Abstract
The pig was introduced more than 20 years ago in drug development following attempts of finding a species that shares better homology with human than the dog, based on biophysiological parameters. However, miniaturization, standardized breeding, and health status control were required before the pig could find a broader than niche application in pharmaceutical industry. During the years of experience with minipigs in pharmaceutical research and the science evolving rapidly, the selection of a nonrodent animal species for preclinical safety testing became primarily driven by pharmacological (target expression homologous function), pharmacokinetic, and biophysiological considerations. This offered a broad field of application for the minipig, besides the well-established use in dermal projects in all areas of drug development but also in novel approaches including genetically modified animals. In this article, we look at recent approaches and requirements in the optimal selection of a nonrodent model in pharmaceutical development and critically ask how good a choice the minipig offers for the scientist, how did the testing environment evolve, and what are the key requirements for a broader use of the minipig compared to the other well-established nonrodent species like dog or monkey.
In the earlier days of toxicology research to support pharmaceutical development, the requirements of health authorities in the different global regions (United States, Europe [EU], or Asia) were mainly driven by the generic concept of rodent and nonrodent testing, particularly to cover a broad spectrum of potential biological responses, without specifically asking for pharmacodynamic response in the toxicology species. In the 1980s, the pig or minipig was rarely used in the industry, and a publication by Alder and Zbinden (1988) listed only the Canadian health authorities and Food and Drug Administration (FDA) for considering the pig for topical use (weanling pig). For topical development, most of the other countries were relying on rodents, guinea pig, or rabbit. In 1992, collaboration was starting between a pig breeder and an academic institution in Germany, resulting in what is today the most commonly used minipig in pharmaceutical research, the Ellegaard-Goettingen minipig (Ellegaard 2015a). The European pharmaceutical industry has picked up on the use of the minipig as a “standard” nonrodent species for toxicity testing as early as 1994, running the whole package including oral and dermal chronic toxicity studies with the Goettingen minipig for a product by Novartis, which was approved in 2001 (Elidel 2001). In the mid-1990s, there were a series of attempts to further characterize the minipig for its use in pharmaceutical research (Summerfield, Rziha, and Saalmueller 1996; Zuckermann et al. 1996) and toxicology, for example, reproductive toxicology (Jorgensen 1994). At the European Society of Toxicology in 1997, there was the introduction of the minipig in toxicity by a special symposium dedicated to its use (Svendsen 1998). In the same year, the Organization for Economic Cooperation and Development (1998) included the minipig as an alternative species in guideline 409. In spite of many attempts to justify its use by socioethical considerations and the fact that minipig use may prove less offensive to some groups within the society, it could not deliver the global answer to the moral dilemma of research (Morton 2012) and is not considered to be acceptable for the replacement of dogs and monkeys without further (scientific) arguments (Webster et al. 2010). However, the shorter reproductive cycle and larger litter size of pigs provide opportunities for reduction in animals to be kept for breeding purposes (Bode et al. 2010; Foster et al. 2010).
In EU, the Directive 2010/63/EU voted in 2010 has been implemented in 2013 in the European Medicines Agency (EMA) guidelines, resulting in restrictions in the use of nonhuman primates (NHPs) in biomedical research (EMA 2014). According to chapter 14 of this guidance, “due to their genetic proximity to human beings and to their highly developed social skills, the use of non-human primates in scientific procedures raises specific ethical and practical problems in terms of meeting their behavioural, environmental and social needs in a laboratory environment.”
Eventually, animal testing in drug development has to be based on the scientific rationale that pharmacological or adverse effects observed in a test species would also occur to man. It is obvious that there is still some room for improvement in the predictivity of animal models related to safety issues caused by pharmaceutical development candidates in patients (Cook et al. 2014). And looking at reviews spanning the past two decades, the existing paradigm of nonclinical toxicity testing has proven critical and useful for human risk assessment (Olson et al. 2000; Greaves, Williams, and Eve 2004; Tamaki et al. 2013) and still indicates a need for novel and refined testing strategies (Mangipudy, Burkhardt, and Kadambi 2014).
For a long time, pharmaceutical industry and regulators felt it was appropriate to assure translational safety by a probabilistic approach increasing the chance of success when you ask for at least two animal species that are phylogenetically somewhat apart like a rodent and a nonrodent species. Without necessarily requiring closeness to man, this is still the general rule supported by the current international guidelines like the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use, Guidance M3 (Revision 2; 2009). This is also reflected by the current use of mammals in research as published by the American Physiological Society (2015), showing that over 90% of animals used in biomedical research are rodents, and among the nonrodent species used dogs, cats, or NHPs that account for less than 1%, the pig or minipig is not even considered in that statistics. A recent survey by the International Consortium for Innovation and Quality in Pharmaceutical Development on factors considered in selecting the minipig as a nonrodent species for safety assessment (Colleton et al. in press) confirmed the survey performed in 2010 by the RETHINK group (Bode et al. 2010). The key outcome of this survey presented the drug substance requirements and the associated costs of the toxicology program as major drivers influencing the deselection of the minipig as the nonrodent species for a given project.
A recent overview on the use of minipigs was presented for the years 2002 through 2008 by Ganderup et al. (2012) based on government registration data. The total use of swine and NHPs had increased by 15% and 33%, respectively, but there were substantial differences between the global regions. The authors concluded that the pressure to move away from the dog as a testing species in toxicology was increasing in EU. A reflection on the current use of minipigs across the contract research organization (CRO) industry was performed by a survey which was sent to 8 of the top 10 CROs globally, representing 27 preclinical CRO sites (12 in North America [NA], 13 in EU, and 2 in Asia). The absolute number of nonrodents (pigs, minipigs, dogs, and NHPs) utilized over the last 5 years, detailing the strain of pigs and minipigs as well as the route of administration are presented in a relative manner to fulfill confidentiality obligations.
The relative usage of pigs and minipigs compared to other nonrodents from 2009 to 2014 in the largest CROs (Figure 1) shows different trends in EU and NA. In the European CROs interviewed, a maximum was reached in 2010, where almost 1 porcine was utilized for 2 dogs and 2 primates. While the relative usage of pigs and minipigs dropped almost 30% in EU, it has been growing consistently in NA, thus the global average use of pigs and minipig remained between 7.2% and 10.4% of nonrodents utilized between 2009 and 2014. While the maximum usage of pigs in 2010 may be associated with the momentum created by the RETHINK project (Forster et al 2010), it is surprising to observe a significant drop in 2013, given the limited availability of dogs in EU following the closure of a breeding facility in Italy, (Marshall Farm, Montichiari in September 2012).
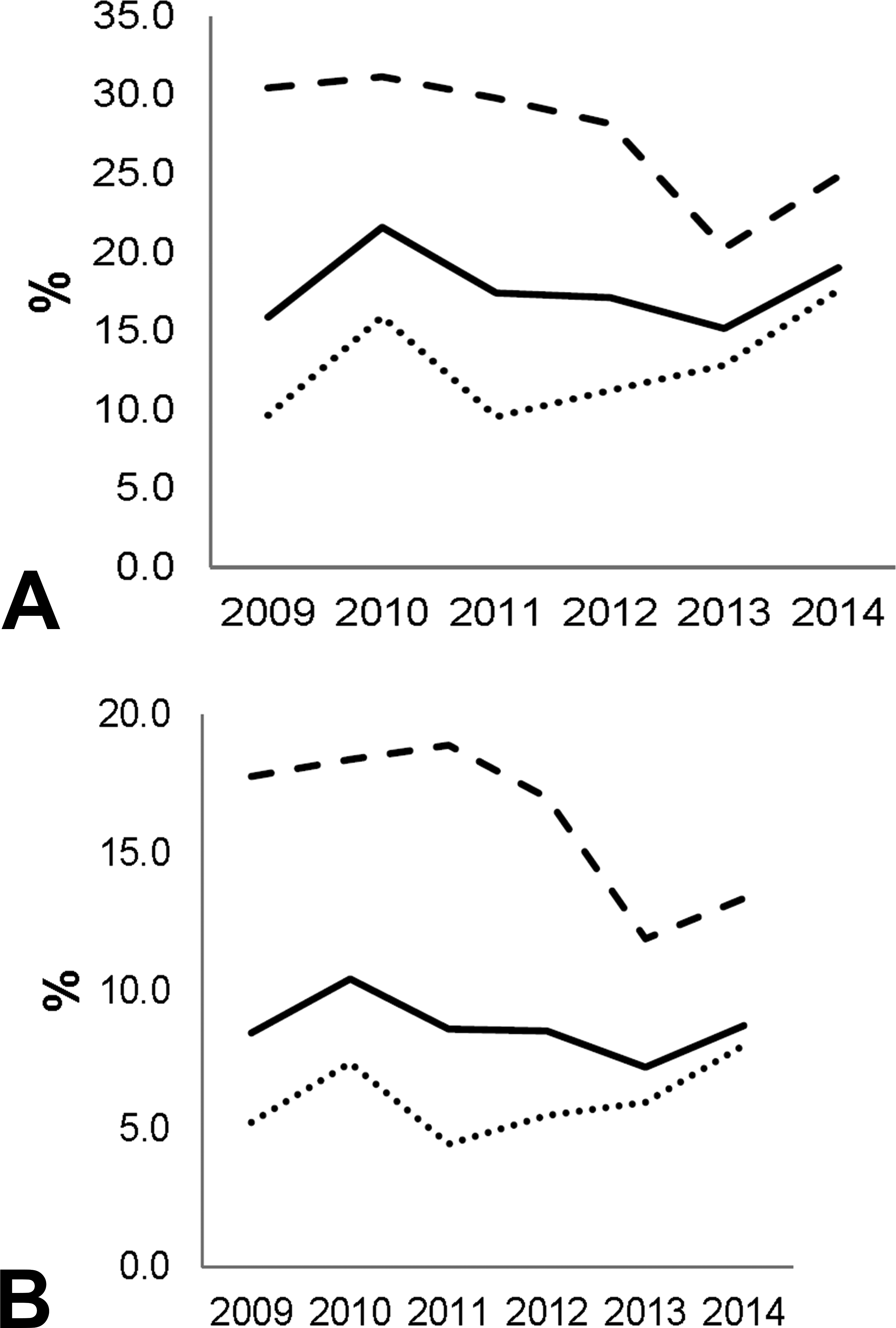
Relative use of (A) pig and minipig versus dog, and (B) pig and minipig versus dog + nonhuman primates, in percentage and per geographical area: North America (……NA), Europe (- - - EU), and globally (continued) among the 27 contract research organization sites interviewed.
As described in Figure 2, the Goettingen minipig remains the main species of choice for preclinical safety assessment globally. The Goettingen pig is currently the only pig strain available globally through breeding facilities that are generally used by pharmaceutical industry, allowing consistency and constantly increasing historical background data for sponsors and CROs in the field of preclinical safety. The farm pig (large white hybrid or Yorkshire pig) is the main alternative to Goettingen, but their use is generally intended for different purposes, for example, animal health, surgical model, devices, or local tolerability. In addition, there are other minipig strains that have been mentioned in this survey which have been used at very low numbers: Yucatan, Sinclair, and micro-pigs.
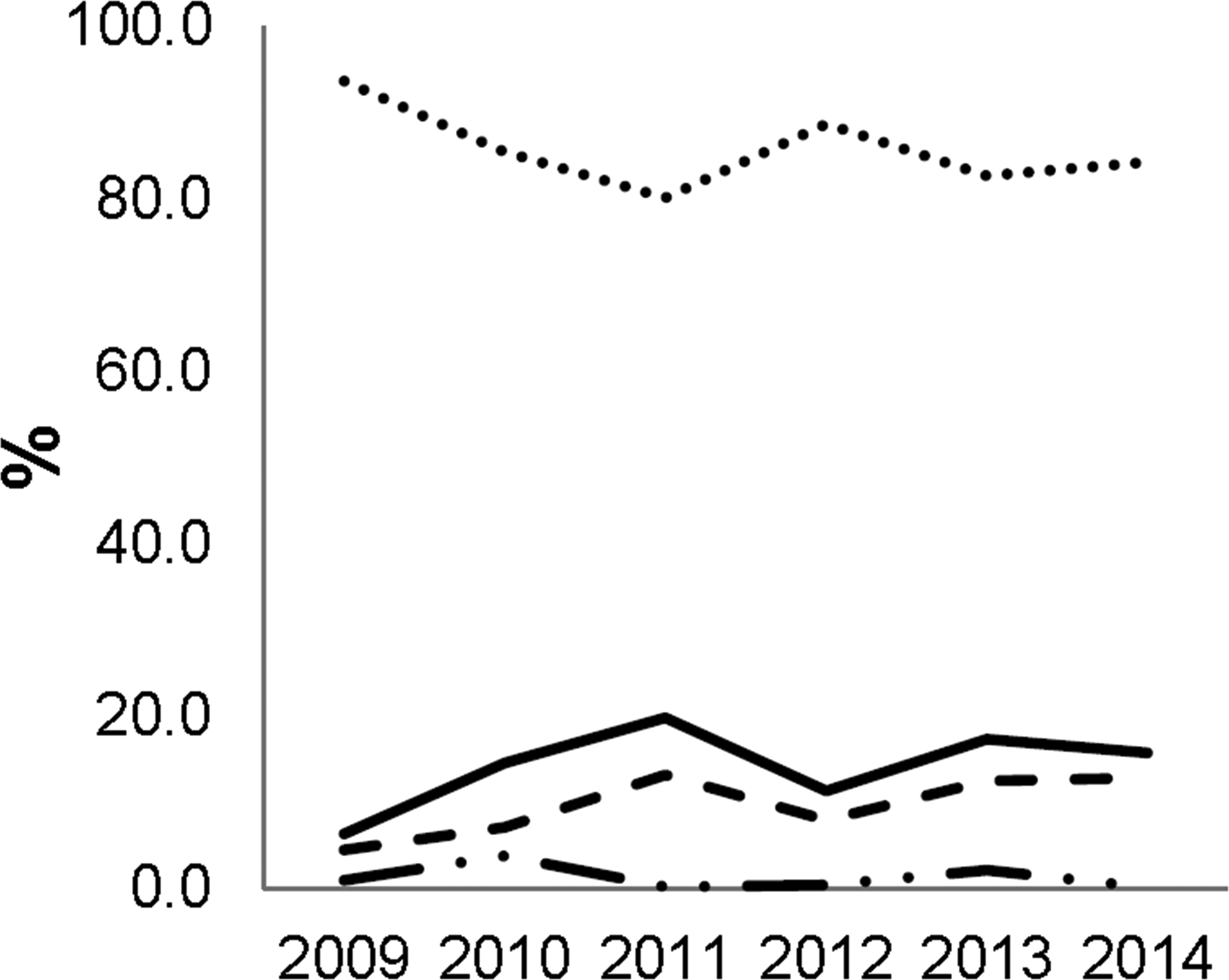
Pig and minipig strains utilized by the 27 contract research organization sites interviewed from 2009 to 2014. Only the Goettingen (……), conventional pig (or farm pig, large white hybrid, or Yorkshire pig; - - - -), and Yucatan (- - . . - - . .) are detailed. The continued line represents the sum of all strains of pig and minipig other than Goettingen minipig.
Looking at the overall usage of the pig, there is a relative increase in programs using the dermal route of administration over the last few years, up to 45.7% of all pigs used in 2014 (Figure 3). This development is mainly driven by the North American providers with more than 60% of the minipigs being dosed dermally in 2014 (data not shown). In EU, the dermal route of administration represented less than 30% in 2014 (data not shown), and the minipig is used for a broader range of administration routes and study types such as oral, intravenous, reproductive, or juvenile toxicity studies.
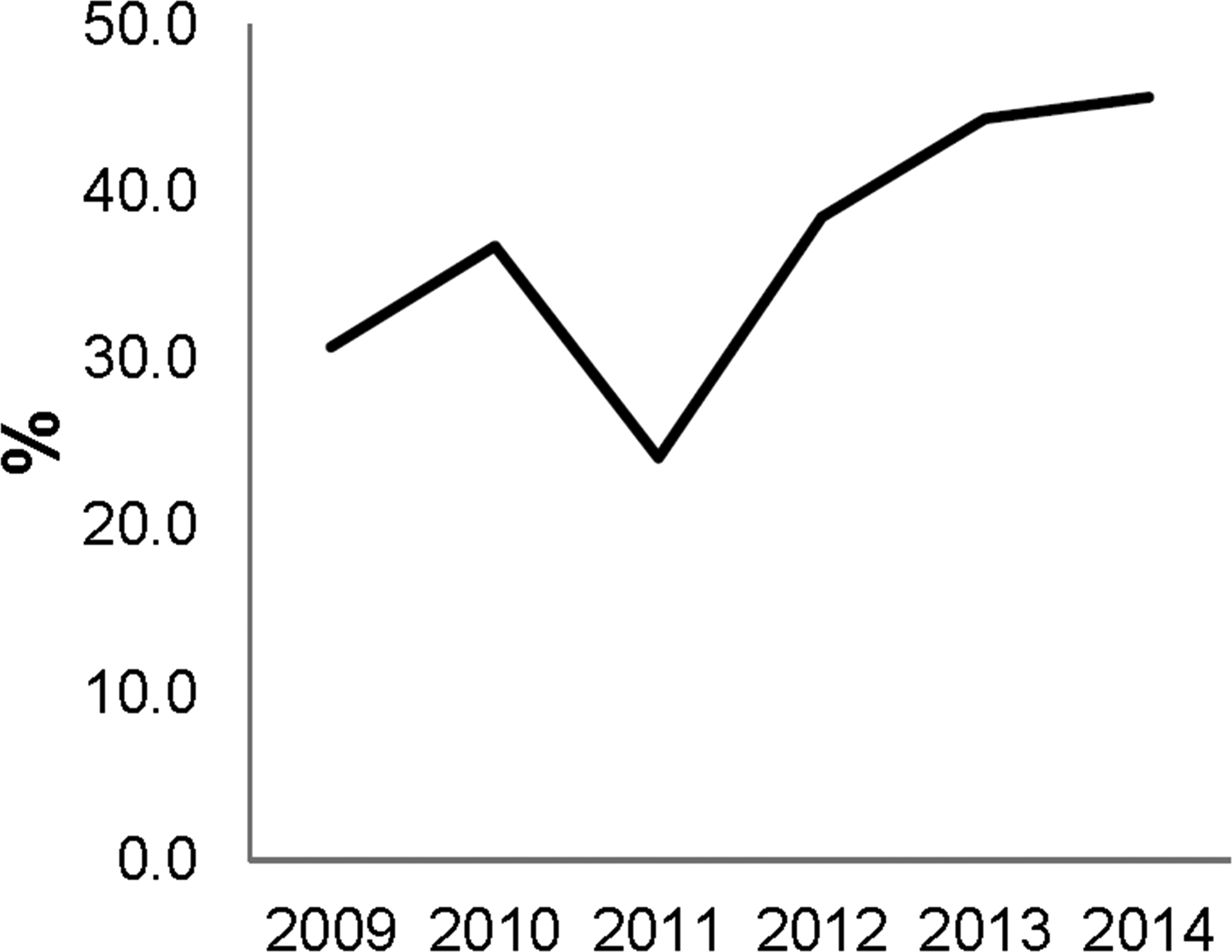
Usage of minipig for dermal route of administration opposed to all other routes of administration at the 27 contract research organization sites interviewed.
As our survey was only reaching out to the larger CROs involved in preclinical safety work, we have compared the annual usage of Goettingen minipigs recorded in this survey with the data on global Ellegaard Goettingen minipig A/S production (including Marshall Bioresources). In 2010, the market share of the CROs interviewed represented up to 60% of the global Goettingen minipig production, while it dropped down to approximately 30% in 2013 (data not shown). With more than 200 customers, the Goettingen minipig is becoming broadly available at universities, pharma, and most CROs. As the minipig becomes a more widespread preclinical model outside the traditional use in pharmaceutical industry, it will be interesting to see whether the proportion of nondermal routes will increase in NA like it is currently happening in EU.
All the major CROs are employing the Goettingen minipig strain, which by its growth rate and sexual maturity trigger its use in toxicology studies, imply a similar body weight range compared to dogs in screening studies and 4-week good laboratory practice (GLP) toxicity studies for first-in-man studies. However for the later development programs supporting chronic use in man, the compound requirements are at least 4-fold compared to a program in dogs (see Figure 4). A recent industry survey (Colleton et al. in press) identified the size of the animals and the consequent test article requirements, an important factor for selecting a nonrodent species. Alternatives are scarce but appear to exist with the “micro minipig” which has been established and proclaims a growth curve which is close to that of a dog as shown in Figure 4.
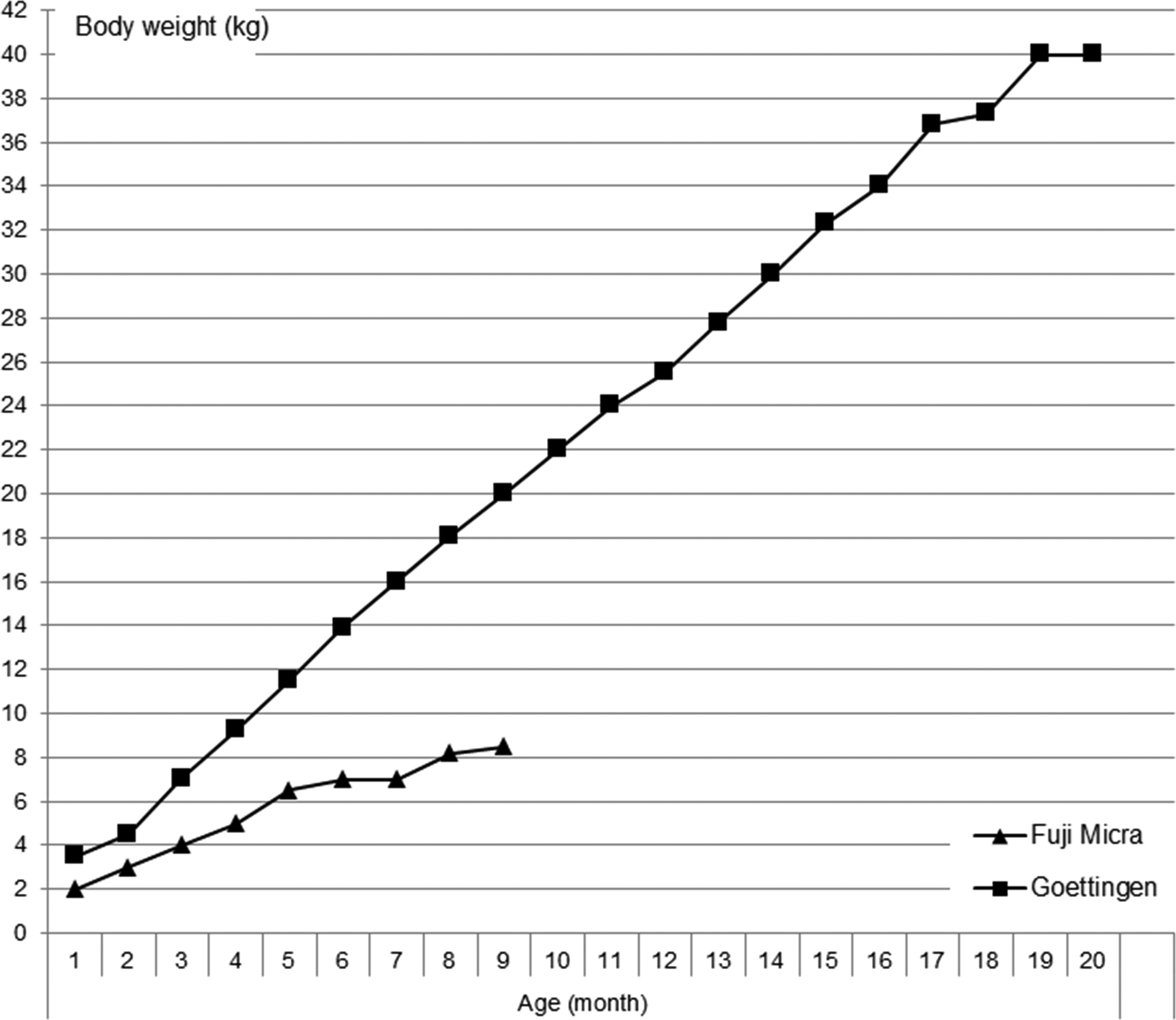
Growth curve of the Goettingen minipig (Ellegaard 2015b) and the Fuji Micro-minipig (Fuji Micra 2015) as presented by the providers in July 2015. Data for the micro-minipig were only available up to the age of 9 months.
Although the usefulness of such model may be shown by some groups (Yoshikawa et al. 2013; Kawaguchi et al. 2012), the drawback in introducing a new strain in safety testing is the need for building up a robust historical database which has posed one of the major hurdles, making the Goettingen minipig an accepted species by the pharmaceutical industry for the last 20 years. New technologies like next gene sequencing may help to bridge data between pig strains but still requires major investments until a new strain would be widely accepted.
Pharmacodynamic response is generally accepted by toxicologists as one of the main driving factors in selecting an appropriate nonrodent species for toxicity testing. This was also confirmed in a recent survey among pharmaceutical industry (Colleton et al. in press) and has been introduced a while ago in a guideline putting a strong focus on demonstrating the relevance of the animal model to predict human safety based on pharmacodynamics principles and pharmacokinetic coherence (EMA 2007). The majority of adverse drug reactions in man are reported to be the so-called type A reactions which are predictable from the known pharmacology of the drug (Park et al. 2000). And such type of reactions were reported to account for approximately 90% of admissions to a department of internal medicine in Sweden (Mjoerndal et al. 2002).
It is important to know which model is not to be used in certain targets and there is an increasing cross-industry effort to share knowledge on animal toxicities more timely (Morris et al. 2010; Goldman, Seigneuret, and Eichler 2015). Interspecies differences may be due to many factors like genetic, biochemical, or metabolic and the disease, genetic setup, and many more complex circumstances in man make it difficult to predict the complete safety profile from animal studies. Among other factors, the availability of tools and technologies to assess safety parameters in a way which is applicable and predictive for man may drive the selection of an animal species for safety assessment. In addition to full capabilities of assessing cardiovascular parameters like electrocardiogram and blood pressure noninvasively and invasively using telemetric devices, the minipig has also been established as a preferred model for urology and neurology research (Huppertz, Tolba, and Grosse 2015). Progress has also been made to establish the pig for research and development of immunomodulatory pathways. Although the NHPs have been used as a preferred animal species to predict the human safety profile of drugs interfering with the immune system, the predictive value has not been overwhelming. This may be partly due to the fact outlined above that there are large differences between different origins of monkeys, for example, relating to major histocompatibility locus genetics. The pig has shown to be of use as an animal model in developing immunosuppressive agents (McDaid et al. 2015), and transgenic colonies with defined histocompatibility complex can be established (Hwang et al. 2015). Based on available knowledge (Bode et al. 2010; Foster et al. 2010) and recently accumulated data, the minipig is a valid alternative nonrodent species for safety testing of immunomodulatory development candidates and the functional similarity and the available tools make it definitely superior to the dog (Kammueller and Rubic in press).
In the past few years, it has become apparent that the characterization of an animal model on the molecular biology and genetic level is a necessity to predict the relevance of a finding in an animal experiment for the human patient. And for NHPs it was shown that the genetic background of the same species, cynomolgus monkey, coming from different regions, can be quite diverse in terms of genetics but also in the variation of the response to drug targets (Moggs 2012). The available data so far are comprising different pig strains like the domestic pig (Freeman et al. 2012; Groenen et al. 2012), a Chinese pig strain (Fang et al. 2012), or the Goettingen minipig (Vamathevan et al. 2013). Closeness to human protein sequences, particularly those linked with multifactorial diseases were postulated (Groenen et al. 2012), and it has been found that the overall structural similarity of pig proteins was significantly closer to human than this was the case for the mouse (Dawson 2012). Comparing genomic information from the different pig strains, it became evident that for biomedical research and safety prediction to man it is important to have a gene atlas available from the exact breed one is using (Larzul 2013). Work is in progress to establish gene maps and data banks of the strains used in biomedical research by pharmaceutical companies (e.g., Novartis, unpublished information). Recent advances in preclinical safety testing have postulated the use of disease models and/or genetically engineered animal models for safety testing including pivotal studies for first-in-human trials, for example, for enzyme replacement therapy (FDA 2015).
The availability of appropriate technologies and experience with setting up transgenic animal models clearly speaks in favor of the minipig. Efficient and reliable techniques for the genetic modification of pigs have been established and can be based on a variety of approaches such as DNA microinjection, sperm-mediated gene transfer, lentiviral transgenesis, and somatic cell nuclear transfer using genetically modified nuclear donor cells (Aigner et al. 2010). Transgenic pigs are increasingly offered as models for human disease in the field of neurology (Alzheimer’s and Huntington’s), ophthalmology (macular dystrophy), cardiovascular, metabolic (liver, diabetes), respiratory (cystic fibrosis), cancer, or immunodeficiency (Aigner et al. 2013; Kemter et al. 2012). Improvement in animal models for human safety assessment will be a major challenge in drug development, and this also applies to models that have been thought to be unsurmountable like the NHP model, which is considered the only relevant species for safety testing of recombinant proteins or monoclonal antibodies and that have demonstrated limited value and predictivity of patients (Van Meer, Graham, and Schuurman 2015).
In combination with the safety read outs from toxicology studies, pharmacokinetics and metabolism and its closeness to the human prediction are one of the major cornerstones of successful safety predictions of animal models. Accumulating data propose the use of the minipig as a species of choice among other nonrodent species like dog or monkey, and certain cases like involvement of aldehyde oxidase or cytochrome P450 enzyme, subclass 2C9-like pathways suggest it as clearly superior to the dog (Dalgaard 2015). For chemical entities that are metabolized via non-cytochrome P 450 enzymes it is important to have appropriate information for the selection of the appropriate nonrodent species, and data are available for pigs and minipigs (Oda et al. 2015). And the picture may slightly differ between strains (Skaanild 2014), which underscores the need to work with the most appropriate and well-characterized pig strain which the investigator needs to determine based on the properties of a given molecule. For a better prediction on how to translate
For parenteral use and biotherapeutics like monoclonal antibodies, the minipig has not yet picked up its share in spite of the scientific community never having disputed its usefulness. As shown by Colleton et al (in press), pharmaceutical industry is still reluctant to generate the necessary supporting data like cross-reactivity and immunogenicity in the discovery phase. The potential to combine general and reproductive toxicity studies into one species with much higher convenience compared to the NHP model could be an incentive for using the minipig. However, the absence of maternally derived antibodies in the newborn piglet did exclude considerations of using minpigs for reproductive toxicity studies with monoclonals. New data are emerging which may lead to changes in strategy as therapeutic antibodies may have higher affinity to neonatal fragment crystallizable (of an antibody) receptor (FcRn), and it was shown that FcRn is expressed in the fetal intestine of minipigs which may enable the uptake of immunoglobulins (Jacobsen et al. in press).
In order to achieve sufficient systemic exposure in toxicity studies, it is important to find appropriate formulations of a potential drug candidate that are well tolerated in the desired animal species. Fortunately, the knowledge on formulations which can be used in minipigs is growing and reached a milestone with the publication by Weaver et al. (in press).
In summary, the minipig has been established in pharmaceutical industry as a valuable nonrodent species for safety testing with convincing scientific arguments demonstrating its closeness to human biophysiology in many aspects and making it highly competitive versus dogs or monkeys. As shown by the survey and the data from the supplier indicating a dilution of the market share held by the larger CROs, the minipig is now broadly available at most preclinical CROs and also outside of EU. Reagents and biomarkers have been developed that make it appear superior to the dog for development of drugs with immune-modulatory properties. The main pitfall for its underusage lies in the fact that the minipig is not routinely included in screens for pharmacological responsiveness or pharmacokinetic behavior of a drug. Based on the accumulating evidence from many labs doing research in pigs, it is high time to close this gap and include pigs as a species to be thoroughly evaluated when it comes to selection of the most appropriate nonrodent species.
Footnotes
Authors’ Contribution
Authors contributed to conception or design (PH, TR); data acquisition, analysis, or interpretation (TR); drafting the manuscript (PH, TR); and critically revising the manuscript (PH, TR). All authors gave the final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
