Abstract
The field of dermal toxicity continues to evolve in order to accurately predict dermal (and systemic) responses in humans to topically applied chemicals. Although the testing methods have undergone extensive refinements, idiosyncrasies and unexpected issues during the conduct of these studies are not unusual due to the plethora of new vehicles available for formulating test substances, changing regulatory requirements, and introducting new strain and/or species of laboratory animals as no single species or method seems to suffice for evaluating skin toxicity. The objective of this article is to illustrate some pragmatic issues that should be considered during the conduct as well as interpretation of dermal toxicity studies. Routine procedure-related issues such as hair clipping, tape stripping, and wrapping the animal’s torso to prevent oral ingestion can influence the interpretation. Excipients used in dermal toxicity studies may be nontoxic when used alone but complex dermal formulations can result in unexpected irritation and toxicity. In conclusion, interpretation and risk assessment of dermal toxicity studies should be done in a comprehensive manner, taking into account procedure-related impact on study results, unique species susceptibility, limitation of gross visual (naked eye) observation for evidence of toxicity, and normal anatomical variation.
Keywords
Introduction
The skin is a significant portal of entry of hazardous agents and a vulnerable target organ system. Most repeat-dose dermal toxicity studies (chemicals applied topically) are intended to characterize irritation potential, cutaneous and/or systemic toxicity associated with topical administration of compounds, and results used to predict human response. Dermal toxicity studies in laboratory animals pose certain unique challenges that are not of significant concern in oral or parenteral studies. For example, animal grooming is often associated with ingestion of test compound (voluntarily or involuntarily), which could increase systemic exposure and toxicity. To prevent ingestion and to mimic clinical administration of dermal test formulations, the application site is usually occluded with gauze and the animals may be wrapped with a cotton sleeve to secure the gauze in place. Wrapping of the torso or body too tightly with bandage or a body sleeve has resulted in unintended consequences. Additionally, several variables in dermal studies may affect results and interpretation including but not limited to the vehicles/excipients used, duration of application, application techniques, preparation of test sites, and species differences in dermal response to chemicals. Apart from these well-known specializations of dermal toxicity studies, a few rather unusual but pragmatic approaches that can impact interpretation and outcome of dermal toxicity studies are outlined in this article. In the context of this article, toxicity and irritation studies have been used interchangeably as some dermal studies are conducted purely to characterize the skin irritation of dermal formulations, while other dermal studies are conducted to characterize potential local and systemic toxicity after dermal administration of the test article.
Hair Clipping, Depilation, and Tape Stripping
In nonclinical species, hair removal is a prerequisite procedure to ensure uniform distribution and adequate contact of test article with the epidermis. Electric clippers are generally used, and due to the rapid regrowth, repeated clipping over the course of an extended (1 month or longer administration) period is often necessary. Minimal epidermal hyperplasia, hyperkeratosis, sebaceous gland hypertrophy, and occasionally focal epidermal pustules (perhaps as a result of subclinical trauma; Figure 1) can occur secondary to the clipping procedure, primarily in rodents. Although similar changes should be present in clipped concurrent controls, therefore minimizing the potential for misinterpretation, those evaluating and/or interpreting findings from dermal studies must be aware of these procedural-related changes. In addition to morphological changes, rapid hair regrowth due to clipping (Matsuo, Mori, and Hashimoto 2003) can also impair drug absorption perhaps due to the physical presence of hairs (Foley et al. 2001).
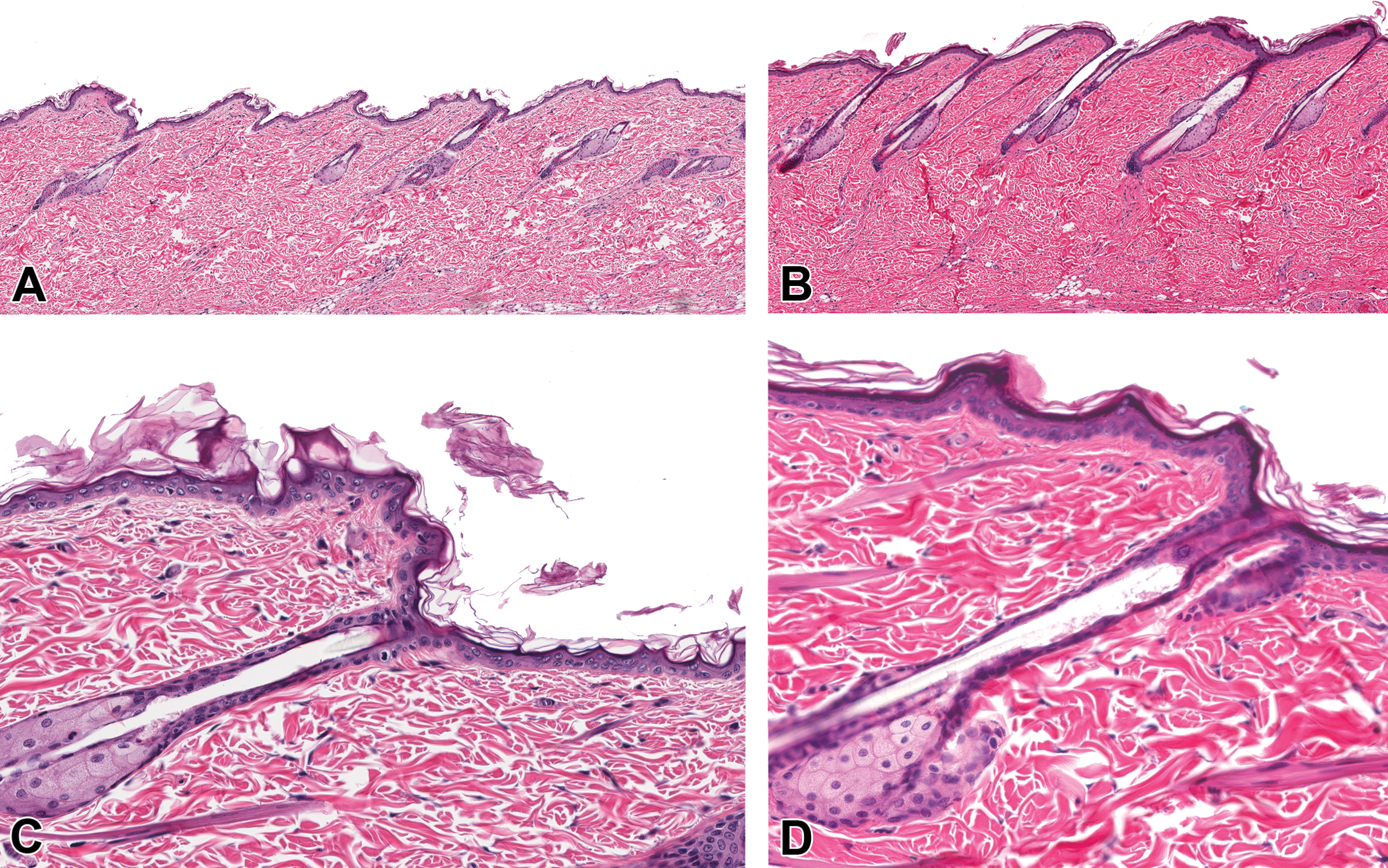
Hair clipping in rats. Minimal epidermal hyperplasia, hyperkeratosis, and hypertrophy of sebaceous glands associated with repeated hair clipping of the dorsal skin in rats (B and D). The naive unclipped skin from the same rat is on the left (A and C). HE.
In addition to electric clippers, hair may be removed using topical application of chemical depilatory agents such as Nair® or Neet®, resulting in a smooth and hairless skin surface. Chemical depilation especially in rats and/or mice with relatively thin epidermis may result in loss or reduction of the stratum corneum, rendering the skin more sensitive to topical agents. It can also result in significantly higher systemic exposure of topically administered compounds compared to clipped skin (Foley et al. 2001), thereby affecting the outcome of a toxicity study. For example, BALB/c mice treated topically with 10% epigallocatechin-3-gallate (EGCG) ointment after chemical depilation had significant toxicity characterized by weight loss (12%), skin ulceration, and acanthosis. However, there was no evidence of toxicity when the same concentration of EGCG was applied to mice when the hair was clipped (Stratton et al. 2000).
Tape stripping, a technique utilized to remove the stratum corneum after application of an adhesive tape is another procedure that is often used to enhance dermal absorption or to remove the epidermal barrier. Hennings and Elgjo (1970) noted a pronounced but transient inflammatory reaction in the skin of hairless mice during the first 2 days after tape stripping returning to normal morphology 6 to 8 days after stripping. Early inflammatory changes associated with tape stripping might have resolved when tissues are examined after the completion of studies longer than a week. A similar procedure in humans, repeated tape stripping (5 times at 24-hr intervals for 10 days) elicited acanthosis, hypergranulosis, parakeratosis, and mixed inflammatory cell infiltrate (Gerritsen et al. 1994). We have observed procedure-related minimal decrease in thickness of the stratum corneum with minimal hypergranulosis (data not shown) in Göttingen minipigs after repeated tape stripping (10 times/day for 6 days) without any overt evidence of inflammatory changes (unpublished data). Similar to tape stripped mice, increased inflammatory cells (due to tape stripping) may be masked by the resident population of mixed inflammatory cells around the superficial dermal vasculature, a normal feature of the Göttingen minipig. In another study, except for procedure-related decrease in stratum corneum, no detectable abnormalities were observed in the skin of Yorkshire pigs 30 min or 14 days after tape stripping 30 times on day 1 (Berrutti, Singer, and McClain 2000). Minimal hypergranulosis observed in our study may be due to repeated tape stripping (6 consecutive days).
Wrapping Procedure
In some repeat-dose dermal toxicity studies, the test article is applied daily and to ensure close contact with the skin and to prevent ingestion and/or self mutilation, the test site is generally semioccluded with gauze and adhesive tape. Occasionally, a full occlusive dressing involving a plastic cover over the test item application site is also done to mimic the clinical protocol. This is followed by application of a stretchable bandage around the middle torso to hold the gauze dressing firmly in place. Wrapping of the torso or body too tightly with bandage or body sleeve can result in unintended consequences. Prolonged thoracoabdominal bandaging of the rats has resulted in hepatic necrosis (Cushman, Richter, and Duke 1989; Nyska et al. 1992; Parker and Gibson 1995; Ramot et al. 2012) and diffuse retinal atrophy (Nyska et al. 1992). This entity, which was termed “wrapping hepatopathy,” was suggested to be analogous to the “corset liver” found in women who wore tight corsets (Popper and Schaffner 1957). It was postulated that the pressure exerted by wrapping leads to systemic vascular congestion that causes centrilobular hepatic degeneration and fibrosis (Nyska et al. 1992).
We have observed hepatic necrosis in rabbit dermal toxicity studies wherein the animals were wrapped for variable (8–23 hr) duration (Figure 2). Hepatic necrosis, observed in both treated and control groups, was acute to subacute, with no zonal distribution, and was occasionally accompanied by correlating increases in serum enzymes (aspartate transaminase/alanine transaminase). The spectrum of changes in individual rabbits ranged from small, discrete necrotic foci surrounded by a rim of vacuolated hepatocytes to large bridging areas with hemorrhage. In some animals, a band of reactive desmoplasia was present around larger lesions. The morphological changes observed in rabbit dermal toxicity studies are most likely due to the wrapping procedure and not due to torsion of the liver lobe, an unrelated condition also reported in rabbits (Wenger et al., 2009; Weisbroth 1975). If extended wrapping procedures (≥6 hr) are required, an additional naive (nonwrapped) control group may help to distinguish between test method and/or test article–related effects if potential hepatic effects are suspected with the test article.
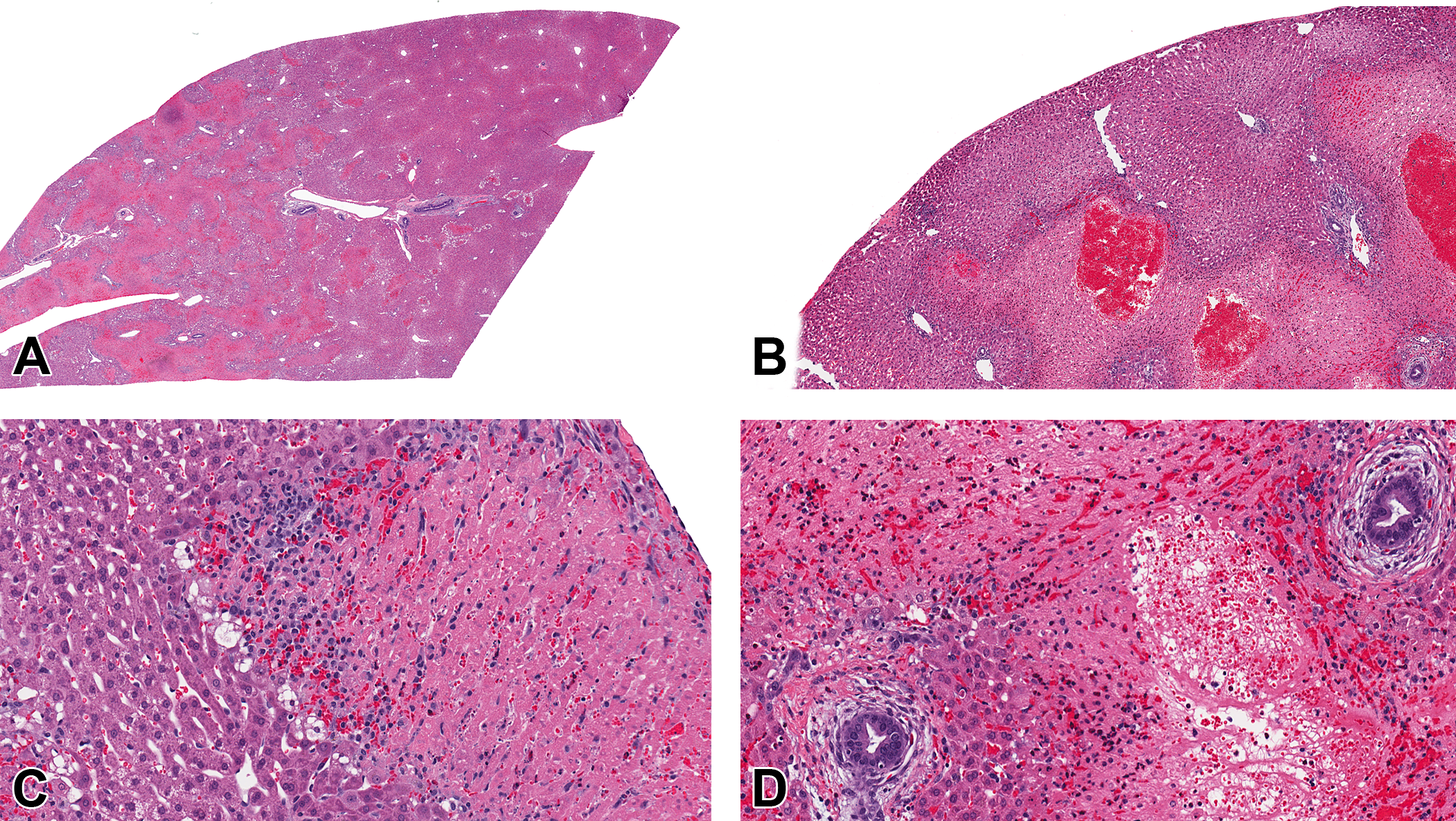
Hepatocellular necrosis in rabbits associated with wrapping the torso. The changes are characterized by bridging coagulative necrosis affecting some portions of a lobe while sparing other regions (A and B). Discrete subcapsular area of necrosis (C) sparing the portal areas (D). HE.
Species and Gender Differences in Dermal Irritation
Variations in toxic responses based on species and gender differences are well known in toxicology after oral or parenteral administration of test articles. Although there are a few reports of gender or species differences in dermal toxicity after systemic administration of compounds (Brown et al. 2008; Shenton et al. 2003), gender and/or species differences in dermal toxicity after topical administration occur infrequently (Dikshith, Raizada, and Srivastava 1990). These gender differences after oral administration of xenobiotics may be either due to differences in systemic exposure or due to increased or decreased susceptibility secondary to different levels of drug metabolizing enzymes among other things. We have observed significant species differences in dermal irritation after topical application of an antibiotic. Topical application of the antibiotic (up to 5% concentration) resulted in dermal irritation in rabbits and rats even at concentrations lower than that used in the clinical formulation (1%). The dermal changes in rats were characterized by epidermal necrosis/ulceration and regenerative hyperplasia and capped by serocellular exudates (Figure 3B and 3D). In contrast, there was no dermal irritation/toxicity in Göttingen minipigs when the antibiotic (at 5% concentration) was applied to abraded skin of Göttingen minipigs for 10 days using the same formulation (Figure 3E and 3F). In addition to species differences, gender differences in dermal toxicity after topical administration of compounds have been observed. Dermal administration of isoproturon for 21 days was more toxic to male rats as evidenced by animal mortality and enzymatic and hematological changes compared to female rats (Dikshith, Raizada, and Srivastava 1990). Conversely, increased sensitivity of female mice to the skin tumor-promoting ability of mirex, a halogenated cycloalkane pesticide has been observed. Since tumor incidence decreased in ovariectomized female mice, ovarian hormones were considered to have a role in tumorigenesis of mirex (Moser, Robinette, and Smart 1993). Differences in systemic exposure to the administered compound could contribute to gender differences in dermal toxicity responses.
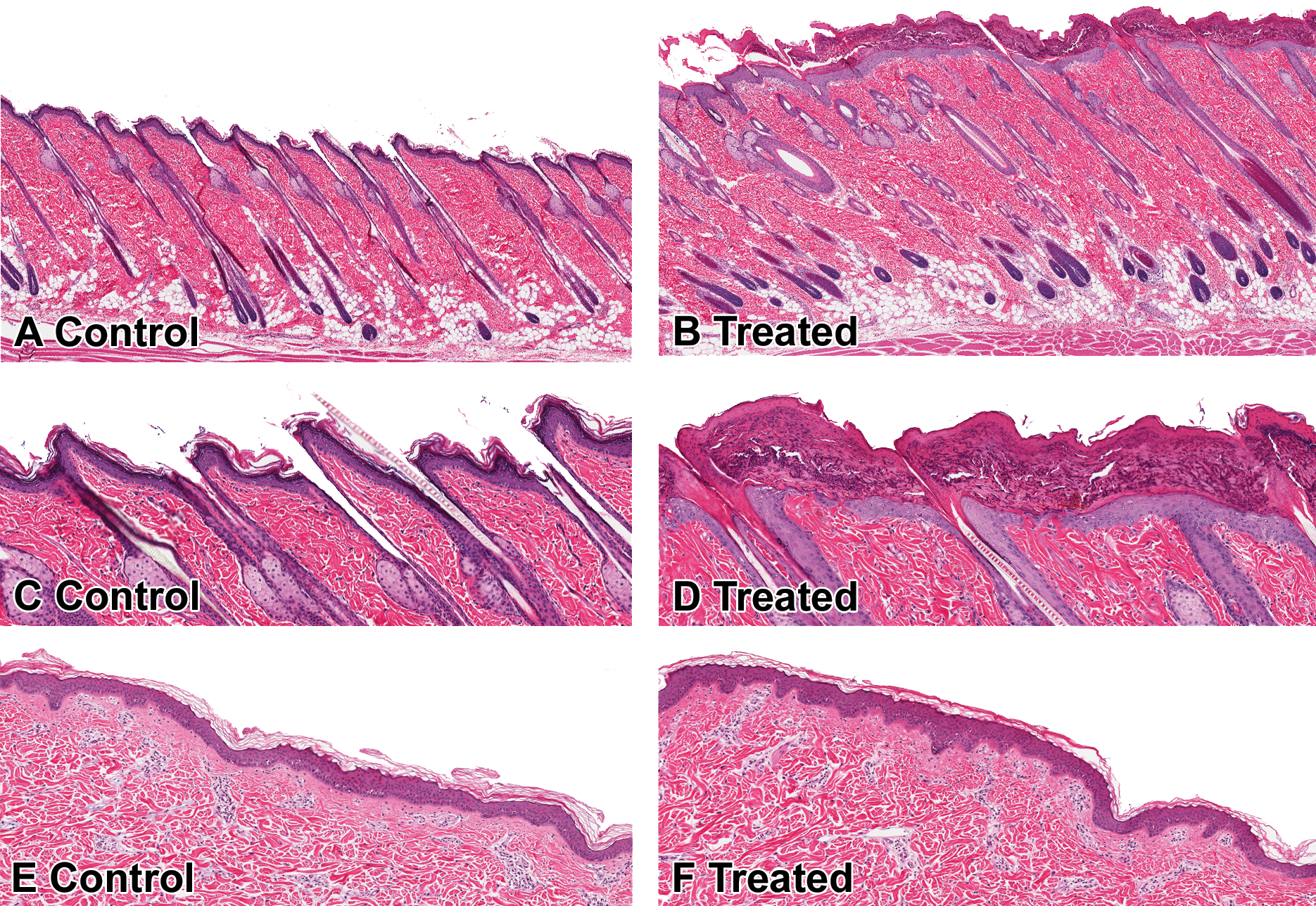
Species differences in dermal response. Compared to vehicle-treated rat (A and C), epidermal hyperplasia and serocellular crusts are present in rats after topical application of an antibiotic (B and D). There is no evidence of skin irritation in minipigs after topical application of the same antibiotic (E control top; F antibiotic treated). HE.
Limitations of Gross Visual Observation
Clinical observations are important in dermal studies. The central tenet of Draize skin irritation testing is largely based on visual inspection of skin surface (to the naked eye) after dermal application of test article (Draize, Woodard, and Calvery 1944; Draize 1959). Briefly, the test material is placed on a specific area of skin (application site) in a conscious animal and let to set for a predetermined amount of time before rinsing it off and recording its effects. Approximately 1 hr after rinsing off, the application site is observed for the presence or absence of erythema and/or edema using a scoring method from 0 to 4 (absent to severe). The animals are observed daily. If the study is repeat dose, the daily erythema and/or edema scores are added to calculate the cumulative irritation score.
Grossly visible irritation scores provoked by different compounds can be different since irritation scores are assessed during the in-life phase over the course of the study (multiple contiguous observations), and a cumulative score for each compound or formulation is calculated at the end of the study. In contrast to a cumulative clinical irritation score, the histologic data to assess irritation are a single static observation since skin is collected at necropsy usually at the end of the study. Although both (in-life irritation scores and histology) end points are important, selecting a compound or a vehicle formulation can be a confounding factor if there is lack of correlation between the in-life observation (Draize score) and the histopathology data. This discrepancy was observed during the conduct of a 7-day dermal irritation study in New Zealand white rabbits using 4 different dermal formulations (formulations A, B, C, and D). Briefly, in this study, the daily erythema and/or edema scores were added to calculate the cumulative 7-day irritation score for each rabbit. The cumulative (day 7) scores were 6, 13, 12, and 27 for formulations A, B, C and D, respectively. However, Histological examination revealed edema, epidermal hyperplasia, hyperkeratosis, and dermal-mixed inflammatory cell infiltrates with identical severity in all rabbits regardless of the irritation score.
Vehicle Toxicity and Irritation
In nonclinical studies, a number of different vehicles, excipients, and/or formulations are employed to administer test article to laboratory animals. The choice of vehicle, excipients, and/or formulation is dependent on the physicochemical properties of the test article, suitability of the formulation for clinical administration, stability, pharmacopoeial acceptability, and so forth. Since species idiosyncrasies often exist, the lack of toxicity and/or irritation in one species does not guarantee safety in a different species. Previously we reported dermal irritation to white petrolatum in New Zealand white rabbits (Chandra et al. 2013), an unexpected finding, considering the innocuous nature of petrolatum and its general use in topical formulations. In another study, striking species susceptibility was observed between rats and hamsters for a topical formulation containing benzyl alcohol and polyethylene glycol 400 (PEG400). Dermal application (50% benzyl alcohol + 50% PEG400) for either 15 or 30 days on the backs of Han-Wistar rats resulted in mild to marked dermal inflammation characterized by multifocal infiltrates of lymphocytes, macrophages, and occasionally giant cells around adnexal structures (Figure 4A), with no evidence of skin changes in clinical examination during the in-life phase of the study. In contrast, there was no evidence of irritation (clinically or histologically) in hamsters after application of an identical formulation (50% benzyl alcohol + 50% PEG400) to the pinnae of Syrian hamsters (Figure 4B). Incidentally, in the same hamster study, dermal application of a topical formulation (25% oleic acid + 25% benzyl alcohol + 25% propylene carbonate + 25% PEG400) to Syrian Hamsters resulted in marked dermal irritation characterized by severe discoloration, thickening of the skin, epidermal hyperplasia, hyperkeratosis, and sebaceous gland hypertrophy in the pinna (Figure 4C). Although it is difficult to isolate a single component as an irritant, based on the published literature oleic acid in the formulation may be responsible (Touitou et al. 2002). Excipients used in dermal toxicity studies may be innocuous when used alone but complex mixtures of individual excipients can result in significant toxicity (Brazeau and Fung 1989a, 1989b). Potential contamination of the formulation or degradation of excipients resulting in irritation is a possibility. Fungal contamination of a vehicle formulation resulting in unexpected skin lesions has been reported in rabbits (Morgan et al. 1985).
Vehicle (dermal formulation)-associated toxicity. Chronic periadnexal inflammation in rats after topical application of a formulation containing benzyl alcohol and PEG400 (A). Lymphohistiocytic inflammatory cell infiltrates around hair follicles (inset). No evidence of any irritation in the pinna of hamsters administered the same (benzyl alcohol and PEG400) formulation (B). Epidermal hyperplasia, hyperkeratosis, and sebaceous gland hypertrophy in the pinna of hamsters administered vehicle containing oleic acid, benzyl alcohol, polypropylene carbonate, and PEG400 (C). HE. PEG400, polyethylene glycol 400.
Normal Anatomical Variation
It is beyond the scope of this article to cover species and/or gender differences in cutaneous morphology; however, a few normal anatomic features that may influence interpretation of dermal toxicity studies are highlighted.
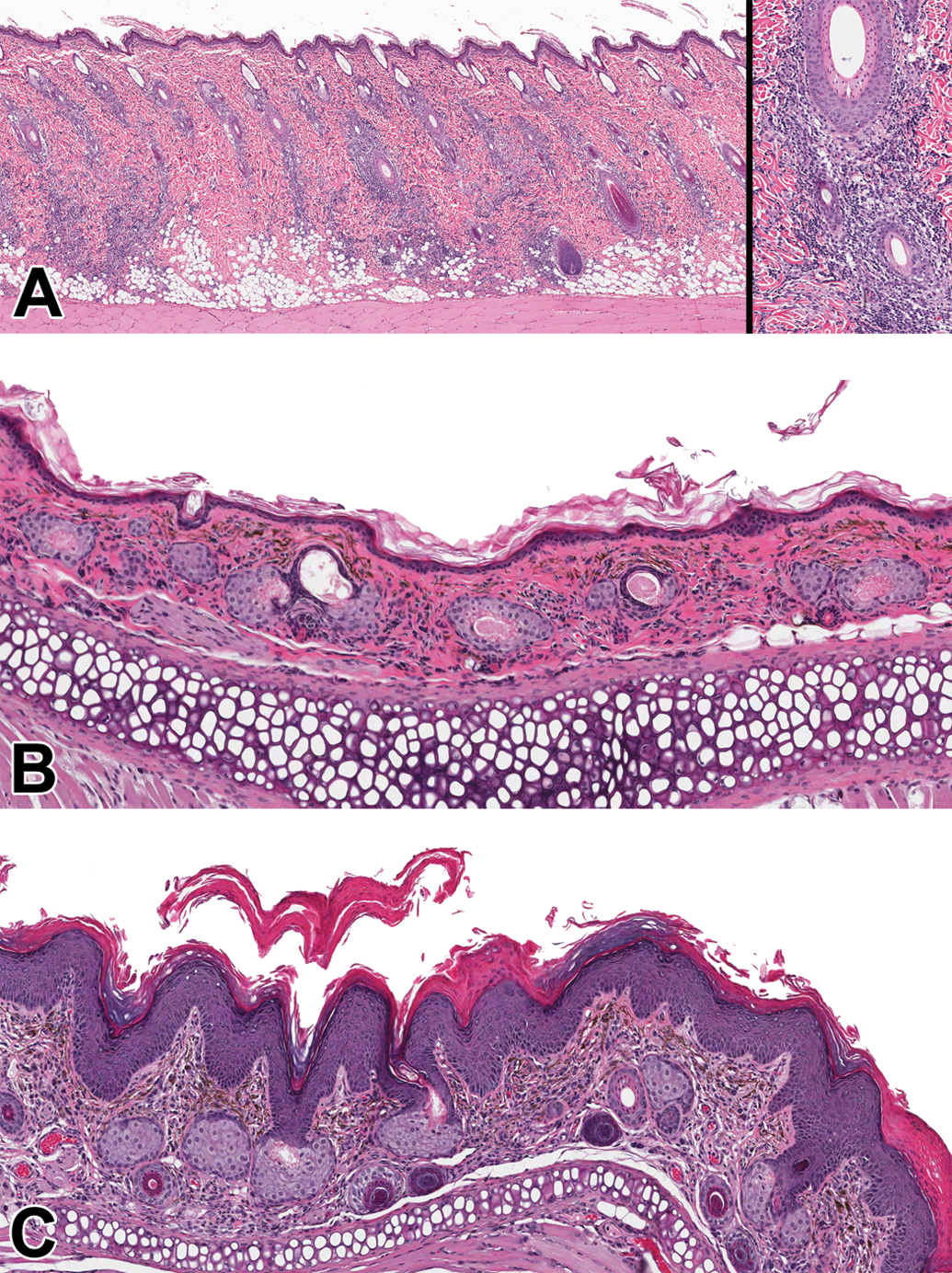
Occasionally, dermal toxicity studies are conducted in pigmented mouse strains such as C57BL/6J mice or Long-Evans rats. The truncal pigmentation of rodents is entirely dependent on their follicular melanocytes. Pigment production is active only during the follicle growth (anagen) phase (Park et al. 2007; Sundberg et al. 2010; Sundberg and Silva 2012). In pigmented mice, rapid hair regrowth after clipping is grossly evident as dark gray/black discoloration of the skin (Figure 5) due to hair follicles entering the anagen phase. The latter observation is also evident on the underside (skin is flipped) when skin is excised and collected at necropsy. Although both control and test article–treated animals will generally have these dark areas of discoloration secondary to the clipping procedure, lack of similar findings in control animals should not be interpreted as a test article–related effect.
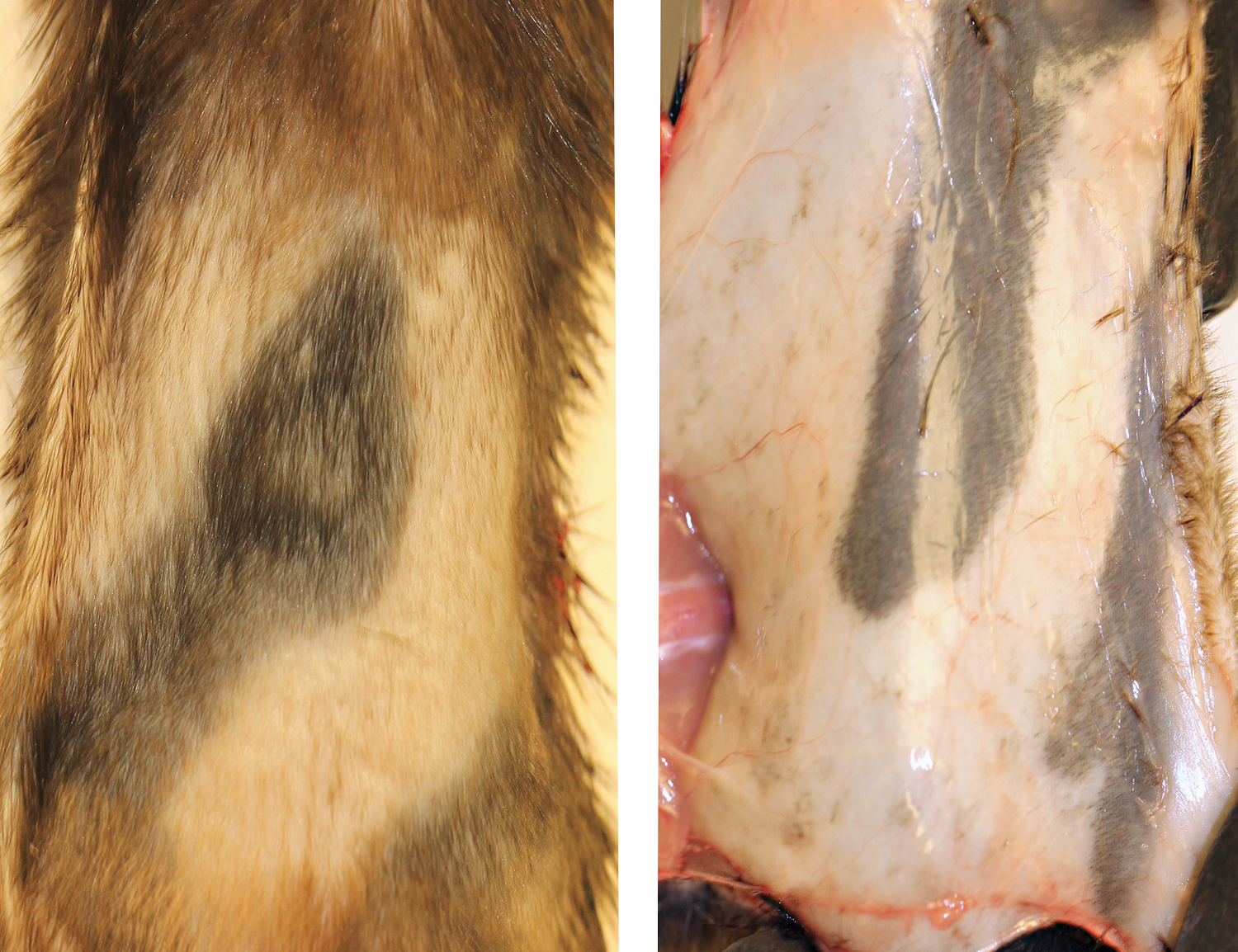
Rapid hair regrowth after clipping in pigmented strains of mice is grossly evident as dark gray/black discoloration of the skin due to hair follicles entering the anagen phase. The latter observation is also evident on the underside (skin is flipped).
Likewise, the Göttingen minipig is a commonly used strain for dermal toxicity studies. Normal biological variability in the skin of Göttingen minipigs includes larger sebaceous glands in the neck (compared to thoracolumbar skin), aggregates of fungal yeast (Malassezia sp.) in the stratum corneum, variable morphology of apocrine sweat glands (cuboidal to tall columnar epithelium), and site differences in dermal fat thickness. Although melanin or melanocytes are not evident in the routine HE-stained skin sections (personal observations), the Göttingen minipig is not an albino (Forster et al. 2010). The retina of Göttingen minipigs is pigmented.
Concluding Remarks
It has been more than 7 decades since the publication of the Draize scoring system (Draize, Woodard, and Calvery 1944), but the field of dermal toxicity continues to evolve in order to accurately predict dermal (and systemic) responses in humans to topically applied industrial chemicals or pharmaceuticals. Although the testing methods have undergone extensive refinements, idiosyncrasies and unexpected issues during the conduct of these studies are not unusual due to the plethora of new vehicles available for formulating test substances, introduction of new strain, and/or species of lab animals as no single species or method seems to suffice for evaluating skin irritants.
The objective of this article was to illustrate some pragmatic issues that should be considered during the conduct as well as interpretation of dermal toxicity/irritation studies. Routine procedure-related issues such as hair clipping, tape stripping, and wrapping the animal’s torso to prevent oral ingestion can influence the interpretation. Dermal toxicity studies are conducted in densely haired animals (rodents/rabbits) with a relatively thin epidermis compared to humans. In general, human and nonhuman primate skin was found to be much less permeable to test substances when compared to rabbit and/or rodent skin (Calabrese 1984; Sidon, Moody, and Franklin 1988; Scott et al. 1991). Further, risk assessment using the Draize test in rabbits has been criticized as the results obtained in rabbits are not always predictive of the human response as the rabbit skin is more readily irritated by chemical agents than human skin (Calvin 1992; Philips et al. 1972; Nixon et al. 1975; Jírová et al. 2010; York et al. 1996; Steinberg et al. 1975). In conclusion, interpretation and risk assessment of dermal toxicity studies should be done in a comprehensive manner, taking into account procedure-related impact on study results, unique species susceptibility, limitation of gross visual (naked eye) observation for evidence of toxicity, and normal anatomical variation.
Footnotes
Acknowledgments
The authors would like to thank Ms. Joanna Barton, Ms. Elizabeth McNeil, and Mr. Kerry Crabb for help with the histological preparations of skin samples.
Author Contributions
Authors contributed to conception or design (DM, KD, SF, RP, KM, RA), acquisition (DM, RP, KM), analysis (DM, RP, KM), interpretation (KD, SF, RP, RA), and critically revised the article (DM, KD, SF, RP, KM, RA). All authors gave final approval, and agree to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Authors’ Note
This study was conducted at GSK in accordance with the GSK Policy on the Care, Welfare and Treatment of Laboratory Animals and was reviewed by the institutional animal care and use committee at GSK.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
