Abstract
To further our understanding of the nonhuman primate kidney anatomy, histology, and incidences of spontaneous pathology, we retrospectively examined kidneys from a total of 505 control Cynomolgus monkeys (
Introduction
The kidney is often a target for large and small molecules in nonclinical safety testing, and drug-induced kidney injury is a safety concern related to clinical attrition. The kidney is particularly vulnerable to drug-induced toxicity due to high volume of blood supply, the presence of drug metabolizing enzymes, and transmembrane transporters that allow exposure to high concentrations of pharmaceuticals and/or their metabolites. 1 Transport across the tubular epithelium and reabsorption processes in cortical tubules may increase drug concentrations to levels much higher than in the plasma. 2 Hence, historically, kidney lesions in both the nonclinical setting and in the clinics have been largely localized to the tubules, with tubular degeneration and or necrosis being the most significant cause of nephrotoxicity-related drug attrition. However, although still relatively uncommon in comparison to tubular injury, glomerular lesions have been reported with increasing frequency in nonclinical toxicity studies, mainly due to the increasing importance of biotherapeutic drugs and oligonucleotide therapies that have the potential to affect glomeruli. 3,4 For studies with these novel biotherapeutic compounds, the nonhuman primate is frequently the pharmacologically relevant species. Toxicologic pathologists are, therefore, frequently tasked with separating potential drug effects from spontaneous occurrences in the nonhuman primate kidney. In addition, with the emerging need to reduce animal use, such that toxicology studies using monkeys especially can have treatment groups with as few as 2 to 3 animals, discerning spontaneous from test article–related findings is even more challenging. Nonetheless, there is a paucity of data on the comparative renal anatomy, physiology, spontaneous pathology, and reference values of traditional renal biomarkers in this species. The primary objective of this study was to investigate, describe, and document the characteristic gross and microscopic morphology of the cynomolgus monkey kidney, and the nature and incidence of spontaneous findings, particularly those that may be confused with test article–related changes. A further objective was to collect and develop a historical control database of background pathology, organ weight data, urinalysis, and renal clinical biochemistry analytes.
Materials and Methods
Animals
Kidney pathology data, and other kidney-related parameters, were obtained from a total of 505 control cynomolgus monkeys (264 males and 241 females) from toxicity studies of 2- to 13-week duration, conducted between 2013 and 2018. All animals were of Mauritian origin, purpose-bred for laboratory use, and obtained from accredited suppliers. Their age at the commencement of the study fell into 2 broad groups, of: (a) sexually mature animals of 4 to 6 years, and (b) young adolescent monkeys of 2 to 3 years, hereafter referred as “mature” and “juvenile” animals, respectively. They received an appropriate veterinary examination, which included serological tests to confirm that they were free of routinely tested infectious agents. They were confirmed free of respiratory infections and the following viral diseases during quarantine: simian immunodeficiency virus,
Study animals were housed in groups of 1 to 3 animals of the same sex and dose group in custom-designed UK Home Office–compliant primate cages (chapter 14, section 21, UK Animals [Scientific Procedures] Act of 1986). The temperature and humidity were automatically controlled at 21°C ± 4°C and 55% ± 10%, respectively, with a minimum of 15 air changes per hour. An automatic light cycle of 7:00
All studies were conducted in accordance with the UK Animals (Scientific Procedures) Act 1986, which conforms to the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (Strasbourg, Council of Europe).
Pathology Procedures
In each individual toxicity study, a detailed necropsy examination was performed on all animals following euthanasia by intravenous injection with sodium pentobarbitone and exsanguination. Prior to fixation, one kidney from each animal was trimmed longitudinally in the midsagittal plane, and the contralateral one, transversely, across the middle. The tissues were preserved in 10% neutral buffered formalin, embedded in paraffin wax, sectioned to a 4- to 5-μm thickness, and stained with hematoxylin and eosin. They were examined microscopically by qualified veterinary pathologists and the findings entered directly onto a computerized database (Provantis; Instem Life Sciences, Stone, Staffordshire, United Kingdom).
Study materials, including microscopic incidence summary tables and individual animal data listings, blocks, and glass slides were retrieved from the archives and analyzed for the subgross anatomy, histology, and spontaneous pathology of the kidney. In addition, a historical control data (HCD) search was performed for pathology findings recorded for the kidneys. Data were available from 505 control cynomolgus monkeys that had been sham-dosed an appropriate vehicle that included: empty or gelatin capsules for the oral capsule route, carboxymethylcellulose or drinking water for the oral gavage route, and phosphate-buffered saline, physiologic saline, and acetic or citrate buffers for intravenous or subcutaneous injection routes, and air, saline, or lactose vehicle via the nose-only method of inhalation exposure.
Clinical Pathology and Organ Weights
For the analysis and comparison of clinical pathology parameters and paired kidney weights, 9 toxicity studies with 104 (52 per sex) sexually mature animals of 4 to 6 years at commencement of the studies, and 10 toxicity studies with 104 (52 per sex) “juvenile” monkeys of 2 to 3 years at commencement of dosing, were selected. Data for absolute and relative (to body weight) kidney weights, serum urea, creatinine, calcium and phosphate, and urine specific gravity were compiled, and the mean, standard deviation, and range analyzed. For each variable, the following comparisons were made—“mature” versus “juvenile” males, “mature” versus “juvenile” females, “mature” males versus “mature” females and “juvenile” males versus “juvenile” females, using Student
Immunohistochemistry
Sections with 4-μm thickness were obtained from paraffin-embedded tissue, mounted on charged slides, air-dried, heated at 60°C for 2 hours, and loaded onto an autostainer (DAKO, Agilent Technologies, Santa Clara, CA). Slides underwent deparaffinization, heat-induced epitope retrieval with either citric buffer (pH 6.0) for aquaporin 1 (AQP1), CD13 and vimentin, or EDTA (pH 9.0) for cytokeratin 19 (CK19), followed by endogenous (30% H2O2) and protein blocking (10% goat serum). Sections were incubated with either anti-AQP1 rabbit polyclonal antibody (Abcam, Cambridge, MA, Cat#ab15080) at 1:1000 concentration, anti-CD13 (EPR4059) rabbit monoclonal antibody (Abcam, Cat#108382) at 1:1000 concentration, anti-CK 19 (EP1580Y) rabbit monoclonal antibody (Abcam, Cat# 52625) at 1:800 concentration, anti-E cadherin (EPR699) rabbit monoclonal antibody (Abcam, Cat# 133597) at 1:1000 concentration, or anti-vimentin (EPR3776) rabbit monoclonal antibody (Abcam, Cat#92547) at 1:500 concentration. Primary antibody incubation was followed by incubation with goat anti-rabbit HRP polymer (Vector lab, IMMPRESS HRP Reagent kit) linked secondary antibody. Normal rabbit IgG (Santa Cruz Biotechnology, Cat# SC-2027) was used as a negative control. The immunoreactivity was visualized by diaminobenzidine (DAKO, Cat#K3468) followed by counterstaining with Mayer’s hematoxylin.
Morphometric Analysis
A selection of 12 midsagittal-sectioned slides of kidneys (7 males and 5 females) were imaged (Aperio AT scanner, Leica, Microsystems GmbH, Wetzlar, Germany), and the thickness of cortex and medulla measured and analyzed using the measurement function of ImageScope (Leica) to calculate values for the cortex/medullary ratio (C:M). Measurements were taken only on those kidneys in which the plane of section passed through the mid-pelvic surface exactly parallel to and centered in the long axis of the papilla as described in the literature. 5 Five measurements were made from each image at different radial directions starting from the middle of the kidney and passing through the tip of the papillae (Figure 1A) and the mean calculated for each. The overall mean was calculated for each sex.
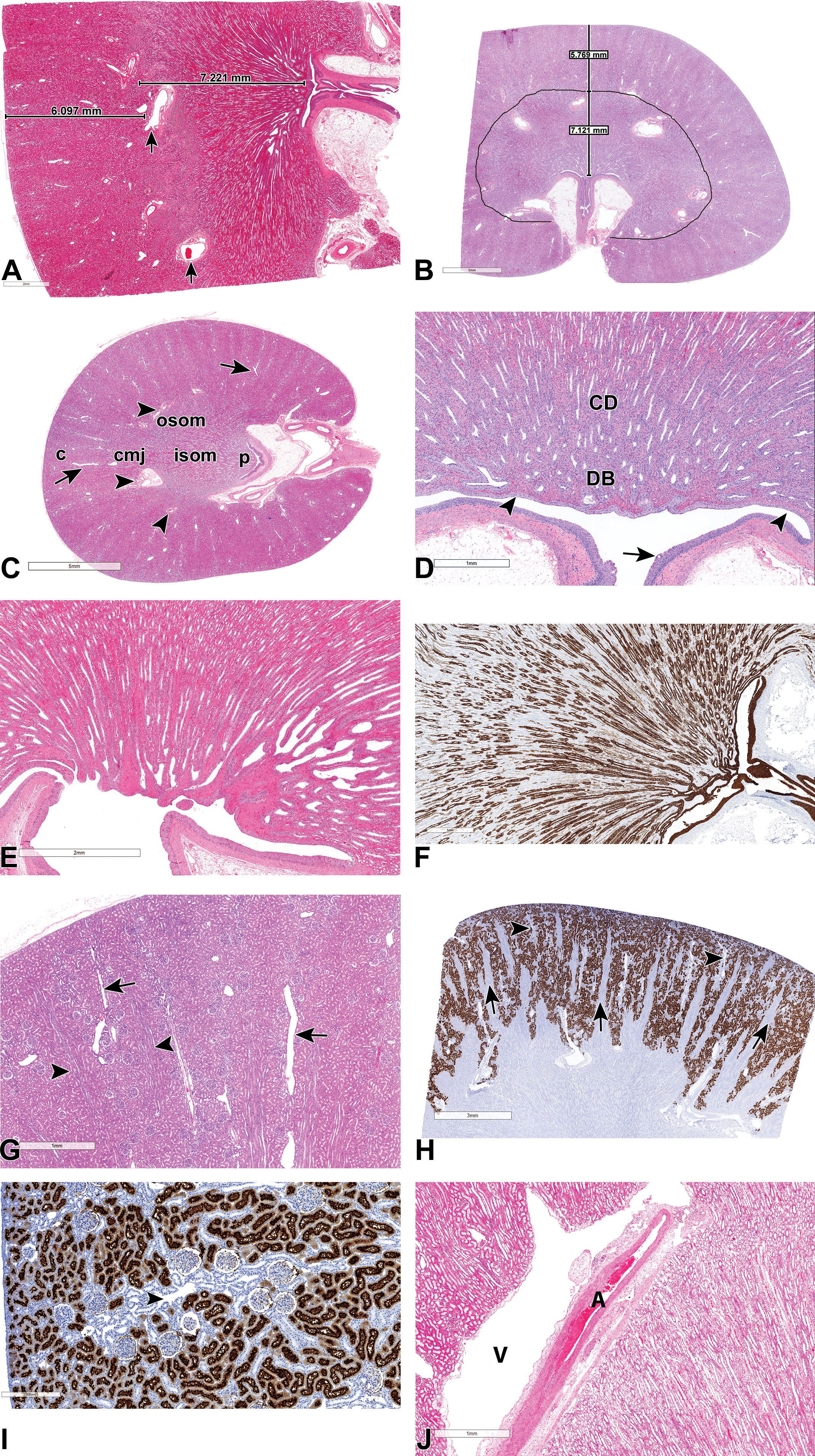
(A-J) Normal, subgross anatomy of the cynomolgus monkey kidney. All sections stained with hematoxylin and eosin, unless otherwise stated. (A and B) Midsagittal sections showing the corticomedullary junction marked by arcuate arteries (arrows) and measurements of the cortical to medullary ratio, from a juvenile monkey (A) and a mature monkey (B). (C) Transverse section showing the cortex (c), arcuate (arrowhead) and interlobular (arrow) blood vessels, the outer stripe of the outer medulla (OSOM), the inner stripe of the outer medulla (ISOM), and a very short papilla (p) and shallow pelvis. (D, E, and F) Longitudinal sections of the papilla showing the more common flat or blunt type (D and E), a slightly more protruding type (F, with cytokeratin 19 which stains for collecting ducts, urothelium, and modified epithelium lining the papillary tip and ducts of Bellini), collecting ducts (CD), ducts of Bellini (DB), a shallow pelvis (arrowhead) with no specialized fornices, and a pelvic urothelium that may occasionally show vacuolation (arrow). (G) Cortex showing medullary rays (arrowheads) and interlobular vessels (arrows) passing through the cortical labyrinth. (H) CD13 staining of the cortex showing positive (brown) staining of proximal convoluted tubules, unstained medullary rays (arrows), and interlobular vessels (arrowheads). (I) Higher power image of the cortical labyrinth showing CD13 positive (brown) staining of proximal convoluted tubules, and negative staining of distal tubules around interlobular vessels (arrowhead). (J) Arcuate vessels at the corticomedullary junction showing an artery (A) and a thin-walled vein (V) that appears as a cystic empty space.
Results
Gross and Subgross Anatomy
An evaluation of the gross and subgross anatomy of the cynomolgus monkey kidney revealed that although the general anatomy is comparable to that of other laboratory animals and humans, several differences exist. As in other laboratory species, the Cynomolgus monkey kidney consists of an outer renal cortex and an inner medulla, with the latter further subdivided into an outer medulla composed of an outer stripe of the outer medulla (OSOM), an inner stripe of the outer medulla (ISOM), and an inner medulla or papilla that extends into the renal pelvis (Figure 1A-F). However, unlike in humans, the kidney of the Cynomolgus macaque is simple and unipapillate as in dogs and rodents, and its medulla is organized into a single ray-shaped renal pyramid with a base at the corticomedullary junction, and a papilla that projects into a single calyx at the hilus (Figure 1A and B). Unlike in dogs and rodents, the monkey inner medulla is relatively short, at approximately a quarter of the thickness of the entire medulla (Figure 1A and B), and the renal pelvis is also shallow and rudimentary with no specialized fornixes (Figure 1A-E). The relatively short papilla is also relatively poorly developed (Figure 1C-F), in comparison to that of other species such as the dog and rat, with scarce or far fewer numbers of well-defined long loops of Henle (Figure 1D-F). On midsagittal sections, the papilla has a characteristic flat or blunt, crest-like configuration (Figure 1C-E) in almost all animals, although some inter-animal variations exist in the degree of flatness, concavity, or prominence (Figure 1F). In addition, some animals occasionally show a pelvic surface that is subdivided into 2 or more secondary or false papillae or papillary grooves of various depth. Measurements of the relative sizes of the cortex to medulla shows that, due to the smaller size of the papilla, the cortex and medulla of Cynomolgus monkeys are of almost equivalent lengths, with the length of the medulla being only slightly greater than that of the cortex (Figure 1A and B). The average ratio of the cortex to the medulla (corticomedullary ratio) in this study was 0.80 (1:1.25), with the female average ratio of 0.84 (SD 0.05) being slightly more than that of males at 0.78 (SD 0.08), and no differences observed between juvenile (Figure 1A) and mature. The OSOM, which as in other species contains the thick ascending limbs (TAL), pars recta, and collecting ducts from juxtamedullary nephrons, is relatively thick in the monkey (compared to humans and dogs) and is of approximately similar thickness to that of the ISOM (Figure 1C).
The monkey renal cortex is subdivided as in other species into, cortical labyrinths bordered by medullary rays (Figure 1G). The medullary rays, which are a projection of the medulla into the cortex, can be visualized grossly as faint longitudinal striations in the cortex in monkeys. The cortical labyrinth, which as in other species is mainly composed of glomeruli and large numbers of proximal convoluted tubules (PCT) and distal tubules, has prominent interlobular vessels that run through the middle and perpendicular to the capsule (Figure 1G and H). In the monkey, the interlobular vessels and are frequently surrounded by a prominent collection of basophilic-staining tubular profiles of distal tubules or connecting ducts (Figure 1G and I), as described further below. The cortical vascular supply is largely similar to that in other species and shows the renal artery branching into interlobar arteries, which in turn give rise to arcuate arteries (Figure 1A-C) that run parallel to the capsule along the corticomedullary junction. Arcuate arteries continue as the perpendicular interlobular arteries (Figure 1C and IG-I) that eventually give rise to afferent arterioles and glomerular capillaries. Arcuate veins and other cortical veins are so thin-walled in monkeys that they may appear as empty cystic spaces (Figure 1J)
Histology
Fine microscopic examination also reveals that, although the basic structure of the monkey kidney is similar to that of other laboratory animals and humans, 6,7 some minor differences and inter-animal variations and peculiarities exist. In the cortex, the tall, cuboidal and eosinophilic epithelium of the PCT is occasionally seen to extend partially or completely onto the parietal surface of the Bowman’s capsule (cuboidal metaplasia; Figure 2A); and thickening of the basement membrane of the Bowman’s capsule with or without hyperplasia of the parietal epithelial cells (PEC) may be observed fairly commonly (Figure 2B). In the middle of the cortical labyrinth, a prominent collection of distal tubules or connecting ducts that are generally more basophilic staining, are frequently observed in between groups of glomeruli, and in close association with interlobular vessels and dilated vascular spaces (Figure 2C and D). This apparently normal feature, which may be easily confused with foci of hyperplastic or regenerative tubules appear to be consistent with either the branched arcades of connecting tubules of juxtamedullary nephrons that ascend through cortical labyrinth before emptying into collecting ducts or distal convoluted tubules (DCT). They stain positive for distal tubule markers (DCT and connecting tubules) such as CK19 and E-cadherin (Figure 2E and F) and are negative for the PCT markers CD13 (Figure 1I) and AQP1 (figure not shown). The macula densa cells of the juxtaglomerular apparatus are also CK19 positive. The proximal tubules are, as in some laboratory species, subdivided into 3 segments, S1, S2, and S3, with the S1 forming a short part of the PCT that connects to the glomeruli, the S2, the largest part of the PCT and a short distance of the pars recta, while the S3 segment forms the rest and majority of the pars recta. In the monkey, the CD13 marker only stains positive for the PCT part of the proximal tubules and is negative in the pars recta (Figure 1I). Aquaporin 1, on the other hand, stains positive in nephron segments compatible with both the PCT and pars recta and also shows positive staining of the squamous epithelium-lined thin descending limbs (TDL), the cuboidal epithelium lining the parietal surface of the Bowman’s capsule (cuboidal metaplasia), and vascular endothelium.
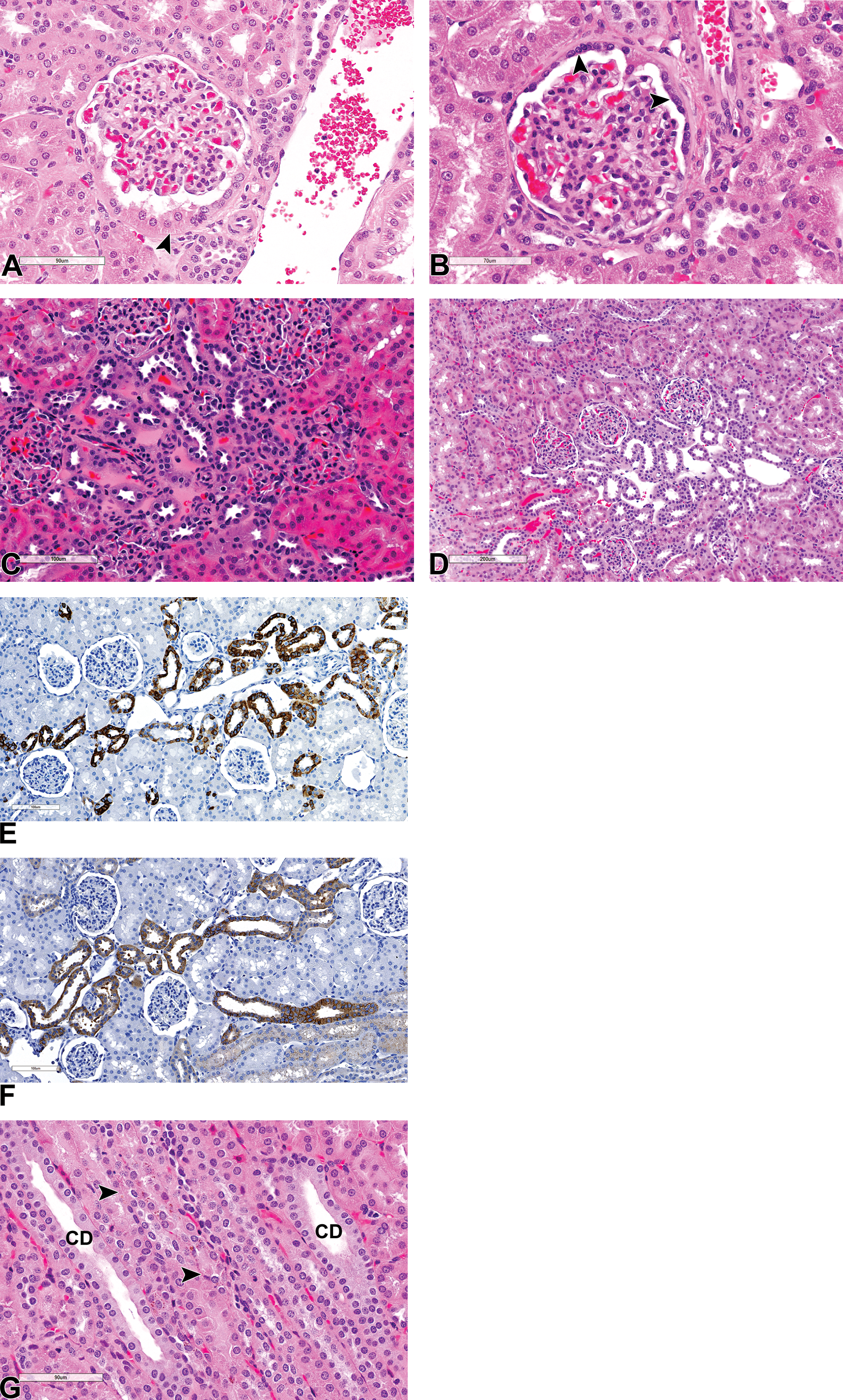
(A-G) The cortex. Salient normal and unique microscopic features of the cortical labyrinth and medullary rays in the cynomolgus monkey kidney. All sections stained with hematoxylin and eosin, unless otherwise stated. (A) Cuboidal metaplasia of the parietal epithelium of the Bowman’s capsule (arrowhead). (B) Thickening of the Bowman’s capsular basement membrane and hyperplasia of the parietal epithelium (arrowhead). (C and D) A collection of basophilic staining distal tubules or connecting ducts associated with interlobular vessels within the superficial cortical labyrinth. (E and F) The distal tubule or connecting ducts complex stains positive (brown) for cytokeratin 19 (E) and E cadherin (F). (G) Medullary rays showing collecting ducts (CD), pars recta, and thick ascending limbs containing golden-brown lipofuscin pigment (arrowhead).
The medullary rays, which are more prominent in the monkey kidney in comparison to other laboratory species, contain collecting ducts, TAL, and the terminal pars recta from superficial nephrons (Figure 2G), as in other animals. The TAL is lined by cuboidal cells with an eosinophilic cytoplasm and round central nucleus as in other species including human, and the cells are smaller than those lining PCT and pars recta cells. However, while in other animals including humans, the cells lining the TDL are squamous, in the Cynomolgus monkeys, the descending limbs and the “U-turn” or hairpin of the long loops are often observed to be lined by a cuboidal epithelium similar to that of the TAL (Figure 3A). As a result, the transition from the terminal pars recta to the TDL appears gradual in the monkey, mainly due to this absence of an abrupt change in the epithelial from a cuboidal to a squamous morphology. In fact, the transition from the cuboidal epithelium to the thin squamous epithelium frequently occurred in the ascending part of the limb rather than the descending part. In addition, the transition from the pars recta in monkeys is frequently characterized by the presence of a golden-brown pigment in the TDL (Figure 3B). The pigment, which stains for lipofuscin, often extends from the TDL to the TAL in the medullary rays via the loop of Henle in normal kidneys (Figure 2G). Apart from the morphological differences in the epithelium of the loops of Henle, closer examination of the inner stripe and the inner medulla also show that most loops from juxtamedullary glomeruli in the Cynomolgus monkey kidney are short, rather than long (as in other animals); as described above, their “U-turn” is most commonly lined by a cuboidal rather than squamous epithelium. As a result, long loops of Henle are rare in the monkey kidney, and the papilla is mostly composed of a high density of collecting ducts and vasa recta and abundant interstitial tissue (Figure 3A and C). Papillary interstitial tissue in the monkey kidney is more prominent in the area cribrosa (Figure 3C and D), where it can present as either as myxomatous or dense collagenous connective tissues with dilated vessels and an edematous appearance (Figure 3C and D), in otherwise normal kidneys, which can be difficult to differentiate from interstitial edema.
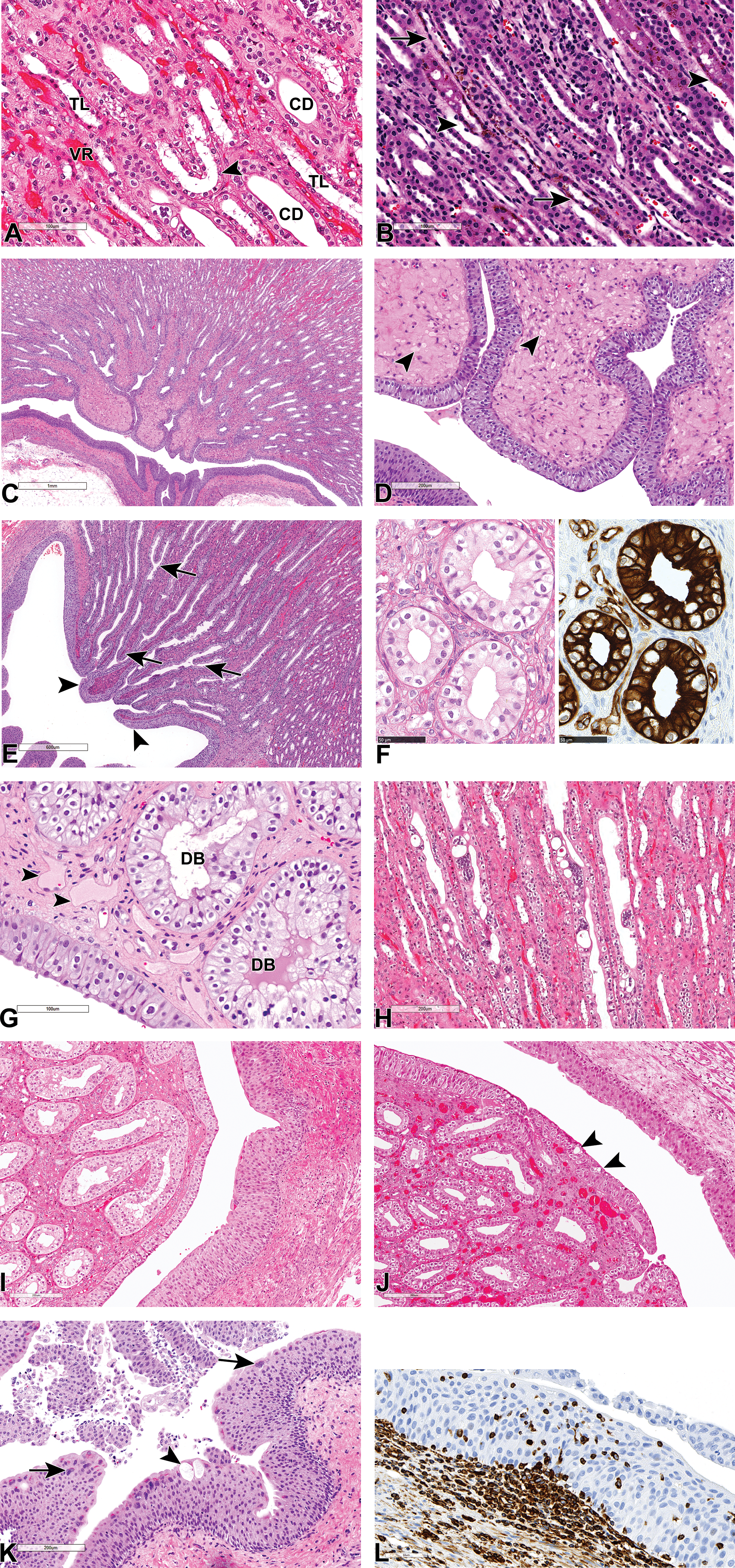
(A-L) Salient normal and unique microscopic features of the inner medulla and pelvis of the cynomolgus monkey kidney. All sections stained with hematoxylin and eosin, unless otherwise stated. (A-H) Inner medulla showing abundant vasa recta (VR), collecting ducts (CD), the scarce long loops of Henle (TL) with some U-turns that are lined by cuboidal epithelium (arrowhead) (A); thin descending limbs (arrows) and thick ascending limbs (arrowheads) containing a golden-brown pigment (B); abundant interstitial tissue at the tip of the papilla or area cribrosa (C) with edema-like expansion of the interstitium and dilatation of vessels (arrowheads) (D); successive fusion of inner medullary collecting ducts (arrows) and their transition into ducts of Bellini, which are lined by a stratified urothelium (arrowheads) (E); the ratio of principal (CK19+) to intercalated cells (CK19-) cells of the inner medullary collecting duct (F); stratified, modified urothelium lining ducts of Bellini (DB), and is contiguous with the epithelium overlying the tip of the renal papilla (G). Note, edema-like dilation of vessels in the areas cribrosa (arrowhead). Multinucleate cells of the collecting ducts containing up to 20 or more nuclei (H). Renal pelvis showing normal pelvic urothelium and a vacuolated (arrowhead) modified transitional epithelium lining the papillary tip (I and J); pelvic urothelium containing brightly eosinophilic cytoplasmic keratohyaline inclusions, vacuolated cells (arrowhead), and multinucleated umbrella cells (arrow) (K); and a vimentin (brown) stain for intraepithelial lymphocytes and macrophages within the urothelium (L).
As in other animals, the monkey collecting ducts successively and dichotomously fuse as they descend into the inner medulla to form ducts of Bellini, which empty into the renal pelvis (Figure 3E). However, due to the shorter nature of the papilla, the inner medullary collecting ducts are very short, and rapidly transition into the ducts of Bellini. Collecting ducts are lined by 2 cell types, the principal (also called light cell) or collecting duct cell, and the intercalated or dark cell, which can occupy the same tubular cross section in the cortex and outer medulla. The ratio of principal (CK19+) to intercalated (CK19−) cells appears to be 3 to 1 in the cortical collecting ducts, and 2 to 1 in the papillary collecting ducts in monkeys (Figure 3F). Therefore, as in other laboratory animals and humans, the principal cell is the more abundant of the 2 in the cortical collecting ducts. However, unlike in other species in which the intercalated cell decreases in frequency as the collecting duct descends into the inner medulla and the principal cells are often the only cell type present in the inner medullary collecting ducts, it appears intercalated cells (CK19−) are also present here in monkeys. Morphologically, the cells of the collecting duct progressively increase in height as the duct descends into the inner medulla, so that the inner medullary collecting duct cells are progressively more columnar (Figure 3D-F). In monkeys, the cells lining the ducts of Bellini are also columnar but represent a stratified/multilayered or modified type of transitional epithelium that is contiguous with the epithelium overlying the tip of the renal papilla (Figure 3D, E, and G). Incidentally, the epithelium of the papillary collecting ducts occasionally exhibits multinucleated or syncytial cells, with as up to 20 intensely dark-staining nuclei per cell, and moderate bulging of the cells into the lumen without light or electron microscopic evidence of viral particles 8 or any other morphological abnormality (Figure 3H).
The Cynomolgus monkey pelvic urothelium has more than 3 cell layers and often shows extensive variability in the number of layers (Figure 3I and J). In addition, it often contains keratohyalin granules which appear as brightly eosinophilic cytoplasmic inclusions (Figure 3K) and frequently has intraepithelial lymphocytes and macrophages (Figure 3L). The urothelium may also show vacuolated or multinucleated umbrella cells (Figure 3K). Vacuolation may also be occasionally observed in the epithelium lining the tip of the papilla (Figure 3J). Some of the more common background anatomic variations, nonlesions, and peculiarities in the monkey kidneys are listed in Table 1.
Some Incidental Microscopic Findings Representing Anatomic Variations or Findings of Little Pathological Significance in the Kidneys of Control Cynomolgus Monkeys, and Their Toxicological Relevance.
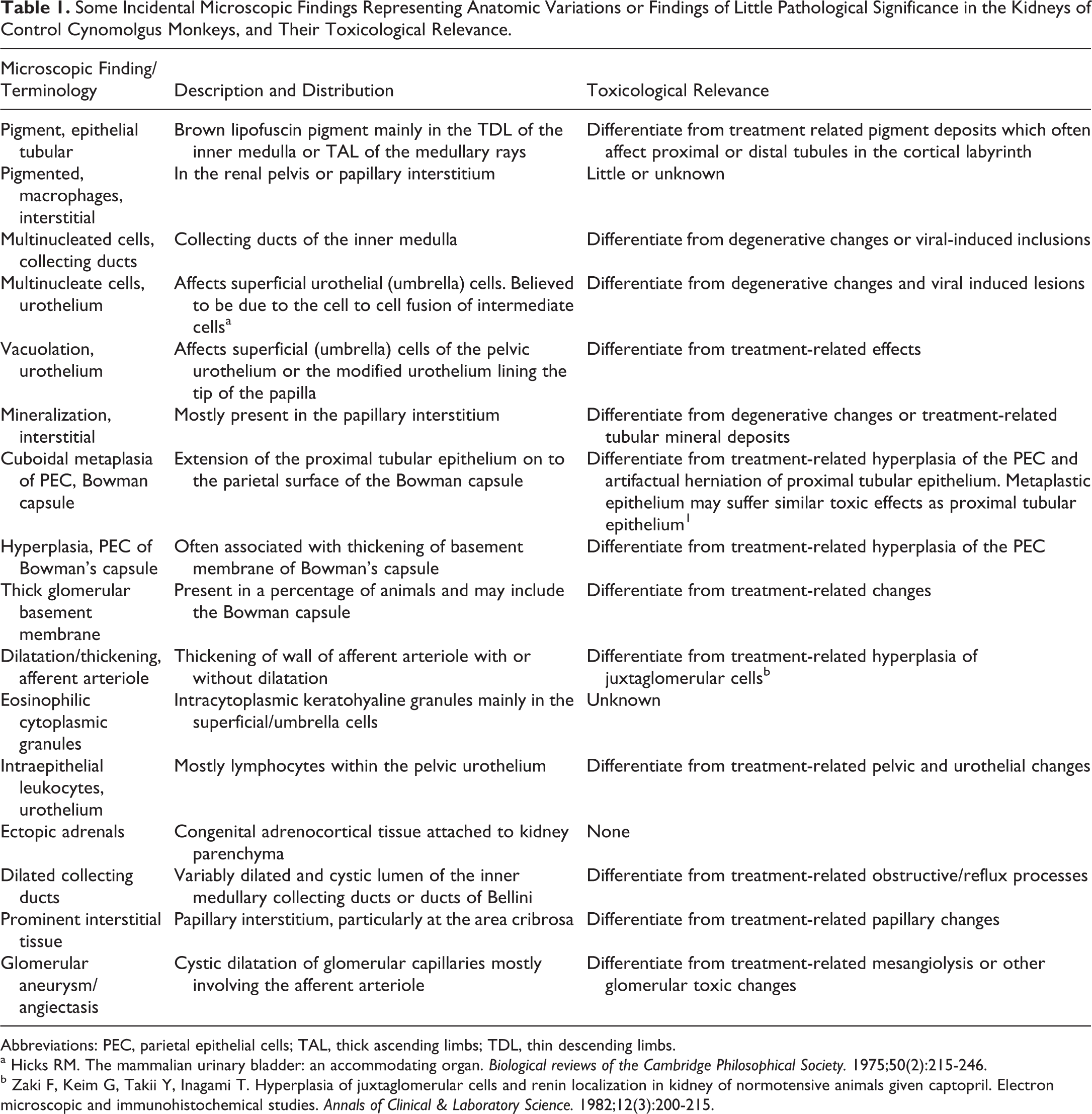
Abbreviations: PEC, parietal epithelial cells; TAL, thick ascending limbs; TDL, thin descending limbs.
a Hicks RM. The mammalian urinary bladder: an accommodating organ.
b Zaki F, Keim G, Takii Y, Inagami T. Hyperplasia of juxtaglomerular cells and renin localization in kidney of normotensive animals given captopril. Electron microscopic and immunohistochemical studies.
Spontaneous Lesions
The most common spontaneous pathology findings recorded in the kidney of control cynomolgus monkeys are listed in Table 2. Since some of these studies were evaluated prior to the Standard for Exchange of Nonclinical Data (SEND)-compliance period, and before the publication of the nonhuman primates International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) documents, some terms were not SEND-compliant and may not reflect the nonhuman primate INHAND terminology. Focal to multifocal interstitial mononuclear/lymphoplasmacytic cell infiltrates (Figure 4A), and/or interstitial inflammation, lymphoplasmacytic (also recorded as interstitial nephritis; Figure 4B and C) were the most commonly recorded findings. The findings were characterized by minimal to moderate accumulation of lymphoplasmacytic infiltrates in the intertubular interstitial tissue with frequent involvement of surrounding tubules or glomeruli (Figure 4A) and occurred more commonly in the outer or subcapsular cortex. In general, the term “interstitial nephritis” (inflammation, interstitial lymphoplasmacytic) was reserved for interstitial inflammatory lesions with more extensive or severe tubular damage, or tubulointerstitial lesions suggestive of primary tubular damage with secondary interstitial inflammation (Figure 4B-D), while “mononuclear or lymphoplasmacytic infiltrates” was applied to lesions with less involvement of surrounding tubules or glomeruli (Figure 4A). Other less common tubular lesions included tubular degeneration/regeneration or “basophilic tubules,” hyaline casts (Figure 4F), granular protein casts (Figure 4G), and tubular dilatation (Figure 4H). Spontaneous tubular dilatation was mostly confined to the superficial cortex, with no discernible distribution pattern, and affected both proximal and distal tubules (Figure 4H). However, focal or cystic tubular dilatation of collecting ducts or ducts of Bellini was occasionally observed in the papilla (Figure 4I and inset).
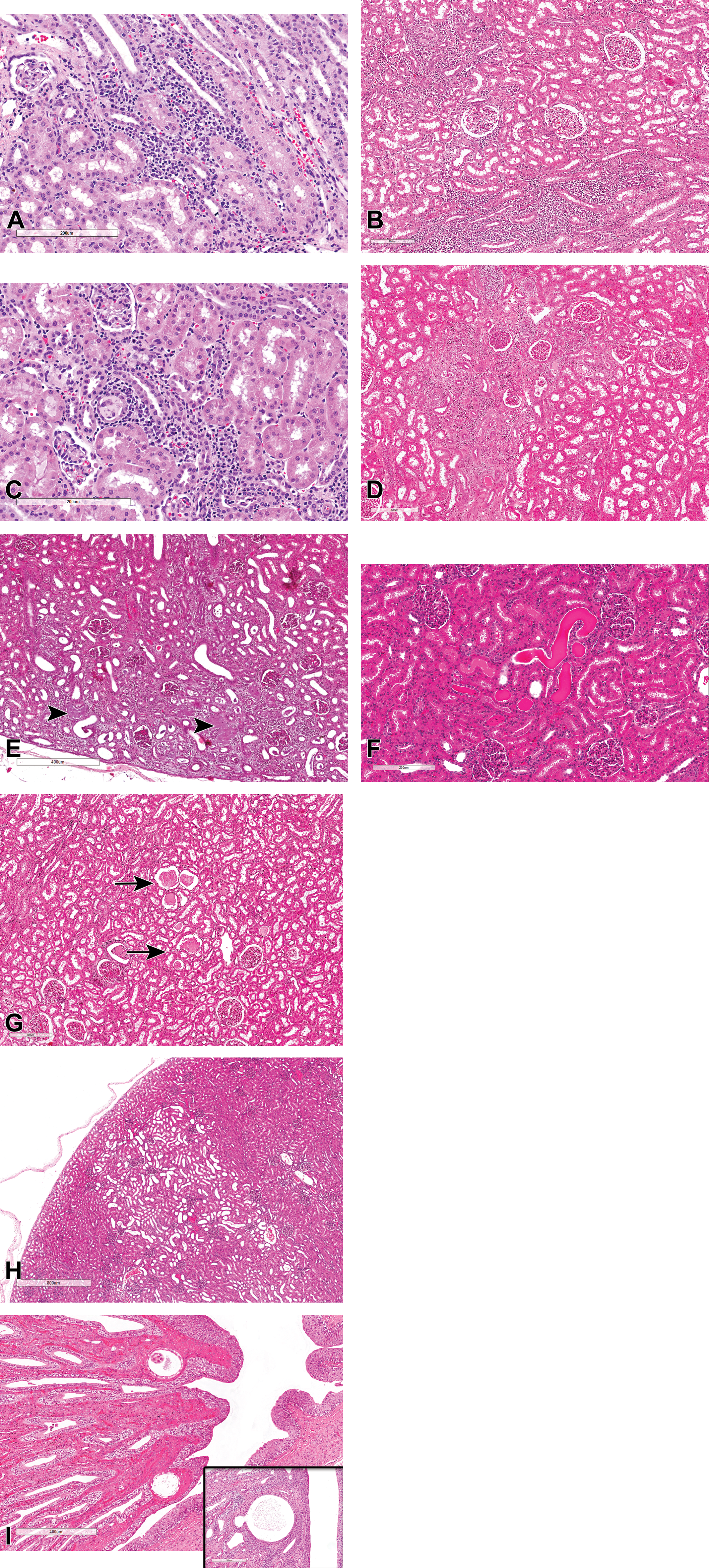
(A-I) Spontaneous lesions of the tubules and interstitium in control cynomolgus monkeys. All sections stained with hematoxylin and eosin. (A) Mononuclear cell infiltrates in the renal cortex with minimal tubular damage. (B and C) Interstitial nephritis (lymphohistiocytic infiltrates, interstitial), with tubular and glomerular inflammation, renal cortex. (D) Localized chronic interstitial nephritis (lymphohistiocytic inflammation, interstitial, cortex) with fibrosis. (E) Tubulointerstitial nephritis of likely bacterial origin showing bacterial colonies (arrowheads). (F) Hyaline casts in the renal cortex. (G) Granular protein casts (arrows) with minimal tubular dilatation. (H) Tubular dilatation, cortex. (I and inset) Dilated/cystic collecting ducts.
Incidences of Pathology Findings in the Kidneys of Control Cynomolgus Monkeys
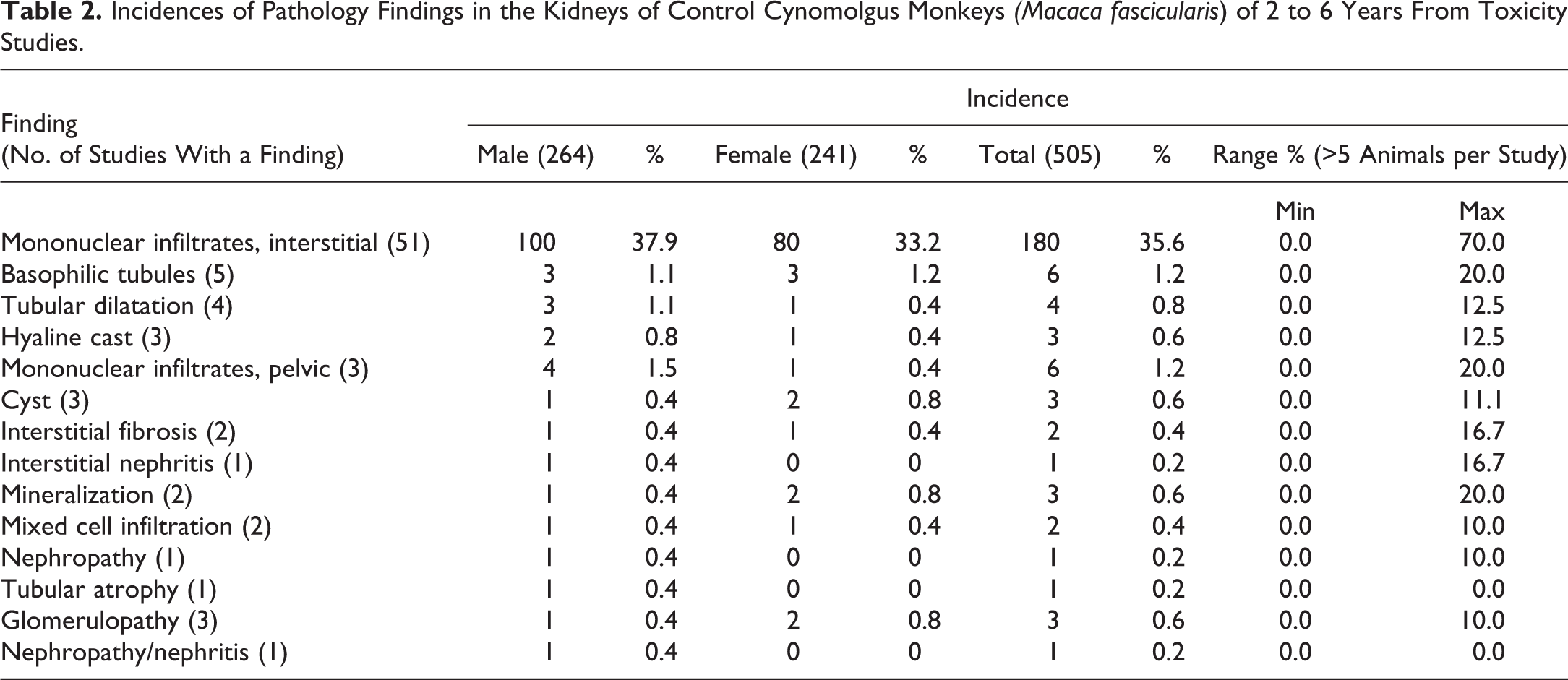
Proliferative lesions of the tubules or interstitium were rare but included cystic tubular epithelial hyperplasia (Figure 5A) and a congenital fibrogenic proliferative lesion (Figure 5B and inset) resembling a medullary fibrous hamartoma 9 or metanephric stromal tumour 10,11 in humans. The later was characterized by medullary proliferation of bland spindle-shaped stromal cells surrounding what appears to be trapped renal tubules (Figure 5B and inset).
(A-G) Spontaneous lesions of the tubules and interstitium in control cynomolgus monkeys. All sections stained with hematoxylin and eosin. (A) Focal tubular epithelial hyperplasia, cystic renal medulla. (B and inset) A fibrogenic proliferative lesion resembling a medullary fibrous hamartoma or metanephric stromal tumor, showing proliferating spindle-shaped stromal cells and trapped renal tubules (arrowheads). (C and D) Interstitial edema with expansion of the interstitium, tubular dilatation (arrowheads), and dilated vessels in the renal papilla. (E) Interstitial edema with expansion of the interstitium and tubular dilatation in the outer stripe of the outer medulla. (F) Mineralization (arrowheads) in the papillary interstitium. (G) Pigmented macrophages (arrowheads) in the papillary interstitium.
Apart from inflammation, the renal medullary interstitium also exhibited edema (Figure 5C, D, and E), mineralization (Figure 5F), and pigmented macrophages (Figure 5G). Mineralization occurred relatively commonly and was almost always confined to the papillary interstitium (Figure 5F), while pigmented macrophages were infrequent and mostly observed around the renal pelvis or the papillary interstitium (Figure 5G). Interstitial edema was most commonly observed in the distal papilla and was characterized by prominently dilated lymphatics and separation of connective tissue (Figure 5C and D). True papillary edema was difficult to differentiate from the edema-like accumulation of myxomatous connective tissue in the area cribrosa, or some postmortem artefact, but the presence of associated tubular lesions and/or interstitial inflammatory infiltrates (Figure 5D) was helpful in the diagnosis. Interstitial edema also occurred in the outer medulla of otherwise normal kidneys and was characterized by increased interstitial space, myxomatous appearance, and tubular dilatation (Figure 5E).
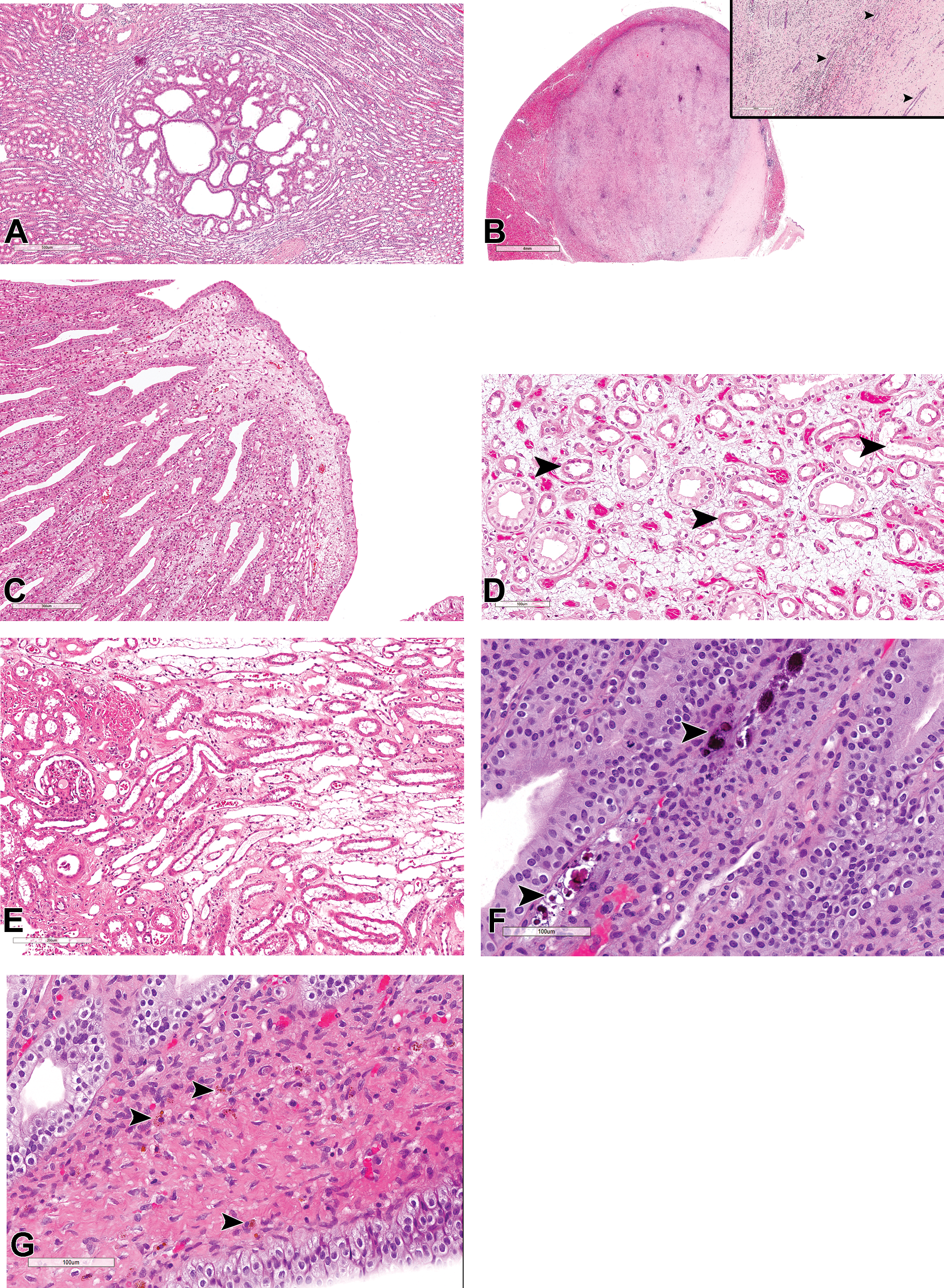
Glomerular lesions were uncommonly recorded in routine evaluation of toxicity studies, but a reevaluation of kidneys showed higher incidences (data not shown) of cases where a single or a minority of glomeruli exhibited chronic glomerulopathy or glomerulosclerosis (Figure 6A and B), or asymptomatic focal to multifocal membranous (Figure 6C and D), mesangioproliferative or membranoproliferative (Figure 6E and F) glomerulonephritis. Apart from thickening of the basement membrane of the Bowman capsule and hyperplasia of the PEC described above, segmental or global glomerulosclerosis and diffuse membranoproliferative glomerulonephritis were the 2 most commonly observed glomerular findings. Glomerulosclerosis was often characterized by segmental or global replacement of the mesangial matrix by a pale eosinophilic, PAS-positive material, initial mesangial expansion and enlargement of the glomeruli, and later periglomerular fibrosis and thickening of the Bowman capsule (Figure 6A). Segmental hyalinization (Figure 6D) or glomerular obsolescence and synechiation (Figure 6C) were occasionally observed. The vast majority of glomerulosclerotic lesions were focal in nature and lacked involvement of a majority of the glomeruli or were multifocally distributed but confined to the subcapsular cortex, in cases of small dysplastic/sclerotic glomeruli (Figure 6G). The latter, also referred to as “subcapsular glomerular sclerosis,” occurred at higher incidences in some studies, affecting several glomeruli in the superficial cortex. The lesion was characterized by small, shrunken, hyalinized, and sclerotic glomeruli that were frequently associated with hyaline casts or other degenerative and fibrotic changes in adjacent tubules (Figure 6G). These were mostly regarded as congenital lesions representing glomeruli that failed to connect to the corresponding nephron.
(A-J) Hematoxylin and eosin stained sections of glomerular lesions in kidneys from control cynomolgus monkeys showing, focal glomerulosclerosis with periglomerular fibrosis (A), and inflammation (B); focal membranous glomerulonephritis with synechia (C); focal segmental membranous glomerulonephritis (D); mild diffuse mesangioproliferative glomerulopathy with tubular casts (arrowhead) and dilated afferent arterioles (arrows; E); a focal moderately severe lesion (E inset); mild diffuse membranoproliferative glomerulonephritis with synechiae (arrowheads; F and inset); small, multifocal, sclerotic glomeruli in the subcapsular cortex (arrowheads), and associated hyaline casts (arrow; G), glomerular lipidosis (H); focal angiectasis of glomerular capillaries (I and inset); and artifactual herniation of proximal tubular epithelium into the glomerular space (arrow) with associated postmortem detachment of tubular epithelium from their basement membrane (arrowhead; J).
Membranoproliferative or mesangioproliferative glomerulonephritis were mostly focal, although minimal to mild diffuse lesions with minimal tubulointerstitial involvement were occasionally observed in some reevaluated sections (Figure 6E and F). Like the sclerotic glomeruli in the superficial cortex, such cases of diffuse membranoproliferative or mesangioproliferative glomerulopathy were observed only in a few studies, but at high incidences in those studies in which they occurred, affecting a majority of the animals in such studies. Affected kidneys showed a diffuse enlargement of glomeruli with complete filling up of Bowman space (Figure 6E and F), proliferation of the mesangium with moderate thickening of the capillary basement membrane, thickening of the mesangial stalk at the hilus with widening of the afferent arteriole or the afferent vascular chamber (Figure 6E), congestion of capillary loops, and proliferation of the PEC. Although the diffuse lesions were of minimal to mild severity, and therefore often considered to be within the expected background variation in Cynomolgus monkey, the severity of the lesions varied between different glomeruli within a section, and focal lesions of up to moderate grades of mesangial proliferation were often present in affected kidneys (Figure 6E and F). Unlike cases of glomerulosclerosis, thickening of the basement membrane of the Bowman capsule was not a major feature of membranoproliferative glomerulonephritis.
Other incidental, focal to multifocal glomerular changes that were inconsistently recorded but observed by the authors during slide review included: glomerular lipidosis (Figure 6H), focal aneurysm or angiectasis of glomerular capillaries (Figure 6I and inset), glomerular mesangiolysis-like diffuse ballooning of glomerular capillaries lesions, cystic dilatation of glomerular filtration space, artifactual herniation of proximal tubular epithelium into the glomerular space (Figure 6J), and artifactual accumulation of eosinophilic/proteinaceous globules in the glomerular filtration space.
Congenital findings included, unilateral renal hypoplasia/atrophy with compensatory hypertrophy of the contralateral kidney, renal dysplasia (Figure 7A and B) with atypical tubular epithelium, persistent renal mesenchyme, persistent metanephric ducts, and hypertrophic arteries; ectopic adrenocortical tissue attached to the cortical surface of the kidney (Figure 7C) and renal cortical cysts (Figure 7D-F). Some cortical renal cysts were associated with glomerular cysts (Figure 7E and F), which were characterized by the cystic dilatation of the Bowman capsule. Such cysts were thought to derive from the glomerulocystic lesions (Figure 7E and F). Vascular lesions were infrequent and included focal arteritis/periarteritis (Figure 7G) and localized wedge-shaped renal infarction with fibrosis (Figure 7H). Other glomerular incidental findings that were considered to represent anatomic variations or considered to be findings of little no known pathological significance are outlined in Table 1.
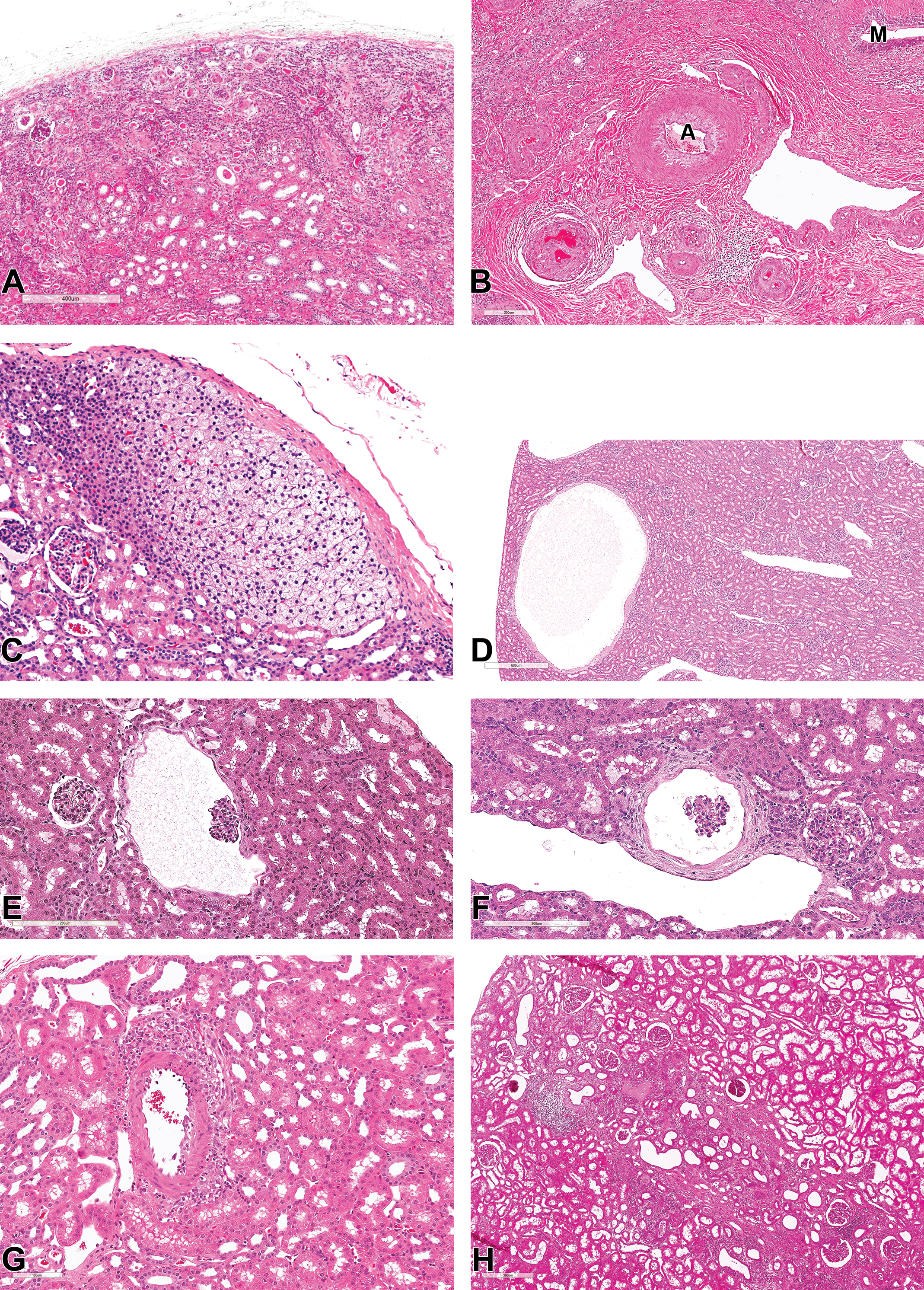
(A-H) Congenital and vascular findings in the kidney of cynomolgus monkeys. All sections stained with hematoxylin and eosin. (A and B) Renal dysplasia showing atypical tubular epithelium, persistent renal mesenchyme, persistent metanephric ducts (M), and hypertrophic arteries (A). (C) Ectopic adrenocortical tissue attached to the cortical surface of the kidney. (D-F) Cortical renal cysts thought to derive from the cystic dilatation of the Bowman capsule (glomerular cyst). (G) Focal periarteritis. (H) Focal wedge-shaped renal infarction with fibrosis.
Clinical Chemistry, Urine Specific Gravity, and Kidney Weights
Mean serum calcium, serum sodium, and urine specific gravity were highly consistent in both age groups and showed no sex-related differences (Tables 3 and 4; Figure 8A-I). Serum urea, on the other hand, showed age-related differences in both sexes, but with no sex difference at both age ranges (Figure 8D). Mature animals (4- to 6-year-old age-group) of both sexes showed slightly lower serum urea when compared with the younger juvenile animals, by a similar magnitude in males and females. Serum creatinine showed both age- and sex-related differences. In both sexes, creatinine levels were higher in mature when compared to juvenile animals, but with a more marked difference being present in males than in females. This was due to the presence of a sex-related difference in serum creatinine in mature animals, with males having higher creatinine values than females, while juvenile’ males and females had near identical mean values. Serum phosphate also showed both age- and sex-related differences, with lower serum levels in mature animals of both sexes than the juvenile group and a more marked difference being present in males than in females. Males from the juvenile animals had higher phosphate levels than females, while mature animals had similar levels between sexes.
Salient Organ Weights, Selected Clinical Chemistry, and Urinalysis Parameters Pertaining to the Kidney in “Juvenile” (2-3 years) and “Mature” (4-6 years) Control Male Cynomolgus Monkeys
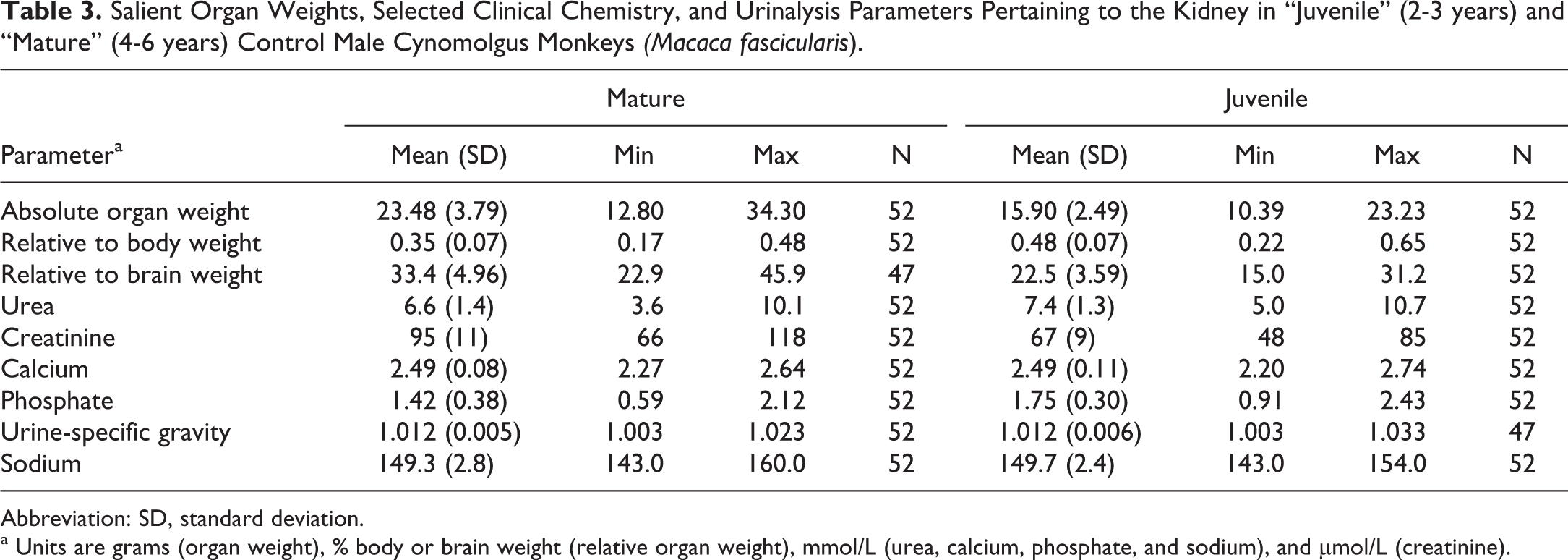
Abbreviation: SD, standard deviation.
a Units are grams (organ weight), % body or brain weight (relative organ weight), mmol/L (urea, calcium, phosphate, and sodium), and µmol/L (creatinine).
Salient Organ Weights, Selected Clinical Chemistry, and Urinalysis Parameters Pertaining to the Kidney in “Juvenile” (2-3 Years) and “Mature” (4-6 Years) Control Female Cynomolgus Monkeys
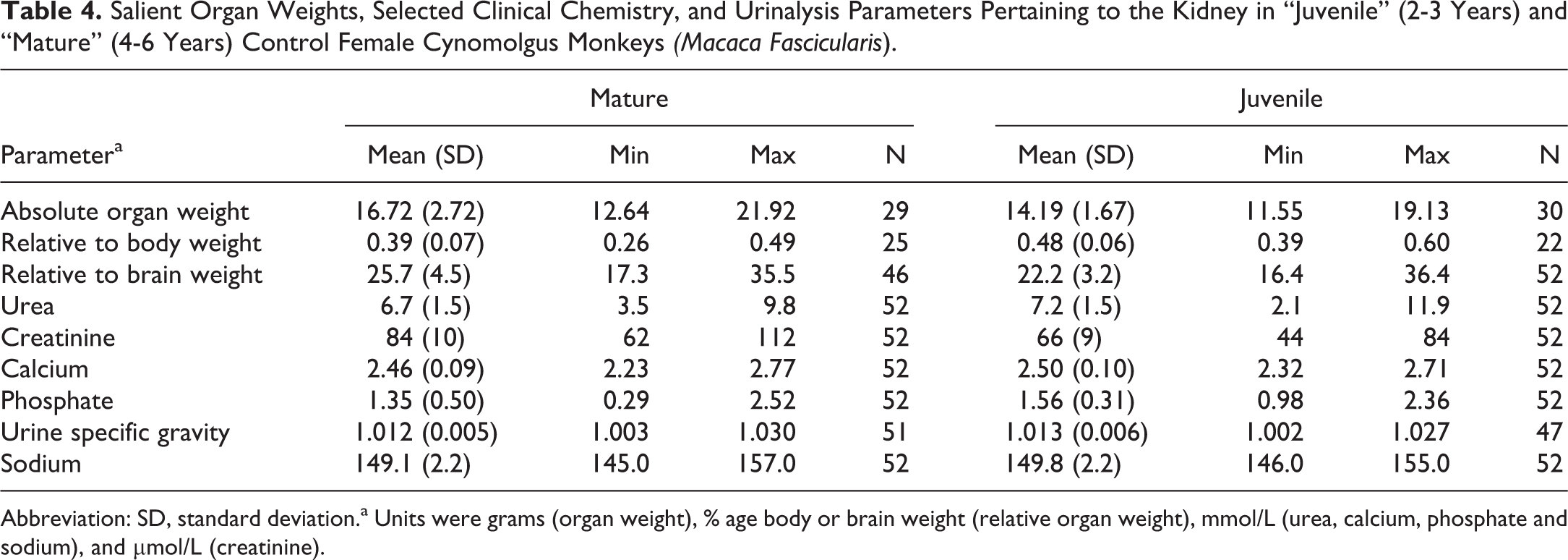
Abbreviation: SD, standard deviation.a Units were grams (organ weight), % age body or brain weight (relative organ weight), mmol/L (urea, calcium, phosphate and sodium), and µmol/L (creatinine).
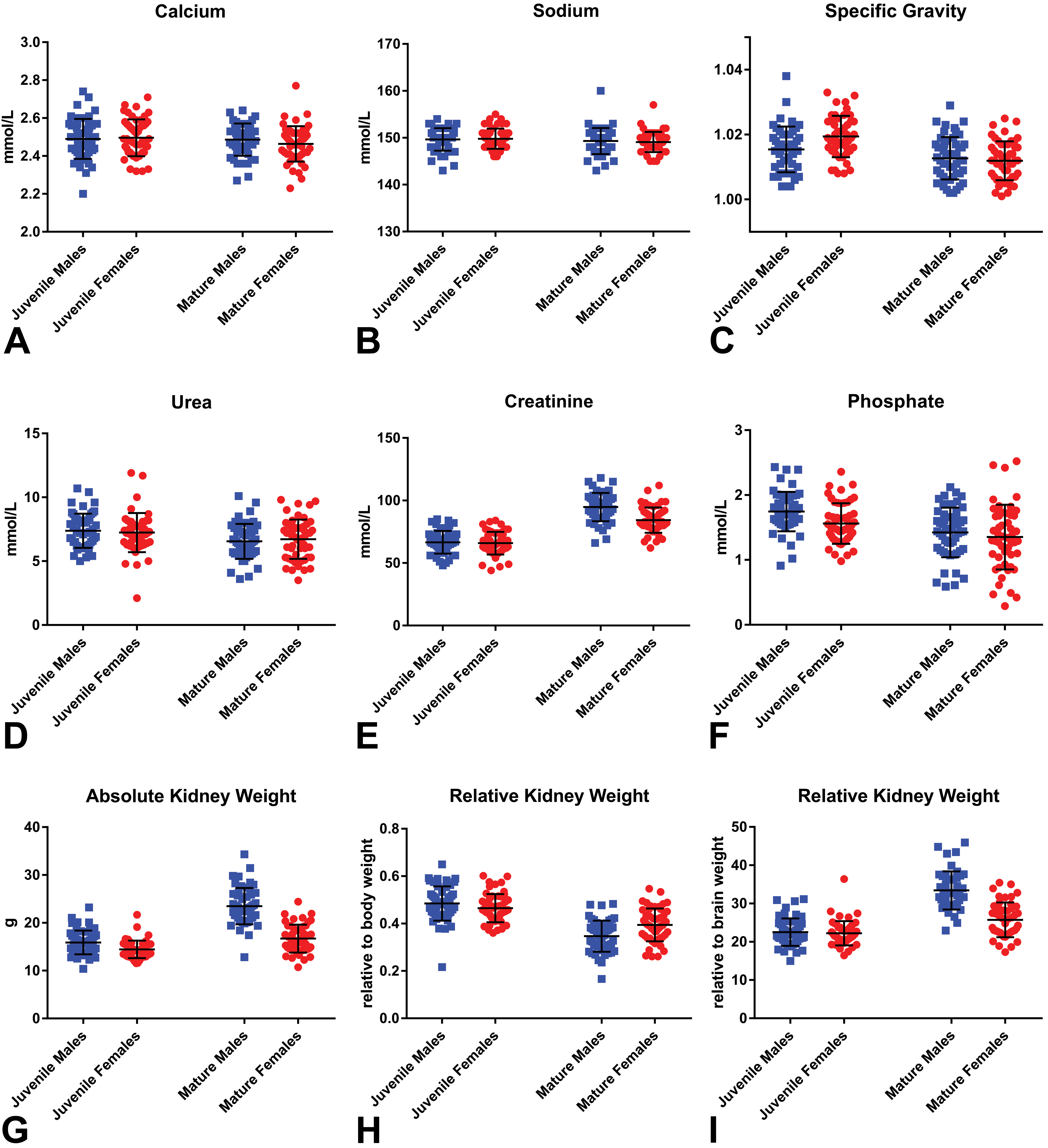
(A-I) Kidney weights and selected renal clinical chemistry parameters in juvenile and mature cynomolgus male and female monkeys. Calcium (A), sodium (B), urine specific gravity (C), urea (D), creatinine (E), phosphate (F), absolute kidney weights (G), kidney weight relative to body weight (H), and kidney weight relative to brain weight (I).
The mean, standard deviation, and range of paired kidney weights for each age/sex combination are shown in Tables 3 and 4. As organ weights are often expressed relative to body or brain weight in safety assessment studies, these values were likewise tabulated. Patterns of differences between the various age/sex combinations were expressed as percentage changes together with the statistical significance (Table 5). As expected, absolute kidney weights were higher in the older/mature group when compared to the younger age group of the same sex, with a greater magnitude of difference present in males. On average, the absolute paired kidney weights in the younger cynomolgus monkeys of 2 to 3 years were 16 grams in males, and 14 grams in females, while the paired relative kidney (to body) weights were on average 0.5% of the body weight in both sexes at this age. In the older or mature animals, the paired kidney weights were on average 24 grams in males, and 17 grams in females, while the mean relative to body weight ratio slightly decreased to approximately 0.3% in males and 0.4% in females. The relationship between paired kidney and body weights, therefore, appeared to become weaker as the animals become older, suggesting a lack of a correlation between body weight and kidney size in older males and females. The relative kidney to brain weights also showed a weaker relationship with age, but with a trend toward an increase in the ratio, due to the kidney growth or weights, outpacing those of the brain with increasing age.
Kidney Weights, Clinical Chemistry, and Urinalysis Parameters in “Juvenile” (2-3 years) and “Mature” (4-6 years) Control Female Cynomolgus Monkeys
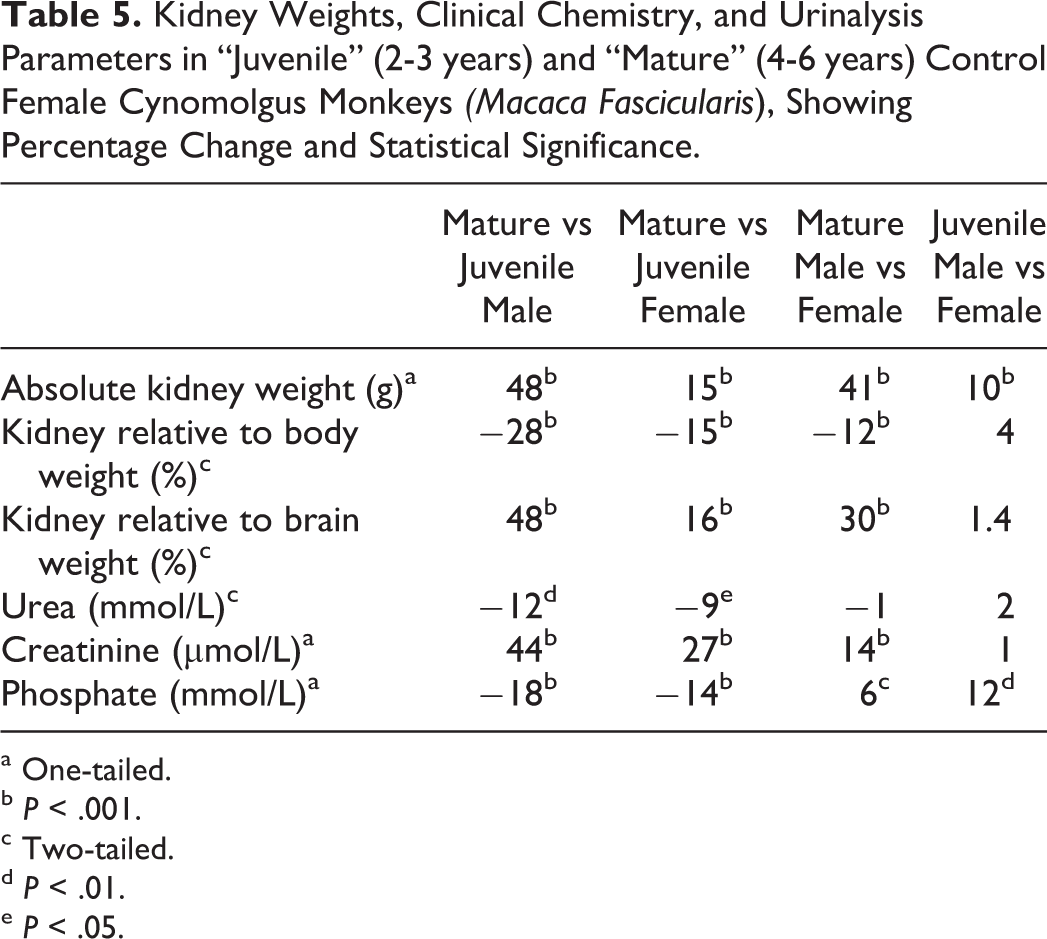
a One-tailed.
b
c Two-tailed.
d
e
Discussion
Anatomical differences notwithstanding, the main physiological functions of the kidney are shared and conserved across nonclinical species and humans. The kidney is a vital organ that plays an important role in maintaining systemic homeostasis, and as such, it is routinely and comprehensively evaluated during the nonclinical safety assessment of new therapeutic agents. Furthermore, the kidney is a unique site of test article accumulation, metabolism and/or excretion implicating active, and passive determinants of renal toxicity and toxicant fate. Therefore, a greater understanding of this organ in the nonhuman primate, a species that is central to nonclinical safety assessment, empowers the toxicologic pathologist to derive decision-driving safety data to influence drug discovery and development.
We have described the characteristic gross and microscopic anatomy of the Cynomolgus monkey kidney, including some unique anatomic features and background variations, and spontaneous pathology findings. The description of the gross and microscopic anatomy of the Cynomolgus monkey kidney in this report closely matches that described for other macaque in the literature published many decades ago. 12 –14 Our findings show that, although the anatomy of the Cynomolgus monkey kidney is generally similar to that of other laboratory animals and humans, there are key differences and unique characteristics that are critical in the evaluation of the organ in nonclinical safety assessment of drugs. Apart from the unipapillate nature of the Cynomolgus monkey kidney, other key gross or subgross anatomic features that a pathologist should be aware of when evaluating this organ include the near equivalence of the cortical to medullary ratio, the scarcity of the long loops of Henle, and the abrupt transformation of collecting ducts into ducts of Bellini; features that are all considered to be related to the shortness of the inner medulla/papilla. Similar consideration should be paid to the flattening or bluntness of the papilla as it protrudes into a shallow and rather rudimentary renal pelvis devoid of specialized fornixes, and the variably increased amount of connective tissue at the area cribrosa. Some of these features, in particular the flattened or concave shape of some papilla, may resemble secondary changes associated with vesicoureteral reflux conditions or hydronephrosis. 15,16 Knowledge of the subgross anatomy in conjunction with IHC can help to identify nephron segments targeted by a toxic compound and to directly localize biomarkers normally expressed in the kidney. 17
Microscopically, routine evaluation and identification of renal nephron segments targeted by toxicants should take into consideration some unique characteristics of the monkey kidney described here, such as loops of Henle lined by cuboidal rather than squamous epithelium, the increased amount of connective tissue in the papilla and the edematous appearance in otherwise normal kidneys, and the collection of distal tubules or connecting ducts that may resemble foci of basophilic tubules. These “normal” morphological features that are unique to the monkey kidney, and some background inter-animal variations, may be mistaken for treatment-related changes, particularly when prominent in just a few animals from the treated groups. Similarly, other background pathology findings of little or no known etiology or pathological significance listed in Table 1, such as the thickening of the basement membrane of the Bowman capsule, cuboidal metaplasia or hyperplasia of the PEC, intraepithelial pigment in the TDL and TAL, and mineralization in the papilla, should be differentiated from drug-induced changes that may closely resemble them. An understanding of the morphological characteristics and anatomic distribution of the background findings is essential in this regard. For instance, in Cynomolgus monkeys, spontaneous mineralization is predominantly interstitial and is most commonly observed in the renal papilla, as opposed to treatment-related mineralization which involves tubules and can potentially occur anywhere. Similarly, whereas spontaneous pigment deposition typically involves the TAL in the medullary rays or the TDL, treatment-related pigment deposition may occur anywhere including PCTs of the cortical labyrinth.
In the renal pelvis, intraepithelial eosinophilic inclusions observed in the urothelium are considered to represent aberrant development of tonofilaments and are frequently found in the supranuclear region of the superficial cells (umbrella cells). They are of no-known pathological significance but are suggestive of a tendency toward squamous differentiation, although the urothelium also stains positive for uroplakin, indicative of true urothelial differentiation. 18
The main spontaneous pathological changes affecting the nonhuman primate kidney have been reported and their significance in the evaluation of toxicity studies discussed. 19 –21 As previously stated, the absence of a standardized or harmonized system of nomenclature and diagnostic criteria for nonhuman primates has resulted in the use of a variety of synonymous terms by study pathologists. However, with the advent of the nonhuman primate INHAND document, and the proposed terminology for monkey glomerular lesions in recent publications, 3 some degree of harmonization of the nomenclature is expected to be achieved. This article will include synonymous terms and some of the suggested nomenclature wherever possible.
Chronic interstitial nephritis (interstitial inflammation, mononuclear, or lymphoplasmacytic), characterized by minimal to moderate accumulation of lymphoplasmacytic cells in the interstitium of mainly the outer cortex of the kidney, is the most common incidental findings in macaques. 20– 22 Interstitial nephritis is also listed as the most frequently observed lesion in a survey of lesions seen in nonhuman primates in zoos. 23 Findings range from minimal, focal, mononuclear, or lymphoplasmacytic infiltration, with little to no associated damage of renal tubular epithelium, to tubulointerstitial inflammation, characterized by large areas of moderate inflammatory-cell infiltration, renal tubular necrosis, and minimal peritubular fibrosis. Mononuclear inflammatory infiltrates associated with negligible or no renal tubular damage should be differentiated from treatment-related immune-stimulatory effects by compounds such as immuno-oncology drugs. While the composition of the inflammatory infiltrate is predominantly lymphoplasmacytic in spontaneous or treatment-related cases, the distribution and severity are slightly different with the latter being predominately perivascular (along the vascular adventitia) or likely to occur in or near vessels, and in both the cortex and medulla. In severe cases, treatment-related lesions may result in undulating elevation of the renal capsule by cellular infiltrates of predominantly lymphocytes. In addition, treatment-related infiltrates often have orthogonal evidence of systemic pro-inflammatory state or immunomodulation which must be considered in the final interpretation.
Likewise, spontaneous tubulointerstitial nephritis or chronic interstitial nephritis (interstitial inflammation, lymphoplasmacytic, chronic) should be differentiated from treatment-related tubulointerstitial inflammation based on a weight of evidence approach that includes an analysis of the distribution and severity of the lesion, localization of the finding to specific tubular segments, and the overall incidence of the finding in the study. In monkeys, as in dogs and humans (but unlike in rats) tubulointerstitial disease or chronic interstitial nephritis may result from treatment-related tubular damage leading to an immediate reaction in the interstitium. 7 The primary tubular lesions may be induced by direct cytotoxicity effects by the parent compound or metabolite to tubules, or by the accumulation of the drug material within the nephron causing obstructive nephropathy with tubular dilatation and/or inflammation. 24 Monkeys appear to be more susceptible to obstructive nephropathy, in particular protein cast nephropathy, when compared to other laboratory species and humans. 25 Protein casts formed by the binding of the compound to monkey Tamm-Horsfall protein (THP), a normal constituent of urine secreted by epithelium of the TAL, may result in tubular dilation, interstitial inflammation and edema, and lesions indistinguishable from other forms of obstructive nephropathy. 25 Typical lesions have been described with antibiotic such as sulphonamides and fluoroquinolones, some small organic carboxylates such as nonsteroidal anti-inflammatory drugs (NSAIDs) containing carboxylic acid 25,26 and biological compounds. 27 –29 It is therefore possible that similar “incidental” lesions that are infrequently observed in controls animals may be due to interventional treatment of monkeys on a study with medicinal compounds such as antibiotics or NSAID, 25,26 particularly in dehydrated animals. 30 The hydration status of an animal is thought to play a major role in facilitating the formation of crystals and THP casts. 25 Drug-induced obstructive nephropathy is not known to be a significant adverse event in humans, and therefore, the clinical relevance of the protein casts with obstructive nephropathy in monkeys is not clear. 25
Treatment-related dilatation is also frequently observed in association with tubular obstruction in the renal medulla as part of obstructive nephropathy or protein cast nephropathy 24 –26 and normally involves tubulointerstitial inflammation or edema with or without evidence of obstruction or protein casts, in addition to dilatation of collecting ducts and TAL in the medullary rays, and proximal and DCTs. Tubular dilatation associated with medullary obstruction or intrarenal reflux is normally characterized by ascending dilatation of collecting ducts, with an upstream linear pattern that follows the nephron. Conversely, spontaneous tubular dilatation is usually either confined to the cortex or occurs as a focal cystic dilatation of the collecting ducts. Several studies reviewed during the compilation of HCD reported tubular dilatation in combination with other induced changes, for example, tubular vacuolation, nephropathy, and glomerulonephritis. However, in one study, tubular dilatation occurred in isolation as a clear induced change with organ weight increase. The presence of tubular vacuolation was not recorded in any animal within the HCD indicating that this finding, either alone or in conjunction with other findings, is likely to indicate a test article effect.
Spontaneous glomerular lesions may be differentiated from treatment-related glomerulopathy by their focal nature and lack of involvement of a majority of the glomeruli or being confined to the subcapsular cortex in cases dysplastic/sclerotic glomeruli. Induced glomerular changes on the other hand frequently occur as diffuse or generalized glomerulonephritis, occasionally accompanied by tubulointerstitial or vascular changes. The majority of treatment-related, generalized glomerulonephritis is immune-mediated and driven by immune complex formation involving antibody and/or complement and therefore more commonly associated with large molecule compounds that induce antidrug antibody (ADA)–related immune-complex disease. 3 It mostly occurs as membranoproliferative glomerulonephritis. Monkeys are prone to immunoreactivity and increased susceptibility to complement activation, 31 and development or persistence of circulating immune complexes. Given that a few cases of spontaneous, generalized membranoproliferative glomerulopathy were observed in this study, differentiating treatment-related glomerulonephritis might in such cases, require consideration of additional factors such as the concurrent presence of glomerulonephritis or vasculitis, levels of ADA within the group, and further investigative techniques such as electron microscopy (EM) and immunohistochemistry (IHC). 3 Other additional factors might include, the safety target assessment, duration of dosing, drug absorption, distribution, metabolism and excretion (ADME) properties, and historical knowledge of the molecular program.
There were several noteworthy age- and sex-related trends in clinical chemistry and organ weight parameters related to the kidney. Higher serum creatinine in juveniles of both sexes was considered to be most likely due to the higher protein metabolism in growing animals or less effective excretion in less mature kidneys, as higher values were recorded in juvenile compared to mature monkeys by a similar magnitude in both sexes. The sex difference observed in serum creatinine in mature animals, with males having higher creatinine than females, was considered to be consistent with the higher expected relative muscle mass in males. Higher serum phosphate levels in juvenile compared to mature animals, and a more marked difference in males than females, were attributed to the relative differences in growth rates and bone turnover between the various groups. Regarding urine-specific gravity, it is interesting to note that despite the paucity of long loops of Henle in macaque monkeys, they are still able to concentrate urine as well as humans and other animals with a well-developed inner medulla, as determined by the range of urine-specific gravity observed in the present study compared with published values for other species. This ability to concentrate urine despite the relatively scarcity of long loops of Henle is considered to be due to the better developed vascular arrangement and higher density of the vasa recta in the inner medulla in monkeys, 32 as observed in this study.
Conclusion
Taken together, this information improves the use and evaluation of the laboratory primate kidney and the assessment of the suitability of nonhuman primate as an animal model for identifying and characterizing drug-induced renal injury and the nuances of diagnosis and interpretation of treatment-related findings in cynomolgus macaques.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
