Abstract
In response to growing concerns that environmental chemicals may have adverse effects on human health by altering the endocrine system, the Endocrine Disruptor Screening Program (EDSP), under the auspices of the United States Environmental Protection Agency (U.S. EPA), recently instituted a Tier I battery of tests including a female pubertal assay. This assay requires dosing of female rats from postnatal day (PND) 22 through PND 42 (or 43), the period of pubertal development in the rat, to identify test articles that may have estrogenic or antiestrogenic effects, or may alter hormones or neurotransmitters. While certain landmarks in female rat reproductive development are published, little is published on the microscopic appearance of the female reproductive tract during prepubertal and pubertal development. In this study, reproductive tissues from three female Sprague-Dawley rats were collected each day from PND 20 through PND 50, such that tissues from a total of 93 rats were collected throughout the prepubertal and pubertal period. Tissues were formalin-fixed, trimmed, paraffin-embedded, sectioned at 5-µm thickness, and examined microscopically. The major histologic features of the female reproductive tract throughout this critical period were described in detail. This information will help pathologists interpret findings observed in female pubertal assays.
Keywords
Introduction
The Endocrine Disruptor Screening Program (EDSP), under the auspices of the United States Environmental Protection Agency (U.S. EPA), instituted male and female
Certain physiologic and anatomic landmarks in development of the female reproductive system of the rat are known. The hormone profiles and patterns during prepubertal and pubertal stages in the rat have been well described (Beckman and Feuston 2003; Davis, Travlos, and McShane 2001). The timing of vaginal opening, estrous cyclicity, and anogenital distances are known (Beckman and Feuston 2003). However, little is published on the microscopic appearance of the female reproductive tract during this time. This article documents the microscopic appearance of the normal developing female rat reproductive tract during prepubertal and pubertal development, which will provide information to pathologists assigned the task of identifying test substance–related findings in the female pubertal assay. Knowing the normal appearance of the developing ovary and uterus will allow one to differentiate otherwise normal reproductive tissues with delayed development (i.e., sexual immaturity) from reproductive tissues with direct or specific test substance–related lesions.
Materials and Methods
Animals
Female Sprague-Dawley (SD) rats (Charles River Laboratories, Inc., Crl:CD (SD) rats) ranging from PND 20 to 50 were used. The rats were randomly selected from the pool of clinically normal rats available in the WIL Research stock colony and did not undergo any treatment regimen. All study procedures and animal handling techniques were performed in compliance with WIL Research’s Institutional Animal Care and Use Committee (IACUC) and the Public Health Service Policy on Humane Care and Use of Laboratory Animals.
Necropsy and Histology
Three female SD rats per day from separate litters, beginning at PND 20 and progressing through PND 50 for a total of 93 rats, were selected for scheduled necropsy. Prior to necropsy, final body weight and clinical observations for each animal were recorded. Animals were euthanized by carbon dioxide inhalation and subjected to gross necropsy examination. At the time of necropsy, ovaries (paired) and uterus (wet and blotted) weights were measured and recorded. The ovaries, uterus, cervix, and vagina were collected and placed in 10% neutral buffered formalin. After approximately 24 hr, the ovaries and uterus with cervix were rinsed and stored in 70% ethanol until histological processing.
The fixed tissues were trimmed, subjected to routine histologic processing, and paraffin-embedded. Five systematic uniform random 5-µm thick sections were collected from each ovary at 50-µm intervals beginning from a random starting point. Ovarian tissue blocks were faced on a microtome until ovarian tissue was reached, then a randomly generated number between 51 and 100 was used and the block was further faced that number of sections into the block prior to collection of the first section. After collection of the first section, four additional ovarian sections were obtained at 50-µm intervals. One section from each of the following reproductive tissues was also obtained: cross section of each uterine horn, longitudinal section of uterus and cervix, and longitudinal section of vagina. Routine hematoxylin and eosin (H&E) staining was performed on the obtained tissue sections.
Pathology
Detailed microscopic examination of the prepared ovarian, uterine, cervical, and vaginal sections was performed by a board-certified anatomic veterinary pathologist. Ovarian and uterine examinations were performed as described in the U.S. EPA female pubertal assay test guidelines (U.S. EPA 2009). Ovarian histopathological examination included an evaluation of follicular development (including presence/absence of tertiary/antral follicles, presence/absence of corpora lutea, changes in corpus luteum development, and changes in number of both primary and atretic follicles) in addition to any abnormalities/lesions, such as ovarian atrophy. Uterine histopathological examination included an evaluation of changes in uterine horn diameter and myometrial, stromal, or endometrial gland development. Features of the ovaries, uterus, cervix, and vagina throughout the prepubertal and pubertal developmental period were described in detail.
Results
Microscopic Findings in Ovaries
Throughout the prepubertal and pubertal developmental time period, from PND 20 through 50, primordial and primary follicles were present in all animals. Primordial and primary follicles were more readily visualized in the highly immature ovary (i.e., PND 20 to PND 25), and appeared scattered along the cortical periphery and in dense clusters at the ovarian hilus. As the ovary matured and contained more tertiary and Graafian follicles and corpora lutea, the clusters of primordial and primary follicles were less commonly observed. Primordial or primary follicles will not be further mentioned in the results. Noteworthy histologic features of the ovaries are summarized in Table 1.
Microscopic features of developing ovary, uterus, and vagina in Sprague-Dawley rats from PND 20 to 50.
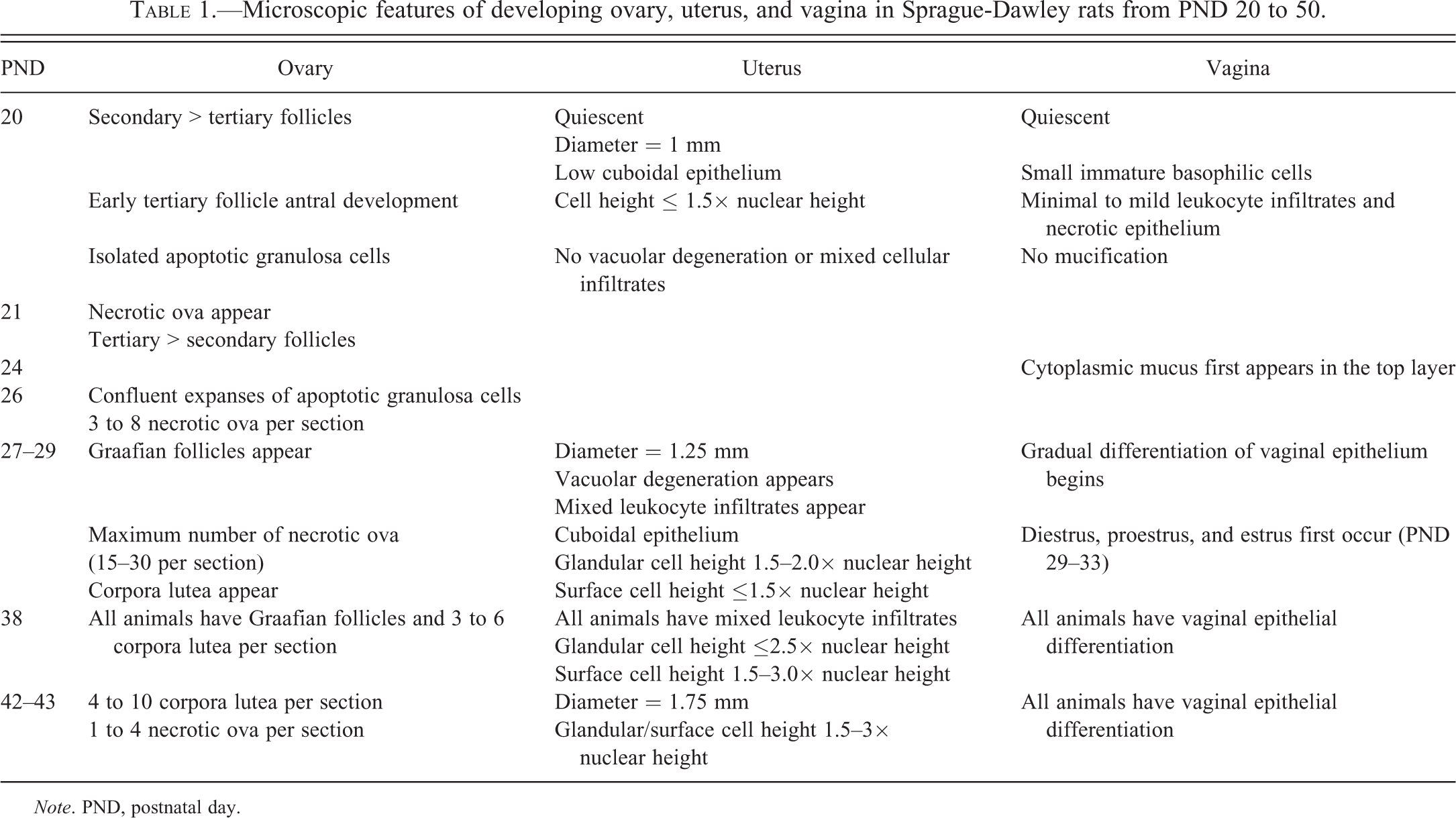
PND 20
The ovary was composed of predominantly secondary follicles and fewer but significant numbers of tertiary follicles with early antral development (Figure 1). Graafian follicles (tertiary follicles that reached the ovulatory stage and had an ovum surrounded by a cumulus oophorus) and corpora lutea were not present. There was little distinction between ovarian cortex and medulla. Isolated collections of apoptotic granulosa cells, indicative of early follicle atresia, were present in the walls of secondary and tertiary follicles (Figure 2). Necrotic ova, another feature of atretic follicles, were not present in these follicles with apoptotic granulosa cells.
PND 21
Necrotic ova were present in small numbers (one to four per ovarian section) in the medulla. There were generally more tertiary follicles than secondary follicles.
PND 22 to 26
Early tertiary follicles continued to expand with fluid. Many of these tertiary follicles had significant apoptosis of granulosa cells, which were most prominent in the area adjacent to the follicular fluid (Figure 3). By PND 26, there was confluent expansion of apoptotic granulosa cells in atretic secondary and early to mid-stage tertiary follicles. In addition, there were 3 to 8 necrotic ova per ovarian section (Figure 4). These necrotic ova were primarily located in the medulla.
PND 27 to 28
There was clear distinction between the outer cortex containing large tertiary follicles (Figure 5) and the center medulla. Graafian follicles were present in the outer ovarian cortex of several animals. Numerous necrotic ova were located in the medulla, and approximately 15 to 30 necrotic ova could be seen in any given section (Figure 6).
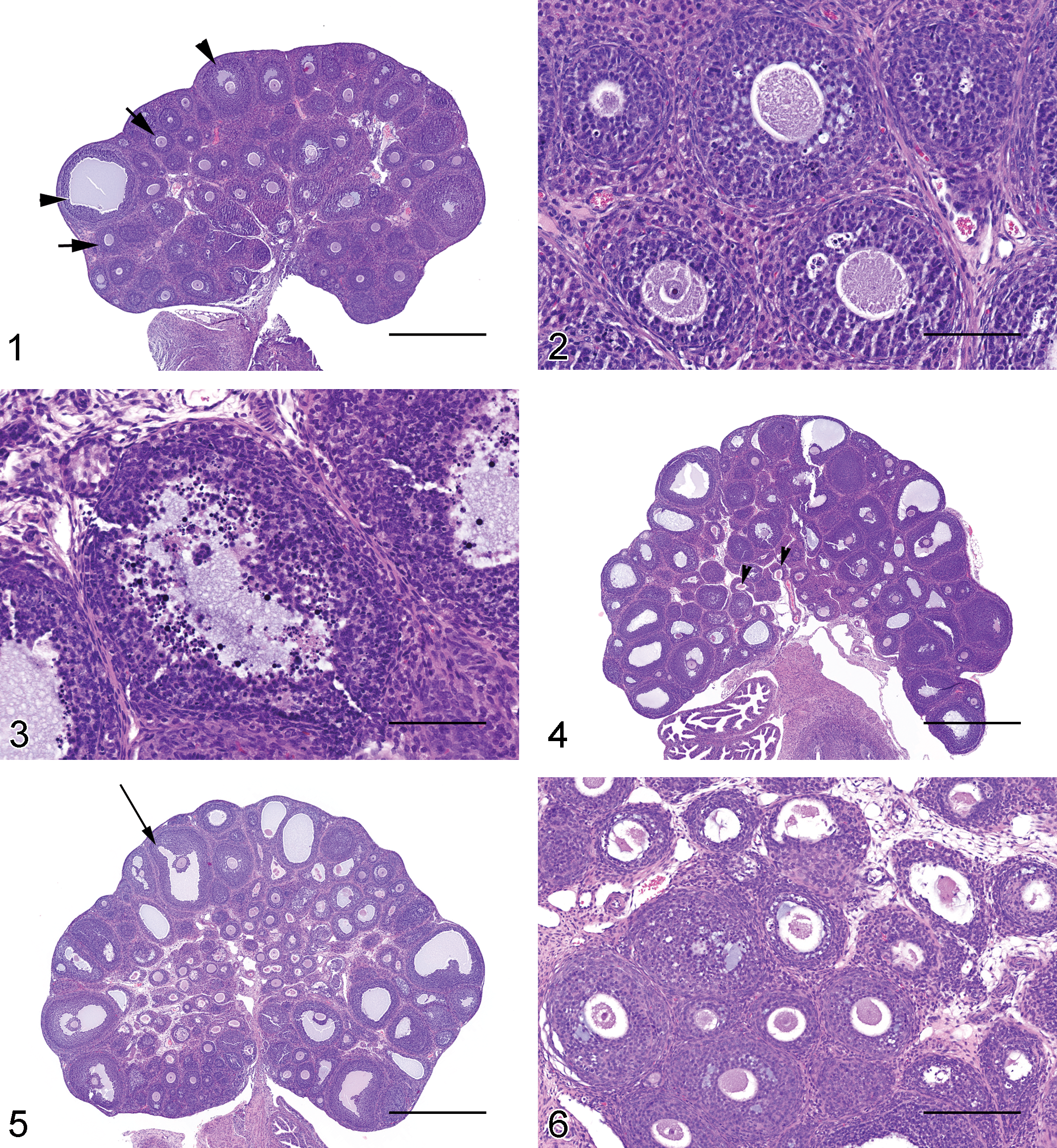
PND 29 to 38
At PND 29, 1 of the 3 animals had 1 to 3 corpora lutea per section (Figure 7). The corpora lutea were in the growing stage of luteal development, characterized by cells with basophilic cytoplasm, few mitotic figures, and an occasional central area containing fluid or hemorrhage. There was a gradual decline in the number of necrotic ova per section of ovary. More tertiary follicles had reached the ovulatory stage. By PND 38, all three animals had several Graafian follicles and 3 to 6 corpora lutea per ovarian section.
PND 39 to 43
From PND 39 to PND 43, the end point of the female pubertal assay, there were approximately 4 to 10 corpora lutea per section of ovary (Figure 8). Necrotic ova were present in low numbers (approximately 1–4 per ovarian section).
PND 44 to 47
The number of corpora lutea increased slightly (Figure 9), ranging from 5 to 15 per section of ovary. The majority of the ovarian parenchyma was composed of corpora lutea in growing (basophilic) and mature (eosinophilic) stages of development (Figure 10), with fewer corpora lutea in the regressing stage (Figure 11). The regressing stage of a corpus luteum was characterized by relatively small cells and a large central area of fibrous connective tissue with necrotic cell debris.
Microscopic Findings in Uterus
Noteworthy histologic features of the uterus are summarized in Table 1.
PND 20 to 26
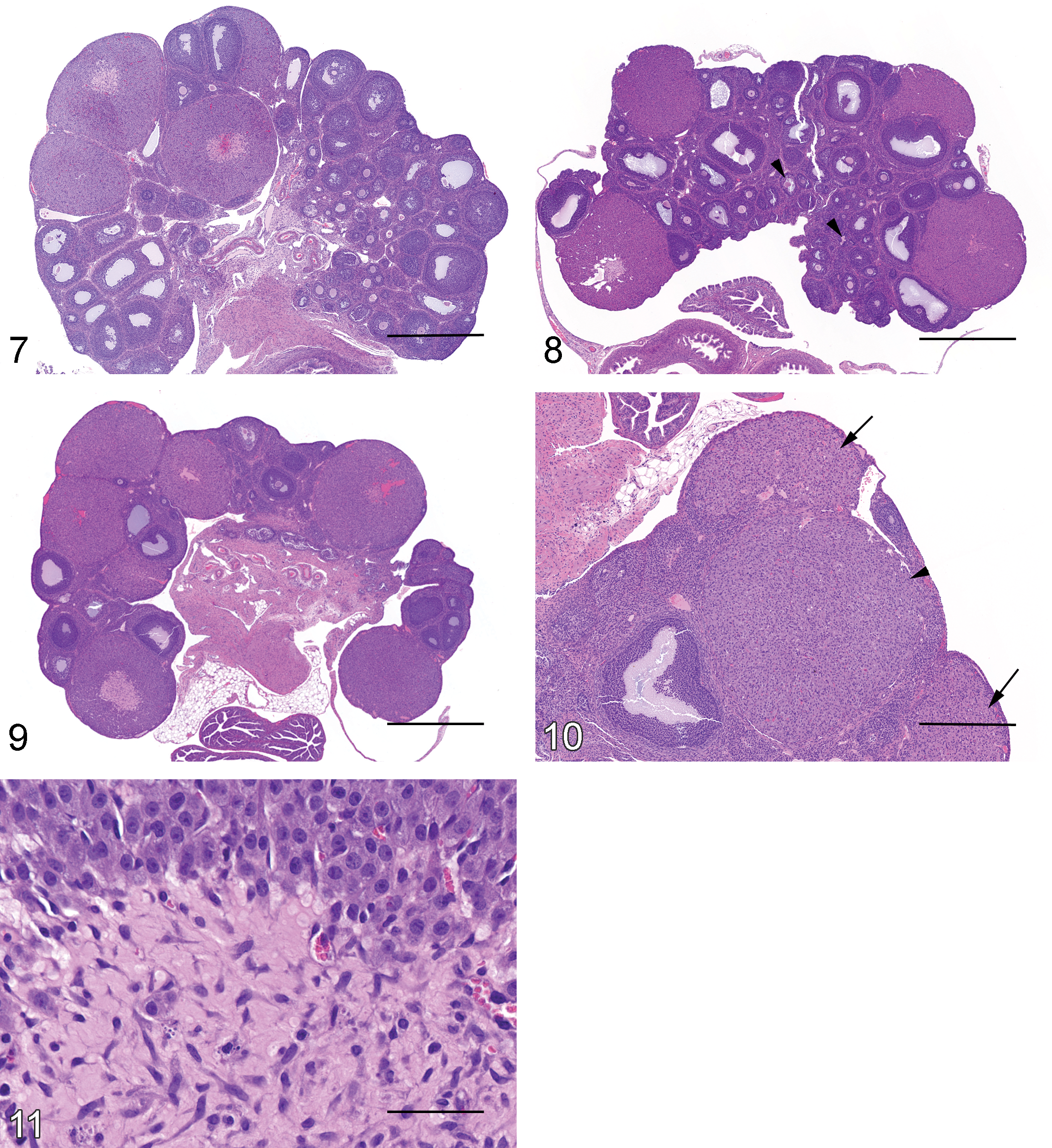
The uterus appeared to be “quiescent” and immature. Cross sections of the uterus were approximately 1 mm in diameter (external diameter of whole uterine cross section) at PND 22 (Figure 12). Vacuolar degeneration of surface or glandular epithelium and leukocyte infiltrates were absent. The uterine endometrial glands and the surface epithelium were lined by a single layer of low cuboidal epithelium with a cell height which was ≤1.5 times the height of the nucleus. A few mitotic figures in the surface epithelium, the endometrial glandular epithelium, or stromal cells were noted in some animals after PND 24.
PND 27 to 28
Cross sections of the uterus were approximately 1.25 mm in diameter at PND 28. Minimal vacuolar degeneration of the endometrial glandular or surface epithelium was first noted at PND 28 (Figure 13). Leukocyte infiltrate was absent at PND 28. The endometrial glandular epithelium was cuboidal with a cell height that was 1.5 to 2.0 times the height of the nucleus, while the surface epithelium remained low cuboidal with a cell height that was ≤1.5 times the height of the nucleus. A few mitotic figures in the endometrial glands or the surface epithelium were noted.
PND 29 to 38
Minimal mixed leukocyte infiltrates composed predominantly of eosinophils with fewer mononuclear cells were noted in one animal, which also had ovarian corpora lutea, at PND 29. There was moderate interindividual animal variability in response to hormonal stimulation of the uterus as evidenced by mild vacuolar degeneration, mixed leukocyte infiltrates, and increased endometrial glandular and surface epithelial cell height in some animals. In other animals, the uterus remained morphologically quiescent up to PND 37, similar to the histomorphology described at PND 20 to 26. The cell height variations were more overt in the surface epithelium than the endometrial glandular epithelium. The uterine surface epithelium cell height varied from ≤1.5 to >3.0 times the height of the nucleus, while the cell height of the endometrial glandular epithelium was ≤2.5 times the height of nucleus. A few mitotic figures in the endometrial glands or the surface epithelium were noted with similar frequency to that noted at PND 24 to 28.
PND 39 to 43
Throughout this time period, there was a minimal mixed leukocyte infiltrate in all animals, and the uterine diameter reached approximately 1.75 mm by days 42 and 43. Minimal vacuolation of the surface and endometrial glandular epithelium and occasional mitotic figures in epithelial cells were also noted. Cellular heights of the surface epithelium and endometrial glandular epithelium were 1.5 to 3.0 times the height of the nucleus (Figure 14). There were no significant changes from PND 43 through PND 50.
Microscopic Findings in Cervix
PND 20 to 50
Histologic features of the proximal and distal ends of the cervix were similar to the uterus and vagina, respectively, throughout the study. Minimal to mild dilatation of the cervical lumen was noted occasionally after PND 29.
Microscopic Findings in Vagina
Noteworthy histologic features of the vagina are summarized in Table 1.
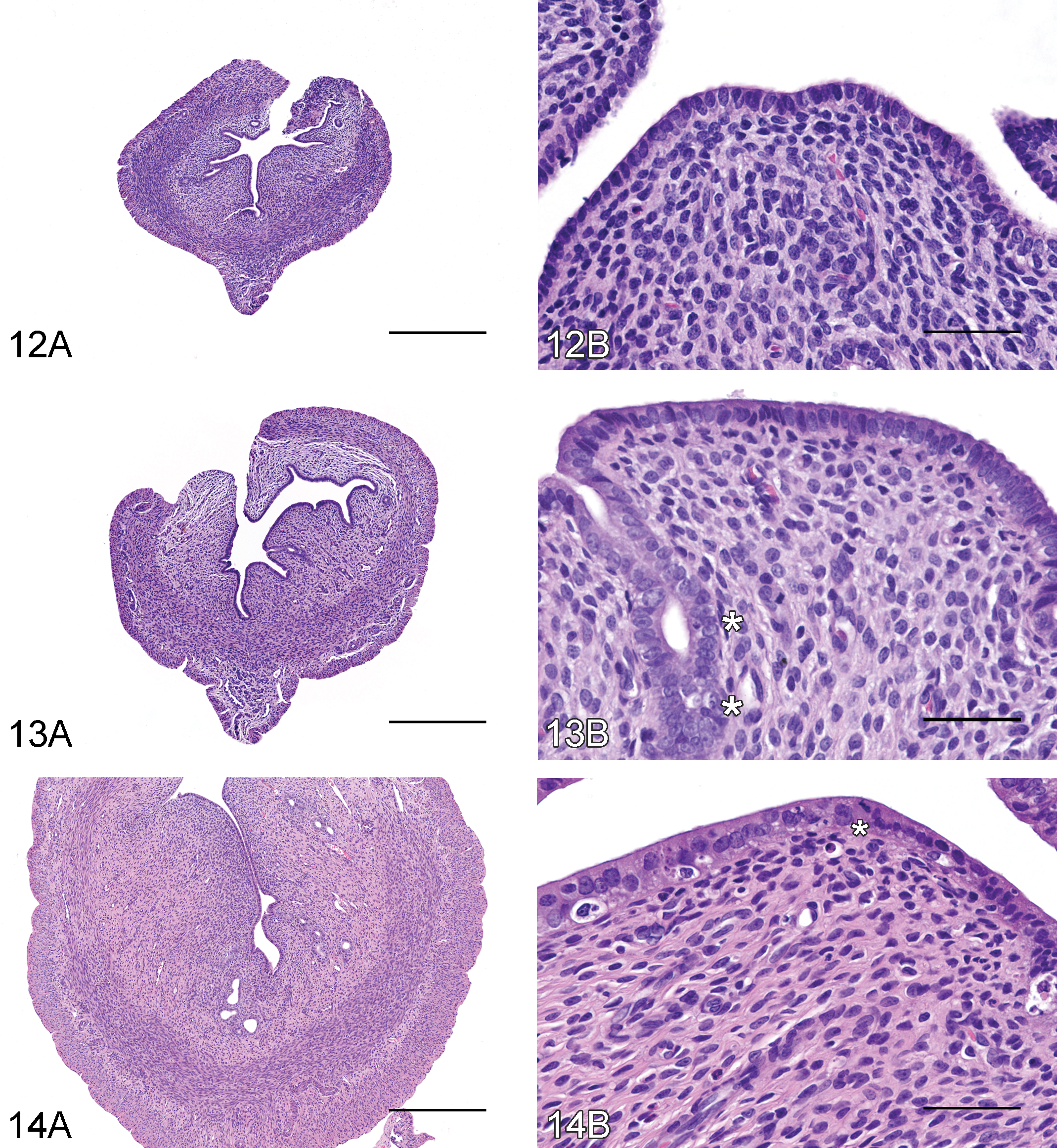
PND 20 to 23
The vagina appeared to be “quiescent” until PND 23 (Figure 15). Three to more than 5 cell layers of the stratum germinativum were present, while the stratum granulosum, stratum corneum, and stratum mucification were absent. The superficial layer of the stratum germinativum was composed of plump cuboidal epithelium. The subjacent layers were composed of immature epithelial cells characterized by a small amount of basophilic cytoplasm. Minimal to mild leukocyte infiltrates in the vaginal lumen and/or mucosa, mitotic figures in the epithelium, and individual or small clusters of necrotic epithelial cells were present.
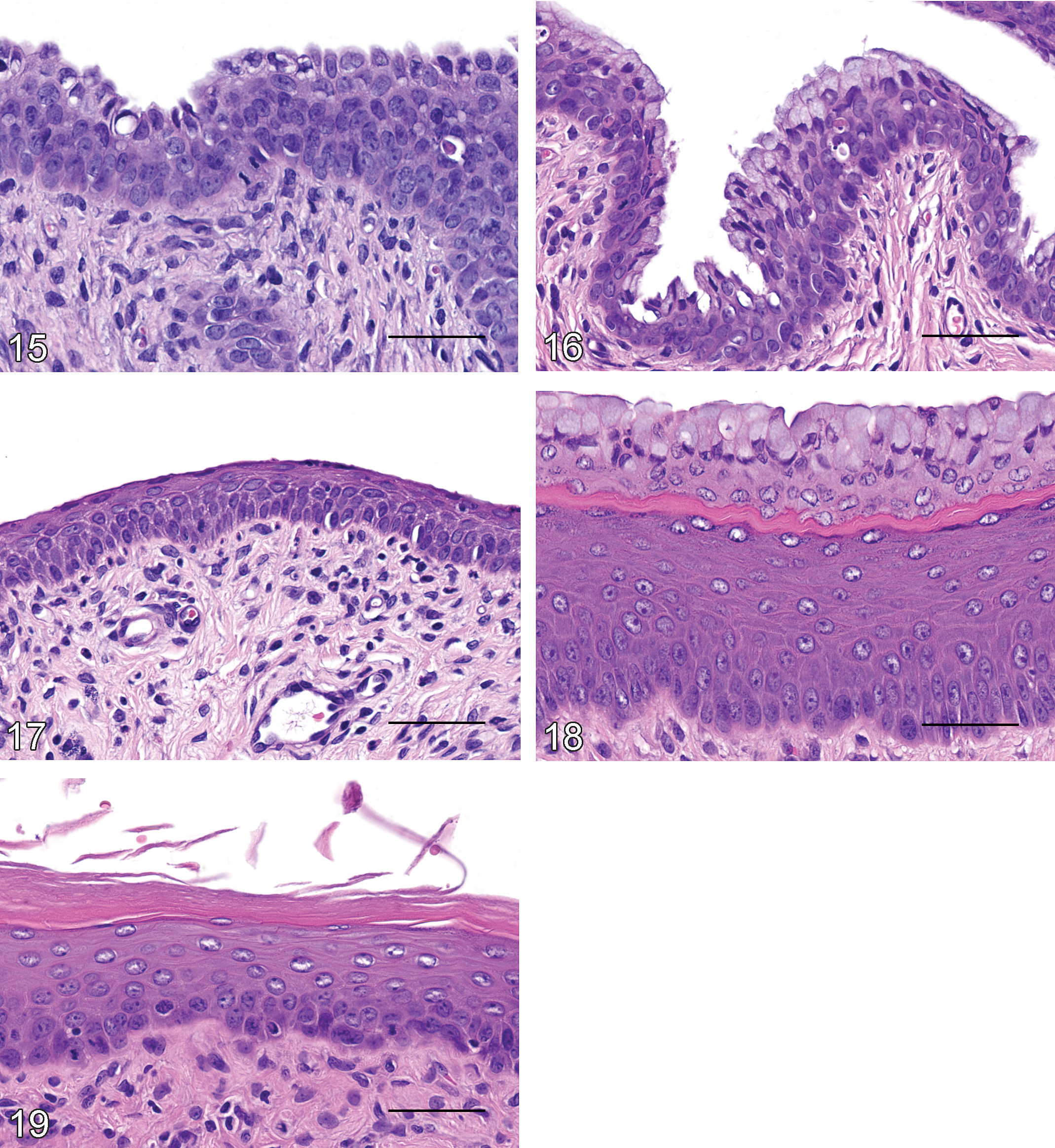
PND 24 to 28
From PND 24 to PND 28, the superficial layer of the stratum germinativum contained multifocal accumulations of pale cytoplasmic mucous material in some animals (Figure 16). The subjacent layers remained immature epithelial cells characterized by a small amount of basophilic cytoplasm. Minimal to mild leukocyte infiltrates in the vaginal lumen and/or mucosa and mitoses and/or necrosis of epithelial cells were present in some animals. The surface epithelium remained cuboidal in most animals, while a few animals had attenuated epithelium.
PND 29 to 50
Histological evidence of estrous cycling began at PND 29 with interindividual animal variability in the stage of estrous cycle at the various time points. Diestrus was characterized by slight flattening of the superficial layer of the stratum germinativum (Figure 17). The animal with ovarian corpora lutea at PND 29 had flattened vaginal surface epithelium. Proestrus was characterized by the appearance of the stratum mucification (Figure 18). Estrus was characterized by the presence of the stratum granulosum and stratum corneum with abundant luminal keratin (Figure 19). The onset of estrous cycling was highly variable, and the vagina remained immature or showed keratinocyte differentiation in some animals from PND 29 to PND 37. In the immature stage, the deep layers of the stratum germinativum consisted of basophilic cells that had scant cytoplasm, without overt distinction or differentiation between the cell layers (Figure 16). By PND 38, cellular differentiation and maturation of each layer of the stratum germinativum was observed in all animals (Figures 17 –19).
Ovarian and Uterine Weights
Final body weight, ovarian weight, and uterine weight (wet and blotted) increased from PND 20 through PND 42 (Table 2 and Figure 20). At approximately PND 42, the ovarian and uterine weights stabilized, albeit there were fluctuations in ovarian and uterine weights to account for estrous cycling. The greatest increase in mean ovary weight occurred between PND 37 and PND 42, when it increased from 52.1 to 97.7 mg. This ovarian weight increase correlated with the period of expansion in both size and number of Graafian follicles and an increase in number of corpora lutea. From PND 42 to PND 50, mean ovarian weights ranged from 85.1 to 99.7 mg.
Final body, ovary, and uterus weights in normal Sprague-Dawley rats from PND 20 to 50.
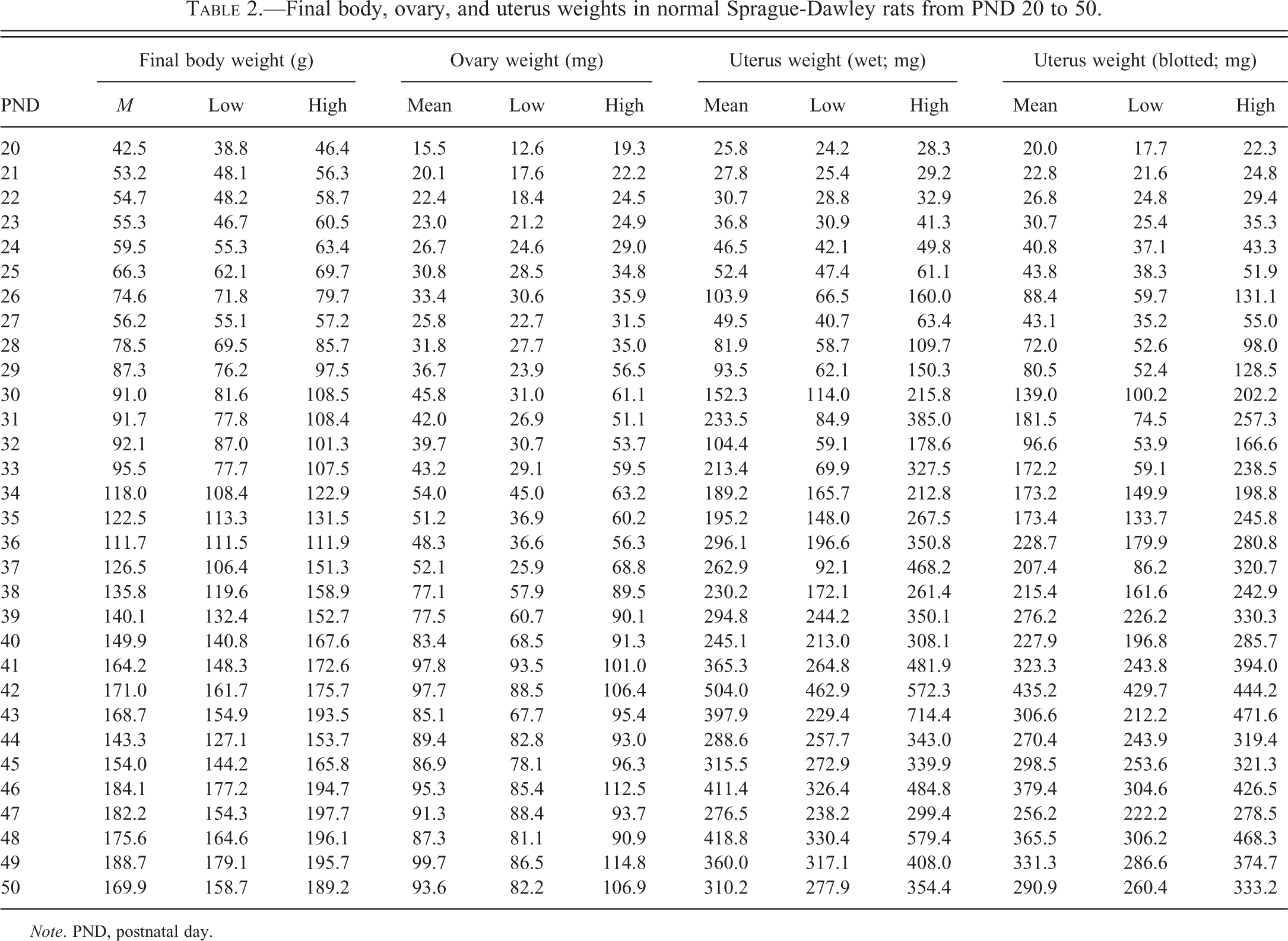
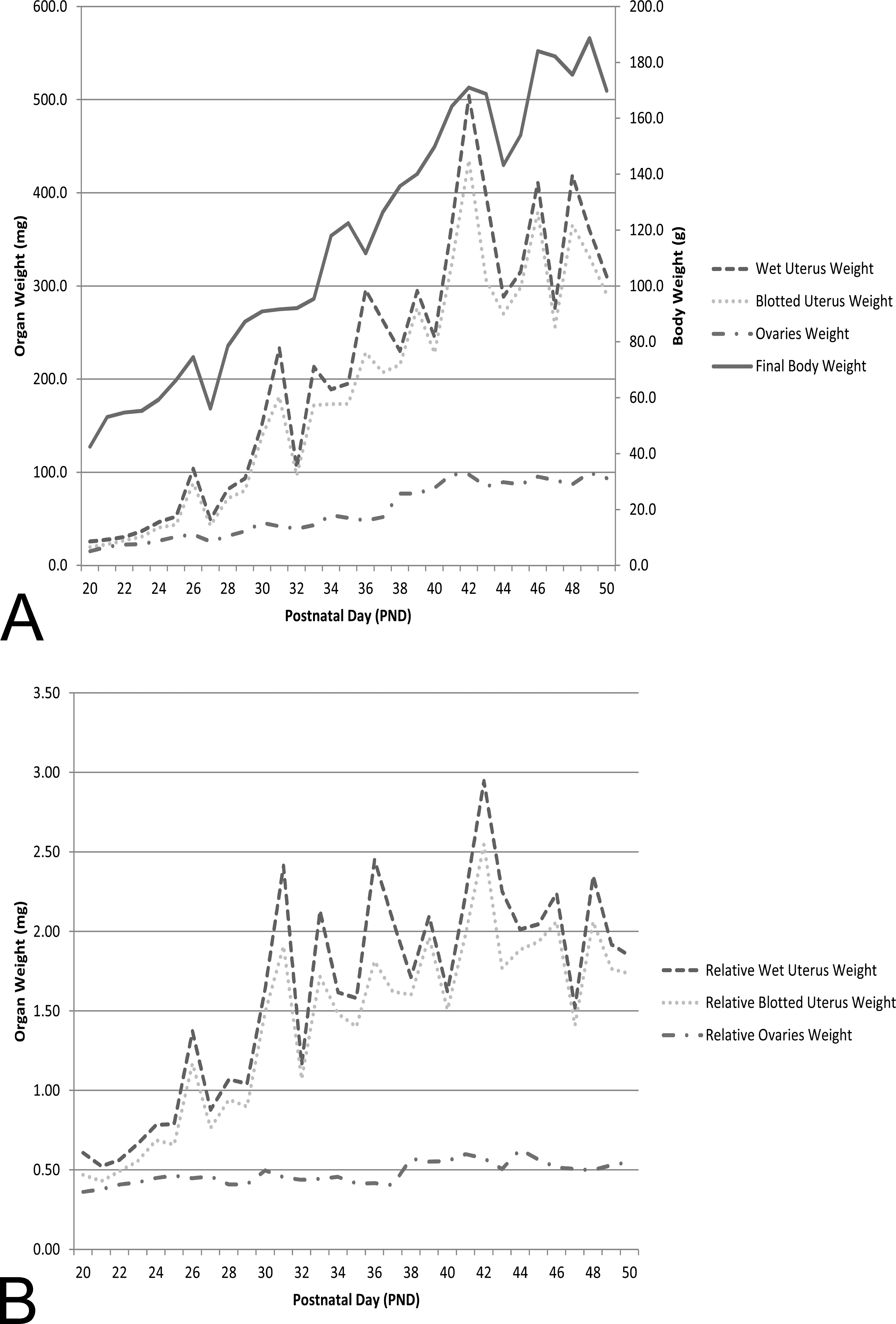
When ovarian and uterine weights were reviewed relative to final body weight, there was an initial increase, with stabilization around the onset of puberty (PND 42) for both uterus and ovary (Figure 20B). Mean group values and low and high individual animal values for final body weight, ovarian weight, and uterine weight (wet and blotted) at each time point are provided in Table 2.
Discussion
Major landmarks of sexual development in the rat are known. The full complement of primordial follicles exists at PND 5 in the rat (Davis, Travlos, and McShane 2001); onset of ovulation varies between reports, but is reported to occur from PND 29 to 38 (Beckman and Feuston 2003); vaginal patency generally occurs at PND 36 to 37 (Beckman and Feuston 2003); and rats reach full sexual maturity at approximately 7 weeks (Kohn and Clifford 2002). Therefore, the 21-day dosing period (PND 22–42) of the female pubertal assay spans the time during which the ovary develops and the rat brain begins to respond to the positive feedback of estrogen, resulting in the LH surge and the occurrence of the first estrous cycle and ovulation (Laws et al. 2012). At PND 43, based on these published reports, normal rats should have ovaries that have undergone 1 or more cycles of ovulation.
Our microscopic findings were in agreement with published literature. The presence of numerous medium-sized secondary and tertiary follicles at PND 20 was consistent with Davis, Travlos, and McShane (2001) and Pelusa (1992). During the infantile period from PND 7 to 21, FSH stimulates the ovary to convert testosterone to estradiol, and follicular development takes place. Granulosa cells of these follicles between PND 7 and 21 have FSH receptors.
The juvenile stage of ovarian development is reported to occur from PND 22 to 32 (Pelusa, 1992). During this time, granulosa cells continue to respond to pituitary gonadotropin to secrete estrogen, while the follicles continue to develop. Since the hypothalamus does not produce sufficient quantities of gonadotropin-releasing hormone during this juvenile stage, the pituitary fails to produce sufficient LH, and the follicles fail to ovulate. LH pulses occur at 40 ng/ml at a rate of 2 pulses/hr during this juvenile time frame (Pelusa 1992). This level of LH is insufficient to cause ovulation, and the developing follicles therefore become atretic during the juvenile time frame. LH insufficiency is the cause of the wave of follicular development and atresia and explains the dramatic increase in necrotic ova at PND 27 to 28. The necrotic ova, which were located in the well-delineated medulla, reached a maximum number at this time (15–30 per section) and then drastically reduced to approximately 1 to 4 per section by PND 42.
The presence of Graafian follicles and ovulation first occurring at PND 28 and corpora lutea first occurring between PND 29 and 38 coincided with that previously reported for onset of ovulation in rats during PND 29 to 38 (Beckman and Feuston 2003) and with the hormonal profile of the peripubertal period lasting 3 days after the juvenile period (Pelusa 1992). Transitioning from the juvenile stage to the peripubertal stage requires a change in LH secretory pattern from pulses of 40 ng/ml to pulses with amplitudes of 80 ng/ml with an additional mini surge of LH between 2 and 4 p.m. The effects of these changes in the pattern of LH secretion results in ovulation (Pelusa 1992). The pulsative pattern of prolactin and GH secretion, as well as the diurnal variation of epinephrine, also influence ovarian function and development. While Pelusa reports that the juvenile stage lasts from PND 22 to 32, with the peripubertal stage lasting 3 days thereafter, we report that ovulation and the peripubertal stage can occur as early as PND 29 in the SD rat.
In addition to the wave of follicular atresia at PND 27 to 28, we report atresia of secondary and tertiary follicles as early as PND 20. Atresia first presented as small collections of apoptotic granulosa cells but by PND 26 progressed to confluent expanses of apoptotic cells. Normal atresia was prominent in secondary and early to mid-stage tertiary follicles, which was consistent with that reported under physiological conditions (Kaipia and Hsueh 1997). Several atretogenic (enhancing apoptosis of granulosa cells) and anti-atretogenic (inhibiting apoptosis of granulosa cells) factors have been identified (Durlinger et al. 2000). It is also known that estrogens suppress follicular atresia and androgens enhance follicular atresia (Durlinger et al. 2000). In addition, during FSH-dependent follicle development, gonadotropins, together with local ovarian growth factors (insulin-like growth factor-1 [IGF-1], epidermal growth factor [EGF]/transforming growth factor α [TGF-α], basic fibroblast growth factor [FGF]), cytokines (interleukin-1 beta [IL-1β]), and estrogens, activate different intracellular pathways to rescue follicles from apoptotic demise. In contrast, TNF-α and Fas ligand, presumably acting through receptors with a death domain, are atretogenic factors (Kaipia and Hsueh 1997).
The first regressing corpora lutea occurred at PND 47, consistent with corpora lutea first appearing between PND 28 and 38 and corpora lutea lasting several estrous cycles in the rat (Yuan and Foley 2002). Regressing corpora lutea in the rat do not commonly present as corpora albicans, since accumulation of fibrous tissue is negligible and rapid and complete resolution of the structure is the rule. The pubertal assay requires pathologists to identify corpora lutea in the growing, mature, and regressing stages. The growing stage corresponds to corpora lutea comprised of basophilic cells. The mature stage is represented by those corpora lutea comprised of plump eosinophilic and finely vacuolated cells. Those corpora lutea with smaller cells, an expanded fibrous center, and necrotic debris are the regressing corpora lutea. Corpora lutea of each cycle grow during metestrus, become mature during the first day of diestrus, and begin regression by the third day of the cycle (Pelusa 1992). Progesterone production by any given corpus luteum generally lasts during metestrus and diestrus. After that time, progesterone secretion declines as the cycle approaches proestrus. In the rat, the uterus secretes a luteolytic factor, presumably prostaglandin F2α, which is responsible for the relatively short life span of the corpus luteum in the rat.
The onset of histologic changes in the uterus was in concert with ovarian changes. The uterus remained quiescent until PND 27 and the earliest vacuolar degeneration of epithelial cells and leukocyte infiltrates (including eosinophils) occurred at PND 28 followed by corpora lutea first appearing in the ovary at PND 29. After PND 38, all animals had leukocyte infiltrates in the uterus, which correlated with the presence of Graafian follicles and corpora lutea in the ovary. There was continuous growth of the uterine diameter throughout the study. The diameter was approximately 1.0 mm, 1.25 mm, and 1.75 mm on PND 22, 28, and 43, respectively. The increase in uterine size was accompanied by increased uterine weight from PND 22 to PND 42.
Histologic changes in the vagina were also in harmony with changes observed in the ovaries and uterus. The vagina remained histologically quiescent until PND 23 with scant basophilic cytoplasm and plump cuboidal superficial epithelium. The earliest change, multifocal mucification, was noted on PND 24; 4 days prior to the onset of corpora lutea in the ovary and vacuolar degeneration of surface and glandular epithelial cells and leukocyte infiltrate into the uterus. Histological evidence of estrous cycling began at PND 29 with interindividual animal variability in the stage of estrous cycle at the various time points. This finding was similar to a previous study that indicated cytological evidence of onset of estrous cyclicity started from PND 31 (Kim et al. 2002). The vaginal changes in our study were in concert with the first appearance of corpora lutea in ovary at PND 29. The deep layer of the stratum germinativum remained immature with scant basophilic cytoplasm in all animals at PND 28, started to show differentiation in some animals from PND 29 to PND 37, and by PND 38 all animals showed characteristic features of vaginal epithelial differentiation into basal, parabasal, intermediate, precornified, and cornified layers. This finding is consistent with the histologic evidence of corpora lutea (i.e., ovulation) in all animals after PND 38.
In conclusion, the major histologic features of the female reproductive tract throughout the critical time period of prepubertal and pubertal development were described in detail. This information will assist pathologists in the interpretation of findings observed in pubertal assays in female rats.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was fully funded by WIL Research.
