Abstract
Mifepristone, which is an orally active synthetic steroid with antiprogesterone activity, is known as an ovarian toxicant. Because the available data regarding the histopathologic characteristics of ovarian toxicity in nonhuman primates are limited, the present study was undertaken in order to investigate detailed histopathologic changes accompanying mifepristone-induced ovarian toxicity and its relationship to changes in menstrual cycle and circulating sex steroid hormone. Twenty mg/kg of mifepristone was orally administered daily to 4 cynomolgus monkeys for 2 months. Mifepristone inhibited the cyclic increases in circulating estradiol-17β and progesterone levels with associated absence of menstruation. Histopathologically, the ovary in the treated animals showed follicular phase without changes in the percentage of atretic antral follicles, and reduced endometrial thickness was noted in the uterus. These changes indicated that a certain degree of antral follicle development had been retained in spite of the menstrual cycle having been arrested in mifepristone-treated animals. Our investigation suggested that it is important to perform detailed histopathologic examination of reproductive organs with precise knowledge of the characteristics of each menstrual stage to detect ovarian toxicity in nonhuman primates. Monitoring menstrual signs and circulating sex steroid hormone levels provides additional evidence for the investigation of the mechanism of ovarian toxicity.
Introduction
Numerous drugs and chemicals are known to interfere with female reproductive function (Yuan and Foley 2002). The female reproductive organs are regulated by the hypothalamic–pituitary–gonadal (HPG) system through a complex feedback loop across the menstrual cycle. For female reproductive safety assessment in preclinical toxicity studies, nonhuman primates provide significant information compared to other species, since their ovarian cycle shows close similarities to that in women. Unlike rodents, primates have a comparatively long life span of the corpus luteum (CL; about 2 weeks), prolactin does not play a definitive role during the luteal phase, and luteolysis does not involve a signal of prostaglandin F2α from the uterus (Weinbauer et al. 2008).
In rodents, toxicity data on the female reproductive organs are abundant, and useful practical guidances for histopathologic examination have been issued (Westwood 2008; Sanbuissho et al. 2009; Yoshida et al. 2009). In nonhuman primates, however, limited literature to support the evaluation of female reproductive organ (ovarian) toxicity are available (Watanabe et al. 2006; Weinbauer et al. 2008; Sato, Nasu, and Tsuchitani 2016), and more investigation is required.
Mifepristone, which is an orally active, synthetic steroid with antiprogesterone and antiglucocorticoid activities, is used for early termination of pregnancy and is known as an ovarian toxicant (Sitruk-Ware 2006; Tamura et al. 2009). Historically, in rats, there were frequent estrus and histopathologic changes in the ovaries, including unruptured luteinized follicles and luteinized cysts, which indicated ovulatory failure in 2-week and 4-week studies (Tamura et al. 2009). In nonhuman primates, progesterone (P4) antagonist induced a decrease in serum P4, suppression of menstruation, and atrophy of endometrium following 4-week or 6-month treatment, but limited information is available regarding the histopathologic changes of reproductive organs (Deraedt, Vannier, and Fournex 1985; Slayden and Brenner 2004).
The present study evaluated the detailed histopathologic characteristics of mifepristone-induced ovarian toxicity in monkeys with close monitoring of menstrual signs and circulating sex steroid hormone levels.
Materials and Method
Animals
Female, 4-year-old, cynomolgus monkeys were purchased from Shin Nippon Biomedical Laboratories, Ltd., Kagoshima, Japan (Breeding site: Angkor Primates Center, Inc., Kingdom of Cambodia). Animals were individually housed in a stainless steel cage, and environmental conditions were maintained at a temperature of 20–26°C, a relative humidity of 35 to 75%, with a 12-h light cycle. The study protocol was approved by the Laboratory Animal Care and Use Committee and was performed in compliance with the Laboratory Animal Policy of Eisai Co., Ltd.
Chemicals and Reagents
Mifepristone was purchased from Tokyo Chemical Industry Co., Ltd. (M1732; Tokyo, Japan) and was suspended in 0.5w/v% methyl cellulose 400 solution (Wako Pure Chemical Industries, Ltd., Osaka, Japan) for a dose volume of 5 mL/kg body weight.
Experimental Design
Twenty mg/kg mifepristone was administered to 4 animals, once per day for 2 months (54 days) via oral gavage. The effective dose on the female reproductive system was selected based on the relevant literature (Deraedt, Vannier, and Fournex 1985; Danforth et al. 1989; Sitruk-Ware and Spitz 2003). The 3 control animals received an equivalent volume of vehicle solution. General condition, including menstruation, was checked for all animals daily, and body weight was measured once a week during the experimental period. Menstruation was confirmed by menstrual bleeding of the perineal area or menstrual blood on the pan. Prior to beginning the study from days −125 to −1, menstrual blood on the pan under the cage was checked and noted in most of the animals including all mifepristone-treated animals (animal no. 101, days −34 to −33; animal no. 102, none; animal no. 103, none; animal no. 201, days −124 to −122, −93 to −90, and −64 to −62; animal no. 202, days −85 to −83 and −48 to −45; animal no. 203, days −112 to −111; animal no. 204, days −124 to −122 and −51 to −46). Blood samples (approximately 2 mL) for the hormone measurement were collected from a femoral vein without anesthesia in all animals 3 times a week during the dosing period. Serum samples were obtained after centrifugation (3,000 rpm, 4°C, 10 min) and stored at −15°C until the analysis. Clinical pathology examination (hematology and blood chemistry) was performed for all animals on day −1 and day 55 of the dosing period. On the day following the last treatment (day 55), animals were euthanized by exsanguination from the cephalic vein, while under sodium pentobarbital anesthesia, and necropsied. The ovary, uterus, vagina, pituitary, adrenal, and mammary gland were fixed in 10% neutral-buffered formalin, routinely processed with hematoxylin and eosin staining, and histopathologically examined. Both sides of each ovary were transversely dissected into 12 different sections for the detailed histologic examination. The number of antral follicles and antral atretic follicles were manually counted in each section. Follicle with antral cavity was defined as antral follicle, and antral atretic follicle was characterized by nuclear pyknosis in granulosa layers often accompanied by oocyte involution and irregular shape of the antral follicle or oocyte (Gougeon 1996). Histologic classification of the menstrual stages was based on well-established descriptions in the literature for female reproductive organs of nonhuman primates (Koering 1969; Watanabe et al. 2006).
Hormone Measurement with Mass-spectrometry System
Concentrations of estradiol-17β (E2) and P4 in serum samples were measured using a liquid chromatograph coupled with a tandem mass spectrometer (LC-MS/MS) system. Briefly, 200 μL of plasma was diluted with 400 μL of 5% methanol followed by adding 5 μL of the internal standard (deuterated P4 [50 ng/mL]; deuterated E2 [2 ng/mL]), then the steroids were extracted by mixing with a vortex. The mixture was purified by solid phase extraction using an (SPE) column and Oasis® HLB 1 cc (30 mg) SPE cartridges. After washing with 1 mL of 30% methanol, the steroids were eluted sequentially with 0.5 mL of methanol followed by 0.5 mL of 50% ethyl acetate in hexane. The solution, including extracted steroids, was vacuum-dried and resuspended in 35 μL of dancyl chloride solution (2 mg/mL in acetonitrile) followed by the addition of 65 μL of 10 mM sodium bicarbonate aqueous solution (pH 9.4). Then, the sample solution was incubated for 30 min at 60°C to derivatize E2. Following the derivatization steps, the sample solutions were directed to LC-MS/MS analysis (50 μL injection volume). The LC-MS/MS system consists of an ultra performance liquid chromatography (ACQUITY UPLC; Waters Corporation, Tokyo, Japan) with an L-column (C18 5 μm, 2.6 × 150 mm; Chemicals Evaluation and Research Institute, Tokyo, Japan) at a flow rate of 0.1 mL/min (50°C) and triple quadrupole tandem mass spectrometer (Xevo TQ MS; Waters Corporation, Tokyo, Japan). The mass spectrometer was operated in ESI positive-ion mode, and detection of the ions was performed in multiple reaction monitoring mode. Water was used as an artificial matrix to prepare calibration samples for generating calibration curves. As internal standards, 17β-Estradiol-16,16,17-d3 (Toronto Research Chemicals Inc., Ontario, Canada) was used for E2, and progesterone-d9 (Toronto Research Chemicals Inc., Ontario, Canada) was used for P4. The quantification range for each steroid was as follows: E2, 6 to 6,000 pg/mL; P4, 2 to 2,000 ng/mL.
Statistical Analysis
Differences in the number of antral follicles and percentage of atresia in antral follicles were evaluated between the control and the treatment groups by the Mann–Whitney U test. The level of significance was set at 0.01.
Results
General Observations and Clinical Pathology
There was no mortality or changes in body weight in any animal throughout the experimental period (data not shown). In clinical signs, menstruation was noted in all control animals once or twice during the dosing period (animal no. 101, days 3 to 4 and 41 to 42; animal no. 102, days 13 to 14; animal no. 103, days 23 to 24), whereas no menstruation was observed in any mifepristone-treated animal. In hematology and blood chemistry, there were no changes in any of the parameters following repeated administration (day 55) compared to values at pretest (day −1), values in control group, or background data of the facility (Online Supplementary Tables 1 and 2).
Time Course Changes of Circulating E2 and P4 Levels
Figure 1 shows the time course changes in circulating E2 and P4 levels for each animal. All 3 control animals exhibited a regular cyclic pattern of E2 and P4, which was characterized by periodic E2 surges and subsequent sustained elevation of P4 levels for approximately 2 weeks. In the 3 mifepristone-treated animals (animal nos. 201, 202, and 203), periodic E2 surges and the subsequent P4 elevation completely disappeared and levels remained at a basal or low during the treatment period. In another animal (animal no. 204), although P4 elevation, shortly after a rapid fall in high E2 level was observed, its duration (9 days) was shorter than that of controls (13 to 15 days).
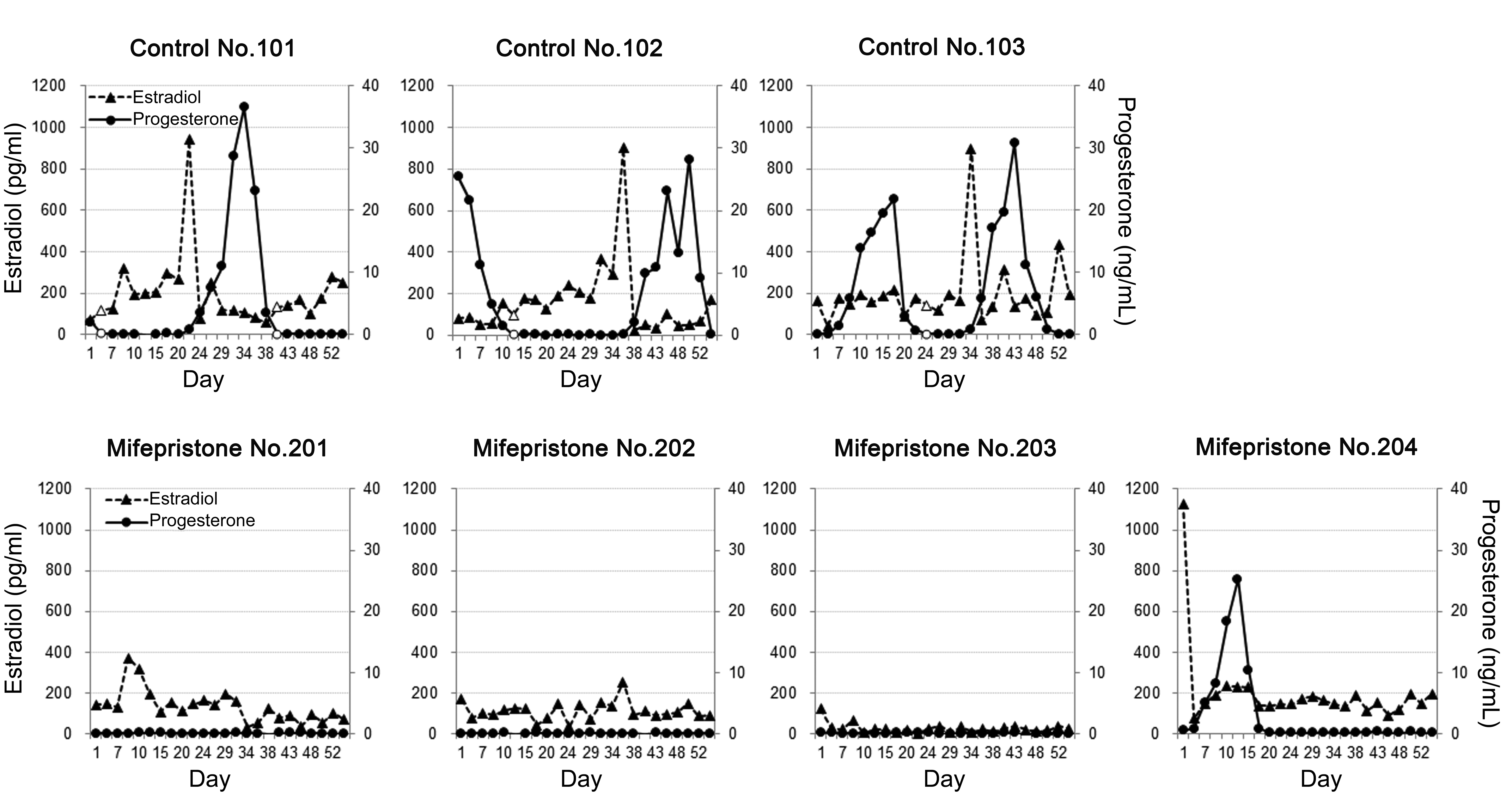
Time course changes of circulating estradiol-17β (E2) and progesterone (P4) levels in each animal during the treatment period. Triangle plots with dotted line show E2 and circled plots with solid line show P4. White plots showed the period of menstruation.
Histopathologic Changes in the Female Reproductive Organs
Histopathologic findings in the ovaries and uterus are shown in Figures 2 and 3, respectively. The 3 control animals showed different phases of menstrual cycle. In all phases of the ovaries, there were various stages of small, growing, antral, and atretic follicles and old corpus luteum (OC), which was characterized by dense nucleus, occasional cytoplasmic pigment, and obvious collagen between luteal cells (Figure 2g). Animal no. 101 was in the late follicular phase; one side of the ovary had a dominant large antral follicle (LF; Figures 2a and 2b). The endometrium thickened with abundant stroma and proliferating glandular cells formed a straight glandular structure (Figure 3a). Animal no. 102 was in the menstrual phase; a regressive corpus luteum (RC) with small vacuoles in lutein cells was present on one side of the ovary (Figures 2c and 2d). Hemorrhage and shedding of the surface of endometrial layer were present in the uterus (Figure 3b). Animal no. 103 was in the early follicular phase; one side of the ovary contained a RC with coarse vacuoles in lutein cells (Figures 2e and 2f). The uterus exhibited thin and poorly developed glandular structure with shedding of superficial endometrium (Figure 3c). In the ovary of all mifepristone-treated animals, there were no LF, RC, or functional corpus luteum, but there were various small and antral follicles and OC, corresponding to the follicular phase (Figures 2g and 2h). In the uterus, endometrial thickness was greatly reduced due to the absence of functional layer of the endometrium with stromal compaction and lack of proliferation of the gland in 3 of the 4 animals (animal nos. 201, 202, and 204; Figure 3d). The uterus of the other animal (animal no. 203) showed an early follicular phase with the same histologic characteristics as control animal no.103. The number of antral follicles and percentage of atretic antral follicles are shown in Table 1. The number of antral follicles was significantly higher in the mifepristone group compared to the controls. There was no difference in the percentage of atretic antral follicles between the control and mifepristone groups. There were no histopathologically noteworthy changes in the vagina, pituitary, adrenal, and mammary glands in any animal.
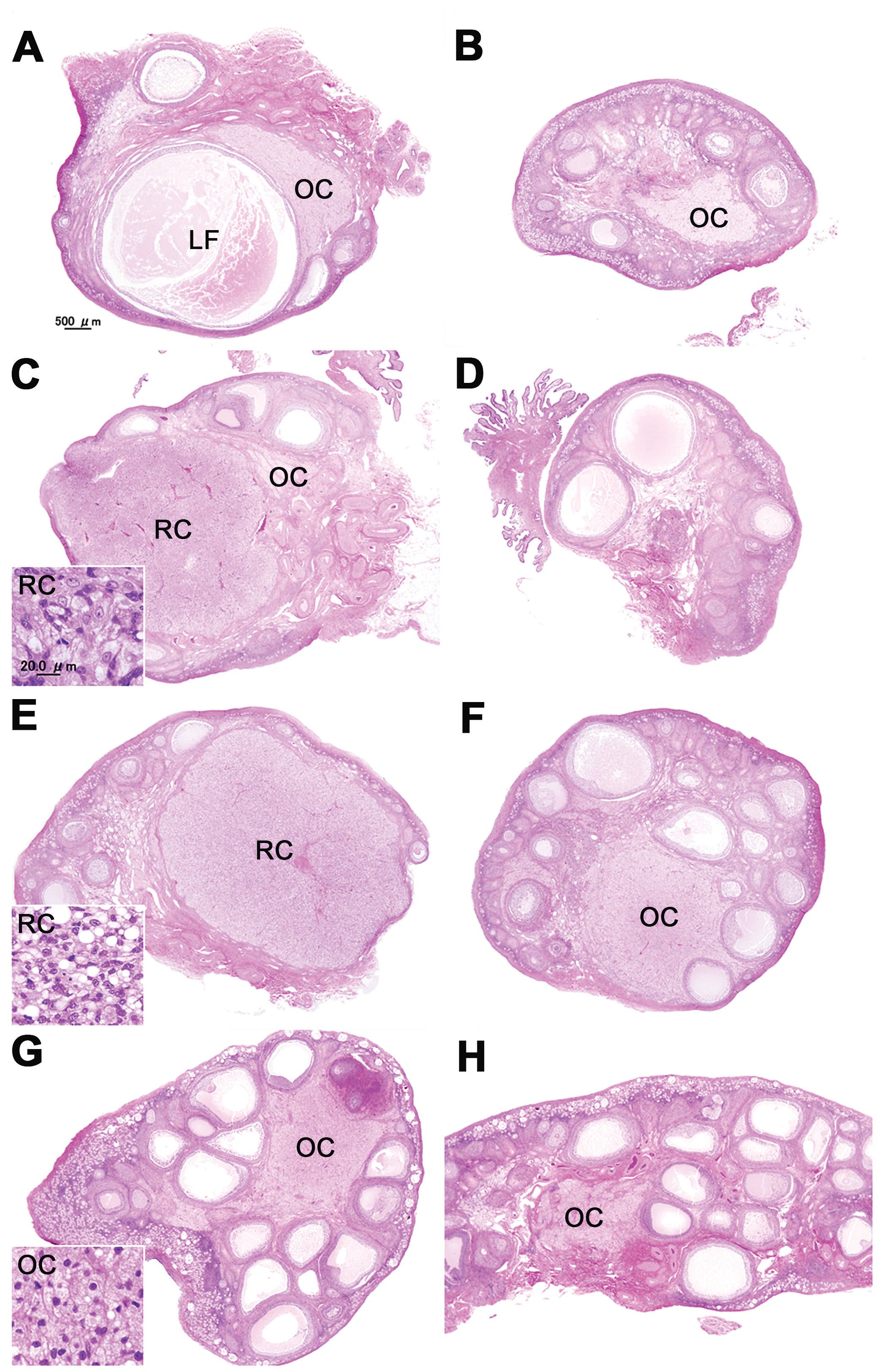
Histologic characteristics of the ovaries in the control (a, b, c, d, e, and f) and mifepristone-treated animals (g and h). In all menstrual phases, there were various stages of small, growing, antral, and atretic follicles and old corpus luteum (OC). In the late follicular phase, a dominant large antral follicle (LF) was unilaterally present (a and b). The ovaries of menstrual phase showed the regressive corpus luteum (RC) with small vacuoles in lutein cells on one side (c and d). In the early follicular phase, the RC was unilaterally noticed with coarse vacuoles in lutein cells (e and f). The ovaries of mifepristone-treated animals showed follicular phase, which was no LF, RC, or functional corpus luteum with various small and antral follicles and OC (g and h). Hematoxylin and eosin (HE) staining.
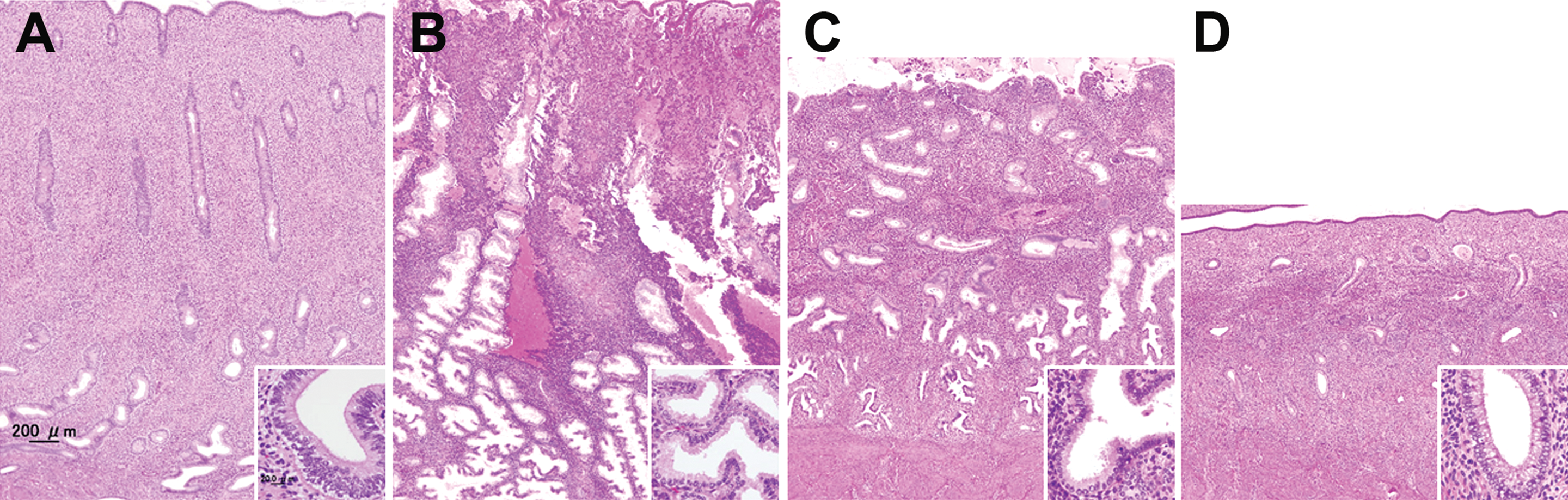
Histologic characteristics of the uterus in the control (a, b, and c) and mifepristone-treated animals (d). In the late follicular phase, the endometrium thickened with swollen and proliferating glandular cells in the uterus (a). The uterus of menstrual phase showed hemorrhage and shedding of the surface of endometrial layer (b). In the early follicular phase, thin and poorly developed glandular structure with shedding of superficial endometrium was observed (c). Mifepristone treatment resulted in greatly reduced endometrial thickness with the absence of functional layer of the endometrium, increase in stromal compaction, and lack of proliferation of the gland (d). Inset pictures showed high-power images of the endometrial glands. Hematoxylin and eosin (HE) staining.
Parameters of Antral Follicles.
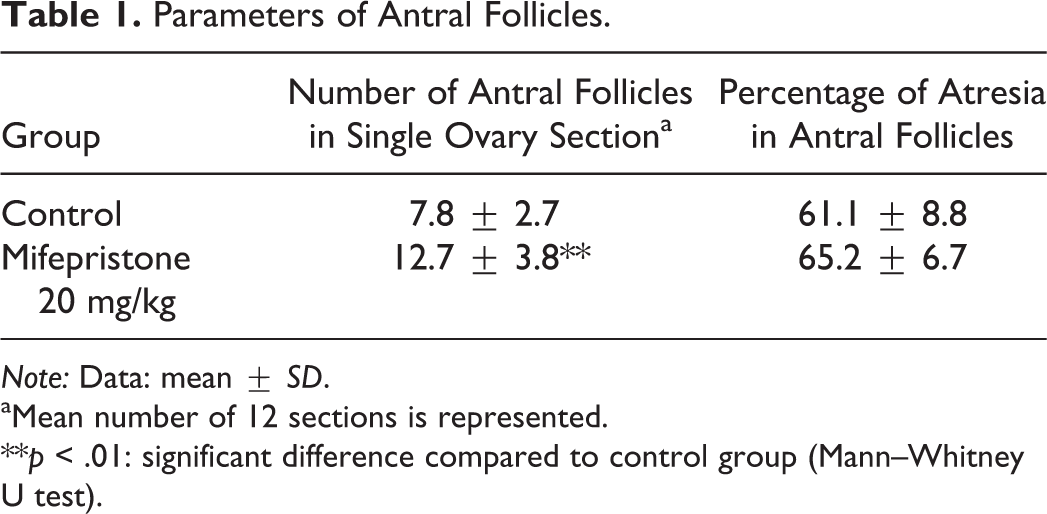
Note: Data: mean ± SD.
aMean number of 12 sections is represented.
**p < .01: significant difference compared to control group (Mann–Whitney U test).
Discussion
Absence of menstruation and hormonal disruption of E2 surge and P4 elevation by mifepristone treatment indicated that mifepristone arrested the menstrual cycle in nonhuman primates, which was consistent with previous reports (Danforth et al. 1989; Remohi et al. 1988; Sitruk-Ware and Spitz 2003). The LC-MS/MS plasma analysis elucidated the time course of circulating E2 and P4 levels in monkeys as previously reported in other experimental animals and humans (Keski-Rahkonen et al. 2011; Koal et al. 2012; Riffle, Henderson, and Laws 2013).
All mifepristone-treated animals showed histologic characteristics of the ovaries consistent with the follicular phase of normal ovaries, without affecting the ratio of atretic antral follicles, despite the uterus showing reduced endometrial thickness. This result indicated that a certain degree of antral follicle development had been retained during the dosing period in spite of the menstrual cycle having been arrested. In monovular species, including monkeys, selection of the dominant follicle, which develops to LF and proceeds to ovulation, occurs once in the early- to mid-follicular phases (Weinbauer et al. 2008; Baerwald, Adams, and Pierson 2012). Since LF was not observed in any mifepristone-treated animal, mifepristone was thought to inhibit the dominant follicle selection or development in monkeys. Regarding the ratio of atretic antral follicles, insufficient or absence of follicle-stimulating hormone (FSH) is said to induce follicular atresia in monkeys (Gougeon 1996). Since there was no effect on atresia of antral follicles in the present study with 2-month treatment, it is speculated that a certain level of FSH seemed to have been sustained throughout the dosing period as in the previous reports (Danforth et al. 1989; Remohi et al. 1988). In nonhuman primates, there is a large individual variation in the number of antral follicles (3 to 22 follicles/ovary) depending on various factors including age, menstrual stage, and ovarian size (Morgan et al. 1987; Nichols et al. 2005; Appt et al. 2006; Bishop et al. 2009). It is considered that the difference in the number of antral follicles between control and mifepristone groups is within the range of individual variation. In rodents, mifepristone inhibited the ovulatory process and induced unruptured, luteinized follicles and luteinized cysts with increase in large atretic follicles (Gaytan et al. 2003; Tamura et al. 2009). These findings are due to the effects of mifepristone on HPG axis in rodents which increased basal LH level, stimulated P4 production before LH surge, and decreased in proestrus LH surge (Curry and Nothnick 1996; Tamura et al. 2009). Differences in the histopathologic features of ovaries in monkeys and rodents were considered to be due to the different effects of mifepristone on the HPG axis or the difference on the control of the reproductive organs during menstrual or estrous cycle in each species (Danforth et al. 1989; Remohi et al. 1988; Weinbauer et al. 2008).
In the uterus, mifepristone treatment resulted in the absence of functional layer of the endometrium. This antiproliferative effect of mifepristone on endometrium was due to the inhibition of E2 and P4, which have critical roles for endometrial growth, differentiation, and proliferation (Slayden and Brenner 2004). Rodents did not show the endometrial antiproliferative effects of P4 antagonists, rather exhibited endometrial hyperplasia due to E2 effect (Chwalisz et al. 2000). In addition to the different effects of mifepristone on the HPG axis, the difference in uterine response between monkeys and rodents was probably caused by the different role of P4 and P4 receptor in controlling the E2 responsiveness of uterine epithelium and the different endometrial physiology, such as presence or absence of periodic bleeding and spiral arteries, different type of endometrial vascular bed, or different thickness of myometrial walls (Chwalisz et al. 2000).
For the detection of ovarian toxicity in nonhuman primates, several evaluation guides are available (Watanabe et al. 2006; Weinbauer et al. 2008; Sato, Nasu, and Tsuchitani 2016). They had 5 recommendations: (1) examine the largest cross sections of ovaries containing CL or large follicles, (2) measure the circulating sex hormone levels, especially E2 and P4, since they are the main regulators of the primate ovarian cycle, (3) monitor menstrual signs and select animals with regular menstrual cycles, (4) conduct comparatively long-term studies, and (5) start dosing at a fixed menstrual phase. The present study mainly focused on points 1 and 2 with some consideration of point 3. The ovaries and uterus of mifepristone-treated animals were histologically inconsistent with regular menstrual stages, with the absence of menstruation and disruption of cyclic sex hormonal pattern. These results suggest the importance of detailed histopathologic examination of reproductive organs, including bilateral examination of ovaries coupled with understanding the histologic characteristics of each menstrual stage. In addition, the time course analysis of circulating E2 and P4 provides evidence and useful data for investigating the mechanism of ovarian toxicity in nonhuman primates. For the monitoring of menstrual cycle, daily assessment of vaginal smears is known to provide a convenient, reliable, and minimally invasive approach (Weinbauer et al. 2008). More reliable and appropriate methods for monitoring the menstrual cycle including taking vaginal smears will be considered in the further experiments. In the human, mifepristone induced a fall in E2 with a regression of the dominant follicle and induced inhibition of glandular secretory activity, degenerative changes in glandular cells, reduction of stromal edema, and stromal extravasation in the uterus (Sitruk-Ware and Spitz 2003). These changes are consistent with the present results in monkeys and nonhuman primates considered to be an appropriate species for ovarian toxicity evaluation of mifepristone. The present mifepristone, ovarian toxicity was detected and histologically characterized following 2-month dosing in 4-year-old monkeys. Compared to rodents, it seems to take a longer dosing period in order to detect ovarian toxicity in nonhuman primates, since they have a substantially longer menstrual/estrous cycle interval than rodents, and most ovarian toxicants are known to require at least several cycles to detect the toxicity with accuracy (Sanbuissho et al. 2009; Yoshida et al. 2009). Additional evidence and examinations are needed during an appropriate dosing period for ovarian toxicity evaluation in nonhuman primates.
In conclusion, mifepristone-treated monkeys showed follicular phase in the ovaries and reduced endometrial thickness with the absence of menstruation and disruption of cyclic E2 and P4 elevation patterns. These data highlighted the importance of a detailed histopathologic examination of female reproductive organs with precise knowledge of the characteristics of each menstrual stage for ovarian toxicity evaluation in nonhuman primates. In addition, monitoring menstrual signs and circulating sex steroid hormone analysis provide the evidence to investigate the mechanism of ovarian toxicity.
Supplemental Material
Supplemental Material, DS1_TPX_10.1177_0192623318763586 - Histopathologic Characterization of Mifepristone-induced Ovarian Toxicity in Cynomolgus Monkeys
Supplemental Material, DS1_TPX_10.1177_0192623318763586 for Histopathologic Characterization of Mifepristone-induced Ovarian Toxicity in Cynomolgus Monkeys by Yoshikazu Taketa, Kanta Horie, Tetsuya Goto, Etsuko Ohta, Kyoko Nakano-Ito, Kazuhiro Hayakawa, Yuki Seki, Aya Goto, and Satoru Hosokawa in Toxicologic Pathology
Supplemental Material
Supplemental Material, DS2_TPX_10.1177_0192623318763586 - Histopathologic Characterization of Mifepristone-induced Ovarian Toxicity in Cynomolgus Monkeys
Supplemental Material, DS2_TPX_10.1177_0192623318763586 for Histopathologic Characterization of Mifepristone-induced Ovarian Toxicity in Cynomolgus Monkeys by Yoshikazu Taketa, Kanta Horie, Tetsuya Goto, Etsuko Ohta, Kyoko Nakano-Ito, Kazuhiro Hayakawa, Yuki Seki, Aya Goto, and Satoru Hosokawa in Toxicologic Pathology
Footnotes
Acknowledgments
I would like to deeply thank Mr. Hideki Sakurai for the contribution of HPLC–MS/MS hormone analysis and Ms. Michiyo Shimada, Mr. Takaya Shimomura, Mr. Katsuya Enjyoji, Mr. Yuki Tanaka, Mr. Kazuhiro Someya, and Mr. Shota Yokota (Sunplanet Co., Ltd.) for their excellent technical assistance. I also appreciate Ms. Kathy Vanderhoof (Eisai, Inc.) for her critical review and helpful comments.
Authors’ Contribution
Authors contributed to conception or design (TY, HKan, GT, OE, NK, HKaz, SY, GA, HS); data acquisition, analysis, or interpretation (TY, HKan, GT, OE, NK, HKaz, SY, GA, HS); drafting the manuscript (TY, HK, GT, OE, NK); and critically revising the manuscript (TY, HKan, OE, NK, HKaz, SY, GA, HS). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
