Abstract
Spontaneously occurring proliferative lesions of the male accessory sex glands are infrequent in various strains of rats. In rodents, the ampullary glands are embedded in the prostate. Although 2 spontaneous cases of atypical hyperplastic lesions at the ampullary gland were previously described in Wistar rats, adenocarcinoma and/or adenoma in this gland have not been reported. This study describes adenocarcinomas in the bilateral ampullary glands in a 52-week-old intact male Sprague-Dawley rat housed as part of a control group in a toxicological experiment. At necropsy, the body weight (644.4 g) and the weight of the prostate with ampullary gland (2.75 g) were similar to others of the same control group, and it had a normal gross appearance. Histopathologically, both ampullary glands revealed microinvasive adenocarcinoma without vascular invasion. The morphological characteristics of the neoplasm varied in different regions of the gland. Other parts of the male accessory sex glands did not show proliferative lesions.
Introduction
Adenocarcinomas of the prostate are some of the most commonly diagnosed carcinomas in adult men and are second only to lung cancer in cancer deaths (Kumar, Abbas, and Fausto 2005). Anatomical similarities between the human and rodent prostates have helped support the application of murine models for studies of pathological alterations that accompany the normal development of the organ, as well as the progression of prostate carcinoma (Polland and Lucker 1986; Huss, Maddison, and Greberger 2001).
Spontaneous neoplasms of the prostate lobes, coagulating gland, and seminal vesicles have been described in a few cases in rodents (Mitsumori and Elwell 1988; Bosland et al. 1998; Shirai et al. 2000; Creasy et al. 2012). Although the ampullary glands are physically located within the prostatic complex (Figure 1), the spontaneous neoplasms of ampullary glands have not been previously reported.
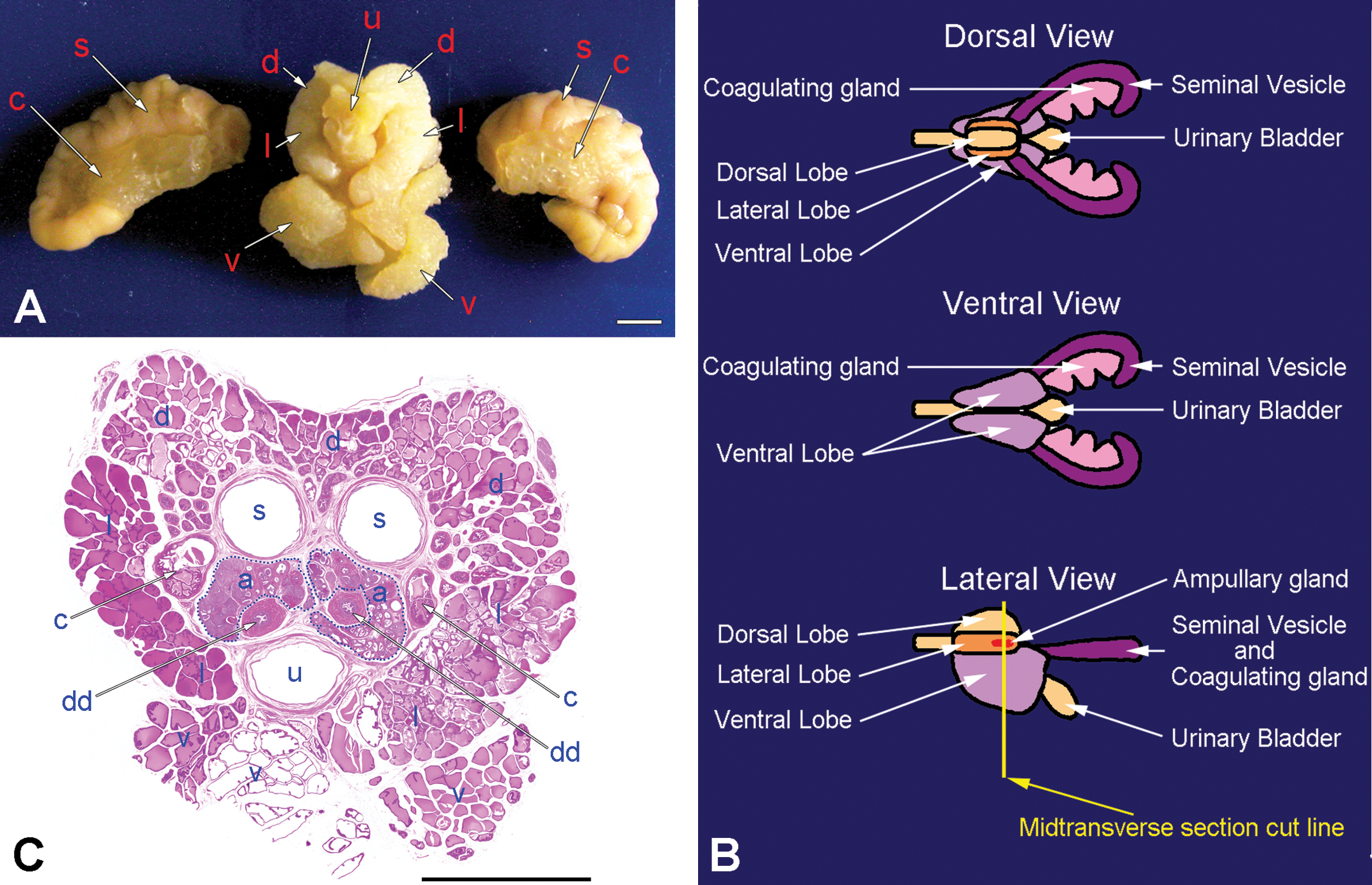
(A) Gross appearance of caudal view of the rat prostatic complex, with a part of the seminal vesicles and coagulating glands cut off. (B) Pattern diagram of the dorsal, ventral, and lateral view of rat male accessory glands that modified figure 15 of Suwa et al. (2001). (C) Low power view of midtransverse section micrograph of prostatic complex cutting by yellow line bar in (B) showed that the ampullary glands (circled with blue dotted lines) were indicated to embed in the prostate, hematoxylin and eosin (H&E). (a) Ampullary gland; (d) dorsal prostatic lobe; (l) lateral prostatic lobe; (v) ventral prostatic lobe; (u) urethra; (dd) deferent duct; (s) seminal vesicle; (c) coagulating gland. Bar = 5 mm.
Assessment of the potential toxicological and carcinogenic effects of a chemical compound on male rodents’ accessory sex glands, composed of dorsal–lateral–ventral prostate lobes, coagulating glands, seminal vesicles, and ampullary gland, may be difficult if the incidence of spontaneous histopathological background lesions is unclear. Previous reports of spontaneously occurring proliferative lesions of the male accessory sex glands in rodents are infrequent, except for a number of chemically induced neoplasms (Bosland et al. 1998; Creasy et al. 2012). Thus, the male rat accessory sex glands have been used as an experimental model to analyze the functions of androgenic hormones (Lee and Holland 1987) and in 2-year carcinogenicity and toxicity studies (Solleveld, Haseman, and McConnell 1984; Bosland 1992; Suwa et al. 2001).
Terminology and diagnostic criteria, as well as the general features and incidence of proliferative lesions of the male accessory sex glands of rats and mice, have been previously published by World Health Organization/International Agency for Research on Cancer (WHO/IARC) and Standardized System of Nomenclature and Diagnostic Criteria (SSNDC; Squire et al. 1978; Frith and Ward 1988; Mitsumori and Elwell 1988, 1994; Boorman, Chapin, and Mitsumori 1990; Faccini, Abbott, and Paulus 1990; Bosland 1992; Gordon, Majka, and Boorman 1996; Alison et al. 1997; Bosland et al. 1998; Rehm et al. 2001). Moreover, the International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice (INHAND) project provides an internationally accepted nomenclature for proliferative and nonproliferative lesions of the male reproductive system in rats and mice (Creasy et al. 2012). However, a recent review (Creasy et al. 2012) excluded a description of the ampullary gland in the male accessory sex glands, and the incidence of proliferative lesions of this organ is unclear.
Materials and Methods
The progenitor colony of the 52-week-old male Sprague-Dawley (slc) rat (Japan SLC, Japan), which from Charles River Inc., USA, in 1968, and its genetic strain was thought to be similar to that of Crl:CD(SD) rats, was housed at the Research Institute of Bioscience, Azabu University, Kanagawa, Japan, as part of a control group in toxicology experiments. The rat was housed in a plastic cage on hardwood-chip bedding in an environment-controlled room on a 12-hr light/12-hr dark cycle at 22 ± 2°C and 55 ± 5% relative humidity, with a conventional diet (F-1, Funabashi Form, Japan). All experimental procedures were conducted following approval of the Animal Care and Use Committee of the Azabu University School of Veterinary Medicine. Guidelines set by the National Institutes of Health and the Public Health Service Policy on the Humane Use and Care of Laboratory Animals were followed at all times. The rat did not show abnormal behavior and was euthanized by an intravenous injection of pentobarbital. We performed a complete necropsy on the rat and conducted gross and histopathological examinations of major organs including the prostatic complex. Seminal vesicles and coagulation glands were cut off at each root, and transverse sections through the urethra that included both prostatic dorsal–lateral–ventral lobes and ampullary glands were cut (Figure 1). Tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 3 μm, and stained with hematoxylin and eosin (H&E). Selected sections were stained with periodic acid Schiff (PAS). We performed an immunohistochemical stain using the anti-keratin monoclonal antibody EAB 903 (“basal cell-specific antibody,” Enzo Biochemical, New York; Brawick and Mardi 1983; Cleary, Choi, and Ayala 1983; Wakui et al. 1989; Hayward et al. 1996), because the normal ampullary glands consisted of the secretory cells and the basal cells (Cleary, Choi, and Ayala 1983; Brawick and Mardi 1983; Hayward et al. 1996).
Results
The body weight of the rat was 644.4 g, and 3 grossly recognizable prostate lobes with ampullary glands measured 22 × 16 × 20 mm and weighed 2.75 g. These were similar to those of the other rats housed in the same environmental condition. However, macroscopic observation revealed no specific abnormalities (Figure 1).
Histopathologically, both ampullary glands revealed microinvasive adenocarcinoma without vascular invasion. The morphological characteristics of the neoplasm varied in different regions of the gland. Other parts of the male accessory glands, prostate lobes, coagulating glands, and seminal vesicles did not show any proliferative lesions.
There were adenocarcinomas that disrupted the architecture, and marked cellular atypia and invasive growth were present. The tumor cells were enlarged and their cytoplasm was eosinophilic; their nuclei were pleomorphic, enlarged round to oval shapes with hypochromatic and small nucleoli (Figure 2). The adenocarcinomas had invaded the surrounding stroma, probably by rupture of the basement membrane (Figure 2). However, these relatively small lesions showed morphological characteristics of carcinoma except for their large predominantly invasive growth, which might justify classifying them as early carcinoma; therefore, we considered these lesions as microinvasive adenocarcinomas.
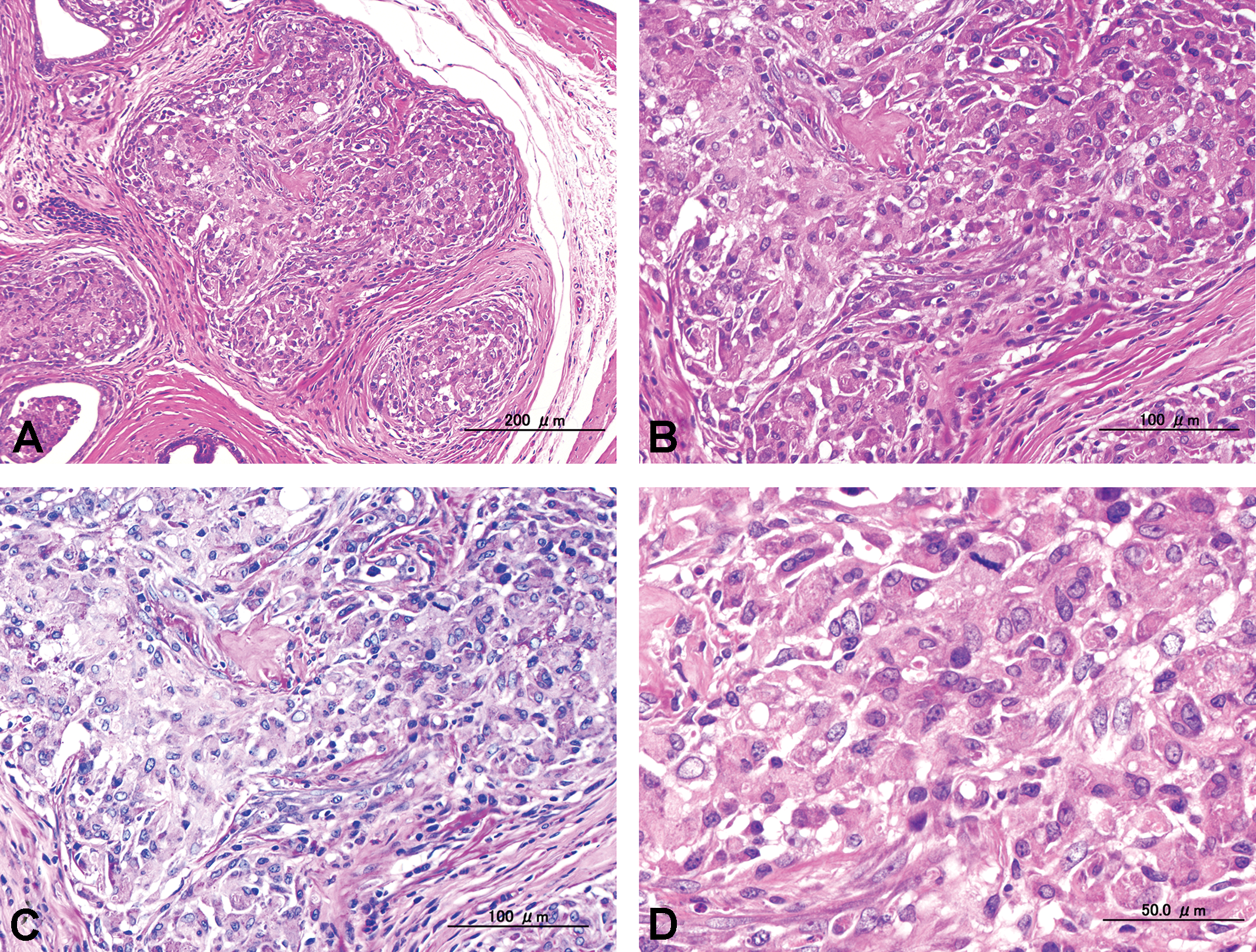
(A) Low power view of microinvasive adenocarcinoma on rat ampullary gland, hematoxylin and eosin (H&E). (B) High power view of (A), H&E. (C) Serial section of (B). Tumor cells apparently invaded interstitial space with ruptured basement membrane, periodic acid Schiff (PAS). (D) Higher power view of (B). Tumors were composed of pleomorphic cells with enlarged nuclei, and mitotic figures are also observed, H&E.
The morphological characteristics of the adenocarcinoma varied in different areas of the glands (Figures 3–5). Proliferative lesions occasionally obliterated the alveolar lumen and were composed of tumor cells that had lost their polarity and had enlarged round to oval hypochromatic nuclei, while basal cells were absent (Figure 3). Other areas showed loss of normal cellular polarity, eosinophilic cytoplasm, and enlarged nuclei with hypochromatin and a small nucleolus, but basal cells were present. There were no mitotic figures. We observed 2 types of growth pattern: the cribriform type (Figure 4) and the intra-alveolar type (Figure 5). Both possessed basal cells that were positive for EAB 903 (Figures 4 and 5).
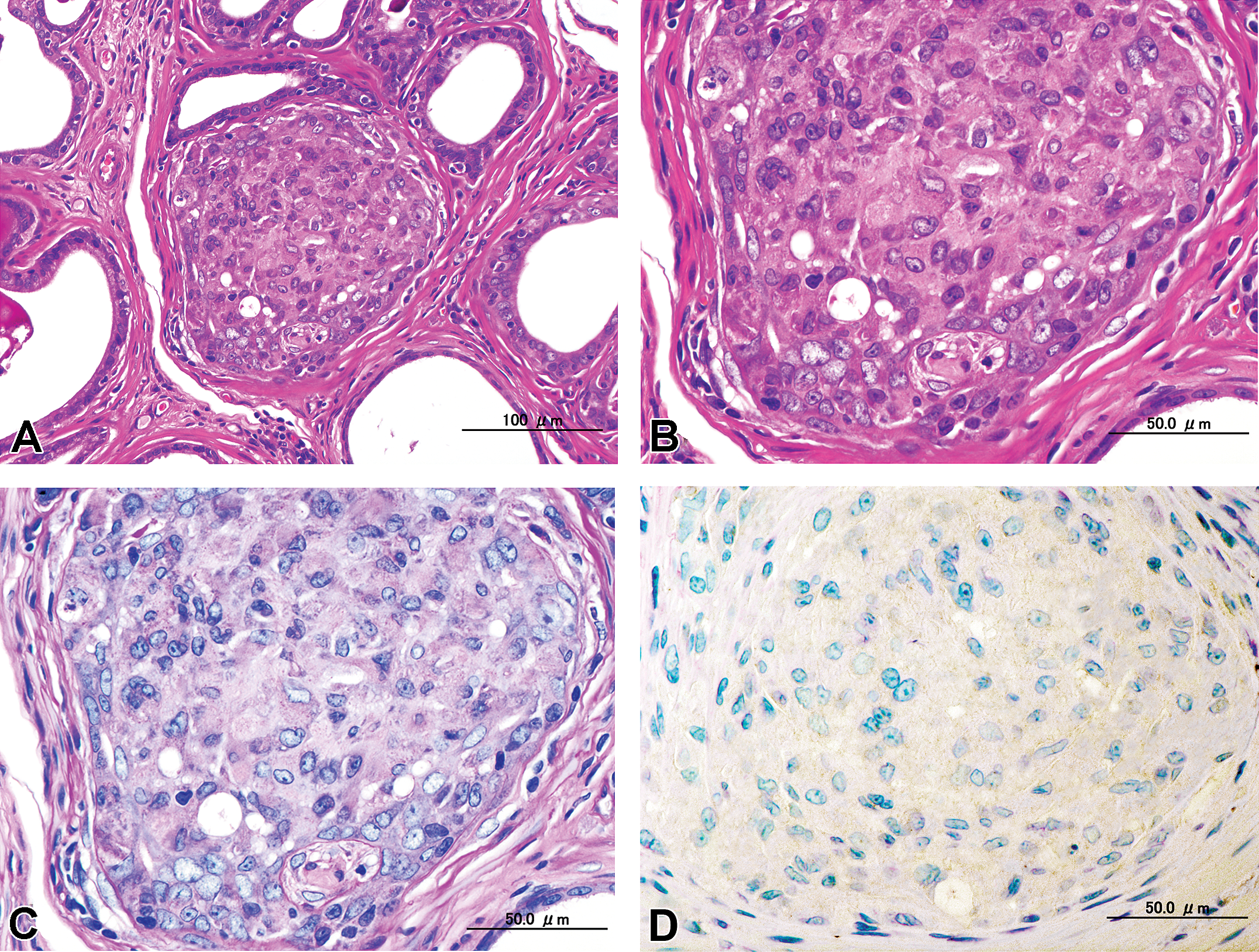
(A) Low power view of intra-alveoli epithelial proliferation with a solid to cribriform pattern filling each lumen of alveoli with distribution of normal glandular architecture, hematoxylin and eosin (H&E). (B) High power view of (A), H&E. (C) Serial section of (B). Tumor cells were not invasive, and the basement membrane of the adenomatous alveoli remained intact, periodic acid Schiff (PAS). (D) Contiguous section of (C). Immunohistochemical staining with anti-keratin EAB 903 was negative.
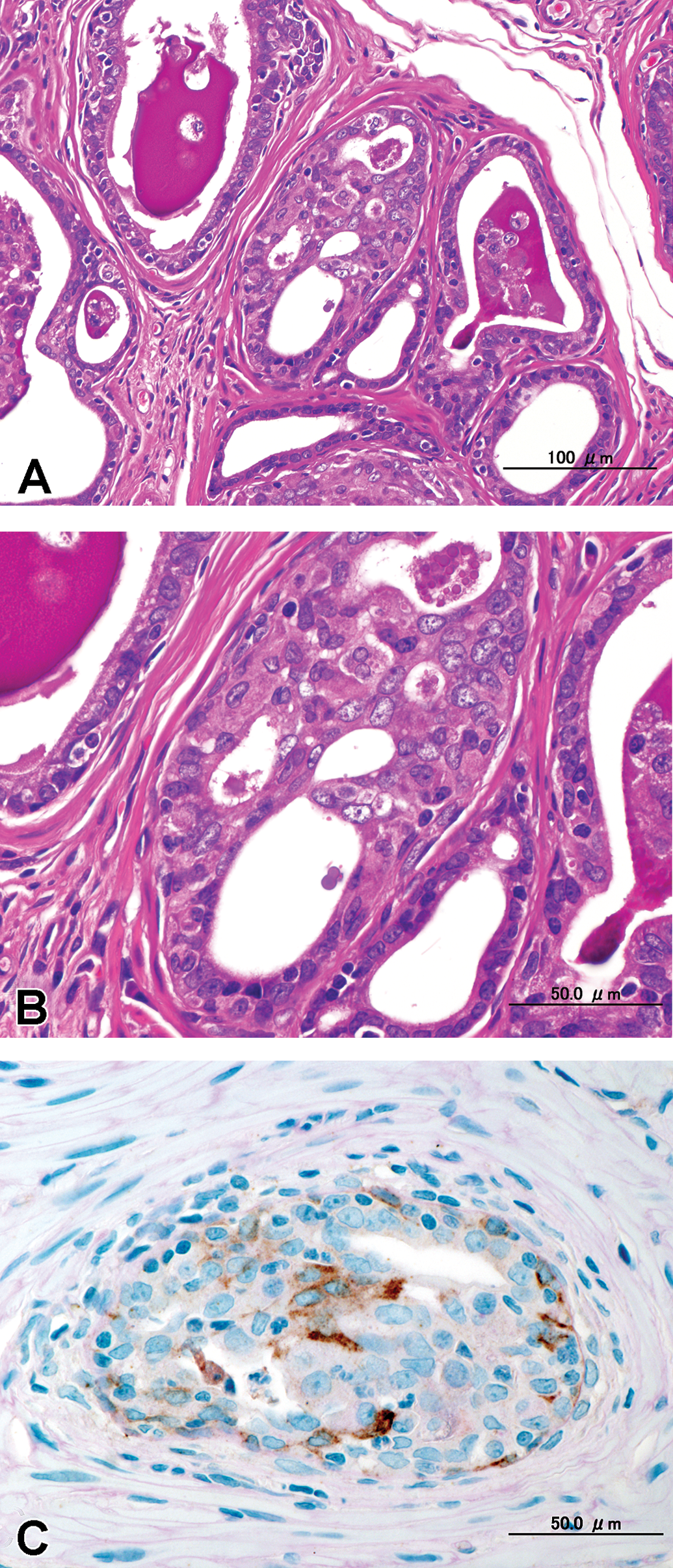
(A) Cribriform proliferation inside the alveolar glandular lumen that was composed of multilayered atypical epithelial alveoli without distorted normal architecture or complete obliteration of the alveolar lumen. The surrounding area was not compressed by the lesions, and there was no inflammation; hematoxylin and eosin (H&E). (B) High power view of (A), H&E. (C) Contiguous section of (B). Immunohistochemical staining with anti-keratin EAB 903 showed brown-colored reactivity selectively in the basal cell cytoplasm.
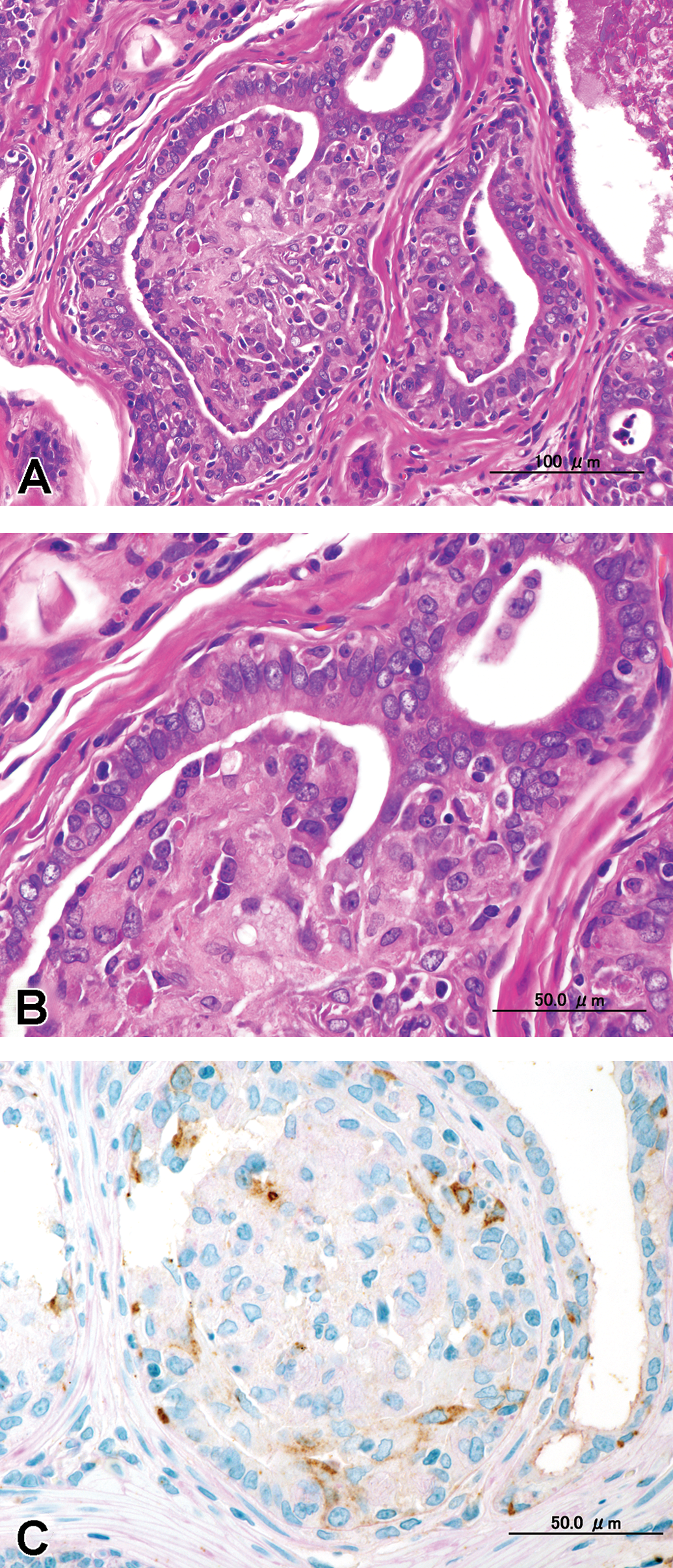
(A) Intra-alveolar growth that was composed of multilayered atypical epithelial alveoli without distorted normal architecture or complete obliteration of the alveolar lumen. The surrounding area was not compressed by the lesions, and there was no inflammation; hematoxylin and eosin (H&E). (B) High power view of (A), H&E. (C) Contiguous section of (B). Immunohistochemical staining with anti-keratin EAB 903 showed brown-colored reactivity selectively in the basal cell cytoplasm.
Discussion
The ampullary gland in rats is structurally the smallest gland in the male rodents’ accessory sex glands, and it differs from the other glands by opening directly into the ampullary portion of the deferent duct rather than into the other glands (Chow and Pang 1989; Aumüller et al. 2012). In rats, because sperm are released from the deferent duct along with an emission from the ampullary gland, it has been considered that the ampullary gland may be involved with sperm maturation and might be important in the evaluation of male reproductive toxicity.
Normal ampullary glands in the rat encircle the deferent ducts. The alveoli are large, uniform in size, and surrounded by a few smooth muscle cells and connective tissue stroma. Strongly eosinophilic secretions are frequently observed in alveoli. The epithelium is flattened to cuboidal secretory cells with centrally located nuclei and some basal cells (Cleary, Choi, and Ayala 1983; Brawick and Mardi 1983; Hayward et al. 1996).
Proliferative nonneoplastic lesions of the male rodents’ accessory sex glands, including ampullary glands, are infrequent (Bosland et al. 1998; Creasy et al. 2012). Two cases of atypical hyperplasia in the ampullary gland were previously reported, but the lesions were found in aged rats treated with a carcinogen and testosterone: an 85-week-old Wistar rat (Bosland and Prinsen 1990) and 1.5-year-old Wistar rat (HsdCpb: WU; Bosland et al. 1998). This study revealed spontaneous adenocarcinoma in an aged SD rat. Although the morphological features of the overall lesion varied in the different glandular alveoli, it was considered a locally invasive adenocarcinoma based on its cellular atypia, loss of cellular polarity, and evidence of invasion. Basal cells are a normal cellular component of the ampullary gland but are absent in adenoma and adenocarcinoma. Utilizing the immunohistochemical stain for EAB 903, we demonstrated that basal cells were absent in some parts of the tumor but present in others.
Spontaneous neoplasms of the male accessory sex glands have been described in a limited number of cases or species in rodents (Mitsumori and Elwell 1988; Bosland et al. 1998; Shirai et al. 2000; Creasy et al. 2012). For example, incidences of spontaneous prostate tumors are less than 1% for mice and 2% for hamsters (Riverson and Silverman 1979). In rats, the incidence of prostate carcinoma varies by strain from 4% for F344 rats (Shirai et al. 2000) to 70% for ACI/Seg rats after 33 months of age (Ward et al. 1980). Lobund-Wistar rats also exhibit spontaneous prostate carcinoma at over 20 months of age, but the incidence is only around 10% (Ward et al. 1980; Polland and Lucker 1986; Shirai et al. 2000). In addition, a limited number of spontaneous epithelial hyperplasia and adenoma, as well as adenocarcinoma, have been reported in the seminal vesicles in F344 rats (Suwa et al. 2001) and B3C3F1 mice (Suwa et al. 2002).
Although the seminal vesicles and the ampullary glands have been considered to be of similar embryological origin, different from the prostate lobes (Cunha et al. 2001), spontaneous adenocarcinoma and/or adenoma have not been reported in the ampullary gland in nontreated or carcinogen-treated rats and mice (Bosland 1992; Bosland et al. 1998; Suwa et al. 2001; Suwa et al. 2002). In addition, although secondary tumors metastasizing in the ampullary gland of rats have been reported (Boorman, Chapin, and Mitsumori 1990; Bosland 1992), little is known about primary lesions in the rat ampullary gland (Suwa et al. 2001). To our knowledge, the present case described here is the first report of ampullary gland neoplasms in a rat.
Footnotes
Acknowledgments
The authors are grateful to M. Sigomoto, M. Sakurai, M. Ohwada, and M. Hayashi for technical assistance, and thank Dr. Katherine Ono and Bioscience Editing Solutions for critically reading and editing the manuscript.
Author Contribution
M. Motohashi substantially contributed to conception and design, analysis of data, drafted the manuscript, gave final approval, and agrees to be accountable. S. Wakui substantially contributed to conception and design, analysis of data, drafted the manuscript, gave final approval, and agrees to be accountable. H. Takahashi substantially contributed to conception and design, analysis of data, drafted the manuscript, gave final approval, and agrees to be accountable. A. Yoshida substantially contributed to conception and design, analysis of data, drafted the manuscript, gave final approval, and agrees to be accountable. T. Mutou substantially contributed to conception and design, analysis of data, drafted the manuscript, gave final approval, and agrees to be accountable. M. Ikegami substantially contributed to conception and design, analysis of data, drafted the manuscript, gave final approval, and agrees to be accountable. M. Asari substantially contributed to conception and design, analysis of data, drafted the manuscript, gave final approval, and agrees to be accountable. T. Inomata substantially contributed to conception and design, analysis of data, drafted the manuscript, gave final approval, and agrees to be accountable.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by a research project grant award by the Azabu University Research Services Division, and the Promotion and Mutual Aid Corporation for Private Schools of Japan, Grant-in-Aid for Matching Fund Subsidy for Private Universities, provided funding for this project. This study was supported by Grant-in-Aid (C) #25450473 from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
