Abstract
The development of bleomycin-induced pulmonary fibrosis (BLEO-PF) has been associated with differences in genetic background and oxidative stress status. The authors’ aim was to investigate the crosstalk between the redox profile, lung histology, and respiratory function in BLEO-PF in C57BL/6, DBA/2, and BALB/c mice. BLEO-PF was induced with a single intratracheal dose of bleomycin (0.1 U/mouse). Twenty-one days after bleomycin administration, the mortality rate was over 50% in C57BL/6 and 20% in DBA/2 mice, and BLEO-PF was not observed in BALB/c. There was an increase in lung static elastance (p < .001), viscoelastic/inhomogeneous pressure (p < .05), total pressure drop after flow interruption (p < .01), and ΔE (p < .05) in C57BL/6 mice. The septa volume increased in C57BL/6 (p < .05) and DBA/2 (p < .001). The levels of IFN-γ were reduced in C57BL/6 mice (p < .01). OH-proline levels were increased in C57BL/6 and DBA/2 mice (p < .05). SOD activity and expression were reduced in C57BL/6 and DBA/2 mice (p < .001 and p < .001, respectively), whereas catalase was reduced in all strains 21 days following bleomycin administration compared with the saline groups (C57BL/6: p < .05; DBA/2: p < .01; BALB/c: p < .01). GPx activity and GPx1/2 expression decreased in C57BL/6 (p < .001). The authors conclude that BLEO-PF resistance may also be related to the activity and expression of SOD in BALB/c mice.
Introduction
Pulmonary fibrosis (PF) is a severe, chronic, and restrictive interstitial disease that results from inflammation-induced parenchymal injury followed by excessive extracellular matrix deposition (Cottin and Cordier 2008; Odajima et al. 2010). PF can be caused by different types of stimuli, such as persistent acute respiratory distress syndrome (Lamy et al. 1975), long-term exposure to silica (Driscoll et al. 1990), or cytotoxic treatment with bleomycin (Jones 1978). However, not all individuals exposed to the known stimuli indeed manifest PF (Fernandez Perez et al. 2010; Raghu et al. 2006).
Susceptibility to PF appears to be related to the interaction between multiple factors, such as genetic predisposition (Garcia 2011), oxidative stress (Homer et al. 2011; Kliment and Oury 2010), and type of inflammatory response (Homer et al. 2011). Several animal models have been widely used to investigate these interactions in the pathogenesis of PF, and many studies have used the bleomycin-induced PF (BLEO-PF) mouse model (Chung et al. 2003; Garcia-Prieto et al. 2010; Harrison and Lazo 1987; Lemay and Haston 2005; Martins et al. 2009; Moeller et al. 2008).
Bleomycin, which is widely used as an adjuvant in chemotherapy treatment, not only has cytotoxic anticancer properties but also presents oxidative properties mediated by copper, iron, and reactive oxygen species (Fyfe and McKay 2010; Muller and Zahn 1977). Studies have shown that patients undergoing bleomycin treatment who develop lung fibrosis present an imbalance between tissue inhibitors of metalloproteinase (TIMPs) and metalloproteinase (MMPs) activities (Manoury, Caulet-Maugendre, et al. 2006; Manoury, Nenan, et al. 2006) and upregulation of both Wnt (Konigshoff et al. 2009) and TGF-b-induced Notch signaling, which leads to the excessive deposition of collagen in the lung parenchyma (Aoyagi-Ikeda et al. 2010). However, little is known regarding the differences in bleomycin-related susceptibility in humans.
We have demonstrated that the differences in the effects of cigarette smoke depend on the mouse strain studied, suggesting that genetic background influences susceptibility to the effects of cigarette smoke on wound healing (Cardoso et al. 2009, 2007). Mouse strains and individual organs display a range of susceptibilities to cigarette smoke–induced oxidative stress. The BALB/c and C57BL/6 strains appear to be the best experimental models for studying the effects of cigarette smoke on the liver and lung, and the C3H and C57BL/6 strains are the best models for the effects of cigarette smoke on the brain (Rueff-Barroso et al. 2010). Studies have shown that C57BL/6 mice are more susceptible to PF and BALB/c mice are resistant, and these responses are mainly due to Th-mediated IFN-γ/IL-12 and IL-4/IL-5/IL13 cytokine release, respectively (Chung et al. 2003; Kikuchi et al. 2010).
Th1 cytokines are associated with the increased oxidant production that induces damage of key molecules, such as lipids, proteins, and DNA. The balance between oxidants and antioxidants can be maintained by the activity of antioxidant enzymes, such as catalase (CAT), superoxide dismutase (SOD), and glutathione peroxidase (GPx) (Russo et al. 2010). Oxidative stress occurs when a redox imbalance, defined by alterations in antioxidant enzyme activities, is followed by oxidative damage (Rahman and Kilty 2006). In this case, antioxidant treatments have been shown to decrease bleomycin-induced oxidant production (Chen and Stubbe 2005) and inflammation (Day 2008; Kikuchi et al. 2011; Russo et al. 2011; Tanaka et al. 2010).
Therefore, understanding the crosstalk between the redox profile, lung histology, and respiratory function in BLEO-PF in C57BL/6, DBA/2, and BALB/c mice may help to identify the missing links in the pathogenesis of PF.
Materials and Methods
Animals
Male 8-wk-old C57BL/6, DBA/2, and BALB/c mice (weight: 20–24 g) were purchased from the Division of Clinical Research, National Cancer Institute (Rio de Janeiro, Brazil). Mice were housed (10 per cage) in a controlled environment with a 12-h light/12-h dark cycle (lights on at 6 p.m.) and an ambient temperature of 25 ± 2ºC (humidity 40–60%). The animals had free access to water and food. The animals were acclimatized during the 2 weeks before the experimental procedures. All animal care and experimental protocols were conducted following the guidelines of the institutional care and use committee (Committee for Evaluation of Animal Use for Research from the Federal University of Rio de Janeiro, CAUAP-UFRJ) and the NIH Guide for the Care and Use of Laboratory Animals (ISBN 0-309-05377-3). The protocols were approved by CAUAP-UFRJ (under registry CEUA-IBCCF108).
Experimental Design
Intratracheal administration (i.t.) of bleomycin (Sigma-Aldrich, St. Louis, MO, USA) was performed on mice under intraperitoneal (i.p.) ketamine anesthesia as previously described (Manoury, Nenan, et al. 2006). After tracheostomy, bleomycin was instilled at a dose of 0.1 U in a final volume of 50 µL. The saline groups received saline by i.t. administration. This protocol was performed 3 times, and the number of animals and samples used in each experiment or assay is indicated in the Results section. Survival rates were analyzed throughout 21 days after bleomycin instillation. The remaining mice were subjected to a mechanical ventilation protocol and were sacrificed by exsanguination.
Pulmonary Mechanics
The animals were sedated with diazepam (1 mg i.p.), anesthetized with sodium pentobarbital (20 mg/kg body wt i.p.), paralyzed with pancuronium bromide (5 mg/kg body wt) intravenous (i.v), and mechanically ventilated (Samay VR15, Universidad de la Republica, Montevideo, Uruguay) with a frequency of 100 breaths/min, tidal volume of 0.2 mL, flow of 1 mL/s, and positive end-expiratory pressure of 2.0 cm H2O. The anterior chest wall was surgically removed. A pneumotachograph (1.5-mm ID; length = 4.2 cm, distance between side ports = 2.1 cm) was connected to the tracheal cannula for the measurement of airflow (V=), and changes in lung volume were obtained by digital integration of the flow signal. The pressure gradient across the pneumotachograph was determined by means of a Validyne MP-45-2 differential pressure transducer (Validyne Engineering, Northridge, CA, USA). The equipment resistive pressure (= Req·V’) was subtracted from pulmonary resistive pressure to obtain the intrinsic values. The transpulmonary pressure was measured with the Validyne MP-45 differential pressure transducer. Lung resistive (ΔP1) and viscoelastic/inhomogeneous (ΔP2) pressures, total pressure drop after flow interruption (ΔPtot = ΔP1 + ΔP2), lung static (Est) and dynamic (Edyn) elastance, and ΔE (Edyn − Est) were computed by the end-inflation occlusion method. ΔP1 selectively reflects the airway resistance, and ΔP2 reflects stress relaxation or the viscoelastic properties of the lung (Faffe and Zin 2009). Lung mechanics were measured 10–15 times in each animal.
Bronchoalveolar Lavage Fluid (BALF)
The right ventricle was perfused with saline to remove blood, and the BALF was obtained from all mice by injecting phosphate buffered saline (PBS) 3 consecutive times to a final volume of 1.5 mL in the right lung. The fluid was withdrawn and stored on ice. The total protein content in the samples was determined by the method of Bradford (1976).
BALF Analysis for IFN-γ and IL-6
IFN-γ and IL-6 levels were determined using a standardized ELISA technique—enzyme-linked immunosorbent assay kits (Quantikine, R&D Systems, Inc., Minneapolis, MN, USA) following the instructions of the manufacturer. All samples were measured in duplicate.
BALF Determination of Total Collagen
The presence of hydroxyproline (OH-proline) was determined using a colorimetric method based on the reaction of chloramine-T, perchloric acid, and p-dimethylbenzaldehyde. The color that developed was measured at 557 nm (Woessner 1961).
Tissue Collection, Histology, and Stereology
After BAL, the right lung was ligated, and the left lung in all mice was inflated by instilling 4% formalin buffer at 25 cm H2O pressure for 2 min, ligated, removed, and weighed. The inflated lungs were fixed for 48 h before embedding in paraffin. Serial sagittal sections were obtained for histological and morphometric analyses. To obtain uniform and proportionate lung samples, 18 fields (6 nonoverlapping fields in 3 different sections) were randomly analyzed using a video microscope (Zeiss-Axioplan—20× objective lens and JVC color video camera linked to a Sony Trinitron color video monitor; Carl Zeiss, Oberkochen, Germany) and a cycloid test-system superimposed on the monitor screen. The reference volume was estimated by point counting using the test points system (PT). The points that contacted the air spaces (PP) were counted to estimate the volume densities (Vv) of the structures (V = PP/PT) (Weibel et al. 2007). A total area of 1.94 mm2 was analyzed to determine the volume density of air spaces (Vv alveoli and septum) in sections stained with hematoxylin and eosin (H&E). Two investigators that performed the measurement counted non-identified sections. The morphometric method was adapted from Vlahovic et al. (1999). Collagen organization was evaluated in tissue sections stained with Sirius red observed under polarized light where thick collagen fibers are strongly birefringent and yellow to red, whereas thin collagen fibers are weakly birefringent and greenish.
Oxidative Stress Markers—Antioxidant Enzymes Assay
The left lungs were immediately removed and homogenized on ice with 10% (w/v) PBS (pH 7.3) using an Ultra-Turrax® T8 homogenizer (Toronto, Canada) and centrifuged at 600 g for 5 min. The supernatants were kept at –20ºC until analysis of the antioxidant enzyme activities. The total protein content in the lung homogenate samples was determined by the method of Bradford (1976).
The CAT, SOD, and GPx activities were determined in the lung homogenates. CAT activity was measured by the rate of decrease in hydrogen peroxide concentration at 240 nm (Aebi 1984). SOD activity was assayed by measuring the inhibition of adrenaline auto-oxidation by absorbance at 480 nm (Bannister and Calabrese 1987). GPx activity was measured by monitoring the oxidation of NADPH at 340 nm in the presence of H2O2 (Flohe and Gunzler 1984).
Oxidative Damage Assay—Malondialdehyde Assay
As an index of lipid peroxidation, we used the thiobarbituric acid-reactive substances (TBARS) method for analyzing malondialdehyde products during an acid-heating reaction, as previously described by Draper and coworkers (1993). Briefly, samples from lung homogenates were mixed with 1 mL of 10% trichloroacetic acid and 1 mL of 0.67% thiobarbituric acid; the samples were then heated in a boiling water bath for 30 min. TBARS levels were determined by absorbance at 532 nm and are expressed as malondialdehyde equivalents (nm/mg protein).
Nitrite Assay
Nitrite, a subproduct of NO metabolism, was measured using the Griess reaction (Valenca et al. 2009). BAL samples were reacted with 50 µL of 1% sulfanilamide solution for 10 min and mixed with 50 µL of 0.1% naphthyl ethylenediamine solution. Formation of the stable azo compound with a purple color was measured spectrophotometrically by absorbance at 540 nm. The method was standardized with known concentrations of nitrite. Nitrite concentrations are expressed in µMol/mg protein.
Western Blotting
Twenty micrograms of lung homogenate protein were subjected to electrophoresis on 8% PAGE gels and transferred onto nitrocellulose membranes (Amersham, Arlington Heights, IL, USA). The nitrocellulose membrane was blocked with 5% BSA and subsequently incubated overnight with rabbit polyclonal IgG SOD-3, CAT, and GPx 1/2 antibodies (1:1,000 dilution; Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA). After 3 washing steps (10 min each), the levels of protein were detected using a goat anti-rabbit antibody (1:10,000 dilution) conjugated to horseradish peroxidase (Dako Cytomation, Santa Barbara, CA, USA), and bound complexes were detected using the enhanced chemiluminescence method (ECL; Amersham). Molecular weight standards were used to identify the target band (Precision Plus Protein All Blue Standards, Bio-Rad Laboratories, Hercules, CA, USA).
Statistical Analysis
Survival rates were expressed as percentages, and a Mantel-Cox log-rank test (χ2 test) was used to detect differences in mice survival. Data are shown as the mean ± SEM. Following a normality test, parametric data were tested using one-way ANOVA and Tukey’s post-hoc test. Nonparametric data were tested using Kruskal-Wallis and Dunn’s post-hoc test. T-test was used for densitometric analysis of Western blotting bands. Statistical significance was defined as p < .05.
Results
Survival Rate
The survival curves of the groups are shown in Figure 1. There was no effect of bleomycin administration on the survival rate of BALB/c mice. Compared with BALB/c mice, higher mortality rates were found in both C57BL/6 and DBA/2 mice, which was represented by a gradual decline in the survival rates of the mice starting at 24 hours; there was a mice mortality rate of 50% in C57BL/6 and 20% in DBA/2 mice at the end of the experiment (n = 20 for each group).
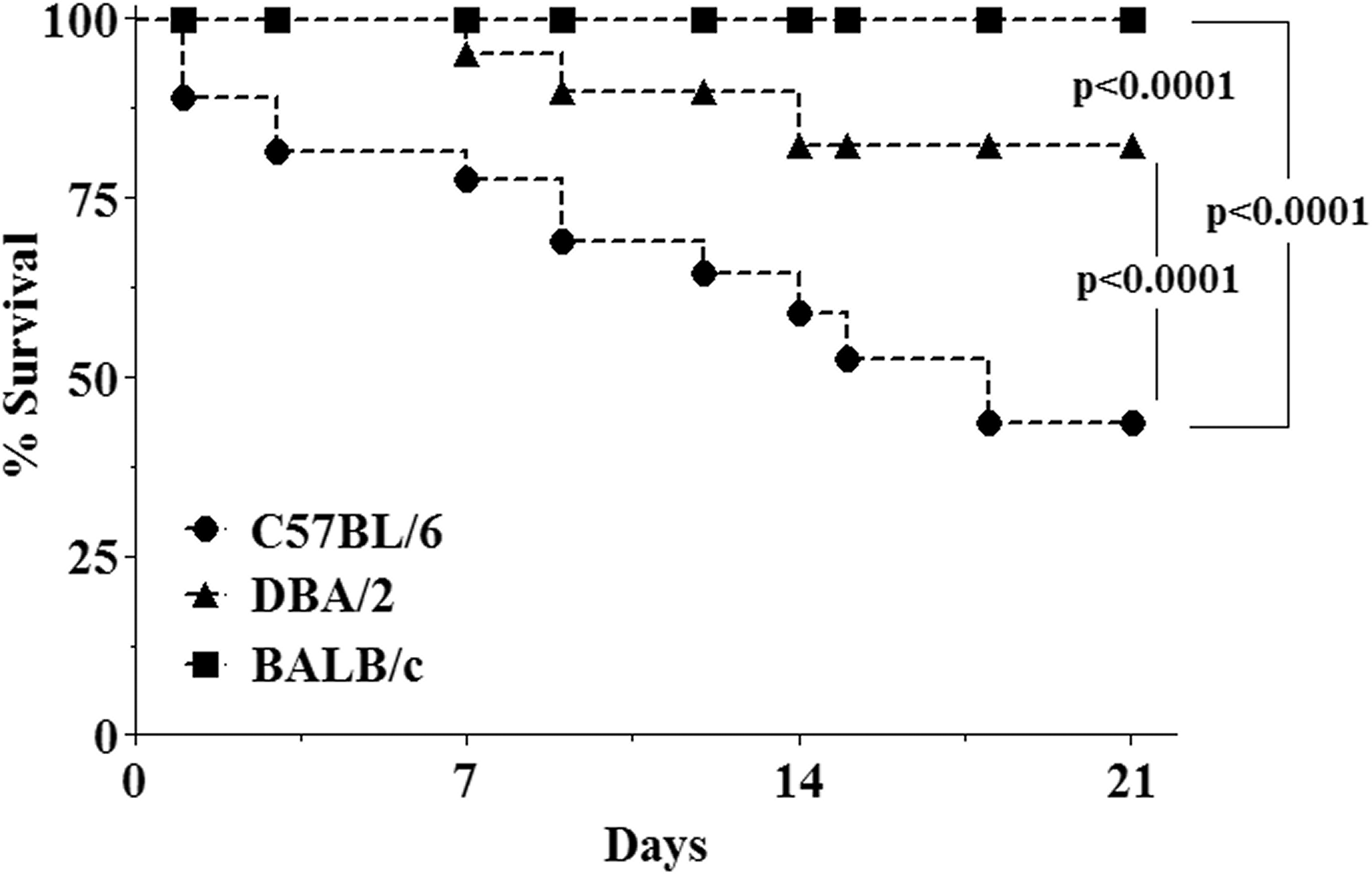
Survival curve of mice 21 days after intratracheal administration of bleomycin. The administration of bleomycin increased mortality in both C57BL/6 and DBA/2 mice. Survival rates are expressed as percentages, and the Mantel-Cox log-rank test (χ2 test) was used to detect differences in survival rates among mouse strains (p < .05). Dark squares: BALB/c strain; dark triangles: DBA/2 strain; dark circles: BALB/c.
Pulmonary Mechanics
The values for lung mechanics during spontaneous breathing for each group are shown in Table 1. C57BL/6 mice exhibited increases of 53% in Est (p < .001), 35% in ΔP2 (p < .05), 26% in ΔPtot (p < .01), and 35% in ΔE (p < .05) 21 days following bleomycin administration. No statistical differences in the respiratory mechanics were observed in bleomycin-instilled BALB/c and DBA/2 mice compared with the paired saline controls (n = 10 for each group).
Mechanical ventilation of mice 21 days following bleomycin administration (0.1 U/mouse).
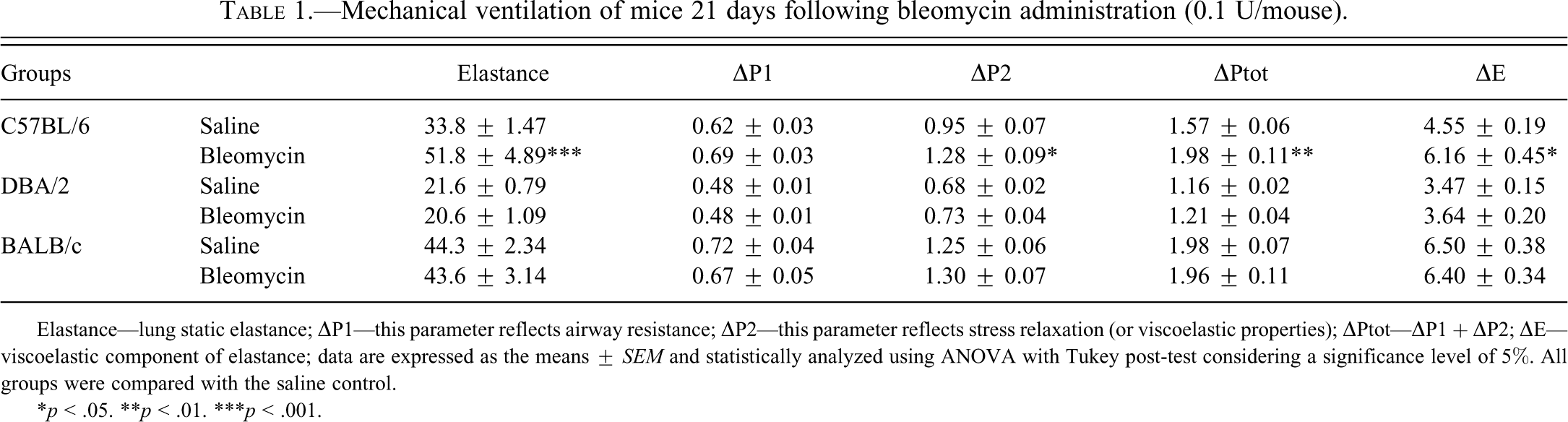
Elastance—lung static elastance; ▵P1—this parameter reflects airway resistance; ▵P2—this parameter reflects stress relaxation (or viscoelastic properties); ▵Ptot—▵P1 + ▵P2; ▵E—viscoelastic component of elastance; data are expressed as the means ± SEM and statistically analyzed using ANOVA with Tukey post-test considering a significance level of 5%. All groups were compared with the saline control.
*p < .05. **p < .01. ***p < .001.
Inflammatory Response
The inflammatory cells and levels of IFN-γ, IL-6, and OH-proline are presented in Table 2. The number of inflammatory cells increased in C57BL/6, DBA/2, and BALB/c mice 1 day following bleomycin administration (p < .001). The levels of IFN-γ were reduced by 43% in C57BL/6 mice (p < .01). There was a 76% increase in the IL-6 levels in C57BL/6 mice (p < .01), whereas the IL-6 levels were decreased by 40% in DBA/2 mice (p < .05). The OH-proline levels were increased in C57BL/6 and DBA/2 mice compared with the respective saline groups (112%, p < .05 and 104%, p < .05, respectively) (n = 10 each group).
Inflammatory cells, pro-inflammatory cytokines, and OH-proline from bronchoalveolar lavage of mice treated with the bleomycin 0.1 U/mice.
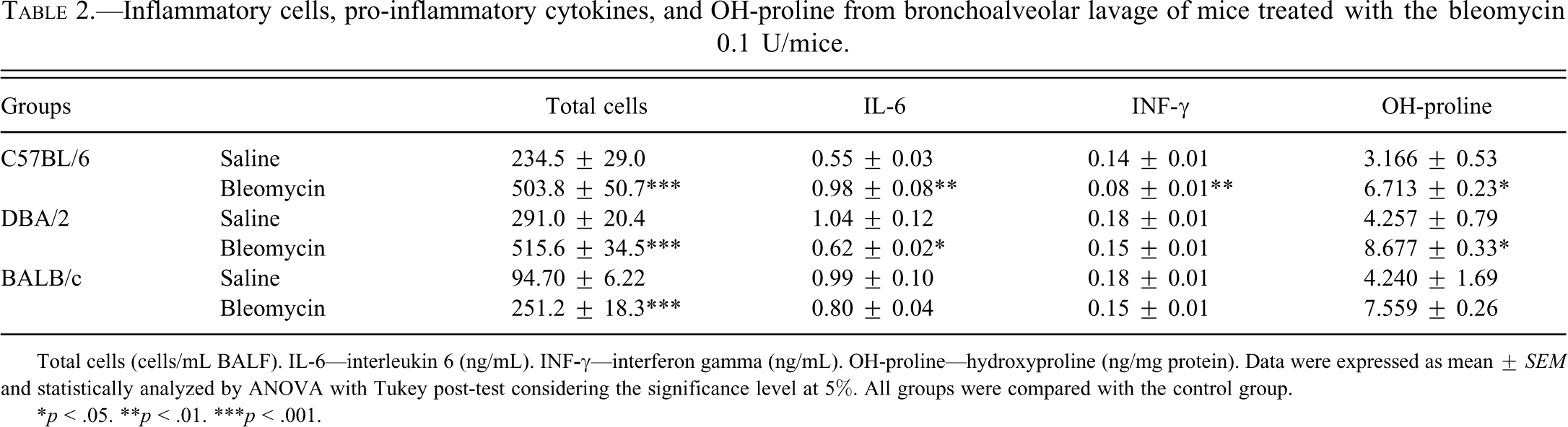
Total cells (cells/mL BALF). IL-6—interleukin 6 (ng/mL). INF
*p < .05. **p < .01. ***p < .001.
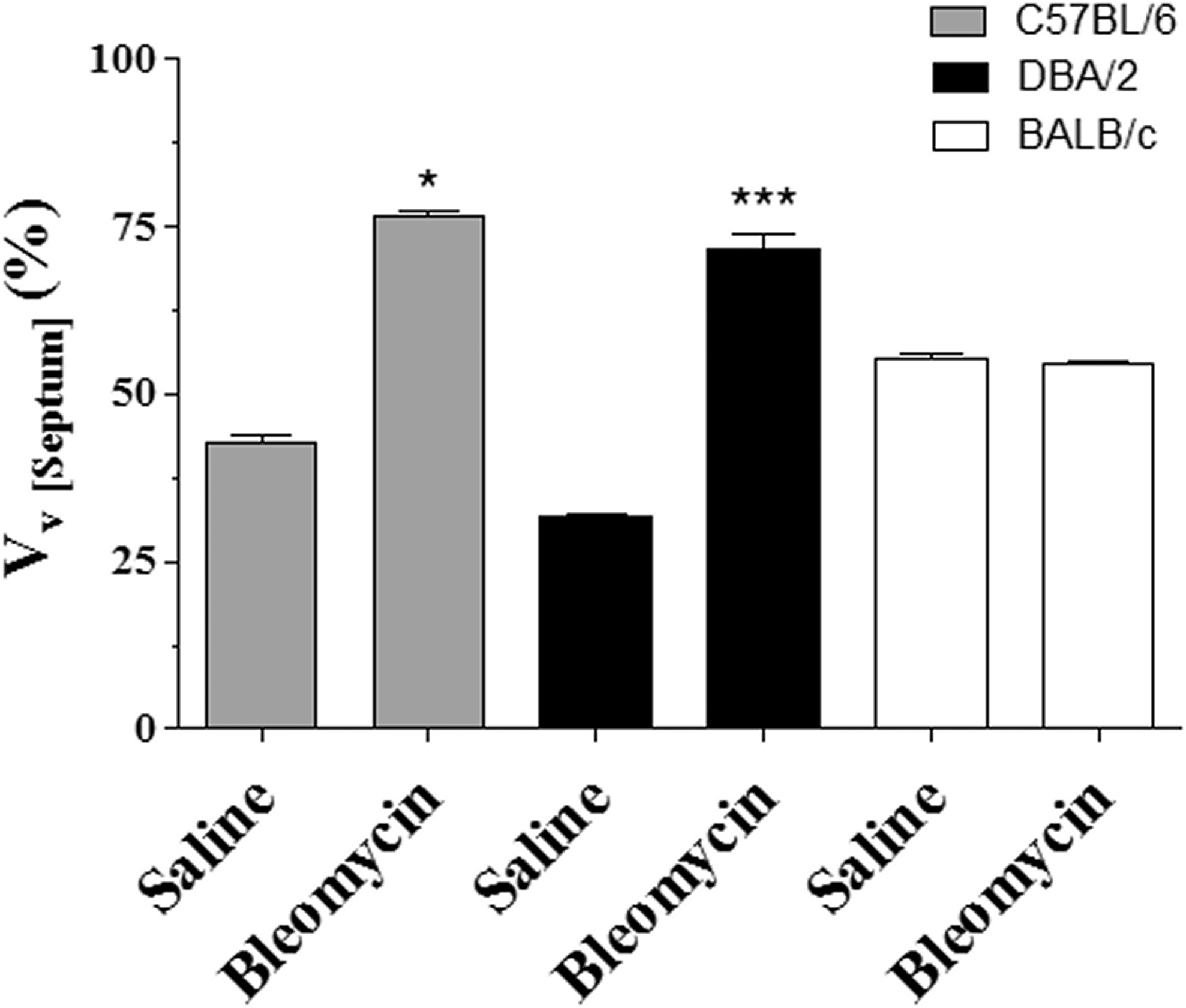
There was an increase in the extent of fibrosis in lung histological samples from both C57BL/6 and DBA/2 mice 21 days following bleomycin administration (Figure 2). The volume density of septa was increased by 79% (p < .05) and 126% (p < .001) (Figure 3A), and the volume density of alveoli was decreased by 59% (p < .05) and 58% (p < .001) (Figure 3B) in C57BL/6 and DBA/2 mice, respectively, compared with the saline groups (n = 10 for each group).
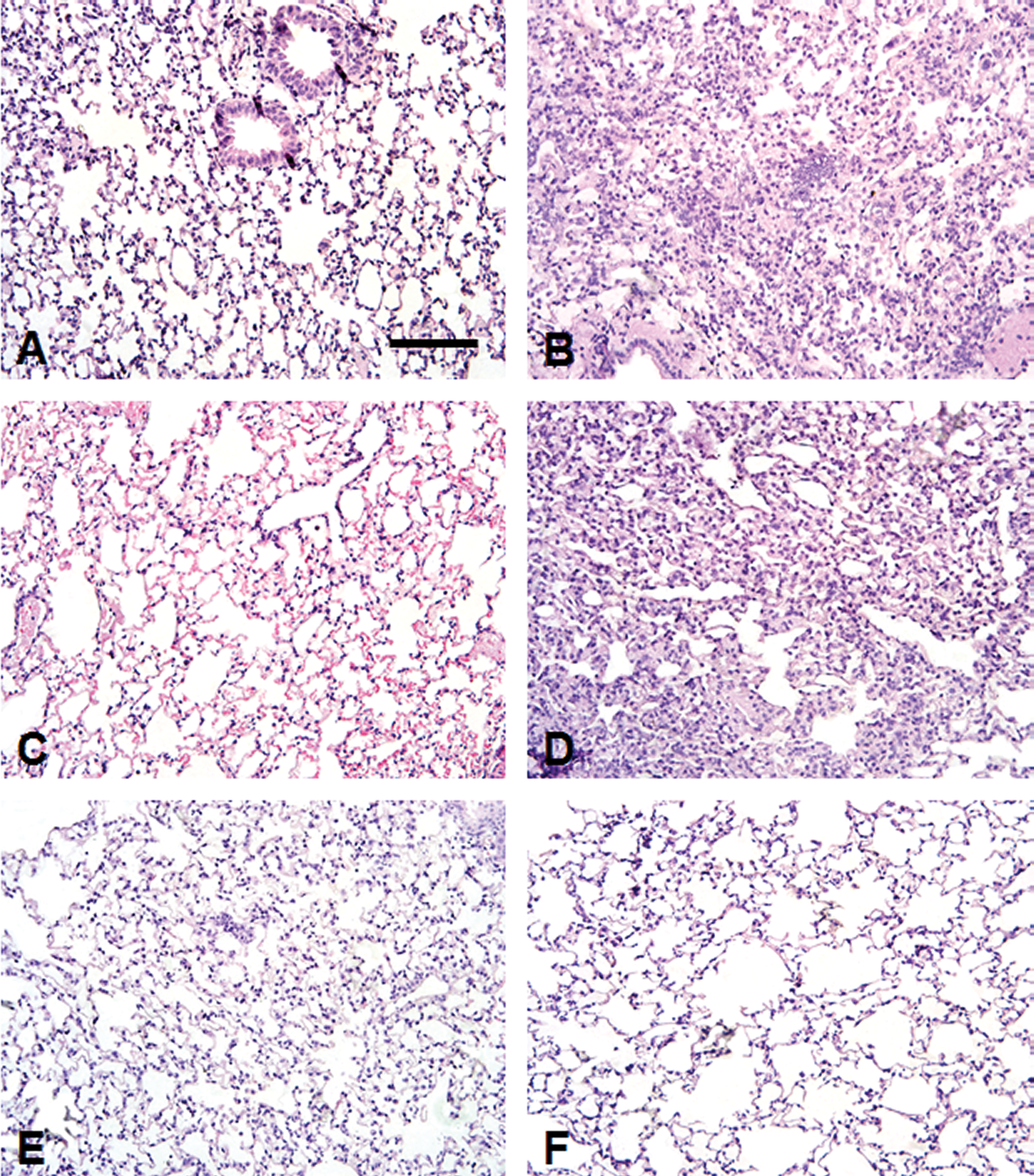
Photomicrographs of lung sections stained with hematoxylin and eosin from mice 21 days after intratracheal administration of 0.1 U of bleomycin in different strains (right column). Mice receiving saline were considered as controls (left column). A and B: C57BL/6; C and D: DBA/2; E and F: BALB/c. Extensive areas of fibrosis of the alveolar septa in both C57BL/6 and DBA/2 (B and D). There were no histological alterations in BALB/c following bleomycin administration. Bar 50 µm.
Stereological analysis of lung parenchyma. Histological changes were demonstrated by H&E staining (original magnification, X20). (
The hallmark characteristic of fibrosis is the excessive deposition of an extracellular matrix, such as collagen. In our experimental condition, bleomycin-induced excessive deposition of extracellular matrix proteins that were revealed by Red Picro Sirius in C57BL/6 (Figure 4B and 4D) mice and DBA/2 (Figure 4F and 4H). However, BALB/c mice showed no histological changes after administration of bleomycin (Figure 4J and 4M) with delicate collagens fibers in alveolar septa and in the wall of bronchiole.
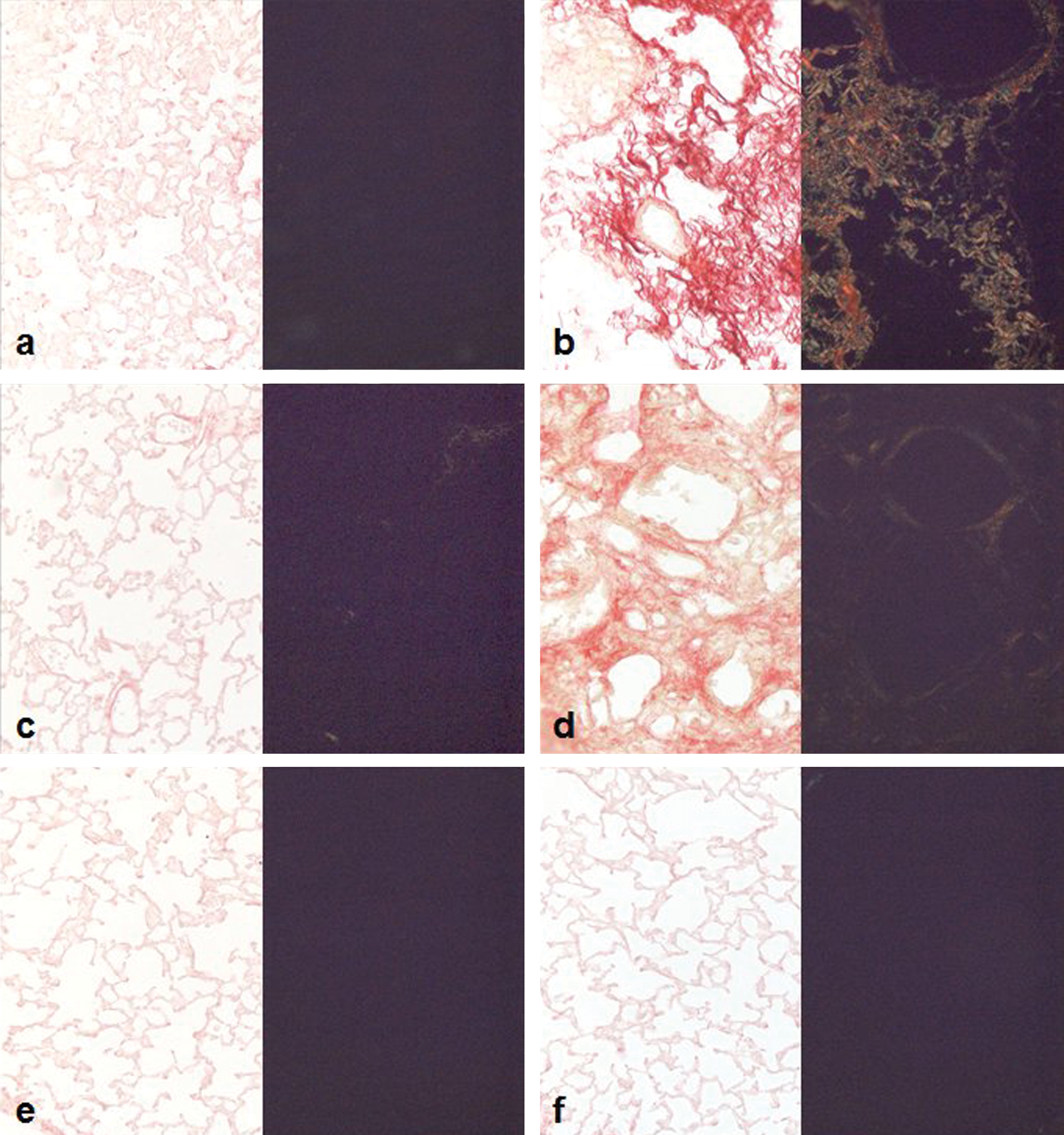
Lung photomicrographs of Sirius red stained sections (original magnification, ×20). Saline groups (a—C57BL/6; c—DBA/2, and e—BALB/c) with delicate collagens fibers in alveolar septa and in the wall of bronchiole and half view with polarized light. Lung sections 21 days after intratracheal administration of 0.1 U of bleomycin in different strains (b—C57BL/6 and d—DBA/2) with irregular collagen fibers in thickened septa, mainly in C57BL/6; half view with polarized light. There were no histological alterations in BALB/c following bleomycin administration (f). Bar 50 µm.
Activity of Oxidative Stress Markers
The activities of oxidative stress markers are shown in Table 3 (n = 10 for each group). A marked redox imbalance was observed in all strains studied. SOD activity was reduced in C57BL/6 and DBA/2 mice (76%, p < .001 and 24%, p < .001, respectively), whereas CAT activity was reduced in all strains 21 days following bleomycin administration compared with the saline groups (C57BL/6: 53%, p < .05; DBA/2: 33%, p < .01; BALB/c: 45%, p < .01). Although there was a reduction in GPx activity in C57BL/6 (31%, p < .05) and in BALB/c mice (26%, p < .01), there was an increase of 56% on the GPx activity in DBA/2 mice (p < .01) compared with the respective saline group. Nitrite levels were increased in C57BL/6 and BALB/c mice (129%, p < .001 and 150%, p < .001, respectively). Malondialdehyde (MDA) levels were increased in C57BL/6 (163%, p < .001) and were reduced in BALB/c mice (44%, p < .001) 21 days following bleomycin administration compared with the respective saline groups.
Biochemical analyses of oxidative stress and oxidative damage markers in lung tissue homogenates and bronchoalveolar lavage samples from mice 21 days following bleomycin administration (0.1 U/mouse).
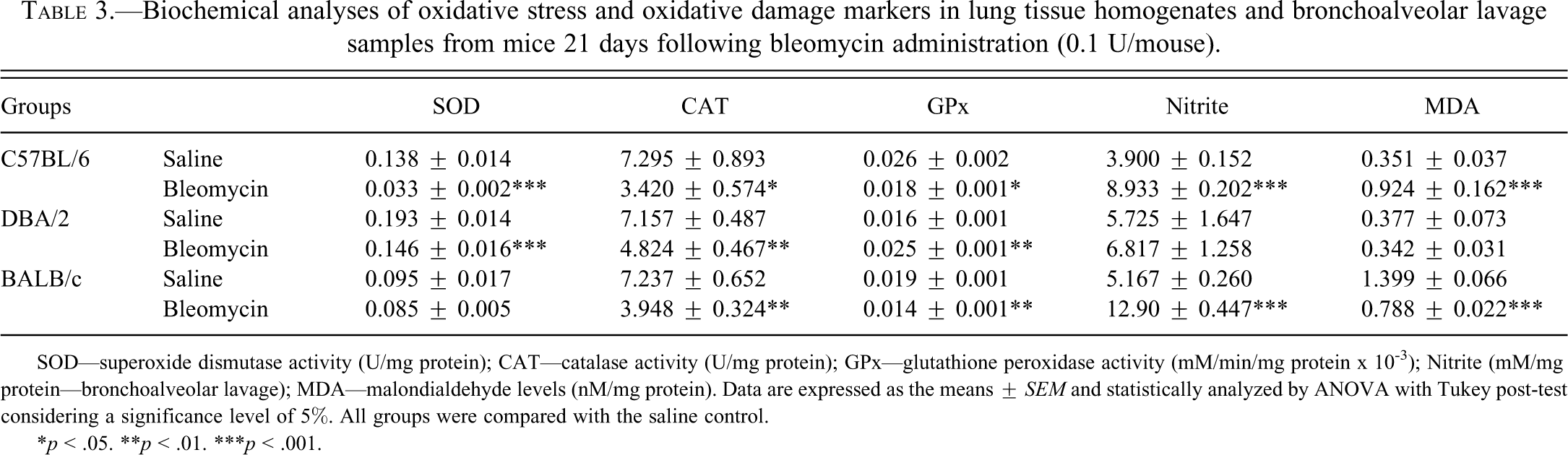
SOD—superoxide dismutase activity (U/mg protein); CAT—catalase activity (U/mg protein); GPx—glutathione peroxidase activity (mM/min/mg protein x 10-3); Nitrite (mM/mg protein—bronchoalveolar lavage); MDA—malondialdehyde levels (nM/mg protein). Data are expressed as the means ± SEM and statistically analyzed by ANOVA with Tukey post-test considering a significance level of 5%. All groups were compared with the saline control.
*p < .05. **p < .01. ***p < .001.
Expression of Oxidative Stress Markers
SOD-3 expression was decreased in both the C57BL/6 and DAB/2 groups, but SOD-3 expression was increased in the BALB/c group (Figure 5A). CAT expression was increased in the C57BL/6 group but was deceased in the DBA/2 and BALB/c groups (Figure 5B). GPx expression was decreased in both the C57BL/6 and BALB/c groups 21 days following bleomycin administration (Figure 5C).
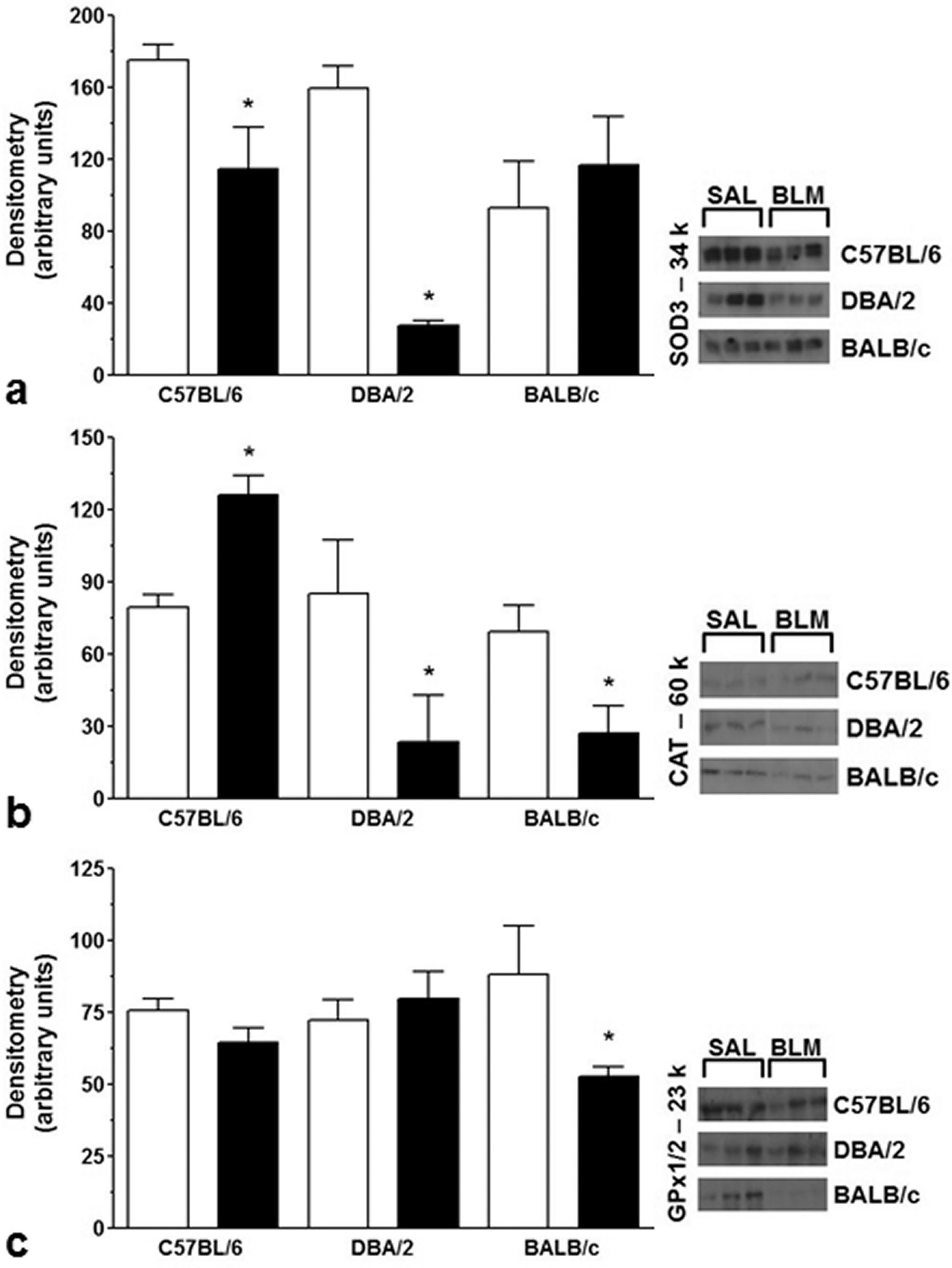
Expression levels of antioxidant enzymes in protein extracts from lung homogenates (Western blot analysis).
Discussion
Pulmonary fibrosis occurs following abnormal lung repair that results in pulmonary dysfunction (Pardo and Selman 2002). The mechanisms involved in the progression of pulmonary fibrosis include fibroblast activation and chronic inflammation (Meneghin and Hogaboam 2007). Oxidative stress has been associated with a number of diseases, including pulmonary fibrosis (Day 2008), as bleomycin, along with Fe2+, acts as is an exogenous source of oxidants due to Fenton’s reaction (Chen and Stubbe 2005). Studies have suggested that genetic background may play an important role in human PF susceptibility because only 10% of the patients undergoing chemotherapeutic regimens develop PF (Abid et al. 2001; du Bois 2002). Therefore, investigating the crosstalk between the redox profile, lung histology, and respiratory function in BLEO-PF in C57BL/6, DBA/2, and BALB/c mice may help to identify the missing links in the pathogenesis of PF.
Although C57BL/6 and BALB/c mice have been shown to be prone to oxidative damage in the lungs following long-term exposure to cigarette smoke (Rueff-Barroso et al. 2010), the oxidative parameters following bleomycin administration observed in the present study showed that BALB/c mice were resistant to oxidative damage, in contrast to the C57BL/6 and DBA/2 strains.
We showed that both SOD activity and expression are associated with the incidence of bleomycin-induced PF in mice. In the antioxidant enzyme system, SOD converts O2− into H2O2, whereas CAT and GPx convert H2O2 into water. Along with this system, the GSH/GSSG pair is thought to be the major redox buffer in the cell, as it controls not only the cellular level of H2O2 but also the levels of other organic hydroperoxides (Keller and Menzel 1989; Meister and Anderson 1983). Due to an impairment in the activity of antioxidant enzymes, increased oxidant production leads to lipid peroxidation, protein carbonylation, and DNA damage (Rahman and MacNee 1996). As lipid peroxidation products may result in dehydration and cell death, they have been considered to be important factors for the development of PF. Our results showed that MDA levels, which are representative of lipid peroxidation, were increased only in C57BL/6 mice. Increased lipid peroxidation has been described to be a direct oxidative product of hydroxyl radical attack in BLEO-PF (Sato et al. 2008). As previously described (Iraz et al. 2006; Kalayarasan et al. 2008), our results also show that the increased lipid peroxidation found in C57BL/6 mice was associated with reductions in the SOD, CAT, and GPx activities.
Three gene isoforms for SOD have been described in mammals: cytosolic SOD—SOD-1, mitochondrial SOD—SOD-2, and extracellular SOD—SOD-3 (Zelko et al. 2002). As SOD-3-deficient mice showed increased bleomycin-induced inflammatory responses in the lungs, extracellular matrix-located SOD seems to play an important role in protection against PF (Fattman et al. 2003). In the present study, C57BL/6 mice showed not only decreased SOD activity but also decreased SOD-3 expression in the lung. The decreased SOD activity observed in C57BL/6 mice 21 days after bleomycin administration could have resulted from enzyme inactivation by oxidants (De Raeve et al. 1997). As nitrite levels were found to be increased in C57BL/6 mice, O2− might have first been converted into peroxonitrite (ONOO−), as the rate constant is over 3.5 times faster than the dismutation of O2− by SOD (Dweik 2005). In addition, our data suggest that the increase in both SOD expression and activity might be associated with the resistance of BALB/c mice to PF.
Unlike SOD, CAT activities decreased in all groups following bleomycin administration; however, CAT expression was increased in C57BL/6 mice. The present data suggest that reduced SOD activity resulted in lower levels of hydrogen peroxide, which thus directly affected CAT activity, even though the expression of this enzyme was increased in C57BL/6 mice. In addition, reduced GPx activity impairs membrane phospholipid protection, which also results in lipid peroxidation, as observed in C57BL/6 mice.
Acute lung injury and normal repair of the alveolar-capillary membrane result in rapid restoration of tissue integrity and function following a variety of insults. Although acute inflammation and normal repair represent a complex interplay between humoral, cellular, and extracellular matrix networks, this process usually occurs in a sequential, yet overlapping, manner (Strieter 2008). The presence of myofibroblasts in patients with pulmonary fibrosis is documented in lung tissues and also in animal models of the disease (Phan 2002). It is established that fibroblasts transdifferentiate to myofibroblasts that express elevated levels of α-smooth muscle actin and, consequently, display a markedly enhanced ability to secrete extracellular matrix proteins (Phan 2002). Among several mediators, TGF-β is the major cytokine associated with pulmonary fibrosis; it is involved in the transition of fibroblasts into myofibroblasts (Strieter 2008), the synthesis of matrix proteins, and collagen degradation inhibition (Higashiyama et al. 2007; Moore and Hogaboam 2008). Studies have shown that a decrease in IFN-γ is associated with increased fibroblast activation and, therefore, increased extracellular matrix deposition, which leads to increased airway resistance in patients with idiopathic pulmonary fibrosis (Segel et al. 2003; Ziesche et al. 1999). In the present study, PF in C57BL/6 mice was associated with reduced levels of IFN-γ and increased levels of OH-proline. Moreover, C57BL/6 mice showed impaired pulmonary function and, for the most part, increased elastance and resistance, probably due to increased extracellular matrix deposition in mouse lungs (Faffe and Zin 2009).
In the present study, bleomycin administration induced PF and increased OH-proline in DBA/2 mice. Although the SOD and CAT activities were found to be reduced in these mice, no lipid peroxidation was observed, probably due to the elevation in GPx activity in these mice. In addition, DBA/2 mice showed no alterations in mechanical ventilation parameters, even though histological quantification showed clear indications of PF. Previous data have shown that a single intratracheal instillation of bleomycin resulted in reversible fibrosis in DBA/2 mice, whereas multiple doses resulted in persistent and prolonged fibrosis (Chung et al. 2003). The present data suggest that DBA/2 mice may be at an early stage of PF resolution or that the elastic components of the lung parenchyma have been preserved, as described by Manoury and coworkers, as pulmonary mechanics were unaltered 21 days following bleomycin administration (Manoury, Nenan, et al. 2006).
The present results also show that BALB/c mice, conversely, did not develop PF or show lipid peroxidation and showed no alterations in both IL-6 and IFN-γ levels and pulmonary mechanics parameters. Although the levels of TGF-β were not measured in this study, the lack of signs of fibrosis in BALB/c mice may be related to the maintenance of IFN-γ in these mice, as the production of IFN-y by Natural Killer T cells (NKT) and the subsequent production of TGF-β are pivotal for the development of BLEO-PF in mice (Kim et al. 2005).
BALB/c mice have been shown to present a significantly reduced number of myofibroblasts, which results in an inefficient production of collagen following wound repair processes (Cardoso et al. 2009). This may, in part, explain the observation that even following bleomycin administration, BALB/c mice did not develop PF.
As SOD acts as the first line of antioxidant defense in cells (Kaarteenaho-Wiik and Kinnula 2004), both CAT and GPx activities were probably reduced in BALB/c mice due to the absence of substrates (mainly H2O2). In addition, as the irreversible differentiation of myofibroblasts from fibroblasts is mediated by both ROS and TGF-β production through NADPH oxidase activation (Amara et al. 2010; Hecker et al. 2009), we suggest that this mechanism was active in BALB/c mice.
In addition, SOD has been described as a key enzyme in the process of BLEO-PF, as either intratracheal or inhaled administration of a hybrid phosphatidylcholine-SOD (PC-SOD: lecithinized superoxide dismutase) reduced collagen deposition in mouse lungs following bleomycin administration (Tanaka et al. 2010). Therefore, SOD seems to play a pivotal role in the development of BLEO-PF, as indicated by the maintenance of both SOD activity and expression in BALB/c mice.
The present results showed that MDA levels were reduced in BALB/c mice following bleomycin administration. As ROS, mainly H2O2, have been implicated in increasing cell death in the lungs of mice with BLEO-PF (Kasper and Barth 2009; Tanaka et al. 2010), the present data seem to indicate that the low susceptibility of BALB/c mice to oxidative damage protected pulmonary parenchyma.
Taken together, the present results suggest that PF development is associated with both SOD activity and SOD-3 expression in C57BL/6, DBA/2, and BALB/c mice following bleomycin administration.
Footnotes
This work was supported by grants from FAPERJ and CNPq. A.C.N. and M.L. received bursaries from CAPES. M.A.S.S. received a bursary from CNPq. S.S.V. was supported by the Visiting Professor Program of UERJ.
