Abstract
Developmental toxicity testing of therapeutic antibodies is most often conducted in nonhuman primates owing to lack of cross-reactivity in other species. Minipigs may show cross-reactivity for some humanized antibodies but have not been used for developmental toxicity testing due to an assumed lack of embryo-fetal exposure. Unlike in humans, maternal IgGs do not cross the porcine placenta to reach the fetus. Some humanized IgGs, however, have a higher affinity for the neonatal Fc receptor (FcRn) and are more likely than endogenous antibodies to cross the placenta of animals. The major site of prenatal IgG transfer is the placenta, though FcRn in fetal intestine could also uptake maternal IgGs from swallowed amniotic fluid. Using immunohistochemistry and in situ hybridization in this experiment, FcRn was found in minipig placenta and fetal intestine during early, mid-, and late gestation. To date, however, fetal exposure to maternally administered IgGs has never been demonstrated in the minipig.
Various model species have been used or proposed for the developmental toxicity testing of therapeutic antibodies, including nonhuman primates (NHPs), rodents, and rabbit (Bowman et al. 2013). While the minipig is a suitable species for the teratology testing of drugs (McAnulty 2013), it has been largely excluded from consideration for the developmental toxicity testing of therapeutic antibodies due to an assumed lack of embryo-fetal exposure owing to the absence of a prenatal transport system for immunoglobulins (Pentsuk and van der Laan 2009). This assumption was based on the observation that the minipig has little or no circulating titers of maternal antibodies at birth, unlike humans and the species cited earlier. Nonetheless, trace levels of IgG—thought to be maternal origin—have been reported in porcine fetuses from gestation day (GD)50 onward, which then decline in germ-free piglets within 1 week after birth (Butler et al. 2002).
The difference in prenatal transport of maternal antibodies to the fetus is influenced by the differences in placentation between species. Primates, rodents, and rabbits have a hemochorial placenta, while pigs have an epitheliochorial placenta. The porcine placenta has the most superficial implantation with the greatest degree of separation between the maternal and the embryonic circulation consisting of 6 epitheliochorial layers: endothelium of maternal blood vessels, surrounding connective tissue, overlying endometrium in close interdigitation with the fetal trophectoderm, and underlying connective tissue with embedded fetal blood vessels. In contrast, the hemochorial placenta has a much more invasive implantation in which all maternal tissue layers have eroded, allowing a direct connection between the three-layered chorion (trophoblasts, embryonic connective tissue, and capillary endothelium) and maternal blood (DeSesso et al. 2012; Furukawa, Kuroda, and Sugiyama 2014). With 6 layers of epitheliochorial separation, the placenta of the pig is thought to be impermeable to the passage of immunoglobulins throughout the whole period of gestation. There are essentially no maternally derived antibodies in the newborn piglet, and maternal antibodies are transferred via the colostrum during the first 6 hr after birth (Bode et al. 2010). The neonatal Fc receptors (FcRn) are required for the active uptake and transport of IgGs. To date, FcRn receptors have not been reported in the minipig placenta or conceptus, which would discount prenatal IgG transfer. The major site of prenatal IgG transfer is the placenta, though FcRn receptors in the fetal intestine could also uptake maternal IgGs from the amniotic fluid swallowed by the fetus (DeSesso et al. 2012).
Materials and Methods
FcRn profiling investigations were conducted in minipig placenta and fetal jejunum samples collected at different time points of gestation. Sexually mature female Göttingen minipigs (donated by Ellegaard Göttingen Minipigs A/S, Denmark) of 6–7 months of age were housed in pairs in double-spaced indoor kennels in an air-conditioned animal room (22°C ± 3°C) with a 12-hr light/dark cycle environment and background music coordinated with light hours in an Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)-accredited facility (WIL Research Europe-Lyon, France). They were provided with standard minipig diet and tap water ad libitum. All animals were observed at least during the feeding period to ensure they were in good health. All procedures were in accordance with national and European animal health and welfare guidelines (Decree n° 2013-118; Directive 2010/63/EU). For mating, the females received Regumate (Altrénogest, 20 mg/animal) in the diet for at least 18 days during the acclimatization period. This was stopped 5 days before mating with a male of similar age. The first day of mating was considered the first day of gestation (GD0). After sedation, 3 females each were euthanized at early gestation (GD35), mid-gestation (GD60), and at the end of gestation (GD108 or GD109) by intravenous injection of sodium pentobarbitone followed by exsanguination. Samples were collected from the placenta and the jejunal intestine of up to 2 fetuses per sow. The tissues were fixed in 10% buffered formalin, routinely embedded in paraffin, and 5-µm sections were cut and deparaffinized. Serial sections were taken for immunohistochemistry (IHC) and in situ hybridization (ISH).
The protein expression of FcRn was evaluated by IHC. Slides were stained with the Ventana Omnimap DAB Detection Kit using the avidin–biotin peroxidase complex method (brown staining) involving a biotinylated secondary donkey anti-rabbit antibody in a Ventana Discovery XT® immunostainer (Ventana Medical Systems, Inc., Tucson, AZ). The primary antibody used was a prediluted (1:50) polyclonal rabbit anti-FcRn antibody (Novus biological, NBP1-89128). The slides were counterstained with hematoxylin. For negative control, the primary antibody was omitted.
The transcription of the corresponding messenger RNA (mRNA) of Fc fragment of IgG receptor transporter alpha (FCGRT) was detected using RNAscopeVS® (Advanced Cell Diagnostics, Inc., Hayward, CA), an RNA ISH method that permits signal amplification and background suppression. A cocktail of FCGRT-specific RNA target probe sets for the pig (Sus scrofa) were provided by the manufacturer (Entrez Gene ID 397399, targeted base pairs 371 through 1,482 of the FCGRT complementary DNA sequence). In brief, after pretreatment of slides with protease according to the manufacturer’s instructions, hybridization was performed at 43°C for 3 hr. Following signal amplification, slides were stained with Fast Red (red staining) and counterstained with hematoxylin. All steps of this procedure were carried out using a Ventana Discovery Ultra® immunostainer (Ventana Medical System, Tucson, AZ). Probes to the bacterial gene dihydrodipicolinate reductase (DapB) and the endogenous peptidylprolyl isomerase B (PpiB) mRNA were used as negative and positive controls, respectively. Specific staining signals were identified as red, punctate dots present in the cytoplasm. The results are summarized in Table 1 and illustrated in Figures 1 and 2.
Results of IHC and ISH.
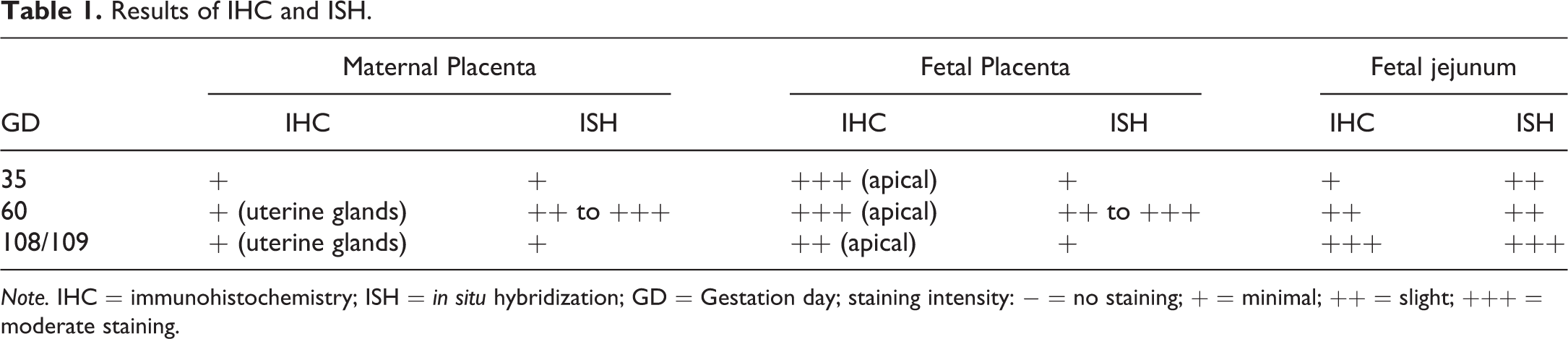
Note. IHC = immunohistochemistry; ISH = in situ hybridization; GD = Gestation day; staining intensity: − = no staining; + = minimal; ++ = slight; +++ = moderate staining.
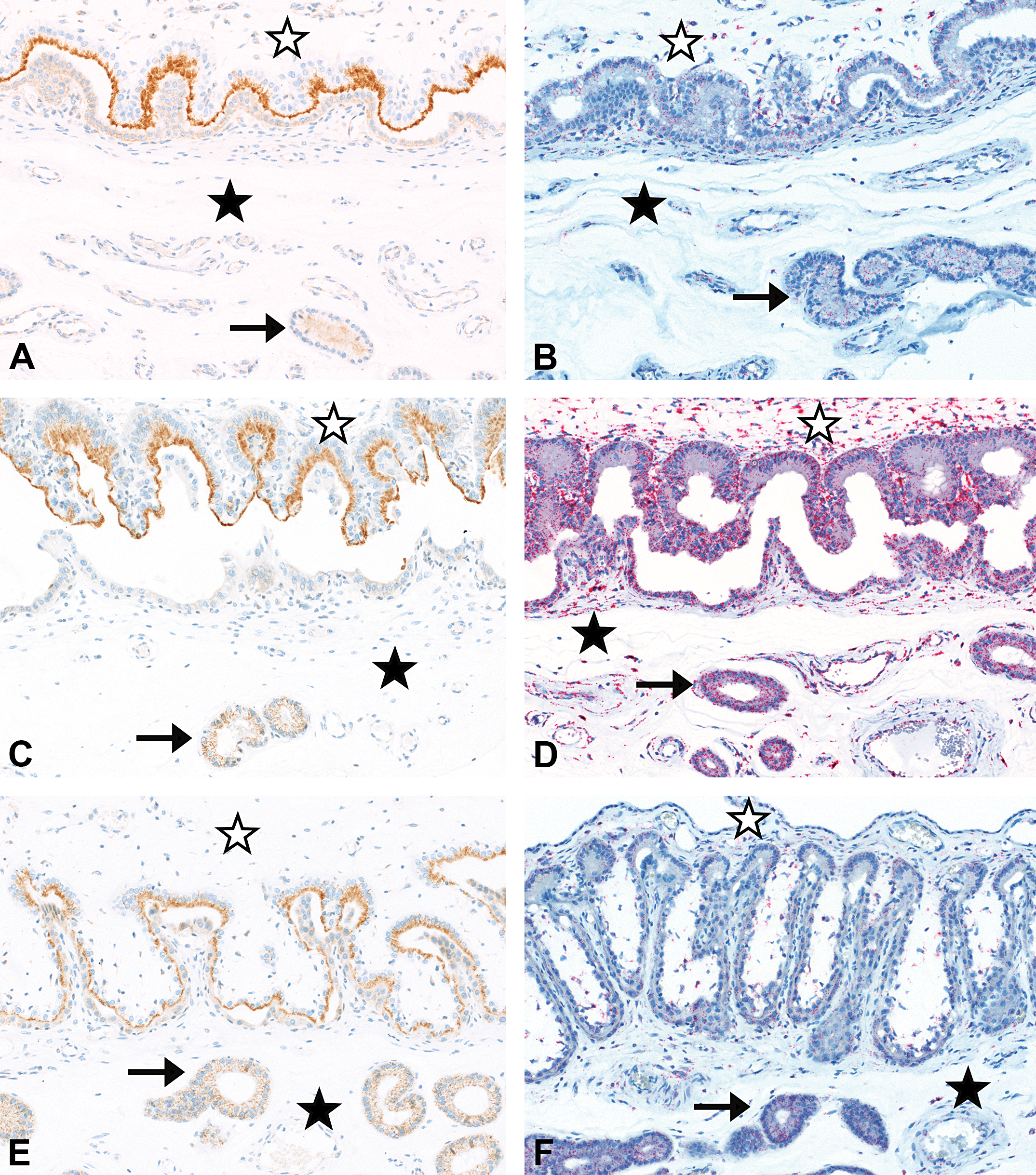
Placenta from gestation day (GD)35 (A and B), GD60 (C and D), and GD108 (E and F). Moderate (A and C) to slight (E) staining of FcRn in apical part of fetal trophoblasts by immunohistochemistry (IHC); no to minimal staining in maternal endometrium and minimal staining in uterine glands epithelium (A, C, and E). Minimal (B and F) and slight to moderate (D) staining of Fc fragment of IgG receptor transporter alpha (FCGRT) by in situ hybridization (ISH) in fetal and maternal placenta including uterine glands. IHC with an antibody directed against FcRn, ISH with a probe directed against porcine FCGRT, maternal placenta (filled asterisk), uterine glands (arrow), and fetal placenta (empty asterisk). Original objective used 20×.
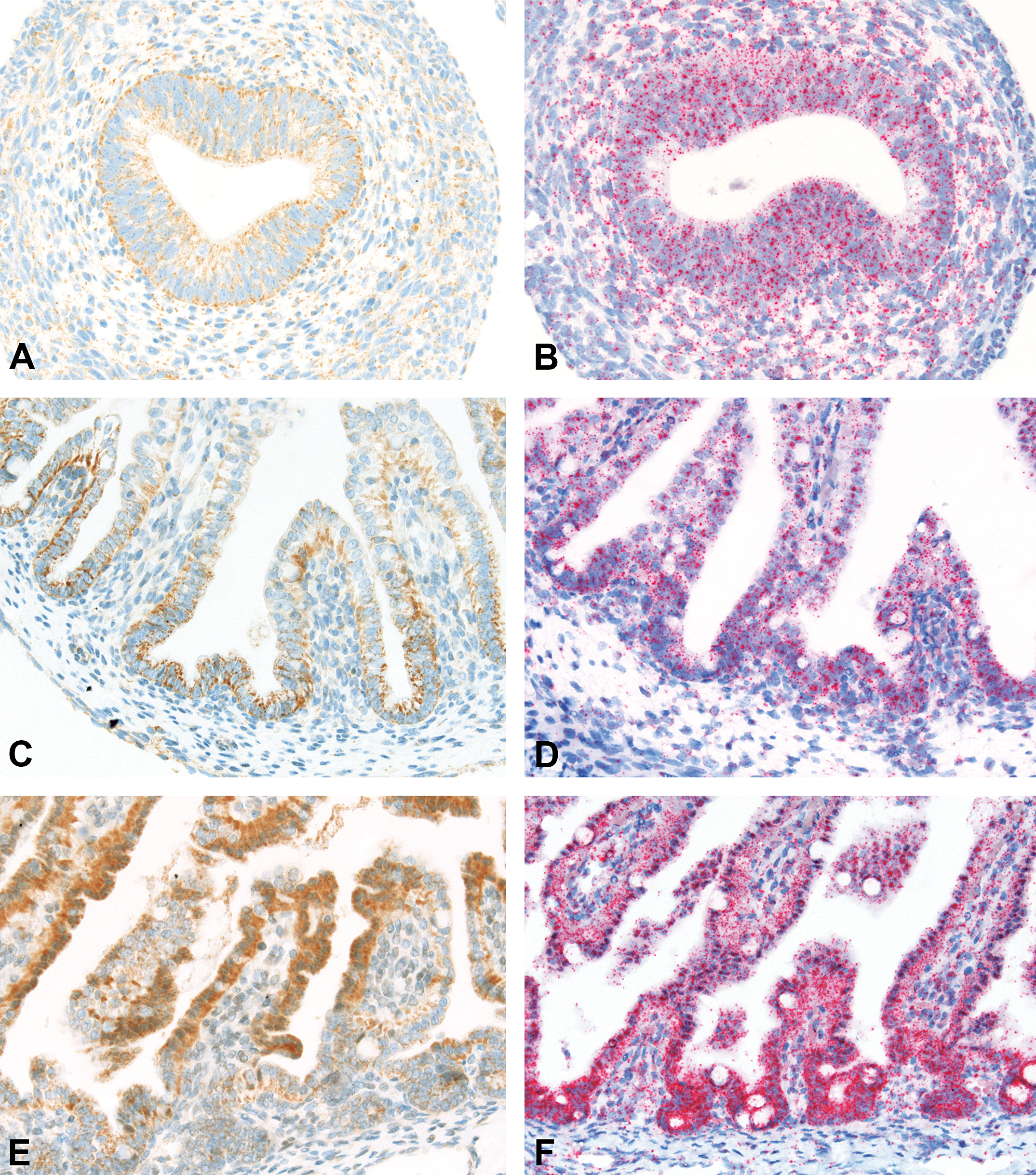
Jejunal intestine from fetuses at gestation day (GD)35 (A and B), GD60 (C and D), and GD108 (E and F). Minimal (A), slight (C), and moderate (E) staining of FcRn in jejunal enterocytes by immunohistochemistry (IHC). Slight (B and D) and moderate (F) staining of Fc fragment of IgG receptor transporter alpha (FCGRT) in jejunal enterocytes by in situ hybridization (ISH) with additional minimal staining of individual cells in the underlying lamina propria. IHC with an antibody directed against FcRn, ISH with a probe directed against porcine FCGRT. Original objective used 40×.
In the fetal placenta, IHC revealed the presence of FcRn in the fetal trophoblasts at all 3 gestation time points (Figure 1). The staining was in the apical part of the trophoblasts with a moderate intensity on GD35 and GD60 and a slight staining intensity on GD108/109. In contrast, no or only minimal FcRn was detectable by IHC in the endometrium of the maternal placenta. From GD60 onward, there was a minimal staining present in the epithelium of the uterine glands (Figure 1).
ISH revealed the presence of FCGRT mRNA in both fetal and maternal placenta including uterine glands at all 3 gestation time points (Figure 1). The staining intensity was minimal on GD35, slight to moderate on GD60, and minimal again on GD108/109.
In the fetal jejunum, IHC revealed the presence of FcRn in the enterocytes at all 3 gestation time points, with increasing staining intensity toward the end of gestation (Figure 2). FcRn staining was present in the crypts of the epithelial cells from GD35, becoming more intense on GD60 and migrating to the proximal ends of the villi by GD108/109.
By ISH, FCGRT mRNA was present in the jejunal enterocytes and in the individual cells in the underlying lamina propria at all 3 gestation time points investigated (Figure 2). The staining intensity was slight on GD35 and GD60, and moderate at GD108/109 with a more pronounced staining in the crypts at the latter time point.
The protein expression of FcRn and transcription of the corresponding mRNA of FCGRT were demonstrated by IHC and ISH, respectively, in the minipig placenta and fetal intestine. To our knowledge, this is the first report of FcRn expression in the minipig placenta and fetal intestine. This previously unreported presence of FcRn in the porcine placenta could explain the trace levels of maternal IgGs found in minipig fetuses (Butler et al. 2002).
The presence of FcRn in placenta and fetal intestine in the minipig is comparable to that in species with a hemochorial placenta, including the human. FcRn has been shown to be present in rodent and human syncytiotrophoblasts from the late first trimester onward (Simister 2003). Similarly, FcRn has been shown to be present in enterocytes of human fetuses (Shah et al. 2003).
In the pig, there are essentially no maternally derived antibodies in the newborn piglet, and endogenous maternal antibodies are transferred to the piglet postnatally in the colostrum (Bode et al. 2010). FcRn was shown to be present in the mammary gland of the sow, where it assures the transport of IgGs into the milk (Schnulle and Hurley 2003), and a corresponding FcRn expression is necessary in the neonatal intestine of the piglet to assure absorption of the maternal IgGs. The current evaluation demonstrates the prenatal expression of FcRn in the fetal intestine by IHC and ISH. This early FcRn expression may provide a nonplacental route by which maternal IgGs could be absorbed by the fetus following swallowing of the amniotic fluid (Shah et al. 2003; DeSesso et al. 2012).
Recent experiments have shown that some humanized therapeutic antibodies have a higher FcRn binding affinity in rodents and NHPs than the endogenous equivalent (Andersen et al. 2010; Giragossian et al. 2013). Thus, the titers of endogenous antibodies at birth in model species may underestimate the extent of embryo-fetal exposure to maternally administered humanized IgGs (DeSesso et al. 2012). There are, however, only limited data available on the binding affinity of porcine FcRn for human or humanized antibodies (Zheng et al. 2012) and none for the relative affinity of endogenous porcine IgG versus humanized antibodies. Nonetheless, the presence of FcRn in the minipig placenta revealed in the current investigation raises the possibility of transplacental transport of therapeutic IgGs in the minipig.
There was a discrepancy between FcRn protein demonstrated by IHC and FCGRT mRNA demonstrated by ISH. Although mRNA detected by ISH does not necessarily indicate protein expression, the discrepancy could also be due to a greater sensitivity of ISH over IHC. Standard fixation could cause antigenic alterations resulting in a decreased sensitivity of IHC to detect protein expression (Pauletti et al. 2000). Nonetheless, the presence of FcRn in the epithelium of the uterine glands shown by IHC provides the possibility for transfer toward the fetal placenta. To date, however, functional prenatal transport and fetal exposure to maternally administered immunoglobulins have never been demonstrated in minipigs. Further work is therefore required to assess the levels of placental or intestinal transfer of antibodies during pregnancy and thereby confirm or dismiss the suitability of the minipig as a model species for prenatal developmental toxicity testing of Fc-containing biopharmaceuticals.
Footnotes
Acknowledgments
The authors would like to thank Christelle Zundel, Rachel Neff, and Virginie Ott for their excellent technical support for IHC and ISH, and Ellegaard Göttingen Minipigs A/S, Denmark, for donating the minipigs.
Author Contribution
Authors contributed to conception or design (BJ, MH, LR, AH, PB); data acquisition, analysis, or interpretation (BJ, MH, LR, AH, PB); drafting the manuscript (BJ); and critically revising the manuscript (MH, LR, AH, PB). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
