Abstract
The skin irritating, sensitizing, and acute dermal toxicity potential of a novel combinational prophylactic transdermal patch, mainly composed of eserine and pralidoxime chloride as active pharmaceutical ingredients, against (±) anatoxin-a poisoning were investigated in rabbits, guinea pigs, and rats in compliance with the Organisation for Economic Cooperation and Development guidelines. In primary skin irritation test, rabbits were dermally attached with the therapeutically active transdermal patch or with a placebo patch for 72 hours. The transdermal patches did not induce any adverse reactions such as erythema and edema on intact skin sites. The active patch was classified as a practically nonirritating material based on the score in the primary irritation index. In the Buehler test, guinea pigs were sensitized by the active or placebo transdermal patches attached for 24 hours. The patches did not induce any sensitization reactions in contrast to a severe sensitization reaction that occurred in the positive control. Therefore, the active patch and placebo patch were both graded as weak in sensitization score and rate. Acute dermal toxicity test in rats did not produce any overt signs of toxicity following a 14-day treatment period. Taken together, these findings suggest that the transdermal patch does not cause skin irritation, skin sensitization, or dermal toxic effects following dermal application.
Introduction
(±) Anatoxin-A, a guanidinemethyl phosphate ester isolated from the freshwater cyanobacterium (blue-green algae), is a potent irreversible inhibitor of acetylcholine esterase (AChE). (±) Anatoxin-A has been shown to be an active site-directed inhibitor of AChE, which is resistant to reactivation by oxides because of the enzyme–oxime adduct formation. In vivo pretreatment with eserine and high concentrations of pralidoxime chloride were the only effective antagonists against a lethal dose of (±) anatoxin-A poisoning. 1 For this reason, eserine has been suggested as an alternative prophylactic against AChE poisoning, because it is an unquaternized carbamate that penetrates the central nervous system. 2 However, eserine has a short plasma half-life and a narrow therapeutic index, which necessitate the use of a sustained release formulation. 3 Hence, we developed a sustained release combinational prophylactic transdermal drug-in-adhesive matrix-type therapeutic system containing eserine and pralidoxime chloride (2-PAM) for the delivery to the skin.
The human skin is the major organ of exposure to transdermal therapeutic system, because adhesion of such materials to the superficial organs has the potential to be a major route of exposure during the application, use, and disposal of such therapeutic system. In this study, the prophylactic transdermal patch composed of eserine, 2-PAM, and inactive ingredients was tested for acute dermal irritation in rabbits, skin sensitization in guinea pigs, and acute dermal toxicity in rats. To date, our knowledge of the toxicological effects of combinational prophylactic transdermal patches is still limited. Only a few reports are available on skin irritation and sensitization study for combination prophylactic transdermal patches. 4,5 Therefore, the present study was conducted to close a (knowledge/data) gap. We evaluated the potential for acute dermal irritation, skin sensitization, and acute dermal toxicities of transdermal patch or its placebo patch when administered on skin. Blood parameters and serum chemistry, using various animal models and the reversibility of its toxic effects, identify the target organs and thus provide experimental evidence of the safety of the test agent in compliance with Organization for Economic Cooperation and Development (OECD) guidelines.
Materials and Methods
Animal Husbandry and Maintenance
Healthy, adult New Zealand rabbits (weighing 1.2-2.5 kg, 17 weeks of age, male); healthy adult guinea pigs (weighing 650-750 g, 5-6 weeks of age, male); and healthy adult Wistar strain albino rats (weighing 210-250 g, 5-8 weeks of age, male and female) were obtained from Central Animal Resources, Defence Research Laboratory, Defence Research and Development Organization, Tezpur, Assam, India. The animals were placed in polypropylene cages with free access to standard laboratory diet (Pranav Agro Industries Limited, Sangli, Maharastra, India) and provided water ad libitum. Each individual animal was clinically examined upon arrival and identified by fur marked with picric acid. The females were nulliparous and not pregnant at the time of arrival. Animals were grouped and housed in an environmentally controlled room with temperature of 22°C ± 3°C and 40% to 70% relative humidity with a 12-hour light–dark cycle and ventilation of 15 to 21 air changes/h for an acclimation period of 7 days to laboratory conditions prior to the beginning of the experiment in order to adjust the new environment and to overcome stress incurred during their transit. Only healthy animals were assigned for these studies. Approval to carry out these studies was obtained from the Institutional Animal Ethics Committee under a subproject, and an experiment was performed in compliance with the Principles of Laboratory Animal Care (NIH Publication 85-23, revised 1985). All of the animal experimental protocols were in accordance with the guidelines of the committee for the purpose of control and supervision of experiments on animals, Ministry of Forest and Environment, Government of India.
Test Articles and Fabrication Protocol
Drug-in-adhesive matrix-type combinational prophylactic transdermal patches composed of eserine and 2-PAM and its placebo patches were tested for acute dermal irritation, skin sensitization, and acute dermal toxicity study. Detailed in vitro physicochemical evaluations and skin permeation studies were performed before commencing these dermal evaluations. Eserine and 2-PAM were provided by Sigma-Aldrich Chemical, Co, St Louis, Missouri. All biochemical kits were obtained from Coral Clinical Systems, Verna, Goa, India. All other reagents were of analytical grade and purchased commercially.
Experimental Design
Acute dermal irritation study
The acute dermal irritation/corrosion study was carried out in accordance with the OECD Guideline 404 6 method. Eighteen healthy rabbits with intact skin were used. One day before the first induction, the rabbits were assigned to 1 of 3 groups: a positive control group (n = 6) that received 0.8% w/v aqueous solution of formaldehyde as a standard irritant, a control group (n = 6) that received placebo patch, and a treated group (n = 6), that received drug-loaded transdermal patch. On day 0 of the test period, hair was clipped from the back of each rabbit. About 25 cm2 area were shaved from the back surface of each rabbit. Rabbits were caged individually and left undisturbed for 3 days. On day 1 of the test period, rabbits were treated with a placebo patch, drug-loaded transdermal patch, or a standard irritant by evenly applying the respective ingredients on to the shaved area. Rabbits were then returned to their cages. After 72 hours of exposure, the patches were removed and the test site was rinsed with double-distilled water. The animals were examined for the presence of erythema and edema according to Draize dermal irritation scoring system (0 = no erythema or no edema; 1 = barely perceptible erythema or edema; 2 = well-defined erythema or slight edema; 3 = moderate to severe erythema or moderate edema; and 4 = severe erythema or edema) at grading intervals of 24, 48, and 72 hours. Parallelly, cage side observations were made daily for signs of clinical toxicity throughout the test period.
Primary irritation index (PII) was also calculated. It was then classified according to Draize method of classification using the PII scoring as nonirritant (if PII <0.5), slightly irritant (if PII <2), moderately irritant (if PII ≤ 2-5), and severely irritant (if PII >5). For each animal, the dermal response scores (sum of the scores for erythema formation and edema formation) at 24, 48, and 72 hours after the removal of the patches were summed up and then divided by 3 to obtain a mean irritation score per time point. The mean scores at 24, 48, and 72 hours were summed and derived the average to obtain the PII.
Skin sensitization test
The skin sensitization test was carried out in accordance with the OECD Guideline 406 7 and modified per Buehler 8 method. Forty healthy guinea pigs were used for this study. One day before the first induction, the guinea pigs were assigned to 1 of the 3 groups: a positive control group (n = 10) that received0.1% w/v 1-chloro-2,4-dinitrobenzene (CDNB) in 10% propylene glycol as a standard skin sensitizing agent, a placebo group (n = 10), and a transdermal patch-treated group (n = 20). The left flank of each guinea pig was shaved using electric clippers and an electric shaver. Only those animals without injury or irritation of the skin were used in the test. During the induction phase of this assay, transdermal patch was applied to the shaved flanks of the animals. Control groups were treated with placebo transdermal patches. Treatment sites were left open between applications. On day 0, the first day of the first stage of induction, 0.1% w/v of sensitizing agent, CDNB, was evenly spread on the shaved skin. Challenge reactions were assessed at 24 hours after application. The intensity of all skin reactions was graded on a sensitization score of 0 to 3, with 0 indicating no reaction, 1 indicating scattered mild redness, 2 indicating moderate and diffuse redness, and 3 representing an intense skin reaction that included erythema and edema with eventual deeper skin damage. 9 Percentage sensitization rate, sensitization grade, sensitization classifications, and sensitization reaction were also made (Table 1).
Rate, Grade, and Classification of Sensitization.

Acute dermal toxicity study
An acute dermal toxicity study in Wistar albino rats was conducted in accordance with the OECD Guideline number 402. 10 A total of 24 (12 males and 12 females) healthy Wistar albino rats of both sexes weighing 210 to 250 g were divided into 2 groups and assigned as 1 treated group, that is, transdermal patch and 1 control/nontreated group. Each group consists of 6 male and 6 female animals. One day prior to commencing the acute dermal toxicity study, the backs of rats were clipped with an electric clipper. Each rat was caged individually and left undisturbed for 24 hours. On the test day, transdermal patch preparation was applied evenly to the exposed skin. Rats were then returned to their cages. The animals were observed twice daily for 14 days for signs of irritation, general behavior, and possible mortality. Body weight measurement, food, and water consumptions were taken daily for 14 days. On the 15th day, the rats were killed and the organs were carefully taken out and weighed. Histopathological examination of animals was performed at the termination of the study on day 15.
Clinical observation and mortality
Throughout the acute dermal toxicity study, all the animals were observed daily for clinical signs of irritation, general behavior, toxicity, morbidity, and mortality. Detailed clinical observations were recorded daily.
Terminal body weight trends
In acute dermal toxicity study, the terminal body weight of each rat was measured at the initiation of treatment and once a week (7th and 14th day) throughout the treatment period.
Food and water consumption
Food and water consumption of each rat caged individually were measured at the start of treatment and on a weekly basis up to the 14th day after treatment. A preweighed amount of food and water was provided for each cage and their remnants were measured the next day to calculate the difference, which was considered as daily food (g/kg body weight/day) and water consumption (mL/kg body weight/day), respectively.
Hematology
All animals were fasted overnight prior to necropsy and blood collection (14th day of acute dermal toxicity study). Blood samples were collected through orbital sinus vein puncture technique from retro orbital sinus of rats by 75 mm heparinized capillary tube (Haematocrit capillary, Himedia Laboratories Private Limited, Mumbai, Maharastra, India). The blood samples were collected in nonvacuum blood collection tubes containing K3 EDTA (Peerless Biotech Pvt Ltd, Chennai, Tamil Nadu, India) and analyzed within 60 minutes. The hematological parameters including white blood cell (WBC) count, WBC differential counts, that is, lymphocyte (Lym), monocyte (Mon), neutrophill (Neu), eosinophil (Eo), and basophil (Ba) counts, red blood cell (RBC) count, mean corpuscular volume (MCV), hematocrit (Hct), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), hemoglobin (Hb) concentration, and platelet (Pct) were examined by Automatic Hemato analyzer (MS-4) (Melet Schloesing Laboratories, Osny, France).
Serum biochemistry
Blood for clinical chemistry was placed in vacuum blood collection tubes (Peerless Biotech Pvt Ltd) devoid of anticoagulant (serum tube) and allowed to clot at room temperature. Blood samples were centrifuged at 3000 rpm (604
Necropsy and organ weight
All surviving animals were killed on day 15 by cervical dislocation under anesthesia. Necropsy of vital organs of all animals was carried out and the weights of the liver, brain, and kidney were recorded. The absolute and relative (organ to body weight ratios) weights of major organs and tissues including the liver, kidney, and brain were measured.
Histopathology
After necroscopy, the following samples of major organs (liver, brain, and kidney were preserved in neutral, phosphate-buffered 10% formalin. The samples were routinely processed, embedded in paraffin, and sectioned at 4 to 6 µm. The sections were stained with hematoxylin and eosin for microscopic examination. All organs taken from all animals in the vehicle control and treated groups were examined microscopically.
Statistical Analysis
Data on irritation were presented as visual scores based on the Draize method of erythema and edema grading system, and PII was calculated. Data obtained from hematology analysis, serum biochemistry, and the body and organ weight measurements were expressed as mean ± SD and evaluated by unpaired
Results
Acute Dermal Irritation Study
The results of acute dermal irritation study of the transdermal patch and the placebo patch in rabbits are presented in Table 2. No dermal responses, including erythema or edema, were found in rabbits treated with either the transdermal patch or its placebo compared to the negative control counterparts. The PII was calculated to be 0 in both the control and the treated groups, whereas in the group treated with 0.8% w/v aqueous solution of formaldehyde, all rabbits exhibited severe erythema after 72 hours. The PII was calculated to be 9.77 in this group, indicating severe irritation. Cage-side observation did not reveal any observable signs of systemic toxicity in any of the treated groups.
Acute Dermal Irritation Study of Transdermal Patch or its Placebo Patch in Rabbits.
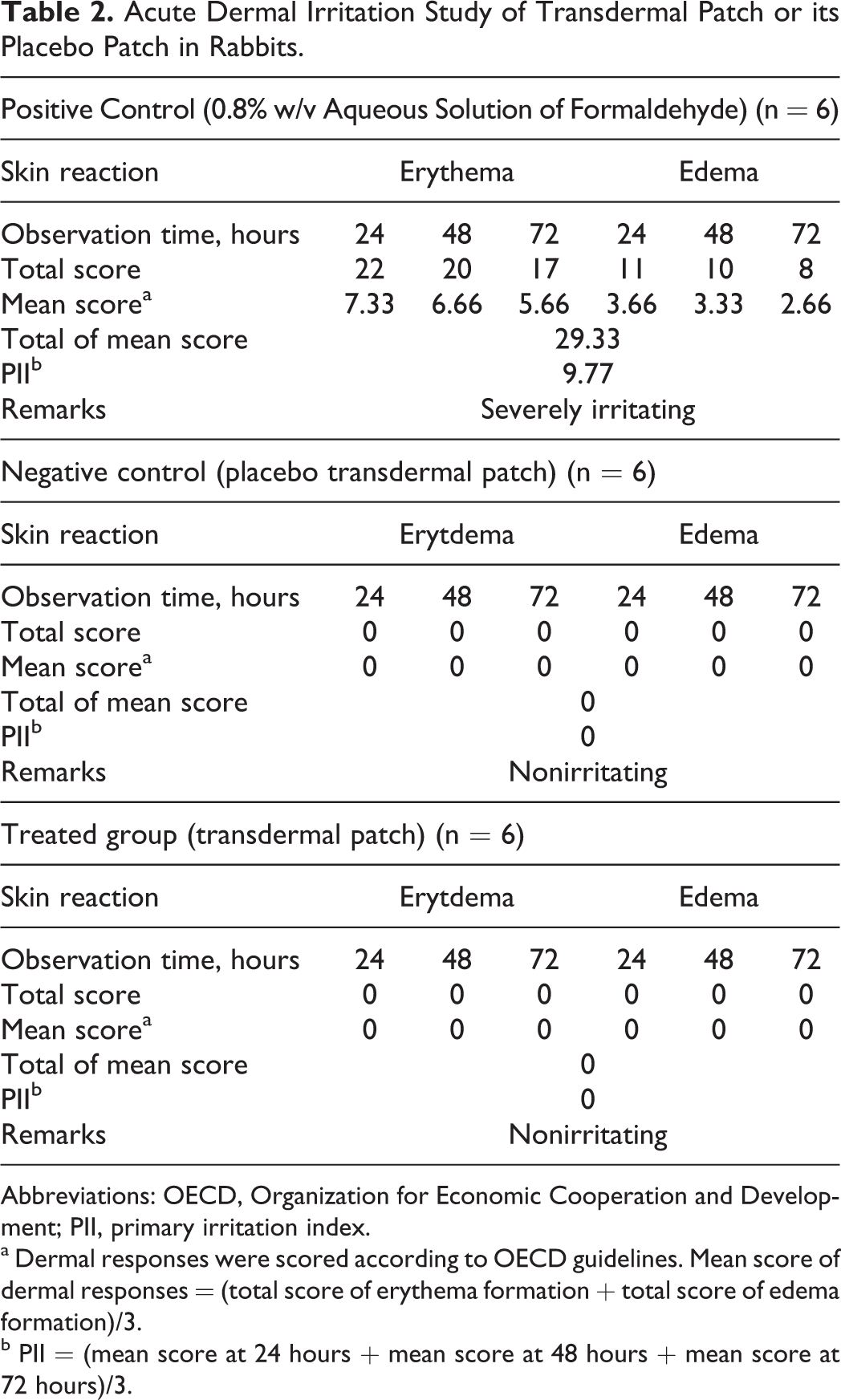
Abbreviations: OECD, Organization for Economic Cooperation and Development; PII, primary irritation index.
a Dermal responses were scored according to OECD guidelines. Mean score of dermal responses = (total score of erythema formation + total score of edema formation)/3.
b PII = (mean score at 24 hours + mean score at 48 hours + mean score at 72 hours)/3.
Skin Sensitization Test
The results of sensitization score, rate, grade, and classification of transdermal patch and its placebo after 24 hours of dermal application in guinea pigs are shown in Table 3. The skin sensitization experiments were validated using the positive control group (0.1% w/v CDNB), where positive dermal sensitization responses were observed. No sensitization was noted among guinea pigs that were challenged with transdermal patch or the placebo patch. Erythema and edema were not observed after the challenge in this study (treated and untreated).
Sensitization Score, Rate, Grade, and Classification of Challenge Reaction for 24 Hours After Dermal Application of Transdermal Patch in Guinea Pigs.
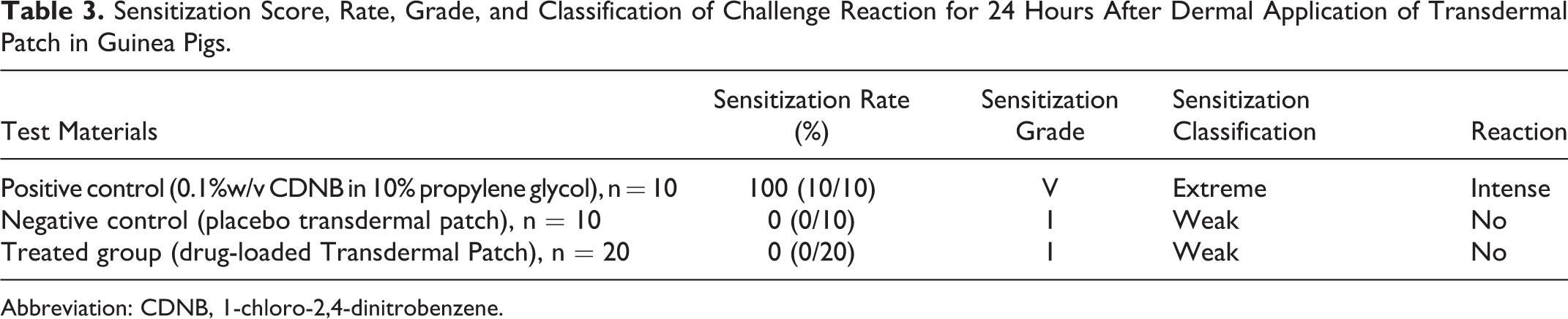
Abbreviation: CDNB, 1-chloro-2,4-dinitrobenzene.
Acute Dermal Toxicity Study
Wistar albino rats were divided into 2 groups (control and transdermal patch-treated group). A total of 6 rats/sex/group were used.
Clinical observation and mortality
None of the animals showed any clinical signs, and none showed any overt signs of toxicity from the first day until the end of the experiment. The skin of the animals appeared normal, and no erythema or edema was noted. The locomotor behavior was also normal, and there were no signs of toxicity detected in the treated rats.
Terminal body weight trends
In this study, no treatment-related changes were noted in the terminal body weights of rats when compared to their negative control counterparts. There were no statistically significant mean weight differences in body weights between the control and the treated groups from the first day of patch application through the end of the experiment (Table 4), thus indicating that the transdermal patch had no effect on the change in body weight of either sex.
Group Mean Terminal Body Weight (g) of Control and Treated Rats for 14 days.
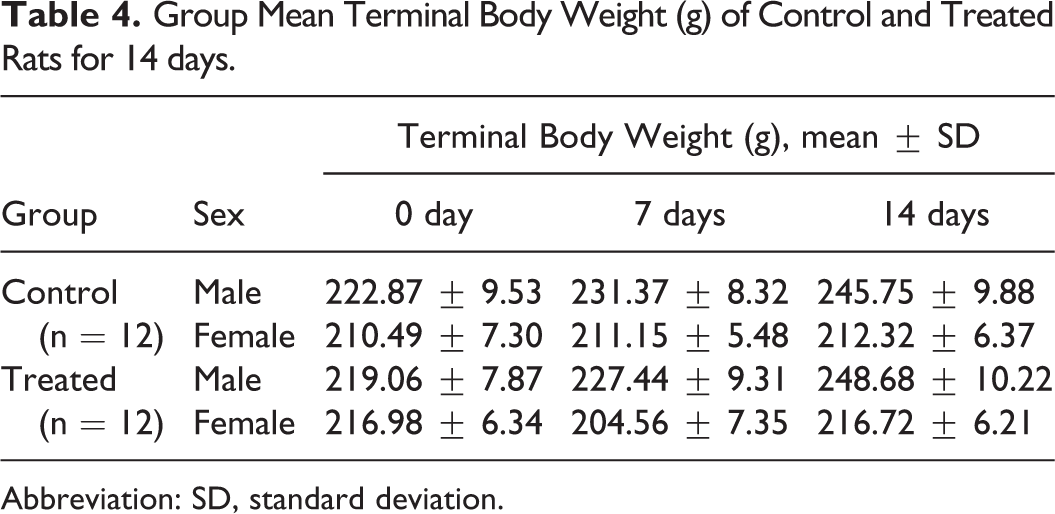
Abbreviation: SD, standard deviation.
Food and water consumption trends
No statistically significant differences were noted for food and water consumption among treated and control groups from the first day of patch application through the end of the experiment. No sex differences were noted among the treated groups (Tables 5 and 6).
Group Mean Food Consumption (g/kg body weight/day) Trends for Male and Female Wistar Albino Rats for 14 days.
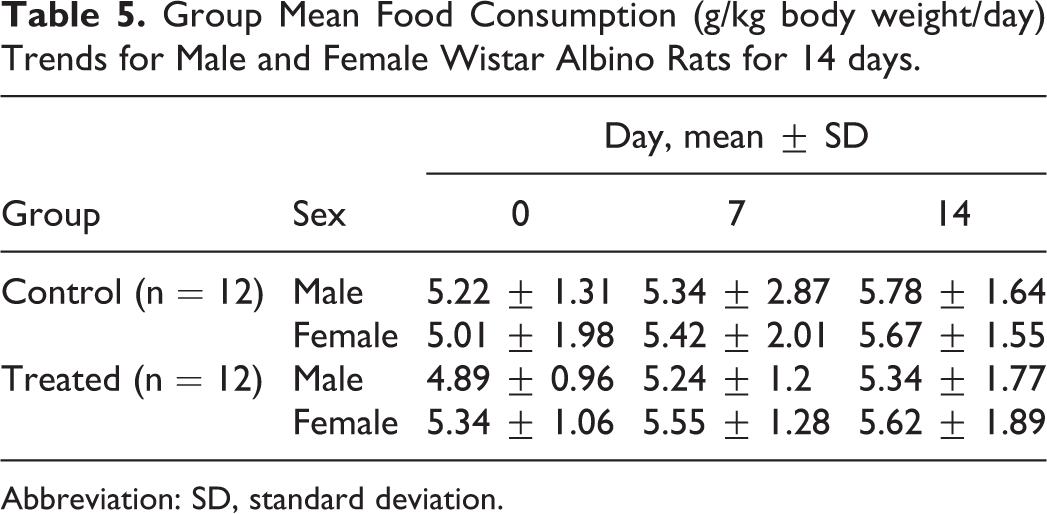
Abbreviation: SD, standard deviation.
Group Mean Water Consumption (mL/kg body weight/d) Trends for Male and Female Wistar Albino Rats for 14 days.
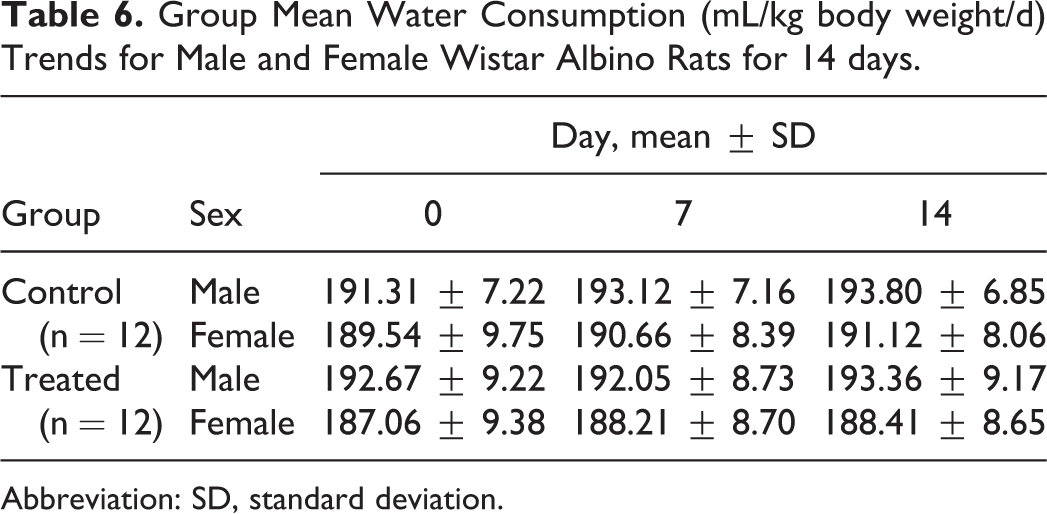
Abbreviation: SD, standard deviation.
Hematology
Hematological analysis was conducted for all treated and control groups. Various hematological parameters were tested including WBC count, WBC differential counts, that is, Lym, Mon, Neu, Eo, and Ba counts, RBC, MCV, Hct, MCH, MCHC, Hb, and Pct in both control and treated groups (see Supplementary Table 1). Hematology analyses did not show significant statistical difference between control and drug-loaded patch-treated group.
Serum biochemistry
Clinical chemistry parameters including LDL, HDL, GLUC, UREA, TP, UA, TGL, CHOL, CRE, ALT/SGPT, and AST/SGOT were evaluated in both sexes and each treatment group. Groups treated with transdermal patch did not show significant statistical differences between control and treated groups in either sex (see Supplementary Table 2). Transaminases, that is, ALT/SGPT and AST/SGOT are good indices of liver damage. 11 The transdermal patch did not affect the levels of transaminase, indicating that the patch does not negatively impact the liver.
Necropsy and organ weight
Following necropsy, no macroscopic changes were observed in the internal organs of all treated animals. The absolute and relative organ weights of rats showed no statistically significant difference between test and control groups (see Supplementary Table 3 for absolute and relative organ weights).
Histopathology
Analysis of the toxic potential of a chemical agent on target organs is incomplete without gross and histopathological evaluation. Histopathological examination of selected organs of both treated and control animals showed normal architecture, suggesting no abnormal findings in the histological evaluation.
Discussion
Acute dermal irritation study of the transdermal patch and the placebo patch in rabbits showed no dermal responses, including erythema or edema, compared to their negative control counterparts. No skin sensitization was also observed among guinea pigs that were challenged with transdermal patch or the placebo patch. It is also evident from the results of acute dermal toxicity study that none of the animals in each group showed any clinical signs (such as making noise immediately after treatment, struggling, etc) and any overt signs of toxicity from the first day until the end of the experiment. There was no treatment-related mortality in any of the groups following patch administration in either male or female rats for 14 days. Loss of body weight is an important marker of gross toxicity. Drastic toxicity or interference with absorption of nutrients will be reflected in body weight reduction. The body weight in the transdermal patch-treated groups did not differ significantly from the control group. Hence, it can be inferred that the formulation has no tendency to produce drastic tissue destruction nor does it seem to interfere with absorption of the nutrients. Food and water consumption among treated and control groups were almost similar without any significant statistical differences. The effects of treated group on hematological, biochemical, and histological parameters were also considered in this study. The acute toxicity experiment showed that the patch-treated group did not show any hematological and biochemical changes up to 14 days of treatment period. Of the 13 hematological parameters studied, the percentage lym count of the treated groups of both sexes was found to be higher when compared to the levels noted in the control group. No adverse effects were noted in the levels of CRE, UREA, and UA in serum of the treated group when compared with negative controls, thus confirming the lack of nephrotoxic potential of the transdermal patch. After postmortem examination, the gross examination of the vital organs such as liver, kidney, and brain showed no noticeable hemorrhage and abnormal conditions. After the necropsy, the relative weights of these 3 vital organs were recorded, and no significant changes were observed in the treated group when compared to the control group. The present study also clearly showed that dermal contact with the patch did not cause any major histopathological abnormalities in the liver, kidney, and brain of the animals. This study has also emphasized that the treated groups exhibited no organ toxicity signs compared to control groups. If we consider the necropsy data together with the histopathological findings, it can be inferred that this prophylactic transdermal patch is a safe therapeutic system and may be used against (±) anatoxin-A poisoning without any notable toxicity.
Conclusion
In this study, we have shown that the placebo transdermal patch and the drug-loaded transdermal patch were not irritating to the skin of several species. None of the treatment group exhibited skin sensitization effects except for the positive control group. Regarding acute dermal toxicity study, the overall results indicate that the dermal administration of transdermal patch did not result in any toxicological effect on the vital organs such as the liver, kidney, or brain in both male and female Wistar albino rats. There were no significant changes in hematology, serum biochemistry, or terminal body weight following a 14-day observation period. No detectable abnormalities were found in the histopathology of the selected organs. These preliminary toxicological results suggest that, under the conditions of this study, short-term treatment of rat with this transdermal patch does not have any detectable adverse effects. It can be concluded from the findings of the present study that the initial safety profile for this transdermal patch looks promising as a safe prophylactic transdermal therapeutic regimen against (±) anatoxin-A poisoning but warrants further research in terms of antidotal and neuroprotective efficacy study.
Footnotes
Acknowledgments
One of the authors Subham Banerjee is grateful to Defence Research and Development Organisation, Ministry of Defence, Government of India for providing the research fellowship for this work. Subham Banerjee is also thankful to Defence Research Laboratory, Tezpur, Assam, India, for providing necessary instrumental facilities for this research work and the administration of the Birla Institute of Technology, Mesra, Ranchi, India, for providing necessary administrative support for carrying out his PhD work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
