity, determined by the presence of ovarian corpora luteal, which correlates with uterine weight not ovarian weight (Tortereau, Howroyd, and Lorentsen 2013). Note that the lack of reports of immature reproductive organs in females older than 2 months in either Hanford or Yucatan breeds in this study may reflect a decision to not record this finding in general toxicity studies rather than a true assessment of sexual maturity in these animals.
Leydig cell hyperplasia is a difficult diagnosis in Göttingen minipigs, as these cells may be quite prominent, especially if there is tubular atrophy or hypoplasia (Jeppesen and Skydsgaard 2015). This results in some facilities not using the diagnosis or using it sparingly. Assessment of the maturity of the testes also differs between sites, as some sites assume the testes are immature at younger ages and do not record it. The diagnosis of hypoplasia/atrophy may be part of a continuum and may just be immaturity in the testes. However, a recent study in Göttingens reported that most males are sexually mature at 8 weeks of age (2 months). Factors to consider are housing with mature boars and conspecifics which accelerate maturation (Navratil et al. 2014).
In Hanford minipigs, sexual maturity was complete in most animals between 2 and 6 months, and no immature reproductive organs were reported in animals older than 6 months.
This study reports a case of porcine stress syndrome (from EU), even though the Göttingen minipig breed has been tested and does not have the mutation of the ryanodine receptor gene and is assumed to not be susceptible to porcine stress syndrome, including malignant hyperthermia. However, there has been a newly identified mutation in the dystrophin gene which leads to loss of animals due to transport stress, and pathological findings are similar to porcine stress syndrome (Nonneman et al. 2012).
Pigmentation within macrophages of the kidney, liver, lymph nodes, and pigment deposition in renal tissues has been associated with iron administration months earlier (Rinke 1997). Pigmentation was not reported in EU but was prevalent in NA Göttingen animals and in the Yucatan minipigs younger than 2 months. This is consistent with the iron administration protocol of the supplier, which differs between NA and EU Göttingen breeding sites. In both EU and NA, the same dose is administered within 48 hr of birth. In NA, a second dose is administered at a later time point at breeding facilities for all minipigs (Göttingen, Hanford, and Yucatan), which is not done in the EU Göttingen colonies. Another difference between the NA and EU Göttingen breeding facilities is that EU animals do not receive vaccines, but NA pregnant sows are prophylactically vaccinated with Clostridium perfringens Types C and D toxoid twice during pregnancy.
Granulomatous inflammation is reported in multiple organs in this study, with no obvious reason for their presence. Granulomatous inflammation has previously been reported in pigs with no obvious cause and while rare does occur and is not associated with an identified specific pathogen or disease process (Baba et al. 2006). Mineralization of tissues has been reported numerous times in pigs. This has been hypothesized to be an inflammatory lesion associated with finely ground feed as well as heterotopic calcification due to dietary imbalances and hypervitaminosis D3 (Gilka et al. 1978; Gilka and Sugden 1981).
Chronic necrotizing cholecystitis/hypoplasia/aplasia has been reported previously as a background finding and has been described as diffuse necrotizing inflammation and hemorrhage with granulomatous inflammation extending into the muscular layer (Dincer and Skydsgaard 2012; Jeppesen and Skydsgaard 2015). Although necrosis of the gallbladder was not reported in any animals included in this report, the subacute/chronic inflammation calls may be part of the necrotizing cholecystitis lesion commonly reported in current literature.
Kidney tubular basophilia, degeneration, and regeneration had previously been combined as the same change. In rodents, it has recently been reported that these are different pathophysiological processes. Tubular basophilia in rodents may occur without a reparative process and that it may be early atrophy or persistent low-grade toxic injury (Frazier et al. 2012). Similarly, in minipigs, tubular basophilia is common and appears to be an early change but may be seen without other renal changes.
One of the drawbacks of this study is that findings were not associated with the route of administration of test article or venipuncture procedures to determine if these experimental procedures contributed to the prevalence of findings. The route of administration would be a major factor to consider in properly interpreting the incidence of some changes. For example in oral dosing, pulmonary alveolar macrophages and tongue lesions may be more commonly observed and skin changes could be more common in dermal/topical studies.
A few new findings and trends in the incidences of some of the findings which had no apparent explanation were noted in this study. These included inflammation and inflammatory cell infiltrates within the GI tract of Göttingen minipigs, which decrease with age. In addition, many of the renal lesions were more prevalent in females relative to males, and serous atrophy of the bone marrow fat was only seen in the EU but not NA animals, although the incidence appeared to be decreasing.
Conclusions
Pathological evaluation of toxicity studies is concerned not only with the recognition of lesions caused by treatment directly but also with the identification of spontaneous changes that may have increased in severity and/or incidence in treated animals. It is, therefore, vital to be aware of the background and incidentally occurring lesions that can be observed during the examinations of organs from laboratory animals in routine toxicity studies. Although the use of minipigs has aroused increased interest during recent 15 years, there is still a paucity of basic pathological background information. Particular similarities to humans include its cardiovascular anatomy and physiology, skin, GI system and digestion, renal, and immune systems. These similarities make the minipig an attractive model for translational medicine but also a viable alternative to dogs or nonhuman primates in toxicology studies in support of human clinical trials. The most common breed of minipigs currently used in toxicology and pharmacology research are the Göttingen, Yucatan, Sinclair, and Hanford. As demonstrated in the present report, there are many background findings in minipigs, some are more common than others.
Additionally, there are consistent differences between Göttingens of EU and NA origin, which have not been previously characterized, and between Göttingens and other breeds of pigs. Since all pigs are not biologically identical, this needs to be taken into account in the planning and evaluation of toxicological studies.
It is important to ensure notation of severity of lesions, as many background lesions are present only at low levels of severity and increased severity may represent a pathologic response. The standardization of terminology through the INHAND initiative will be of great benefit in consistent evaluation and comparison of findings, as the current wide range of terminology in use renders this a more difficult interpretive process.
We believe the background pathology presented in this report, which combines data from multiple sites in NA and EU, complements and expands the scope of existing literature by providing more recent (2004–2015) background findings which would serve as a useful reference material for pathologists, toxicologists, and regulators in the field of safety assessment.
Footnotes
Author Contribution
Authors contributed to conception or design (KH, KN, AS, and CH); data acquisition, analysis, or interpretation (KH, KN, AS, BJ, SM, JH, VV, MG, AS, NN, DS, and CH); drafting the manuscript (KH); and critically revising the manuscript (KN, AS, BJ, SM, JH, VV, MG, AS, NN, DS, and CH). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
