Abstract
Liver fibrosis is associated with increased mortality and morbidity. However, there is not effective treatment so far. Vinpocetine (Vinpo) is a synthetic derivative of vinca alkaloid vincamine. Limited previous reports have shown some beneficial effects of Vinpo in different organ fibrosis, but the ability of Vinpo to inhibit liver fibrosis induced by thioacetamide (TAA) has not been reported, that is why we investigate the potential ability of this vinca alkaloid derivative to attenuate liver fibrosis. Hepatic fibrosis was induced in male Sprague Dawley rats by TAA (200 mg/kg; ip; 3 times/week) for 6 weeks. Daily treatments with Vinpo (10–20 mg/kg/day; orally) ameliorated TAA-induced hepatic oxidative stress and histopathological damage as indicated by a decrease in liver injury markers, LDH, hepatic MDA, and NOx levels, as well as increase anti-oxidative parameters. Besides, the anti-fibrotic efficacy of Vinpo was confirmed by decreasing hydroxyproline, and α-SMA. Also, the anti-inflammatory effect of Vinpo was explored by decreasing IL-6 and TNF-α levels. Our novel findings were that Vinpo decreased VEGF/Ki-67 expression in the liver confirming its effect on angiogenesis and proliferation. These findings reveal the anti-fibrotic effect of Vinpo against TAA-induced liver fibrosis in rats, and suggest the modulation of oxidative stress, inflammation, angiogenesis and proliferation as mechanistic cassette underlines this effect.
Introduction
Liver fibrosis is a serious public health problem that results in liver cirrhosis, liver cancer, and death. 1 Thioacetamide (TAA) is one of the most prevalent hepatotoxic agents used to induce liver fibrosis by repeated administration. TAA metabolites covalently bind to the liver macromolecules leading to a decrease in antioxidant capacity and an increase in lipid peroxidation by increasing reactive oxygen species (ROS) production. 2
The pathogenesis of liver fibrosis is complex, in which different factors and pathways are involved. Oxidative stress has been postulated as a major molecular mechanism involved in TAA-induced liver fibrosis. ROS are shown to cause prolonged NF-κβ DNA binding activity. 3 This resulted in enhanced production of pro-inflammatory cytokines tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6). Inflammation slowly leads to the motivation of the pro-fibrogenic stimuli. 4,5 Augmentation of cellular injury caused by various inflammatory mediators, which have angiogenic action and caused elevation of vascular endothelial growth factor (VEGF) for neovascularization. Pathologic angiogenesis is detected in most progressive chronic liver diseases such as liver fibrosis. 6,7 Also, VEGF leads to the upregulation of Ki-67, a marker of proliferative activity. 8 In the fibrotic model, there are strong links between oxidative stress, inflammation, fibrogenesis and angiogenesis. 9
Vinpocetine (Vinpo) is a synthetic ethyl ester of vinca alkaloid vincamine that is an alkaloid extracted from the periwinkle plant, it is well known worldwide as cerebroprotective agent contributes to improve and maintain brain functions so indicated for treatment of memory disturbances, cognitive impairment, stroke and dementia. 10 It has an excellent safety profile, no reports of any toxicity for long-term use at a therapeutic dose up to date. 11 Vinpo is a multi-action agent with different pharmacological targets, it acts as a vasodilator, inhibitor of cyclic nucleotide phosphodiesterase 1 (PDE1), voltage-gated sodium channels, calcium channels and increases glucose and oxygen utilization in the brain. It also has anti-oxidation, anti-inflammatory, anti-thrombosis effect. 10,12 –15
Limited data is available studying the role of Vinpo in the pathogenesis of the liver disease, especially of liver fibrosis. Therefore, the present study was carried out to evaluate the effects of Vinpo on established liver fibrosis and oxidative stress induced by TAA.
Materials and methods
Drugs and chemicals
Thioacetamide (TAA) was ordered from Sigma Aldrich Chemical Co (St. Louis, MO, USA). Vinpocetine (Vinpo) was purchased from the pharmacy as a Vinporal tablet (5 mg) and was suspended in 0.5% Carboxymethyl cellulose (CMC) to be given orally to rats.
Animals
Adult male Sprague-Dawley rats, with an average age of 8 weeks weighing 180 ± 20 g were employed for this study. The animals were purchased from the “Medical Experimental Research Center (MERC), Faculty of Medicine, Mansoura University”. Animals were allowed free access to standard laboratory food and water. All procedures have been approved by the ‘‘Research Ethics Committee’’ of Faculty of Pharmacy, Mansoura University, Egypt which is following Laboratory Animal Care (NIH publication No. 85-23).
Experimental protocol
Rats were divided into five groups: Control (n = 6), rats received no treatment only 0.5% CMC orally for 6 weeks. Vinpo (n = 6), rats received Vinpo (20 mg/kg/day, orally for 6 weeks). TAA (n = 12), rats were injected by Thioacetamide (200 mg/kg, i.p.) three-time per week for 6 weeks.
16
TAA+Vinpo10 mg/kg (n = 6), rats received Vinpo (10 mg/kg/day, orally) for 6 weeks concurrently with TAA injection. TAA+Vinpo 20 mg/kg (n = 6), rats received Vinpo (20 mg/kg/day, orally) for 6 weeks concurrently with TAA injection.
The doses of Vinpo used were chosen from the published literature. 12,15
After 6 weeks, rats were weighed; aneasethised by thiopental (40 mg/kg, ip) and blood was collected from the retro-orbital puncture for measurement of biochemical parameters. The animals were then sacrificed and their livers were removed and weighed to calculate the liver weight to body weight ratio. Portions of hepatic lobes were divided into a) liver tissue which fixed in 10% buffered formalin for histopathological and immunohistopathological analysis; B) liver tissue which was used for hydroxyproline (HP) analysis. Additional lobe of liver was homogenized using (10%w/v) phosphate-buffered saline (50 mM K2HPO4, pH 7.4) for the assay of oxidative stress markers and enzyme-linked immunosorbent assay (ELISA) experiments.
Serum biochemical analysis
Serum was used to measure albumin, bilirubin, lactate dehydrogenase (LDH), Gamma-glutamyl transferase (GGT), Aspartate aminotransferase (AST), Alanine aminotransferase (ALT), and Alkaline phosphatase (ALK-p) according to the manufacturer’s instructions using commercial kits (Biodagnostics, Giza, Egypt). They were analyzed automatically by COBAS INTEGRA 400 plus analyzer (Roche Diagnostics, Switzerland).
Determination of oxidative stress biomarkers in liver homogenate
Liver tissue supernatants were used to determine the levels of glutathione (GSH), and superoxide dismutase (SOD) according to the method described previously, 17,18 respectively. Besides, total nitrite/nitrate (NOx) products, an indicator of NO synthesis, and malondialdehyde (MDA) according to the method described previously 19,20 respectively.
Determination of cytokines in liver homogenate
IL-6 and TNF-α levels were estimated by using ELISA kit (Thermo scientific., Rockford, USA) and (AssayPro, St. Charles, MO) respectively, following the given procedures.
Determination of fibrotic biomarker
The hydroxyproline assay was carried out based on the previously described method. 21
Histopathological and immunohistochemically examination
Fibrosis and necroinflammation changes were evaluated in sections (5 µm thick) stained with Masson’s trichrome and hematoxylin-eosin, respectively, in a blind manner using the Ishak-modified histological activity index scoring system. 22
The following scores were applied to quantify fibrosis:
cirrhosis, probable or definite
marked bridging (p-p and/or p-c) with occasional nodules (incomplete cirrhosis)
fibrous expansion of portal areas with marked portal bridging (p-p) as well as portaltocentral (p-c)
fibrous expansion of most portal areas with occasional portal-to-portal (p-p) bridging
fibrous expansion of most portal areas with or without short fibrous septa
fibrous expansion of some portal areas with or without short fibrous septa
no fibrosis
The necroinflammation scores are calculated as the sum of the four categories of necroinflammation. The final numerical score is the result of multiplying the number of rats per grade by the grade, adding these products and dividing by the total number of rats per group. (0–6) confluent necrosis (0– 4) periportal or periseptal interface hepatitis (0– 4) focal (spotty)lytic necrosis.apoptosis and focal inflammation (0– 4) portal inflammation
The hepatic parenchyma was examined for the presence of disorganization of hepatic cords and pseudo-lobular formation. The hepatocytes were examined for the presence of the following lesions: vacuolar degeneration, ballooning degeneration, steatosis, proliferation of new bile ductules as a reactive process, apoptosis, dysplastic cells (eosinophilic nuclear inclusions), cholangiocarcinoma composed of deranged and irregular, duct forming tissue with few mitotic figures, together with a dense inflammatory infiltrate.
Immunohistochemical assessments (IHC)
Using polyclonal antibodies (Thermo Fisher Scientific Anatomical Pathology 46360 Fremont Blvd., CA 94538, USA). liver tissue expression of alpha-smooth muscle actin (α- SMA), tumor growth factor beta (TGF-β), vascular endothelial growth factor (VEGF), and Ki-67 was determined via immuno-staining utilizing Avidin-Biotin Complex (ABC) method. 23 The severity of staining was graded semi-quantitatively and each sample was assigned a score according to. 24,25
Statistical analysis
Data are expressed as mean ± SEM. The level of significance was set at a p < 0.05. Statistical evaluations of the results were carried out utilizing one-way analysis of variance (ANOVA) followed by Tukey Kramer’s test. Non-parametric Kruskal-Wallis test followed by Dunn’s multiple comparison post-hoc test was used for the analysis of histopathology scores. Statistical analysis and graphing were carried out using GraphPad Prism V 6.02 (GraphPad Software Inc., San Diego, CA, USA).
Results
Vinpocetine (Vinpo) administration for 6 weeks had no significant effect on all parameters assessed in the current study when compared to control rats; therefore, (Vinpo group) results were omitted throughout the results section for simplicity.
Vinpocetine significantly alters thioacetamide-induced increase in mortality, and liver/body weights ratio
Administration of thioacetamide (TAA) alone caused 42% mortality in rats. Concurrent administration of Vinpo (10 or 20 mg/kg) completely abolished TAA-induced mortality (Figure 1A). Upon compared body weights of the animals at the beginning and the end of the experiment and L/BW ratio, TAA caused a significant decrease in body weights (Figure 1B) and an increase in L/BW ratio (Figure 1C). Both changes were significantly inhibited by Vinpo in both doses (10–20 mg/kg), the overall weight for each rat was nearly maintained upon continuous administration of Vinpo in the treatment groups than that recorded with the diseased ones.

Effect of Vinpocetine on Survival ratio, Body and Liver/body weights ratio:
Vinpocetine protected rat liver from thioacetamide-induced hepatic injury
The injection of TAA caused severe liver damage characterized by significant increases in serum ALT, AST, ALK-p (Figure 1A), and GGT activities (Figure 2B), as well as total bilirubin level (Figure 2C), compared with the control group. Although Vinpo treatment in both 10–20 mg/kg doses caused a reduction in ALT, AST, ALK-p, and bilirubin levels compared with the TAA group. Albumin; an indicator for the synthetic function of the liver was significantly reduced in the TAA groups. In both Vinpo groups, albumin level was significantly elevated from TAA groups (Figure 2D). The level of LDH was significantly elevated with TAA injection. Vinpo significantly reduced its level in the Vinpo groups, with a significant reduction recorded with a small dose than the large one (Figure 2E).
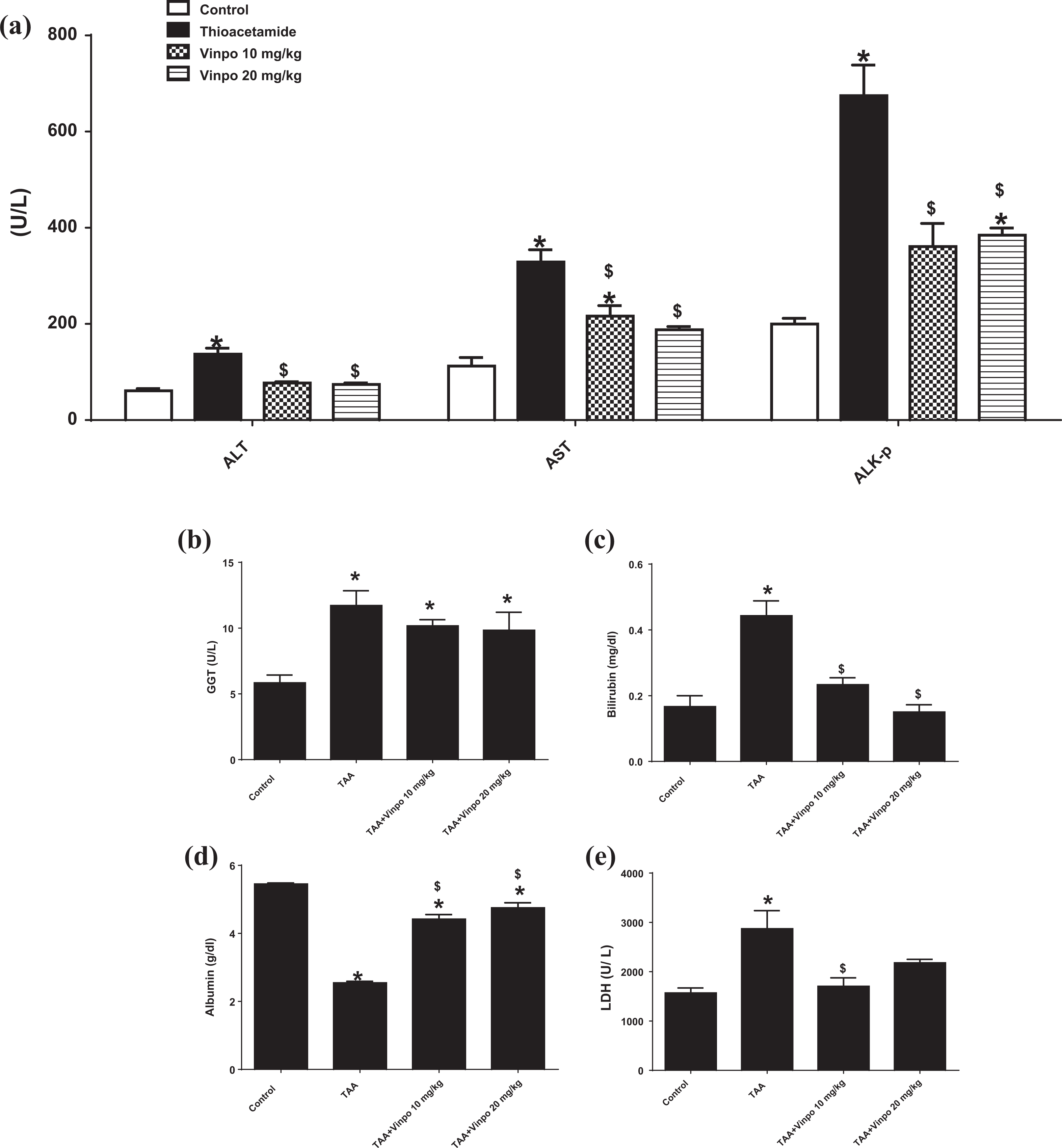
Effect of Vinpocetine on liver injury biomarkers: A: Alanine aminotransferase (ALT), Aspartate aminotransferase (AST), and Alkaline phosphatase (ALK-p), B: Gamma-glutamyl transferase (GGT), C: Bilirubin, C: Albumin, D: Lactate dehydrogenase (LDH). Control, rats treated with vehicles (0.5% CMC); TAA, rats treated with thioacetamide (200 mg/kg/3 time weekly for 6 weeks, ip); TAA+Vinpo, rats treated with thioacetamide and Vinpocetine (10 or 20 mg/kg/day for 6 weeks, orally). Data are expressed as mean ± SEM (Control, Vinpo n = 6, TAA n = 7). Data were statically analyzed using One-Way ANOVA followed by Tukey-Kramer multiple comparisons test (p < 0.05). * $ significantly different from control groups and TAA groups. Vinpo, Vinpocetine; TAA, Thioacetamide.
Vinpocetine repressed the oxidative stress on liver tissue
Hepatic MDA content (Figure 3A) and NO level (Figure 3B) were significantly increased in TAA treated rats, compared with the control rats, but this increase was attenuated in rats treated with Vinpo. The GSH level was nearly diminished in the TAA groups. In a small dose group (Vinpo 10 mg/kg), GSH level was significantly increased with marked value than the TAA rats (Figure 3C). No significant difference in SOD levels in diseased or Vinpo groups than the control group (Figure 3D).
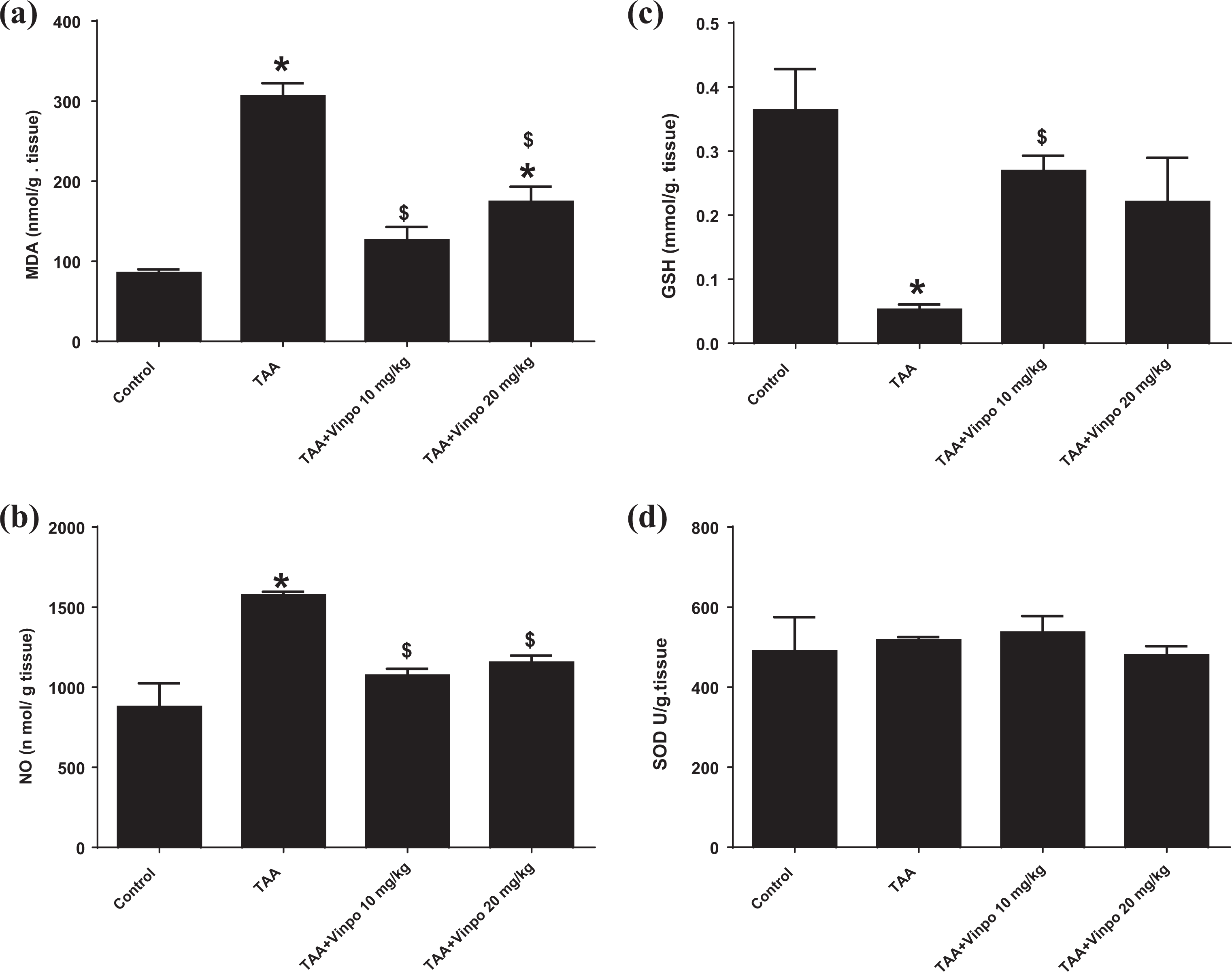
Effect of Vinpocetine inhibit the oxidative stress on liver tissue. A. MDA: Malondialdehyde; B. NO: Nitric Oxide; C. GSH: Reduced Glutathione; D. SOD: Superoxide Dismutase Control, rats treated with vehicles (0.5% CMC); TAA, rats treated with thioacetamide (200 mg/kg/3 time weekly for 6 weeks, ip); TAA+Vinpo, rats treated with thioacetamide and Vinpocetine (10 or 20 mg/kg/day for 6 weeks, orally). Data are expressed as mean ± SEM (Control, Vinpo n = 6, TAA n = 7). Data were statically analyzed using One-Way ANOVA followed by Tukey-Kramer multiple comparisons test (p < 0.05). *$significantly different from control groups and Thioacetamide groups. Vinpo, Vinpocetine; TAA, Thioacetamide.
Vinpocetine alleviated thioacetamide-induced liver inflammation
Both the inflammatory cytokines, TNFα, a cell-signaling protein (cytokine) involved in systemic inflammation, and IL-6, a pro-inflammatory cytokine, were significantly elevated with high value in the TAA groups, and markedly reduced by Vinpo. The large dose of Vinpo 20 mg/kg caused a significant reduction in TNFα compared to the small dose of 10 mg/kg (Figure 4).

Effect of Vinpocetine on liver inflammation markers: Interleukin-6 (IL-6) and Tumor Necrosis Factor- α (TNF-α) Control, rats treated with vehicles (0.5% CMC); TAA, rats treated with thioacetamide (200 mg/kg/3 time weekly for 6 weeks, ip); TAA+Vinpo, rats treated with thioacetamide and Vinpocetine (10 or 20 mg/kg/day for 6 weeks, orally). Data are expressed as mean ± SEM (n = 4). Data were statically analyzed using One-Way ANOVA followed by Tukey-Kramer multiple comparisons test (p < 0.05). * $ # significantly different from control, TAA and TAA+Vinpo10 mg/kg groups. Vinpo, Vinpocetine; TAA, Thioacetamide.
The effect of vinpocetine on thioacetamide-induced histopathological changes
Microscopic pictures of H&E stained liver sections from experimental groups showing score 0 (no inflammation) in the control group, score 4 in TAA, score 3 (Figure 5B) in both doses treated group. (Arrows point to inflammation that consisted mainly of mononuclear cells). X:100 bar 100, (Figure 5A, upper panel) (Table 1).

The effect of vinpocetine on Thioacetamide induced histopathological changes using H&E and Masson’s trichrome staining (X: 100 bar 100):
Histopathological examination for the presence of other lesions.

Control, rats treated with vehicles (0.5% CMC); TAA, rats treated with thioacetamide (200 mg/kg/3 time weekly for 6 weeks, ip); TAA+Vinpo, rats treated with thioacetamide and Vinpocetine (10 or 20 mg/kg/day for 6 weeks, orally). Vinpo, Vinpocetine; TAA, Thioacetamide.
Microscopic pictures of Masson’s trichrome stained liver sections from experimental groups showing score 0 (no fibrosis) in control group, score 5 in the TAA group (Figure 5B), score 5 in Vinpo 10 mg/kg treated groups, score 1 in Vinpo 20 mg/kg treated group. (Arrows point to blue-stained fibrous tissue). X: 100 bar 100, (Figure 5A, lower panel).
Vinpocetine attenuated thioacetamide-induced liver fibrosis
The hydroxyproline (HP) level; an amino acid present in collagen fibers was significantly elevated with marked value in the TAA liver groups, and markedly reduced by Vinpo with a significant reduction recorded with a large dose than the small one (Figure 6A).

Effect of Vinpocetine on fibrosis markers Hydroxyproline (HP), liver Alpha-smooth muscle actin (α-SMA) and Tumor growth factor beta (TGF-β) expression following injection of Thioacetamide (200 mg/kg) in rat using (IHC). Control, rats treated with vehicles (0.5% CMC); TAA, rats treated with thioacetamide (200 mg/kg/3 time weekly for 6 weeks, ip); TAA+Vinpo, rats treated with thioacetamide and Vinpocetine (10 or 20 mg/kg/day for 6 weeks, orally). A: Bar graph showing changes in HP level; B: Photomicrographs of α-SMA (left panel) and scatter-dot plot showing the score of α-SMA (right panel); C: Photomicrographs of TGF-β (left panel) and scatter-dot plot showing the score of α-SMA (right panel). Control group; revealed negative staining of α-SMA and TGF-β. Thioacetamide (TAA) group; revealed very strong expression of α-SMA and TGF-β. TAA+Vinpo 10–20 mg/kg; revealed weak positive of α-SMA and TGF-β. The median is indicated by a line. @, % p < 0.05 compared with control and TAA groups, respectively. Statistical analysis was performed using Kruskal–Wallis followed by Dunn’s Multiple Comparison test (n = 5).
Effect of Vinpocetine on liver Alpha-smooth muscle actin (α-SMA) and Tumor growth factor beta (TGF-β) expression following injection of thioacetamide (200 mg/kg) in rat using (IHC)
Figure 6B, left panel, showed a microscopic picture of immunostained liver against α-SMA. Brown staining means positive and indicated by arrows. No immunoreaction is seen in control group that scored 0, very strong expression of α-SMA is detected in portal areas and fibrous strands dividing hepatic parenchyma into multiple cirrhotic nodules in TAA groups that scored 4 (Figure 6B, right panel), moderate positive reaction for α-SMA that scored 2 in Vinpo 10 mg/kg treated group and very mild positive reaction in Vinpo 20 mg/kg treated group that scored 1. IHC counterstained with Mayer’s hematoxylin. X:100 bar 100.
Figure 6C, left panel, Microscopic pictures of immunostained liver sections against TGF-β showing score 0 in control group, strong positive brown reaction score 4 (Figure 6C, right panel) in TAA group, moderate positive brown reaction score 3 in Vinpo 10 mg/kg treated group, mild positive brown reaction score 1 in Vinpo 20 mg/kg treated group. IHC counterstained with Mayer’s hematoxylin. Black arrows point to positive reaction. X:100 bar 100
Effect of Vinpocetine on liver Vascular endothelial growth factor (VEGF), and Ki 67 expression following injection of thioacetamide (200 mg/kg) in rat using Immunohistochemistry (IHC)
As shown in Figure 7A, upper panel, Microscopic pictures of immunostained liver against VEGF. Brown staining means positive and indicated by arrows. No immunoreaction is seen in the control group that scored 0, marked expression of VEGF in many hepatocytes in TAA groups that scored 3 (Figure 7B, right panel), a weak positive reaction for VEGF in few hepatocytes that scored 1 in both treated groups Vinpo 10–20 mg/kg respectively. Note the reaction is mainly detected on the periphery of cirrhotic nodules and close to the inflammatory zone. IHC counterstained with Mayer’s hematoxylin. X:400 bar 50.
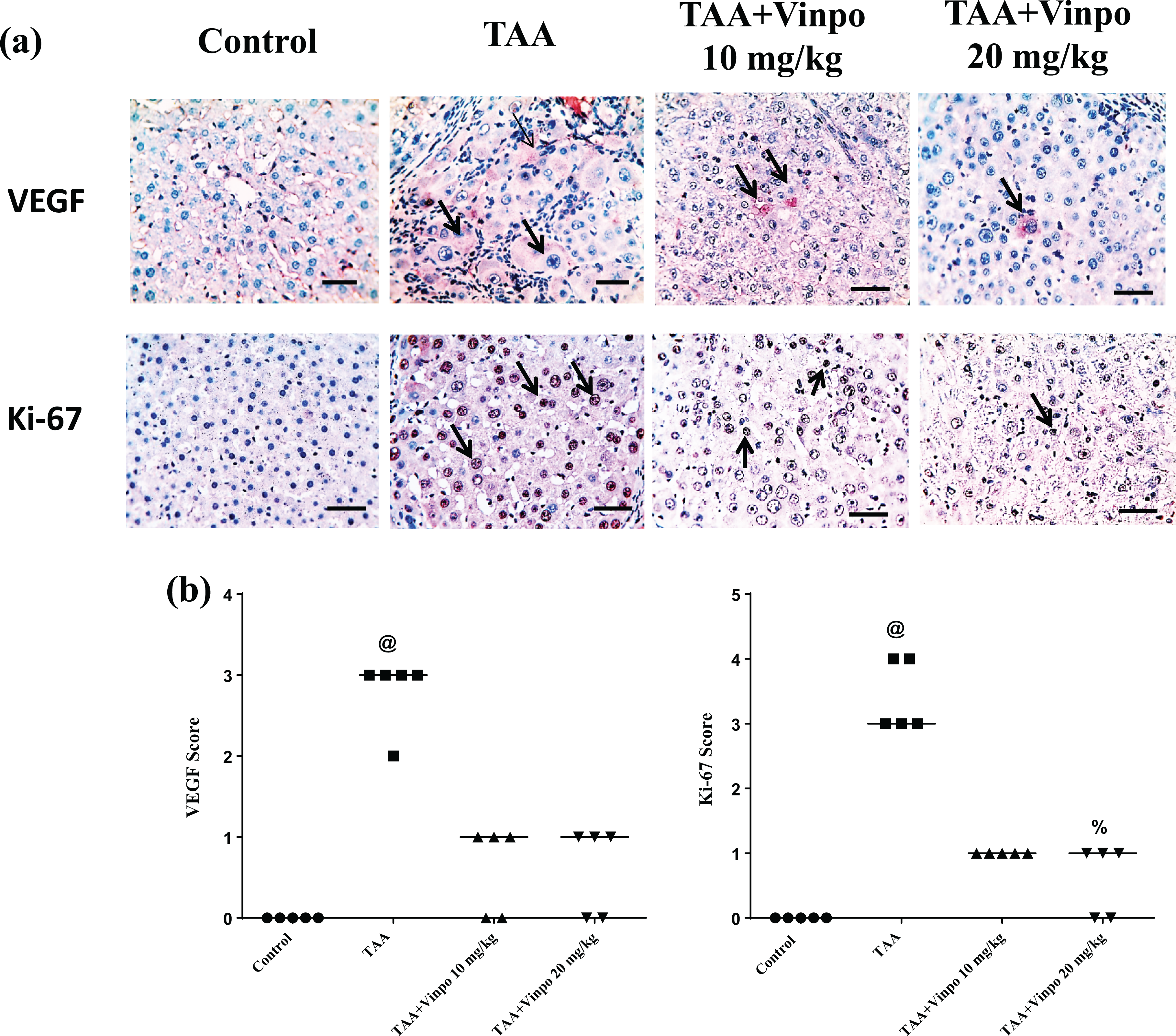
Effect of Vinpocetine on liver Vascular endothelial growth factor (VEGF), and Ki 67 expression following injection of Thioacetamide (200 mg/kg) in rat using (IHC). Control, rats treated with vehicles (0.5% CMC); TAA, rats treated with thioacetamide (200 mg/kg/3 time weekly for 6 weeks, ip); TAA+Vinpo, rats treated with thioacetamide and Vinpocetine (10 or 20 mg/kg/day for 6 weeks, orally).
As shown in Figure 7A, lower panel, Microscopic pictures of immunostained liver against Ki-67. Brown nuclear staining means positive and indicated by arrows. No immunoreaction is seen in a control group that scored 0, very strong expression of Ki-67 in all hepatocytes in TAA groups that scored 4 (Figure 7B, left panel) and weak positive reaction in few hepatocytes that scored 1 in both treated groups (Vinpo 10–20 mg/kg) respectively. IHC counterstained with Mayer’s hematoxylin. X:400 bar 50.
Discussion
To our knowledge, this is the first study demonstrating the possible hepatoprotective action of orally applied Vinpo in attenuating thioacetamide (TAA) induced liver fibrosis in rat models. The main findings of this study were that (a) Vinpo functionally, biochemically, and histopathologically reduced TAA-induced liver fibrosis; (b) Vinpo significantly inhibited oxidative stress biomarkers (MDA and NOx), and inflammation biomarkers (TNF-α and IL-6); (c) Vinpo significantly inhibited biomarkers of pro-fibrogenesis (HP, TGF-β, and α-SMA), angiogenesis (VEGF), and proliferative (Ki-67) (Figure 8).
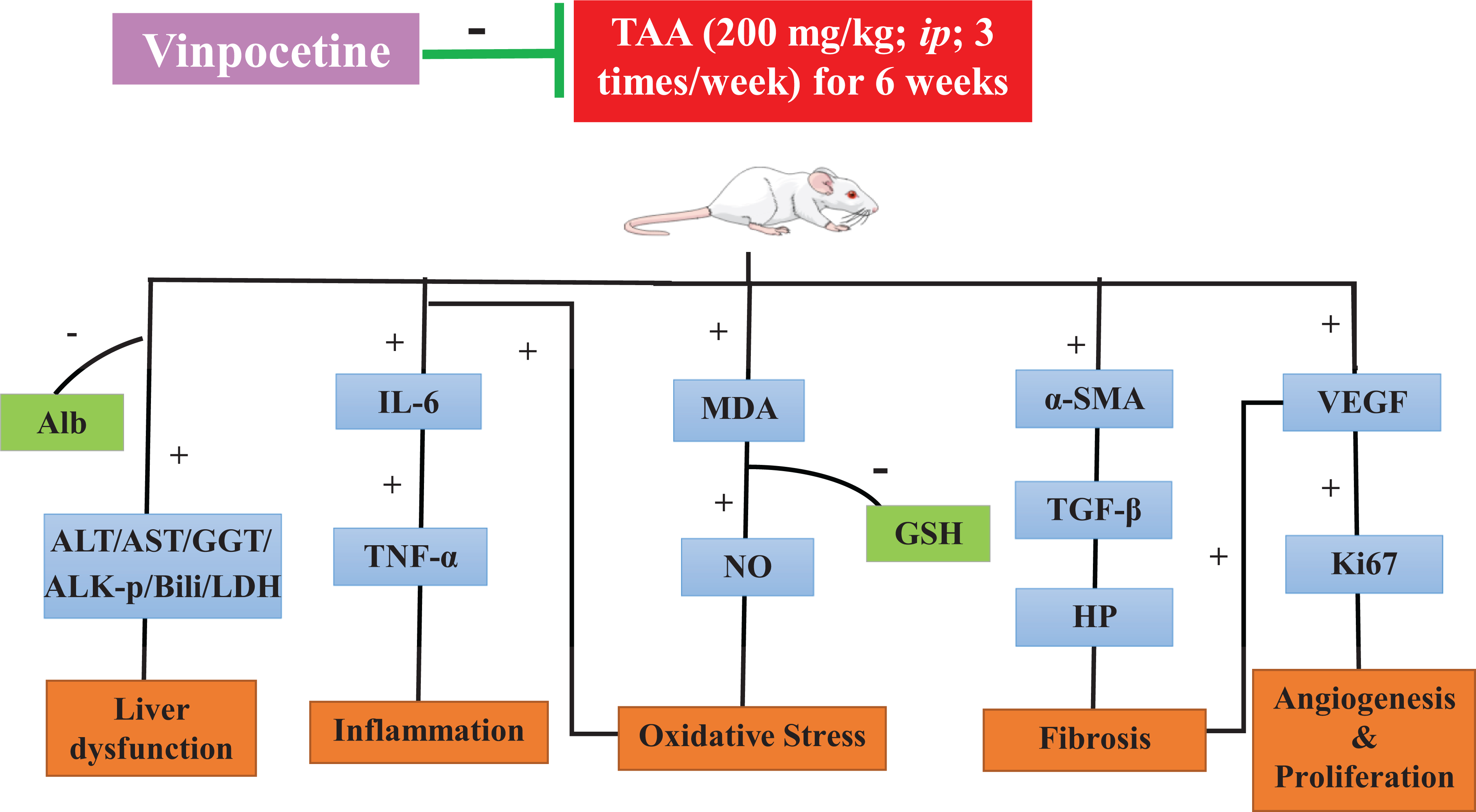
Schematic diagram summarizing the possible mechanisms through which Vinpocetine can ameliorate thioacetamide-induced liver fibrosis. Control, rats treated with vehicles (0.5% CMC); TAA, rats treated with thioacetamide (200 mg/kg/3 time weekly for 6 weeks, ip); TAA+Vinpo, rats treated with thioacetamide and Vinpocetine (10 or 20 mg/kg/day for 6 weeks, orally). ALK-p: Alkaline phosphatase, GSH: Reduced Glutathione, GGT: Gamma-glutamyl transferase, IL-6: Interleukin-6, LDH: Lactate Dehydrogenase, MDA: Malondialdehyde, NO: Nitric Oxide, ROS: Reactive oxygen species, SOD: Superoxide Dismutase, TNF-α: Tumor Necrosis Factor- α, VEGF: Vascular endothelial growth factor, α-SMA: Alpha-smooth muscle actin.
Liver fibrosis is a serious public health problem that results in liver cirrhosis, liver cancer, and death. Induction of liver fibrosis in rodents by hepatotoxins, such as dimethylnitrosamine, furan, and carbon tetrachloride is often associated with major drawbacks, such as low reproducibility resulting from severe inflammatory reactions, high mortality rate during induction, and low resemblance to humans’ fibrotic livers. 26 Compared to other experimental models, TAA-induced liver fibrosis is an ideal model to test potential antifibrotic drugs as the hepatotoxin TAA is well known to produce a hardly reversible fibrosis in rodents similar to that of humans. 27
In our study, in TAA group, there was a significant increase in AST, ALT, ALK-p, GGT, and bilirubin and reduction in albumin which indicate damage and worsening of hepatic functions and suppress synthetic and detoxifying functions of the liver. Also, TAA caused an elevation in the liver index (ratio of liver/ body weight) due to the accumulation of extracellular matrix (ECM) in the fibrotic liver. These findings came in line with previous studies. 6,28 –31 Vinpo preserved hepatic functions and opposed TAA actions which indicate improvement of liver functions. These results supported previous notions that backs the hepatoprotective action of Vinpo. 12,13,15,32 Also Vinpo showed no toxic effect on liver markers, and produced no significant change from negative control rat as previous reported. 15 Vinpo at therapeutic doses has shown no significant side effects or toxicity and it is considered a safe drug for long-term use 11
Oxidative stress has been postulated as a major molecular mechanism involved in TAA- induced liver fibrosis. The hepatic metabolism of TAA release ROS and other toxic metabolites resulting in hepatic necrosis, centrilobular damage affecting cell membranes releasing MDA as lipid peroxidation product. 33,34 The link between ROS and inflammation and fibrosis has been recognized in many diseases such as hepatic diseases. In liver fibrosis, both oxidative stress and inflammatory mediators play a role in the activation of hepatic stellate cells (HSCs), and, conversely, activated HSCs could, in turn, enhance the generation of ROS and inflammation and inhibit antioxidant defense. 35,36 It has been previously reported that both TNF-α and IL-6 promoted hepatic fibrosis. 4,5 Till now, there are no established anti-fibrogenic agents. Therefore, previous studies showed that agents exhibit anti-oxidant or anti-inflammatory effect are considered to be promising for attenuating liver fibrosis. 37 Therefore, TNF-α and IL-6 signal pathways have been checked in this paper. Vinpocetine in the present study was found to suppress the accumulation of oxidative and inflammatory markers (MDA, NOx, IL-6, and TNF-α), and restored anti-oxidative activity by increasing GSH levels in rats showing strong anti-oxidant and anti-inflammatory activity opposing the actions of TAA. These results are concord with other results of Vinpo. 12,13,15
In our fibrotic model, hydroxyproline content, very important amino acid entering in collagen formation, 38 and expression of TGF-β (main fibrogenic cytokine and a potent activator of HSC) and α-SMA (a unique marker for HSC) 39 were elevated in response to TAA injection. These observations indicated an increase in the number of activated HSCs that express α-SMA and further produce TGF-β, collagen and other ECM proteins. Our results agreed with other results. 28,31,40 Previous studies have demonstrated that inhibition of the TGF-β/α-SMA signaling pathway attenuates liver fibrosis. 31,41 Our results support this hypothesis as fibrotic markers (Hp, TGF-β and α-SMA) downregulated by Vinpo administration, indicating the anti-fibrotic activity of Vinpo. Previous experimental studies have also reported that Vinpo possesses anti-fibrotic activity by decreasing fibrotic markers in cardiac fibrosis 14 and liver fibrosis induced by carbon tetrachloride 13 and diethylnitrosamine. 15
Regarding the functional link between liver fibrosis and angiogenesis, anti-angiogenesis is considered a target for fibrosis therapy. 42 As a result of oxidative stress and liver hypoxia that results from disrupted blood vessels, increase expression of Vascular endothelial growth factor (VEGF), which induces new blood vessels formation termed angiogenesis. 9 VEGF, a major angiogenic factor, was found to increase in TAA induced-liver fibrosis. 6,7,43 Moreover, in this study, TAA showed a significant increase in VEGF providing further support on the link between liver fibrosis and angiogenesis. Vinpo significantly attenuated the rise of VEGF expression in TAA-treated animals confirming its antiangiogenic activity. According to our knowledge, no previous study reported the anti-angiogenic activity of Vinpo in the fibrotic model.
Previous studies have pointed out a clear relation between VEGF and Ki-67. Ki-67 is a marker of proliferative activity and carcinogenicity of liver expressed in many cell cycle phases except the G0 phase 44 which was associated with higher mitotic activity in hepatocellular carcinoma lesions. 45 An increase in Ki-67 hepatocyte proliferation index is probably induced by the upregulation of VEGF and other growth factors, 8 and it has a relation with fibrosis. 46 Beside, Ki-67 expression are reliable markers of the fibrotic transformation of the liver tissue. 47,48 In this study, Ki-67 expression was upregulated in TAA rats, coincided with the results of the previous liver fibrosis model. 49,50 Previous studies reported that decreased Ki-67 expression, anti-proliferative effect, used as a potential therapeutic for fibrosis model. 51,52 The anti-proliferative effect of Vinpo on Ki-67 expression was reported in this study, which may be due to the antiangiogenic (as demonstrated by decreased VEGF) and anti-fibrotic effect of Vinpo (as demonstrated by reduced α-SMA, TGF-β, and HP). According to our knowledge, this is the first study reported the effect of Vinpo as an anti-proliferative on ki-67 in the fibrotic model.
Conclusion
Vinpocetine ameliorated liver fibrosis induced by TAA in rats via exhibiting strong anti-oxidant (suppressed oxidative stress ROS and NOx), anti-inflammatory (suppressed LDH, TNF-α, and IL-6) and anti-fibrotic (suppressed HP and αSMA), anti-angiogenic (suppressed VEGF), anti-proliferative (suppressed Ki-67), activities and improved liver functionally, biochemically, and histopathologically. Vinpo is cheap, available and could be a good candidate for clinical trials for protection against liver fibrosis (Figure 8).
Limitations
Several limitations of this study should be considered. First, the absence of investigation the effect of Vinpocetine on the Cytochromes P450 enzyme. Second, Vinpocetine administered to rats as a prophylactic agent versus therapeutic drug. Finally, the absence of comparison between the effect of Vinpocetine and other anti-fibrotic agents (silymarin) against thioacetamide-induced liver fibrosis.
Footnotes
Highlights
൦ Vinpo significantly inhibited biomarkers of profibrogenesis and inflammation. ൦ Vinpo significantly inhibited Ki67/VEGF axis in a rat model of TAA-induced liver fibrosis. ൦ Vinpo functionally, biochemically, and histopathologically abrogated TAA-induced liver fibrosis
Abbreviations
Acknowledgments
Great thanks to Walla F. Awadin, Associated professor of Pathology, Faculty of Veterinary Medicine, Mansoura University for her help to complete the histopathological and IHC parts of this study.
Author contributions
All authors (AE, AN, AA, IK, RE, and RA) carried out the experiment. AE participate in designed the experiment and wrote the discussion part. AN wrote the results part. AA wrote the material and method part. IK participate in wrote the discussion part. RE wrote the introduction part. RA designed, carried out the experiment, designed the graphical abstract, statistically analyzed data, participate in writing all parts of the manuscript, revised and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The author(s) received no financial support for the research, authorship, and/or publication of this article.
