Abstract
We report renal tubular adenomas and a carcinoma in 26-week Tg.rasH2 mouse carcinogenicity studies, which have not been reported to date either at our facility or in other published data. However, during the year 2014, renal tubular tumors were present in 4 studies conducted at our facility. Because of their morphological similarity to the amphophilic–vacuolar (AV) phenotypic variant of renal tubule tumors noted in Sprague-Dawley and Fischer 344 rats, which are thought to be familial, as well as the genetic homogeneity of Tg.rasH2 mice, we tracked the parents of these mice with tumors in each study. The origin of these tumors could not be traced back to any of the parents or even an animal barrier, and these tumors were not attributed to the vehicle or test article. Although the exact mechanism of tumorigenesis was not known, based on the available information, the development of renal tumors in these mice was considered random and spontaneous.
Keywords
The 2-year rodent carcinogenicity assays involving conventional rats and mice have been conducted for over 3 decades. As an alternative to the 2-year rodent carcinogenicity bioassays, 26-week short-term carcinogenicity bioassays were approved using genetically modified mouse strains, including Tg.rasH2 (International Conference on Harmonisation 1998). The Tg.rasH2 model, which can be used for both genotoxic and nongenotoxic compounds, has gained popularity and its use has increased over the years, and currently more than 75% of mouse carcinogenicity studies are conducted in Tg.rasH2 mice (Jacobs and Brown 2015). The Tg.rasH2 model predicts neoplastic findings relevant to human cancer risk assessment, produces fewer nonbiologically significant neoplastic outcomes, and is thus preferable to a 2-year rodent study (Morton et al. 2014). We recently published the largest historical control database for neoplastic lesions in Tg.rasH2 mice (Paranjpe et al. 2013). Historical control data on Tg.rasH2 model have also been published by others (Nambiar, Turnquist, and Morton 2012; Morse et al. 2015). None of these papers have reported primary renal tumors (either adenomas or carcinomas) in Tg.rasH2 mice. The material and methods pertaining to animals, housing, environmental conditions, regulatory requirements, and retrospective analysis were identical to the ones that we have published before (Paranjpe et al. 2013). After each study was completed, tumor data statistical analysis was performed on each study separately referencing the draft Food and Drug Administration Guidance for Industry (2001). The incidence of tumors for each study was separately analyzed by mortality-prevalence method, without continuity correction, incorporating the context (a) benign vs. malignant, (b) primary vs. metastatic vs. multicentric, (c) incidental vs. fatal, and (d) single vs. multiple, in which tumors were observed (Peto et al. 1980). There were no statistically significant differences for any of these renal tumors or any other tumors and in the survival in the individual studies when the test article groups were compared to the control group. Each study was administered a different control and test article; the test articles in individual studies were not related or members of a particular family of compounds. All tumors described in this article were amphophilic with lobular appearance and morphologically similar to AV tumors noted in Sprague-Dawley and Fischer 344 rats, which are considered familial in nature (Crabbs et al. 2013; Hard et al. 2008). However, none of the tumors exhibited both amphophilic and vacuolar patterns. When the parents of the mice used in these 4 individual studies were tracked, the mice with renal tumors could not be linked to any particular set of parents or animal barrier to establish a familial pattern. The mice with renal tumors reached terminal sacrifice and were less than 9 months old at the time of terminal sacrifice. Similar to the rats, there was no sex predilection (Hard et al. 2008).
The distribution of renal tumors noted in the 4 studies is presented in Table 1. There were 25 mice per sex per group and included 4 dose groups designated as vehicle, low, mid, and high in each study. These tumors were observed only in a single control male (in 2 of 4 studies), in a single high-dose male (in 2 of 4 studies), and a single high-dose female (in 3 of 4 studies). No renal tumors were noted in the low- and mid doses of either sex, and the renal tumors were not simultaneously observed in the control and high-dose groups of the same sex in each study. The interesting feature of these tumors was that they were all multiple, 2 or more tumors were present either in the same kidney or in both the left and right kidneys. Figure 1 demonstrates renal adenoma with amphophilic nature, in which the cells were well differentiated. Some of the tumors demonstrated cystic and/or papillary pattern. In the same kidney of 1 male mouse in a single study, multiple solid pattern amphophilic adenomas as well as a carcinoma, diagnosed based on the anisokaryosis, anaplasia, giant cells, and high degree of mitosis (Figure 2), were present. There were no hyperplastic lesions in any of the kidneys, and there were no other remarkable lesions or other tumors in these individual animals. Although the exact mechanism of tumorigenesis was unclear from the limited data available, the authors believe the development of renal tumors in these mice was random and spontaneous based on the characteristics described above.
Incidence of Renal Tumors in Tg.rasH2 Mice.
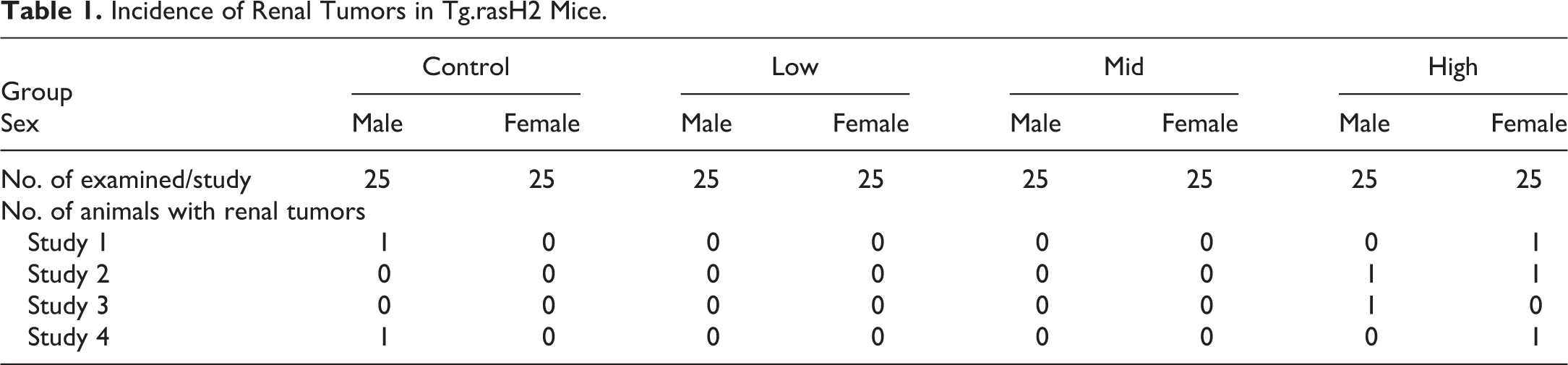
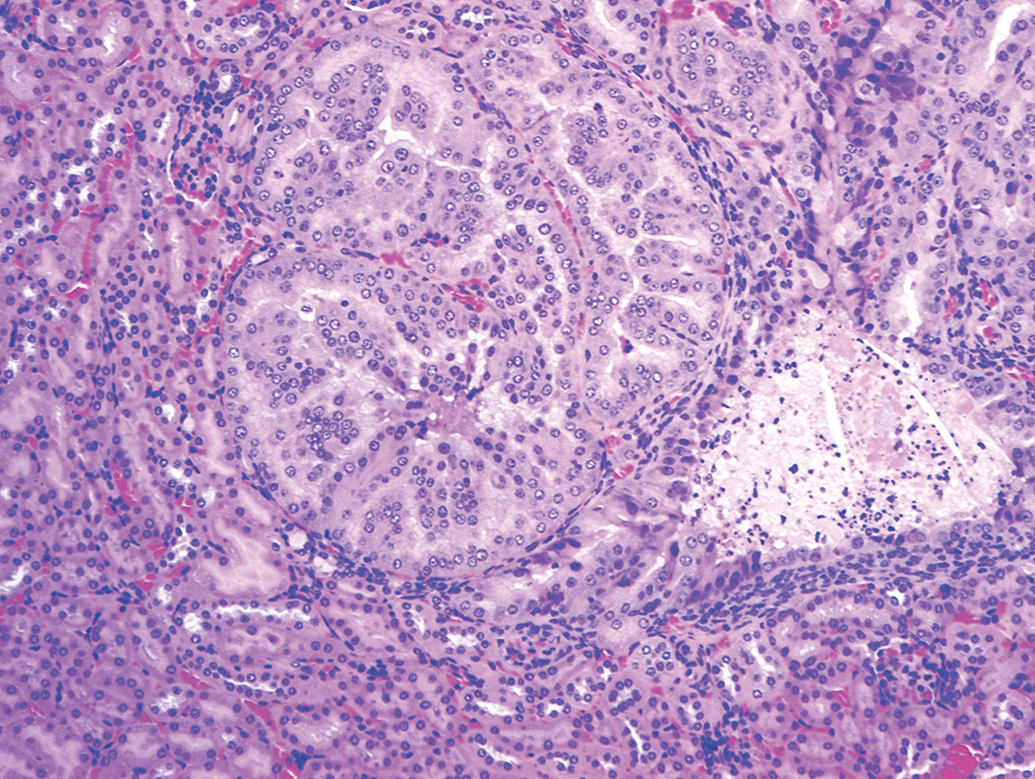
Renal tubular adenoma, amphophilic nature, hematoxylin and eosin stain, 10× original objective.
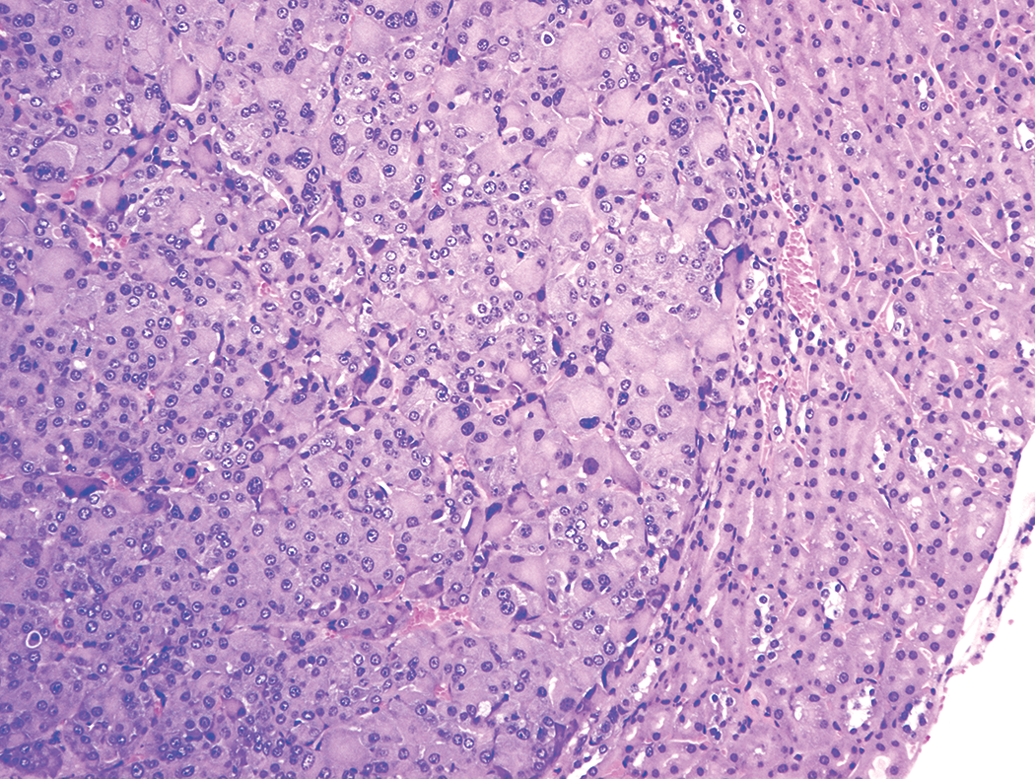
Renal tubular carcinoma, hematoxylin and eosin stain, 10× original objective.
Footnotes
Author Contribution
Authors contributed to conception or design (MP, JB, MM, RE, PM, GH, JS); data acquisition, analysis, or interpretation (MP, JB, MM, RE, PM, GH, JS); drafting the manuscript (MP, JB); and critically revising the manuscript (MM, RE, PM, GH, JS). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
