Abstract
Minipigs have been used for dermal drug development studies for decades, and they are currently more frequently considered as the second nonrodent species for pivotal nonclinical studies, in lieu of the dog or nonhuman primate, for compounds delivered via standard systemic routes of administration. Little is known about the tolerability of different excipients in minipigs; sharing knowledge of excipient tolerability and compositions previously used in nonclinical studies may avoid testing of inadequate formulations, thereby contributing to reduced animal usage. This article reviews vehicles employed in the Göttingen® minipig based on the combined experience from a number of pharmaceutical companies and contract research organizations. The review includes vehicles tolerated for single or multiple dosing by the Göttingen minipig, some of which are not appropriate for administration to other common nonrodent species (e.g., dogs). By presenting these data for dermal, oral, subcutaneous, and intravenous routes of administration, studies to qualify these vehicles in minipigs can be minimized or avoided. Additionally, investigators may more frequently consider using the minipig in place of higher species if the tolerability of a vehicle in the minipig is known.
Introduction
Safety in drug discovery is paramount and must ensure that safety liabilities associated with the primary target, secondary pharmacology, and/or specific chemical features of the development candidate are identified early and subsequently mitigated (Hornberg and Mow 2014). In this context, the choice of the most appropriate animal species to correctly predict the therapeutic index of a drug is paramount, and the minipig is more frequently being used for oral and/or parenteral drug development programs (Ganderup et al. 2012). Administration of pharmaceutical development candidates in preclinical safety studies requires a formulation that is itself well tolerated, while a broader choice of vehicles may be suitable for acute pharmacokinetic studies, including vehicles needed to prepare a single-dose intravenous formulation. Formulations can themselves profoundly impact drug release, absorption, and metabolism, thereby affecting the pharmacodynamic response (Bhattachar et al. 2015). The minipig has demonstrated its value in the past for improved gastrointestinal tolerability of new formulations, especially compared to the dog, as well as a model species in toxicity testing of dermal products since pig skin better models human skin compared to that of animals with fur (Bode et al. 2010).
Information on formulations tolerated by the minipig has not been broadly shared within the scientific community (Ganderup 2012). Only two vehicles are cited on a publically available database (http://www.gadconsulting.com/vehicles.htm, accessed August 20, 2015). While there is a member-only database that lists vehicles for many preclinical species, excipients for minipig studies via all routes of administration are limited in number to 35, representing 4% of the 2013 database entries (http://www.lhasalimited.org/products/vitic-excipients.htm, accessed August 21, 2015).
Scope
The present publication summarizes the Göttingen® minipig experiences of numerous laboratories to address this knowledge gap and place the information into the public domain. The studies were approved by the Institutional Animal Care and Use Committee and were conducted according to local regulations. Formulations that were generally well tolerated are listed along with the duration of treatment and dose volume, when available. All dose volumes were within the good practice guidances put forth by Diehl et al. (2001). Although the Göttingen minipig was used for all the tabulated formulations, they are likely suitable for all swine species, given the similar physiology among minipigs and standard pigs (Bode et al. 2010). Formulations that resulted in adverse clinical signs, histopathologic changes, or other adverse events are listed separately. As a reference for the following sections, the average weight of a mature Göttingen minipig is 35 kg (range: 20–40 kg; Simianer et al. 2010), however, toxicology studies typically use younger animals with weights ranging from approximately 8 to 10 kg at the beginning of the study.
Oral Excipients
Oral excipients utilized by the authors are listed in Table 1. The dose volumes range from 2 to 10 ml/kg, though 5 ml/kg, approximately 60 ml based on a 12-kg animal, is the most frequently administered dose volume. Oral vehicles often included viscosity enhancers and/or suspending agents, using the same excipients frequently used for other species. Oral vehicles were sometimes preserved with the same agents commonly used in commercial formulations. With the exception of Captisol® (27% w/v), there were no oral excipients reported as causing distress to the minipig or resulting in clinical chemistry, urinalysis, or pathologic changes when used for up to 1 year of daily administration.
Oral Excipients.
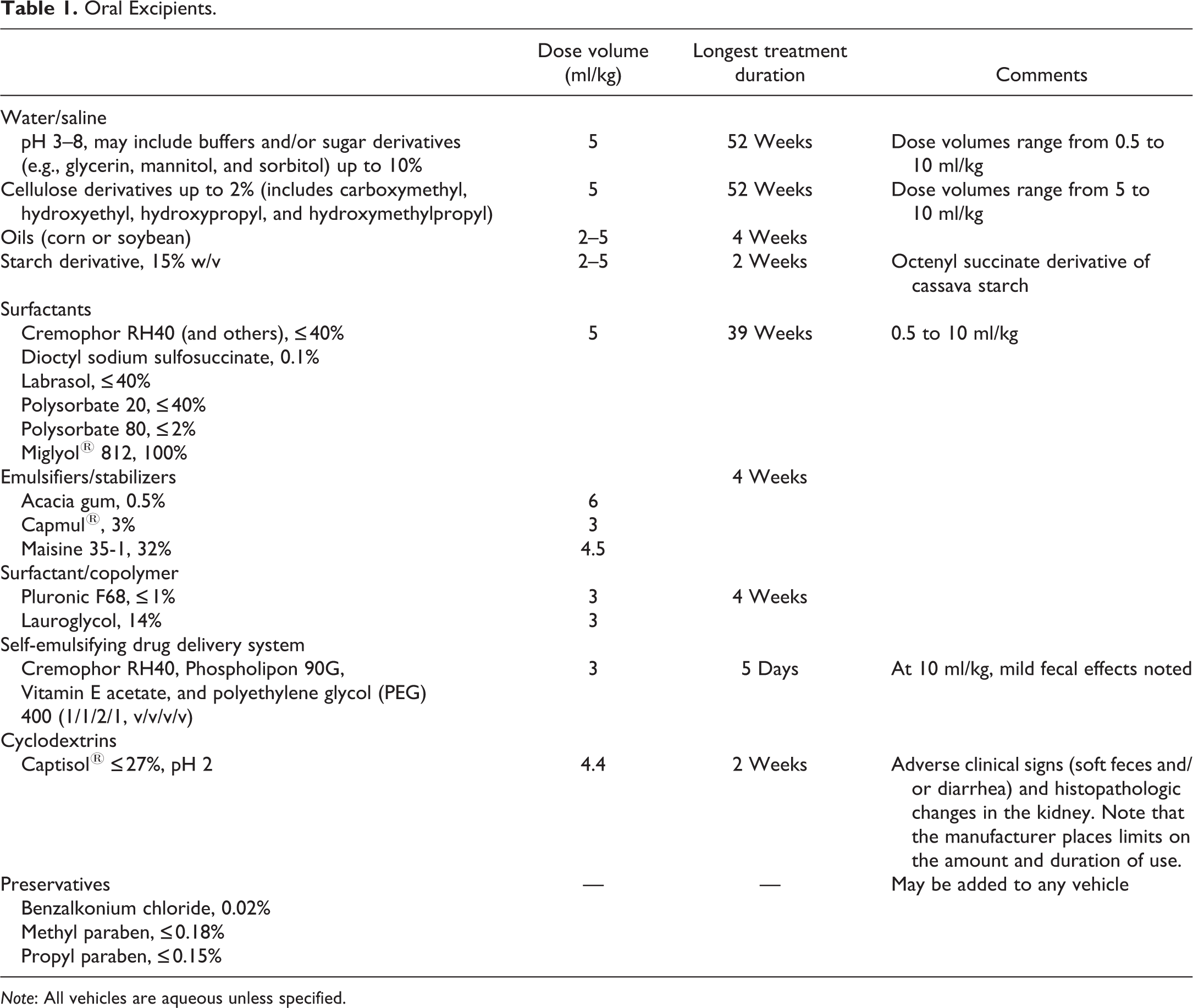
Note: All vehicles are aqueous unless specified.
Parenteral Excipients
Table 2 lists vehicles used for intravenous bolus administration. The dose volumes ranged from 0.33 ml/kg to 10 ml/kg; however, 2.5 ml/kg is considered the best practice. The duration of dosing was shorter than the oral dose experience, as 13 weeks was the longest treatment duration; however, longer treatments may be feasible, given the lack of adverse effects for most excipients, including polysorbate and polyethylene glycol (PEG), which are not well tolerated in dogs (Montaguti, Melloni, and Cavalletti 1994; Gough et al. 1982; Torres-Arraut, Singh, and Pickoff 1984). Cremophor also appears to be better tolerated by the minipig where minimal effects were reported following up to 13 weeks of dosing, while dogs experience anaplylaxis following a single Cremophor administration (Gaudy et al. 1987). At least two vehicles are not routinely used by all institutions, dimethyl sulfoxide (DMSO) and n-methyl-2-pyrrolidone (NMP), since they are not suitable for human formulations. Captisol, a polyanionic beta-cyclodextrin derivative with a sodium sulfonate salt separated from the lipophilic cavity by a sulfobutylether, presents a special case as the conditions of its use, as to the amount and duration of treatment, are governed by independent contractual agreements between the manufacturer and the users. Vacuolation of renal tubular epithelium has been reported and is consistent with the findings reported in the Captisol Drug Master File for other species. Several vehicles are not recommended due to adverse clinical signs or histopathologic changes. It is not possible to confirm the excipient/s responsible for the adverse events with certainty since in addition to one or more individual components that may not be tolerated, the combination of several lipophilic agents may also contribute to adverse events. By a process of elimination, it appears that dimethyl isosorbide (DMI, ≥20%) and vitamin E
Bolus Intravenous Dose Excipients.
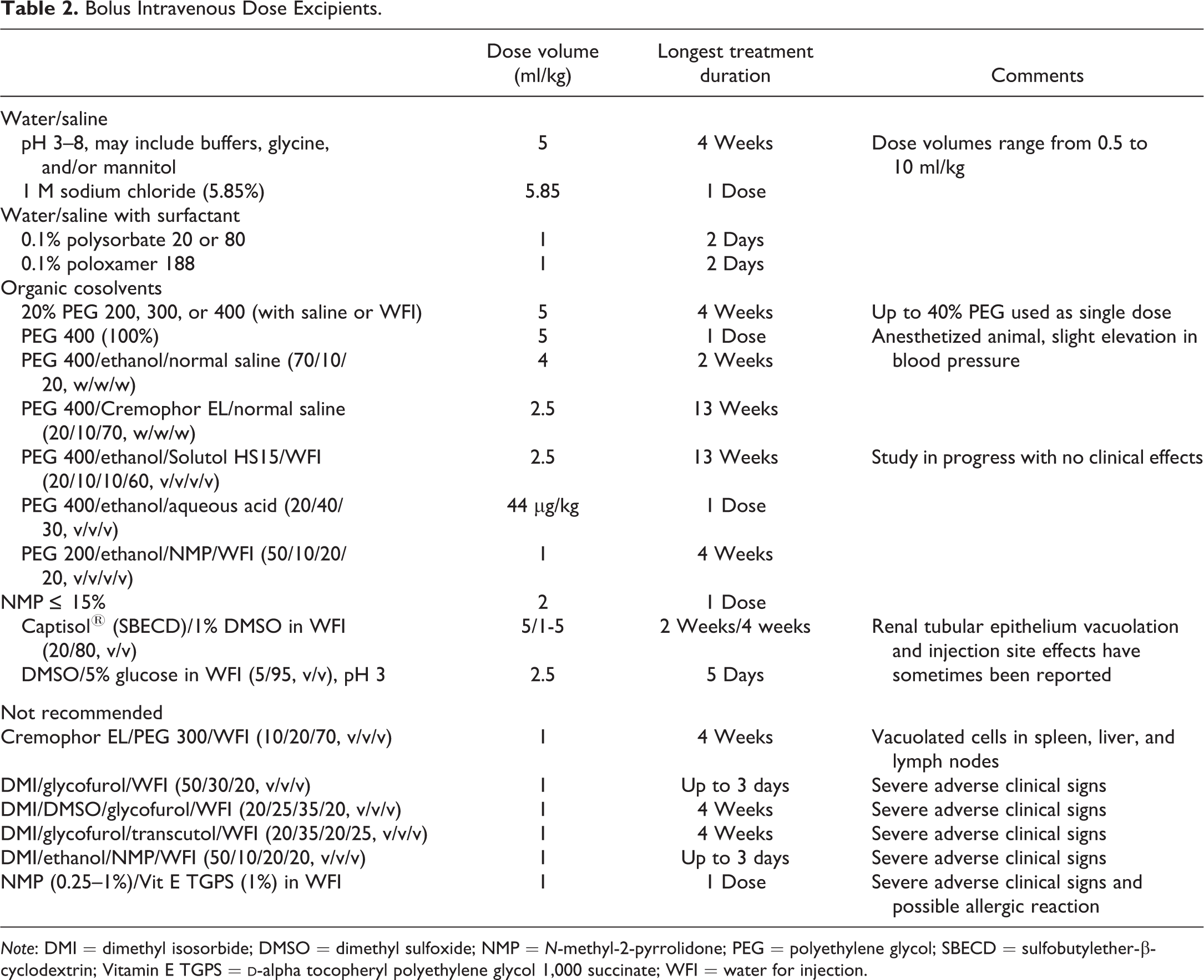
Note: DMI = dimethyl isosorbide; DMSO = dimethyl sulfoxide; NMP = N-methyl-2-pyrrolidone; PEG = polyethylene glycol; SBECD = sulfobutylether-β-cyclodextrin; Vitamin E TGPS =
Intravenous infusion studies in the minipig have been successfully executed by the authors using the vehicles listed in Table 3. There is a large variability in both the infusion duration (5 min to 24 hr) and dosing duration (single dose up to 6 weeks); however, most of the experience was for a week or less. Polysorbate 80 used at a 3% concentration, far above its critical micelle concentration, produced mild, transient itching and anxiety. In the dog, intravenous administration of polysorbates 20 or 80 was associated with acute effects on blood pressure, heart and respiration rates, plasma fibrinogen as well as a marked increase in histamine levels and alterations in complement factors (Moghimi et al. 2004; Marks and Kolman 1971; Qui et al. 2013). Changes to these parameters were identified as pseudoanaphylactic reactions. Submicellular concentrations of 0.1% polysorbate 20 and 80 were tolerated when administered as slow bolus intravenous doses. DMSO and dimethylacetamide are notable excipients not used by all authors due to toxicological concerns (Smith, Hadidian, and Mason 2006; European Chemicals Agency 2014). While these solvents (including PEG and ethanol) can significantly solubilize poorly water-soluble compounds when used as a pure solvent, once the solution is diluted with aqueous media, the poorly water-soluble compound often precipitates en masse (Stella and He 2008). This may cause serious adverse effects, including death, when dosed intravenously.
Intravenous Infusion Dose Excipients.
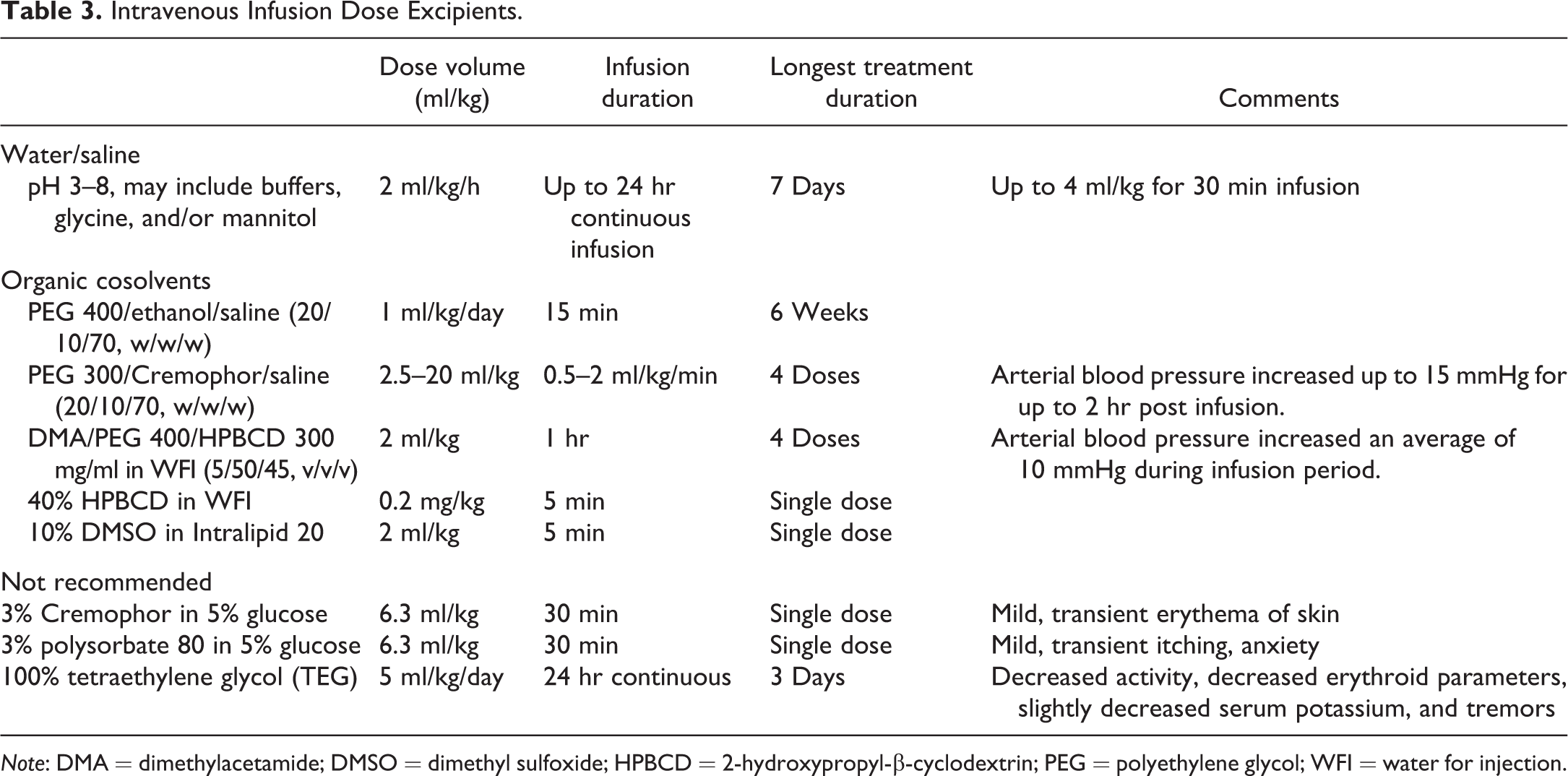
Note: DMA = dimethylacetamide; DMSO = dimethyl sulfoxide; HPBCD = 2-hydroxypropyl-β-cyclodextrin; PEG = polyethylene glycol; WFI = water for injection.
Subcutaneous (SC) studies of up to 26 weeks have been performed in the minipig as either repeated dose toxicity or local tolerance studies using the vehicles cited in Table 4. The dose volumes range from 0.2 to 2 ml/kg. The pig or minipig is often used for SC local tolerance studies due to the similarities in texture and anatomy between human and porcine skin, and the background lesions observed in pig skin were recently reported (Jeppesen and Skydsgaard 2015). Several vehicles were associated with minimal to moderate injection site changes observed at a microscopic level ranging from bruising and inflammatory cell infiltration to ulceration, fibrosis, and necrotic changes. Specifically, olive oil and Solutol HS15 are not recommended as SC vehicles in the minipig. Since injection site changes are often observed in SC dose toxicology studies in other species, it is unlikely the effects described herein would limit the use of any of the other vehicles listed.
Subcutaneous Dose Excipients.
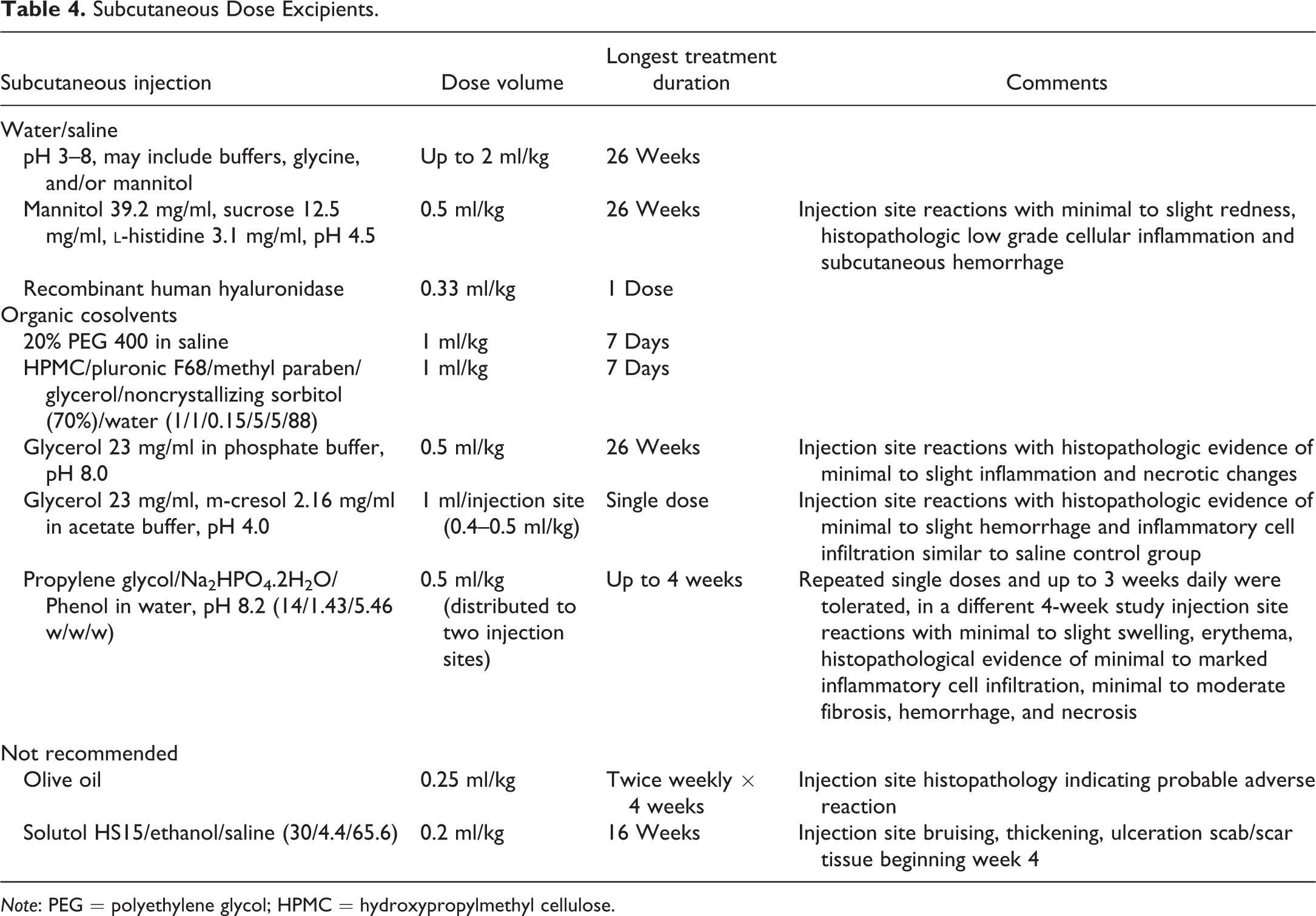
Note: PEG = polyethylene glycol; HPMC = hydroxypropylmethyl cellulose.
Dermal Excipients
The authors have used numerous dermal vehicles on intact minipig skin, in various permutations of excipients approved for use in human formulations, for up to 1 year. There is a large variety of excipients including gels, creams, sprays, and ointments, with or without emollients, surfactants, preservatives, and buffers. Among the reported vehicles, only DMSO-containing formulations yielded adverse reactions and would not be recommended for use in minipig studies. Erythema and edema were observed following a single application of DMSO/ethanol (50/50, v/v), while the skin reaction progressed to include red nodules and ulceration by week 32 following application of DMSO/glycofurol/water (25/25/50, v/v/v). None of the formulations using excipients approved for use in human formulations exhibited more than slight desquamation on treated skin, suggesting there is a wide choice of topical vehicles available for studies in the minipig. Nevertheless, it is recommended that the tolerability of any unknown or untested excipients be evaluated in a limited number of animals before using them in preclinical safety studies.
The minipig has also been used for wound healing studies. In these studies, the skin is incised to create a partial or full thickness wound. Several generally accepted dermal excipients are not compatible with such open wounds. In particular, 0.5% sodium lauryl sulfate used as an excipient yielded excessive oozing that appeared to be serum transudate, not infection, in those wounds when applied for 6 hr. A formulation containing 100% PEG (4% PEG6000 and 96% PEG300) impaired wound healing with daily application. In contrast, formulations containing 20% PEG300, 1% polysorbate 80, and 1% hydroxypropylmethyl cellulose were well tolerated with daily application to wounds for 3 weeks.
Summary
In summary, future studies in the minipig may benefit by the experiences with the numerous vehicles presented herein. By reviewing these excipients, it is hoped that suitable vehicles may be identified and that dedicated studies to confirm vehicle tolerability in the minipig may be avoided. Furthermore, toxicologists and pharmacologists engaged in minipig studies are encouraged to publish their in-house experience with excipients and excipient combinations not available in the public domain to minimize the need for additional tolerability testing and to avoid using nontolerable vehicles.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
