Abstract
Hepatic macrophages play crucial roles in hepatotoxicity. We investigated immunophenotypes of macrophages in liver injury induced in rats by thioacetamide (TAA; 300 mg/kg, intraperitoneal) after hepatic macrophage depletion; hepatic macrophages were depleted by liposomal clodronate (CLD; 10 ml/kg, i.v.) one day before TAA injection. Samples were obtained on post-TAA injection days 0, 1, 2, 3, 5, and 7. TAA injection induced coagulation necrosis of hepatocytes on days 1 through 3 and subsequent reparative fibrosis on days 5 and 7 in the centrilobular area, accompanied by increased numbers of M1 macrophages (expressing cluster of differentiation [CD]68 and major histocompatibility complex class II) and M2 macrophages (expressing CD163 and CD204) mainly on days 1 through 3. TAA + CLD treatment markedly decreased the numbers of M1 and M2 macrophages mainly on days 1 through 3; CD163+ Kupffer cells were most sensitive to CLD depletion. In TAA + CLD–treated rats, interestingly, coagulation necrosis of hepatocytes was prolonged with more increased levels of hepatic enzymes (aspartate transaminase, alanine transaminase, and alkaline phosphatase) to TAA-treated rats; reparative fibrosis was incomplete and replaced by dystrophic calcification in the injured area, indicating the aggravated damage. Furthermore, in TAA + CLD–treated rats, inflammatory factors (monocyte chemoattractant protein [MCP]-1, interferon-γ, tumor necrosis factor-α, and interleukin-10) and fibrosis-related factors (transforming growth factor-β1, matrix metalloproteinase-2, tissue inhibitor of metalloproteinase-1) were decreased at messenger RNA levels, indicating abnormal macrophage functions. It was clearly demonstrated that hepatic macrophages have important roles in tissue damage and remodeling in hepatotoxicity.
Introduction
Macrophages are versatile cells, playing crucial roles in regulation of inflammation and repair in various pathological settings (Fujiwara and Kobayashi 2005; Beljaars et al. 2014; Pellicoro et al. 2014; Tacke and Zimmermann 2014). In damaged tissues, they display functions such as modulation/recruitment of inflammatory cells, killing of cells, and debridement of necrotic tissues; furthermore, myofibroblasts, capable of producing extracellular matrices (ECMs), are induced by factors released from resident and infiltrating macrophages, resulting in reparative fibrosis after tissue injury (Wynn 2008; Wynn and Barron 2010; Xu et al. 2014). Immunophenotyping studies recently have demonstrated the existence of divergent types of macrophages, which could be identified by specific molecules that they express depending on microenvironmental conditions evoked by cell-to-cell or cell-to-matrix interactions. For example, the phagolysosomal membrane protein, cluster of differentiation (CD)68, is highly expressed by infiltrating macrophages with activated phagocytosis; although CD163 and CD204 are expressed in resident macrophages (Kupffer cells in the liver) in normal rat livers, their expressions in injured tissues may imply the activation of different scavenger receptors recognized by respective CD163 and CD204; antigen-presenting macrophages such as dendritic cells express major histocompatibility complex (MHC) class II molecule not only in normal tissues but also in pathological lesions, and MHC class II molecule expression may be related to subsequent complicated immune response (Damoiseaux et al. 1994; Polfliet et al. 2006; Conrad and Dittel 2011; Kiyanagi et al. 2011; Golbar et al. 2012). Recently, macrophages are functionally classified as classically activated macrophages (M1) and alternatively activated M2 macrophages (Sica and Mantovani 2012; Martinez and Gordon 2014; Ong et al. 2015). Macrophages expressing CD68 and CD163 in injured areas are regarded as representative M1 and M2 types, respectively (McGuinness et al. 2000; Mantovani et al. 2002; Wijesundera et al. 2014).
Previously, we showed that infiltrating macrophages had central roles in tissue injury and subsequent fibrosis in thioacetamide (TAA)-induced rat hepatic lesions; the lesions at early stages are characterized by coagulation necrosis and infiltration of a lot of macrophages with heterogeneous antigens in the centrilobular area; at mid and late stages, the damaged areas were replaced by reparative fibrosis (Wijesundera et al. 2014). We hypothesized that the hepatic lesions induced in rats by TAA may be altered in the absence of resident and infiltrating macrophages. To further clarify the functional roles of these macrophages, we investigated the immunophenotypes of macrophages and subsequent reparative fibrosis in TAA-induced hepatic lesions of macrophage-depleted rats; hepatic macrophages were depleted by preinjection of liposome (Lipo)-encapsulated clodronate (CLD). The encapsulated CLD causes damage to hepatic macrophages via apoptosis, resulting in their depletion (van Rooijen and Sanders 1994; van Rooijen and Hendrikx 2010). It was found, as a result, that the hepatic macrophage depletion aggravated tissue damages in TAA-induced rat hepatic lesions, indicating importance of reactive macrophages in hepatotoxicity.
Materials and Methods
Animals and Experiment
Forty-eight 5-week-old male F344 rats (100–120 g body weight) purchased from Charles River Japan (CRJ, Hino, Shiga, Japan) were used in this experiment. The rats were housed in an animal room under controlled environment at 21°C ± 3°C and with a 12-hr light–dark cycle; they had free access to standard rodent chow (DC-8; CLEA Japan, Inc., Tokyo, Japan) and tap water. After acclimatization of 1 week, the animals were randomly divided into two groups; macrophage depletion group (designated as TAA + CLD; 24 rats) and control group with normal condition (designated as TAA + Lipo; 24 rats). Lipo-encapsulated CLD is readily engulfed by macrophages, particularly leading to depletion of hepatic macrophages (van Rooijen and Sanders 1994; van Rooijen and Hendrikx 2010). A CLD suspension of 5 mg/ml (Foundation Clodronate Liposomes, Amsterdam, the Netherlands) was injected intravenously once via tail vein in TAA + CLD group at the dose of 10 ml/kg body weight; 1 day after CLD injection, 20 rats were injected intraperitoneally once with TAA (Wako Pure Chemical Industries, Osaka, Japan) at the dose of 300 mg/kg (Wijesundera et al. 2014); four rats were euthanized under deep isoflurane anesthesia on post-TAA injection days 1, 2, 3, 5 and 7, respectively, and the remaining 4 rats received physiological saline instead of TAA and sacrificed on day 0 (injection day). Rats in TAA + Lipo group received empty Lipo, instead of CLD, followed by injection of saline (on day 0) or TAA; they were sacrificed accordingly on postinjection days 1, 2, 3, 5, and 7. Blood samples were collected from abdominal aorta at necropsy and separated sera were subjected to biochemical assay for aspartate transaminase (AST), alanine transaminase (ALT), and alkaline phosphatase (ALP) by SRL Inc. (Tokyo, Japan).
The animal experiment was conducted in compliance with the institutional guidelines and with prior approval of protocol by the ethical committee of Osaka Prefecture University for the Care and Use of Experimental Animals.
Histopathology and Immunohistochemistry
Tissues from the left lateral lobe of the livers were fixed in 10% neutral-buffered formalin, Zamboni’s fixative (0.21% picric acid and 2% paraformaldehyde in 130 mM phosphate buffer, pH 7.4), and periodate-lysine-paraformaldehyde (PLP) solutions (Golbar et al. 2011). These tissues were dehydrated, embedded in paraffin, sectioned (4 μm), and stained with H&E for histopathology and von Kossa’s stain for calcium staining.
Zamboni’s solution—or PLP-fixed sections—were used for immunohistochemistry with mouse monoclonal anti-CD68, anti-CD163, anti-MHC class II, anti-CD204, anti-vimentin, anti-desmin, and anti-α-smooth muscle actin (α-SMA) antibodies. Briefly, after pretreatments (Table 1), tissue sections were treated with 3% H2O2 and 5% skimmed milk, respectively. The sections were allowed to react with primary antibody overnight at 4°C and with secondary antibody (Histofine Simple Stain MAX-PO, Nichirei, Tokyo, Japan) at room temperature for 30 min. Positive reactions were visualized with 3, 3′-diaminobenzidine (DAB substrate kit; Vector Laboratories, Burlingame, CA, USA). The sections were counterstained lightly with hematoxylin.
Primary Antibodies Used for the Immunohistochemistry.
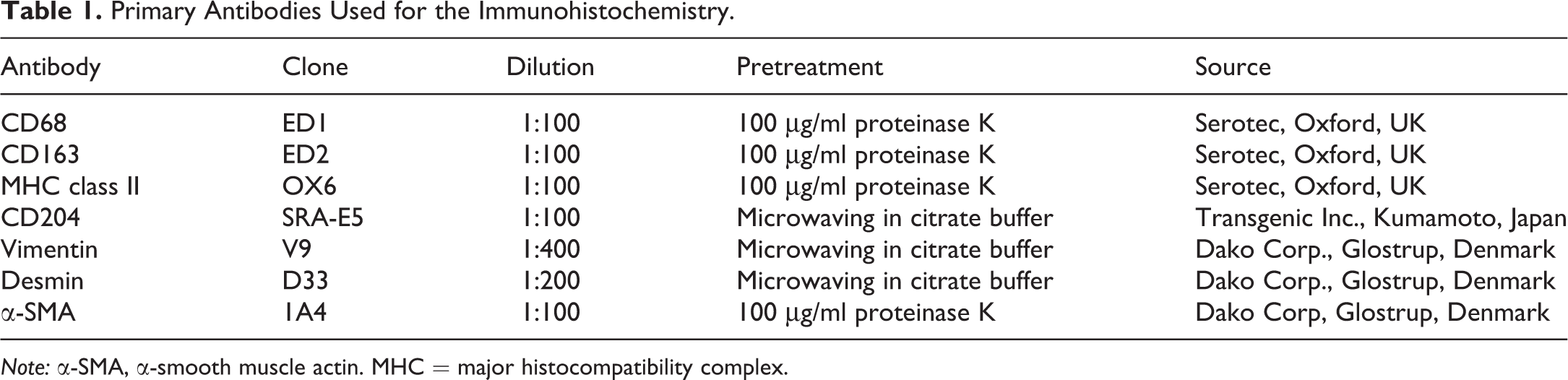
Note: α-SMA, α-smooth muscle actin. MHC = major histocompatibility complex.
The nuclei of cells immunopositive for CD163, CD68, MHC class II, and CD204 were counted at 5 randomly selected areas (0.2 mm2) of each rat at the centrilobular area of hepatic lobules at a magnification of ×400 using ImageJ software (http://rsbweb.nih.gov/ij/). Cells immunopositive for vimentin, desmin, and α-SMA in the same area were evaluated semiquantitatively (Table 2).
Semiquantitative Evaluation of Mesenchymal Cells/Myofibroblasts.
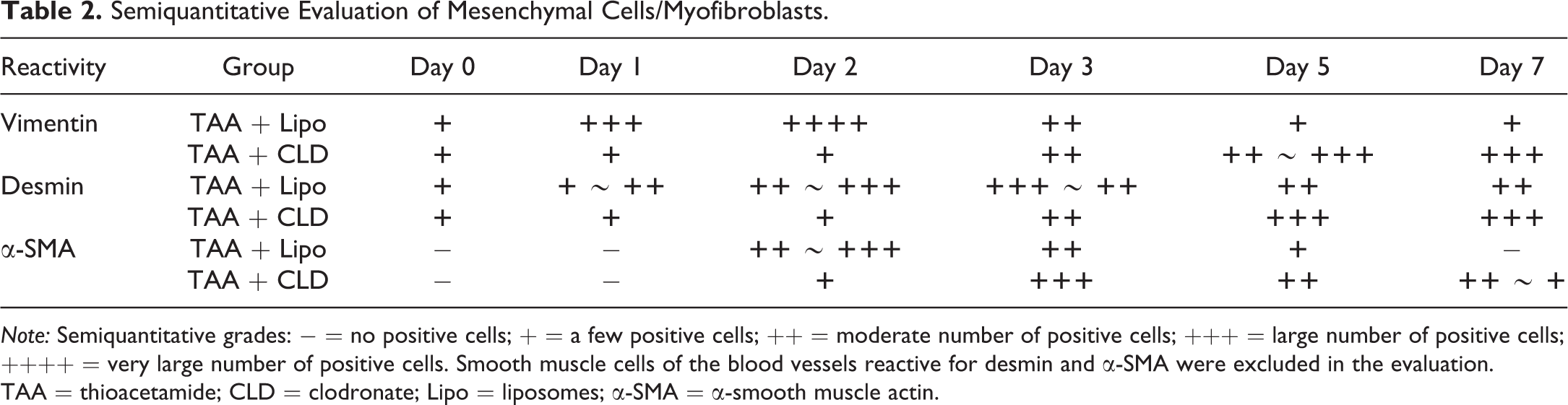
Note: Semiquantitative grades:
Reverse Transcriptase Polymerase Chain Reaction
Liver tissues from left medial lobe were immersed in RNAlater® (Qiagen GmbH, Hilden, Germany) overnight at 4°C and stored at −80°C until use. Extraction of total RNA was carried out with an SV total RNA isolation system kit® (Promega Corporation, Madison, WI, USA) per the manufacturer’s instruction and concentration of RNA was measured on a nanodrop1000™ spectrophotometer (Thermo Scientific, Wilmington, DE, USA). One microgram of RNA was reverse transcribed to complementary DNA with the SuperScript®VILOTM (Life Technologies, Carlsbad, CA, USA) and amplified for rat-specific monocyte chemoattractant protein (MCP)-1, interferon-γ, tumor necrosis factor (TNF)-α, transforming growth factor (TGF)-β1, interleukin (IL)-10, IL-4 (TaqMan assay ID: Rn01456866_m1; Life Technologies, Carlsbad, CA, USA), matrix metalloproteinase (MMP)-2, tissue inhibitor of metalloproteinase (TIMP)-1, and 18s rRNA (control gene) with the SYBR® Green Real-time PCR Master Mix (Toyobo Co. Ltd., Osaka, Japan) by a PikoReal®96 real-time PCR system (Thermo Scientific); the oligonucleotide sequences used are presented in Table 3.
Oligonucleotide Sequences Used in the Real-time PCR.
Note: PCR = polymerase chain reaction; MCP-1 = monocyte chemoattractant protein-1; IFN-γ = interferon-γ; TNF-α = tumor necrosis factor-α; TGF-β1 = transforming growth factor-β1; IL-10 = interleukin-10; MMP-2 = matrix metalloproteinase-2; TIMP-1 = tissue inhibitor of metalloproteinase-1; 18S rRNA = 18S ribosomal RNA.
Statistical Analyses
The obtained data are presented as mean ± standard deviation. Statistical analysis was performed using Tukey’s test with IBM®SPSS® grad pack 21.0 software or Student’s t-test. A P < .05 was considered significant.
Results
Serum Biochemistry and Histopathology
In TAA + Lipo group, the values of AST (Figure 1A), ALT (Figure 1B), and ALP (Figure 1C) were significantly increased on days 1 and 2, except for ALP on day 2. In TAA + CLD group, similarly, the values of AST, ALT, and ALP were significantly increased, with prolonged period on days 1, 2, and 3, and the increased levels, particularly on days 2 and 3, were significantly greater than those in TAA + Lipo group (Figure 1A–C). In addition, the value of ALP on day 0 was significantly increased in TAA + CLD group in contrast to TAA + Lipo (Figure 1C).
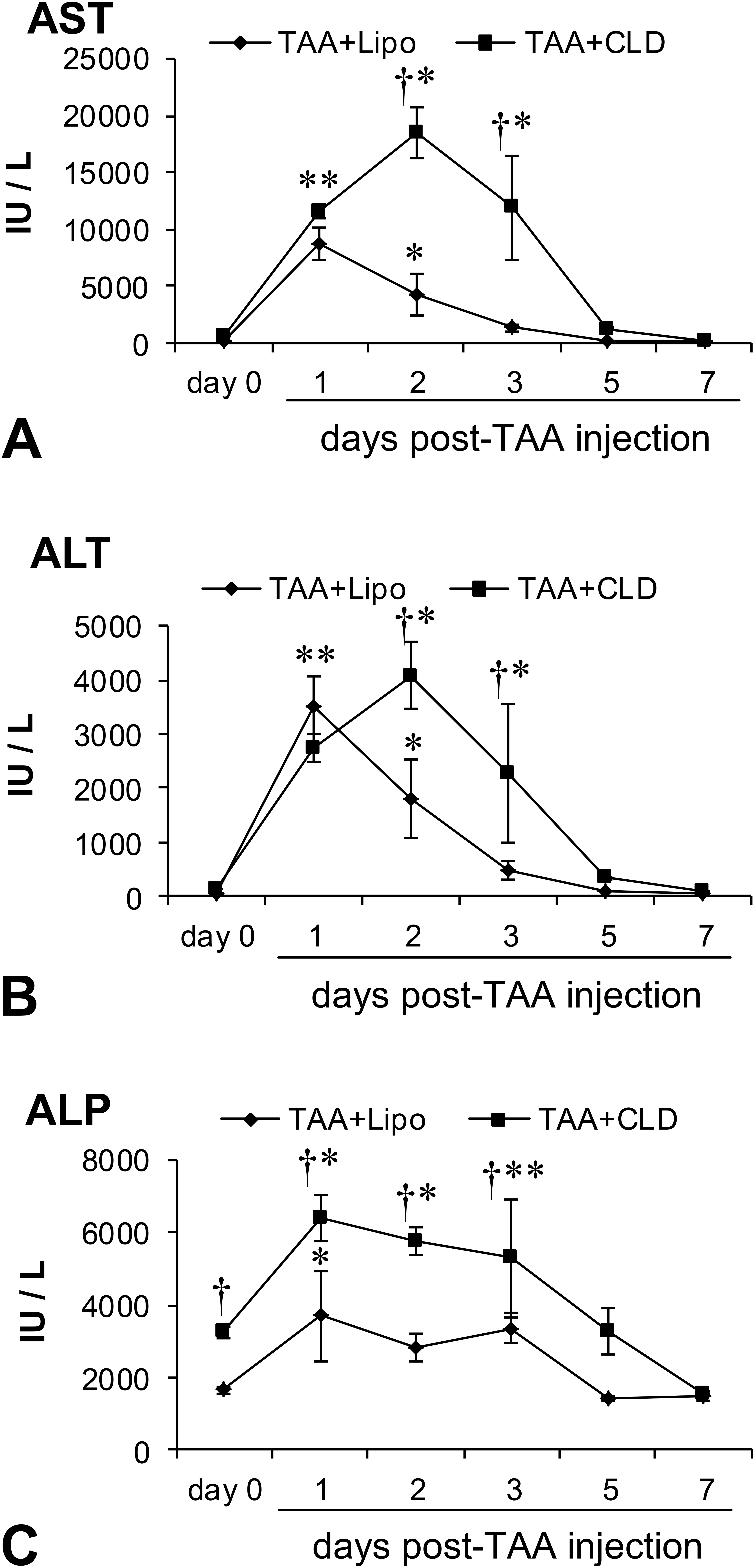
Biochemical analyses on the serum levels of aspartate transaminase (AST; A), alanine transaminase (ALT; B), and alkaline phosphatase (ALP; C) in control (TAA + Lipo) and macrophage-depleted (TAA + CLD) groups. Compared to day 0, the levels of AST and ALT are significantly increased on days 1 and 2 and ALP on days 1 and 3 in TAA + Lipo group, while AST, ALT, and ALP are increased significantly on days 1–3 in TAA + CLD group. Note that there are significant differences in AST and ALT levels on days 2 and 3 and ALP on days 0–3 between TAA + Lipo and TAA + CLD groups. Tukey’s test *P < .05, significantly different from day 0 in TAA + Lipo and TAA + CLD groups; † P < .05, significantly different between TAA + Lipo and TAA + CLD groups at respective examination points. Note: TAA = thioacetamide; CLD = clodronate; Lipo = liposomes.
Histopathologically, livers on day 0 in TAA + Lipo (Figure 2A) and TAA + CLD (Figure 2B) groups showed normal architecture similar to each other, regardless of macrophage depletion in TAA + CLD group. On day 1 after the TAA injection, coagulation necrosis developed in centrilobular areas in both TAA + Lipo and TAA + CLD groups. In TAA + Lipo group, thereafter, inflammatory cell infiltrates, mainly macrophages as mentioned below, were seen on days 2 (Figure 2C) and 3, and these cells were quickly decreased on day 5, being replaced by reparative fibrosis; on day 7, the injured centrilobular areas were recovered with regenerating hepatocytes (Figure 2E). On the other hand, in TAA + CLD group, hepatocyte injury (Figure 2D) and coagulation necrosis were continuously seen on days 5 and 7 without marked inflammatory cell reactions, indicative of prolonged damage; more interestingly, on days 5 and 7, dystrophic calcification developed in the necrotic areas (Figure 2F), which were demonstrated by the von Kossa’s stain, whereas calcification was not seen in TAA + Lipo group.
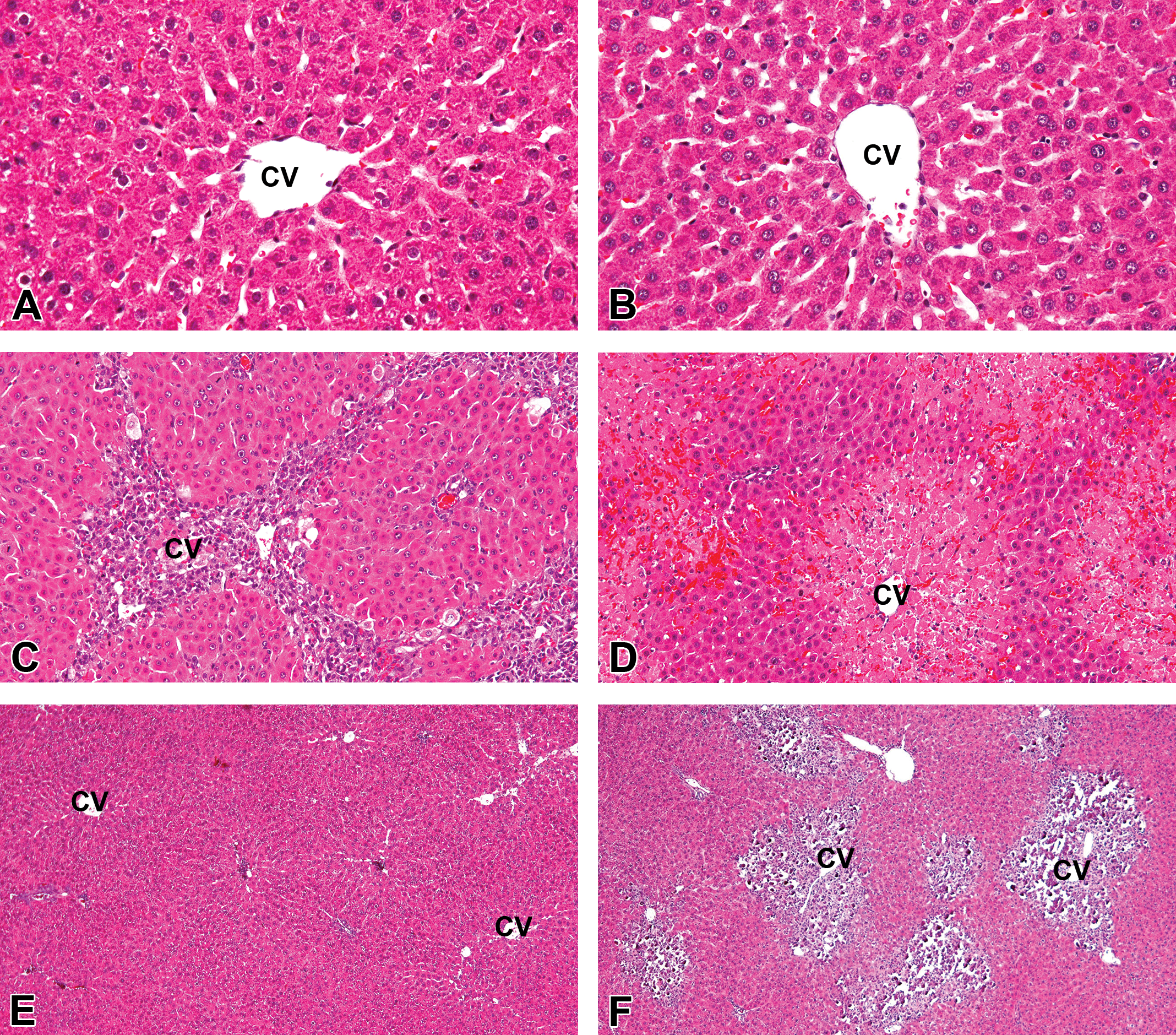
Representative liver sections showing histopathology in TAA + Lipo (A, C, and E) and TAA + CLD (B, D, and F) groups. On day 0, there are no apparent histopathological differences between TAA + Lipo (A) and TAA + CLD (B) (regardless of the hepatic macrophage depletion) groups. The inflammatory infiltrates are more severe in TAA + Lipo group (C) compared to TAA + CLD group (D) on day 2. On day 7, the injured area is almost completely recovered by regenerating hepatocytes in TAA + Lipo group (E), whereas coagulation necrosis of hepatocytes accompanied by severe dystrophic calcification is seen still in TAA + CLD group (F). CV, central vein; H&E (A–F). Note: AA = thioacetamide; CLD = clodronate; Lipo = liposomes.
Kinetics of Macrophages in TAA-induced Hepatic Lesions
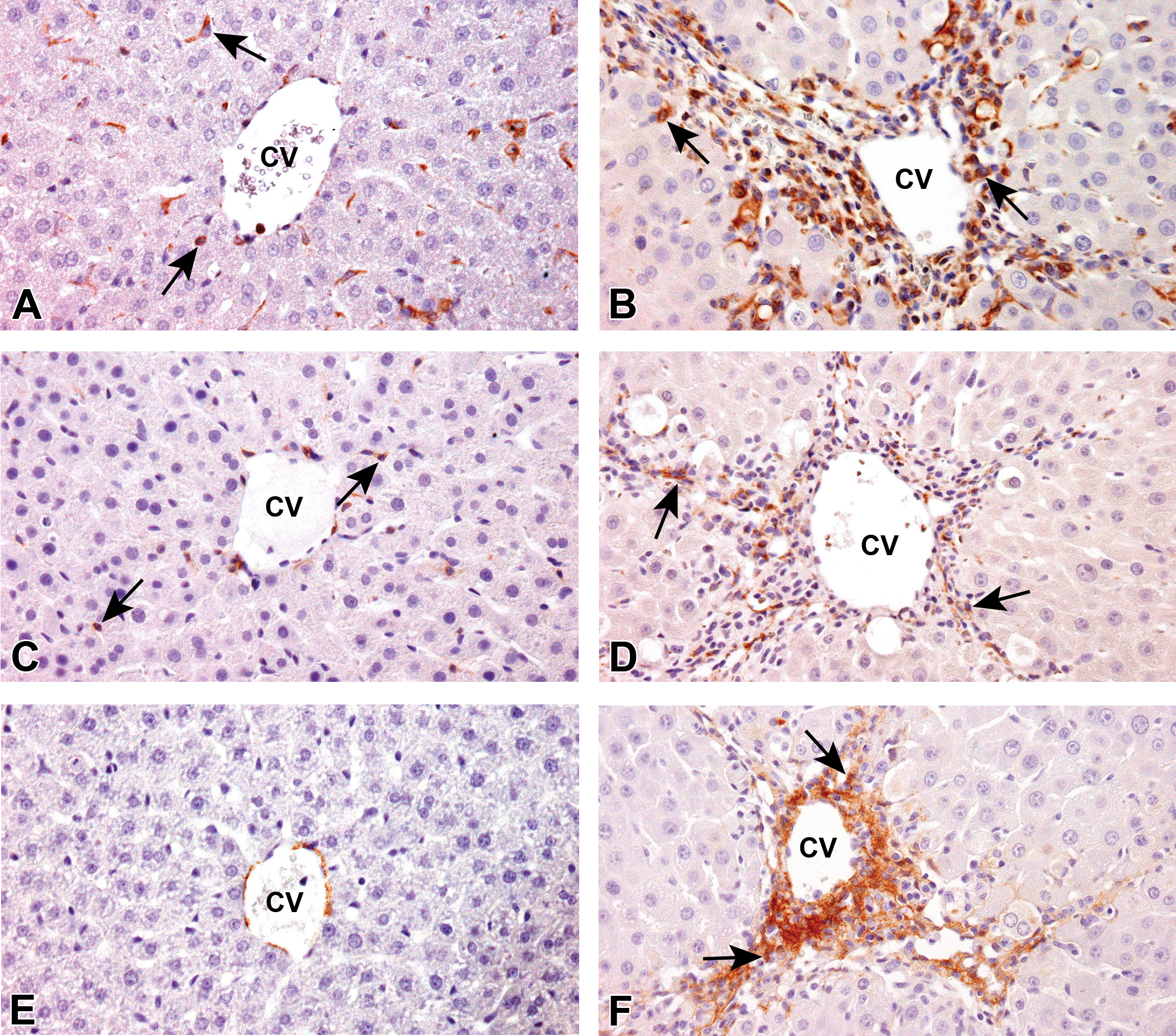
CD163 is expressed in Kupffer cells in normal rat livers (Golbar et al. 2012). In comparison between TAA + CLD and TAA + Lipo groups on day 0 before TAA injection, Kupffer cells located along the sinusoid were significantly decreased in TAA + CLD group (Figures 3A, 4A, and 5A). The decreased number of Kupffer cells was continued during 7-day observation period, regardless of TAA injection (Figures 3A and 5B); therefore, the number of CD163+ cells on days 1, 2, 3, 5, and 7 was significantly increased in TAA + Lipo group than in TAA + CLD group, partly because TAA injection significantly increased the number of CD163+ cells in TAA + Lipo group on days 1, 2, 3, 5, and 7 (Figures 3A, 4B, and 5B). On the other hand, in the numbers of cells reacting to CD68 (Figures 3B, 4C, and 5C), MHC class II (Figures 3C, 4E, and 5E), and CD204 (Figure 3D), there were no significant changes on day 0 in TAA + Lipo and TAA + CLD groups. The TAA injection significantly increased the number of CD68+ (Figures 3B and 4D), MHC class II+ (Figures 3C and 4F), and CD204+ (Figure 3D) cells on days 1, 2, 3, 5, and 7 in TAA + Lipo group; in TAA + CLD group, although the TAA injection also increased the number of these cell types, the increased levels were significantly decreased, particularly on days 1, 2, and 3 (Figures 3B and 5D, 3C and 5F, and 3D, respectively); interestingly, conversely, the CD68+ cell number on days 5 and 7 was greatly increased in TAA + CLD (Figure 3B) group when compared with that in TAA + Lipo group, presumably due to prolonged hepatic lesions (coagulation necrosis) as mentioned above (Figure 2F).
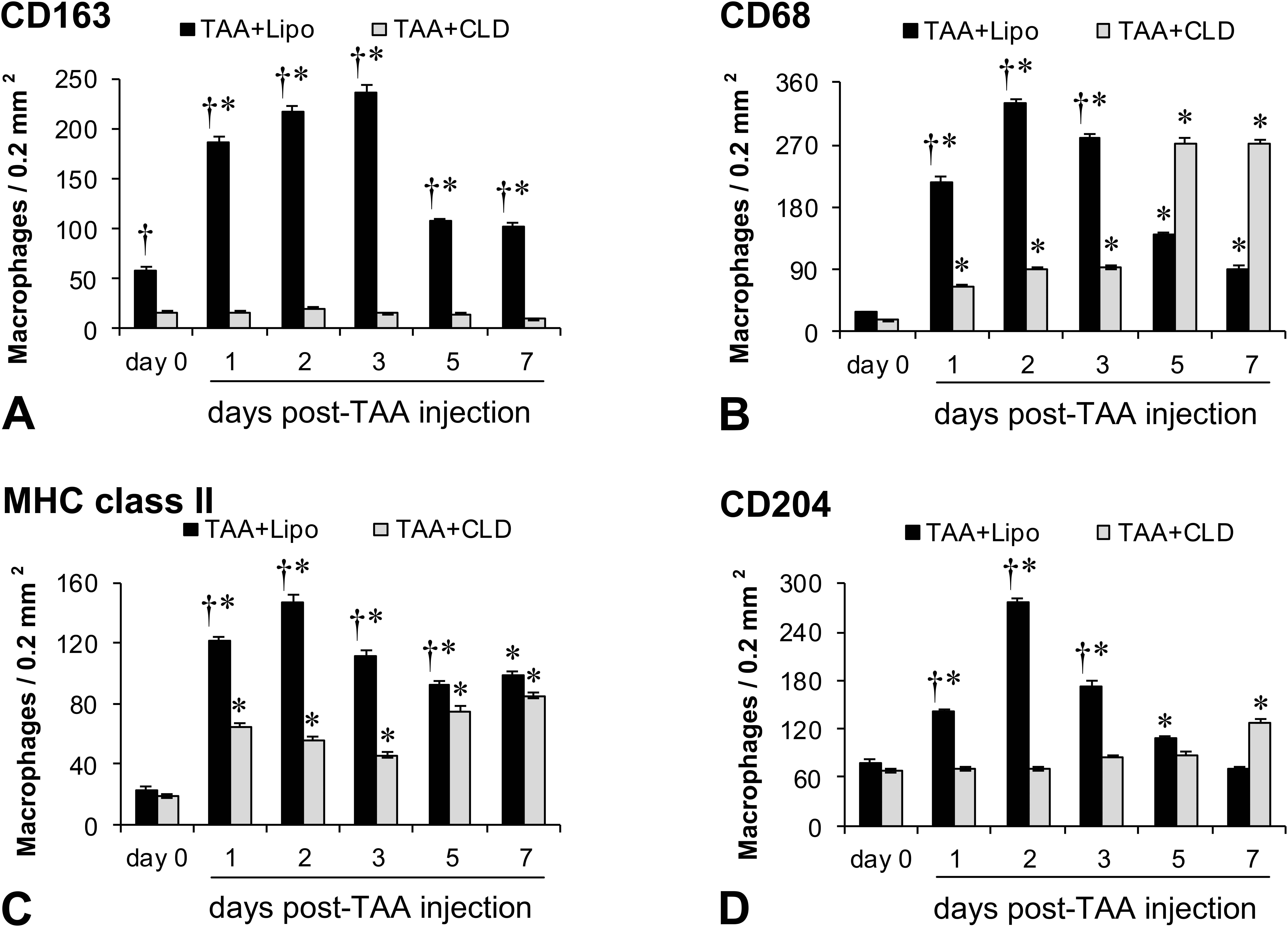
The kinetics of macrophages immunopositive for CD163 (A), CD68 (B), MHC class II (C), and CD204 (D) in TAA + Lipo and TAA + CLD groups in the centrilobular areas. With respect to day 0, there are significant increment of macrophages immune reactive for CD163, CD68, and MHC class II on days 1–7 and CD204 on days 1–5 in TAA + Lipo group. On the other hand, those reactive for CD68 and MHC class II are increased significantly on days 1–7 and CD204 on day 7 in TAA + CLD group. Note the significant differences between TAA + Lipo and TAA + CLD groups on days 0–7 for CD163, days 1–3 for CD68 and CD204, and days 1–5 for MHC class II. Tukey’s test; *P < .05, significantly different from day 0 in TAA + Lipo and TAA + CLD groups; † P < .05, significantly different between TAA + Lipo and TAA + CLD groups at respective examination points. Note: TAA = thioacetamide; CLD = clodronate; Lipo = liposomes; MHC = major histocompatibility complex; CD = cluster of differentiation.
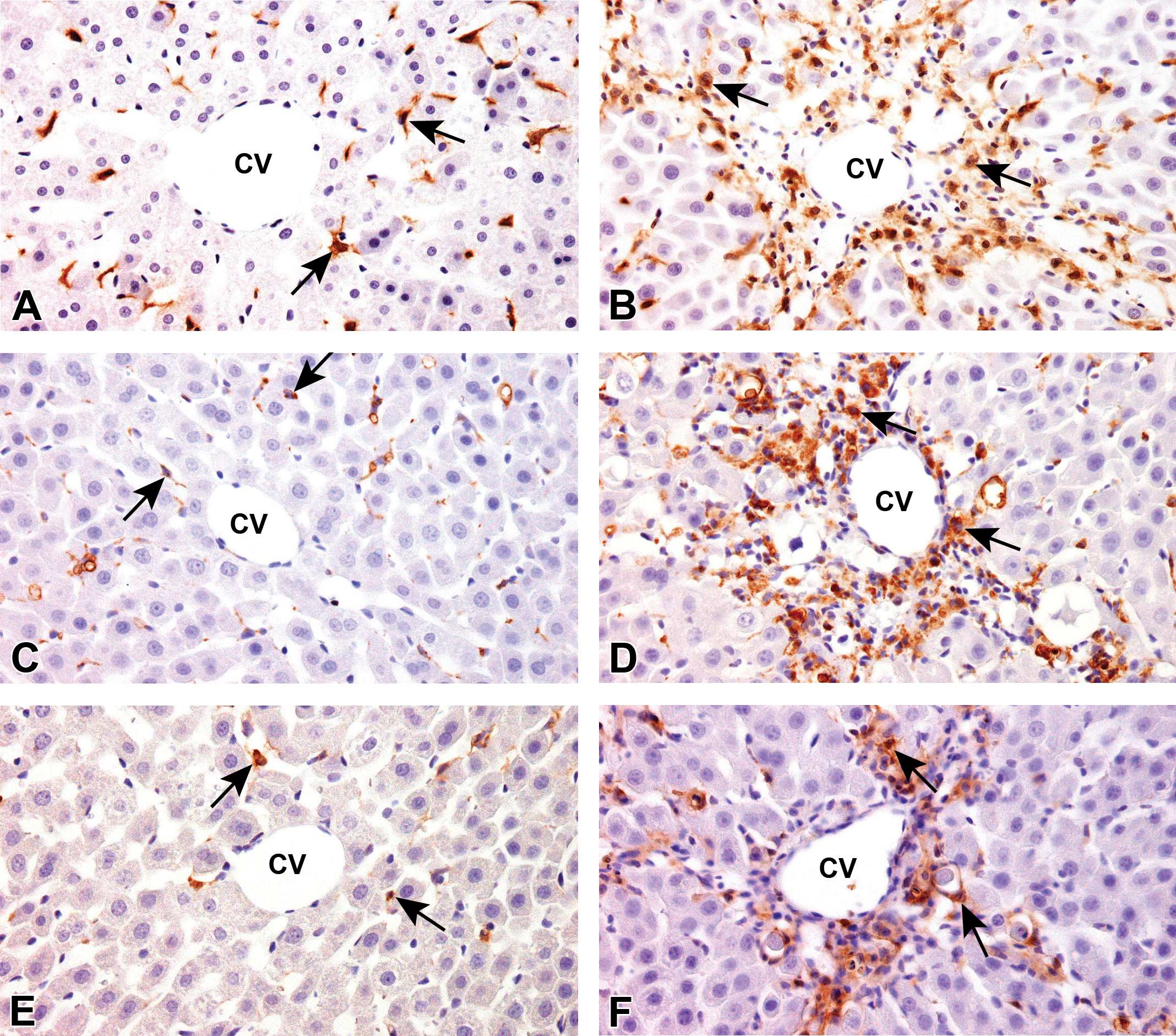
Distribution of macrophages in the centrilobular areas of liver parenchyma in TAA + Lipo group, showing immune reaction for CD163 (A and B), CD68 (C and D), and MHC class II (E and F). Macrophages immune reactive for CD163 (A), CD68 (C), and MHC class II (E) with normal distribution on day 0 and those for CD163 (B), CD68 (D), and MHC class II (F) with increased numbers in the injured centrilobular areas on day 3 are seen. Immunohistochemistry, counterstained with hematoxylin. Arrows indicate representative immunopositive cells. Note: CV = central vein; TAA = thioacetamide; Lipo = liposomes; CD = cluster of differentiation; MHC = major histocompatibility complex.
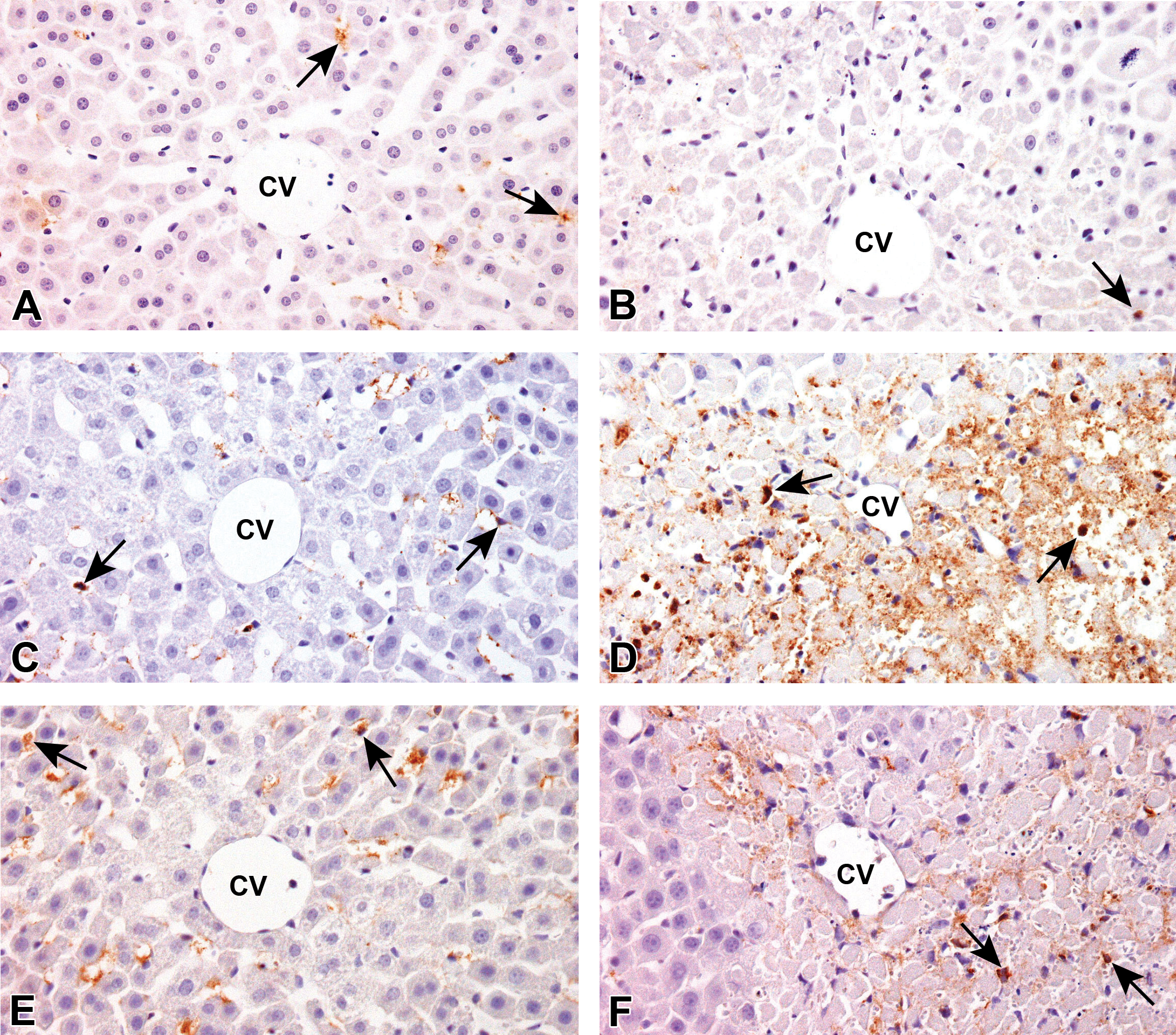
Distribution of macrophages in the centrilobular areas of liver parenchyma in TAA + CLD group, showing immune reaction for CD163 (A and B), CD68 (C and D), and MHC class II (E and F). Macrophages immune reactive for CD163, CD68, and MHC class II are rarely seen on day 0 (A, C, and E) and day 3 (B, D, and F), respectively. Immunohistochemistry, counterstained with hematoxylin. Arrows indicate representative immunopositive cells. Note: CV = central vein; TAA = thioacetamide; CLD = clodronate; CD = cluster of differentiation; MHC, major histocompatibility complex.
Expression of M1-/M2-related Cytokines
Messenger RNAs (mRNAs) of MCP-1 on day 1 (Figure 6A), TGF-β1 on days 1 and 2 (Figure 6B), IFN-γ on day 1 (Figure 6C), IL-10 on days 0 and 1 (Figure 6D), and TNF-α on day 1 (Figure 6E) were significantly increased in TAA + Lipo group; in TAA + CLD group, mRNAs of these factors did not show any significant change (Figure 6A–E). There was no significant change in IL-4 mRNA at any point between TAA + Lipo and TAA + CLD groups (Figure 6F). mRNAs of MMP-2 on day 3 (Figure 7A) and TIMP-1 on days 1 and 2 (Figure 7B) in TAA + Lipo group showed a significant increase in contrast to those in TAA + CLD group; TAA + CLD group did not reveal significant change in mRNAs of MMP-2 (Figure 7A) and TIMP-1 (Figure 7B) at any examination point after TAA injection.
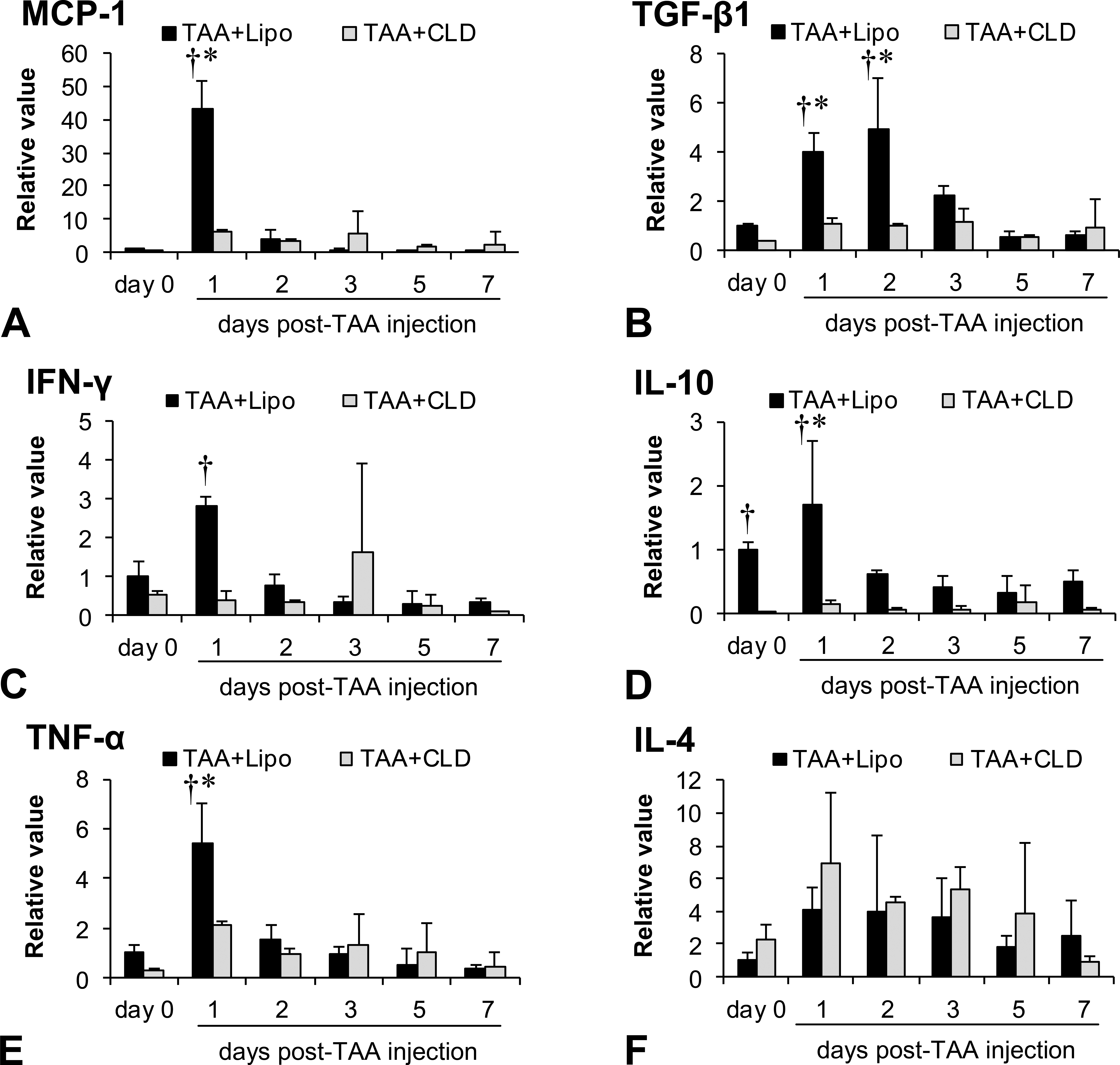
Real-time RT-PCR analyses for MCP-1 (A), TGF-β1 (B), IFN-γ (C), IL-10 (D), TNF-α (E), and IL-4 (F) in TAA + Lipo and TAA + CLD groups. The expressions of mRNAs for MCP-1, TNF-α, and IL-10 on day 1 and TGF-β1 on days 1 and 2 are significantly increased with respect to day 0 in TAA + Lipo group and those for MCP-1, IFN-γ, TNF-α on day 1, TGF-β1 on days 1 and 2, and IL-10 on days 0 and 1 are significantly different between TAA + Lipo and TAA + CLD groups. Tukey’s test; *P < .05, significantly different from day 0 in TAA + Lipo and TAA + CLD groups; † P < .05, significantly different between TAA + Lipo and TAA + CLD groups at respective examination points. Note: TAA = thioacetamide; CLD = clodronate; Lipo = liposomes; MCP = monocyte chemoattractant protein; IFN = interferon; TNF = tumor necrosis factor; IL = interleukin; mRNA = messenger RNA; TGF = transforming growth factor; RT-PCR = reverse transcriptase–polymerase chain reaction.
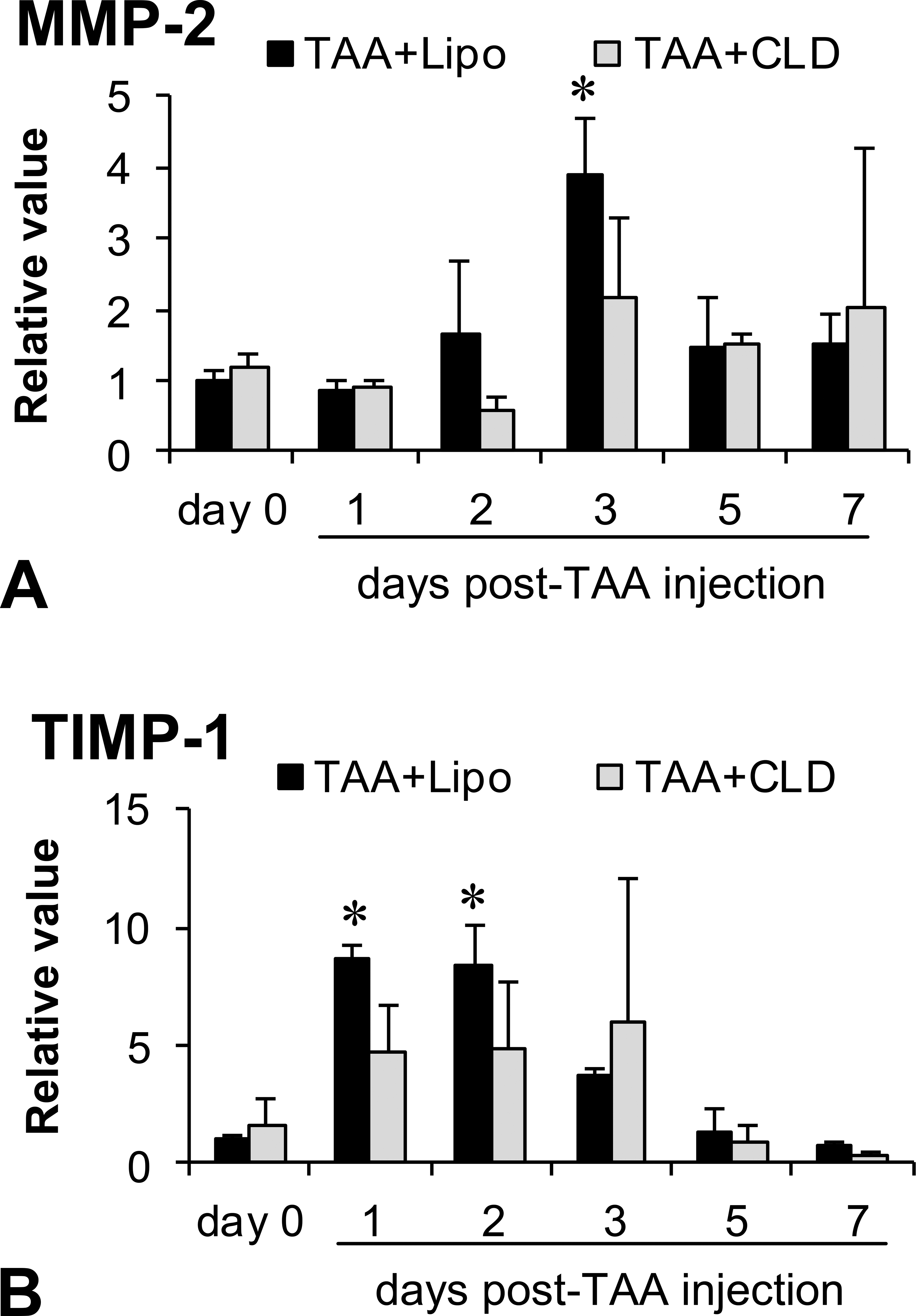
Real-time RT-PCR analyses for MMP-2 (A) and TIMP-1 (B) in TAA + Lipo and TAA + CLD groups. The expressions of mRNAs for MMP-2 on day 3 and TIMP-1 on days 1 and 2 are significantly increased in TAA + Lipo group. Tukey’s test; *P < .05, significantly different from day 0 in TAA + Lipo and TAA + CLD groups. Note: TAA = thioacetamide; CLD = clodronate; Lipo = liposomes; MMP = matrix metalloproteinase; mRNA = messenger RNA; TIMP = tissue inhibitor of metalloproteinase; RT-PCR = reverse transcriptase–polymerase chain reaction.
Immunohistochemistry for Myofibroblasts
Myofibroblasts can express various cytoskeletons in the development; vimentin immunoreaction was seen at early stages, followed by desmin expression at later stages; α-SMA is expressed in well-developed myofibroblasts at later stages which can produce excessive ECMs (Bataller and Brenner 2005; Golbar et al. 2013a). The myofibroblasts are considered to be derived from hepatic stellate cells and are characterized by expression of vimentin and desmin; however, α-SMA was not expressed in the absence of inflammation on day 0 (Figures 8A, C, and E; 9A, C, and E ; Table 2). In TAA + Lipo group, myofibroblasts expressing vimentin (Figure 8B), desmin (Figure 8D), and α-SMA (Figure 8F) showed a peak on days 2 and 3, and gradually disappeared by day 7 (Table 2), indicative of reparative fibrosis. Very interestingly, in TAA + CLD group, the appearance of myofibroblasts reacting to vimentin (Figure 9B), desmin (Figure 9D), and α-SMA (Figure 9F) was delayed with a peak on day 3 for α-SMA and on days 5 and 7 for vimentin and desmin, as compared with those in TAA + Lipo group (Table 2); on day 7 when fibrosis almost disappeared in TAA + Lipo group, myofibroblasts reacting to vimentin, desmin, and α-SMA still remained in necrotic areas with dystrophic calcification.
Distribution of mesenchymal cells/myofibroblasts in the centrilobular areas of liver parenchyma in TAA + Lipo group, showing immunoreactivity for vimentin (A and B), desmin (C and D), and α-SMA (E and F). Mesenchymal cells/myofibroblasts expressing vimentin (A) and desmin (C) are rarely seen on day 0 and their numbers are increased greatly on day 3 (B and D, respectively). Although, α-SMA-expressing cells (E) are not detectable on day 0, the number is increased on day 3 (F). Immunohistochemistry, counterstained with hematoxylin. Arrows indicate representative immunopositive cells. Note: CV = central vein; TAA = thioacetamide; Lipo = liposomes; SMA = smooth muscle actin.
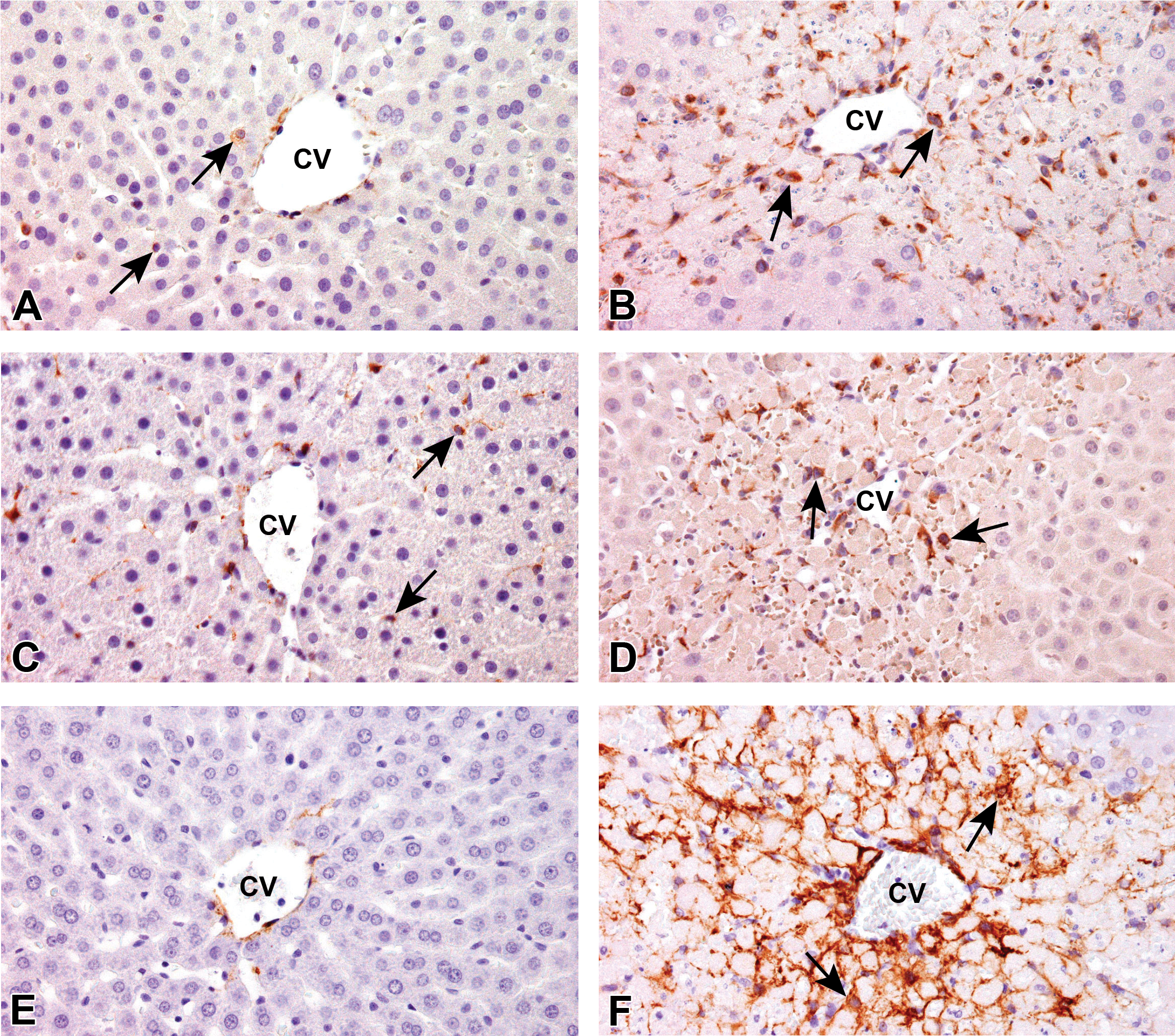
Distribution of mesenchymal cells/myofibroblasts in the centrilobular areas of liver parenchyma in TAA + CLD group, showing immunoreactivity for vimentin (A and B), desmin (C and D), and α-SMA (E and F). Mesenchymal cells/myofirbroblasts reacting to vimentin (A) and desmin (C) are occasionally seen on day 0 and their numbers are increased on day 3 (B and D, respectively). α-SMA-expressing cells are not seen on day 0 (E) and the number is greatly increased on day 3 (F). Immunohistochemistry, counterstained with hematoxylin. Arrows indicate representative immunopositive cells. Note: CV = central vein; TAA = thioacetamide; CLD = clodronate; SMA = smooth muscle actin.
Discussion
Generally, it is well known that macrophages play important roles in tissue damage and fibrosis through phagocytosis, cytokine/growth factor production, and antigen presentation (Schümann et al. 2000; Henderson and Forbes 2008; Lech and Anders 2013). The dose of TAA (300 mg/kg body weight, a single injection) used in this study induced coagulation necrosis on days 1 and 2 and subsequent reparative fibrosis on days 3–7 in the centrilobular area in rats (Mori et al. 2009; Wijesundera et al. 2014). There were a lot of macrophages infiltrating in the injured area in TAA + Lipo group. Macrophages are recently classified as classically activated macrophages (M1) and alternatively activated macrophages (M2) (Sica and Mantovani 2012; Martinez and Gordon 2014; Ong et al. 2015). It is reported that CD68 is expressed mainly in M1 macrophages, whereas M2 macrophages work through CD163 expression (McGuinness et al. 2000; Mantovani et al. 2002; Wijesundera et al. 2014). CD68 is a glycoprotein on lysosomal membranes, particularly on the phagosomes in macrophages; its increased expression implies enhanced phagocytosis (Damoiseaux et al. 1994; Mori et al. 2009; Golbar 2013b). CD163, the hemoglobin scavenger receptor, is expressed by activated macrophages in pathological lesions; increased CD163 expression is associated with the phagocytosis and production of inflammatory factors (Polfliet et al. 2006; Mori et al. 2009; Yamate et al. 2009; Zizzo et al. 2012). We previously analyzed the M1/M2 polarization of different macrophage immunophenotypes in TAA-induced acute liver injury using various cell surface markers such as CD68, CD163, CD204, and MHC class II (Wijesundera et al. 2014). Interestingly, MHC class II macrophages have the M1 polarization, whereas CD204 macrophages undergo the M2 polarization. MHC class II molecule is expressed on antigen-presenting cells including dendritic cells and activated macrophages, and they play pivotal roles in interaction between macrophages and lymphocytes in the immune response (Conrad and Dittel 2011). CD204 is an antigen of scavenger receptor type A (Beatty et al. 2011; Wijesundera et al. 2014). Based on the notion of M1/M2 macrophage polarization, in the present study, we evaluated macrophage functions and subsequent fibrosis in TAA-induced lesions in the absence of macrophages induced by CLD injection.
Clodronate-Lipo Deplete M1 and M2 Macrophages in TAA-induced Rat Liver Lesions
Because different macrophage phenotypes appeared in TAA-induced hepatic lesions, it was considered that heterogeneous macrophage populations could contribute to the hepatotoxicity (Yamate et al. 2000; Tacke and Zimmermann 2014). In this study, the numbers of M1 macrophages (expressing CD68 and MHC class II molecule) and M2 macrophages (expressing CD163 and CD204) were significantly decreased on days 1 to 3 in the TAA + CLD group livers. However, on days 5 or 7, macrophages reacting to CD68, MHC class II, and CD204 recovered to levels of TAA + Lipo group liver, although CD163+ macrophages remained significantly decreased during the observation period. CD163 is expressed in Kupffer cells in normal rat livers (Golbar et al. 2012). These findings clearly indicate that CLD-Lipo could deplete both M1 and M2 macrophages at the early stages in TAA-induced rat livers, with the most sensitive of Kupffer cells. The depletions of M1 and M2 macrophages might imply the insufficient state of macrophage functions.
It is interesting to note that on day 0 before TAA injection, CD163+ Kupffer cells were already depleted by CLD treatment in TAA + CLD group (Figure 3A). Increased value of ALP and decreased mRNA level of IL-10 on day 0 might be related to the depleted Kupffer cells in TAA + CLD group, indicating the roles of Kupffer cells on liver homeostasis via clearance or production of such factors (Smit et al. 1987; Radi et al. 2011).
Depletion of M1 and M2 Macrophages Results in Prolonged Tissue Injury in TAA-treated Livers
Generally, M1 macrophages exert tissue injury through antiproliferative and cytotoxic activities, which result from the release of reactive oxygen species and reactive nitrogen species and proinflammatory cytokines (IFN-γ and TNF-α) (Soehnlein and Lindbom 2010; Laskin et al. 2011). The decrease in M1 macrophages (CD68+ and MHC class II+) was seen in TAA + CLD group at early stages on days 1 to 3. The prolonged and continued centrilobular injury (coagulation necrosis) in TAA + CLD group livers might be due partly to depletion of M1 macrophages. Therefore, coagulation necrosis of hepatocytes seen in TAA + CLD group should be caused by toxic effects of TAA itself but not by macrophage-produced cytotoxic factors. The findings that the values of hepatic enzymes such as AST, ALT, and ALP on days 2 and 3 were greater in the TAA + CLD group might have been due mainly to aggravated hepatocyte damage or partly to decreased clearance of hepatic enzymes by macrophages (Smit et al. 1987; Radi et al. 2011). The action of M1 macrophages is regulated by M2 macrophages, which are primarily involved in downregulating inflammation and initiating reparative fibrosis (Martinez et al., 2008; Laskin et al. 2011; Sindrilaru and Scharffetter-Kochanek 2013). M2 macrophages release anti-inflammatory cytokines (IL-4 and IL-10), which contribute to the resolution of inflammation by phagocytizing apoptotic cells and synthesizing growth factors (TGF-β1) important in tissue remodeling (La Flamme et al. 2012; Martinez 2011; Novak and Koh 2013). Generally, dystrophic calcification occurs in injured tissues such as coagulation necrosis. The dystrophic calcification was observed on days 5 and 7 in the centrilobular lesion in TAA + CLD group. In addition to depleted M1 macropahges on days 1 to 3, CD163+ M2 macrophages (mainly Kupffer cells) were still decreased on days 5 and 7. Although IL-4 mRNA did not change between TAA + CLD and TAA + Lipo groups, mRNAs of TGF-β1 and IL-10 were less in the TAA + CLD group when compared with those in the TAA + Lipo group. Cell debris of necrotizing hepatocytes could not completely be removed by M1 macrophages; furthermore, M2 macrophages did not work for reparative fibrosis, because the appearance of myofibroblasts reacting to vimentin, desmin, and α-SMA was delayed, which may be due to insufficient level of fibrogenic factors such as TGF-β1. Such pathological conditions where M1/M2 macrophages could not work resulted in dystrophic calcification in TAA-induced hepatic lesions, indicating aggravated damage. It has been reported that tissue lesions and subsequent fibrosis under depletion of macrophages resulted in unsuccessful healing (Duffield et al., 2005). Further studies to know the functions of hepatic macrophages are needed by other hepatotoxicants.
In conclusion, the present study showed that the injection of CLD-Lipo caused depletion of both M1 and M2 macrophages in TAA-induced liver injury. The depletion of these macrophages gave rise to the prolonged coagulation necrosis with more increased levels of hepatic enzymes at early stages and resulted in unsuccessful reparative fibrosis and dystrophic calcification at late stages, indicating the aggravated damage. It was clearly demonstrated that hepatic macrophages have important roles in tissue damage and remodeling in hepatotoxicity. The functional analyses of hepatic macrophages would be useful to know the pathogenesis of hepatotoxicity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported partly by the captioned research program funded by Food Safety Commission Japan (FSCJ; 2014), by Kobayashi International Scholarship Foundation (2014–2015), and by JSPS KAKENHI Grant Numbers (26292152 to Yamate and 24·02095 to Golbar).
