Abstract
The minipig has long been identified as the nonrodent animal of choice for preclinical safety evaluation of topically applied materials. This article reviews types of topical applications, study designs, and practical considerations. Dermal administration to the minipig presents multiple challenges. Issues to be considered are the area of exposure, the nature of the test article, the need for covering of the dose site, preparation of the dose site, and procedures for administration. The potential for cross-contamination (exposure of control animals to test article) is very high in topical studies and appropriate safeguards to prevent this are discussed. Topical administration to the intact skin is the most commonly utilized form of safety evaluation, but procedures have also been developed for intradermal administration and for the use of nonintact/wounded skin; these will be discussed. Evaluation of local (topical) effects is critical and can become complicated. Considerations in evaluations of the skin are interobserver variability, use of different scoring systems and discrepancies between in vivo observations, necropsy observations, and microscopic pathology findings. Causes for apparent discrepancies and suggestions for practical resolution through appropriate procedures are discussed. Practical issues in necropsy and histotechnology procedures and techniques to optimize preservation of skin are also discussed.
Introduction
The pig is the nonrodent of choice for evaluation of topical products because of the similarity of porcine and human skin anatomy and biochemistry (Mortensen, Brinck, and Lichtenberg 1998; Montiero-Riviere and Riviere 1996). This article is an overview of the use of the minipig in safety evaluation of topical products that discusses regulatory and study design issues and special considerations that may be of particular interest to anatomic pathologists.
Why Minipigs?
Characteristics of porcine skin more closely resemble those of human skin than do those of other common nonrodent models used in safety evaluation (dogs and nonhuman primates). The rabbit was previously the nonrodent model of choice for dermal evaluations but is seldom used currently because of the poor anatomical and physiological correlation of rabbit and human skin and the tendency for studies in rabbits to overpredict effects in humans. The minipig is preferred over the domestic pig because of its manageable size. Several strains of minipig are commercially available; the Sinclair (Hormel), the Yucatan, the Hanford, and the Göttingen. These strains vary in hair coat and growth patterns. Hair coat and color in the Sinclair are variable, ranging from white to dark brown. The Yucatan generally has gray/dark pigmentation and little or no hair, although one line is hairless with white skin. The Hanford has white skin and hair. The Göttingen minipig has pale pink skin and white or nonpigmented hair, which make dermal effects easy to evaluate. Body weight growth rates are slowest in the Göttingen and most rapid in the Hanford (Swindle 2007). Based on the pale skin and slow growth rate, the Göttingen is the minipig of choice in many laboratories. Because of the widespread use of minipigs, many laboratories have established historical control databases; the Göttingen supplier (Ellegaard, Dalmose, Denmark) also has historical data collected over several years, thus making this strain an attractive choice.
Regulatory Requirements/Guidance
Regulatory guidances exist for preclinical safety evaluation of pharmaceuticals (drugs and biologics) intended for human use. However, there are no specific regulatory guidelines for preclinical testing of pharmaceuticals by topical administration (with the exception of some guidelines for special cases such as chronic ulcers, burns, and wounds). Regulatory guidelines are intentionally nonspecific in order to allow for flexibility and a tailored approach. Regulators can provide informal guidance and often present specifically on dermal administration at scientific meetings (Jacobs 2003 [Society of Toxicology]; Wang 2014 [American College of Toxicology]) or contribute to nongovernment publications (Jacobs and van der Laan 2012). In the United States, dermal products are regulated by the Division of Dermatology and Dental Products in the Office of New Drugs in the Center for Drug Evaluation and Research.
The only regulatory guidelines that specifically refer to dermal administration are those published by the U.S. Environmental Protection Agency (EPA) and the Organization for Economic Cooperation and Development (OECD) for safety evaluation of materials with a high potential for accidental/inadvertent topical human exposure (chemicals). These guidelines focus almost exclusively on in vitro studies or on safety evaluation in rodents, with essentially no testing in nonrodents and are, therefore, beyond the scope of this article. However, they do contain useful information about the body surface area to be covered by topical products (see Discussion section, under Dose Sites, and reference to Table 1).
Dose Area for Dermal Studies—Regulatory Guidelines.
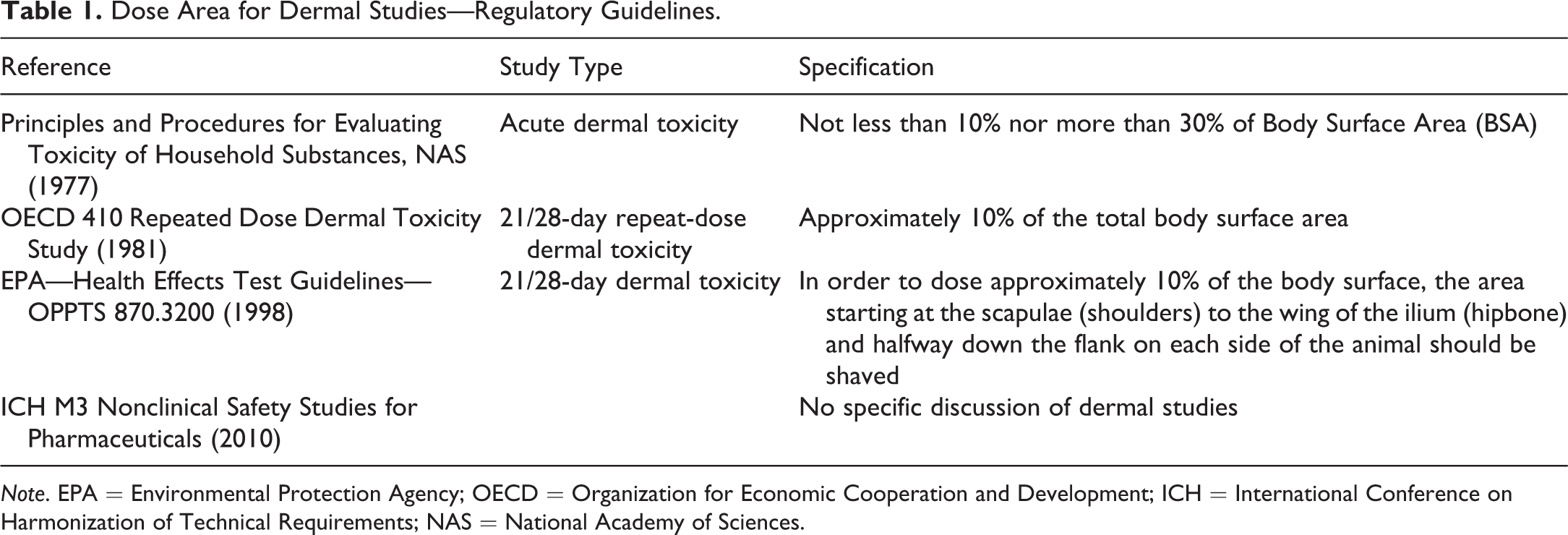
Note. EPA = Environmental Protection Agency; OECD = Organization for Economic Cooperation and Development; ICH = International Conference on Harmonization of Technical Requirements; NAS = National Academy of Sciences.
Human exposure, through topically applied drugs or biologics, is regulated in the United States by the Food and Drug Administration (FDA), in Europe by the Committee for Proprietary Medicinal Products of the European Agency for the Evaluation of Medicinal Products, and in Japan by the Ministry of Health, Labor and Welfare. Guidelines are harmonized internationally by the International Conference on Harmonization of Technical Requirements (ICH). Regulatory guidelines for safety evaluation of small molecules are presented in the ICH M3 Guidance for Industry (Nonclinical Safety Studies for the Conduct of Human Clinical Trials for Pharmaceuticals; ICH 2010). Similar guidance for biopharmaceuticals is presented in the ICH S6 guidelines (ICH 2011). However, very few biopharmaceuticals are developed for topical administration and these studies are designed on a case-by-case basis. This article will, therefore, focus on study designs for small molecule products.
Study Designs
Studies are designed with varying durations based on the anticipated human exposure in clinical trials. Evaluations for definitive studies are described as: acute (single exposure), subchronic (generally 2 weeks to 3 months), and chronic (6 of the 9 months).
Requirements for duration of Good Laboratory Practices (GLP) preclinical studies are summarized in Table 2. Range-finding studies (non-GLP), ranging from single-dose “up and down” studies to 14-day studies, are frequently conducted to select doses for subsequent definitive studies.
Duration of Repeated Dose Toxicity Studies in Nonrodents to Support Clinical Trials.

aAs an alternative to 2-week studies, single-dose toxicity studies with extended examinations can support single-dose human trials in the United States.
Topical products, like all pharmaceuticals, are evaluated for systemic toxicity. Because of the topical route of administration, evaluations of local (dermal) toxicity are also necessary for these materials. Systemic and dermal evaluations are generally incorporated into a single study, but this distinction should be kept in mind when designing a safety evaluation program.
Acute Studies
Evaluation for acute systemic toxicity is part of a standard drug development program. A typical program for a topically administered material would include single-dose studies that would be used to evaluate irritation and systemic toxicity potential and select doses for repeat-dose studies. Single-dose GLP studies with extended examinations (usually for up to 2 weeks) can be used to support single-dose administration to humans in some cases, as described in Table 2.
If no previous information about the toxicity of the test article is available, a preliminary range-finding/maximum tolerated dose study may be conducted in a small number of animals. One or 2 minipigs per sex (or 1 or 2 animals of 1 sex) may be dosed in a sequential (up and down) pattern, with doses increased or decreased based on results of previous doses, to establish doses for further evaluations. The end point of these studies may be local effects (for materials with known irritation potential) or systemic effects of the absorbed drug. Frequently, there is no significant absorption, and acute studies of topically administered materials are limited to determination of the maximum feasible dose (MFD), the largest volume that can be administered of the highest concentration of test article that can be manufactured.
MFD Determination
This is a non-GLP evaluation performed early in a program to establish the maximum dose that can be administered. The MFD determination is generally performed using a single animal, and it is done during preliminary evaluations often during the pretest period of a repeat-dose study. The dose site is prepared as it will be used in the study and the vehicle (assuming it is of the same consistency as the test formulation) is applied over the area. An initial volume of 0.5 ml/kg is applied and coverage of the dose site is evaluated. If this volume does not completely cover the site, additional volumes of 0.5 ml/kg are added, stepwise, until a volume that covers the dose site but is not excessive (does not run off or extend beyond the site) is determined. Dose volumes of up to 2.0 ml/kg may be evaluated, but the maximum volume is generally 0.5 to 1.0 ml/kg. Although some FDA reviewers have been quoted as saying that 2.0 ml/kg is a required volume, this volume (20 ml for a 10-kg animal) is excessive for most materials and is rarely considered “feasible.”
Subchronic Studies
Once appropriate doses have been established with acute studies, a range-finding study or a repeat-dose toxicity study may be performed. A 7-day range-finding study is a non-GLP study designed to evaluate repeat-dose toxicity in a small number of animals (1 or 2 animals per sex in control and test article–treated groups) and may or may not be needed based on the preliminary studies. A 14-day, 28-day, or 3-month repeat-dose study is considered a definitive GLP study for regulatory purposes; duration would be determined by proposed clinical duration (Table 2). Typically, groups of 3 or 4 minipigs per sex, with controls (untreated/sham treated and vehicle treated) and 3 or more treated groups, are used. The use of 2 control groups is a regulatory expectation to clarify any effects that may be related to the vehicle. Treatment would be daily topical administration. Duration of daily exposure and presence or absence of dose site coverings are discussed below. Routine evaluations (clinical signs, body weight measurements, food consumption evaluations, ophthalmology, electrocardiograms, clinical pathology, organ weight measurements, and macroscopic and microscopic pathology) as well as evaluations of dermal responses are performed.
Collection of electrocardiograms can be a challenge if animals are jacketed. Currently, the standard procedure is to collect single-point leads over a short interval (1 min). Leads are placed predose for a pretest evaluation and are left in place (with the cables disconnected) for dose administration and covering/jacket application. At the designated postdose time, jackets are removed or are moved to gain access, cables are reapplied, and electrocardiograms are obtained. Our laboratory is currently working with a jacket manufacturer (Lomir, Malone, NY) to design a dermal jacket with a pocket for an EMKA (Falls Church, VA) transmitter that can be used to obtain continuous electrocardiograms in telemetered animals. Toxicokinetic evaluations are generally performed after the first dose and near the end of the treatment period. Recovery from any effects is evaluated by holding some animals from each group, or minimally from the control and high-dose groups, for a treatment-free postdose period, generally of the same duration as the treatment period.
Chronic Studies
Chronic testing (6 or 9 months) in nonrodents is required for pharmaceuticals with a potential for chronic human exposure. Chronic toxicity study durations of 9 months for nonrodents are accepted internationally (ICH 1998), although some countries require only 6-month studies. Designs are similar to those for subchronic studies, described above, generally with 4 or 5 minipigs per sex per group (Auletta 2014).
Animal Welfare Issues
Because of the potential for pain and skin damage resulting from topically applied materials, careful monitoring of dermal irritation and toxicity studies is mandated by animal welfare regulations (Auletta 2004). In the United States, the care and use of minipigs are regulated by the U.S. Department of Agriculture under the Animal Welfare Act (AWA, amended 1985). The AWA states that skin irritancy testing is an example of a procedure which can be expected to cause more than momentary or slight pain. It states that the dosing procedure itself is generally not painful, but the reaction caused by the product being tested may cause pain. In the study protocol submitted for prestudy approval by an institution’s Institutional Animal Care and Use Committee (IACUC), it is important to provide complete information on any potential effects and to clarify steps to be taken if severe irritation is seen. The following conditions are considered painful: severe erythema, erosions, ulcers, abscesses, and necrosis. The AWA further states: “Animals exhibiting signs of pain, discomfort or distress, such as adverse reactions to touching inoculated areas, open sores/necrotic skin lesions, abscesses, are expected to receive appropriate relief unless written scientific justification is provided in the animal activity proposal and approved by the IACUC.” In actual practice, reactions such as those described above are unacceptable for a material designed for human use and would not be tolerated in a dermal toxicity evaluation of a pharmaceutical. If unexpectedly severe effects are seen and there is good scientific rationale to continue the study, options to provide pain relief are limited. Topical pain relief medication, which would interfere with test material administration and absorption and would mask effects, is unacceptable. Therefore, options available are euthanasia of affected animals, administration of systemic analgesics, decreasing the dose (with application to undamaged skin), or discontinuing the dose/study.
Study Procedures
Doses/Exposure
The majority of dermal safety evaluation studies in minipigs are conducted by topical application to the skin. Doses are selected based on regulatory guidelines (ICH M3(R2) 2010, Section 1 E, High Dose Selection for General Toxicity Studies) or on information available from preliminary studies (described above) and toxicokinetic information. Doses should provide appropriate multiples of human exposure, ideally causing no systemic effects and minimal or no topical effects at the lowest dose and some systemic and, possibly, topical effects at the highest dose. Some topical effects may be tolerated, and the FDA advises that evaluation of dermal irritation should be incorporated into the repeat-dose studies (rather than conducting stand-alone irritation studies). However, the primary goal of these studies is to evaluate systemic toxicity; severe topical effects are not considered acceptable. Because of the limitations of the size of the dermal dose site and the ability to prepare highly concentrated dose formulations, high-dose selection is often limited to the MFD, as discussed previously. Varying doses should be achieved by varying the drug concentration and administering the same volume to all animals, although in some cases, it may be necessary to administer varying doses of a formulated product. Doses can be administered on the basis of body weight (milliliter per kilogram or milligram per kilogram) or body surface area covered (square centimeter). Frequency and duration of exposure vary depending on the proposed clinical exposure. Frequency of application is usually daily but may be more frequent (2 or more times per day) in order to mimic clinical use and maximize exposure. In most cases, exposure is continuous, but a limited exposure period (4–6 hr per day) may be used with removal of material after the designated interval. “Continuous” exposure is actually 20 to 22 hr per day with a “rest” period between administrations during which any residual material from the previous application is removed.
The volume that can be administered to a minipig varies with the nature of the material, for example, it may be feasible to apply a larger volume of a gel than a liquid. Maximum volumes are generally 0.5 to 1.0 ml/kg of body weight. A determination of the MFD can be conducted on 1 to 2 animals in a preliminary study or as a preliminary component of a repeat-dose study as described previously.
Dose Site
The site for the application of the test formulation is a single continuous area over the dorsum and approximately halfway down the sides of the trunk. The skin in this area is generally the least accessible for oral ingestion. Regulatory guidelines (summarized in Table 1) generally state that the dose site should be at least 10% of the body surface area; only 1 reference (EPA—Health Effects Test Guidelines—OPPTS 870.3200, 1998) designates the anatomical landmarks to be used: in order to dose approximately 10% of the body surface: “The area starting at the scapulae (shoulders) to the wing of the ilium (hipbone) and half way down the flank on each side of the animal should be shaved.” This definition, using anatomical landmarks, is commonly used for designation of the dose site. However, some laboratories calculate the dose site area using a body surface formula. Many formulas exist; the one most commonly used for minipigs is the following (Spector 1956):

Using this formula, 10% of the BSA for minipigs would be 394 cm2 for a 10-kg animal and 451 cm2 for a 12-kg animal. This is similar to the areas measured using anatomical landmarks, 320 and 437 cm2, respectively (Ramani et al. 2014).
Dose Site Preparation
On the day before initiation of dermal application, the hair is closely clipped and shaved from the trunk (dorsal surface and sides) with an electric clipper and shaver. Clipping alone is often considered adequate, but the additional close shaving procedure produces a better dermal surface for test article absorption. Care is taken to avoid abrading the skin. Each corner of the clipped/shaved dose site is marked by tattooing prior to Day 1 of the dosing phase. It is assumed that the area marked will increase as the animal grows during the study and the dose area will be constant relative to the animal’s body weight. During the study, the dose site is reclipped/shaved as needed based on hair regrowth, generally weekly. Clipping/shaving procedures should be conducted after dermal irritation evaluation and at least 30 min prior the start of the dermal application procedure for the next dose. Dose formulations are generally applied to intact skin, although, in some cases, abrasions or tape stripping may be used to simulate damaged skin, based on the clinical use/therapeutic intent of the test formulation. In special cases (therapeutics intended for treatment of wounds), wounds may be created in the skin (Sullivan et al. 2001).
Wounding
For products specifically intended for treatment of wounds, the application site may be prepared for dosing by creating surgical wounds in the skin. In dosing, handling and evaluation of wounds, adequate pain management, and coordination with an attending veterinarian and the company’s IACUC are essential. A specific FDA Guidance for Industry discusses considerations for evaluation of chronic cutaneous ulcer and burn wounds (FDA, 2006).
Surgical wounds can be created by full thickness incision, partial thickness removal of some of the dermis with the overlying epidermis, and punch biopsy. Partial thickness wounds can be created with a dermatome, a surgical instrument used to create skin grafts in humans. Some familiarity with the dermatome is needed to produce wounds of uniform dimensions, so practice on cadaver animals is recommended. Dermatome wounds can be covered individually by over-the-counter heavy duty adhesive bandages. Individually bandaged wound sites can then be further protected by a semiocclusive dressing (see below). Small full thickness wounds can be created with biopsy punches of various diameters.
Dose Application
Dose formulation is applied to the clipped/shaved skin of the animal using a syringe or a spatula and rubbed over the appropriate dose site as evenly as possible. If the formulation is too viscous to draw through a syringe, the syringe may be filled by removing the plunger and filling through the barrel end of the syringe or by filling a smaller syringe with a larger syringe using a 3-way stopper (Figure 1). Dose formulation can remain on the dose site between 6 and 20 hr per day.
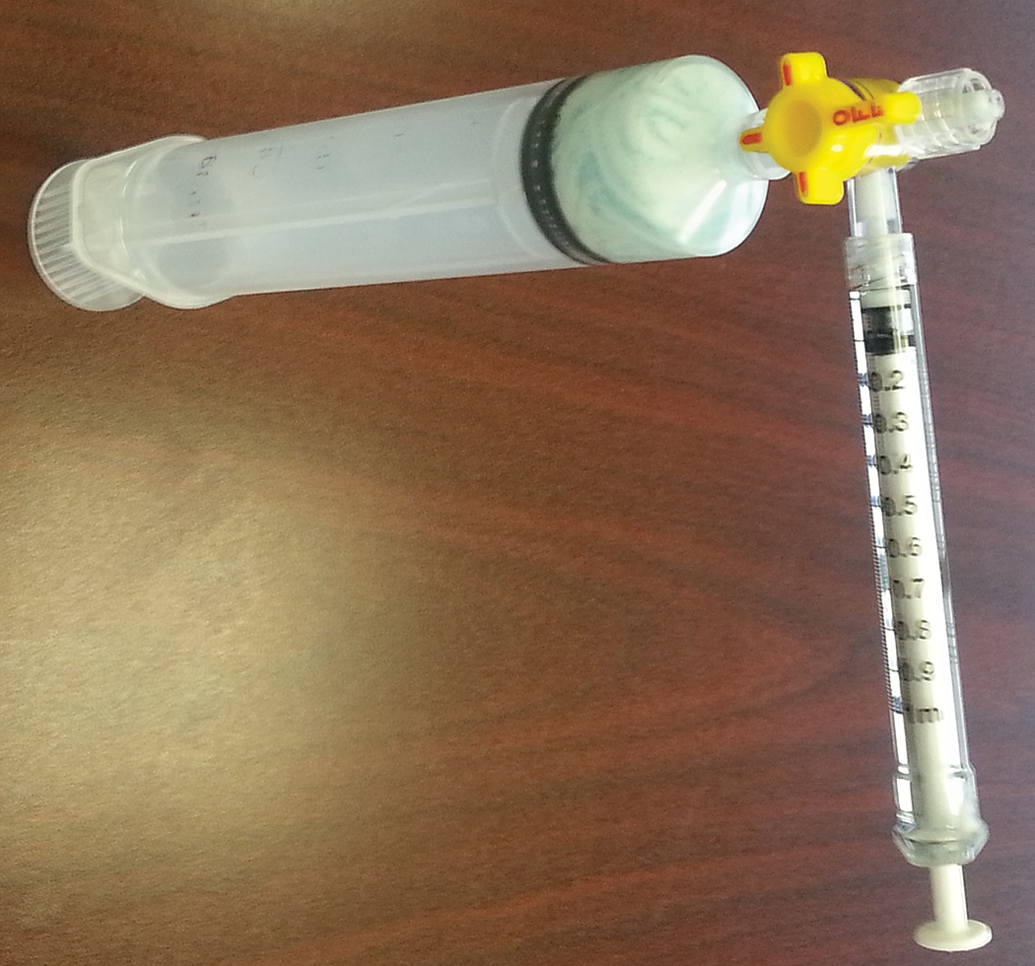
Application tool for dosing dermal formulations.
Covering the Dose Site
Open application (no covering) may be an option where the test formulation dries rapidly and the clinical exposure is to open skin. However, the dose site is usually covered after dermal application to minimize the risk of oral ingestion and mimic the clinical exposure. The most common covering is semiocclusive, using a breathable dressing to cover the dose site. This dressing most closely represents the clinical situation in which dermal formulations are covered with (semiocclusive) clothing. A nonbreathable (fully occluded) dressing is generally not well tolerated, especially on a repeated basis, is more likely to produce dermal irritation and foster microbial growth, and may increase absorption. Animals are habituated to the semiocclusive dressing for at least 2 occasions of varying duration prior to initiation of dosing to confirm the maximum duration that the covering can be tolerated.
There are many ways to prepare and apply a semiocclusive dressing. All have the same goals of covering the dose site without causing unnecessary distress and resistance from the animal and of assuring that the covering stays in place. Some coverings are manufactured commercially, such as Lomir® T-shirts (Figure 2). Many laboratories use coverings constructed by their technical staff using commercially purchased components. The “ideal” covering varies from laboratory to laboratory and is often achieved after much trial and error. The covering used at Envigo (Figure 3) consists of gauze material secured with Durapore tape (Staples catalog No. 112520). The minipig is then dressed with tubular elastic netting (Staples catalog No. NONNET06 or NONNET07) and a protective, cushioning collar (head band, Target catalog No. 14870148) secured with Velcro. Use of the semioccluded dressing may be eliminated or discontinued if skin abrasion/erythema or other signs occur and may be resumed when affected skin areas are adequately healed.

Lomir® T-shirt.
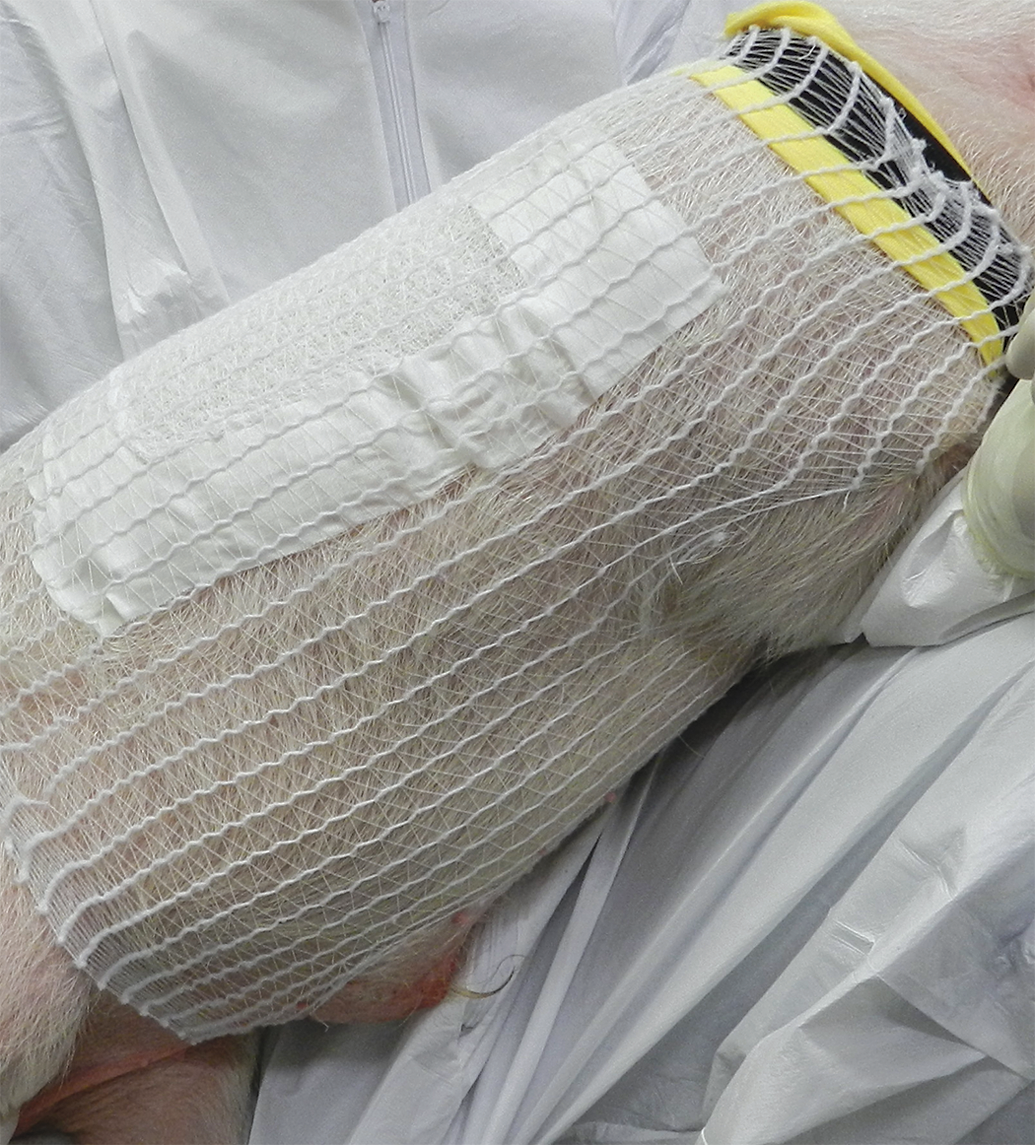
Envigo dose covering.
Washing
After completion of the required duration of exposure, the semiocclusive dressing is carefully removed and the dose area is gently washed with a mild cleanser (e.g., Cetaphil), followed by rinsing with tap water (at least twice) and blotting dry with cotton gauze. This procedure may be repeated if deemed necessary.
Potential for Cross-contamination
Because of the external nature of dermal application and the propensity for minipigs to rub against the sides of their pens, cross-contamination, especially contamination of control animals with the test formulation, is a major concern for studies with topically applied materials. In order to mitigate potential cross-contamination of the dose formulation, the following procedures should be used: (1) animals should be spatially separated by an empty pen between control and test formulation-treated animals or by housing control animals in a separate room; (2) trafficking and study procedures should be conducted in ascending group order, starting with control animals; (3) personnel must frequently change protective clothing/gloves between dose groups or even between animals; (4) personnel must avoid touching any surfaces unnecessarily; (5) separate dedicated equipment should be used for control and each dose group; and (6) a defined cleaning solution and/or method must be established and used for the duration of the study. It is important to adequately train personnel and assure strict adherence to these procedures.
Intradermal Injection
Although not commonly performed in minipigs, test materials can be administered to the skin by intradermal injection at specific locations. Intradermal injection is not possible in the skin on the back and sides because the dense weave of the dermal collagen fibers resists the injection of fluid. However, intradermal injection is possible in the more pliable skin behind the ear and in the axillary and inguinal areas (Figures 4 and 5). The dermis is less dense at these sites and easily accepts injection of a small volume of fluid (Ramani et al. 2013). It is necessary to sedate minipigs in order to access these areas.
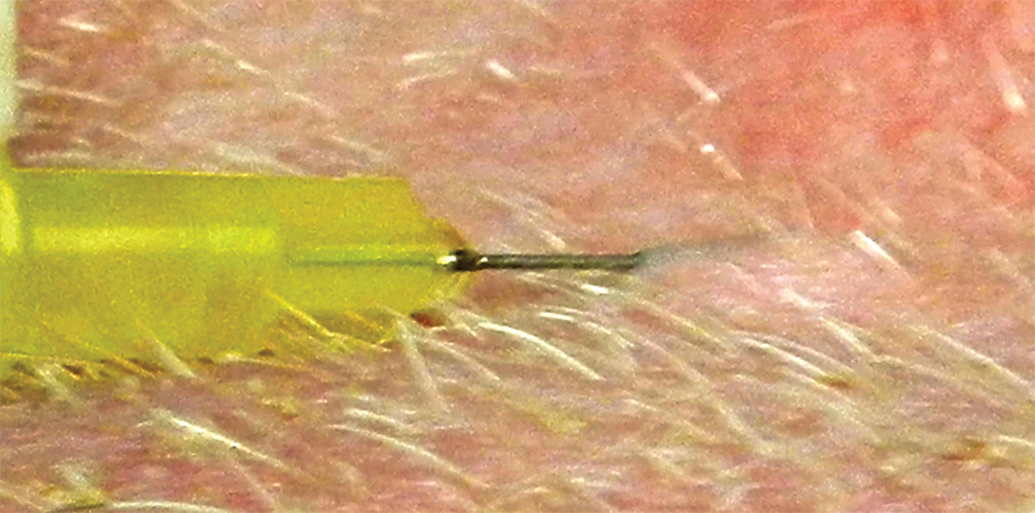
Needle inserted just below skin surface in axillary skin.
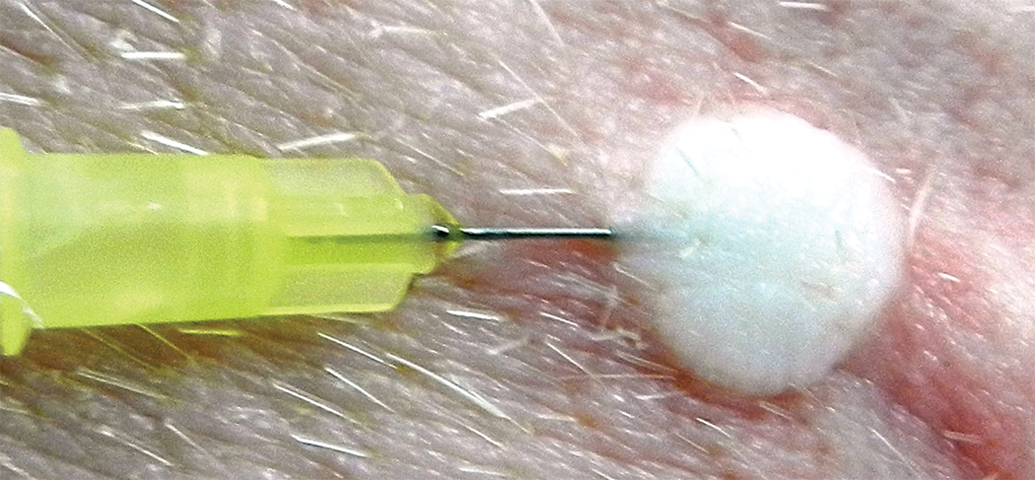
Intradermal injection of 0.1 ml of saline creates a small wheal in axillary skin.
In-life Observations and Evaluation of Effects
Systemic effects of dermally applied materials are evaluated in the same way as for materials administered by other routes. Local effects in the skin are generally evaluated using scoring systems. The “Draize” system is often specified for evaluation of dermal irritation effects. However, this is an outdated system (Draize, Woodard, and Calvery 1944) that was originally designed for categorizing dermal hazards of chemicals (household substances) based on a single exposure (Draize 1959). A generic Draize system, rather than the literal Draize system, is suggested as a more appropriate way to categorize dermal effects of topical pharmaceuticals in current safety evaluations. This is a more generalized and simplified system (Table 3) that allows for adequate evaluation while eliminating some sources of individual variability among the technical staff who make dermal observations. For example, the potentially confusing 4 Draize categories for erythema of very slight (barely perceptible), well-defined, moderate to severe, and severe are replaced by the more easily understood and commonly used categories of slight, moderate, and severe. This system also provides for some additional dermal findings frequently seen in repeat-dose studies. Variability in evaluations using the Draize system was demonstrated dramatically in a classic study by Weil and Scala (1971), which compared scores for the same materials evaluated in different laboratories and found a high degree of interlaboratory variability. Providing a comprehensive training program with periodic recertification and limiting the core technical staff to a small number of technicians using similar evaluation criteria can help provide consistent results within a laboratory.
Grading of Skin Reactions.

Frequency of dermal evaluations varies with the test material and study type. Frequent observations, generally specified in testing guidelines, are required for single-dose irritation studies. For long-term, repeat-dose studies, irritation potential has often been established in early studies and only a general, periodic evaluation is needed. Decreasing the frequency of dermal evaluations over time is suggested, depending on study duration and the type of irritation seen. Initial evaluations may be made daily, if necessary, to establish any potential unexpected responses. Subsequent evaluations may then be performed weekly and may be incorporated into routine physical examinations. Scores may be assigned (Table 3) or a notation of presence or absence of any dermal abnormalities may suffice.
Necropsy/Histology
Necropsy
At necropsy, the skin should be examined carefully to identify macroscopic findings and correlate them with in-life observations before the skin surface is disturbed. Scabs and superficial debris can be mechanically dislodged or altered by contact with fluids during the necropsy procedure. Red discoloration due to vascular congestion will disappear after exsanguination, so an in-life observation of erythema may not be confirmed upon microscopic examination. The appearance and nature of skin lesions often change with fixation; red discoloration may turn brown, fade, or disappear, and test material and superficial secretions may dissolve away.
Macroscopic observations should be described in enough detail to ensure that they can be accurately identified after fixation. Orientation marks (left, right, dorsal, and/or ventral) can be made on the skin with permanent marker to help orient the specimen(s) at trim. Untreated skin should be sampled from the same general body area as treated skin to minimize variations in skin thickness and follicle density. Protocol skin is typically taken from the inguinal area and is used to evaluate systemic effects on the skin. Inguinal skin is not a suitable comparator for treated skin from the dorsum or sides because histological differences in the skin from each site would confound identification of test article–related changes. To ensure adequate fixation, skin specimens should be placed into a formalin container large enough to maintain a 1:10 skin to formalin ratio.
If required for toxicokinetic evaluations, full thickness skin samples may be collected to quantitatively assess test article concentrations. These samples should be collected prior to placing the rest of the skin specimen(s) into fixative. The time of necropsy/skin collection should be recorded for each individual animal to better match toxicokinetic plasma exposure to skin exposure.
Histotechnology
In the ideal skin section, hair follicles are oriented vertically along their long axis. This allows the pathologist to evaluate the entire hair follicle and determine its stage in the hair cycle, if necessary. Skin must be trimmed with the grain of the hair (in the direction of hair growth) to achieve this orientation (Ruehl-Fehlert et al. 2003). Because the application site is typically shaved, the grain of the hair may be difficult to perceive and it also varies from one location to another. In addition, the hairs may be white or transparent, making them difficult to see. Using the orientation markings made at necropsy, the trimmer should be able to identify the anterior, dorsal, posterior, and ventral edges of the application site and establish the general direction of hair growth. Magnification is helpful, both to determine the direction of hair growth prior to trimming and to confirm that the hair follicles are properly oriented on the cut surface of the skin section. Use of a handheld illuminated magnifier (DermLite; 3Gen Inc., San Juan Capistrano, CA) is a quick and easy way to examine the cut surface. Because hair follicles are widely spaced (Figure 6), a large enough skin sample must be taken to ensure that at least a few follicles are present.
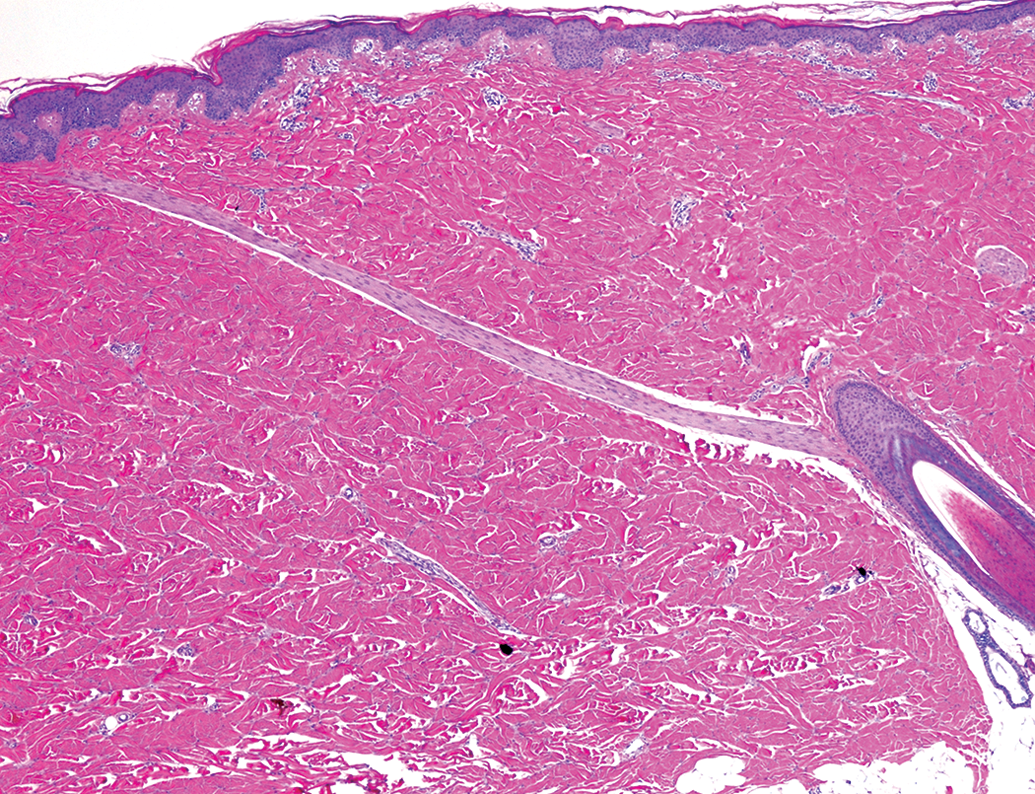
A long erector pili muscle originates from a hair follicle in the lower right and passes diagonally through the dermis to the upper left. A cross section of another erector pili muscle is visible along the right-hand edge of the photograph halfway between the epidermis and the hair follicle. Note the interlacing collagen bundles in the compact dermis and the absence of other hair follicles.
Histology of Minipig Skin
Most topical materials are applied to the skin of the dorsum and sides, so histologic features of the skin in these regions are of greatest relevance to histopathological evaluation of dermal studies. In the standard application area, the dermis is thick and compact and is composed of tightly interwoven collagen bundles (Meyer, Neurand, and Radke 1982; Figures 6 and 7). Hair follicles are sparse. Many follicles are relatively large and have large erector pili muscles (Figure 6), which are conspicuous features in the dermis. Simple tubular apocrine glands are associated with hair follicles (Figure 7) and their ducts open directly onto the skin surface near follicular orifices. Apocrine glands become active at puberty and are not involved in thermoregulation (Mortensen, Brinck, and Lichtenberg 1998). Sebaceous glands are associated with hair follicles in some regions of the skin. The subcutis can be very extensive, depending on the age and nutritional status of the animal. Additional details about porcine skin are available in the literature (Meyer, Schwarz, and Neurand 1978, Montagna and Jeung 1964).
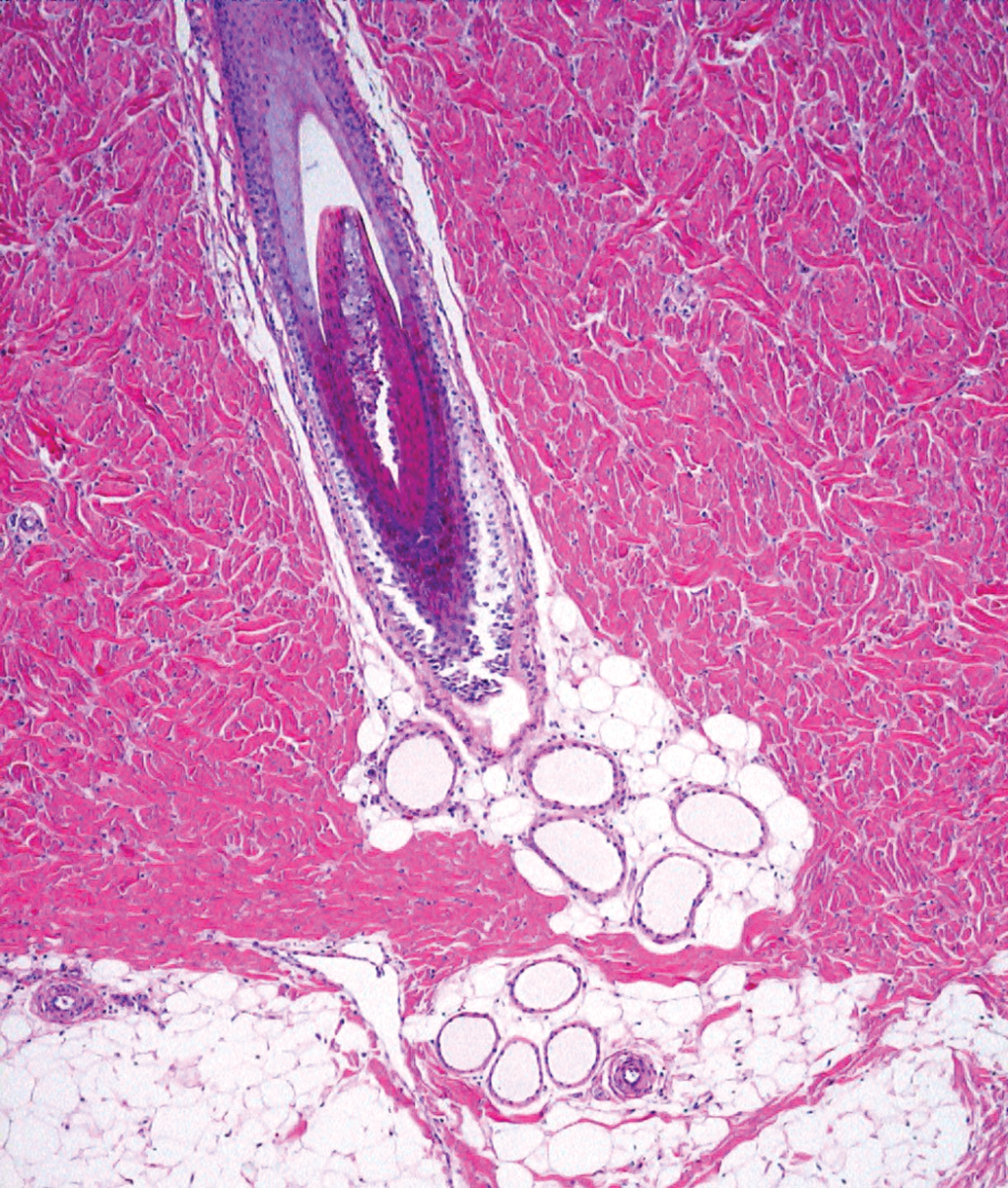
Cross sections of an apocrine gland in adipose tissue adjacent to a hair follicle.
Concluding Remarks
This article describes some of the challenges in performing safety evaluation of dermal products in minipigs and provides guidance for procedures, both in-life and postmortem, to successfully accomplish this evaluation. There is continued need for evaluation of topical products, and it is expected that the minipig will continue to play an important role in product safety assessment. The authors hope that this article will be useful for researchers in this area.
Footnotes
Acknowledgment
The authors like to thank Brian Poirson for designing the application tool for administering viscous formulations. The authors would also like to thank Christina Savidge for designing modifications to the Lomir Jackets that would be fit for purpose use.
Author Contribution
All authors (CW, TR, CA) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
