Abstract
Cutaneous adverse drug reactions (CADRs) in patients are not uncommon, and they are difficult to predict from nonclinical safety studies. Nonhuman primates (NHPs) are predestinated for a high predictivity of adverse drug reactions, and we postulate that this may also be true for CADRs, if skin findings in NHPs are thoroughly worked up, following the diagnostic approach in clinical veterinary dermatology. This article proposes a systematic approach to describe, analyze, and report skin findings that occur in NHP toxicity studies. Implementing this approach may increase the likelihood to differentiate between test item–related cutaneous findings and those that are independent of the test item. This will eventually result in increased relevance of skin findings identified in the scope of an NHP regulatory toxicity study for the risk assessment process to safeguard patients in clinical trials and beyond.
Keywords
Introduction
Cutaneous adverse drug reactions (CADRs) in patients are not uncommon. Among hospitalized patients, the incidence of adverse drug reactions concerning the skin ranges from 1% to 3% (Svensson, Cowen, and Gaspari 2000), but the actual prevalence is much higher, as many mild forms of CADRs do not become reported. CADRs can show a high variety in clinical presentation and in clinical severity, ranging from mild focal erythema to severe—even life-threatening—extensive epidermal necrosis. Understandably, the underlying pathogenesis is manifold and often incompletely understood.
It is a general goal of nonclinical safety studies conducted with medicinal products under development to predict adverse reactions to a drug in patients. While there is controversy on how predictive general toxicity studies in animals are (Olson et al. 2000; Shanks, Greek, and Greek 2009), it has been demonstrated that such studies are particularly weak in predicting CADRs (Olson et al. 2000). This generally accepted dogma is possibly related to the differences in skin anatomy and physiology between rodents and humans, but probably a major factor to this is the insufficient rigor and knowledge of terminologic standards that is applied to the clinical and pathological documentation and management of skin findings.
Nonhuman primates (NHPs) are frequently used as a relevant species for the nonclinical safety evaluation of medicinal products under development. Their close proximity to humans in terms of phylogeny and gene or protein homology predestinate them for a high predictivity of adverse drug reactions. We postulate that this may also be true for CADRs, if skin findings in NHPs are thoroughly worked up, following the diagnostic approach in clinical veterinary dermatology.
This article proposes a systematic approach to describe, analyze, and report skin findings identified in the scope of an NHP regulatory toxicity study. Implementing this approach should increase the likelihood to differentiate between test item–related cutaneous toxicity and cutaneous findings that are unrelated to the test item. This would eventually result in increased relevance of an NHP study for the risk assessment process to safeguard patients in clinical trials and beyond.
Description of Grossly Visible Skin Findings
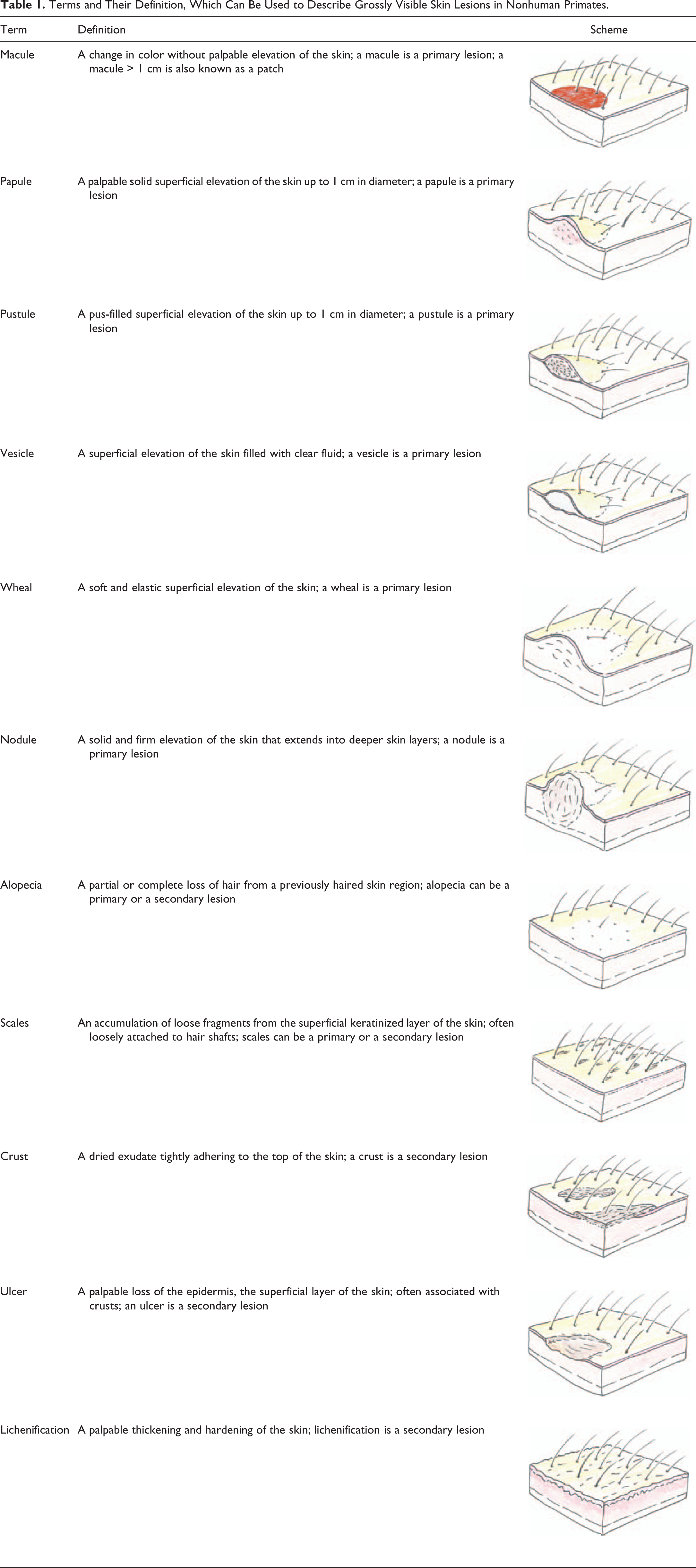
The use of a reproducible, coherent, and professionally recognized terminology is of paramount importance for the description of grossly apparent skin abnormalities. Experience has shown that skin changes in NHP toxicity studies, as well as in studies with other species, are described with a wealth of heterogeneous terms, mostly used by technical staff with no medical training and not sufficiently trained in clinical veterinary dermatologic diagnostics and terminology. This leads to study reports containing large tables of clinical observations which do not allow an appropriate evaluation, correlation, or interpretation by pathologists or toxicologist. In order to be reproducible, the terms that are used to describe grossly visible skin lesions need an accurate definition. Standard definitions need to be used by the staff who are involved in recording of lesions. Experience has demonstrated that iterative training is required if the use of such terminology is to be kept consistent over time. The literature on clinical dermatology, particularly veterinary dermatology, provides a comprehensive list of terms that are routinely used among clinical dermatologists to describe morphologic characteristics of skin lesions (Miller, Griffin, and Campbell 2012). It is recommended that every laboratory conducting NHP toxicity studies establishes a list of such standard terms that are to be used by the staff inspecting the animals. As mentioned above, training is needed to assure that the terminology is appropriately used. Consideration should be given to the concept that some morphologies represent primary skin lesions (i.e., the initial eruption of a lesion), while others represent secondary lesions (i.e., lesions that evolve from primary lesions by external factors such as self-induced trauma or bacterial infection). Some further skin lesions may represent either primary or secondary lesions. Table 1 contains a list of terms and their definition, which can be used to describe the vast majority of grossly visible skin lesions in NHPs.
Terms and Their Definition, Which Can Be Used to Describe Grossly Visible Skin Lesions in Nonhuman Primates.
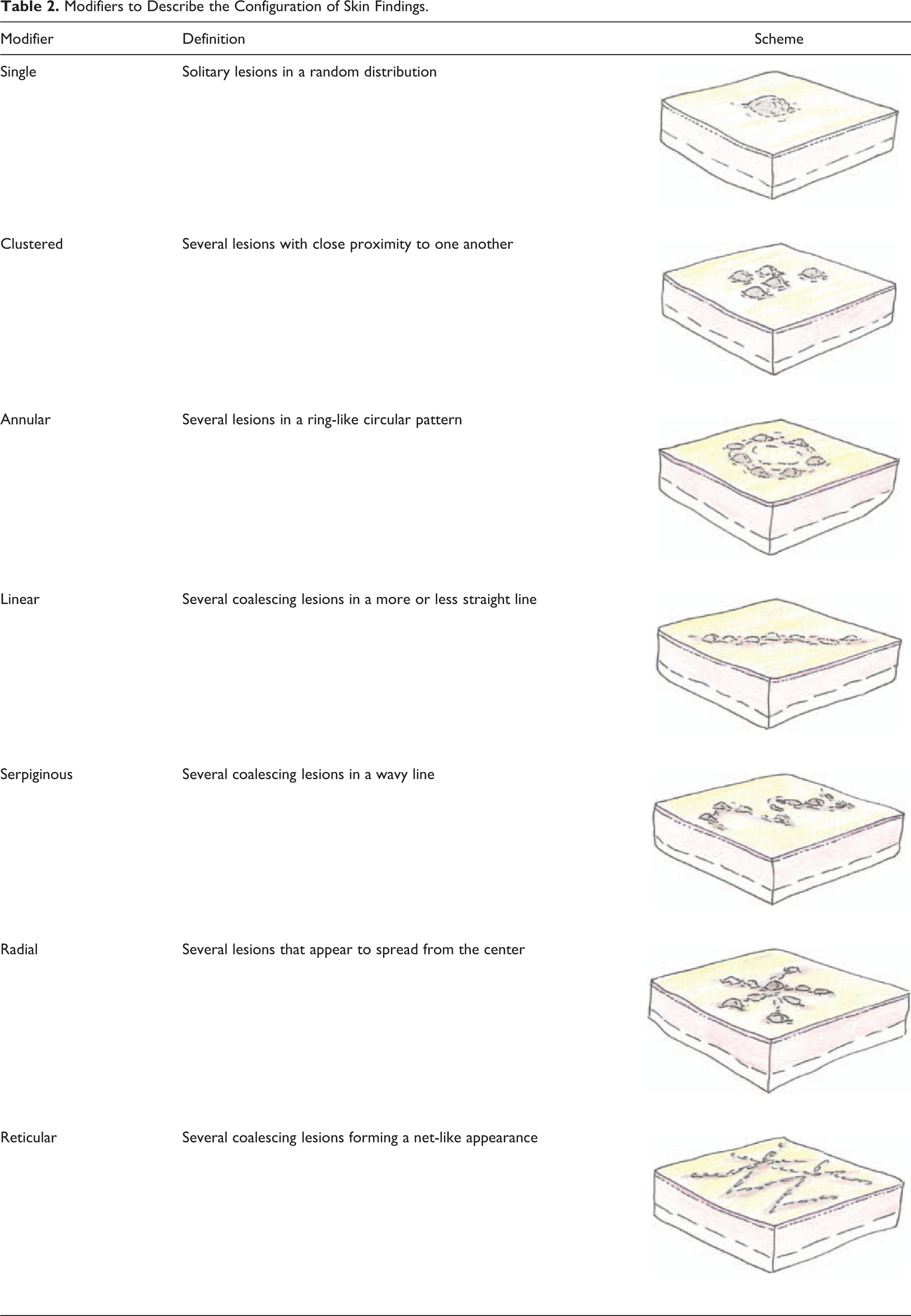
The above terms represent only one aspect of the cutaneous abnormality, but the description remains incomplete without modifiers. Modifiers should describe the configuration of the lesion (Table 2), the size of the lesion (given in mm), and the precise anatomical localization of the lesion. In order to assure a consistent use of anatomic terms, it is recommended to provide all staff involved in the description of skin findings with a schematic depiction of anatomic sites. An example is shown in Figure 1.
Modifiers to Describe the Configuration of Skin Findings.
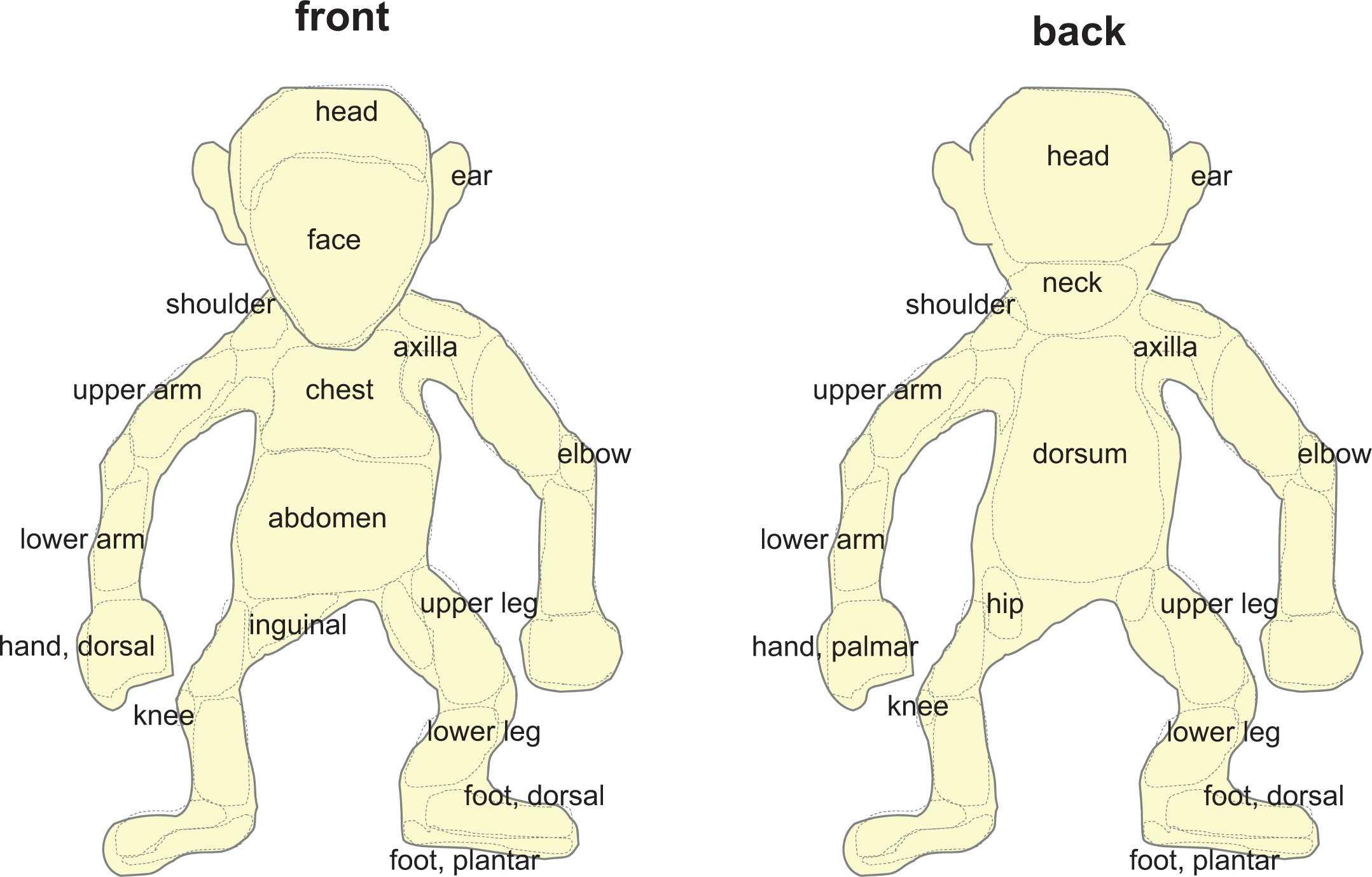
Schematic depiction of anatomic sites in nonhuman primates.
Surveillance and Advanced Examination of Skin Findings
Animals in toxicity studies are grossly inspected at regular time intervals as stated in the study protocol. Every effort should be undertaken to ensure that skin findings are described in a consistent manner. This includes the evolution of the descriptive terminology over time within a given study, as skin lesions tend to evolve rather rapidly due to secondary factors (e.g., trauma, infection, etc.; Ackermann 1999). Consequently, it is to be expected that a primary lesion, for example, a vesicle, eventually ruptures and forms an ulcer and/or a crust.
While some cutaneous observations may just represent common background findings in NHPs with no further implication for the study or the animal’s health (e.g., focal alopecia at sites of rubbing or hair loss associated with aging), the majority of findings will require inspection by a veterinarian. The respective veterinarian should be trained in clinical veterinary dermatology and should be able to conduct advanced clinical examinations such as skin scrapings, trichograms, harvesting of smears for cytological examination, or sampling for bacterial or fungal cultures. This should ensure rapid and accurate diagnosis of infectious diseases such as pyoderma or dermatophytosis which, although rare, may occur in captive macaques (Baker, Bradford, and Montes 1971; Pal et al. 1991; Novak and Meyer 2009). It will also ensure diagnosis of spontaneous viral diseases in macaques which can affect the skin (see below).
Although rarely conducted, the collection of skin biopsies during the in-life phase of an NHP toxicity study should be taken into consideration, particularly if skin lesions are uncommon and unclear with regard to their pathogenesis and relevance. As alluded to before, the gross morphology of cutaneous findings changes over time and so does the histopathology associated with it. Timing of skin biopsies is therefore an important aspect that should be discussed in close collaboration between the study director, the veterinarian in charge of the animal’s health, and the study pathologist. Details on how to collect biopsies are given below. Since the electronic data capture systems that are routinely used in large-scale Good Laboratory Practice (GLP)-accredited laboratories are usually not set up to include biopsy collection during the in-life phase of a study, consideration should be given how to document and report the sampling and the histopathological results obtained on these biopsies.
Gross Pathology and Sampling for Histopathology
The skin is a standard protocol organ that is to be sampled for histopathological analysis (Bregman et al. 2003). Such sampling is conducted in a standardized manner, and in most organizations conducting NHP toxicity studies, the skin that is histologically examined is sampled from the abdominal skin. While this sampling pattern might be considered appropriate for routine studies with no cutaneous findings, it certainly is insufficient for any animal in which grossly visible skin findings have been recorded. Any skin finding that has been observed during the in-life phase of a study should be verified for its presence at the time of necropsy. Naturally, this requires that the anatomic localization of the finding is precisely documented and that this documentation is available to the personnel conducting the necropsy.
It should be general consensus that any skin finding should be sampled for histopathology. This requires that the necropsy personnel is familiar with the technique how to sample diseased skin. In principle, there are two different techniques of sampling skin for histopathology: one is a wedge-shaped excisional sample (Figure 2). This technique should be routinely used for the majority of skin lesions. It is important to orient the sample in a way that it contains a portion of normal unaffected skin, the margin of the lesion, and ideally the center of the lesion. If skin findings imply involvement of deeper layers of the skin (such as in nodules or some wheals), caution should be taken that the skin samples taken at necropsy contain sufficient subcutaneous fat and fascial soft tissue. If alopecia is the primary character of the lesion, a sample should contain both haired and nonhaired skin, and it should be oriented in a way that the longitudinal site is parallel to the orientation of hair shafts emerging from the skin (Figure 2).
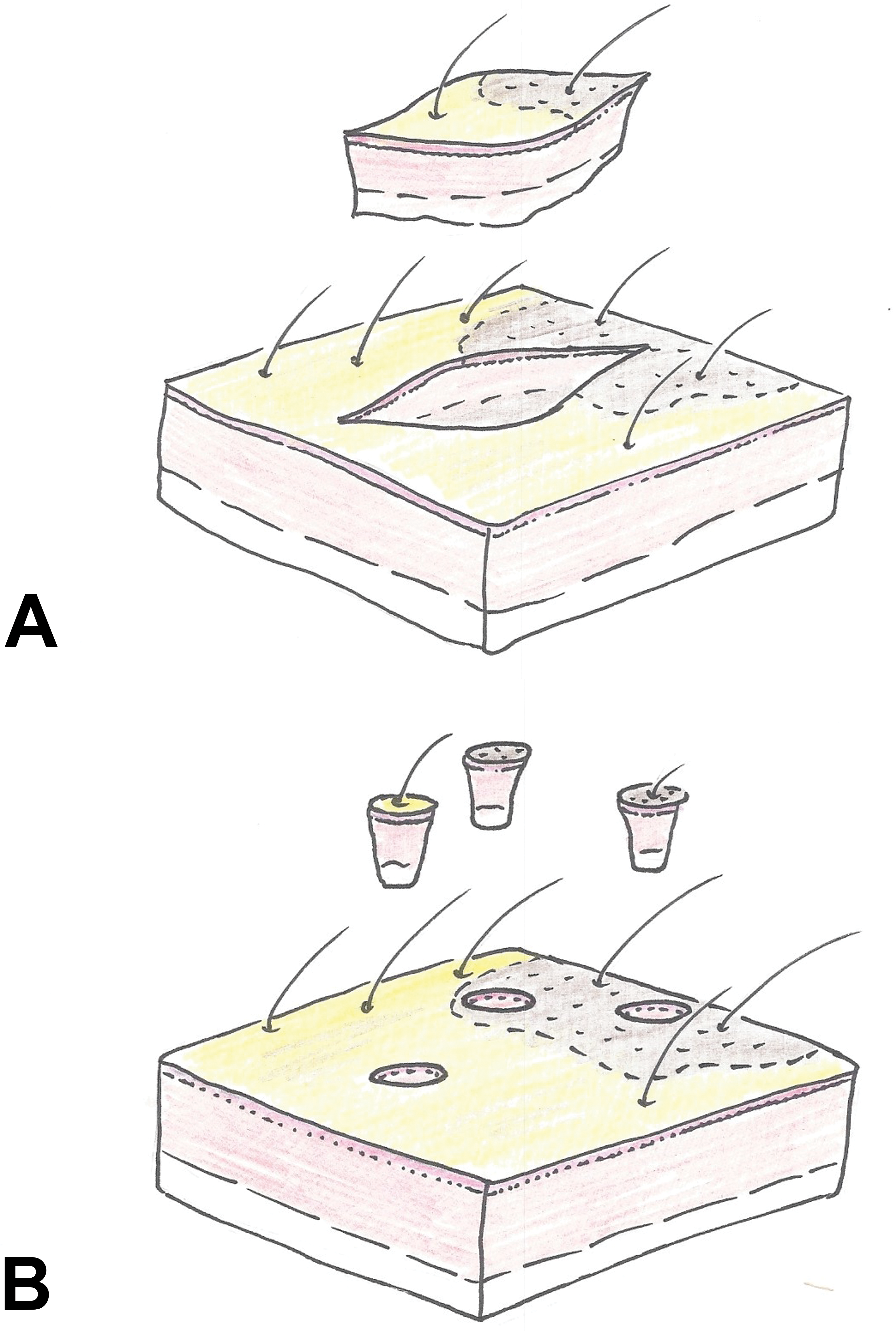
Schematic representation of sampling techniques. (A) Wedge-shaped excisional skin sample. (B) Punch biopsy. Consideration should be given to always sample the center of a lesion, the periphery, and adjacent unaffected skin. The longitudinal axis of wedge-shaped samples should be oriented in the same direction as the hair shafts emerging from the skin.
The second technique to obtain skin samples is the use of round biopsy punches (Figure 2). These come at different sizes, and 6- or 8-mm punches have proven most useful in a clinical and a necropsy setting. Punch biopsies should be obtained when lesions are very small or otherwise difficult to excise. Punch biopsies are also the gold standard with regard to skin sampling during the in-life phase of a study or in conditions where several samples of similar lesions need to be obtained (e.g., if biopsies from the same area need to be fixed differently for different analytical methods). Caution should be given to orient the punch in a way that the lesion is in the center of the biopsy. Whatever technology is used for sampling skin lesions, it needs to be stressed that adequate controls should be included. This often requires sampling of adjacent nonaffected skin or skin from the same anatomical region of the contralateral site.
The sampling of skin lesions at necropsy needs to be thoroughly documented, not only because this is required by GLP. It is of paramount importance that the pathologist analyzing the skin samples understands their anatomic origin. This is crucial to enable correlation between the histopathological findings, the gross observations, and those recorded during the in-life phase. We have made the experience that the data recording systems, which are routinely used in large GLP-accredited laboratories, may represent an obstacle as they only contain one protocol organ designated as “skin.” We have found it useful to include nonprotocol organs named Skin 1, Skin 2, and so on, into the glossaries and to make annotations during necropsy what samples were assigned to these organs.
Histopathology
Pathologists are trained to recognize specific patterns related to pathogenetic mechanisms and draw conclusions from them. While the analysis of patterns is a rational approach in all fields of pathology, it is particularly used in dermatopathology to classify morphological findings. The majority of skin diseases can be diagnosed with specificity using the scanning objective (1.0–2.5×) of a conventional microscope (Ackermann 1999). Most pathologists, who nowadays evaluate slides from NHP toxicity studies, have undergone a thorough education in veterinary diagnostic pathology and are well familiar with the specific terms that are used in dermatopathology. The reader is referred to standard texts on dermatopathology and to the nomenclature suggested by the International Harmonization of Nomenclature and Diagnostic criteria (INHAND) project (Mecklenburg et al. 2013), which can be used for NHPs as well. While there are individual preferences regarding the splitting or lumping of terms, a thorough description of cutaneous histopathological findings will quickly result in a large amount of terms that show up in summary tables of the pathology report. For the sake of readability and clarity, it is recommended to merge terms where appropriate, either in the original data recording or in a separate table to be included in the anatomic pathology report. It is the study pathologist’s responsibility to provide clarity about the correlation of cutaneous histopathological findings to cutaneous gross lesions at necropsy and to skin findings described during the in-life phase of the study.
Interpretation of Results
The interpretation of cutaneous findings in NHP toxicity studies requires some knowledge about the nature of CADRs in patients. While a detailed description of CADRs is beyond the scope of this article, the following principles should be taken into consideration:
Direct cutaneous toxicity, that is, toxicity originating from a physical or chemical insult to the skin, is generally separated from indirect toxicity, that is, toxicity that is mediated via an immune reaction. The latter can be divided into immediate reactions (urticaria) and delayed reactions. The most common types of CADRs in patients are irritant contact dermatitis (ICD) and allergic contact dermatitis (ACD). Both are very similar in terms of gross characteristics and histopathology, being characterized by erythematous (red) macules and vesicles with underlying intercellular edema in the epidermis and superficial dermal inflammation (Ale and Maibach 2008). While ICD occurs rapidly after the insult mediated via the release of preformed mediators from injured cells, ACD is a type IV hypersensitivity reaction with delayed onset. Urticaria is another common CADR with immediate onset characterized by the occurrence of wheals. Other forms of immune-mediated CADRs are less common and are generally classified according to their clinical pattern (Goncalo and Bruynzeel 2008). Examples are drug-induced rash with eosinophilia and systemic symptoms (Cacoub et al. 2011), acute generalized exanthematic pustulosis (Saissi et al. 2003), erythema multiforme (EM), fixed drug eruption, Stevens–Johnson syndrome (SJS), and toxic epidermal necrolysis (TEN; Yager 2014). EM has been described in many species other than man, and it has been seen in cynomolgus macaques (personal observation). EM is likely mediated by the deposition of immune complexes within the microvasculature, and it involves skin as well as mucous membranes. EM minor is restricted to less than 10% of the skin of the extremities, while EM major affects extremities with more than 10% of epidermal detachment. SJS is a severe form of EM with the trunk and face being affected in addition to the extremities, and TEN is a life-threatening condition in which more than 30% of the epidermis are necrotic and detach. The histological pattern of EM and TEN has been well described in the veterinary dermatopathology literature, and descriptions of the other conditions can be found in human case reports.
Drug-induced cutaneous findings in NHP toxicity studies need to be differentiated from spontaneous skin diseases in NHPs. This is difficult and in some cases a clear distinction may be impossible, particularly since the description of spontaneous skin diseases in NHPs is still fragmentary. The presence of specific antiviral antibodies in serum (e.g., measles virus) may not have any relevance for the current skin condition (personal observation). The pathologist should be aware of viral diseases that may cause skin lesions in NHPs: macaques in captivity might become infected with the human measles virus through contact with humans carrying this virus. The virus is highly contagious through aerosolization and may induce immunosuppression in macaques, followed by various secondary opportunistic infections such as candidiasis or cytomegalovirus infection. One typical clinical feature of human measles virus infection in macaques is the occurrence of well-circumscribed erythematous macules on the abdomen. These macules may be associated with scales, correlating to epidermal necrosis and orthokeratotic hyperkeratosis in histology (Choi et al. 1999). Simian varicella virus is a naturally occurring herpesvirus of Old World primates responsible for sporadic epizootics in biomedical research facilities (Hukkanen et al. 2009). Infection is associated with erythematous macules, papules, and vesicles on the abdomen that might spread to the proximal hind legs and/or the oral mucosa. The typical lesion shows a radial configuration with a centripetal spread. Histology will show epithelial necrosis associated with amphophilic to eosinophilic intranuclear inclusion bodies. Simian immunodeficiency virus causes progressive immunosuppression in macaques. In addition, infection is frequently associated with erythematous macules and papules, often affecting the axillary skin (Wilk, Lewis, and Lukas 2008). Orthopoxvirus infections in monkeys (monkeypox) can cause multiple papules, which are characterized by epidermal proliferation and necrosis, associated with typical eosinophilic intracytoplasmic inclusion bodies. These lesions eventually result in ulcers.
Reporting
The importance of a consistent use of appropriate terminology has been emphasized before. In addition to the accuracy of data, its presentation in a report determines its applicability in a risk assessment for patients. It is widely accepted that tables are most useful in providing an overview about complex data sets. However, images can transport even more complexity than can tables, and according to the dogma, “a picture is worth a thousand words,” one should consider to use images when communicating the character, configuration, distribution, and incidence of cutaneous findings in NHP toxicity studies. An example is given in Figure 3.
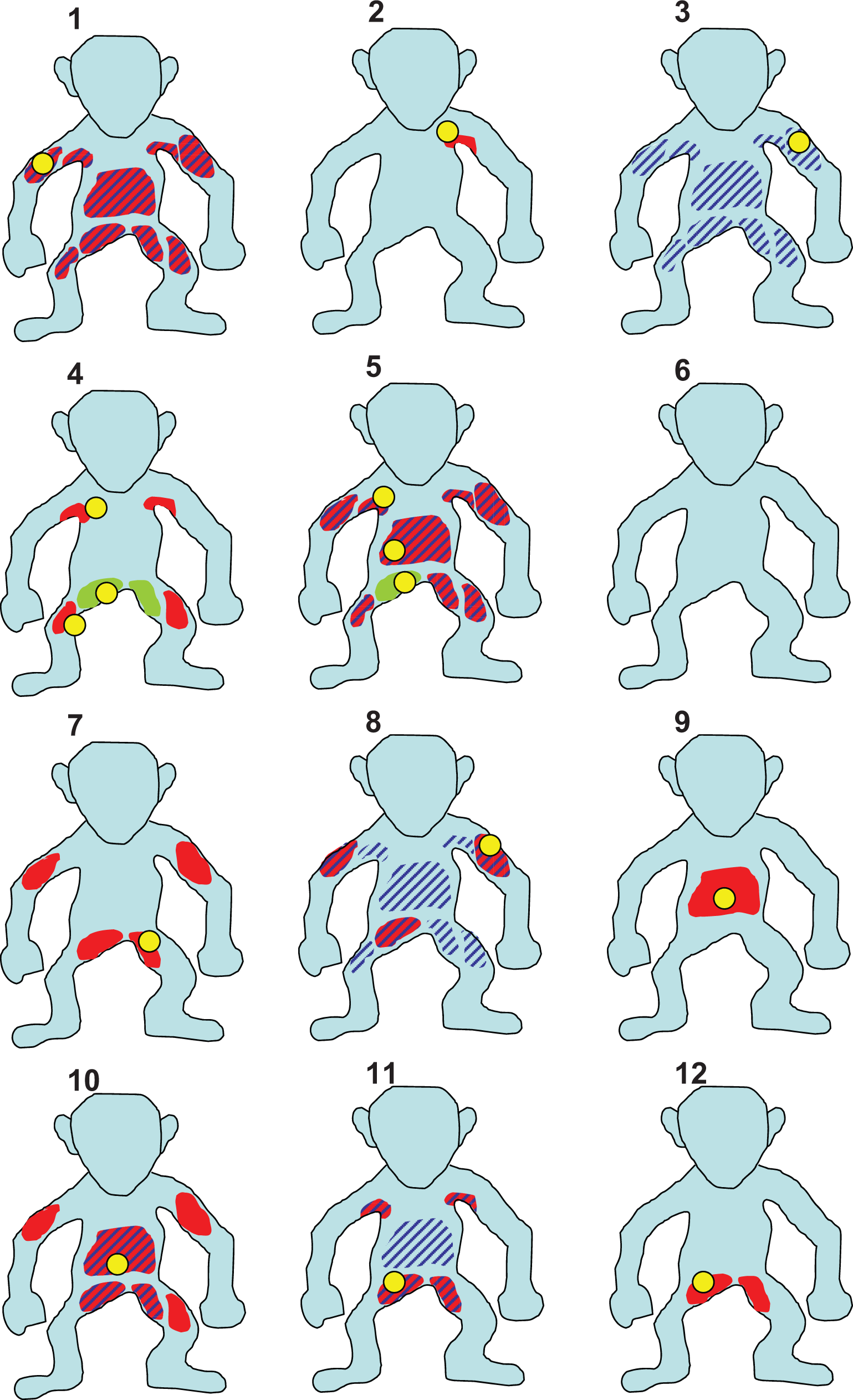
Example of how to illustrate character and distribution of skin findings in relation to the site of sampling. Yellow: site of sampling, red: erythema, green: pustule, stripes: scales.
Summary and Conclusion
Toxicity studies in NHPs represent an opportunity to identify and characterize potential CADRs in patients. However, in order to detect such lesions, a systematic diagnostic approach is needed comparable to the procedures in clinical veterinary dermatology. The goal is to detect and characterize grossly visible skin findings, to monitor and further analyze them throughout the in-life phase of a study, to document and sample them thoroughly at necropsy, and finally to correlate them with histopathological findings. A stringent approach as is suggested in this article will assist in the interpretation of skin lesions encountered in NHP toxicity studies and will eventually provide added value to the safety assessment of medicinal products under development.
Footnotes
Authors’ Contribution
Authors contributed to conception or design (LM, AR); data acquisition, analysis, or interpretation (LM, AR); drafting the manuscript (LM); and critically revising the manuscript (AR). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
