Abstract
Vanadium is a metal present in particulate matter and its reprotoxic effects have been demonstrated in males and pregnant females in animal models. However, the effects of this metal on the reproductive organs of nonpregnant females have not been sufficiently studied. In a vanadium inhalation model in nonpregnant female mice, we found anestrous and estrous cycle irregularity, as well as low serum concentrations of 17β-estradiol and progesterone. A decrease in the diameter of secondary and preovulatory follicles, as well as a thickening of the myometrium and endometrial stroma, was observed in the vanadium-treated mice. There was no difference against the control group with respect to the presence of the estrogen receptor α in the uterus of the animals during the estrous stage. Our results indicate that when vanadium is administered by inhalation, effects are observed on the female reproductive organs and the production of female sex hormones.
Introduction
Epidemiological studies have shown the negative impact of air pollution on human fertility, mainly by particulate matter (PM) 1 ; further, it has been established that the fertility rate decreases by 2% for each increment of 10 µg/m3 of PM2.5. 2 Regarding female infertility, Mahalingaiah et al 3 reported the association between the incidence of infertility in women and living near main roads with exposure to PM2.5-10. It has been suggested that air pollution can be fetotoxic in humans; specifically, maternal exposure to PM can result in early pregnancy loss and miscarriages, low birth weight, preterm birth, and neonatal mortality. 4,5 Other reports of laboratory animals show damage by PM in nonpregnant females. These alterations affect the duration of the estrous cycle and involve a decrease in the number of antral follicles, 6 as well as ovarian hemorrhage and congestion; 7 in pregnant female mice, a loss of embryo implantation and a lower fetal weight 6 were observed. Another aspect to consider is the relationship between PM and the alterations in endocrine functions, including the inhibition of the activity of enzymes involved in the synthesis of hormones, as is the case of aromatase, which enables the synthesis of estrogens from androgens in the placenta cells. 8,9 Other studies suggest that PM is endocrine disruptor either by the particles themselves or by the compounds adhered to the surface of PM.
Particulate matter with the smaller aerodynamic diameter are the most toxic because after they are inhaled, they remain longer in the alveoli, 10 facilitating its contact with the alveolar surface, liberating the metals adhered to them, crossing the air–blood barrier, and entering the systemic bloodstream; these particles are generated mainly by the burning of fossil fuels in motor vehicles and for industrial use. 11 The toxicity of the suspended particles depends not only on their size but also on their composition, and it has been determined that the major components of PM2.5 and PM1 are metals. 12 Vanadium is a metal emitted into the atmosphere by the combustion of fossil fuels with high levels of this metal. 13 Pentavalent vanadium compounds are the most toxic; the inhalation of vanadium induces organ and tissue injury, oxidative stress, and genotoxicity. 14 In animal models, the oral or intraperitoneal administration of different vanadium compounds have increased resorptions and pre- or postimplantation losses 15 and caused fetal death and reduced fetal weight; also, teratogenic effects have been observed. 16 However, the studies are focused in the effects on pregnant females, fetuses, or newborns and the data are very limited, particularly regarding the toxic effects of vanadium on the female reproductive system of a nonpregnant female. Therefore, the aim of this article is to describe the effects of vanadium on the estrous cycle, the ovaries, and the uterus of mice exposed to this metal by inhalation.
Materials and Methods
Animals
A 46-day-old CD-1 female mice weighing 30 to 35 g were housed in a temperature controlled (22°C ± 2°C), 12-hour light/dark cycle room, and were allowed to acclimate for 1 week prior to experimentation. Animals had ad libitum access to water and pelleted food (Purina rodent chow). The study was approved by the Ethics Committee of the Faculty of Medicine, UNAM (#04-2005), and the Mexican Guideline for Animal Welfare (NOM-062-200-1999). After the week of acclimatization, the animals underwent a daily vaginal exfoliative cytology for 2 weeks, with the aim of determining the regularity of the estrous cycle in each animal, and therefore, to be able to exclude those mice that did not have a regular cycle.
Inhalation Model
Forty mice were randomly divided into 2 groups, control (n = 20) and exposed group (n = 20). The exposed animals were placed in an acrylic box with a volume of 3.3 L, connected to a nebulizer (Ultraneb 99, De Vilbiss, Somerset, Pennsylvania) with vanadium pentoxide (V2O5) in saline (0.02 M) for 1 hour with a constant flow of 10 L/min, twice a week per 4 weeks. The vanadium concentration inside the chamber was 1.44 mg/m3. 17 The concentration selected for this experiment was greater than the concentration reported in workplaces (0.2-0.5 mg/m3) by International Agency for Research on Cancer, 18 but in the range of those noted by the World Health Organization 19 (0.01-60 mg/m3).
Based on the previous determinations of the diameter of the particles and the specifications from the nebulizer manufacturer, the diameter range of the aerozolized particles was 0.5 to 5 μm. 14,17 Control mice inhaled only saline vehicle under the same conditions. The animals were weighed daily to assess possible changes in body weight in response to the treatments.
Exfoliative Vaginal Cytology
To evaluate whether the females presented a normal estrous cycle, we performed the cytology every day at the same hour. According to Caligioni, 20 the micropipette was filled with 25 µL of phosphate-buffered saline (PBS) to perform vaginal washes and to obtain the vaginal smears. The fluid was placed on a glass slide and stained with methylene blue (0.1%) and then viewed under the microscope (CH30; Olympus, Miami, Florida). The proestrous is characterized by the presence of a great number of nucleated cells; in the estrous, irregular cornified epithelial cells with no visible nucleus and granular cytoplasm were observed; during the metestrus, leukocytes and a few nucleated and cornified cells were observed; in the diestrous, there were leukocytes in the vaginal smear. The exfoliative cytology was performed after the final inhalation exposure.
Blood Collection
After the fourth week of inhalation, animals were anesthetized by an intraperitoneal injection of sodium pentobarbital (PiSa Agropecuaria, S.A. de C.V., Guadalajara, Jal., Mexico; 35 mg/kg body weight). To quantify the concentration of estradiol and progesterone, we obtain a blood sample directly from the atrium with a 1-mL syringe and immediately placed it in a tube containing no anticoagulant and allowed it to clot at room temperature. The serum was separated by centrifugation, 2 times for 10 minutes at 4,500 rpm, and was stored at −70°C until it was used for the quantification of 17β-estradiol and progesterone by radioimmunoassay.
Radioimmunoassay for 17β-Estradiol and Progesterone
Each sample was analyzed in duplicate for the quantification of estradiol and progesterone by solid-phase radioimmunoassay (Coat-A-Count Estradiol and Coat-A-Count Progesterone kits; Siemens Medical Solutions Diagnostics, Los Angeles, California, USA) Briefly, 100 μL of test serum and 1,000 μL of estradiol or progesterone (depending on the case) were labeled with the 125I isotope and placed in tubes, afterward, they were shaken and incubated at room temperature for 3 hours; then the supernatant was decanted and the radioactivity was counted per minute in a γ counter. To determine the concentration of serum hormones, a curve was made (in duplicate) of each labeled hormone, where the known concentrations of this hormone were used to obtain its equivalent in radioactivity. 21
Ovary and Uterus Collection
After obtaining the blood samples, the animals were perfused intracardially with 0.9% saline solution followed by 4% paraformaldehyde (PFA) in PBS (pH 7.4). The organs were then removed, sectioned, and postfixed in 4% PFA/PBS, dehydrated, and paraffin embedded. Three-micrometer thick sections were obtained, some were placed on poly-
Immunohistochemistry
An immunohistochemical staining for the estrogen receptor α (ERα) in the uterus was performed by using a streptavidin–biotin–peroxidase complex method. Paraffin of 3-µm thick sections were dewaxed and rehydrated, the antigen retrieval was performed with Diva Decloaker, 1:20 for 3 minutes at 15 psi (Biocare Medical, Concord, California) and the endogenous peroxidase was quenched using H2O2 0.9% for 30 minutes at room temperature. Estrogen receptor α primary antibody (Santa Cruz Biotechnology, Santa Cruz, California; 1:200) was incubated for 45 minutes at ambient temperature afterward; universal antibody (Biotinylated Link Universal; DAKO, Carpinteria, California) was incubated for 30 minutes, and thereafter streptavidin–horseradish peroxidase (DAKO) was also incubated for 30 minutes. DAB-plus substrate kit (Invitrogen, Carlsbad, California, USA) was used to reveal the peroxidase reaction. Finally, the samples were counterstained with hematoxylin.
The evaluation of the immunoreactivity was assessed on tissue sections by color densitometry. Photomicrographs were taken of 30 fields of 180 μm2, each randomly selected per group, with an Olympus BH2 microscope (Olympus) adapted with a digital camera Canon T4, 18 mpx (Canon USA, Inc, Huntington, New York, USA). The photomicrographs were processed with an image analysis software (Adobe Photoshop CC, San Jose, California, USA) in a color model CMYK (Cyan, Magenta, Yellow and Key) using the yellow channel. The densitometry was analyzed with a software developed in MATLAB (R2012b) and color intensity was reported in pixel units.
Histomorphometry
Serial sections from an ovary and a uterus of each mouse were stained with H-E and they were evaluated with a light microscope equipped with a micrometric ocular lens (CH30; Olympus). For the ovary morphometry, 10 primary follicles, 10 secondary, and 10 preovulatory follicles were measured to determine the largest diameter of each follicle; one of the conditions was that the nucleus of the oocyte had to be visibile. The total diameter included oocyte, granulosa, and theca cells. In some cases, the nucleus was observed in more than 1 section, so its diameter was measured in each section; the determination of the maximum diameter of each follicle was made in the section where the nucleus was observed in its largest diameter. In the case of the uterus, 10 serial longitudinal sections of 1 of the uterine horns per animal were observed with a light microscope, and the maximum thickness of the epithelium and myometrium of 30 fields of 450 μm2 each per group was quantified.
Image Processing
Nikon microscope (model eclipse 90i) and a camera of the same brand (model DS-Ri1) with 12.7 MP adapted to the microscope and an image analyzer software (NIS-Elements, Melville, New York, USA) were used. The images were processed with Adobe Photoshop CS4 software. The processing was turning the background completely white.
Statistical Analysis
Data were presented as means ± standard error of the mean. Statistical significances of body weight, exfoliative cytology, radioimmunoassay, and immunohistochemistry were determined by 1-way analysis of variance and post hoc Tukey for multiple comparisons. For the histomorphometry studies, means between 2 groups were compared with a Student t test. The differences were considered statistically significant when P was <0.05 using a Graph-Pad Prism 5.3 software (GraphPad Software, La Jolla, California).
Results
Body Weight
There were no measurable differences in body weight between the control animals and the females exposed to vanadium (Figure 1).
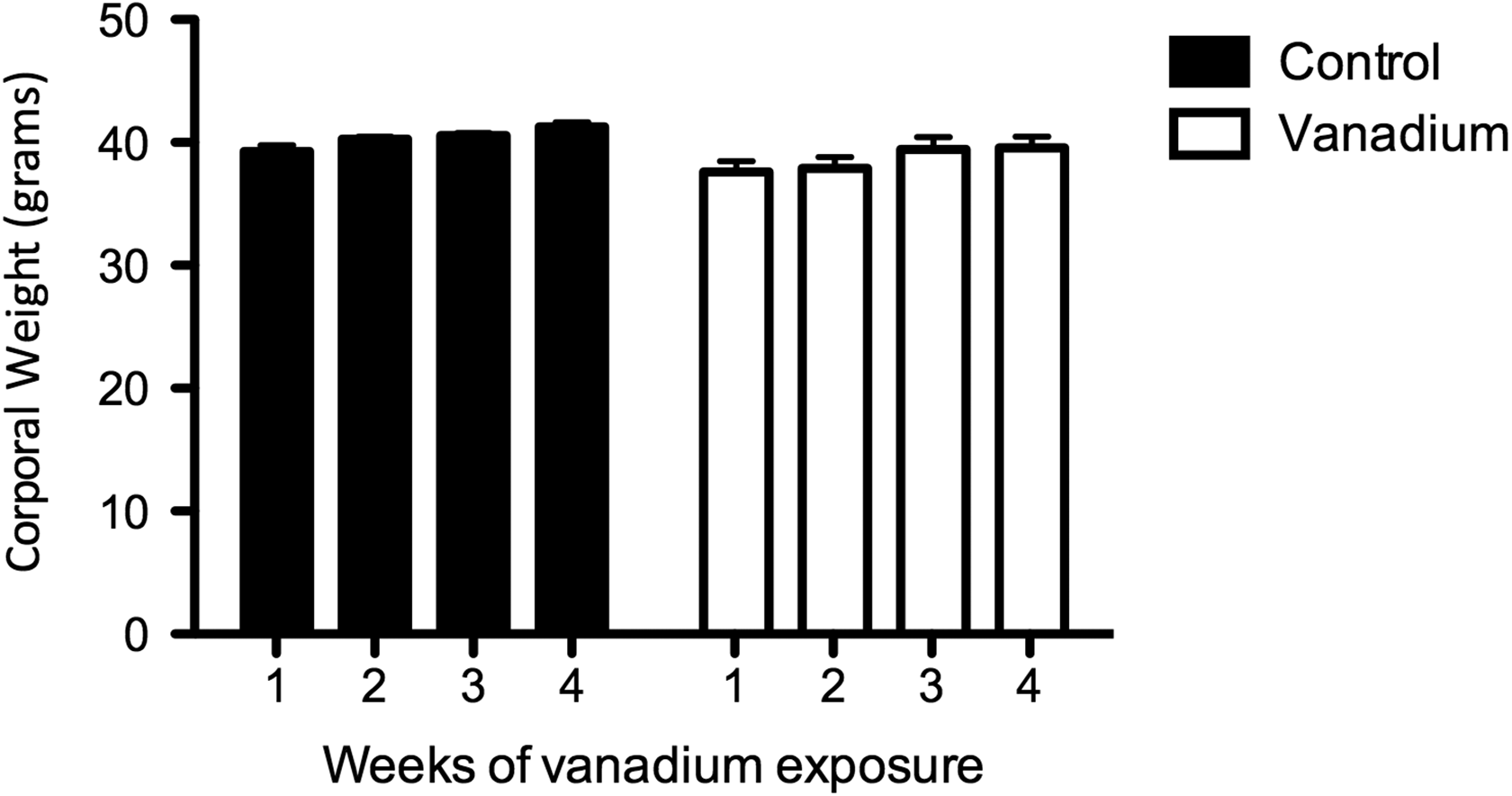
Average body weight in grams of the control (n = 20) and vanadium (n = 20) mice over 4 weeks. There were no significant differences between the groups or during the weeks of experimentation.
Estrous Cycle
The control group presented a normal estrous cycle with periodical cycles in proestrous, estrous, metestrus, and diestrous during the 4 weeks (Figure 2). In contrast, on the third week of inhalation, 50% of the females from the vanadium group presented parabasal and basal cells in vaginal smears, which means that they were in anestrous. During the fourth week of vanadium inhalation, 72.3% of the females exposed remained in anestrous (Figure 3). In the vanadium group, during weeks 3 and 4, daily vaginal cytology showed that the females that were not in estrous, only presented the metestrus and diestrous stages.
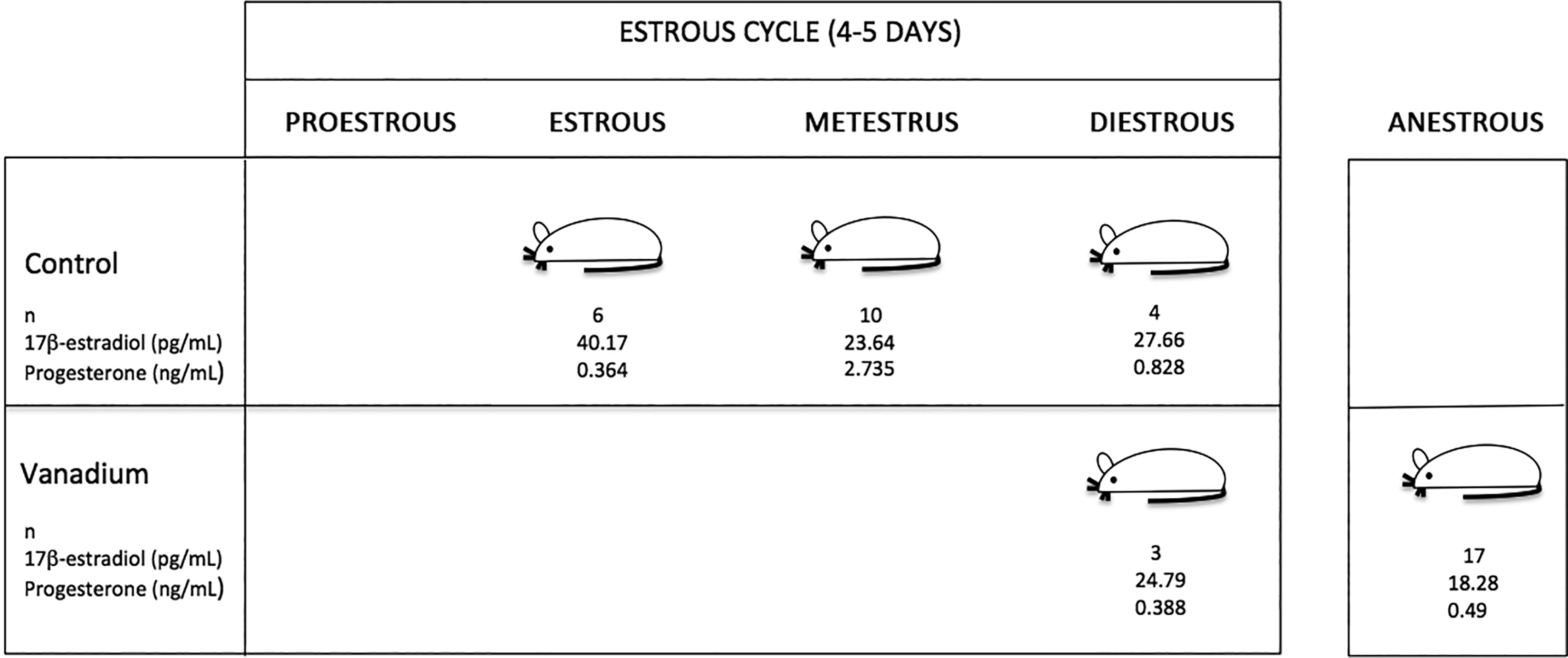
Estrous cycle, blood 17-β estradiol, and progesterone concentrations in female mice after the final vanadium inhalation exposure (n = number of animals per stage of the estrous cycle).
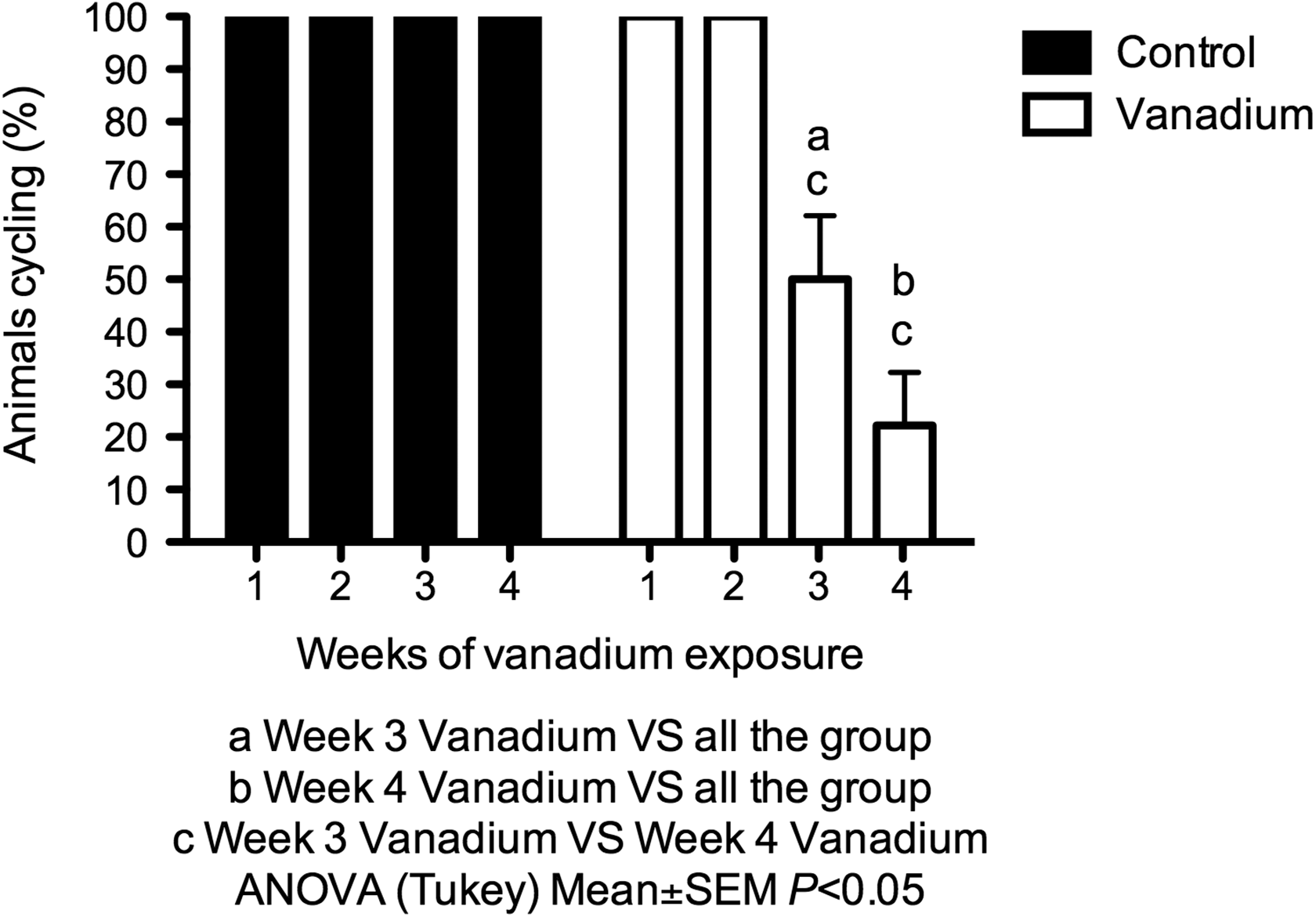
Percentage of mice that presented regularity in the estrous cycle in the control (n = 20) and vanadium (n = 20) groups during the 4 weeks of the treatment.
Serum 17β-Estradiol and Progesterone
The measured concentrations of 17-β estradiol in the blood of the female mice in the control group at estrous were 40.17 ± 1.39 pg/mL, 23.64 ± 0.39 pg/mL during metestrus, and 27.66 ± 1.19 pg/mL in diestrous; while the female mice exposed to vanadium had a concentration of 24.79 ± 0.48 pg/mL in diestrous and 18.28 ± 0.39 pg/mL at anestrous (Figure 4). As a consequence of the vanadium exposure, estrus and metestrus were not observed, and thus blood samples were not evaluated for 17-β estradiol.
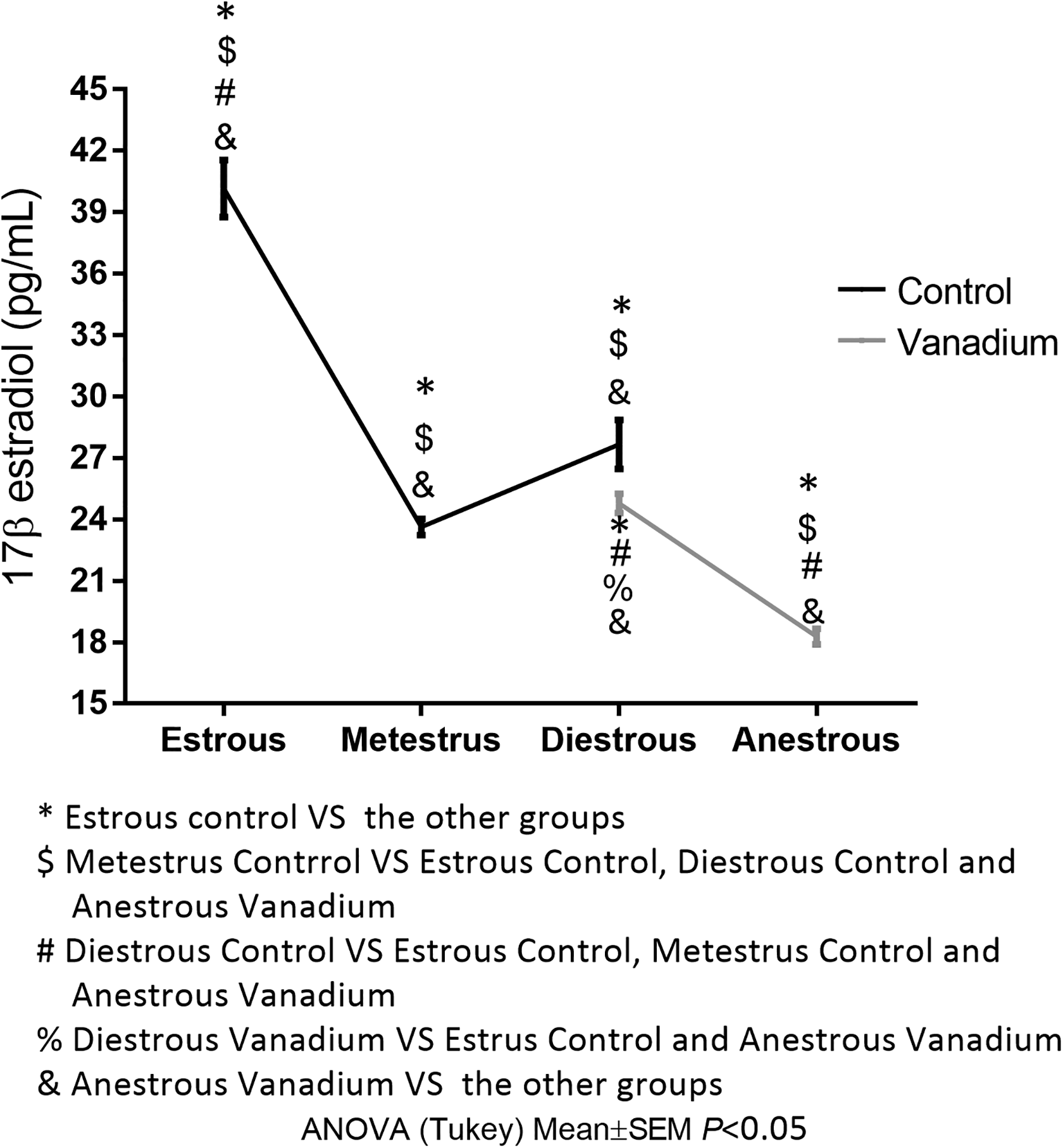
Mean concentration of 17-β estradiol in serum per stage of the estrous cycle at week 4 of the treatment. Control group: estrous n = 6, metestrus n = 10, diestrous n = 4. Vanadium group: diestrous n = 3, anestrous = 17.
Progesterone levels of the control group were 0.364 ± 0.018 ng/mL during estrous, 2.735 ± 0.27 ng/mL in metestrus, and 0.828 ± 0.035 ng/mL in diestrus; the values of the vanadium group were 0.388 ± 0.002 ng/mL during diestrous and 0.49 ± 0.06 ng/mL in females at anestrus (Figure 5). As a consequence of the vanadium exposure, estrus and metestrus were not observed, and thus blood samples were not evaluated for progesterone.
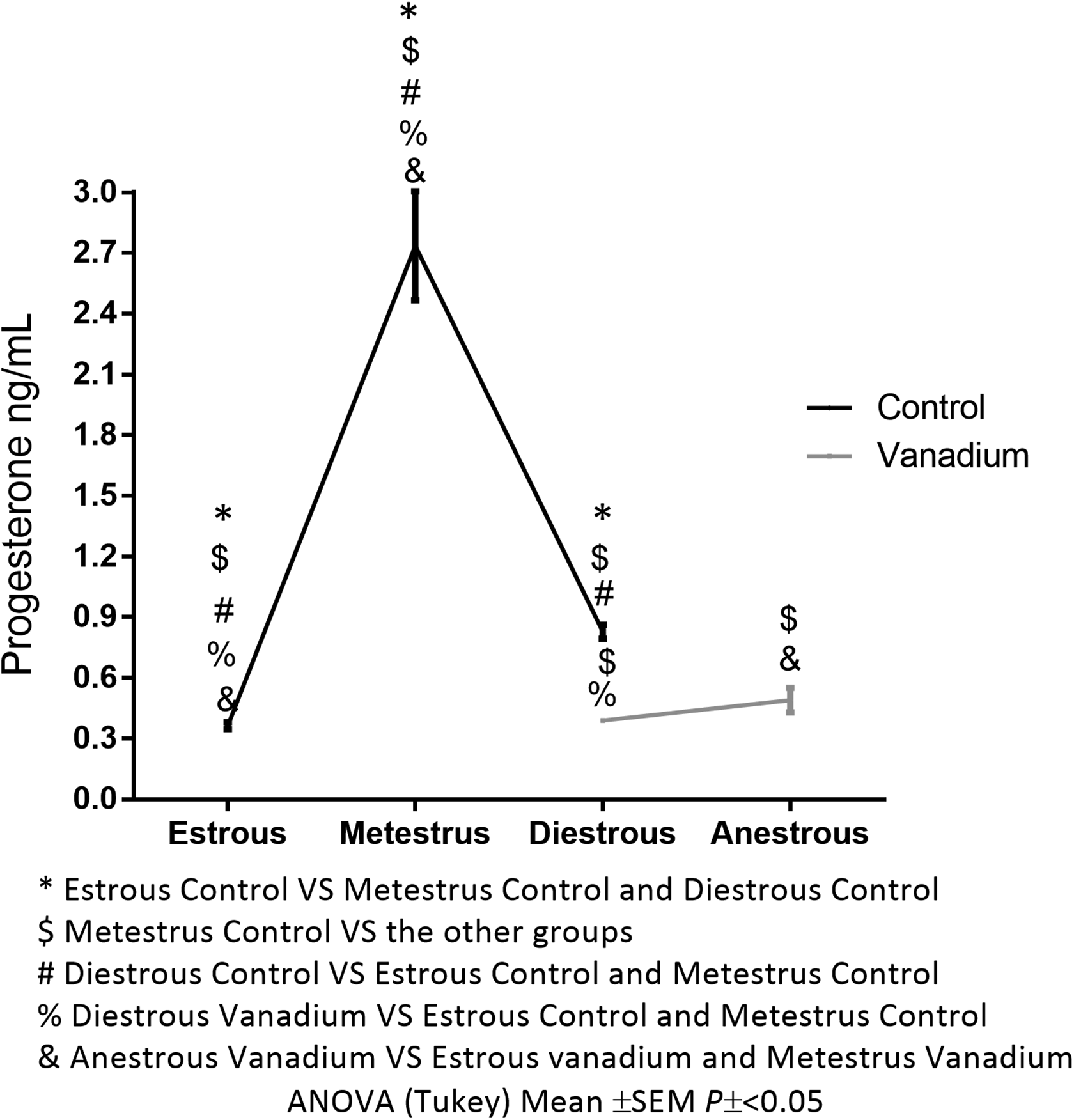
Mean concentration of progesterone in serum per stage of the estrous cycle in week 4 of the treatment. Control group: estrous n = 6, metestrus n = 10, diestrous n = 4. Vanadium group: diestrous n = 3, anestrous = 17.
Estrogen Receptor α in Uterus
The presence of the receptor in the uterine epithelium was 1,746,000 ± 106,158 pixel units and 687,413 ± 71,661 pixel units in the myometrium of the control group. In the vanadium group, 1,941,000 ± 165,134 pixel units in epithelium and 844,986 ± 76,401 pixel units in the myometrium were measured, and there were no statistically significant differences between the groups (Figure 6).
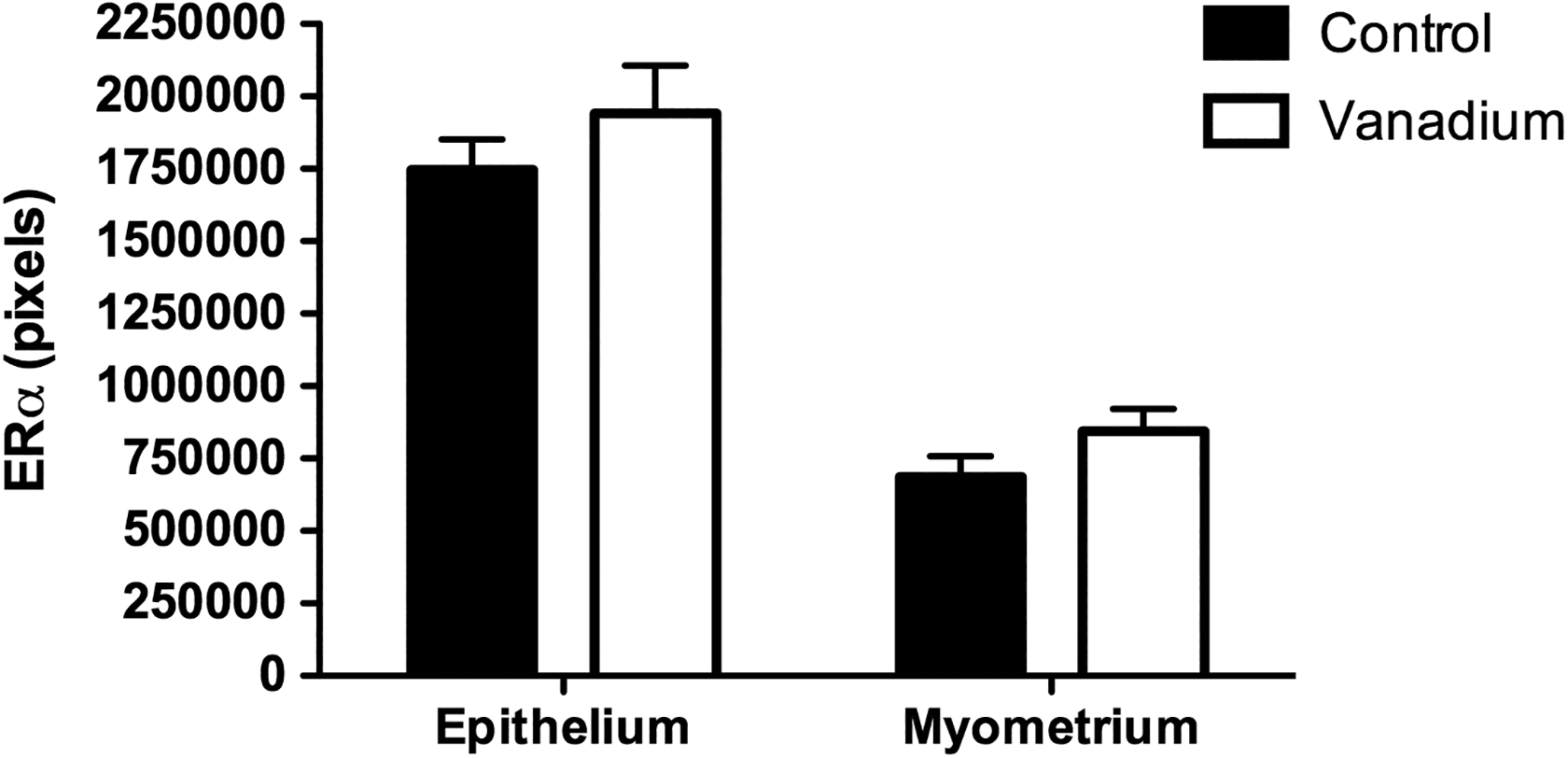
Immunoreactivity for Erα in the epithelium (n = 30 fields) and in the myometrium (n = 30 fields) of the uterus of the control and vanadium groups at the fourth week of exposure. There was no difference between the groups. Erα indicates estrogen receptor α.
Histological Evaluation
Figure 7 shows representative images of the histological sections of the ovary of an animal in the control group during estrous (Figure 7A) and an ovary of an animal in the vanadium group in the anestrous stage (Figure 7B). Figure 8 shows the mean follicular diameter per group and per follicle class. As previously observed with primary follicles, there is no statistically significant difference between control and vanadium groups (100.6 ± 4.89 µm and 104.8 ± 3.76 μm, respectively). The diameter of the secondary follicles (212.9 ± 11.76 μm) and the preovulatory follicles (313.0 ± 5.5 μm) of the vanadium group was smaller than the diameter of the secondary follicles (250.4 ± 12.8 μm) and of the preovulatory follicles of the control group (370.3 ± 4.40 μm).
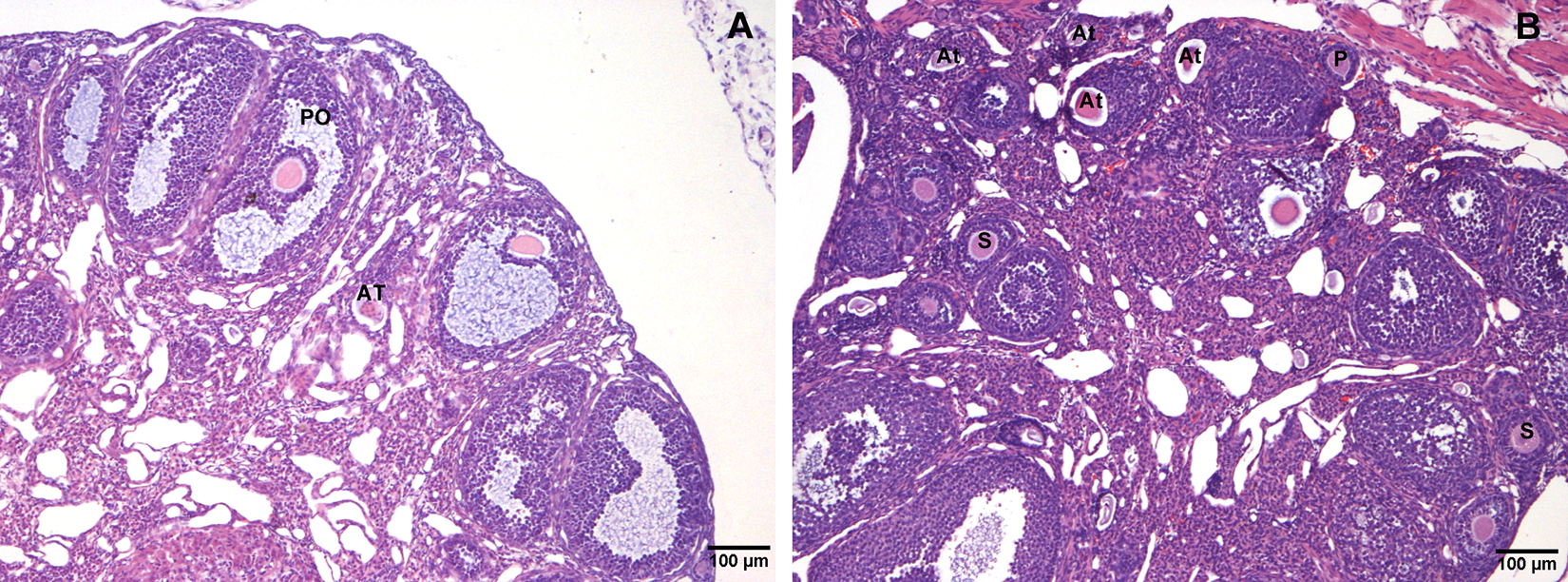
Representative ovary histology sections from control (A) during estrous and vanadium (B) groups during anestrous stage. In addition to the decrease in the size of the ovarian follicles, the vanadium group presented a higher frequency of atretic follicles (At). Primary (P), secondary (S), and preovulatory (PO) follicles.
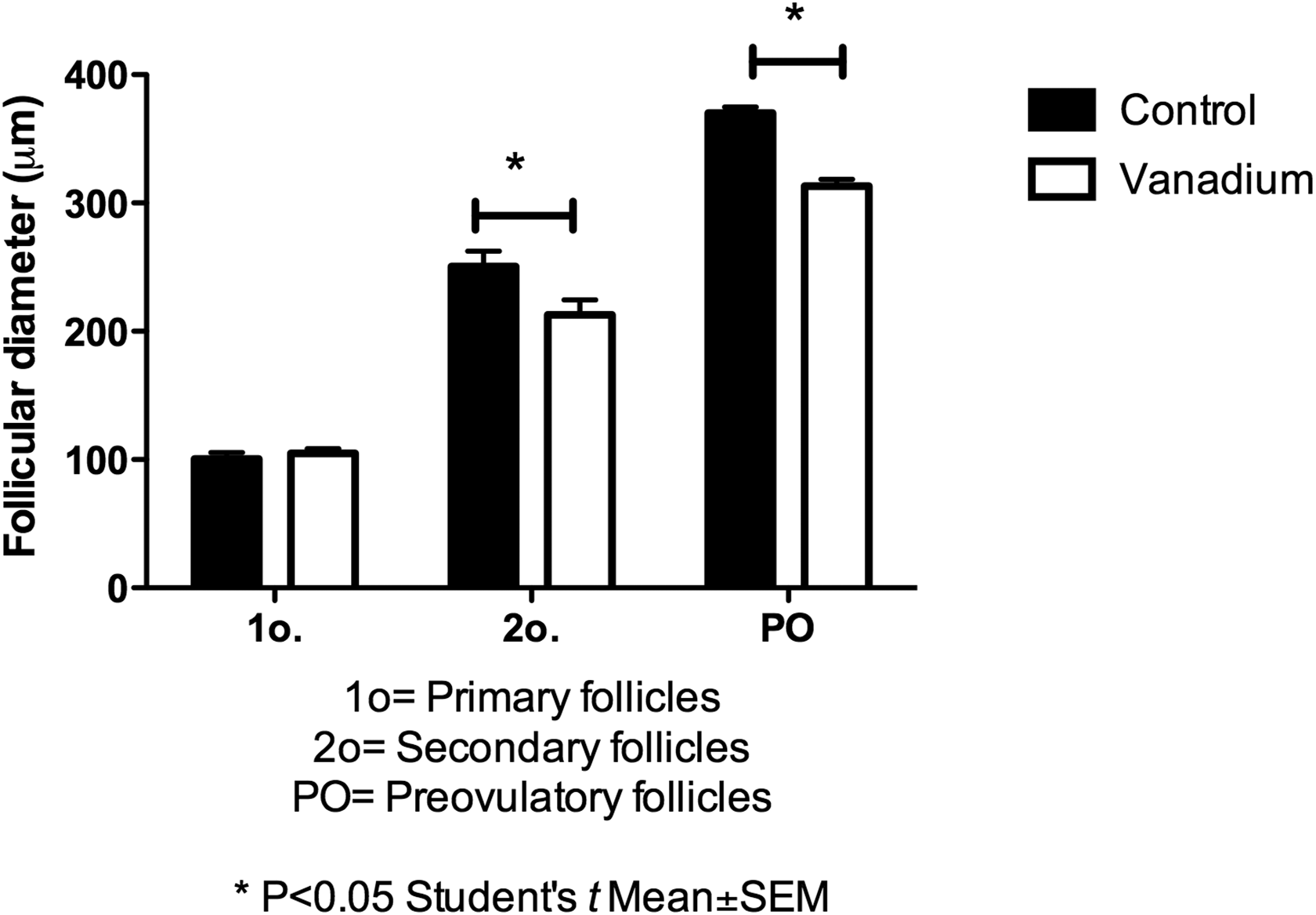
Diameter of the primary follicles (n = 10), secondary (n = 10), and preovulatory(n = 10) in the ovaries of the control and vanadium groups at the fourth week of exposure.
Longitudinal sections of mouse uterus revealed distinct histological morphologies consistent with the stages of the estrous cycle; these changes were more evident in the diestrous (Figure 9A) and in the estrous (Figure 9B), the uterine wall was thicker due to the development of the endometrial glands and the edema of the stroma. However, we observed that in the vanadium group (Figure 9C), the uterine wall was thicker even when compared with the samples of the estrous females of the control group because of the presence of inflammatory infiltrate. We did not find any statistically significant differences in the thickness of epithelium between groups, 16.5 ± 0.46 µm in the vanadium group and 16.1 ± 0.37 µm in the control group. The stroma from vanadium group was thicker (178.8 ± 11.41 µm) than the stroma of the control group (129.6 ± 3.34 µm); also myometrium thickness was greater in vanadium group (131 ± 5.5 µm) than control group (66.79 ± 2.02 µm; Figure 10).
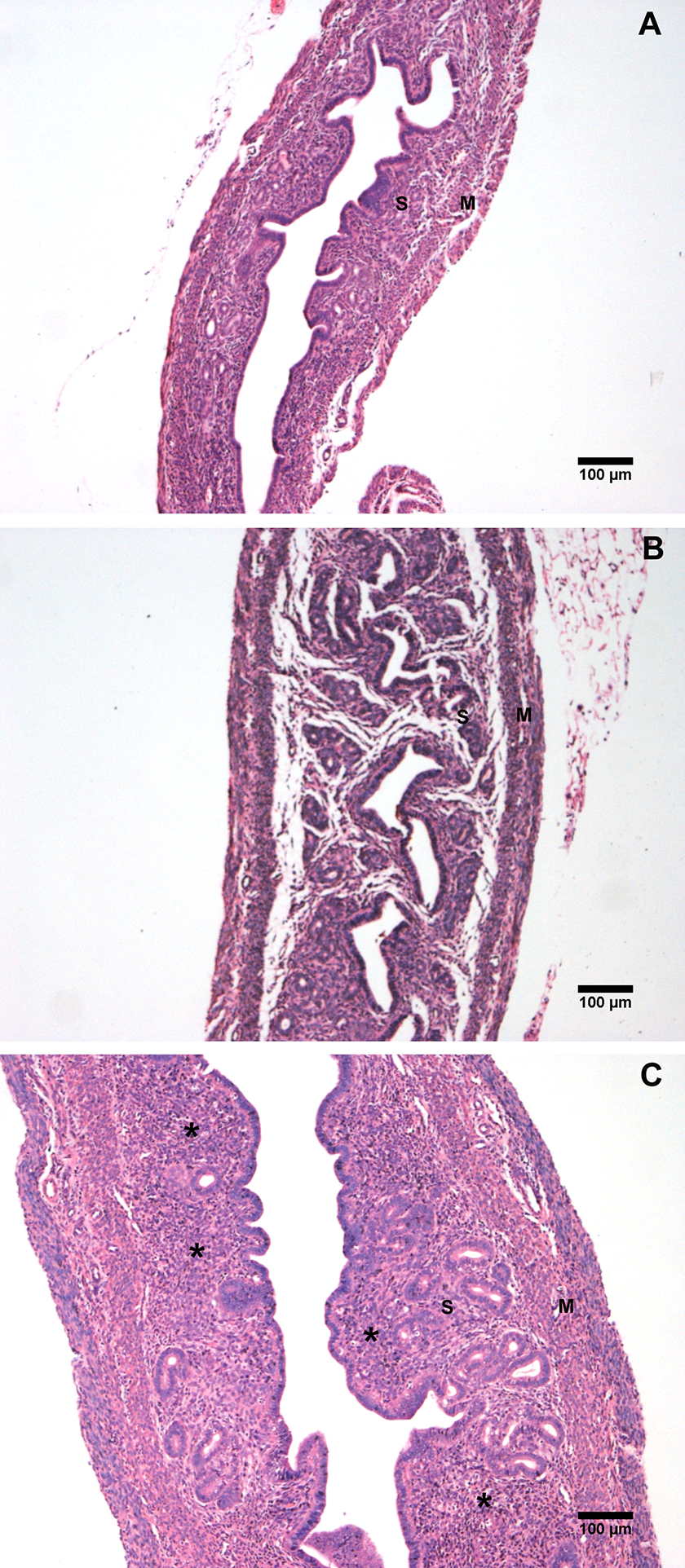
Histological evaluation of uterine longitudinal tissue sections derived from diestrous (A) and estrous (B) stages of the estrous cycle of control group and anestrous (C) of vanadium group. Sections reveal gross changes in the morphology of endometrial stroma (S) and myometrium (M) (*) inflammatory infiltrate.
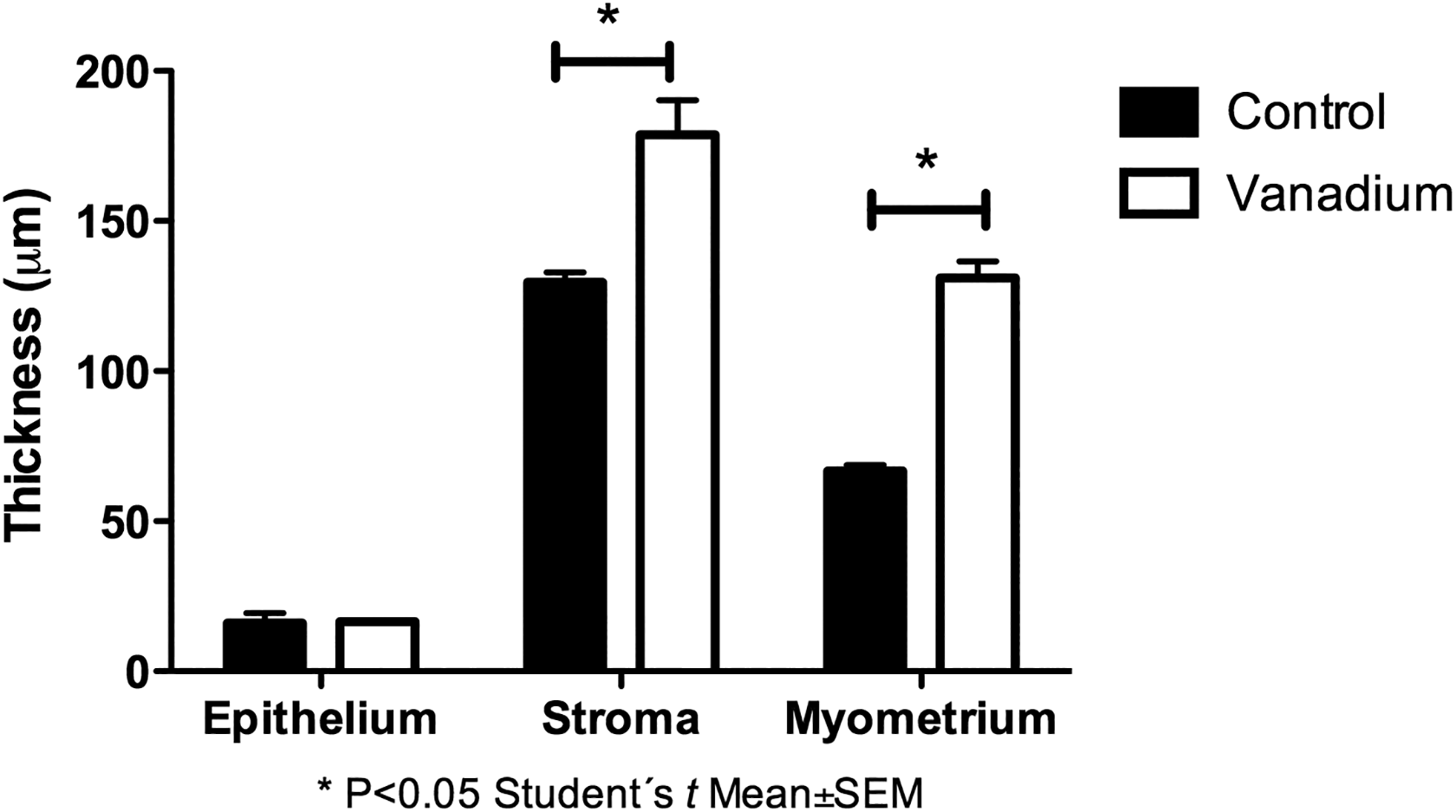
The maximum thickness of uterine epithelium (n = 30 fields) and myometrium (n = 30 fields) in the uterus of control and vanadium mice at the fourth week of exposure.
Discussion
After 4 weeks of vanadium inhalation, more than 70% of the females in this group had immature vaginal desquamation cells (basal and parabasal), which is an indicator of anestrus, or inhibition of the estrous cycle. 22 The exposure to adverse conditions can modify the regularity of the estrous cycle and body weight. For this reason, during the 4-week experiment, body weight was included as an indicator of the adequate management of the animals, and there were no differences in the weight between the experimental and control groups. This finding supports that the findings in this experimental protocol were due to the experimental exposure.
It is proven that different metals such as lead, chromium, and cadmium cause changes in the duration of the estrous cycle 23 -25 and that some vanadium compounds alter the regularity in the estrous cycle. 15 Even though different metals behave in different ways, they share some common toxic mechanisms such as oxidative stress, cellular uptake, interfere signaling pathways, and mimic other elements such as phosphates and sulphates. All these mechanisms might affect several steps in hormonal regulation of the hypothalamic–pituitary–gonadal axis. 26 Metals such as chromium, cadmium, and iron can alter the hormonal axis indirectly through the generation of oxidative stress. 27 -29 Obianime et al 30 observed a decrease in the blood levels of Luteinizing Hormone (LH), Follicle Stimulating Hormone (FSH), and prolactin in rats, following intraperitoneal administration of ammonium metavanadate; this decrease in LH and FSH caused by vanadium could explain the low levels of estrogen and progesterone in the blood of exposed mice. In normal mice, the hormones synthesized in the ovary are regulated by the pulses of LH and FSH produced in the anterior pituitary gland; it is possible that vanadium is also interfering in the liberation of these hormones by this gland; however, this hypothesis needs further research to explore this possible explanation. In addition, previous reports from our group revealed that vanadium inhalation decreased the presence of tubulin and actin in mice testicular cells, proteins that are essential for the transport of vesicles that carry receptors to the membrane, possibly affecting hormone release or hormone binding. 31,32
We observed that vanadium inhalation stops or inhibits the estrous cycle causing anestrous in almost all the female mice, and since the estrous cycle is regulated by hormones, we asked whether it was possible for the metal to alter sex hormones. An analysis of the concentration of 17-β-estradiol and progesterone in the blood of the females in both groups showed that the levels of both hormones were significantly lower in the group exposed to vanadium during diestrous and anestrous. In the exposed mice, vanadium inhalation inhibited the other 2 stages of the estrous cycle (estrous and metestrus; Figure 2).With this result, we suggest that vanadium can be considered as endocrine disruptor, acting as ligand and binding to the receptors of the target cells, as agonists or antagonists of a variety of hormones; examples of this effect are observed in other metals such as cadmium, vanadium, chromium, lead, nickel, and copper can be linked to ERα by mimicking the function of estrogen in Mcf-7 breast cancer cells exposed to different compounds of these metals. 33 Cobalt interferes with the secretion of progesterone in porcine ovarian granulosa cells, 34 and cadmium alters the synthesis of estrogen and progesterone in human adrenocortical carcinoma cell line NCI-H295R. 35
Particulate matter is also considered as an endocrine disruptor, either by the particles themselves or by the compounds attached to them 9 ; PM is capable of modifying the function of the enzymes necessary for the synthesis of sex hormones, such as the aromatase that catalyzes the aromatization of fetal and maternal androgens into estrogens in JEG-3 (Human choriocarcinoma cell line) human placental cells, where it has been shown that aromatase activity decreases by exposure to PM1 collected from urban areas with intense traffic. 8
The basal cells of the vaginal epithelium need to increase the absorption of calcium for its cornification, 36 which could be inhibited by vanadium since this metal inhibits the enzyme calcium-adenosine triphosphatase and reduces the extracellular calcium flow. 37 Proliferation and cornification of the vaginal epithelium also occur in response to growth factors Heparin-binding EGK-like growth factor (HB-EGF), Epidermal Growth Factor (EFG), Transforming Growth Factor alpha (TGF-α), 38 and vanadium modifies the secretion of HB-EGF. 39 This indicates that this metal could cause changes in vaginal histology this way.
The relationship between ERα with estrogen levels and uterine morphology has been demonstrated 40,41 ; however, we found no difference in the presence or distribution of ERα in the uterus between the experimental groups. This suggests that although vanadium decreased the levels of estrogen in the blood, it does not modify the presence of the receptor but could modify its function. Martin et al 33 demonstrated in vitro that in MCF-7 mammary cancer cells treated with sodium vanadate, the estrogen binding to ERα was inhibited and, at the same time, the cell proliferation and the expression of estrogen-regulated genes increased. Vanadium also alters signaling pathways related to hormones such as insulin, 42 and we found in a previous study with our inhalation model that vanadium induces Janus/kinase/ signal transducer and activator of transcription (JAK/STAT) signaling pathway activation in megakaryocytes, which is regulated under normal conditions by thrombopoietin. 43 Another characteristic of this metal is the generation of oxidative stress, 44 and it has been shown that in the adipocytes subjected to oxidative stress, the binding capacity between glucocorticoids and their receptor decreases even though the receptor is functional. 45
The development of the ovarian follicles depends on the hypothalamic–pituitary–gonadal axis, and the hormones produced by these organs modulate intracellular events; among them are the dynamic changes in the extracellular matrix and it has been shown that metalloproteinases (MMPs) participate in the processes of atresia, ovulation, and growth. 46 In our vanadium inhalation model, we reported that there was an increase of MMP2 in the brain structures of the exposed animals 47 ; and it is possible that vanadium modifies the function of MMPs in the ovary and therefore alters the development of secondary and preovulatory follicles. Another event to consider is the interaction between granulosa cells and the oocyte, which is determined by several elements of the cytoskeleton. 48 It has been reported that γ-tubulin, 31 actin, 32 and connexin 43, 49 decrease in Sertoli cells, Leydig cells, and germ cells in CD-1 mouse testis exposed to vanadium inhalation in a time-dependent manner, which indicates the possibility of damage in the ovarian cell cytoskeleton that could disrupt the follicular growth.
We found that the thickness of the endometrial stroma and of the myometrium was greater in the females in the vanadium group than in the animals on the control group. Uterine alterations have been related to metal exposure; Tchernitchin et al, 50 when administering cadmium (subcutaneously) to rats, observed an increase in eosinophilia and edema in the endometrium and that in the absence of estradiol, cadmium causes cellular hypertrophy in the myometrium. Our results indicate that in the vanadium group the thickness of the endometrial stroma and the myometrium increased and that it was related to the presence of inflammatory infiltrate. Vanadium was also shown to induce the expression of inflammatory interleukins (ILs), such as IL-6 and tumor necrosis factor-α 51,52 and also promotes the expression of IL-8 in human bronchial epithelial cells exposed to residual oil fly ash–containing vanadium. 53
Conclusions
In recent years, there has been a great interest in studying the effects of air pollutants on health, particularly on reproductive health, due to the increase in the incidence of infertility cases. The studies about reprotoxicity are mainly focused on the effect pollution has on the male reproductive system, and few reports are found in the literature about the effects of air pollution in the female reproductive system. The data obtained in this study indicate that the exposure to air pollutants has a direct and evident effect on the female reproductive organs, which could cause fertility problems due to several mechanisms summarized in Figure 11.
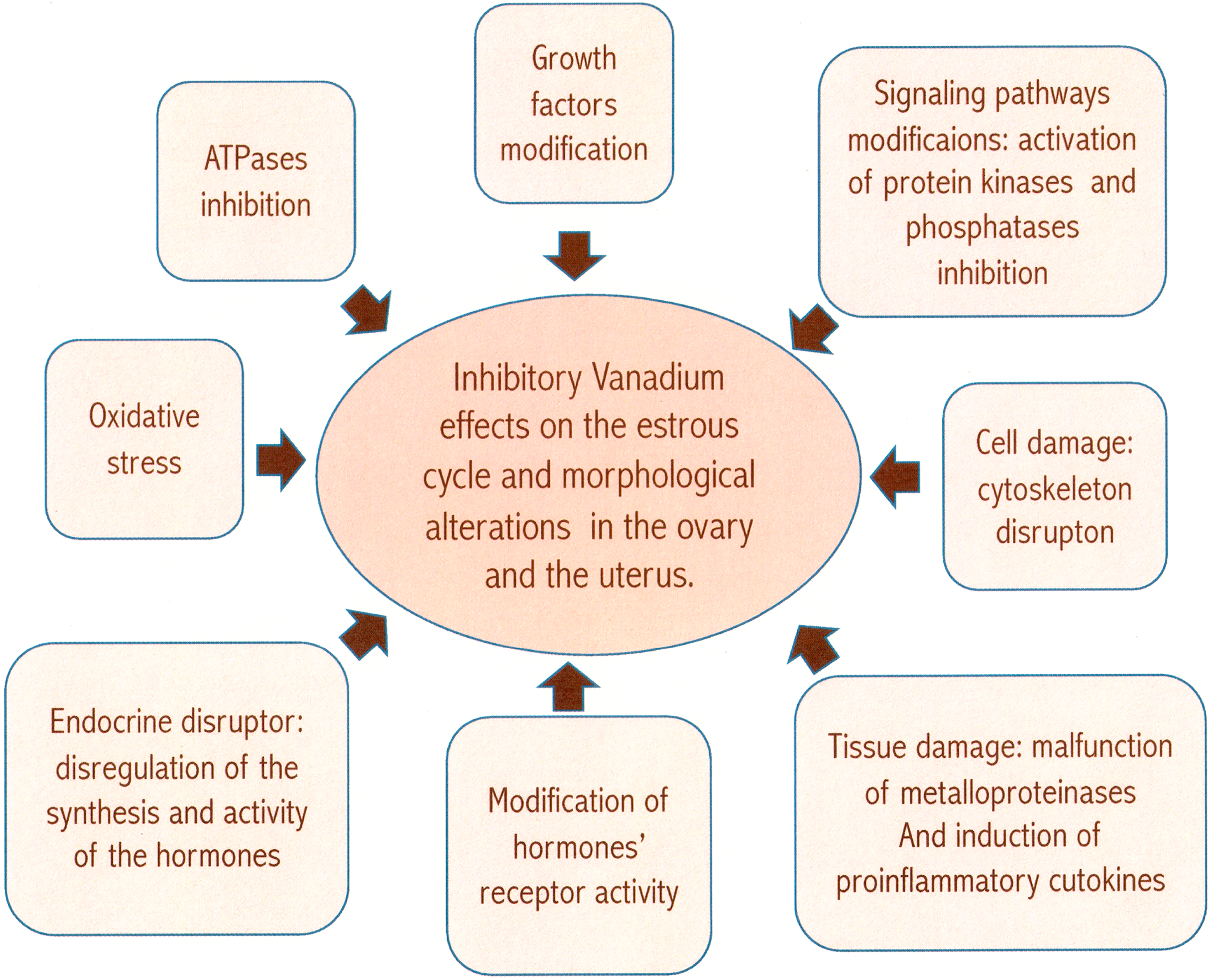
The proposed mechanisms to explain the effect of inhaled vanadium on the reproductive system of the female mice.
Footnotes
Acknowledgments
The authors thank Raquel Guerrero-Alquicira and Verónica Rodríguez-Mata, Departamento de Biología Celular y Tisular, Facultad de Medicina, UNAM, for the histological tissue processing, also Armando Zepeda-Rodríguez and Francisco Pasos-Nájera for artwork with the figures. The authors also appreciate the technical assistance of Enrique Pinzón-Estrada and Ismael Torres-Saldaña, members of the vivarium staff of the Facultad de Medicina, UNAM. Alejandra Núñez-Fortoul edited the English of the final version of the manuscript.
Author Contributions
N.M-.G. substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation of data. F.G-.I. contributed to conception and design, contributed to acquisition. P.B-.N., N.L-.V., A.G-.V., T.I.F. contributed to conception and design, contributed to acquisition, analysis, and interpretation. M.E.A-.E and F.G-.V. contributed to design, contributed to acquisition and analysis. M.R-.L. contributed to interpretation. All authors drafted manuscript, critically revised manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Nelly López-Valdez was supported by the CONACyT scholarship (CVU 346684).
