Abstract
Genetically engineered pigs are increasingly recognized as valuable models for the study of human disease. Immunohistochemical study of cellular markers of disease is an important tool for the investigation of these novel models so as to evaluate genotype and treatment differences. Even so, there remains a lack of validated markers for pig tissues that can serve as a translational link to human disease in organs such as the lung. Herein, we evaluate markers of cellular inflammation (cluster of differentiation [CD]3, CD79a, B cell lymphoma [BCL] 6, ionized calcium-binding adapter molecule [IBA]1, and myeloperoxidase) and those that may be involved with tissue remodeling (alpha-smooth muscle actin, beta-tubulin-III, lactoferrin, mucin [MUC]5AC, MUC5B, and cystic fibrosis transmembrane conductance regulator [CFTR]) for study of lung tissues. We compare the utility of these markers between pig and human lungs to validate translational relevance of each marker. Our results suggest these markers can be a useful addition in the pathological evaluation of porcine models of human disease.
Introduction
Pigs have traditionally been a useful species to model pathophysiology to better understand human biology (Swindle et al. 2012). In particular, the pig lung has been valuable to study normal physiology (Sommerer et al. 2004) as well as developmental (Glenny et al. [1985] 2007), infectious (Rajao and Vincent 2015), and environmental/toxicological diseases (Gushima et al. 2001; Grainge et al. 2010). The similar lung size, anatomy, and physiology to humans along with the longevity of pigs compared to smaller rodents make the pig a useful species to model human lung diseases (Rogers, Abraham, et al. 2008; Aigner et al. 2010).
In recent years, development of genetically modified pigs has been reported for several human diseases including cystic fibrosis (CF; Ostedgaard et al. 2011; Rogers, Stoltz, et al. 2008; Stoltz et al. 2010; Stoltz et al. 2013; Olivier, Gibson-Corley, and Meyerholz 2015), cancer (Sieren et al. 2014), muscular dystrophy (Klymiuk et al. 2013), atherosclerosis (Davis et al. 2014), and diabetes mellitus (Renner et al. 2010). The success of these models has spurred further interest in pigs as a model species for translational studies; however, one potential limitation, compared to small rodent models, is the relative lack of available and validated reagents, assays, and techniques for translational use in pigs with comparison to humans (Olivier et al. 2012). Antibodies and validated immunohistochemistry techniques are useful for pathological evaluation.
In some cases, human markers have been successfully applied to pig tissues (Meyerholz et al. 2010), and these techniques can serve as a guide for use in investigational studies. For antibodies, commercial vendors will often list species that are compatible for immunohistochemistry; however, these endorsements are frequently based upon predicted sequence identity or on anecdotal reports from the vendor’s clients—both of these approaches lack robust substantiation. Importantly, there is currently a lack of direct comparison of lung tissue markers between pigs and humans. To this end, we have begun optimizing several immunohistochemical techniques for direct translational use in pig and human tissues.
Investigational studies of the lung often evaluate cellular inflammation (Gauger et al. 2012; Mirakaj et al. 2014; Posa et al. 2013) and tissue remodeling as parameters (Aguayo 1994; Wright, Meurs, and Dekkers 2014) for end point assessment. Accordingly, we used optimized markers of cellular inflammation (cluster of differentiation [CD]3, CD79a, B cell lymphoma [BCL]6, ionized calcium-binding adapter molecule [IBA]1, and myeloperoxidase) and remodeling (alpha-smooth muscle actin, beta-tubulin-III, lactoferrin, mucin [MUC]5AC, MUC5B, cystic fibrosis transmembrane conductance regulator [CFTR]) in the pig lung to evaluate their utility for translational applications to human lung.
Materials and Methods
Tissues
All pig and human tissues were collected from archival formalin-fixed paraffin-embedded blocks. Pig tissues came from studies that had received the University of Iowa Institutional Animal Care and Use Committee (IACUC) approval. Human tissues were acquired through the Cell Culture Core Repository (University of Iowa), which has received institutional approval from the University of Iowa Institutional Review Board (IRB #:199507432) for collection of human tissues. Comparisons for each cellular marker were made from at least 3 samples from respective porcine (2–6 months of age) and human tissues (adults, 3rd–4th decade). Unless otherwise specified, tissues came from healthy individuals lacking overt clinical disease. Tissues were selected for each marker that was deemed most useful to demonstrate efficacy (e.g., tonsil to highlight CD3 staining) and when possible the “healthy” lung (lacking clinical evidence of overt or chronic disease) was chosen as the tissue of choice. CF lung tissues immunostained for neutrophils came from a 2-month-old pig that had a homozygous null mutation and from a human that was homozygous for the ΔF508 mutation.
Immunohistochemistry
All tissues included in the study were routinely fixed in 10% neutral buffered formalin for ∼4–7 days, with the first few days on a rotating plate to better fix the samples (Olivier et al. 2012). Tissues were routinely processed and paraffin embedded and then sectioned (∼4 µm) onto glass slides (Superfrost Plus Microscope Slides; Fisher Scientific Co., Pittsburgh, PA) for antigen retrieval and application of immunohistochemistry (IHC) reagents (Supplemental Table 1). Tissue chromogen (3,3′-Diaminobenzidine [DAB], brown color) and counterstain (Surgipath Harris Hematoxylin; Leica Biosystems, Richmond, IL) were applied and then followed by routine dehydration and the coverslip was placed on it. Tissues were evaluated and scored following the principles of histopathological scoring and by using an ordinal scoring system (Gibson-Corley, Olivier, and Meyerholz 2013).
Results
Evaluation of lymphoid markers in lymphoid tissues showed CD3+ cells in T-cell-rich zones, CD79a+ cells in B-cell-rich and germinal center zones, and BCL6+ cells exclusively in germinal centers (Figure 1A–F). These markers appeared to be very similar between species with the exception of slightly less-intense staining by CD79a in porcine B cells (Table 1).
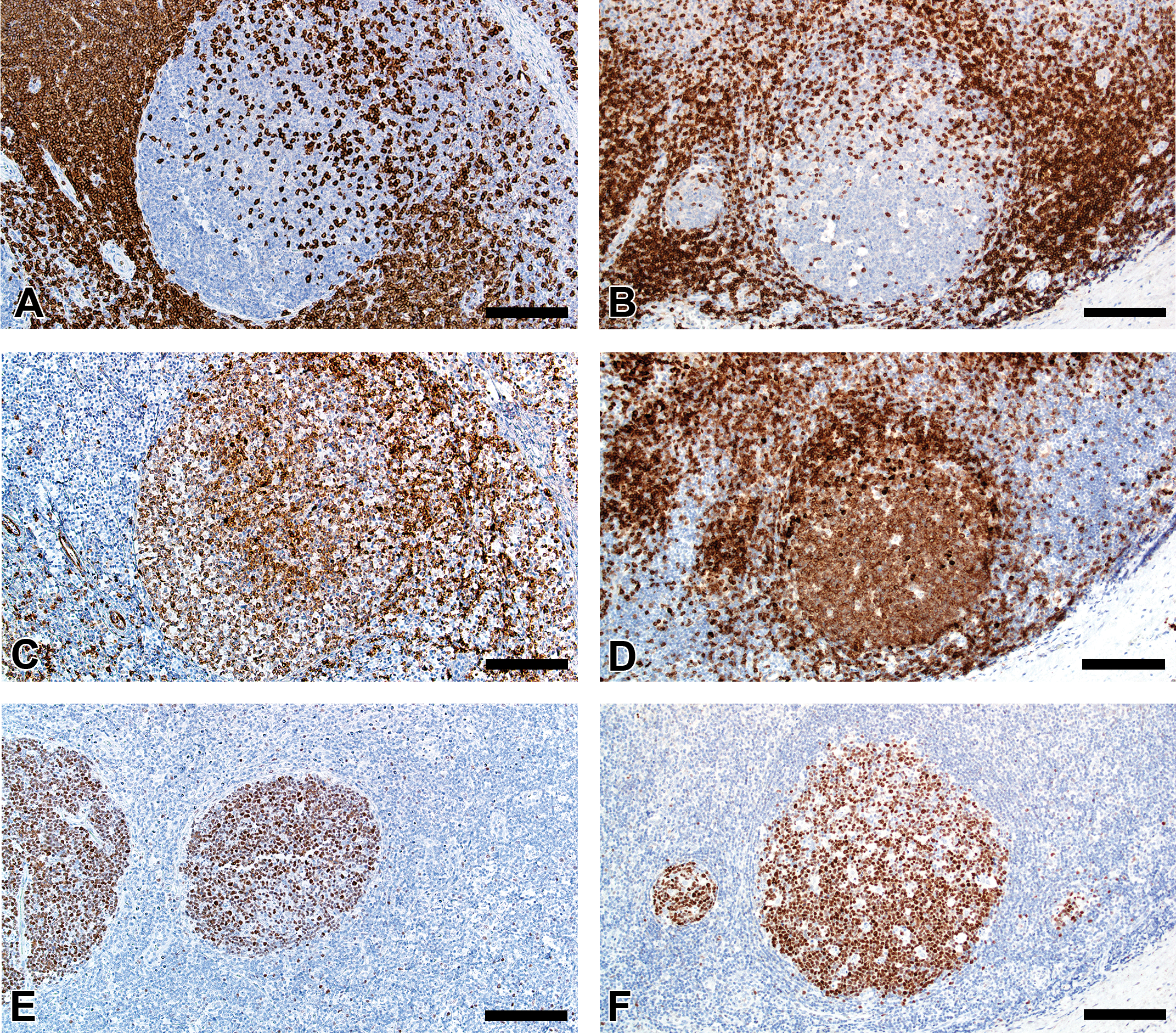
Immunostaining of healthy pig lymph node (A, C, and E) and human tonsil (B, D, and F) for CD3 (A and B), CD79a (C and D), and BCL6 (E and F). Bars = 160 μm.
Immunohistochemical Markers, Tissues, Immunostaining Scores, and Localization.
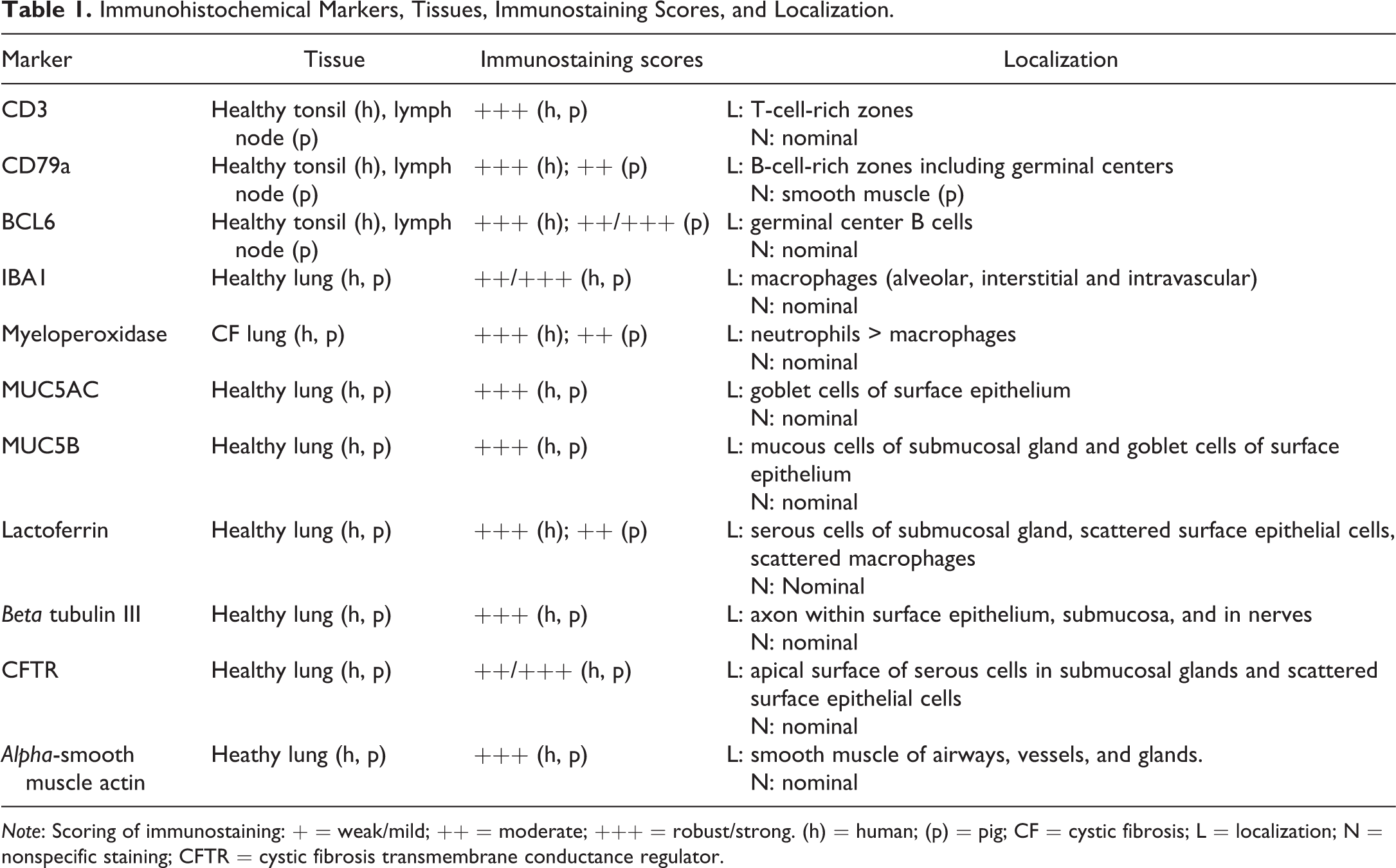
Note: Scoring of immunostaining: + = weak/mild; ++ = moderate; +++ = robust/strong. (h) = human; (p) = pig; CF = cystic fibrosis; L = localization; N = nonspecific staining; CFTR = cystic fibrosis transmembrane conductance regulator.
In CF lungs, myeloperoxidase had robust staining at sites of neutrophilic inflammation (Figure 2A–B) and in areas of macrophages, findings consistent with both being myeloid lineage leukocytes (Table 1). IBA1 had moderate to intense immunostaining of alveolar macrophages within the airspaces (Figure 2C–D), and staining within the septal walls consistent with interstitial or intravascular macrophages (Table 1; Cai et al. 2014; Winkler 1988).
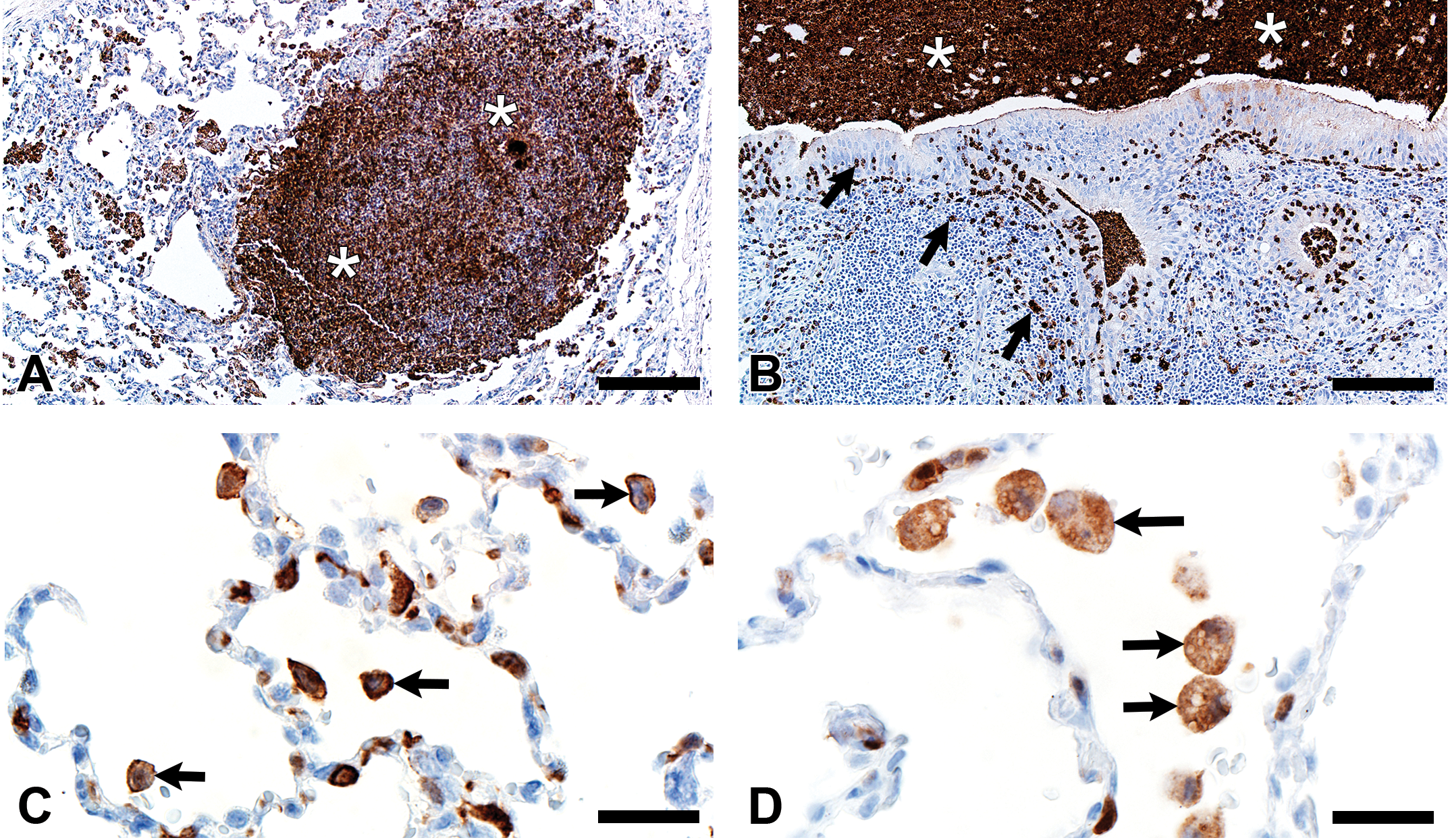
Immunostaining of lung from pig (A and C) and human (B and D). A and B, Aggregates of myeloperoxidase+ (MPO+) neutrophils were detected within airspaces (asterisks, A and B) of cystic fibrosis lungs. Note the MPO+ neutrophils exocytosing across the surface epithelium and airway wall (arrows, B). C and D, IBA1+ alveolar macrophages (arrows) were localized within airspaces and IBA+ cellular staining was also detected within the septal walls of healthy lungs. Bars = 26 μm.
Mucus and submucosal gland secretions are common contributors to several airway diseases including CF, asthma, chronic obstructive pulmonary disease (COPD), and chronic mucus hypersecretion associated with smoking (Rajavelu et al. 2015; Stoltz, Meyerholz, and Welsh 2015; Dijkstra et al. 2015). MUC5AC immunostaining was found in goblet cells of the surface epithelium (Figure 3A–B, Table 1), while MUC5B was seen in mucous cells of submucosal gland and goblet cells of the surface epithelium (Figure 3C–D). In contrast, lactoferrin was detected principally in serous cells of the submucosal glands (Figure 3E–F) and in scattered epithelial cells of the surface epithelium.
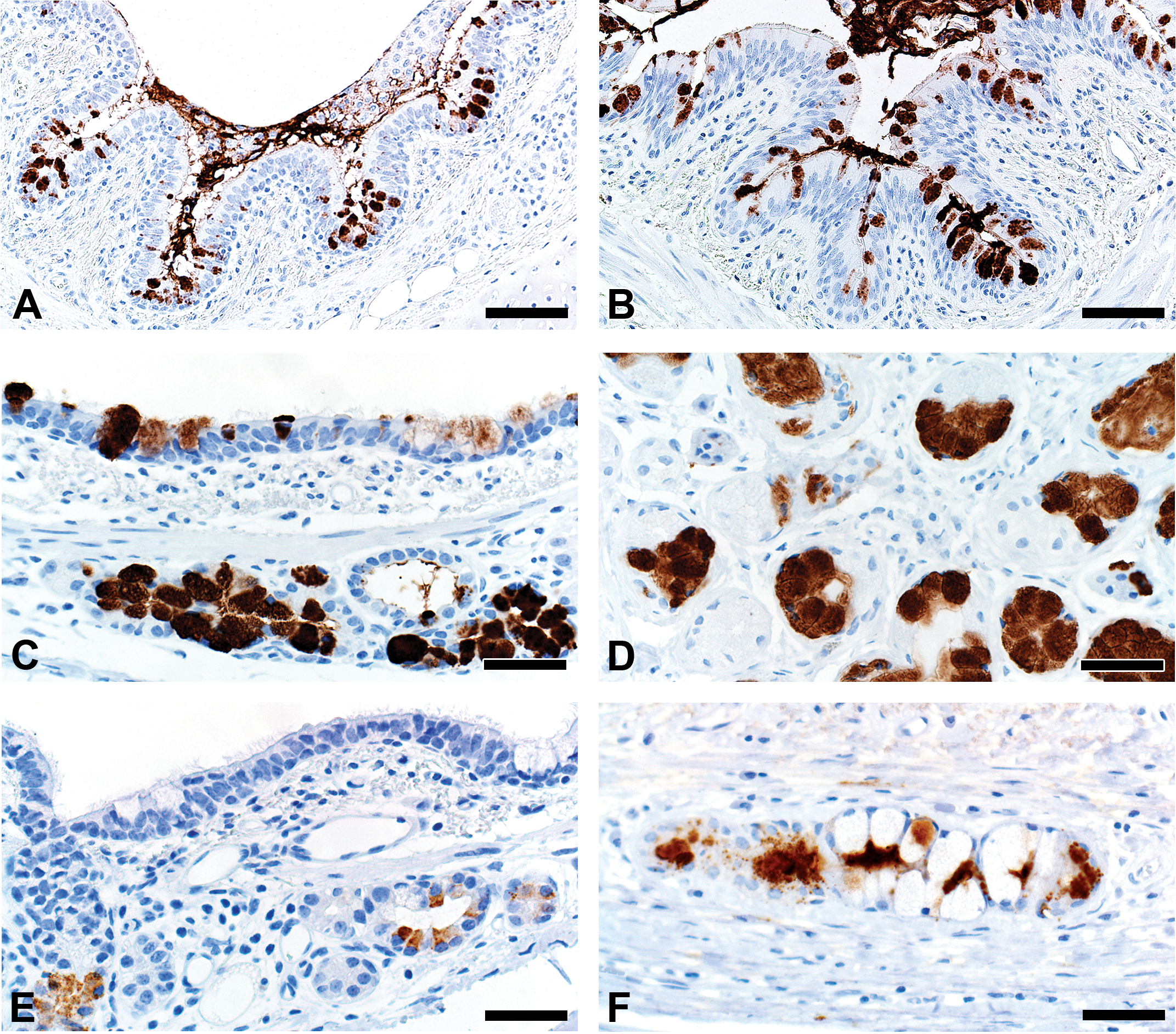
Immunostaining of healthy lung from pig (A, C, and E) and human (B, D, and F). A and B, MUC5AC immunostaining of goblet cells of the surface epithelium. C and D, MUC5B immunostaining of mucous cells in the submucosal gland (C and D) and goblet cells in the surface epithelium (C). E and F, Lactoferrin immunostaining of serous cells in the submucosal gland. Bars = 80 (A,B) and 40 (C-F) μm.
Alpha-smooth muscle actin was detected in airway smooth muscle (Figure 4A–B, Table 1) as well as the tunica media of vessels of both pig and human tissue. Beta-tubulin-III, a marker of axons, was detected in axons within nerves and scattered within submucosal tissues and within the epithelium (Figure 4C–D) where it extended to near the luminal surface. CFTR, an anion channel, was detected on the apical surface of scattered surface epithelial cells and apically on serous cells of the submucosal glands (Figure 4E–F).
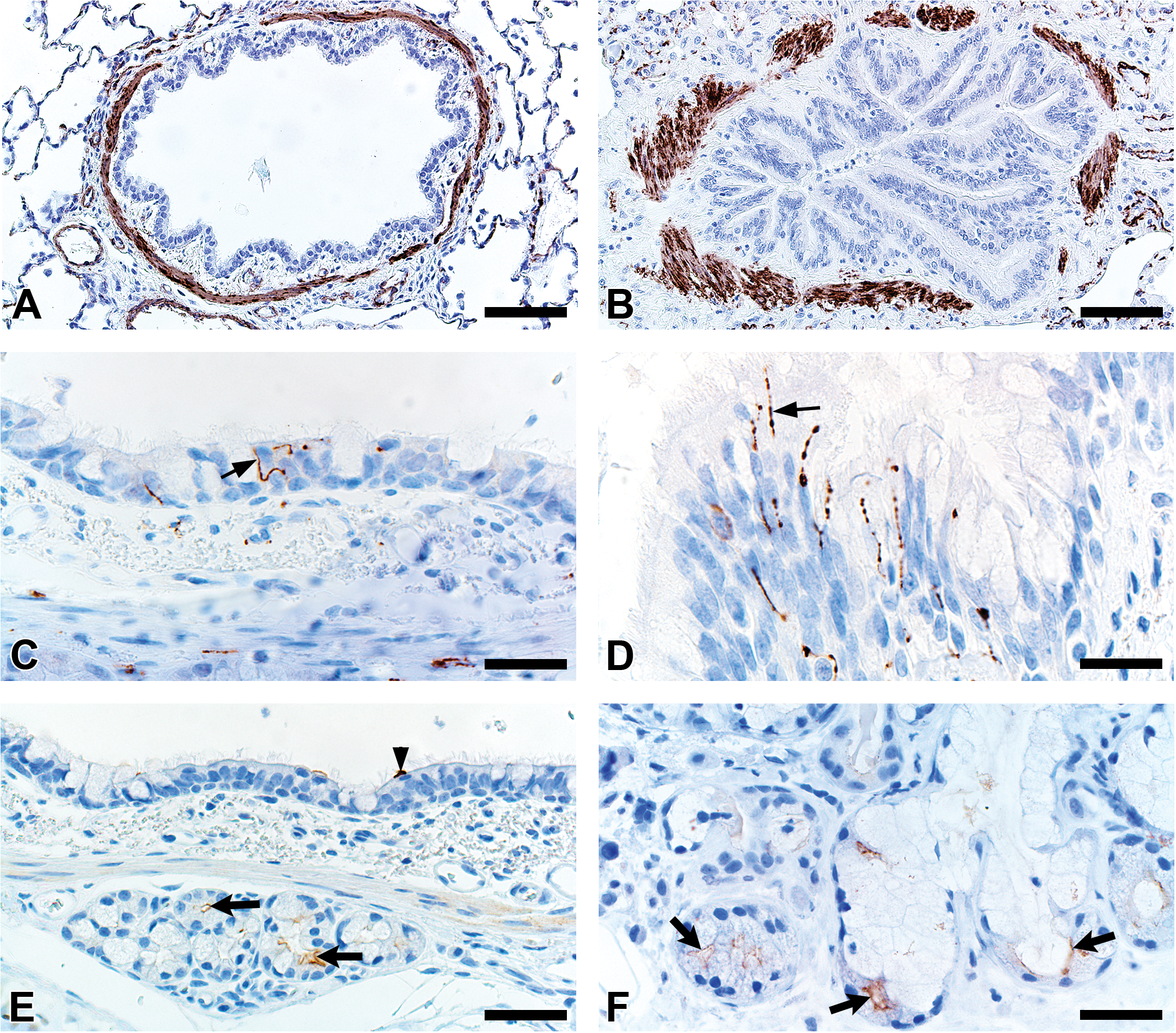
Immunostaining of healthy lung from pig (A, C, and E) and human (B, D, and F). A and B, Airway smooth muscle and tunica media of vessels were immunostained by alpha-smooth muscle actin. C and D, Beta-tubulin-III immunostaining detects axons in the surface epithelium (arrows) and in the submucosa. E and F, Cystic fibrosis transmembrane immunostaining was detected in submucosal glands on the apical cytoplasm/membrane of serous cells (arrows, E and F) and scattered surface epithelial cells (arrowhead, E). Bars = 80 (A,B), 26 (C,D) and 40 (E,F) μm.
Discussion
Most of the immunohistochemistry markers that were optimized in the pig gave comparable immunostaining in human tissues. Lymphoid leukocyte subsets composed of T cells (CD3+), B cells (CD79a+), and germinal center B cells (BCL6+) can contribute to the inflammatory changes within the lung, bronchus-associated lymphoid tissue, and pulmonary lymph nodes following lung injury or disease (Chvatchko et al. 1996; Pabst and Gehrke 1990; Valheim et al. 2011). Lymphoid markers were very comparable in localization and staining intensity (Table 1). Neutrophils and macrophages are important leukocytes to assess for lung injury and disease (Richeldi et al. 2004). Myeloperoxidase, a myeloid leukocyte marker, immunostained neutrophils as well as macrophages as might be predicted. Neutrophils and activated macrophages appeared to have more robust immunostaining than unstimulated macrophages. While neutrophils and macrophages can often be distinguished by their morphologic appearance, the presence of overlapping staining in these cell types can be problematic when doing morphometry or when examining sites with degenerate cells. In contrast, IBA1, while more commonly used in the brain, was surprisingly useful to detect many macrophage/monocyte lineage cells in pigs and humans; further investigations are ongoing.
Submucosal glands can serve as a significant site of airway remodeling through excessive mucus production, defective mucus release into airways, or by gland hypertrophy in chronic disease (Hoegger et al. 2014; Stoltz et al. 2013; Stoltz, Meyerholz, and Welsh 2015). Thus, evaluation of the submucosal glands can be important. Lactoferrin is produced by serous cells of the submucosal glands and some surface epithelial cells but is not expressed by macrophages (Vareille et al. 2011). However, lactoferrin is bound and taken up by macrophages which are speculated to hold a recirculating pool of lactoferrin (Britigan et al. 1991); this could explain apparent macrophage immunostaining. MUC5AC and MUC5B are mucins found in distinct compartments of the airway wall (Meyerholz et al. 2010), with MUC5AC in surface epithelium, and MUC5B in mucous cells of submucosal glands and variably in surface epithelium. The mucus and serous markers were effective in both pig and human tissues.
Environmental exposure to toxicants or airway diseases like asthma, COPD, and CF can lead to remodeling of airway smooth muscle (e.g., hypertrophy) and decreased lung function (McCuaig and Martin 2013; Stoltz, Meyerholz, and Welsh 2015; Wylam et al. 2015). Alpha-smooth muscle actin immunostaining worked well for detecting smooth muscle around airways. Additionally, tunica media of vessels was also immunostained in both pigs and humans—which could possibly confound evaluation of smooth muscle in the walls of distal airspaces.
Activation of autonomic and sensory nerves innervating the airway can elicit cough, bronchoconstriction, mucus secretion, and apnea (Canning [1985] 2006; Undem, McAlexander, and Hunter 1999). Several studies have found increased neural innervation to the airway in experimental models of asthma in both mouse and nonhuman primates (Aven et al. 2014; Kajekar et al. 2007; Yu et al. 2008). Beta-tubulin-III effectively identified pig and human axons within nerves and clearly demonstrated axons extending toward the lumen within the surface epithelium.
CFTR encodes an anion channel that when mutated causes CF (Stoltz, Meyerholz, and Welsh 2015). However, recent evidence suggests that acquired CFTR dysfunction can be observed in COPD and following exposure to cigarette smoke (Courville et al. 2014; Rasmussen et al. 2014). CFTR is often found in the apical membrane of serous cells in submucosal glands and also in scattered surface epithelial cells, similar to what we observed in pig and human tissues.
In conclusion, this study provides direct validation for use of several cellular markers in pathological assessment of porcine tissues for translational investigations. Limitations of this study include the relatively small sample size for each group. Also, we lacked control of the time interval from death to receipt of the fresh human lung tissue—even so, the tissues lacked evidence of autolysis. Advantages of this study included consistent methodology in fixation of tissues for a more standardized final product. Also, the optimization of staining methods in pig tissues as a preliminary step worked well because many of the markers were made for use in humans and so application to the human tissue was often straightforward. Lastly, we were able to directly compare tissues in the same experimental setting and showed that there is remarkable overlap in localization and appearance of tissue markers in pigs and humans. These findings complement and expand upon the existing studies showing that the pig is a useful model for study of human pathobiology.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Institutes of Health (P01 HL051670, P01 HL091842, P30 DK054759, 1K99HL119560, DP2 HL117744), American Asthma Foundation, Cystic Fibrosis Foundation Research and Development Program and Cystic Fibrosis Foundation Mucociliary Clearance Consortium.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
