Abstract
The porcine immune system has been studied especially with regard to infectious diseases of the domestic pig, highlighting the economic importance of the pig in agriculture. Recently, in particular, minipigs have received attention as alternative species to dogs or nonhuman primates in drug safety evaluations. The increasing number of new drug targets investigated to modulate immunological pathways has triggered renewed interest to further explore the porcine immune system. Comparative immunological studies of minipigs with other species broaden the translational models investigated in drug safety evaluations. The porcine immune system overall seems functionally similar to other mammalian species, but there are some anatomical, immunophenotypical, and functional differences. Here, we briefly review current knowledge of the innate and adaptive immune system in pigs and minipigs. In conclusion, more systematic and cross-species comparisons are needed to assess the significance of immunological findings in minipigs in the context of translational safety sciences.
Keywords
Introduction
The use of model organisms is a cornerstone of efficacy and safety studies in drug discovery and development. Historically, rodents, dogs, and nonhuman primates have been the species of choice for drug toxicity testing (P. J. Haley 2003; Helke and Swindle 2013). Ideally, drug safety studies with small molecules or biotherapeutics should be based on pharmacological responsiveness and be question driven, possibly including additional customized (immunological) parameters beyond established safety assessments. Based on anatomical and physiological similarities with humans, domestic pigs (
How much have we advanced in assessing immunocompetence in pigs and minipigs in the context of translational safety sciences? As important domestic and agricultural species, properties of the porcine immune system have been studied for many years, in particular from the perspective of vaccine immunology (McCullough and Summerfield 2009; Wilson and Obradovic 2015) and host resistance to (human) infections (Meurens et al. 2012; Talker et al. 2015). Investigations of immunophenotypical and functional commonalities of the porcine immune system in comparison to other species including man are a field of increasing attention (Saalmüller et al. 1987; Yang and Parkhouse 1996; Hein and Griebel 2002; Piriou-Guzylack and Salmon 2008; Dawson 2012; Dawson et al. 2006; Mach et al. 2013; Mair et al. 2014; Montoya and Merens 2015; Holderness et al. 2013; Summerfield, Auray, and Ricklin 2015; Summerfield, Meurens, and Ricklin 2015). A number of publications have reviewed separate aspects of porcine immunology (Gerner et al. 2015; Mair et al. 2014; Summerfield, Auray, and Ricklin 2015; Summerfield, Meurens, and Ricklin 2015; Wilson and Obradovic 2015). In this brief overview, we review recent advances of cross-species characterization of immunocompetence in pigs and minipigs relating to translational safety sciences, and point to various open questions, requiring further investigation.
Comparative Anatomy of the Immune System
Overall, immune organs and functions of the pig are organized similarly to other mammalian species. Pigs and humans are both omnivores, which may have contributed to anatomical and physiological similarities of for instance the intestinal tracts (P. Haley 2012). With basic constituents of the mucosal associated lymphoid tissue in different anatomical locations being similar across species, it is important to note some differences of form and function in particular of the gut associated lymphoid tissue (P. Haley 2012). The porcine small intestine differs in the length of its segments and in the branching of mesenteric vessels, and there is a continuous ileal Peyer’s patch (Brandtzaeg et al. 2008; Rothkötter, 2009).
Furthermore, in pigs, lymph nodes are histologically unique in having an inversion of the cortex and medulla, differing from humans. The tissue lacks a larger medullary area and is preferentially composed of cortical areas and paracortex (P. Haley 2012; Rothkötter, 2009). Different from other species, the preferred migration route of lymphocytes out of the lymph node is via the high endothelial venules. The low numbers of lymphoid cells in the thoracic duct of pigs is a consequence of the migration pattern (Rothkötter, 2009). Despite these anatomical differences, the overall function of the lymph node does not seem to differ between species.
Circulating white blood cell composition in pigs is very similar to humans (P. J. Haley 2003; P. Haley 2012), and the overall similarity in numbers and function makes extrapolation of pig leukocyte responses to humans appropriate. Similarly to humans and dogs, and differing from rodents and cynomolgus monkeys (P. J. Haley 2003; P. Haley 2012), pigs have high percentages of neutrophils in the peripheral blood (50–70%). Like humans, minipigs are susceptible to bone marrow depletion with cytotoxic drugs and xenobiotics that induce impaired erythropoiesis and granulopoiesis. Although there are many similarities with other mammals, in the context of translational immunosafety assessments, several pig-specific immunobiological characteristics need to be highlighted and require further comparative immunobiological research.
Comparative Immunobiology
Much of our current understanding of immunobiology has come from studies in mice and man. Although mice are used as models to study human immune diseases and pathophysiology, numerous differences between mouse and human immune systems have been described (e.g., balance of leukocyte subsets, antimicrobial peptides, Toll-like receptors [TLR], natural killer [NK] inhibitory receptor families, Fc receptors, immunoglobulin subsets, B cell and T cell signaling pathway components, γδ T cells, cytokines and cytokine receptors, and T helper 1/T helper 2 [Th1/Th2] differentiation) (Mestas and Hughes 2004). The porcine immune system has been studied to elucidate the role of the lymphoid organs during infectious diseases to understand the immune function in transplantation and vaccine research and to generate models for various aspects of human immunology. Availability of immunophenotyping reagents has allowed for delineation of pig lymphoid subsets and their respective functions and has revealed a number of peculiar differences to other mammals (Piriou-Guzylack and Salmon 2008; P. Haley 2012; Gerner et al. 2015; Mair et al. 2014; Summerfield et al. 2015a, 2015b; Wilson and Obradovic 2015). A few are briefly mentioned here (Mair et al. 2014): porcine TLR 7 and TLR 9 expression is restricted to plasmacytoid dendritic cells (pDC) and not expressed on classical DC (cDC) as in mice, greater diversity of interferon type I genes (39 genes vs. 19 in humans), no α-defensin but more cathelicidin genes in pigs (11) than in humans (1) (Sang and Blecha 2009), and a very high frequency of γδ T cells in pigs. Table 1 compares major blood lymphocyte and monocyte subsets (mean percentages) between different species.
Cross-species Comparison of Major Blood Lymphocyte and Monocyte Subsets (Mean Percentages).
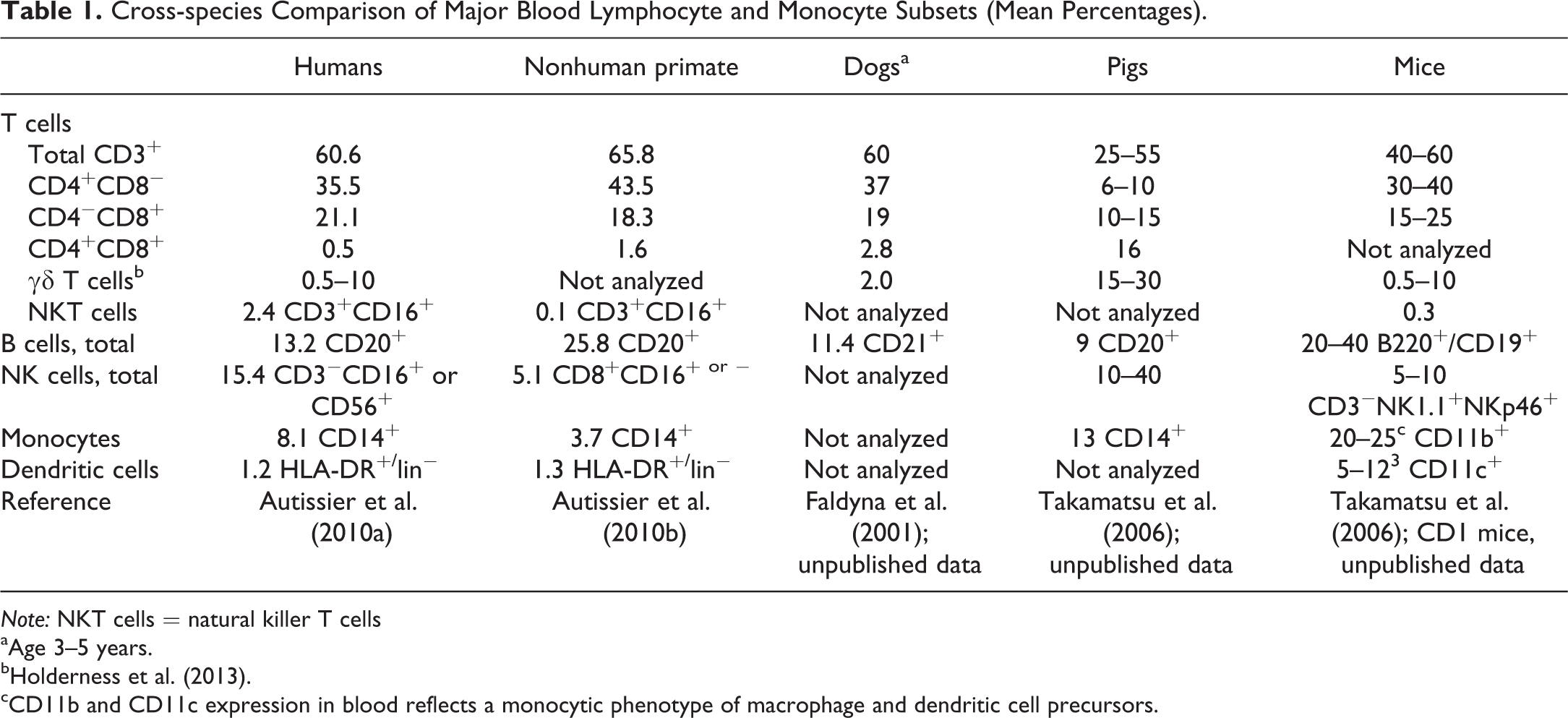
aAge 3–5 years.
cCD11b and CD11c expression in blood reflects a monocytic phenotype of macrophage and dendritic cell precursors.
Current knowledge of important cell populations at the interface of the porcine innate and adaptive immune system such as monocytes–macrophages, DC, and NK cells have been reviewed (Gordon and Taylor 2003; Gerner, Käser, and Saalmüller 2009; Summerfield, Auray, and Ricklin 2015). In view of the complexity of monocytes–macrophage and DC subset identity based on a combination of cell surface phenotyping, expression analysis of hallmark genes, and robust functional assays (Vu Manh, Bertho, et al. 2015; Vu Manh, Elhmouzi-Younes, et al. 2015; Guilliams et al. 2014, 2015), the current classification systems should be seen as work in progress.
In humans, three blood monocyte subsets are distinguished based on expression of human leukocyte antigen DR allele group (HLA-DR), CD14, and/or CD16 (total 9.3%): these include “classical” (CD14+CD16−, 8.1%), “intermediate” (CD14+CD16+, 0.6%), and “nonclassical” monocytes (CD14dimCD16+, 0.4%; Autissier et al. 2010a; Ziegler-Heitbrock et al. 2010; Guilliams et al. 2014). The same nomenclature as proposed for man can be used in other species such as pigs (reviewed by Ziegler-Heitbrock, 2014). Monocytes are functionally characterized by the ability to phagocytize, to produce cytokines, and to present antigen and, under conditions of severe infection, the number of nonclassical and intermediate monocytes may increase (Ziegler-Heitbrock, 2014). Transcriptomic analyses indicate that CD14dimCD16+ human monocytes are the counterparts of LY6Clow nonclassical mouse monocytes and that CD14+CD16− human monocytes are the counterparts of LY6C+ classical mouse monocytes (Guilliams et al. 2014; Ingersoll et al. 2010). A characteristically low level of cell surface expression has been described for chemokine receptor 2 (CCR2) and/or L-selectin (CD62L) in nonclassical monocytes of man, mouse, rat, and pig supporting the homology across species (Ziegler-Heitbrock, 2014). Comparative analyses of monocyte biology between humans, mice, and pigs revealed more concordance of pigs and humans in this regard and supports the relevance of the pig as a model for understanding human monocyte function (Fairbairn et al. 2011, 2013). However, the markers that distinguish mouse and human monocyte subsets cannot readily be applied to the pig (Fairbairn et al. 2013). Studies on monocyte subsets in the pig have utilized the differential expression of CD163 because of the role of CD163 in cellular entry into macrophages of the important pathogen porcine reproductive and respiratory syndrome virus (Chamorro et al. 2000; Fairbairn et al. 2013). CD163+ cells accounted for 5 to 50% of all monocytes, and these cells show slightly increased CD16 and strongly increased major histocompatibility complex (MHC)-class II expression. The CCR2 is low to absent in the CD163+ monocytes, which suggests that CD163+ monocytes are homologous to the CD16+ monocytes in humans (Ziegler-Heitbrock 2014). Moreover, it should be noted that there is a significant interbreed variation in monocyte function in pigs that could be important in innate immunity (Ait-Ali et al. 2007). Fairbairn et al. (2013) showed that among several breeds, CD14 expression varied across a substantial range and varied inversely with CD16. The magnitude of this differential expression was nowhere near as large as in humans, nor was there any population that was genuinely CD14low. With some breeds, for example, large white landrace cross, there appeared to be more distinct populations, whereas other breeds, for example, Pietrain, had more of a continuum of cells with the extremes of the population expressing different levels of CD14 and CD16. In contrast to humans, where the CD16+ population is a minor blood subpopulation, these cells in pigs represented around half of the CD14+ monocytes. In each of the breeds, there was also variable expression of CD163 on the CD14+ monocytes. In cross-bred animals (large white × landrace), CD163 expression divided the monocyte pool clearly in 2 with CD163, like CD16, being inversely related to the level of CD14. In others, for example, the Pietrain, there was no clear separation of a CD14highCD163low population. CD14+ monocytes in all breeds were also uniformly strongly positive for CD172a (signal-regulatory protein alpha [SIRPα]), and there was also no obvious variation in the expression of CD172a among the breeds or individual animals (Fairbairn et al. 2013). Engagement of the inhibitory receptor SIRPα by CD47—present on most cells including erythrocytes and platelets—provides a downregulatory signal that inhibits host cell phagocytosis (Barclay et al. 2014). Overall, there are important differences in monocyte subsets which preclude direct extrapolation, which is of particular importance when preclinical animal studies are used to inform clinical studies in humans (Ziegler-Heitbrock, 2014).
Although remarkable similarities of porcine and human skin structures have been known for a long time, skin immunology and its key players such as dermal DC (DDC)—which play a central role as antigen presenting cells (APC)—are just beginning to be explored in detail in porcine skin (Summerfield, Auray, and Ricklin 2015; Summerfield, Meurens, and Ricklin 2015). MHC class II-positive cells with morphology and skin density similar to APC in man were described in miniature pig skin (Grabbe et al. 1994). These cells were able to both stimulate allogeneic T cell proliferation and trigger a delayed type hypersensitivity response (Grabbe et al. 1994). Transcriptomic meta-analysis established strong homologies between pig DC subset candidates and their putative human and mouse equivalents for pDC, cDC1, and monocyte-derived DC (Vu Manh, Elhmouzi-Younes, et al. 2015). Another recent study investigating porcine skin showed similarities between swine DC subsets and human counterparts (Marquet et al. 2014) and proposed to include porcine skin DC populations into a unified mammalian DC classification (Guilliams et al. 2014). Using comparative transcriptomic analysis, phenotyping as well as
Another recent example showing similarity between human and porcine skin focused on interleukin-33 (IL-33), a novel IL-1 family member, in keratinocytes (Sundnes et al. 2015). In murine epidermis, IL-33 behaved similar to an alarmin, being constitutively expressed in keratinocyte nuclei and rapidly lost during acute inflammation, whereas, human and porcine IL-33 were weakly expressed or absent in keratinocytes of noninflamed skin but induced during acute inflammation (Sundnes et al. 2015). In man, pig, and several other mammals, IL-33 is also constitutively expressed at high levels in the vascular endothelium, whereas it is practically absent from murine blood vessels (Sundnes et al. 2015). IL-33 can be expressed by a variety of barrier tissues, and it signals via the receptor ST2, which is highly expressed on group 2 innate lymphoid cells (ILC2) and Th2 cells (Molofsky, Savage, and Locksley 2015). ILC2 polarize CD4+ T cell responses toward Th2 immunity directly by presenting cognate antigens and secreting Th2-inducing cytokines (von Burg, Turchinovich, and Finke 2015). The receptor ST2 is also expressed on inducible regulatory T (iTreg) cells (Schiering et al. 2014) and although much needs to be learned regarding IL-33 and ILC2 biology, it is clearly associated with helminth infections and allergic responses and plays a role in tissue homeostasis and repair (Molofsky, Savage, and Locksley 2015).
The majority of circulating lymphocyte subsets in humans, nonhuman primates, dogs, mice, but not pigs, are αβ CD3+ T cells, expressing either CD4 or CD8, with γδ T cells representing only a minor fraction in the spleen, lymph nodes, and peripheral blood (approximately 0.5–10% in blood; Autissier et al. 2010a; Sedlak et al. 2014). Table 1 compares major blood lymphocyte and monocyte subsets (mean percentages) between different species and highlights that pigs possess a substantial proportion of γδ T cells in the circulation (15–30%; Gerner, Käser, and Saalmüller 2009; Gerner et al. 2015; P. Haley 2012; Holderness et al. 2013). Absolute numbers of γδ T cells increased from birth until 19 to 25 weeks of age, indicating an important role for these cells during adolescence (Talker et al. 2013). This population declines with increasing age (Gerner, Käser, and Saalmüller 2009; Gerner et al. 2015). Combined with a high proportion of NK cells, circulating porcine lymphocytes may have a much more important role in systemic innate immunity, than in humans and mice (Takamatsu et al. 2006), where γδ T cells show differential tissue tropism for epithelial surfaces (Mestas and Hughes 2004). It is not known why the unique γδ TCR ability to rapidly recognize and respond to unconventional antigens, including nonprocessed ligands in the absence of MHC presentation, seems to be of more importance in pigs and some other domestic animals as compared to humans, nonhuman primates, dogs and rodents (Holderness et al. 2013). Recognition of ligands associated with either stress or infection appear to be common features of human and mouse γδ T cells, but the precise ligands for γδ TCR recognition in domestic animals such as pigs are still poorly described (Holderness et al. 2013). Whereas human γδ T cells produce interferon-γ (IFNγ) and tumor necrosis factor-α (TNFα), as well as cytotoxic granules, for example to kill
The current understanding of porcine αβ T cell phenotypes and function in comparison with human and mouse have recently been reviewed (Gerner et al. 2015). The development of porcine αβ T cells in the thymus appears to be similar to other species, starting from a CD4−CD8− stage via a CD4+CD8+ double positive stage to mature CD4+ and CD8+ single positive stages. However, analyses of porcine T cells in blood revealed that a substantial proportion of CD4+ porcine T cells coexpress CD8α (Saalmüller et al. 1987; Gerner et al. 2015). CD8α expression on CD4+ T cell is related to antigen contact and is expressed on activated and memory CD4+ T cells (Gerner et al. 2015). Furthermore, it was shown that CD27 expression could discriminate porcine T helper cells with functionally distinct properties (Reutner et al. 2013). Naive CD4+CD8α− T cells, which reside predominantly in various lymph nodes, express CD27, whereas CD4+CD8α+ T cells separate into a positive and negative subset. Experiments with sorted CD8α−CD27+, CD8α+CD27+, and CD8α+CD27− T helper cell subsets following polyclonal stimulation revealed the lowest proliferative response but the highest ability for IFNγ and TNFα production in the CD8α+CD27− subset. Therefore, these cells resembled terminally differentiated effector memory cells as described in human. In support of this, analysis of CCR7 and CD62L expression showed a high degree of congruence with CD27 expression. A high proliferative capacity and IL-2 production in the CD4+CD8α+CD27+ subset was reminiscent of central memory T cells (Reutner et al. 2013; Gerner et al. 2015). Magnitude and kinetics of multifunctional CD4+ and CD8β+ T cells have been investigated in pigs infected with swine influenza A virus (Talker et al. 2015). IFNγ+TNFα+IL-2+ multifunctional CD4+ T cells were observed in the blood of all animals at 1 or 2 weeks after primary infection and their number increased in most animals after homologous secondary infection. These cells produced higher amounts of cytokines (IFNy, TNFx, and IL-2) than did CD4+ T cells. The vast majority of cytokine-producing CD4+ T cells expressed CD8α (a marker of activation and memory formation). Based on CD27 expression, the swine influenza A virus-specific CD4+ T cells were composed of both central memory and effector memory populations (Talker et al. 2015).
Treg cells exert their immunosuppressive functions by directly killing or inhibiting T cells and APC (DC, macrophages, and B cells) and by producing suppressive cytokines such as transforming growth factor-β (TGFβ), IL-10, and IL-35 (Hu et al. 2015). Classification and phenotypes of Treg cell subsets are complex in humans and mice. Porcine Treg are defined as CD25+Foxp3+ (Talker et al. 2013). Within the population of Treg, natural Treg (nTreg) and iTreg can be distinguished. A recent study used the transcription factor Helios to identify porcine nTreg (Käser et al. 2015). Thymic Foxp3+ T cells, which arise at the CD4/CD8α single-positive stage of T cell development, are defined as nTregs and express Helios. Porcine iTreg do not express Helios after
Aspects of B cells immunology in pigs (Butler et al. 2009), also in the context of host–microbe interactions have been discussed recently (Wesemann, 2015). All porcine B lymphocytes are CD25low and sIgM+ B cells may differ in the expression of CD2 antigen. CD21 is always present on all mature B cells but can be expressed in at least two differential forms, assigned as CD21a and CD21b (Sinkora, Stepanova, and Sinkorova 2013). Phenotypic and functional features suggest that CD21b+ B cells are less mature than CD21b− (Sinkora, Stepanova, and Sinkorova 2013). Unlike in mice, porcine B cells appear ca. 2 weeks before T cells and progenitors undergo variable heavy chain gene (VDJH) rearrangement at day 20 of gestation in the yolk sac and day 30 of gestation in the fetal liver before entering high-level lymphogenesis in the bone marrow after day 45 of gestation. Early B cells show high proportion of in-frame rearrangements, undergo switch recombination in thymus on day 60 of gestation, and use N-region insertion from the time of the earliest VDJ rearrangement. The genomic repertoire of VH, DH, and JH genes is small compared to mice, and humans and swine appear to depend on junctional diversity for the majority of their repertoire. The limited VH repertoire of swine contrasts with the porcine TCRβ repertoire, which is extensive, extraordinarily conserved, and is similar to that in humans (Sinkora et al. 2005). The porcine fetal B cells are a naive population developing without idiotypic–anti-idiotypic influences and make pigs a good species to study innate and adaptive immunity. B cell repertoire development, especially in the gut-associated lymphoid tissue (Butler, Zhao, et al. 2009; Butler, Lager, et al. 2009), and the intrauterine repertoire development have been studied in detail (Butler et al. 2000). The first B cells (M-chain-positive) occur in the liver on about day 40 of gestation, and surface IgM+ cells are found in the spleen (day 50) and bone marrow (day 60). A small amount of immunoglobulin is secreted by B cells of the spleen and liver beginning with day 50 of gestation, spontaneous isotype switching from IgM to IgG is occurring in the thymus (Cukrowska et al. 1996). Porcine fetal B cell areas react with various conserved molecules and antigens (Tlaskalova-Hogenova et al. 1994; Butler et al. 2000). A recent study analyzed the distribution of lymphoid cells in the small intestine of germ-free and conventional piglets: B cells prevail in the ileum even in germ-free animals, but microbial colonization is a major factor causing an even greater prevalence of B cells in the ileum and further differential representation of lymphoid cells throughout small intestine (Potockova et al. 2015). Effector cells appear only after colonization, including class-switched IgM+IgA+ B cells, primed CD2−CD21+ B cells, antibody-producing/memory CD2+CD21− B cells, and effector/memory CD4+CD8+ αβ T helper cells.
Based on the above, it is obvious that there are many similarities with other mammals, but also several pig-specific characteristics, in particular high proportions of γδ T cell and CD4+CD8+ T cells, which require further comparative immunobiological research to better understand the translational significance of these differences.
Minipigs in Translational Immunosafety Sciences
After the murine and primate immune system, the porcine immune system is probably the best characterized and offers a wide range of established methodologies and tools as highlighted above (Forster, Ancian, et al. 2010; Mair et al. 2014; Gerner et al. 2015). In all cases, the challenge for immunosafety risk assessment remains the interpretation uncertainty of changes of selected immunological end points when assessing immunocompetence and immune dysregulation in translational safety sciences.
Knowledge of species differences in structure and function of the immune system is of importance in species choice for efficacy and safety studies with novel drugs and translational interpretation of hazard and risks relevant to humans. Gene family analysis revealed a good pig–human similarity (Dawson et al. 2013). Moreover, cross-species genome analyses has revealed essential information regarding the presence of human drug target homologues/orthologues and facilitates species selection for regulatory toxicology studies (Vamathevan et al. 2013). Genomic events, such as gene pseudogenization and gene duplication for specific skin drug targets as observed for the minipig, can complicate extrapolation of animal studies to human (Vamathevan et al. 2013). Nevertheless, because the pig’s skin morphology is comparable to that of humans, pigs and minipigs are often used in dermatological investigations (Mahl et al. 2006; Summerfield, Auray, and Ricklin 2015; Summerfield, Meurens, and Ricklin 2015).
Although numerous studies have been published focusing on particular elements of the porcine immune system, there are only few dedicated publications in the field of immunosafety studies with small molecules or biotherapeutics. The utility of minipigs as a model to be used with small molecules in regulatory immunotoxicology studies has been reviewed (Penninks and van Mierlo 2012). Briefly, Göttingen minipigs were treated with either cyclosporin A or dexamethasone up to 40 days, and immunocompetence was assessed with a number of assays developed to evaluate immunotoxicity in other species: clinical signs, body weight, hematology, immunophenotyping (CD3, CD4, CD8 T cells, and B cells), NK cell activity, primary and secondary immunoglobulin responses to the model antigen keyhole limpet hemocyanin, delayed type hypersensitivity responses to keyhole limpet hemocyanin,
Influence of Dexamethasone Treatment on Peripheral Blood Leukocytes and Lymphoid Tissue in Humans and Pigs.
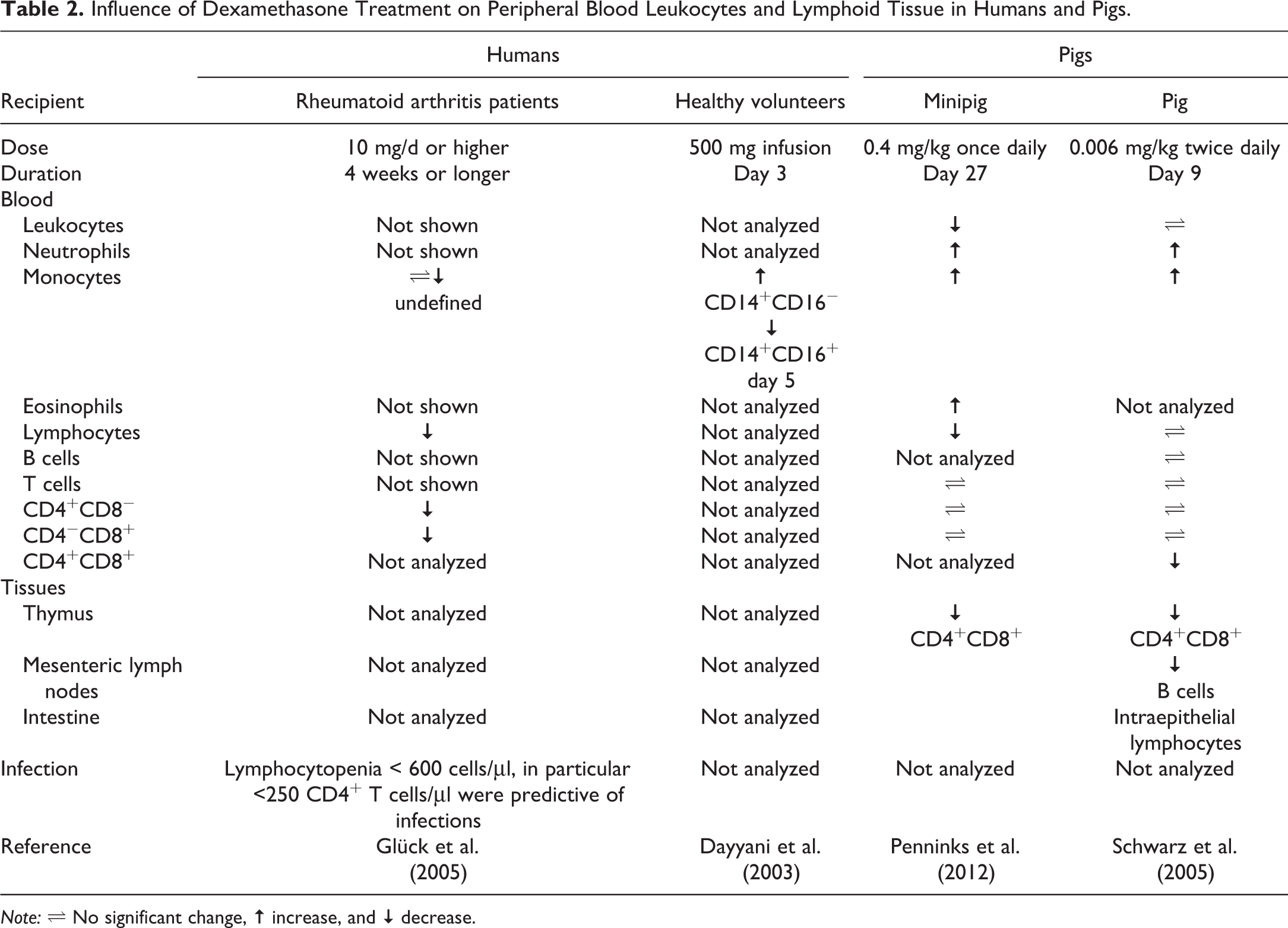
Minipigs have been utilized in safety assessments of biotherapeutics. One study explored Göttingen minipigs as an alternative nonrodent species for immunogenicity testing of the IL-1 receptor antagonist anakinra over 4 weeks (van Mierlo et al. 2013). Minipigs in this study, similar to rhesus monkeys after 4 weeks of daily administration of anakinra, showed no clear treatment-related clinical or pathological findings, except for injection site reactions (van Mierlo et al. 2013). However, all animals developed anti-anakinra antibodies from day 14 onward, which is expected, as anakinra is a recombinant human protein. In another minipig study, the immunogenicity against the TNFα-blocker adalimumab was investigated (van Mierlo et al. 2014). Again, no signs of toxicity of the treatments were observed. Most minipigs treated with adalimumab elicited a detectable antidrug antibody response, which was correlated with decreased plasma levels of adalimumab. As antidrug antibodies against anakinra and adalimumab are less frequent in patients than in minipigs and nonhuman primates (van Mierlo et al. 2013; van Mierlo et al. 2014), despite many similarities between these species, immunogenicity assessments in these species are likely to be overpredictive (Bugelski et al. 2004). Minipigs have also been proposed as a potential alternative model for monoclonal antibody pharmacokinetics after intravenous and subcutaneous administration (Zheng et al. 2012). However, more comparative investigations are needed to support the use of minipigs as an alternative species for safety assessments of biotherapeutics.
Finally, as porcine and human skin have remarkable similarities in structures, pigs have been extensively used as a model for research in dermatology, wound healing, drug delivery, and toxicology. Minipigs have been used to study cutaneous vaccine reactions, and local tolerance to DNA vaccines was compared in domestic pig, minipig, and mouse (Dincer et al., 2006). Furthermore, the acute toxicity and biodistribution of the DNA vaccine against hepatitis B was studied in minipigs (Pilling et al. 2002). The gene delivery was well tolerated, and biodistribution and persistence of the plasmid were acceptable, making the minipig, for this type of vaccine, a good model. More recently, minipigs were used successfully for dermal vaccine delivery of the hepatitis B surface antigen reinforcing the robustness of this model (Ploemen et al. 2014).
Thus, although several publications have described studies with minipigs to assess immunocompetence in translational safety sciences, more systematic and cross-species comparisons are needed to support the translational significance of immunological findings in minipigs. We have begun to explore lymphoid populations and their function in minipigs in more detail (Rubic-Schneider et al., unpublished data), in order to address this gap identified earlier by others (Penninks and van Mierlo 2012).
Future Perspective
As outlined in this brief review, much progress has been made in recent years to further define the immunological characteristics of domestic pigs and minipigs. Fairbairn et al. (2011) and Mair et al. (2014) provide tabulated summaries of the benefits and limitations of pigs as translational animal model to study aspects of immune system competence. Although the minipig is likely to be a valid alternative nonrodent species for safety testing of immunomodulatory development candidates, it is also clear, that the pig has several immunobiological peculiarities, which require additional investigations to further assess the significance of these differences (Mair et al. 2014; Gerner et al. 2015). Based on these assessments, the potential or limitations of minipigs in the context of immune safety assessments can ultimately be determined. Some areas for further investigations in minipigs are linking the significance of γδ T cells and αβ T cells, in particular CD4+CD8+ cell populations, and immune functional end points, with immunocompetence and susceptibility to immune-mediated disorders, such as infections. Neither infections nor immune responses are static processes. Hence, dynamic assessments of lymphocyte counts and immune function parameters by means of repeated testing over time would be advantageous (Fernandez-Ruiz, Kumar, and Humar 2014). Furthermore, the availability of laboratory tools could make the minipig the preferred species before the dog. Pigs, in contrast to dogs, have a digestive tract that is physiologically more similar to humans (P. Haley 2012), which has made it a mechanistically valuable and accepted species in safety studies of food and food additives (Harvey 2012). In addition, research into dietary modulation of the human gut microbiome indicates that human microbiota-associated pigs have the potential to be a valuable model for investigating microbe–host interactions in human health and disease (Heinritz, Mosenthin, and Weiss 2013; Wang and Donovan 2015). Overall, this should make minipigs the species of choice for safety studies with orally administered drugs, in contrast to the more sensitive dog (Morris et al. 2010). Dedicated comparative studies with relevant marketed drugs might further substantiate the benefit of minipigs in this regard.
Footnotes
Acknowledgments
We apologize to those colleagues whose work was not cited owing to space restrictions or our ignorance.
Authors’ Contribution
Authors contributed to conception or design (TRS, DB, and MK); data acquisition, analysis, or interpretation (TRS and BC); and drafting the manuscript (MK). All authors critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
