Abstract
The testes from 136 male cynomolgus monkeys were examined histopathologically in order to investigate the relationship between the development of spermatogenesis and testis weight, age, and body weight. At Grade 1 (immature), Sertoli cells and spermatogonia were the only cell classes in the testis. At Grade 2 (pre-puberty), no elongated spermatids were observed in the testis, although a few round spermatids and small lumen formation were observed. At Grade 3 (onset of puberty), all classes of germ cells were observed in the testis, although seminiferous tubule diameters and numbers of germ cells were small. Slight debris in the epididymis was observed in almost all animals. At Grade 4 (puberty), almost complete spermatogenesis was observed in the seminiferous tubules and it was possible to ascertain the spermatogenesis stage as described by Clermont, although tubule diameters and numbers of germ cells were small. There was less debris in the epididymis than at Grade 3. At Grade 5 (early adult), complete spermatogenesis was observed in the seminiferous tubules. At Grade 6 (adult), complete spermatogenesis in the seminiferous tubules and a moderate or large number of sperm in the epididymis were observed. Moreover, sperm analysis using ejaculated sperm was possible. Logistic regression analysis showed that testis weight is a good indicator of testicular maturity.
Introduction
The cynomolgus monkey is a frequently used nonrodent species in single- and repeated-dose toxicity studies. However, almost all animals used in such studies are relatively young cynomolgus monkeys, in which testicular toxicity cannot be appropriately assessed to estimate potential risk in men. According to an International Conference on Harmonization (ICH) M3(R2) document (available at http://www.ich.org/fileadmin/Public_Web_Site/ ICH_Products/Guidelines/ Multidisciplinary/M3_R2/Step4/M3_R2__Guideline.pdf), an evaluation of male reproductive organs is performed in repeat-dose toxicity studies before inclusion of men in clinical trials. This assessment can be conducted by a histopathological examination of the testis in repeated-dose toxicity studies. When only the nonhuman primate is the relevant species, the potential effects on male reproductive function should be assessed in studies using sexually mature animals. Cynomolgus and rhesus monkeys are widely used in nonhuman primate toxicity studies; however, cynomolgus monkeys are considered to be the more appropriate species because rhesus monkeys are seasonal breeders, which makes it difficult to set the time of year in which to dose the animals and the duration of dosing.
The initiation of spermatogenesis occurs at a similar stage in individual rats, and testicular maturity can be observed histologically in almost all Sprague-Dawley rats at ten weeks of age. Conversely, there are large individual differences in cynomolgus monkeys; both mature and immature testes can be observed in animals at three to five years of age.
In this report, the testes were classified into six grades according to the histopathological findings, and the relationships between the degree of testicular maturity based on histology, body weight, age, and testis weight were investigated.
Materials and Methods
Testes from 136 male cynomolgus monkeys (purpose-bred, source: China) were used for this experiment. These animals, aged thirty-eight to eighty-three months and weighing 3.03 to 6.89 kg, had been used as control animals in repeated-dose toxicity studies conducted at Shin Nippon Biomedical Laboratories, Ltd, Kagoshima, Japan, and they were euthanized between September 2008 and August 2009. All animals had been housed individually in an environment with controlled temperature (23°C–29°C) and humidity (35–75%), and they had been provided with 108 g of solid food daily (HF Primate 5K91 12G 5K9 J, Purina Mills, LLC). Freshly collected testes were weighed individually, fixed in Bouin’s solution, and then re-fixed in 10% neutral buffered formalin. Freshly collected epididymides were weighed and fixed in 10% neutral buffered formalin. In accordance with routine histological methods, these organs were embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H.E.). Additionally, selected sections of the testes were also stained with periodic acid–Schiff (PAS). The testes and epididymides were examined under the light microscope, focusing on spermatogenesis development. The testes were classified into six grades according to the histopathological findings (Table 1), based on available literature (Cho et al. 1973; Dreef et al. 2007; Kluin et al. 1983; Ku et al. 2010; Smedley et al. 2002). The relationships of age, body weight, and testis weight to the testis maturation grade were evaluated.
Summary of testicular maturation in cynomolgus monkeys.
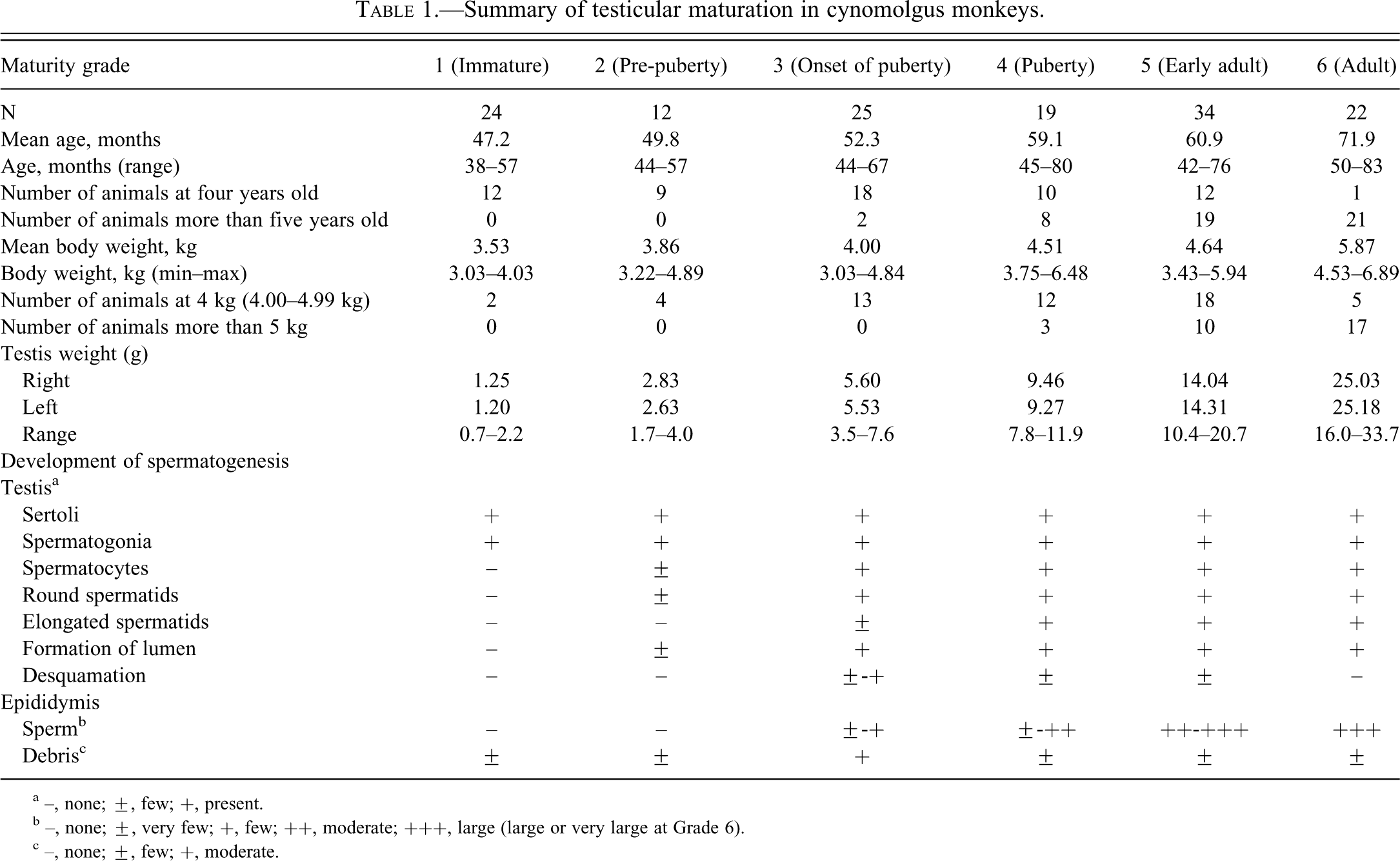
a –, none; ±, few; +, present.
b –, none; ±, very few; +, few; ++, moderate; +++, large (large or very large at Grade 6).
c –, none; ±, few; +, moderate.
The animal care and use practices for the above-mentioned studies had been reviewed and approved by the Animal Care and Use Committee of Shin Nippon Biomedical Laboratories, Ltd, Kagoshima, Japan.
Data Analysis
Each of the relationships of age, body weight, and testis weight to the testis maturation grade was analyzed with logistic regression to investigate whether these parameters are useful indicators of sexual maturity.
Results
Based on the histological findings in the testis and epididymis, testes of 136 cynomolgus monkeys were classified into one of six categories ranging from “immature” to “adult.” Ages, body weights, and testis weights are shown in Table 1.
At Grade 1 (immature, twenty-four monkeys), Sertoli cells and spermatogonia were the only cells observed, and a lumen was not visible (Figure 1A). In the epididymis, no sperm and very slight debris were observed (Figure 2A). At Grade 2 (pre-pubertal, twelve monkeys), no elongated spermatids were observed, although slight presence of round spermatids and lumen were observed. These findings were almost identical in almost all seminiferous tubules examined (Figure 1B). As in the case of cynomolgus monkeys with Grade 1 testes, no sperm and very slight debris were observed in the epididymis (Figure 2B).
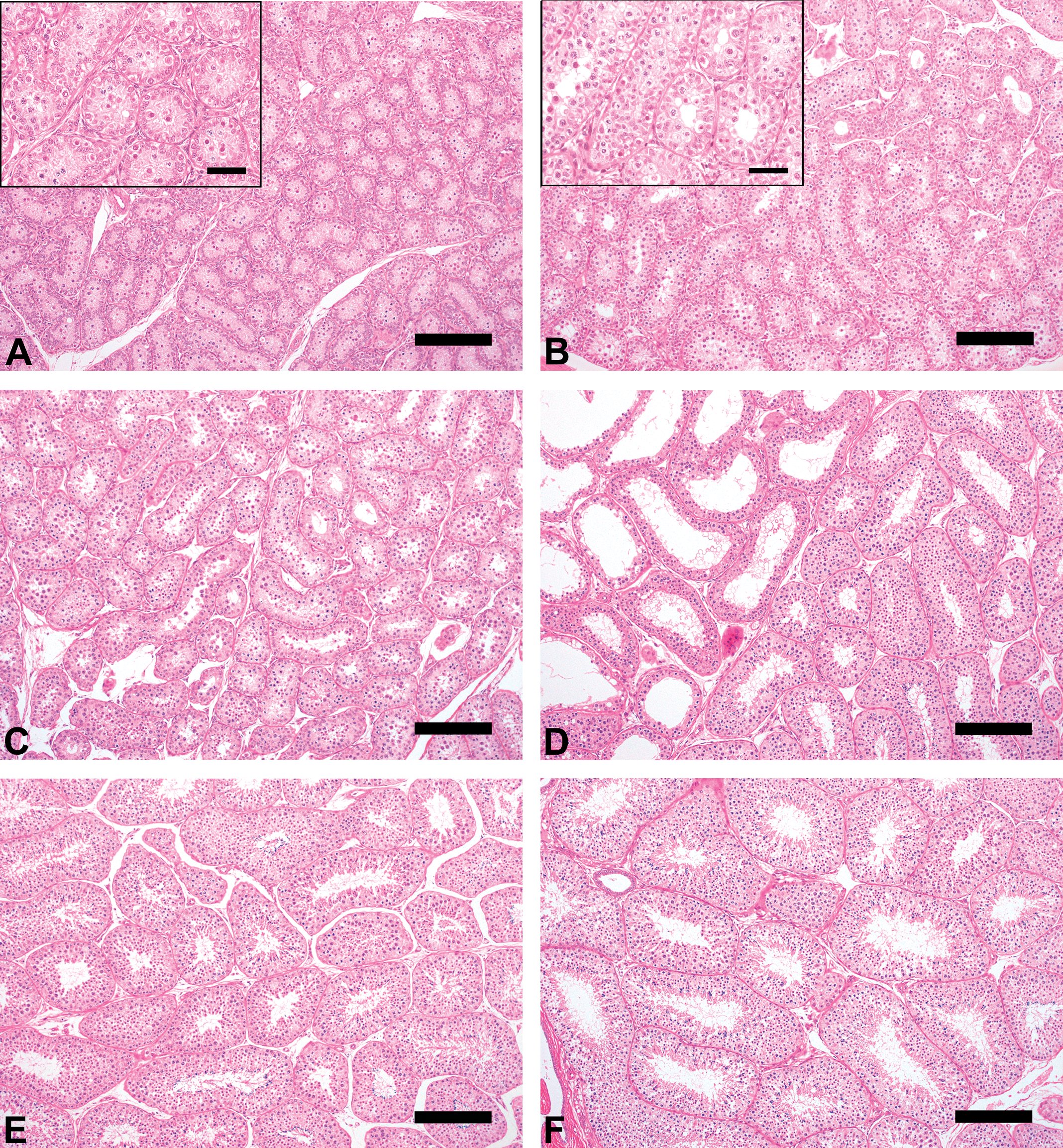
Testis morphologic characteristics (hematoxylin and eosin, Bar = 50 µm [magnification] or 200 µm).
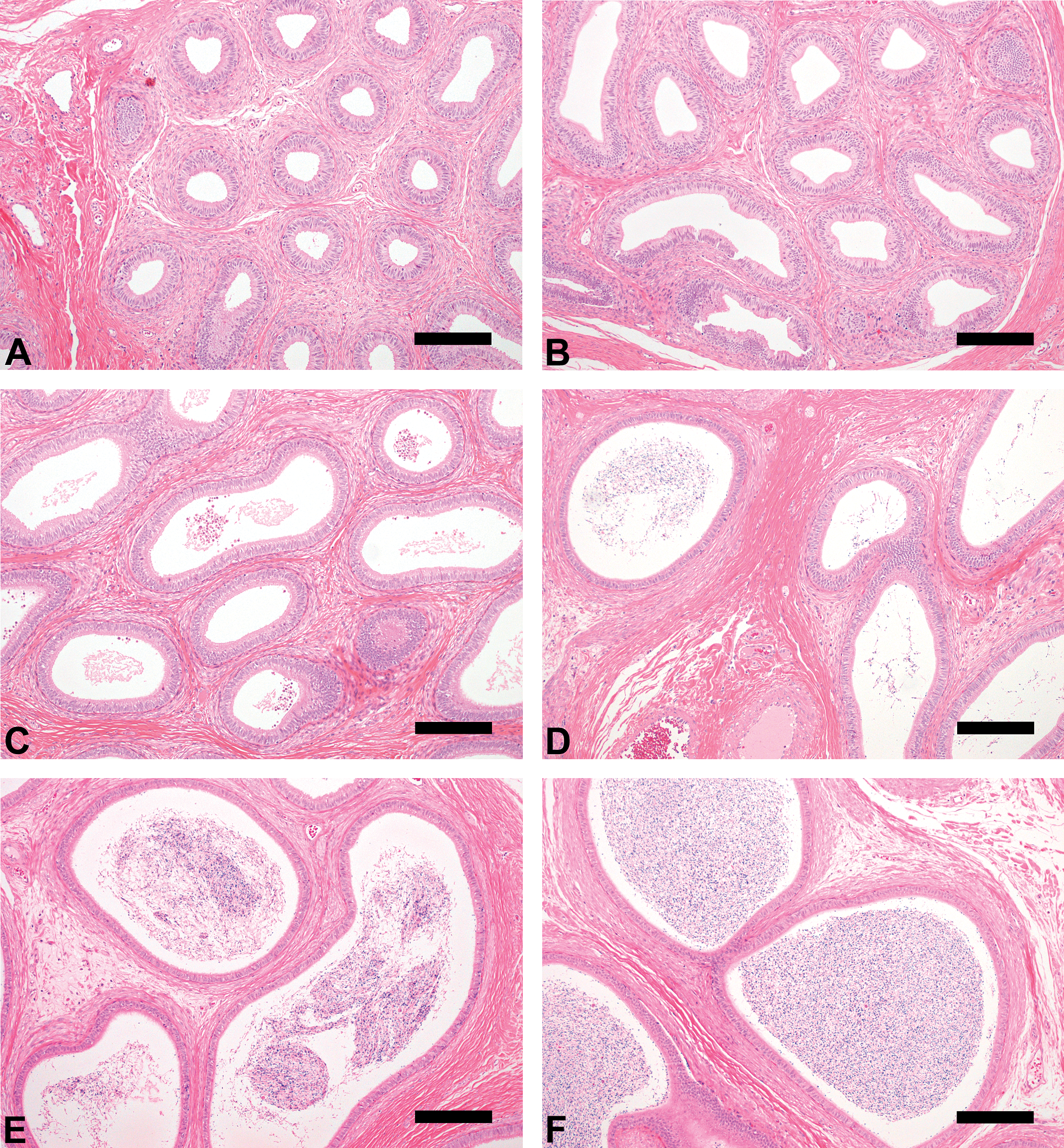
Epididymis morphologic characteristics (hematoxylin and eosin, Bar = 200 µm).
At Grade 3 (onset of puberty, twenty-five monkeys), germ cells of all classes, including spermatogonia, spermatocytes, round spermatids, and elongated spermatids, were observed, although seminiferous tubule diameters were small and few germ cells were observed in cross-sections and a lumen was visible (Figure 1C). Very few or few sperm were observed in the epididymis (Figure 2C). These findings in the testis and epididymis indicated the onset of spermatogenesis in these monkeys. In almost all monkeys, a moderate amount of debris was observed in the epididymis; its extent was greater than for the other grades. This finding suggested immature Sertoli cell function in these monkeys.
At Grade 4 (pubertal, nineteen monkeys), almost complete spermatogenesis was observed in the seminiferous tubules; the spermatogenesis stage as described by Clermont (1972) could be ascertained, although tubule diameters and seminiferous epithelial height were small, and the few germ cells and partial immature seminiferous tubules were observed in cross-sections (Figure 1D). The number of sperm in the epididymis ranged from very few to moderate, and there was less debris than was observed at Grade 3 (Figure 2D). Evaluation of testicular toxicity may be possible in cynomolgus monkeys with testes of this grade.
At Grade 5 (early adult, thirty-four monkeys), complete spermatogenesis was observed in the seminiferous tubules; moderate or large numbers of sperm were observed in the epididymis (Figure 1E and Figure 2E). The characteristics of this grade were considered to be appropriate for the evaluation of testicular toxicity.
At Grade 6 (adult, twenty-two monkeys), complete spermatogenesis in the seminiferous tubules, and large or very large numbers of sperm in the epididymis were observed (Figure 1F and Figure 2F). Moreover, sperm analysis using ejaculated semen was possible for these animals.
For cynomolgus monkeys, there were widespread individual differences in age and body weight, and the testis grades observed in four-year-old monkeys or those weighing 4 kg (4.00–4.99 kg) ranged between Grades 1 and 6. Although 96% (forty-eight of fifty animals) of monkeys more than five years old and all monkeys weighing more than 5 kg had testes that were at Grade 4 or above, approximately 37% (twenty-three of sixty-two) of four-year-old monkeys and more than half of those weighing 4 kg (4.00–4.99 kg) had testes that were also at Grade 4 or above. The testis weight (mean: right and left) at Grades 1–6 were 0.7–2.2 g (1.25 and 1.20 g), 1.7–4.0 g (2.83 and 2.63 g), 3.5–7.6 g (5.60 and 5.53 g), 7.8–11.9 g (9.46 and 9.27 g), 10.4–20.7 g (14.04 and 14.31 g), and 16.0–33.7 g (25.03 and 25.18 g), respectively. The same was true for the individual testis weight. All cynomolgus monkeys in which individual testis weight was less than 8 g had testes at Grade 3 or below, and all cynomolgus monkeys in which individual testis weight was more than 8 g had testes at Grade 4 or above. Moreover, almost all cynomolgus monkeys in which individual testis weight was more than approximately 20 g had testes at Grade 6 (Figures 3 –5).
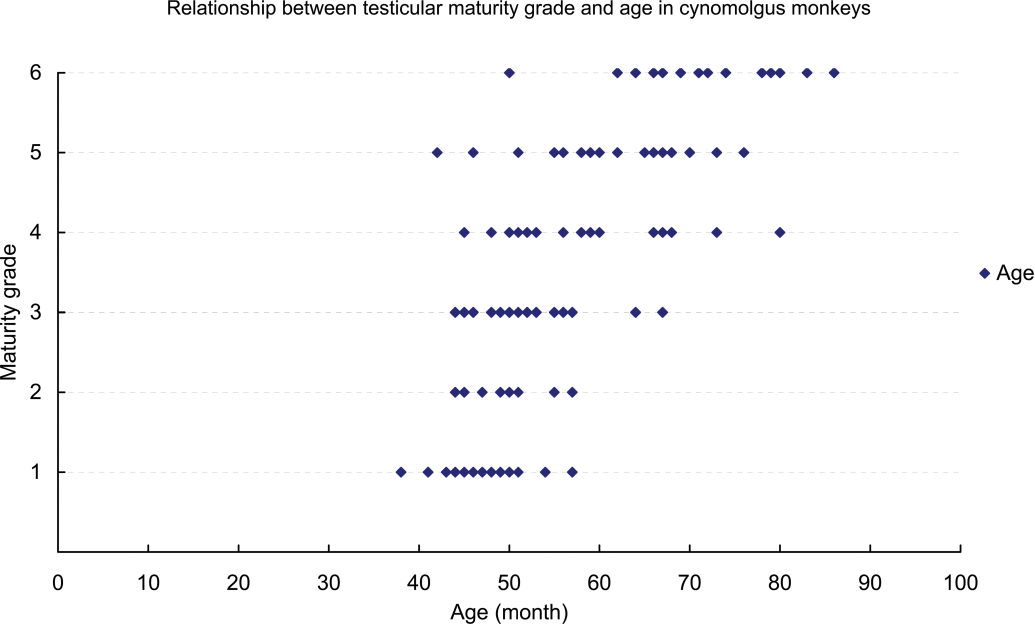
Relationship between testicular maturity grade and age in cynomolgus monkeys.
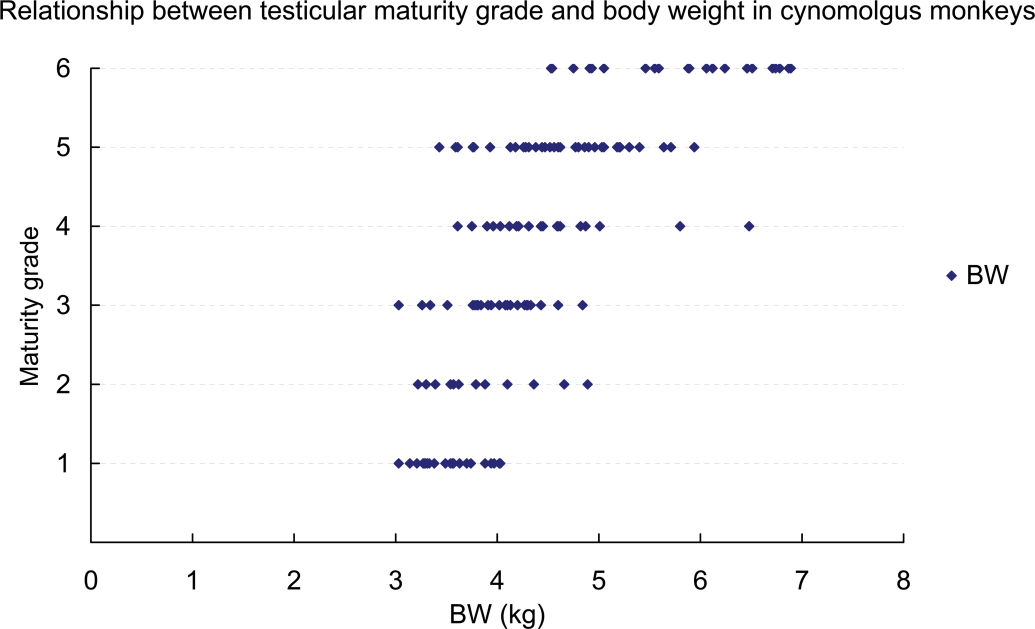
Relationship between testicular maturity grade and body weight in cynomolgus monkeys.
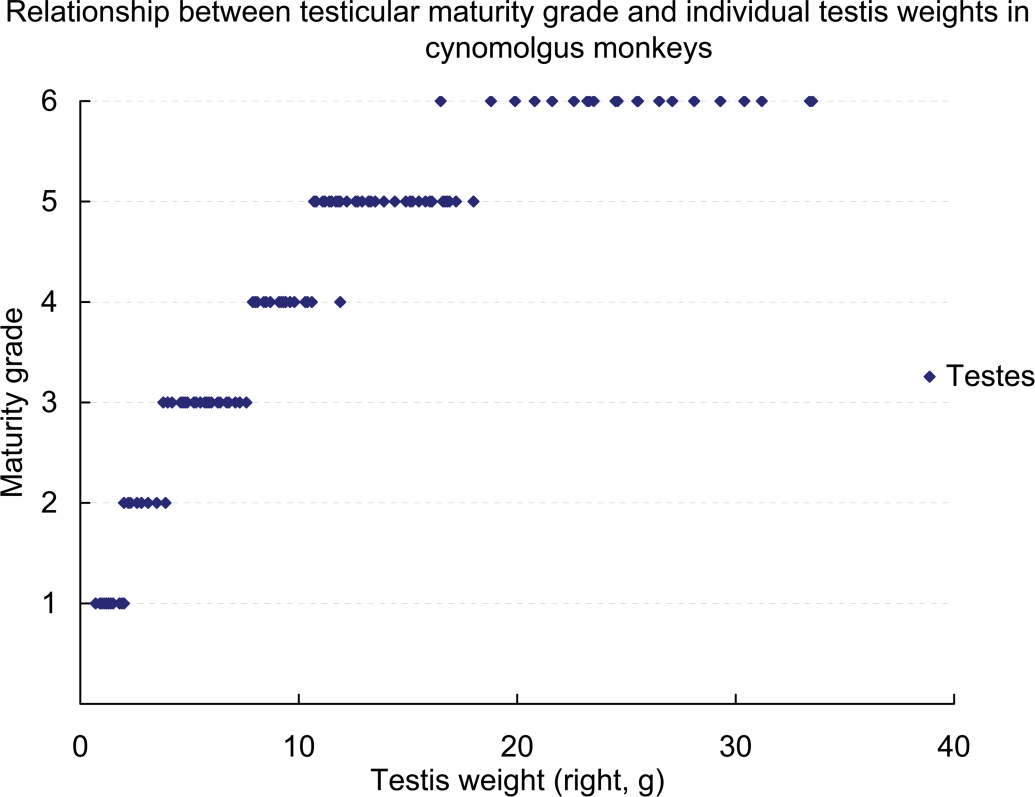
Relationship between testicular maturity grade and testis weight in cynomolgus monkeys.
Each of the relationships of age, body weight, and testis weight to the testis maturation grade was analyzed using logistic regression to investigate whether these parameters are good indicators of sexual maturity. If the regression parameters had not been related to the distribution of the testis maturation grade, the graphs (Figures 6 –8) for these analyses would have shown slopes of the regression curves that tended to be nonsignificant and lines of fit that tended to be horizontal. However, the curves in these graphs differed greatly from the horizontal; therefore, these parameters were strongly related to sexual maturity. In particular, testis weight was found to be a good predictor of testis maturation, because the regression curves were almost vertical lines.
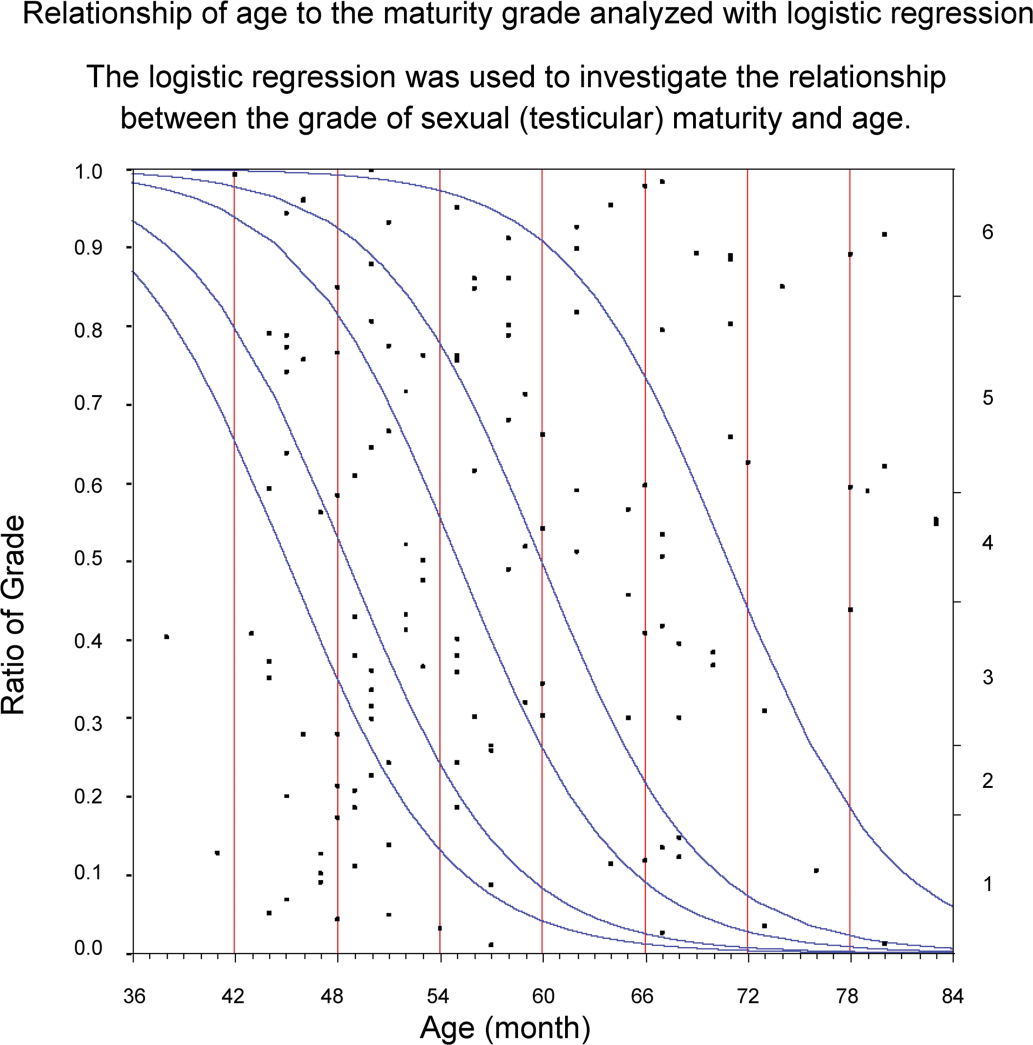
Logistic regression was used to investigate the relationship between the grade of sexual (testicular) maturity and age.
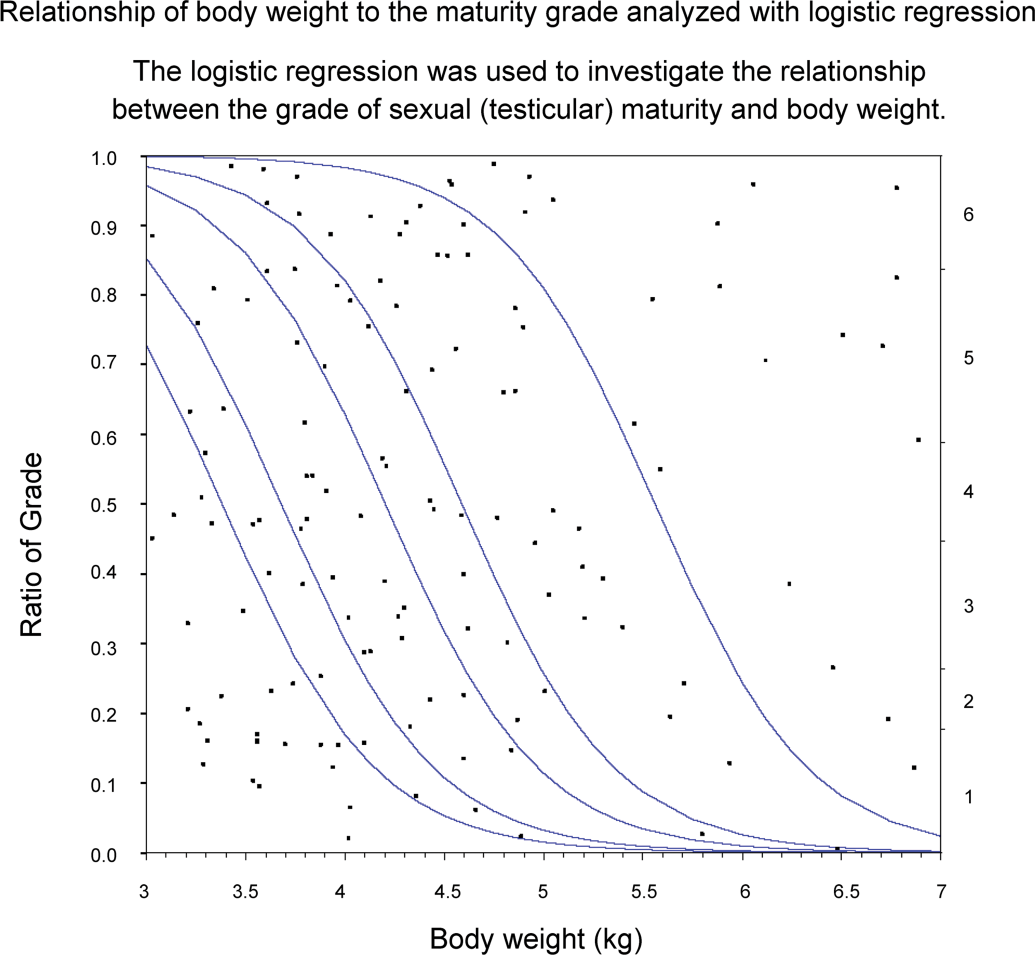
Logistic regression was used to investigate the relationship between the grade of sexual (testicular) maturity and body weight.
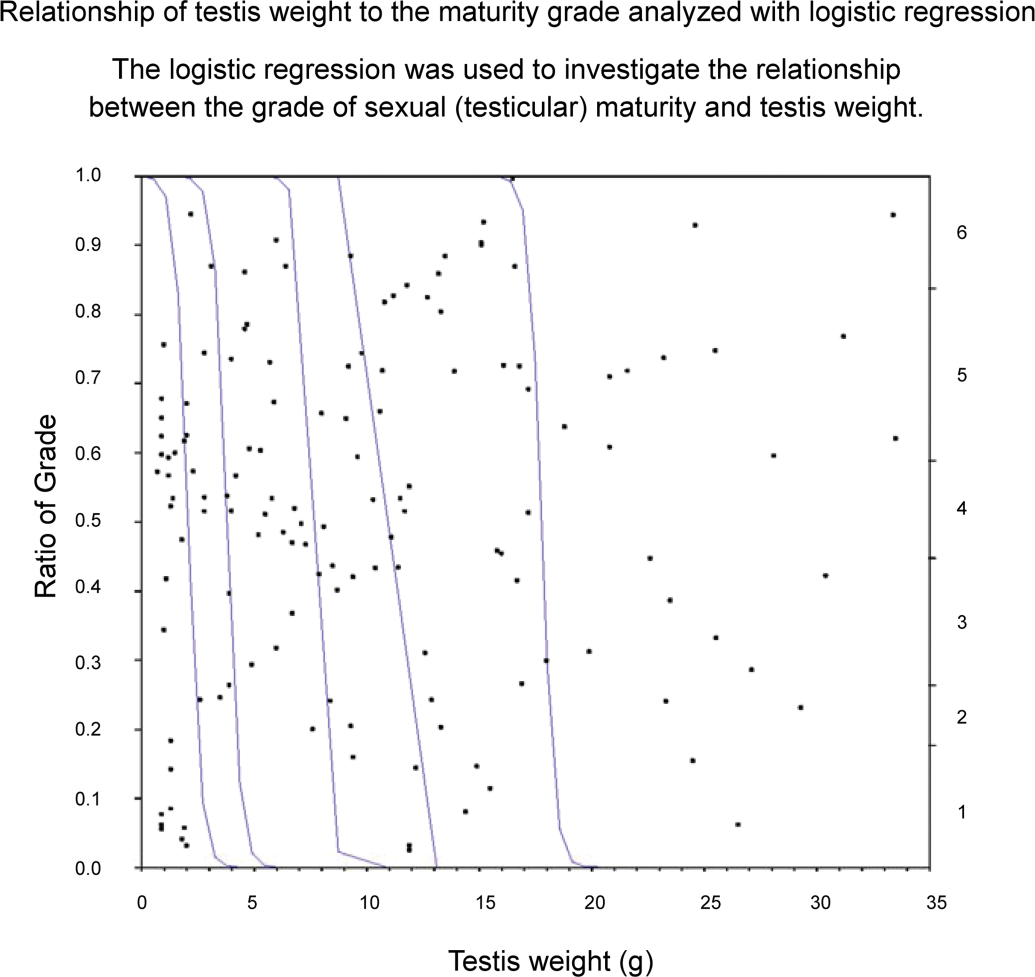
Logistic regression was used to investigate the relationship between the grade of sexual (testicular) maturity and testis weight.
Discussion
Nonhuman primates are frequently used for general or reproductive toxicity studies for biopharmaceuticals because they commonly exhibit pharmacological activities that resemble those in humans (Chellman et al. 2009). Macaque monkeys, such as cynomolgus and rhesus monkeys, are used in such studies; however, selecting sexually mature males is important when evaluating testicular toxicity.
Rhesus monkeys have a seasonal breeding cycle, with reduced spermatogenesis outside of the breeding season. Therefore, it is difficult to evaluate testicular toxicity using rhesus monkeys. On the other hand, with cynomolgus monkeys, as in humans, mature males can produce sperm at any time of the year, and they would provide a suitable model for evaluation of testicular toxicity. However, it can be difficult to obtain a sufficient number of mature males for reproductive or repeated-dose toxicity studies. It was reported that 57 of 126 male cynomolgus monkeys used in a number of repeated-dose toxicity studies were not suitable for the evaluation of testicular toxicity because they were sexually immature or adolescent (Ku et al. 2010).
We reviewed testis and epididymis data from control groups in experiments conducted in our laboratories during an approximate one-year period from September 2008, and the relevant organs were re-examined histopathologically for sexual maturity. In this evaluation, 36 of 136 males showed immature spermatogenesis in the testis and no sperm in the epididymis (Grades 1 and 2). Twenty-five males (Grade 3) showed all classes of germ cells, including step 14 spermatids, in the testis; however, they were considered to be unsuitable for evaluation of testicular toxicity because they showed moderate desquamation of germ cells in the testis and moderate debris (sloughed nucleated germ cells) in the epididymis. Moreover, the morphological stage of spermatogenesis was unclear at this grade because many seminiferous tubules were noted to be at a similar stage. In histological examinations, desquamation of germ cells, multinuclear giant cells, atrophy of the seminiferous tubules, and vacuolization of the Sertoli cells in the testis and debris in the epididymis were observed. Among these findings, desquamation of germ cells in the seminiferous tubules and debris in the epididymis were the most frequently observed, especially at Grade 3.
Ku et al. (2010) reported similar findings in males with Grade 3 (adolescent) testes. These findings indicate that at Grade 3, males have immature testicular development. The desquamation of germ cells and debris observed at this grade were considered to suggest immature Sertoli cell function.
In rats, between postnatal days 15 and 18, numerous cases of apoptosis have been observed in zygotene and pachytene spermatocytes in the seminiferous cords without a blood–testis barrier (Morales et al. 2007). The first spermatogenic wave in rats lasts from postnatal week 1 to 6 (Jahnukainen et al. 2004; Morales et al. 2007). Blood–testis barrier formation occurs during puberty (Kaitu’u-Lino et al. 2007). Tight junctions form between adjacent Sertoli cells at 18 to 20 days of age in the rat, thereby forming the blood–testis barrier; at the same time, the tubular lumen is also formed (Fawcett 1975; Lamb et al. 1982; Means et al. 1976). Sertoli cells continue to proliferate and differentiate until the beginning of puberty, and prior to puberty, the Sertoli cells are immature (Petersen and Soder 2006). The onset of puberty is associated with a rapid and substantial proliferation of Sertoli cells (Marshall and Plant 1996). At the onset of puberty, gonadotropin levels increase again, leading to Sertoli cell maturation and an increase in the rate of germ cell proliferation and differentiation beyond spermatogonia (Rathi et al. 2008). Accordingly, it was considered that the spermatogenic wave in cynomolgus monkeys begins at Grade 3.
A total of 61 out of the 136 males in the present research were judged to be unsuitable for evaluation of testicular toxicity (Grades 1 to 3), a result that is similar to that of the previous report (Ku et al. 2010). At Grades 4–6, there were 75 males, and they showed all classes of germ cells in the seminiferous tubules and sperm in the epididymis. A determination of spermatogenic stage from their seminiferous tubules and histopathological evaluation with a consideration of spermatogenic stage was possible. These males were considered to be suitable for evaluation of testicular toxicity. At Grade 6, males showed a large number of sperm in the epididymis, and sperm analysis using ejaculated semen collected by electroejaculation (Meyer et al. 2006) was possible. The evaluation of both testicular sperm production capability and epididymal sperm maturity was possible with repeated sperm analysis from before treatment to the end of the treatment period for these males.
The use of cynomolgus monkeys that are at least four years old and weigh at least 4 kg has been proposed as a broad standard to evaluate testicular toxicity; however, this standard has been judged to be insufficient because approximately 63% (thirty-nine of sixty-two) of four-year-olds and approximately 35% (nineteen of fifty-four) of those weighing 4 kg (4.00–4.99 kg) in this assessment were at Grade 3 or below. Approximately 96% of animals more than five years old and all of those weighing more than 5 kg were at Grade 4 or above. Although it could be argued that based on the current assessment, evaluation of testicular toxicity is possible with animals aged five years or weighing more than 5 kg (Ku et al. 2010), 85% of sexually mature males weighed less than 5 kg. Furthermore, Lanning et al. (2002) recommended that cynomolgus monkeys four to five years of age should be used in order to ascertain any effect on testicular maturity, and Smedley et al. (2002) reported that sexual maturity in male monkeys had been reached at an age of five years and five months with weights above 5.3 kg. Meyer et al. (2006) reported that sexual maturity in male monkeys had been reached at ages over six years with weights above 5 kg. The wide ranges in age and body weight at each grade of testicular maturity reported here showed that the maturity of the testis did not have a linear relationship with age or body weight in these males, although mean age and mean body weight tended to be higher where the grade of testicular maturity was higher.
On the other hand, there was a clear relationship between grade of testicular maturity and testis weight. All animals in which testis weight was less than 8 g were at Grade 3 or below, and all animals in which testis weight was more than 8 g were at Grade 4 or above. Moreover, almost all animals in which testis weight was more than 20 g were at Grade 6. Logistical regression analysis to evaluate the relationships of the grade of testicular maturity to age, body weight, and testis weight showed that there was a linear relationship between testis weight and maturity grade. Hence, testis weight is a good predictor of testicular maturity compared to the age or body weight of cynomolgus monkeys.
It has been reported that there was a very strong relationship (r 2 = 0.796, p < .01) between testicular volume (measured in-life) and testicular weight (measured at necropsy) in cynomolgus monkeys (Ku et al. 2010). Moreover, testicular volume has been reported as a broad indicator of sexual maturity in humans and monkeys (Bhehre et al. 1989; Chipkevitch et al. 1996; Diamond et al. 2000; Fuse et al. 1990; Hamm and Fobbe 1995; Korte et al. 1995; Meyer et al. 2006; Schiff et al. 2004).
In conclusion, when selecting monkeys for toxicity studies in which testicular toxicity is to be evaluated, testicular volume is a good indicator of testicular weight. In addition, recognizing the grade of testicular development is important for the conduct of toxicity studies and evaluation.
Footnotes
Acknowledgment
The authors gratefully acknowledge Dr. Kok Wah Hew for his critical review of the article.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
