Abstract
The evaluation of inhalation studies in monkeys is often hampered by the scarcity of published information on the relevant nasal anatomy and pathology. We examined nasal cavities of 114 control cynomolgus monkeys from 11 inhalation studies evaluated 2008 to 2013, in order to characterize and document the anatomic features and spontaneous pathology. Compared to other laboratory animals, the cynomolgus monkey has a relatively simple nose with 2 unbranched, dorsoventrally stacked turbinates, large maxillary sinuses, and a nasal septum that continues into the nasopharynx. The vomeronasal organ is absent, but nasopalatine ducts are present. Microscopically, the nasal epithelium is thicker than that in rodents, and the respiratory (RE) and transitional epithelium (TE) rest on a thick basal lamina. Generally, squamous epithelia and TE line the vestibule, RE, the main chamber and nasopharynx, olfactory epithelium, a small caudodorsal region, while TE is observed intermittently along the passages. Relatively high incidences of spontaneous pathology findings, some resembling induced lesions, were observed and included inflammation, luminal exudate, scabs, squamous and respiratory metaplasia or hyperplasia, mucous cell hyperplasia/metaplasia, and olfactory degeneration. Regions of epithelial transition were the most affected. This information is considered helpful in the histopathology evaluation and interpretation of inhalation studies in monkeys.
Keywords
Introduction
Inhalation studies are designed to assess drug or chemical-induced respiratory effects in animals in order to predict the potential risk to human health. For biological pharmaceuticals that are to be given via the inhalation route, nonhuman primates may be the only pharmacologically relevant species due to their close phylogenic relationship to man. For this reason, inhalation studies are often performed in monkeys, with a detailed evaluation of the nasal cavity. However, the evaluation of the monkey nose is often hampered by the scarcity of published information on the gross anatomy, normal histology, and background pathology of the nasal cavities in this species (Renne et al. 2007). A working knowledge of the comparative anatomy and histology of the nasal cavity of the laboratory test species is required for a proper evaluation of toxic effects in the nasal cavity and the extrapolation of the data to human risk assessment (Chamanza et al. 2015; Harkema, Carey, and Wagner 2006). In addition, information on the range and incidences of the more commonly occurring background or incidental pathology findings is of particular importance when evaluating inhalation studies, since some of them may be confused with induced lesions.
For the cynomolgus monkey (Macaca fascicularis), which is the most widely used purpose-bred nonhuman primate in preclinical safety testing, no published information on these parameters is readily available. This study was therefore designed to investigate, describe, and document the characteristic gross anatomical features, morphology and distribution of the epithelium, and the nature and incidence of spontaneous pathological findings in the nasal cavity of young control cynomolgus monkeys in order to produce readily available reference material.
Materials and Method
The Animals
Nasal cavities were obtained from 114 control cynomolgus monkeys (57 animals per sex) from 11 inhalation toxicity studies of 4- to 13-week duration, conducted between 2008 and 2013. All animals used in these studies were of Vietnamese origin, purpose bred for laboratory use, and obtained from accredited suppliers. They were all juvenile monkeys between 27 and 42 months of age (with a full set of deciduous teeth and 1 or 2 permanent erupted molars or incisors) at the start of the studies and weighed between 2.5 and 3.5 kg.
Study animals were housed in groups of 1 or 3 animals of the same sex and dose group in custom-designed U.K. Home Office–compliant primate cages (chapter 14, section 21, U.K. Animals [Scientific Procedures] Act of 1986). The temperature and humidity were automatically controlled at 21°C ± 4°C and 55% ± 10%, respectively, with a minimum of 15 air changes per hour. An automatic light cycle of 7:00 a.m. to 7:00 p.m. (20-hr cycle) was maintained. Each individual gang pen had drinking water and food hoppers, and animals were fed a commercial primate diet (Mazuri diet, Special Diet Service Ltd, Witham, Essex, England). Twice weekly fruit supplements were also offered to all animals. Animals received an appropriate veterinary examination, which included serological tests to confirm that they were free of routinely tested infectious agents. They were confirmed free of respiratory infections and the following viral diseases during quarantine: simian immunodeficiency virus, Cercopithecine herpesvirus 1 (B virus), simian retroviruses type D, rabies, simian T-cell leukemia virus, measles, and filoviruses. Tuberculin tests; other bacteriology screening tests for Shigella, Yersinia, and Salmonella; and parasitological screening (including malaria smears) were carried out on arrival at the test facility.
All studies were conducted in accordance with the U.K. Animals (Scientific Procedures) Act 1986, which conforms to the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (Strasbourg, Council of Europe).
Pathology Procedures
A detailed necropsy examination was performed on all animals following euthanasia by intravenous injection with sodium pentobarbitone and exsanguination. Soon after euthanasia, the heads of the monkeys were removed from the body and the nasal passages flushed with 10% neutral buffered formalin (NBF) retrograde through the pharynx. After flushing the nasal cavities, the brain, mandible, eyes, integument, and musculature were removed from the head, which was then immersed in a large volume of NBF.
Nasal Cavity Trimming of Routine Sections
After removing the lower jaw and the soft tissues, and prior to decalcification, the remaining upper part of the head was fixed to a clamp and serially trimmed transversely, from the vestibule to the nasopharynx, using a water-cooled diamond-bladed band saw. A total of 5 to 6 coronal (transverse) sections were cut in a rostro-caudal direction, using dental and palatal landmarks (Figure 1A), to produce a minimum of 4 tissue slices (levels 1–4). Each slice was then placed with the rostral face down in a cassette.
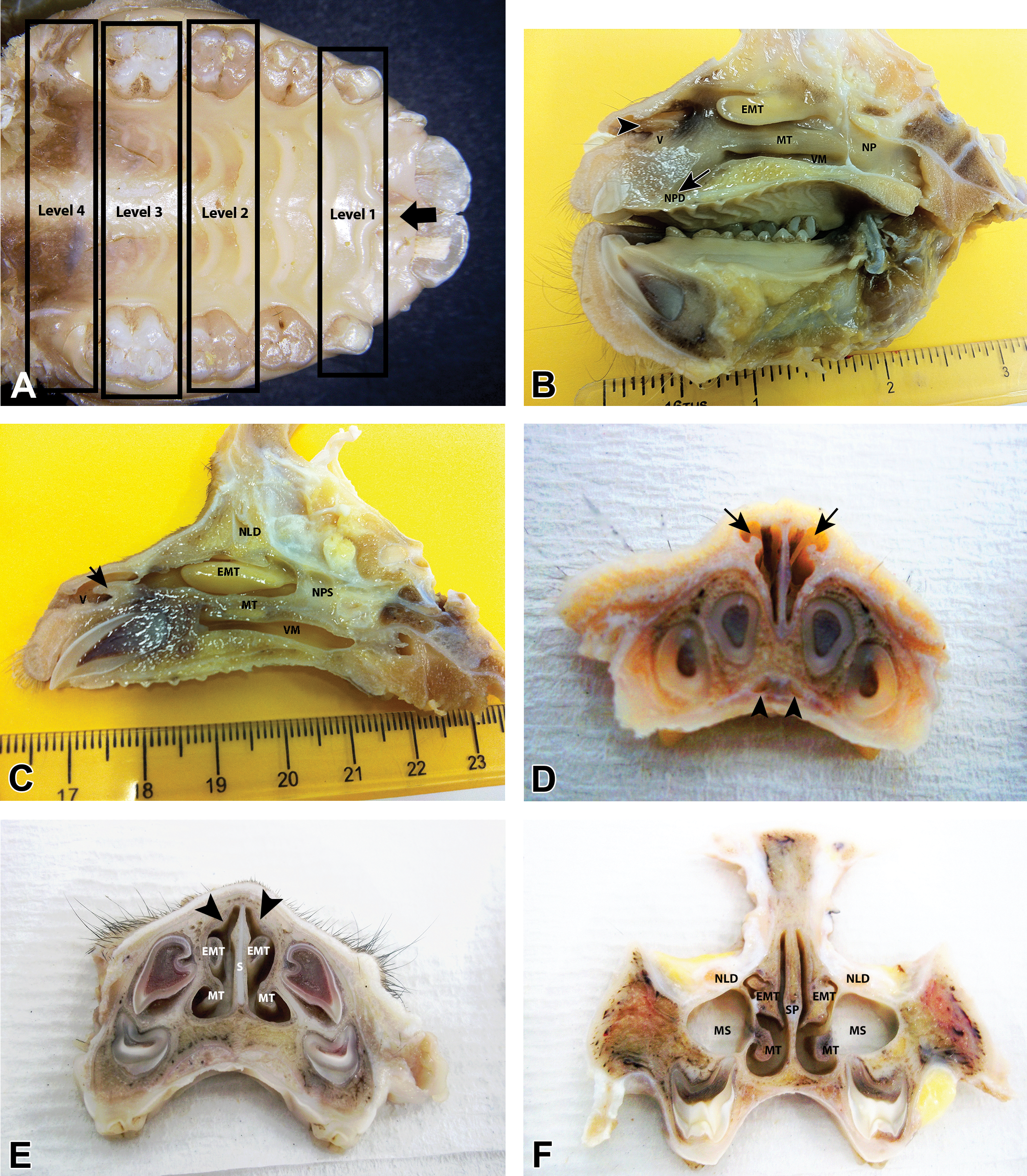
Gross sections of the nasal cavity (NC) of young cynomolgus monkeys (Macaca fascicularis) of 29 to 33 months of age and the landmarks used for nasal trimming. (A) Hard palate from a young female cynomolgus monkey, showing dental structures and palatal ridges used as landmarks for the trimming of coronal sections of the NC. The incisive papilla where nasopalatine ducts (NPDs) exit is shown (arrow). (B) and (C) Parasagittal sections of the NC of young female cynomolgus monkeys showing the vestibule (V), vestibular wings (arrow and arrowhead), maxilloturbinates (MTs), ethmoturbinates (EMTs), NPDs, nasolacrimal ducts (NLDs), the nasopharynx (NP), the ventral meatus (VM), and the nasopharyngeal septum(NPS). (D)–(F) Coronal (transverse) sections of the NC of a juvenile male cynomolgus monkey (note that some teeth are yet to erupt). (D) Rostral V showing NPDs (arrowheads) and how the lateral nasal cartilages or vestibular wings (arrows) create a crude dorsolateral meatus. (E) Rostral NC at level 2 showing MTs, unattached EMTs, nasal septum (S)(SP), and the dorsolateral ridge (arrowheads). (F). Section of the NC at level 3, showing MTs, EMTs, NLD, SP, and maxillary sinuses (MS). The NLDs lie lateral to the EMTs at this level and the septal cartilage is reduced to small piece in the middle of the septum.
Cut 1. Rostral vestibule (not examined on most studies)—Immediately caudal to the second incisor or at the level of the canine or the first palatal ridge.
Cut 2. Level 1—Immediately rostral to the first premolar (or the first deciduous molar) and between the first and second palatal ridges.
Cut 3. Discarded—Immediately rostral to the second premolar (or the second deciduous molar) and between the 3rd and 4th palatal ridges.
Cut 4. Level 2—Immediately rostral to the first (permanent) molar and between the 5th and 6th palatal ridges.
Cut 5. Level 3—Immediately caudal to the first (permanent) molar.
Cut 6. Level 4—Caudal to the second (permanent) molar or the mesiodistal diameter of a molar.
Nasopharynx—Caudal to the last molar and through the soft palate.
The trimmed coronal nasal sections were decalcified using the Ultrasonic Decalcifying Automate (Medite® Medizintechnik GmbH, Burgdorf—Germany) and the Medite USE 33 decalcifying method before embedding. After decalcification, tissues were embedded in paraffin wax, sectioned to a 3- to 4-mm thickness, and stained with hematoxylin and eosin. They were examined histopathologically and the findings entered directly into a computerized database (Pristima Path/Tox, Xybion Medical Systems Corporation, Morris Plains, NJ).
Study materials including histological incidence tables, individual animal data listings, and glass slides were retrieved from the archives and analyzed for the normal histology and spontaneous pathology of the nasal cavity. In addition, a historical control data search was performed for pathology findings recorded under the nasal cavity. Data were available from 114 control animals that had been sham dosed with air, saline, or lactose vehicle via the nose-only method of inhalation exposure.
Nasal Cavity Trimming of Longitudinal Sections
Further investigations of the gross anatomy of the nasal cavity and its associated structures were carried out on 4 heads obtained from control animals on noninhalation studies, from which at least 3 mid- or parasagittal sections were obtained (Figure 2B and C). The sections were obtained by cutting longitudinally (along the long axis of the head) through the right nasal fossae from the nares to the pharynx and exposing the lateral meatus and turbinates.
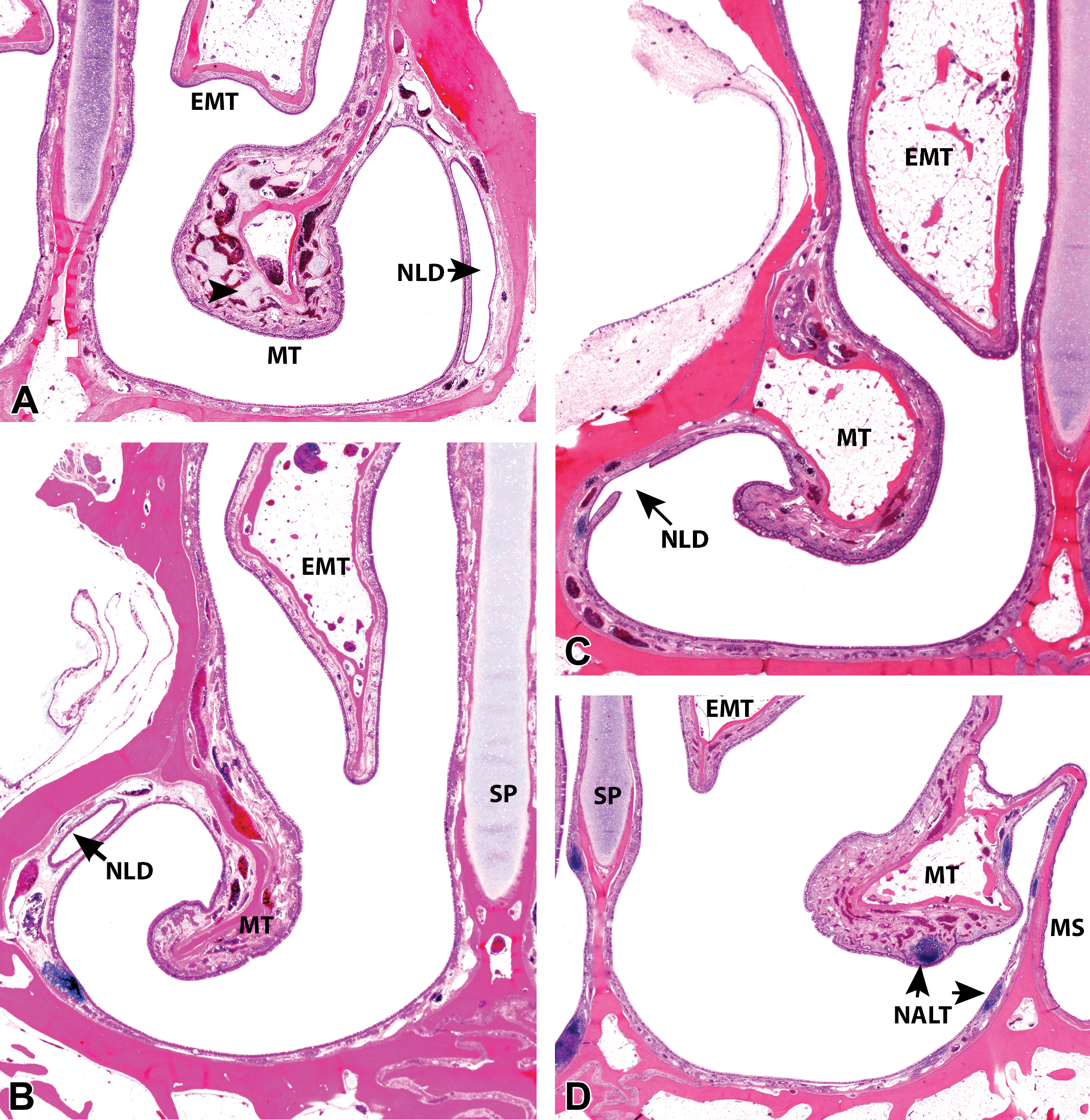
Subgross coronal sections of the ventral nasal meatus of young cynomolgus monkeys (Macaca fascicularis) of 36 to 40 months of age. (A)–(C) Interanimal variations in the size and shape of ethmoturbinates (EMTs) and maxilloturbinates (MTs) in young female cynomolgus monkeys at level 2 of the nasal cavity (NC), which shows the exit of the nasolacrimal ducts (NLD; arrow) in the ventral meatus (VM). Note how at this level the SP is mostly composed of cartilage. Foci of cartilage (arrowhead) and large venous sinuses present in the MTs are also shown. (D) and (E) Interanimal variations in the ventral extension of EMTs in relation to MTs in young female monkeys at level 3 of the NC, which shows the maximum diameter of the maxillary sinus (MS). Note that at this level, the MTs arise from a separate secondary bone that divides the nose into the nasal meatus and the MS. (F) Caudal VM of a young male cynomolgus monkey at level 4, showing that the MS and the NC are separated only by a mucous membrane (arrow) at this level. All images, hematoxylin and eosin, ×1 original objective. SP = nasal septum, NALT = nasal-associated lymphoid tissue.
Results
Gross and Subgross Anatomic Features on Parasagittal Sections (Figure 1B and C)
The gross examination of the cynomolgus monkey nose shows considerable variability in the size and shape of the main nasal structures. The main interanimal differences are mainly in the shape of the turbinates and the overall length of the nasal cavity, and they do not appear to be sex- or age dependent (animals were of a relatively similar age). However, despite this, the general layout of nasal features is relatively uniform between individual animals. Parasagittal sections reveal that, from rostral to caudal, the nasal cavity of the cynomolgus monkey can be roughly divided into a vestibule, a main nasal chamber with 2 dorsoventrally stacked turbinates, and a nasopharynx that is divided by a membranous median septum (Figure 1B and C). The vestibule is relatively large and empty (in comparison to that of rodents and dogs), and there are no well-developed atrioturbinates or alar folds, although a dorsolateral vestibular (alar) wing that forms an atrioturbinates-like projection is present (Figure 1B, C, and D). The vestibule is elevated above the floor of the main nasal chamber due to the prominence of the separate premaxilla. This allows the ventral floor of the main nasal cavity to continue forward (rostroventral) below the floor of the vestibule as the incisive canal or nasopalatine duct (NPD; Figure 1B and D). The NPDs (or Stenson’s ducts) open into the oral cavity at the incisive papillae (Figure 1A).
In the nasal chamber proper, the dorsally located ethmoturbinates (EMTs) can be easily identified by their bright yellow color, which is often retained even after prolonged fixation (Figure 1B and C). They appear as long, horizontal finger-like projections that are attached only at the dorsocaudal end of the nasal cavity (Figure 1B and C). The rostral ends are free on 3 surfaces, for as much as a rostral 3rd of the length of the turbinates. Interanimal variations in the shape of the ventral edge of the EMTs may be appreciated on parasagittal sections, but they are more conspicuous on coronal sections (Figure 2A–F). The variations in the shape of the EMTs represent the greatest interindividual differences in the gross anatomy of the cynomolgus monkey nose and do not seem to be age or sex related. Below the EMTs are the ventrally located pair of maxilloturbinates (MTs), which span about two-thirds of the nasal cavity caudorostrally. The MTs (which correspond to the human inferior turbinates) are the rostral-most turbinates by only a very small margin in monkeys and widen to blend with the lateral walls both caudally and ventrally (Figure 1B and C). The shape of their ventral surface also shows some slight variation on longitudinal sections, but as with EMTs, the interanimal variations are more apparent on coronal sections. There are no analogous structures to the superior nasal turbinates of humans and nasoturbinates are absent, although a small bulge or nasal ridge formed by a small separate bone is often observed on the dorsolateral wall, at the level of the nasal atrium (Figure 3B). In some animals, a poorly defined vestigial structure is occasionally observed attached to the dorsal surface of the EMT.
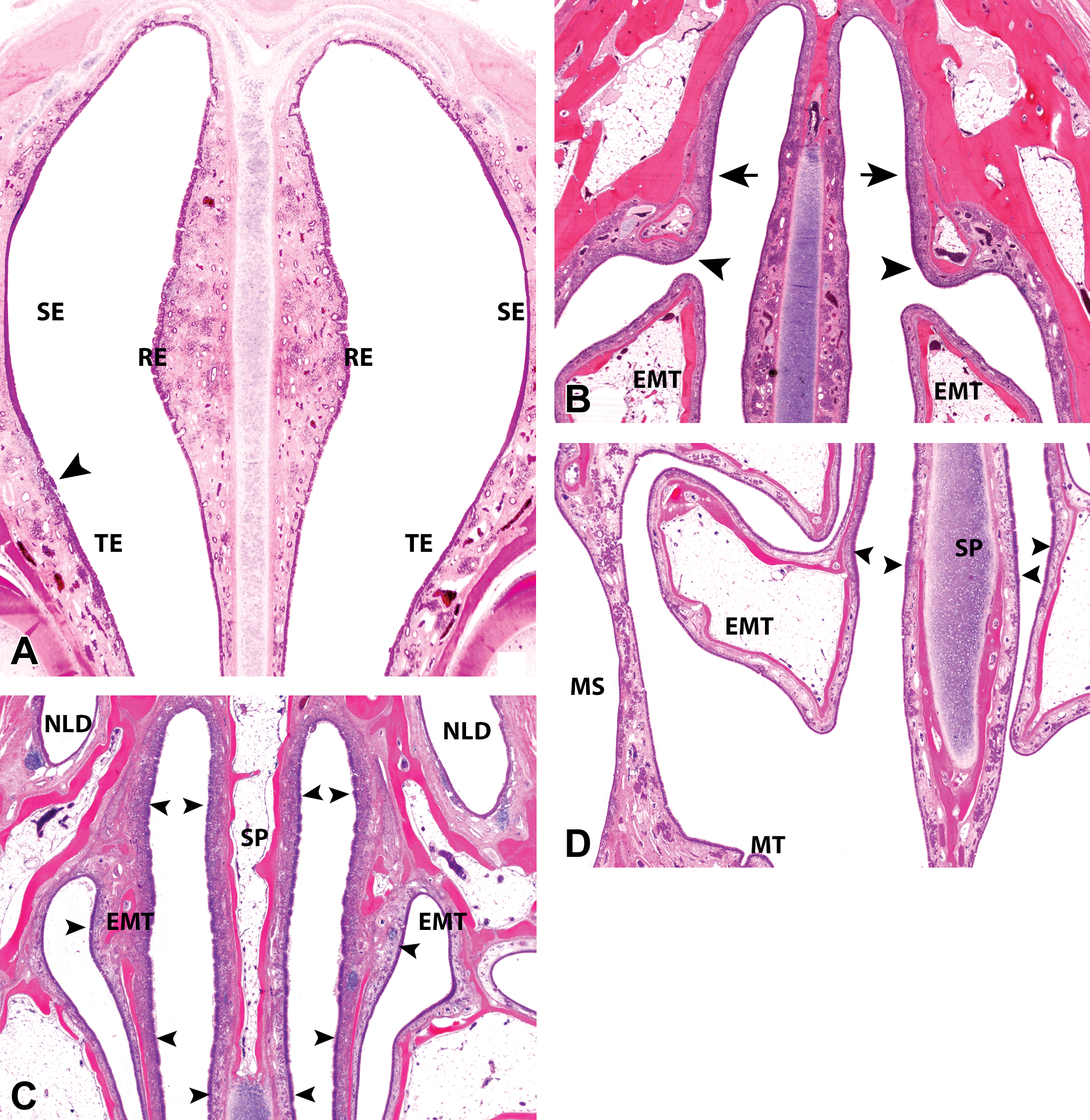
Subgross coronal sections of the dorsal nasal meatus and the nasopharynx (NP) from young cynomolgus monkeys (Macaca fascicularis) of 33–42 months of age. (A) Caudal nasal vestibule of a young female cynomolgus monkeys at level 1 showing the septal bulge and the distribution of squamous (SE), transitional (TE), and respiratory epithelium (RE) in the lateral and septal (SP) walls. Inflammation is present in the lateral wall at the SE and TE junctions (arrowhead). (B) Dorsolateral nasal ridge at level 2 of the nasal cavity (NC) of a young female cynomolgus monkey. The ridge is formed by a small separate bone (arrowhead) and is a region of frequent inflammatory lesions. Dorsal to the ridge at this level is a region of a poorly defined epithelium that resembles both respiratory and olfactory epithelia (OE; arrow). The unattached ethmoturbinates (EMTs) are lined by RE. (C) and (D) Caudal NC of young male cynomolgus monkeys at level 4, showing the dorsal and ventral limits of the OE (arrowheads) on the EMTs and septum (SP) and the location of the nasolacrimal ducts (NLD) dorsal to the NC at this level. Ventrally at this level, the maxillary sinus (MS) and the NC are separated only by a mucous membrane while the maxilloturbinates (MTs) are not anchored by a bony core. (E) EMTs in a young female cynomolgus monkey at level 3, showing regions of frequent background changes (arrowheads) and that the medial and lateral surfaces roughly follow the contours of the adjacent nasal walls. Septal deviation with soft tissue reaction and swelling at the ends of the septal cartilage (SPC) are present (arrow). (F) Level 4 of the NC of a young male cynomolgus monkey showing the caudal edge of the nasal meatus and the beginning of the NP, where the SPC is reduced to its smallest size and is a frequent site for septal deviations. (G) The NP of a young female cynomolgus monkey showing septal tonsillar lymphoid tissue. (H) Level 2 of the NC of a young female cynomolgus monkey showing a rostral dorsal meatus lined by nonolfactory RE and some nasal-associated lymphoid tissue. The distal tips of EMT at this level are a frequent site of spontaneous pathology and are lined by TE and RE. All images hematoxylin and eosin, ×1 original objective.
Gross and Subgross Anatomic Features on Coronal Sections
The main anatomic features on the monkey nasal sections produced by the method described above (Figure 1A) are summarized in Table 1. Minor variations between sections taken at the same level were relatively common and were attributed to a combination of interindividual variability in monkeys and tangential sectioning. However, in general, all coronal sections (Figure 1D–F) reveal a relatively simple nose that is divided into 2 separate and symmetrical passages by a median nasal septum, which is mostly cartilaginous rostrally and bony caudally. The nasal septum divides the nasal cavity from the naris to the choana and past the caudal edge of the vomer into the nasopharynx, where it continues thereafter as a membranous median septum that divides the nasopharynx into 2 equal halves, for as much as two-thirds of its length. A more detailed description of the toxicologically relevant gross and subgross anatomic features on the nasal sections from rostral to caudal is given subsequently.
Main Anatomical Features and Epithelial Types Present on Various Coronary Sectional Levels of the Nasal Cavity of Young Cynomolgus Monkeys (Macaca fascicularis).

Note. OE = olfactory epithelium; RE = respiratory epithelium; SE = squamous epithelium; TE = transitional epithelium.
The coronal section of the vestibule rostral to level 1 reveals how the dorsal and lateral cartilaginous projections of the vestibular wing create a crude dorsolateral meatus of varying sizes (Figure 1C). Caudally, level 1 shows the caudal vestibule or the entrance to the main nasal chamber, which is often wide and empty and devoid of any projections. A nasal ostium as found in humans is not present at the entrance of the main chamber, but a slight narrowing of the nasal passage is achieved rostrally by a bulging of the septum in the middorsal 3rd (Figures 1E and 3A). This midseptal bulge or tubercle is often present from the vestibule to a level just rostral to the tip of the MTs and is limited to the mucosa, without involvement of the cartilage.
Level 2, 3, and 4 sections expose the main nasal chamber and the nasal turbinates. Dorsally at level 2, which is taken slightly caudal to the septal bulge but before the attachment of the EMTs, the dorsolateral wall exhibits a small ridge which is a frequent site of spontaneous pathological changes (Figures 3B and 4H). This dorsolateral ridge continues caudally and may be present on some level 3 sections. Ventrally, there are no vomeronasal organs (VNO) or vestiges of them at the floor of the septum, and only 1 paranasal sinus, the maxillary sinus (MS), is present within the maxilla (Figures 1F and 2D–F) at levels 3 and 4. The relatively large (compared to dogs and rodents) MS has a roughly spherical to polygonal shape, and its floor lies above the roots of the permanent molar teeth while the roof occupies most of the orbital floor (Figure 1F).
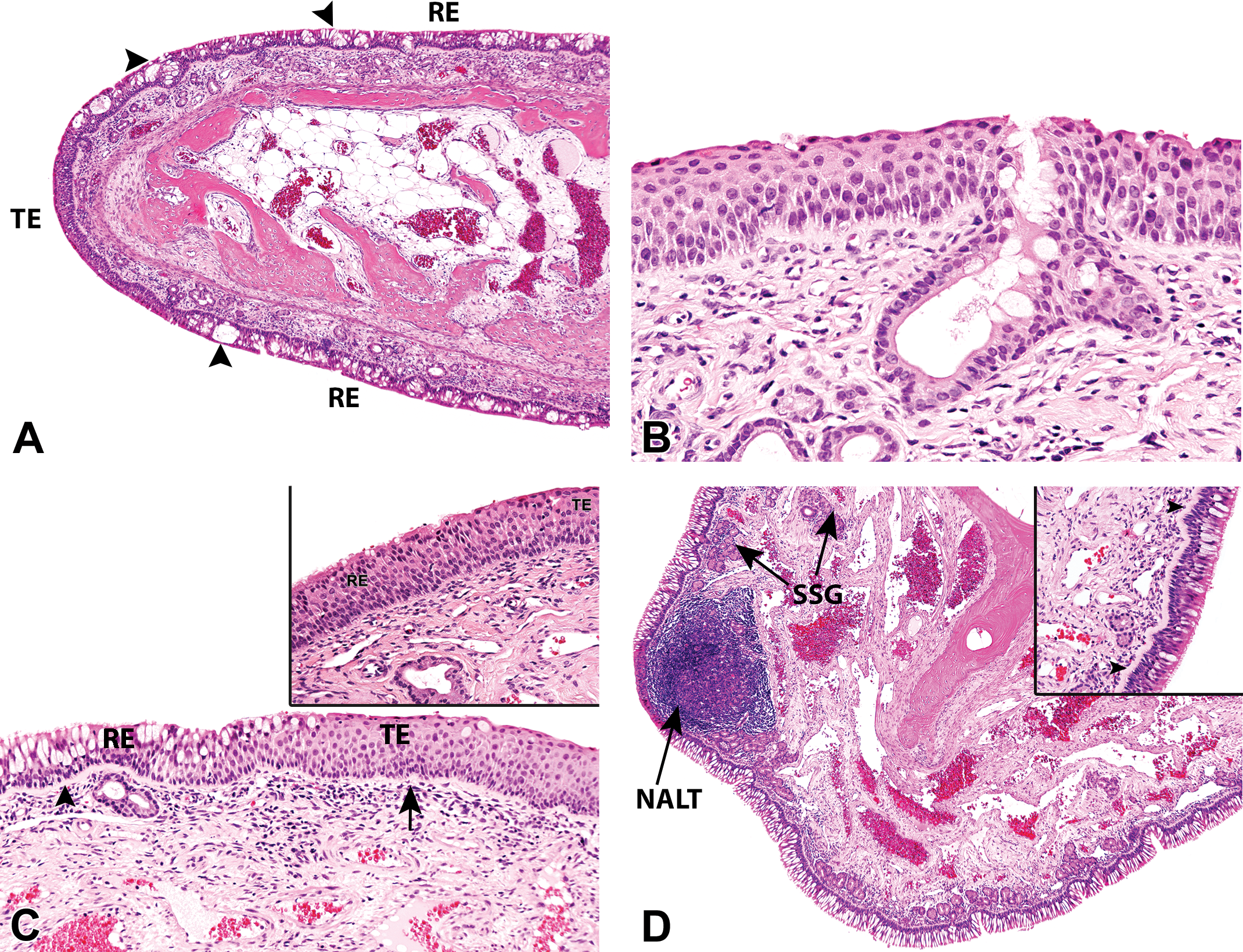
Normal histological appearance of the nasal epithelium in young cynomolgus monkeys (Macaca fascicularis) of 36 to 42 months of age. (A) Transitional (TE) and respiratory epithelium (RE) on the distal ethmoturbinates at level 2 of a male monkey, showing increased goblet cell density with pseudoglands or intraepithelial crypts formation (arrowheads). Hematoxylin and eosin, 4× original objective. (B) TE on the septal bulge at level 1, in a male monkey. Hematoxylin and eosin, 20× original objective. (C) Gradual transition from TE to RE in the rostral lateral meatus at level 2 of the nasal cavity (NC) of a female monkey. There is increased goblet cell density and thickness of the basal lamina (arrow and arrowhead) in the RE. Hematoxylin and eosin, 10× original objective. (C, insert) Gradual transition from TE to RE in the rostral lateral meatus at level 2 of the NC of a female monkey. Higher magnification, hematoxylin and eosin, 20× original objective. (D) The ventromedial surface of the maxilloturbinate of a control female monkey at level 3 of the NC showing a tall, densely ciliated, RE with the highest density of goblet cells in the nose. Submucosal NALT and seromucous glands (SSG), obscured the cartilage and arrowhead are also present. Hematoxylin and eosin, 4× original objective. (D, insert) A higher magnification of 4-D, showing a tall, densely ciliated, RE lying on the thickest basal lamina (arrowheads) of the whole NC. Hematoxylin and eosin, 20× original objective. (E) and (F) Caudal lateral meatus of a young control male monkey at level 3 showing RE with a relatively high goblet cell density and a thick basal lamina (arrowheads). Hematoxylin and eosin, 20× original objective. (G) Ventral maxillary sinus (MS) of a young control male monkey at level 3, showing an increase in goblet cells and the accumulation of intraluminal mucus. Hematoxylin and eosin, 4× original objective. (G, insert) The lateral NC and the adjacent medial surface of the MS of a young control female monkey at level 3, showing the huge difference in the morphology of their respective RE, and the corpora amylacea (arrowhead) in the mucosa of the MS. Besides its characteristic low profile, the RE lining the MS also shows no thick or visible basal lamina. Hematoxylin and eosin, 20× original objective. (H) Septal olfactory epithelium at level 4 of a young control male monkey, showing nuclei of cell types arranged in a roughly lamina pattern from the apical surface to the basal lamina, the sustentacular cell (arrowhead), mature and immature olfactory sensory neurons, and globose and horizontal basal cells (thick arrows). Bowman’s glands and Bowman’s glands/duct cells are also shown. Hematoxylin and eosin, 20× original objective.
The 2 nasal turbinates present in the monkey can be viewed on all coronary sections passing through the main nasal chamber (levels 2, 3, and 4). They appear as 2 pairs of simple and unbranched structures that project ventromedially and dorsally into the meatus from the maxilla and ethmoid bone, respectively (Figures 2 and 3), leaving very little space for free air passage between them and the nasal septum, particularly in the dorsal meatus. Although the MTs appear to arise from the maxilla, subgross histological sections at level 3 reveal that they do not actually arise from the maxilla itself but from a separate, secondary membrane bone that divides the nose into 2 compartments, the nasal meatus and the MS (Figures 1F and 2D). Caudally at level 4, this bony partition is largely absent on the lateral wall of the nasal cavity, so that the MS is separated from the nasal cavity only by a mucous membrane, and the MT has no central bony core (Figures 2F and 3D). Slight interanimal variation may be observed in the shape of the ventral surfaces of the MTs, but the fist-shaped or slightly curved presentation is by far the most common presentation (Figure 2A–F).
Although the EMTs are also present on all sections cutting through the main nasal chamber (levels 2, 3, and 4), at level 2, only the unattached rostral ends, which often float free within the nasal meatus as triangular- or rectangular-shaped structures (Figures 1E and 3B), are present. The interanimal variations in the shape of the ventral surfaces of the EMTs can be appreciated at this level and at level 3 (Figure 2A–C), and these generally show that the long and pointed tip (Figures 1F and 2C–E) is the most common presentation, while the blunt or rounded surface (Figure 2A), or midway in between the two (Figure 2B), are not uncommon.
The largest profile of the EMTs is present on level 3 sections. This level shows that the medial and lateral surfaces of the EMTs have more constant presentations that roughly follow the contours of the septum and the lateral meatus, respectively (Figures 1F, 2B and C, and 3D and E). Therefore, the medial surface of the EMTs consistently appears straight, mirroring the perpendicular shape of the nasal septum, while the lateral surface has a curved appearance that conforms to the curved contours of the lateral nasal wall (Figures 2B and C and 3E), with very little interanimal variation. Besides the variations in the shape, the dorsoventral relationship of the 2 turbinates also differs slightly among individuals, with some EMTs extending further ventrally to overlap or reach the same ventral limits as the MTs (Figure 2E).
Level 4 sections reveal that the monkey’s nasopharynx lies caudal and not ventral to EMTs; therefore, a transverse lamina that divides the EMTs from the nasopharyngeal passageway in other laboratory animals is absent (Figure 3F and G). Near this caudal edge of the nasal cavity, septal deviations may be observed at the bone–cartilage interface, where the septal cartilage is reduced to its smallest size in the middle of the septum (Figure 3E and F). They are differentiated from artifactual damage by the presence of some tissue reaction, including fibrosis and hemorrhage on histology (Figure 3E). A septal window as seen in rodents is not present at this or any other level.
The main gross anatomic differences between the nasal cavities of the cynomolgus monkey and humans can therefore be summarized as the presence of just 1 paranasal sinus (no frontal, sphenoid, or ethmoid sinuses), 2 instead of 3 turbinates, a slightly elevated vestibule, the presence of NPDs with no vestiges of the VNO, and the presence of a median septum that continues to about two-thirds of the nasopharynx, in monkeys.
Histology
The general distribution of epithelial types at each level of the monkey nose is summarized in Table 1. The epithelial distribution at each level can also be influenced by the interanimal variations and tangential sectioning. However, as in other laboratory animals, 4 main types of epithelial populations, squamous (SE), transitional (TE), respiratory (RE), and olfactory epithelium (OE), line the vestibule, nasal chamber, and turbinates in the cynomolgus monkey.
The rostral vestibule (which is not always examined) is completely lined by a stratified SE, but further inward at level 1, the SE often becomes restricted to the dorsal and dorsolateral regions (Figure 3A). The relatively thin SE present in the dorsal regions at this level gives way to a TE mainly in the midseptal, midlateral, and ventrolateral regions (Figure 3A). However, in some individuals, the ventral parts of the vestibule at level 1 may also be lined by a SE that is often thicker than that lining the dorsal regions, possibly due to oblique sectioning.
The SE covering the monkey vestibule is a nonkeratinized- or lightly keratinized-stratified epithelium composed of basal cells lying on a barely visible basal lamina and 5 to 8 layers of more flattened cells toward the surface. In general, there is a rostral to caudal decrease in the thickness of the SE, so that the thinnest SE, with keratohyaline granules and pigmented keratinocytes, is observed in the dorsal regions, where it extends more caudal than in the ventral regions. The thickest SE is present in the most rostral parts and on the midseptal or midlateral regions of the vestibule where it abruptly transitions to TE. This abrupt transition from SE to TE also marks the sudden appearance of a slightly thicker basal lamina lying beneath the TE (Figure 4B and C).
The nasal TE of the cynomolgus monkey is a stratified, nonciliated cuboidal to low columnar epithelium of 4 to 6 cell thickness (Figure 4A–C), which is mostly devoid of goblet cells. It is intermittently present over a relatively wide longitudinal (rostrocaudal) area that extends from as far forward as the vestibule at level 1 to as far back as the middle or rostral 3rd of the nasal cavity at levels 2 and 3. The most commonly affected areas are the septum and lateral meatus of the vestibule (Figure 4B and C), the angular surfaces and rostral ends of turbinates (Figure 4A), much of the rostral nasal septum, and less frequently, the ventral vestibule. Septal TE is most commonly observed in the middle regions, along the septal bulge at level 1, where it is usually at its thickest (Figure 4B), while turbinate TE is most commonly observed on the 2 or more angular surfaces of the unattached turbinates at level 2 (Figures 3B and 4A) or on smaller areas of the ventral and medial surfaces of the caudal EMTs (Figure 3E) at level 3. On the MTs, it is often restricted to a small area at the rostral limits of the turbinate, which is usually not present on any of the sections.
The distribution of TE on the EMTs suggests that in the cynomolgus monkey, TE, or a TE-like nonciliated epithelium with a low profile and scant goblet cells, may be encountered in certain areas of the main nasal chamber away from the rostral region. This TE-like epithelium may also be observed on the dorsolateral ridge, dorsal to the nasal atrium (Figures 1E and 3B), and just before the transition from RE to OE. Since these regions are frequently associated with inflammatory changes in the lamina propria (as regions of high particle impaction), it is often difficult to determine whether the morphology of this epithelium represents a normal appearance or a metaplastic change.
The nasal RE in the cynomolgus monkey is a tall, pseudostratified ciliated columnar epithelium that lines the largest surface area in the nasal cavity and is therefore present on all 4 levels (levels 1–4). It lies on a microscopically visible and strikingly thick basal lamina (Figure 4C–F), which appears as a thick amorphous eosinophilic structure that often obscures cellular detail on tangential sections (Figures 4F and 5H). The basal lamina of the RE is much thicker than that beneath the TE and can therefore be used to demarcate the start of RE at the transition from TE (Figure 4C) or the end of the RE at the RE/OE transition zone. The transition from TE to RE is otherwise gradual and almost imperceptible, although the scarcity of goblet (mucous) and ciliated cells may be used to identify the TE. The RE is also on average taller than the TE.
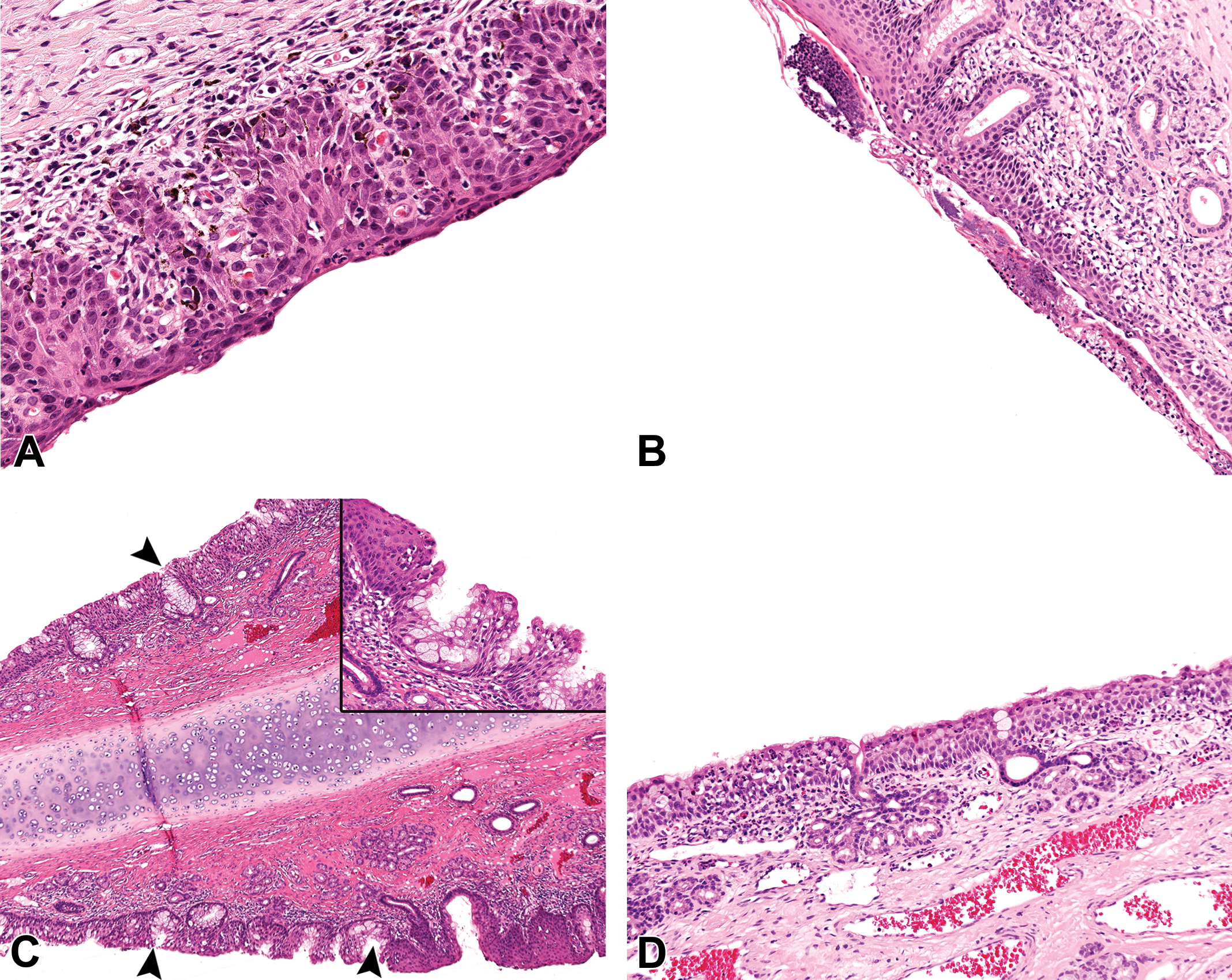
Spontaneous findings of the squamous (SE), transitional (TE), and respiratory epithelium (RE) in the NC of young cynomolgus monkeys (Macaca fascicularis) of 27 to 42 months of age. (A) Rostral vestibule of a young control female monkey, showing inflammation and hyperplasia of the SE with rete pegs formation. Hematoxylin and eosin, 20× original objective. (B) Vestibule of a young control male monkey at level 1, showing inflammation and scabs on the lateral wall at the SE-TE epithelial junction. Hematoxylin and eosin, 10× original objective. (C) The rostral nasal septum (SP) of a young control male monkey at level 1, showing mucous cell hyperplasia/metaplasia of the TE with pseudogland formation (arrowhead and insert) and hyperplasia of the SE. Hematoxylin and eosin, 10× original objective (insert) and hematoxylin and eosin, ×20 original objective. (D) Degeneration and attenuation of the RE with minimal early SE metaplasia in the lateral wall of the rostral nasal cavity (NC) of a young control female monkey at level 2. Hematoxylin and eosin, ×10 original objective. (E) Inflammation and attenuation of the RE on the medial surface of the ethmoturbinate at level 2 of the NC of a young control male monkey. Hematoxylin and eosin, ×20 original objective. (F) Inflammation and glandular microabscess formation in the RE that lies adjacent to the olfactory epithelium on the caudodorsal SP at level 3 of a young control male monkey. Hematoxylin and eosin, ×20 original objective. (G) Early SE metaplasia or attenuation of the RE, in the lateral wall of the rostral NC at level 2 of a male monkey. Note the much thickened basement membrane that identifies this as the RE (arrowheads). Hematoxylin and eosin, ×20 original objective. (H) RE hyperplasia with rosette formation and marked thickening of the basal membrane (arrows) in the caudal dorsolateral angle at level 3 of the NC of a young control female monkey. Hematoxylin and eosin, ×10 original objective.
The RE is composed of basal cells, ciliated columnar cells, goblet cells, and nonciliated columnar cells. Goblet cells are abundant in the RE lining both the nasal meatus and the MS. In the MS, they are more concentrated on the ventral aspect where mucus often collects (Figure 4G). The highest density of goblet cells however is found on the medial wall of the MTs, which also has the tallest and most densely ciliated RE, and the thickest basal lamina (Figure 4D). Goblet cells are also abundant in the RE lining the caudal ventral meatus, the caudal nasal septum, and the nasopharynx. There is therefore a general increase in the density of goblet cells from rostral to caudal, particularly on the septum. Increased goblet cell density with pseudogland formation and mucous cell metaplasia of the submucosal glands are a common feature on the MTs and the septum.
In terms of epithelial height, the RE lining the ventral and the ventrolateral meatus (Figure 4G), and to some extent the septum, is of a relatively lower height than that found on the turbinates and the lateral and dorsal walls, while that covering the MS shows the lowest profile (Figure 4G; less than half the height of the RE on the MTs).
The monkey OE lines only a small part of the caudodorsal meatus and the dorsal attachments of EMTs. Therefore, on nasal sections produced by the method described in this article (Figure 1A), the rostral dorsal meatus and the unattached EMTs at level 2 are usually not lined by OE (Figure 3H), and the neuroepithelium first appears on the level 3 section. However, serial trimming of further coronal sections and examination of parasagittal sections show that the most rostral limits of the OE lie somewhere in between levels 2 and 3; and at this level, OE lines just the dorsal meatus and approximately a dorsal 5th of the septum. It first appears on the EMTs at their attachments to the dorsal meatus and lines the medial surface first, covering approximately nine-tenths of the dorsomedial surface, in addition to the dorsal half of the septum (Figure 3C and D). The lateral surface of the EMTs (dorsal 3rd) and a small part of the dorsolateral wall are lined by OE only in the caudal most regions (Figure 3C) at level 4. Thus, at any given level, the OE lines larger areas of the medial surfaces of the EMTs and the nasal septum, than it does the lateral aspects of the EMTs and the lateral nasal wall. There is therefore on level 3 or 4 sections (mainly level 3), a dorsolateral angle formed by the lateral surface of the EMTs and the lateral wall that is never fully lined by OE, but mainly by a poorly defined epithelium that resembles both RE and OE, and is often a site of epithelial inflammation or vacuolation (Figure 6D).
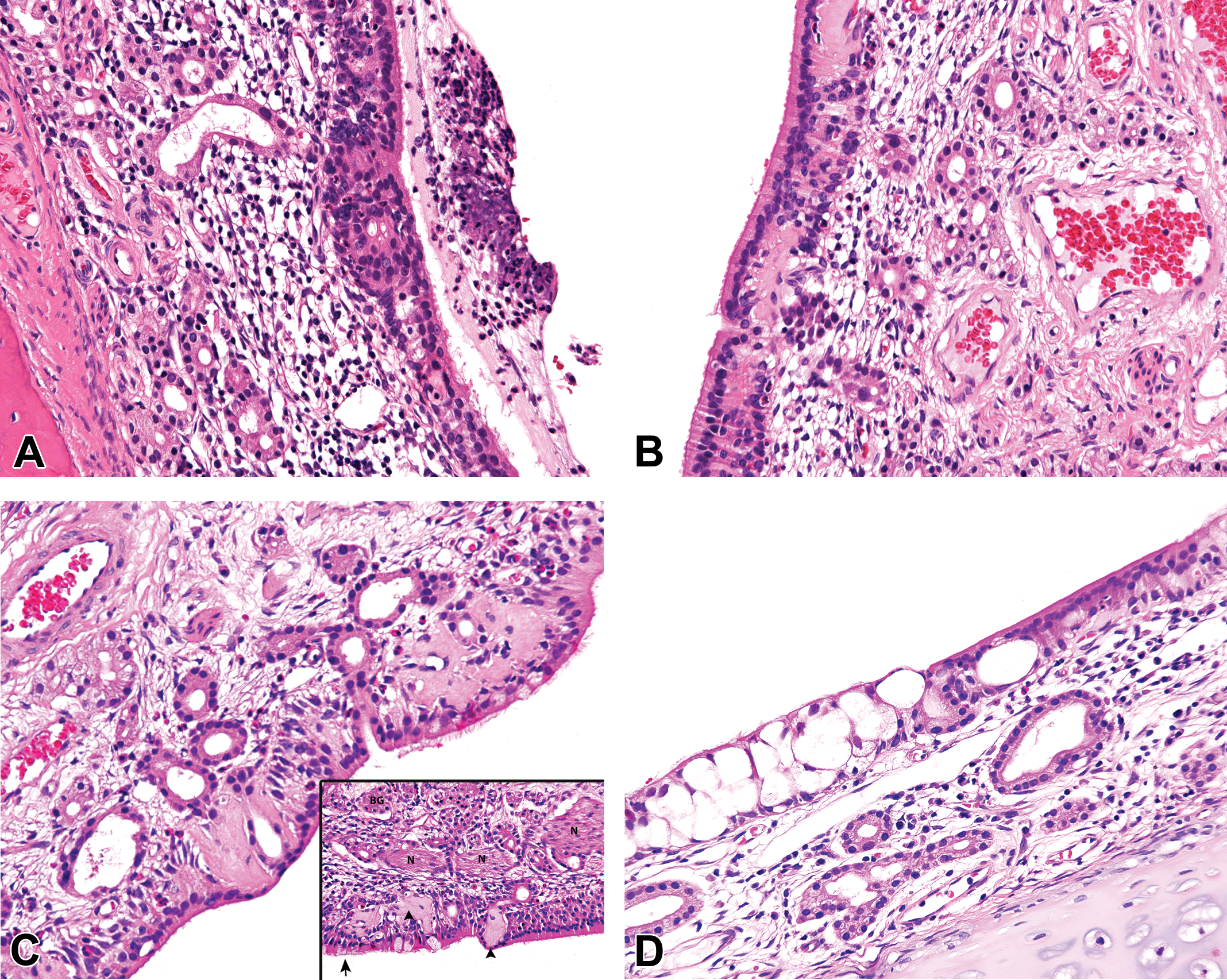
Spontaneous findings of the olfactory epithelium (OE) in the nasal cavity (NC) of young control cynomolgus monkeys (Macaca fascicularis) of 27 to 42 months of age. (A) OE inflammation and degeneration in the dorsolateral meatus of a young control female monkey at level 4. Hematoxylin and eosin, ×20 original objective. (B) OE degeneration, with cell-specific necrosis of olfactory sensory neurons, and sparing of sustentacular cells, in the dorsal meatus at level 4 of the NC of a young female monkey. Minimal inflammation is also present. Hematoxylin and eosin, ×20 original objective. (C, insert). OE degeneration with respiratory epithelial metaplasia and deposition of amyloid-like eosinophilic material in the dorsolateral meatus of a young control female monkey at level 4. Hematoxylin and eosin, ×20 original objective. (D) OE vacuolation at the respiratory–olfactory epithelial border on the dorsal nasal septum of a young female monkey at level 4. Minimal inflammation is also present. Hematoxylin and eosin, ×20 original objective. (E) OE hyperplasia with rosette formation and respiratory metaplasia, at the respiratory–olfactory epithelial border, in the dorsolateral angle of the caudal NC of a young female monkey at level 3. Hematoxylin and eosin, ×20 original objective. (F) Respiratory metaplasia/hyperplasia with hyperplasia of Bowman’s glands, rosette formation, and deposition of amyloid-like material at the respiratory–olfactory epithelial border in the dorsolateral angle of the caudal NC at level 3 of a young male monkey. Hematoxylin and eosin, ×20 original objective. (G) Inflammation at the border between the respiratory epithelium (RE) and OE on the dorsolateral ridge of the caudal NC at level 3 in a young male monkey. Note the thick basal lamina (arrowhead) beneath the RE. Hematoxylin and eosin, ×20 original objective. (H) Pigment in the olfactory mucosa of the caudodorsal meatus at level 4 of the NC in a young control female monkey. Hematoxylin and eosin, ×20 original objective.
In general, the transition from RE to OE is not well defined, and some clusters of ciliated RE cells may be found within the OE, making it difficult to distinguish such normal features from respiratory metaplasia of the OE (Figure 6E and F). Since the basal lamina of the OE is not readily visible, the absence of a thickened basal lamina and the presence of Bowman’s glands and nerve bundles are helpful in identifying metaplastic OE.
The OE of the cynomolgus monkey is a very tall pseudostratified columnar epithelium of up to twice the height of the RE. It is composed of 5 cell types whose nuclei are lined in a roughly lamina pattern in the following order: from the apical surface to the basal lamina, the sustentacular cell, mature and immature olfactory sensory neurons (OSNs), and globose and horizontal basal cells, respectively (Figure 4H). In addition, Bowman’s glands/ducts cells extend through the epithelium from the lamina propria to the apical surface, constituting the 6th cell type. There are no goblet cells and the mucus covering the luminal surface of the OE is produced and secreted by the subepithelial Bowman’s glands.
Three main types of submucosal glands are present as in other animals: serous glands, seromucous glands, and Bowman’s glands of the olfactory mucosa. However, the lateral nasal (Steno’s) glands (of the dog and rodents) are not present. Submucosal glands are more abundant in the rostral septum, lateral walls, and the MTs. Rostral septal glands have ducts that extended rostrally to open in the vestibule, while a few glands are also present in the submucosa of the vestibule itself. Besides the large number of submucosal glands, the submucosa of the MTs also typically contains large and abundant venous sinuses and foci of cartilage as normal features (Figures 2A and 4D). In the olfactory mucosa, dark submucosal pigment is often observed affecting the mucous glands, sustentacular cells, connective tissue cells, nerve bundles, and the Bowman’s gland epithelium (Figure 6H). This pigment is believed to be responsible for the grossly visible yellow color of the olfactory mucosa or EMTs.
Most of the nasopharynx is lined by a low RE, but a TE is present at the level of the exits of the Eustachian tubes. The septal wall of the nasopharynx contains large amounts of nonencapsulated tonsillar lymphoid tissue (Figure 3G) analogous to the Waldeyer’s ring in man. The tonsillar tissue and nasal associated lymphoid tissue (NALT) are also often present in the lateral walls of the nasopharynx. The NALT in the cynomolgus monkey is not confined to a particular region but can be found anywhere throughout the nasal cavity, including the ventral meatus (Figures 2D and 7D), dorsal meatus (Figure 3H), MTs (Figure 2D), or the vestibule (Figure 7C). The NALT of the vestibule often causes partial occlusion of the nasal passages in some animals (Figure 7C).
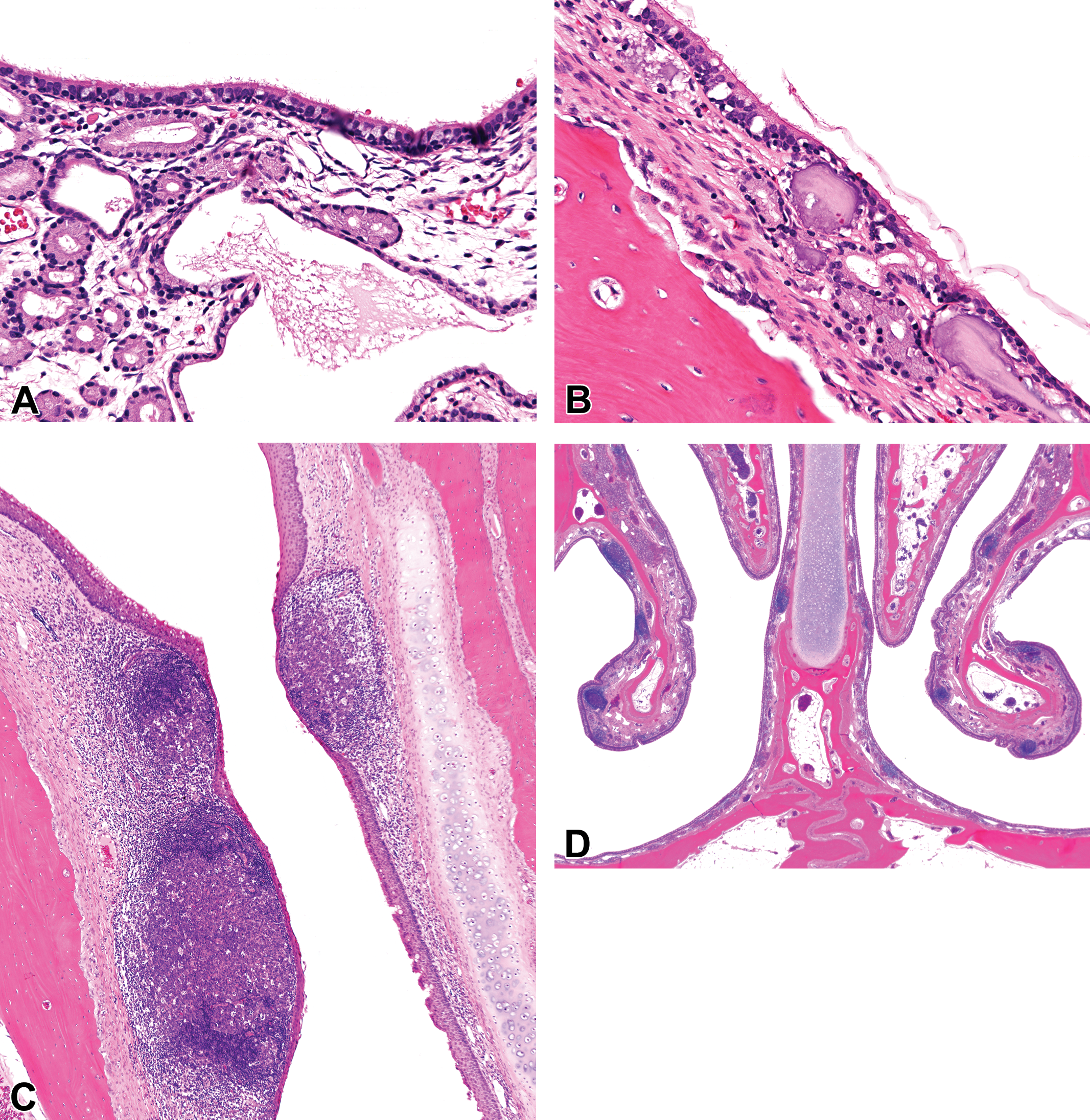
Miscellaneous background findings in the nasal cavity (NC) of young control cynomolgus monkeys (Macaca fascicularis) of 27 to 42 months of age. (A) Dilatation of submucosal glands in the maxillary sinus (MS) of a young male monkey at level 3 of the NC. Hematoxylin and eosin, ×20 original objective. (B) Corpora amylacea in the subepithelium of the MS of a young male monkey at level 3 of the NC. Hematoxylin and eosin, ×20 original objective. (C) Rostral vestibule of a young female monkey showing nasal associated lymphoid tissue (NALT) hyperplasia, with partial occlusion of the nasal passages. Hematoxylin and eosin, ×4 original objective. (D) Multifocal NALT hyperplasia in the nasal cavity of a young male monkey at level 2 of the nasal cavity. Hematoxylin and eosin, ×1 original objective.
Background Pathology
The most common spontaneous pathology findings observed in the nasal cavity of young cynomolgus monkeys are presented in Tables 2 to 5. The findings were recorded on the basis of the anatomic (e.g., ventrolateral meatus) and functional (epithelial type) rather than the section level, in order to avoid repetition. The most common findings were inflammatory lesions, which showed a greater predilection for the rostral nasal cavity, the dorsolateral ridge (Figure 3B), the ventral and medial surfaces of the EMTs, and other regions of epithelial transition. They were invariably associated with other degenerative, reparative, and adaptive changes in the nasal epithelium, which were often not recorded separately. These included squamous or transitional cell hyperplasia (Figure 5A), RE attenuation and squamous metaplasia (Figure 5D, E, and G), mucous cell hyperplasia/metaplasia (Figure 5C), OE degeneration/atrophy (Figure 6A–C), OE vacuolation (Figure 6D), RE hyperplasia (Figure 5H), and olfactory epithelial hyperplasia (Figure 6E). The inflammatory lesions reached high incidences of up to 100% of the animals in the studies where they occurred. However, the prevalence over the 11 studies (data not shown) was not as high as the total incidences within individual studies. The severity grades ranged from minimal to moderate.
Incidences of Pathological Findings in the Transitional Epithelium of the Nasal Cavity in 114 Control Cynomolgus Monkeys (Macaca fascicularis) from 11 Inhalation Studies of 4- to 13-Week Duration.
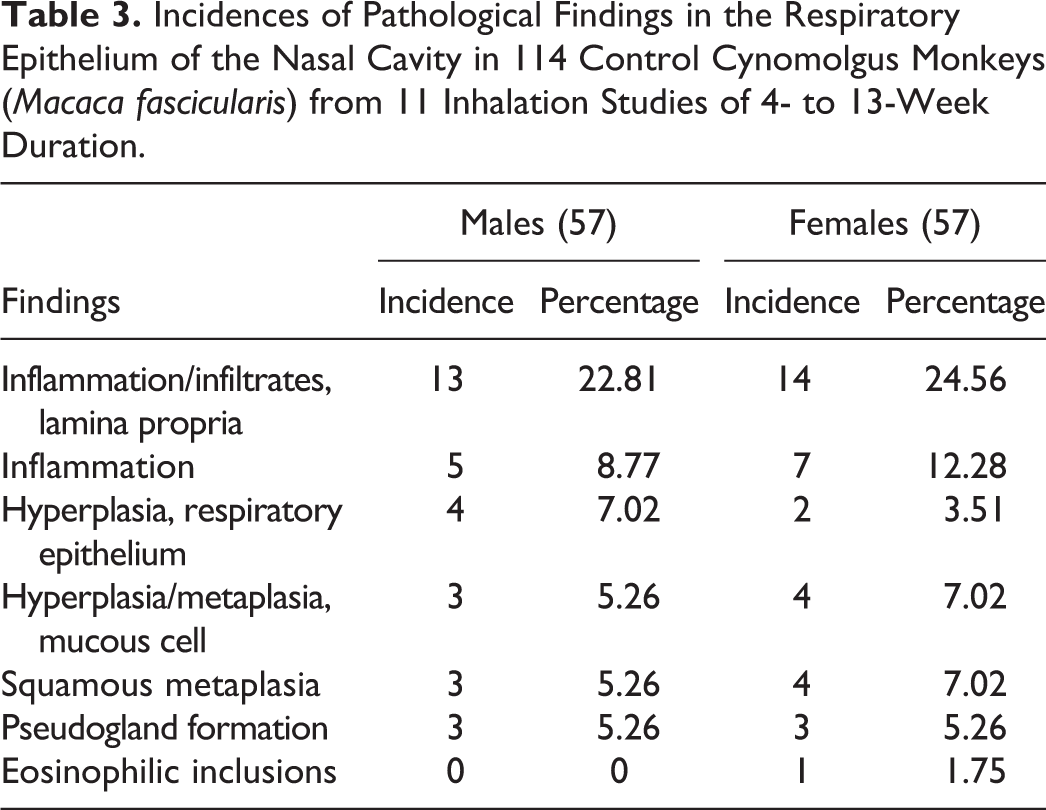
Incidences of Pathological Findings in the Respiratory Epithelium of the Nasal Cavity in 114 Control Cynomolgus Monkeys (Macaca fascicularis) from 11 Inhalation Studies of 4- to 13-Week Duration.
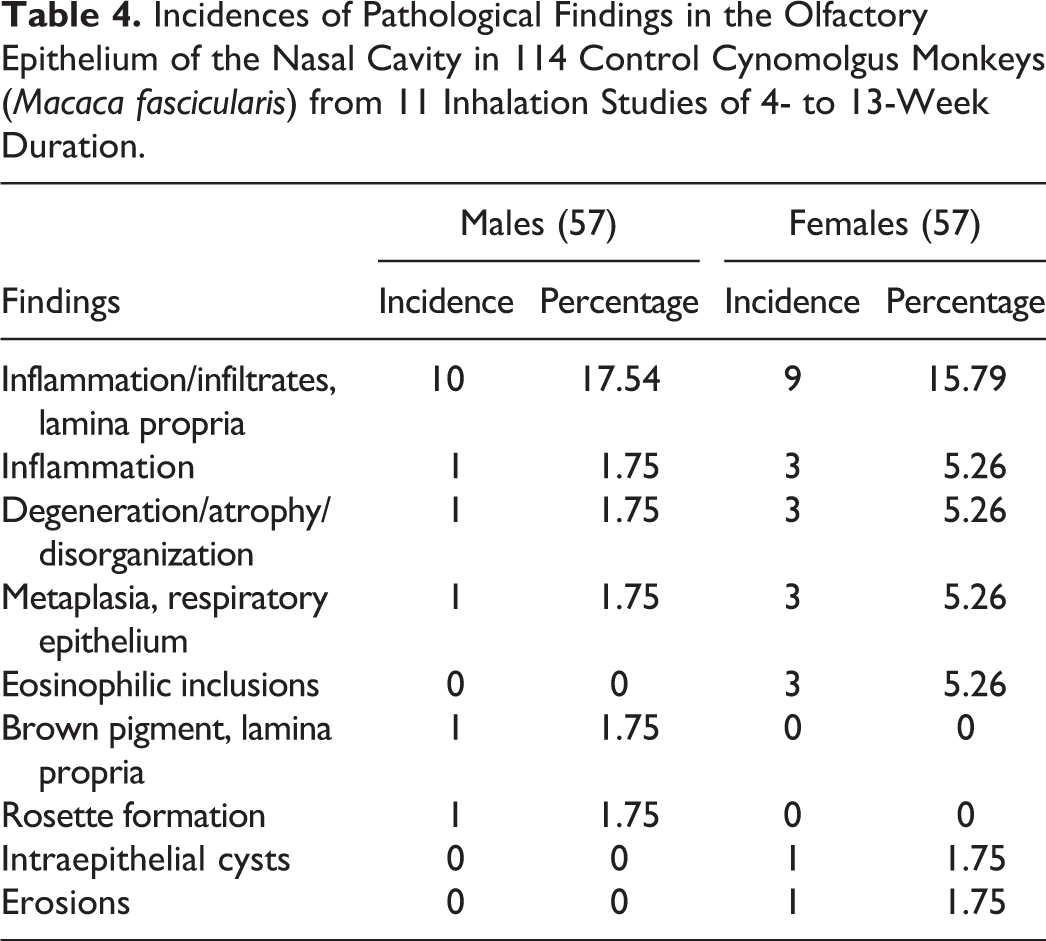
Incidences of Pathological Findings in the Olfactory Epithelium of the Nasal Cavity in 114 Control Cynomolgus Monkeys (Macaca fascicularis) from 11 Inhalation Studies of 4- to 13-Week Duration.
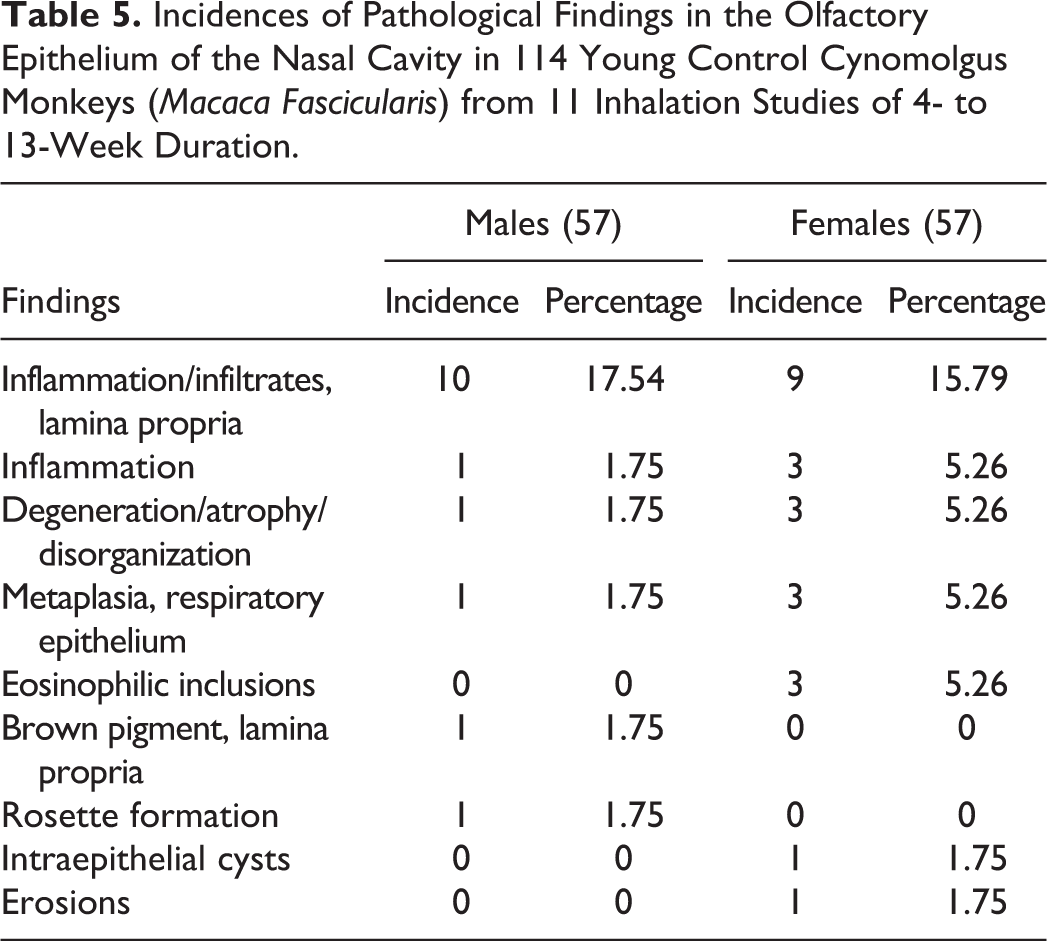
Incidences of Pathological Findings in the Olfactory Epithelium of the Nasal Cavity in 114 Young Control Cynomolgus Monkeys (Macaca Fascicularis) from 11 Inhalation Studies of 4- to 13-Week Duration.
Lesions of the SE
In a large number of the animals, the midlateral or midseptal regions of the vestibule or at the transition from SE to TE at level 1 were covered with scabs or focal inflammatory exudate (Figures 3A and 5B). The lesion at this site was one of the main causes of high incidences of inflammatory lesions in studies and was considered to be associated with nose picking. Associated findings included mixed inflammatory cell infiltrates in the lamina propria, squamous cell hyperplasia with rete peg formation (Figure 5A), and goblet cell metaplasia in the SE/TE transitional zone.
Lesions of the TE and RE
The most common findings in the TE and RE were inflammation, squamous metaplasia, mucous cell hyperplasia/metaplasia, and respiratory hyperplasia. Rostrally, inflammation was most commonly observed in the regions of transition from TE to RE in the midseptum or the septal bulge, the midlateral region, and the rostral ends of the turbinates (Figure 5E). Caudally, inflammation of the RE most commonly occurred in the dorsolateral ridge at level 2 or 3, the regions of transition from RE to OE in the dorsolateral angle or dorsal septum (Figures 3B and 5F), and the ventral/ventromedial surfaces of the EMTs. Inflammation in the dorsolateral ridge and the regions of RE/OE interface was often associated with intraepithelial microabscesses and cysts (Figure 5F), respiratory hyperplasia with rosette formation and further thickening of the basal lamina (Figures 5H and 6F), and epithelial vacuolation (Figure 6D).
Squamous metaplasia (Figure 5G) also occurred commonly in the regions of transition from TE to RE in the midlateral and midseptal regions of the rostral nasal cavity or vestibule, the rostral ends of the turbinates, or on other angular surfaces of the EMTs. In its earliest form, minimal squamous metaplasia was characterized by subtle degenerative changes in the RE such as disorganization and attenuation of the epithelium with superficial cells showing an altered polarity toward a horizontal orientation, loss of cilia, and a reduction in the number of goblet cells (Figure 5D and E). As a result, the RE initially resembled the cuboidal TE lying adjacent to it, giving the impression of an extension of the TE to regions normally occupied by RE. Therefore, some degenerative changes of the RE or early forms of squamous metaplasia in the regions of TE/RE transition were either missed or recorded as TE extension or hyperplasia. Attenuation of the TE and/or RE was also often accompanied by inflammatory cell infiltrations into the lamina propria (Figure 5D and E). Since this finding was considered a precursor to squamous metaplasia, minimal grades of squamous metaplasia or inflammatory cell infiltrates were usually diagnosed.
Goblet cell hyperplasia and metaplasia were often combined as a single term, but in other studies, a distinction was made between the 2 terms. In the latter cases, goblet cell metaplasia was recorded in the TE/RE zones on the septum, where it was characterized by an increase in the number of goblet cells in an epithelium that normally contains scant goblet cells (Figure 5C). Goblet cell metaplasia of the TE was often associated with acute inflammation and influx of neutrophils into the lamina propria, epithelium, or lumen. Goblet cell hyperplasia on the other hand was used to describe an increase in the density of goblet cells in regions which normally have a high goblet cell density, such as the MT, MS (Figure 4D and G), or the caudal ventrolateral meatus (Figure 4E and F). Goblet cell hyperplasia/metaplasia was characterized by mucosal folding and pseudoglands or intraepithelial crypts formation (Figure 5C and F). Rupture of hypertrophic goblet cells in these regions resulted in intraepithelial mucus-filled cysts (Figure 5F).
Lesions of the OE
As with the other epithelial types, inflammatory lesions were the most commonly observed changes in the OE, followed by degeneration/atrophy and respiratory metaplasia. Inflammation and degenerative changes often occurred together in the olfactory mucosa and were usually characterized by varying grades of polymorphonuclear or mixed cell infiltration into the lamina propria or intraepithelial (Figure 6A–E), luminal inflammatory exudate (Figure 6A), OE degeneration, (Figure 6B and C), and the intraepithelial elaboration of an amyloid-like eosinophilic material (Figure 6B and C). The earliest degenerative change observed in the OE was atrophy/disorganization, which was characterized by the disruption of the laminar pattern of the nuclei of the OSNs (Figure 6B) and a vacuolated appearance of the epithelium caused by a nonspecific loss of either OSNss or sustentacular cells. This was followed by thinning or attenuation of the mucosa. An eosinophilic proteinaceous material or cellular debris overlying the attenuated mucosa were usually present in the lumen (Figure 6A) and were often used to differentiate genuine OE degeneration from artifactual thinning of the epithelium. More chronic changes were associated with basal cell hyperplasia, rosette formation, and/or respiratory metaplasia (Figure 6C, E, and f).
Other miscellaneous findings included the frequently observed corpora amylacea in the wall of the MS (Figures 4G and 7B), submucosal glandular metaplasia, and dilatation of submucosal glands of the MS (Figure 7A), NALT hyperplasia (Figure 7C and D), OE pigment, and eosinophilic inclusions in the RE. Corpora amylacea started as mineral deposition in the glands of the MS and was present in almost every animal.
Discussion
The nose is a complex organ with many functions that include olfaction, warming, humidifying, and filtering of the inspired air (Negus 1958). Its anatomy varies greatly between laboratory animals and man. The anatomical differences may affect the occurrence and distribution of lesions following exposure to inhaled nasal toxicants. Differences in the gross anatomy, such as the turbinate structure, folds, or grooves on nasal walls and presence or absence of accessory structures, may influence nasal airflow and therefore species-specific uptake and deposition of inhaled material. In addition, interspecies variations in the morphological composition and distribution of the nasal epithelium may influence the local tissue susceptibility resulting in lesions with a species-specific distribution pattern. The species- and site-specific responses to inhaled compounds may in turn affect the interpretation of results from inhalation toxicity studies and the extrapolation of data from experimental animal studies to human risk assessment.
We have described the normal anatomy and background pathology of the nasal cavity of the cynomolgus monkey, including the characteristic gross features and the interanimal variations, the light microscopic characteristics and distribution of the epithelial types, and the nature and incidences of spontaneous pathological changes. The results indicate that, compared to other laboratory animals, the nasal gross anatomy of the cynomolgus monkey is more similar to that of humans (DeSesso 1993; Lucas 1932; Negus 1958). The main gross anatomic differences to dogs and rodents are that the monkey nose shows the wide interanimal variations in the shape and size; fewer and less complex turbinates; and an absence of a vomeronasal organ, a transverse lamina and olfactory recess, Steno’s glands, and vestibular alar folds or atrioturbinates.
This description of the anatomy of the cynomolgus monkey nose is closely similar to that described for other macaque monkeys in the literature (Harkema, Plopper, Hyde, Wilson, et al. 1987; Kepler et al. 1998; Lucas 1932). Any minor differences between this study and previous studies could be attributed to species differences. For instance, in the rhesus macaque, variations in the shape of the turbinates were observed on the medial surface of the EMTs, while the MTs were reported to be more uniform (Lucas 1932). In this study, the biggest variations in the shape of the turbinates were observed on the ventral surface of both turbinates. The medial and lateral surfaces were uniform and generally followed the contours of the septum and the lateral wall, respectively. Species differences between the rhesus and the cynomolgus macaques could therefore account for these minor differences in the description of the gross anatomy.
Similarly, the histological appearance and distribution of the nasal epithelium is also consistent with what has been reported for other macaque monkeys in the literature (Harkema, Plopper, Hyde, Wilson, et al. 1987; Monticello et al. 1989). The study also confirms that, as with other macaque monkeys, the morphology and distribution of the nasal epithelium in the cynomolgus monkey is closer to that of humans than that of rodents and dogs (Chamanza et al. 2015; DeSesso 1993; Halama et al. 1990; Harkema, Plopper, Hyde, Wilson, et al. 1987). Briefly, the major histological differences between monkeys and rodents include a generally thicker epithelium of all subtypes in monkeys, a wider area lined by TE, and a much reduced area lined by OE.
One of the main objectives of this study was to provide readily accessible reference material on the common background changes in the nasal cavity of control cynomolgus monkeys, which could be used as an aid when evaluating inhalation studies. This included describing the findings, the incidences and the most affected regions, and therefore the possible regions of toxicological significance in the cynomolgus monkey nose. The results indicate that the rostral nasal cavity, areas of particular impaction (bulges and ridges), and areas of epithelial transition were the most affected regions and these included: the midlateral region within the vestibule or rostral nasal cavity, the midseptal regions or septal bulge in the rostral nasal cavity, the dorsolateral ridge (or vestige of the nasoturbinate) that lies dorsal to the nasal atrium, the rostral margins and medial or ventral surface of the EMTs, and the region of transition from RE to OE in the dorsolateral angle or on the adjacent septum.
These regions are largely consistent with those known to be affected by inhaled irritants such as ozone (Harkema, Plopper, Hyde, St. George, et al. 1987) and formaldehyde (Monticello et al. 1989) in monkeys, and the spontaneous pathology changes observed in these regions were also similar to those induced by nasal irritants. Since the lesion distribution patterns associated with nasal irritants are mostly attributable to regional deposition as a result of airflow patterns (Morgan and Monticello 1990) and an inherent susceptibility of the epithelium, such as the reduced mucosubstances in the TE (Harkema, Carey, and Wagner 2006), this suggests that nasal airflow characteristics and tissue susceptibility may also play a role in the distribution of spontaneous lesions in the nasal cavity of the cynomolgus monkey. It also suggests that, although the exact etiology of the spontaneous nasal lesions in juvenile monkeys is not known, exposure to environmental factors such as smog and other impurities in the urban air could be the contributing factors. Ozone, which causes similar nasal responses to those observed in our study (inflammation, epithelial hyperplasia, and mucous cell hyperplasia of the TE) in both laboratory animals and humans (Nikasinovic, Momas, and Seta 2003) is the principal oxidant pollutant in smog. Differentiating these background changes from induced lesions can be a challenge.
Footnotes
Author Contribution
Authors contributed to conception or design (RC); data acquisition, analysis, or interpretation (RC, IT, MG, CH, MS, JG, KG, MA, JS, VM); drafting the manuscript (RC); and critically revising the manuscript (RC, IT, MG, CH, MS, JG, KG, MA, JS, VM). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
