Abstract
Diacylglycerol O-acyltransferase 1 (DGAT1) plays an important role in synthesizing lipids, and inhibitors of DGAT1 have been investigated as potential treatments for diabetes and metabolic diseases. DGAT1 knockout (−/−) mice are resistant to obesity, have increased sensitivity to insulin, and exhibit sebaceous gland atrophy and alopecia. Prolonged pharmacological inhibition of DGAT1 with AZD7687 in mice results in the same skin phenotype, including sebaceous gland atrophy and alopecia, as seen in the skin of DGAT1 (−/−) mice. AZD7687-mediated effects on the skin were dose- and time-dependent and reversible. They occurred only at substantial levels of continuous DGAT1 inhibition. Prolonged treatment of dogs with AZD7687 also resulted in sebaceous gland atrophy but did not result in the more adverse skin changes of hair loss and skin lesions. Our findings highlight a significant risk of generating the same lesions that were seen in mouse skin during clinical development of DGAT1 inhibitors in humans and also reveal a species difference in the effects on the skin, indicating that the mouse may be an especially sensitive species. Therefore, although human therapeutic doses may not have the same influence on skin morphology as seen in mice, monitoring of skin changes will be essential in clinical trials with DGAT1 inhibitors.
Keywords
Introduction
Diacylglycerol O-acyltransferase 1 (DGAT1), a key enzyme that catalyzes triglyceride synthesis from mono- and diglycerides, shows promise as a pharmacological target in the treatment of type 2 diabetes and obesity (Yen et al. 2005; Zammit et al. 2008; Birch, Buckett, and Turnbull 2010). Previous work in DGAT1-deficient mice (−/−) has demonstrated enhanced insulin sensitivity and protection from obesity as a result of increased energy efficiency (Smith et al. 2000; Streeper et al. 2006). However, the skin of DGAT1 (−/−) mice is characterized by atrophy of sebaceous glands, dry fur, and hair loss. Furthermore, their fur lacks wax diesters that are a major component of skin surface lipids in mice (Chen et al. 2002). This observation has highlighted that DGAT1 may also have a significant role in dermatological conditions involving the sebaceous gland and hair follicle, such as acne and alopecia. Further work has indicated that DGAT1 catalyses the synthesis of diacylglycerol, waxes, and retinyl esters (Yen et al. 2005) and acts as an acyl CoA:retinol acyltransferase (ARAT) in the skin of mice where it maintains retinoid homeostasis and prevents retinoid toxicity (Shih et al. 2009). Interestingly, a similar skin phenotype has also been described in mice deficient in the enzyme stearoyl-CoA desaturase 1 (SCD1; Zheng et al. 1999; Sundberg et al. 2000) and in mice that have been subjected to pharmacological inhibition of SCD1 (Meingassner et al. 2013). These mice show a substantial reduction in lipid staining in sebaceous glands, and the absence of normal sebaceous gland lipids has been demonstrated by biochemical analysis. Furthermore, transgenic mice overexpressing apolipoprotein C-I also have sebaceous gland atrophy, scaly skin and hair loss, and reduced triglycerides and wax esters in sebum (Jong et al. 1998). These observations have led to the hypothesis that in the mouse, sebaceous gland lipids play an important role in the development and processing of the hair shaft and hair follicle (Sten 2001; Smith and Thiboutot 2008).
A potential role of DGAT1 in skin and hair development and maintenance is a significant concern in the development of DGAT1 pharmaceutical agents for the treatment of obesity and metabolic diseases since adverse effects on the skin could result in drug-mediated side effects that would render the risk/benefit profile of such a drug unattractive. In this communication, we describe the effects of pharmacological inhibition of DGAT1 on the skin and hair of the mouse and dog, the preclinical species used in the development of candidate drug AZD7687. Our data show that administration of AZD7687 in the mouse and the dog caused sebaceous gland atrophy in both species: this effect being clearly AZD7687-mediated, dose- and time-dependent, and also reversible following cessation of dosing. Inhibition of DGAT1 at levels substantially exceeding the DGAT1 enzyme concentration required to inhibit the enzyme by 50% (IC50) during the dose interval in normal mice for 6 months resulted in the same adverse phenotype described for the skin of DGAT (−/−) mice, including epidermal hyperplasia, hair follicle atrophy, and epithelial erosions, as well as in-life observation of hair loss and skin lesions (i.e., open wounds and scabs). However, in the dog, inhibition of DGAT1 at levels substantially exceeding the DGAT1 enzyme IC50 during the dose interval for 6 months yielded pathological changes in the skin that were restricted to sebaceous gland atrophy and minimal hyperkeratosis of the epithelium covering the infundibula of hair follicles and did not result in overt hair loss or macroscopically apparent skin lesions. Our findings reveal that adverse effects on the skin may be an anticipated outcome of prolonged pharmacological inhibition of DGAT1, but they also show a species difference in the nature and severity of such effects and suggest that skin effects in human may not be a major challenge to clinical development of DGAT1 inhibitors.
Materials and Methods
Test Substance
AZD7687 was synthesized by AstraZeneca R&D (Alderley Park, Macclesfield, UK) and has been confirmed to show selective DGAT1 inhibitory activity (Barlind et al. 2012). In an in vitro recombinant mouse, dog, and human DGAT1 enzyme assay, AZD7687 has an IC50 of approximately 100, 60, and 80 nM, respectively, and an IC50 of >35,000 nM against human acetyl-coenzyme A:cholesterol O-acetyltransferase 1 (ACAT) 1 and ACAT2 enzymes. No activity against human DGAT2 was detected.
For in vivo experiments, AZD7687 was formulated as a suspension at up to 100 mg/ml in water containing hydroxypropylmethycellulose (HPMC)/Tween (0.5% hydroxypropylmethycellulose + 0.1% polysorbate 80; Sigma-Aldrich, UK), which served as the vehicle control. Generally, AZD7687 and vehicle were formulated on the day prior to study start and remade every 7 days where necessary. The suspensions were stirred continuously from preparation throughout use and protected from light.
Experimental Design
Doses and frequency of dosing were selected following consideration of species-specific tolerability and pharmacokinetic parameters. Doses predicted to result in exposures that were likely to cause maximum DGAT1 inhibition without resulting in intolerability were chosen as the highest dose level. Animals were randomized into groups according to their body weight and dosed daily with AZD7687 or vehicle, orally by gavage. Toxicokinetic sampling on the last day of dosing prior to necropsy was carried out to assess the exposure profiles of animals given AZD7687.
Experimental Procedures
Up to 32 (16 male and 16 female) mice, Crl:CD-1(ICR), per dose group were obtained from the AstraZeneca breeding unit. The animals, aged 7 to 8 weeks, were allowed to acclimatize for at least 1 week and were multiple housed, sexes separately, up to 5/cage. Water from the site drinking water supply and RM1 (E) SQC pelleted diet supplied by Special Diets Services Ltd., England, was freely available. Nesting material (Tapvei Aspen Chips, Finland, Tapvei small aspen bricks, sizzle nests and paper shavings, and polycarbonate tunnels; Datesand, UK) was provided. Animals were terminated for scheduled necropsy on the day after the final dose. The mice were euthanized by administration of halothane. AZD7687 was administered orally, once daily, to mice at 100, 300, and 600 mg/kg for 7 days, 0, 10, 80, and 300 mg/kg for 1 month, and 0, 1, 10, 60, and 300 mg/kg for up to 6 months.
In order to compare the effects of AZD7687-mediated DGAT1 inhibition on the skin with skin effects due to genetic DGAT1 deficiency, DGAT1 (−/−) mice were obtained from the AZ breeding unit (AstraZeneca Mölndal, Gothenburg, Sweden). To construct the Dgat1 gene targeting vector, 5.0-, 3.4-, and 1.5-kb genomic DNA fragments were prepared from a bacterial artificial chromosome (BAC) clone, which contained approximately 200-kb C57Bl/6 genomic sequence including the Dgat1 gene (ResGen, Invitrogen Corporation, Carlsbad, CA), for 5′ homology, deletion, and 3′ homology sequences, respectively. The fragments were cloned into a loxP-floxed PGKneo plasmid, using standard cloning techniques. In brief, a 3.3-kb fragment containing exons 3 to 17 of Dgat1 was inserted between an isolated loxP site and loxP-flanked PGK-Neomycin resistance cassette and then inserted between the 2 homology sequences. The Dgat1 targeting vector was linearized and electroporated into C57Bl/6 (PRX-B6N #1, Primogenix, St. Louis, MO) mouse embryonic stem (ES) cells, and ES cell clones were selected with G418 (300 µg/ml). Candidate ES cell clones were screened by polymerase chain reaction (PCR), and targeting was confirmed by Southern blot analysis using standard protocols. Targeted ES cells were injected into BALB/cAnNCrl blastocysts, and injected embryos were implanted into the uteruses of pseudopregnant BALB/cAnNCrl mice. Chimeric males were mated with C57BL/6 females, and the resulting heterozygous F1 males were bred with Rosa26Cre transgenic mice (Gt(ROSA)26Sortm1.1(cre)Jphe) on a C57BL/6 background to delete the loxP-floxed region, including exons 3 to 17 in Dgat1, and the Neomycin cassette. DGAT1 (−/−) mice (B6-Dgat1tm1Aztc) were obtained by heterozygote intercrossing, and the mutations were confirmed by reverse transcriptase-PCR analysis. Animals were kept in pathogen-free environment and routinely screened for pathogens.
Up to 8 (4 male and 4 female) beagle dogs per treatment group were obtained from the AstraZeneca breeding unit (Alderley Park, Macclesfield, UK). The animals, aged 12 to 24 months, were allowed to acclimatize for at least 1 week.
AZD7687 was administered orally, once daily, to dogs at 100 and 1,000 mg/kg/day for 7 days; and twice daily at 0, 20, 160, and 2,000 mg/kg/day for 14 days; 0, 4, 20, 140, and 500 mg/kg/day for up to 3 months; and 0, 4, 16, and 100 mg/kg/day for up to 6 months. All animals were observed daily for clinical signs of toxicity. Body weight and food consumption were measured daily prior to dosing. Animals were single penned, with access to an exercise area with others of the same sex and group, for the major part of each day. Animals had free access to water from the site drinking water supply. A set weight of SDS Dog-D3 (E) SQC, supplied by Special Diet Services Ltd., England, was provided daily, in the afternoon, access being allowed for up to approximately 2 hr. Toys (e.g., bones) were provided for additional environmental enhancement. Animals were terminated for scheduled necropsy on the day after the final dose. The dogs were killed by intravenous administration of sodium pentobarbitone and then exsanguinated. All studies were conducted in strict adherence to the U.K. Home Office regulations for animal welfare (Animal Scientific Procedures 1986 Act).
For pharmacokinetic analysis, blood samples were collected into lithium heparin tubes from animals on either the first or penultimate day of the study. Plasma was separated by centrifugation at 4°C and stored for analysis at −20°C. Plasma concentrations of AZD7687 were determined by liquid chromatography-mass spectrometry, and pharmacokinetic parameters of maximum plasma drug concentration (Cmax) and minimum plasma drug concentration at the dose interval (Cmin) were calculated. Exposure data are presented as free drug concentration in blood plasma required to inhibit the microsomal DGAT1 enzyme by 50% (FDM) and are mean data for each dose level. Due to a significant (~1.5–3×) sex difference in mice, separate data points are given for male and female mice. There was no sex difference in dogs, and data points are mean data of males and females combined.
At the end of each experiment, animals were terminally anesthetized, and representative samples of skin (mammary gland region for mice, abdominal region for dogs) were taken at necropsy, placed in 10% buffered formalin, and processed for histopathological assessment. Conventional processing into wax and H&E staining of sections preceded their light microscopic examination.
Light microscopic examination and histology scoring were performed by a certified pathologist, and findings were peer reviewed by a second pathologist. Each section of skin contained from 15 to 25 hair follicles in different stages of differentiation with associated sebaceous glands that were cut in multiple planes. There was no difference in sebaceous gland appearances within or between tissue sections from control animals, that is, there was no apparent alteration in sebaceous cell morphology related to stage of hair follicle differentiation. Therefore, assessment of sebaceous cell/gland atrophy was based on their appearances irrespective of the stage of differentiation of the associated hair follicle in all instances. Severity scores ranged from no change by comparison with controls to severe, which was defined as the total absence of sebaceous glands in all skin tissue sections examined from an individual animal. Minimal severity was defined as sebaceous cell numbers per gland being as that seen in controls but cell size was uniformly reduced, mild as a small reduction in sebaceous cell numbers but with substantially reduced (and variable) cell size, and moderate, as very few cells remaining that appeared shrunken.
Results
Effects of DGAT1 Inhibition in the Mouse
The DGAT1 inhibitor AZD7687 was administered to mice, orally, once daily, for 7, 28, or 180 days at doses predicted to cause minimal to substantial DGAT1 inhibition over the dose interval. Toxicokinetic analysis revealed that Cmax was above the mouse microsomal DGAT1 enzyme IC50 at all dose levels, ranging from 4× IC50 in the 1 mg/kg/day dose group in the 180-day study to 1,581× IC50 in the 600-mg/kg/day dose group in the 7-day study. For all dose levels and durations, Cmin was above the DGAT1 IC50 for the duration of the dose interval, ranging from just 1× IC50 in the 1-mg/kg/day dose group in the 180-day study to 766× IC50 in the 600-mg/kg/day dose group in the 7-day study, respectively.
In the mouse, AZD7687 administration was well tolerated in all studies. However, significant drug-induced sebaceous gland atrophy in the skin characterized by a reduction in size of sebaceous glands attached to each hair follicle was detected upon histopathological examination of skin samples (Figure 1). Histopathological data from the 1- and 6-month studies indicated that sebaceous gland atrophy did not occur when group mean exposures were ≤6× FDM at Cmax and 1.5× at Cmin during the dose interval. Sebaceous gland atrophy of minimal severity occurred at mean exposures of 22× FDM at Cmax and 10× Cmin, indicating the necessity of a high level of continuous DGAT1 enzyme inhibition for this morphological change to occur. At higher exposures, minimal to mild sebaceous gland atrophy was noted at 50 to 260× FDM at Cmax and 7 to 40× FDM at Cmin, and mild to moderate sebaceous gland atrophy was noted at an exposure of 226× FDM at Cmax and 46× FDM at Cmin. The relationship between FDM (Cmax and Cmin) during the dose interval and severity of sebaceous gland atrophy is shown in Figure 2.
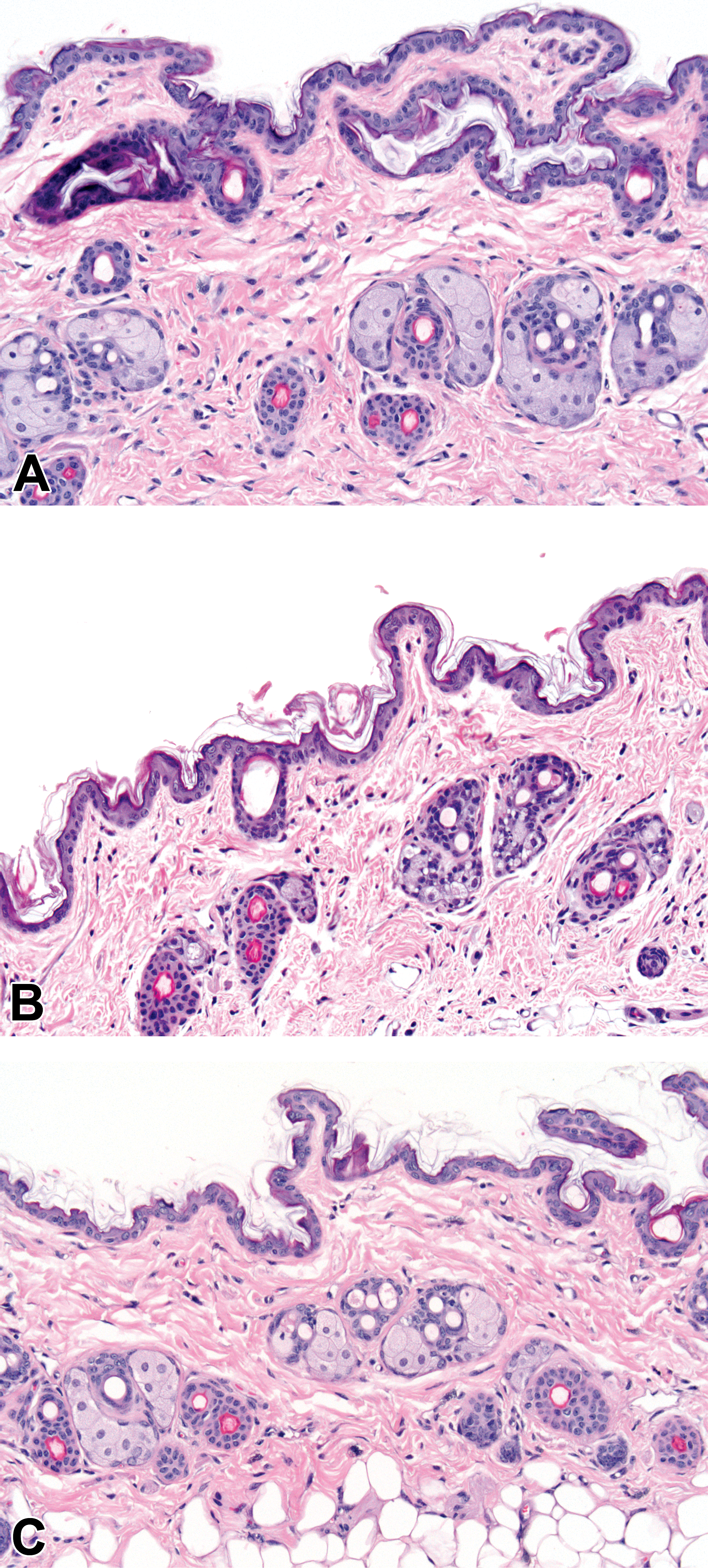
Photomicrographs of skin from (A) a control mouse, (B) a mouse dosed with 300-mg/kg/day AZD7687 for 28 days, and (C) a mouse dosed with 300-mg/kg/day AZD7687 for 28 days followed by a drug-free recovery period of 28 days. Note the normal appearance of sebum-filled sebaceous glands in (A) and (C), and the severely atrophic sebaceous glands with substantially reduced sebum in (B). (H&E; original objective lens magnifications ×20.)
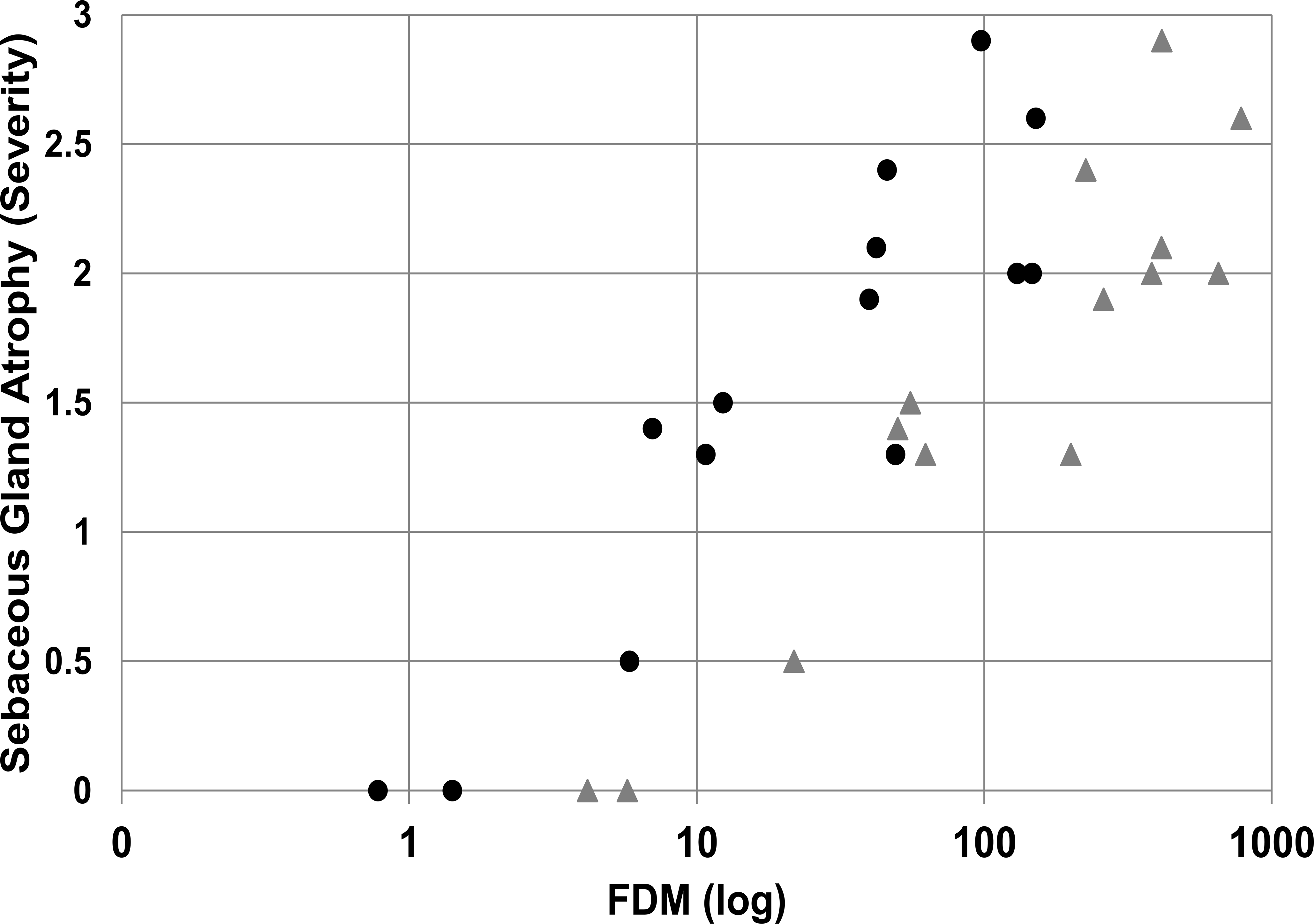
Effects of different degrees of AZD7687-mediated DGAT1 inhibition, expressed as FDM (log), on sebaceous gland atrophy at Cmax (gray triangles) and Cmin (black circles) in mice. A dose-related increase in sebaceous gland atrophy is demonstrated. The score for severity of sebaceous gland atrophy is 0 (absent), 1 (minimal), 2 (mild), and 3 (moderate). Data are group means for males and for females (n ≥ 10/sex/group for histopathology data and n = 3/sex/group for exposure data) and are taken from the 1- and 6-month studies. Cmax = maximum plasma drug concentration; Cmin = minimum plasma drug concentration at dose interval; DGAT1 = diacylglycerol O-acyltransferase 1; FDM = free drug concentration in blood plasma required to inhibit the microsomal DGAT1 enzyme by 50%.
In addition to a relationship between AZD7687 exposure and severity of sebaceous gland atrophy, an association between duration of exposure and severity of this finding was observed. Although sebaceous gland atrophy was absent in mice treated with high doses of AZD7687 for up to 7 days, the group mean severity of this finding ranged from minimal to mild after 1 month (28 days) of dosing, which increased to minimal to moderate severity after 6 months (180 days) of dosing (Figure 3). Following a 4-week drug-free period, the observed sebaceous gland atrophy noted after 1 month of treatment had resolved, indicating complete reversibility of this finding (Figure 1). However, reversibility was not assessed following 6 months of dosing.
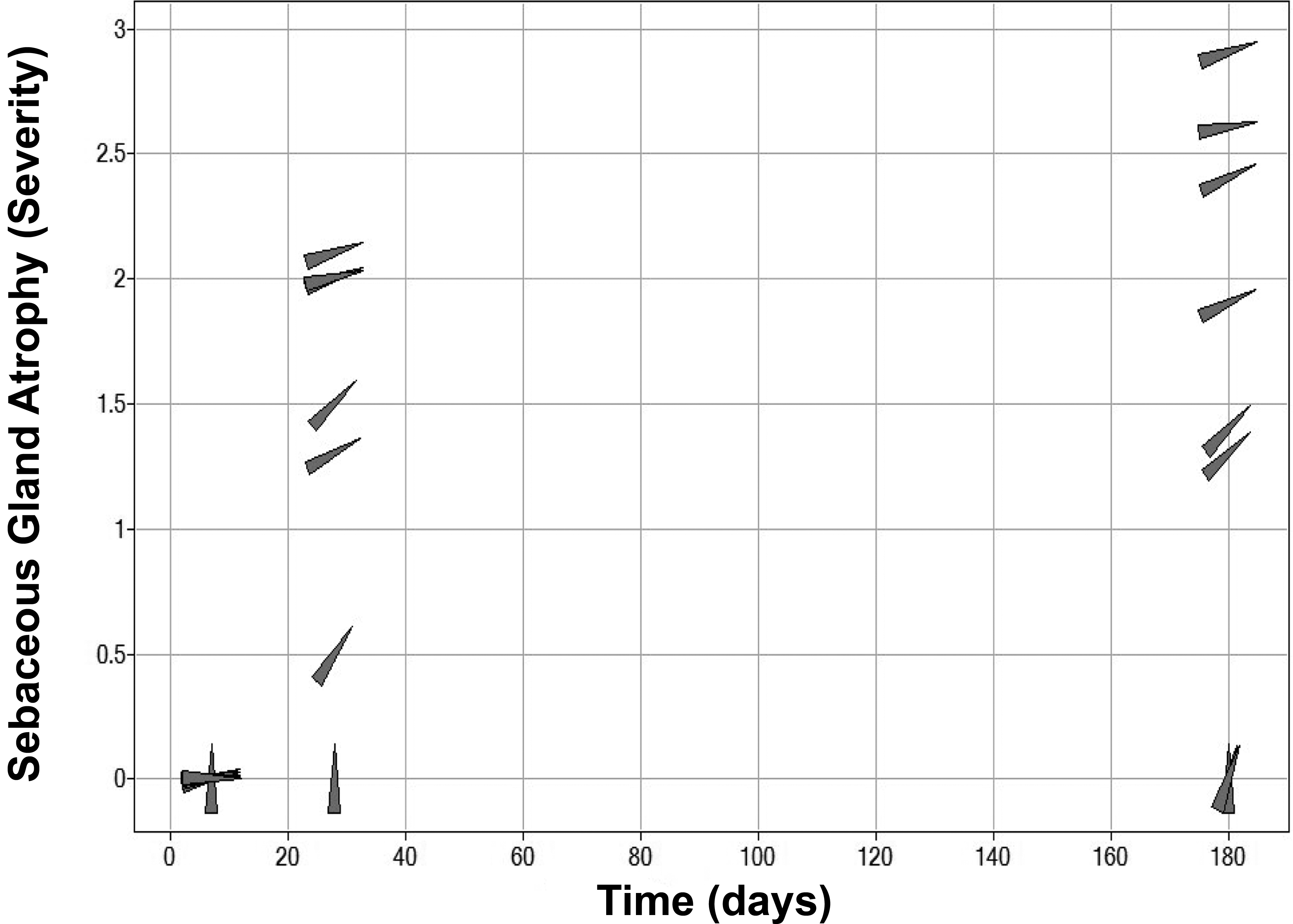
Effects of the duration of AZD7687 dosing and different levels of DGAT1 inhibition, expressed as FDM, on sebaceous gland atrophy in mice. A time- and dose-related increase in sebaceous gland atrophy is demonstrated. Data points (gray triangles) are rotated to the right by increasing level of DGAT inhibition. The score for severity of sebaceous gland atrophy is 0 (absent), 1 (minimal), 2 (mild), and 3 (moderate). Data are group means for males and females (n ≥ 10/sex/group for histopathology data and n = 3/sex/group for exposure data at Cmax). Cmax = maximum plasma drug concentration; DGAT1 = diacylglycerol O-acyltransferase 1; FDM = free drug concentration in blood plasma required to inhibit the microsomal DGAT1 enzyme by 50%.
Although histopathological findings in the skin, following administration of AZD7687 for 1 month, were restricted to sebaceous gland atrophy only, after dosing for 6 months, this finding was also associated with epidermal hyperplasia, occasional hair follicle atrophy, as well as in-life observation of hair loss and skin lesions (i.e., open wounds and scabs). These additional skin findings occurred at exposure levels of at least 50× FDM at Cmax and 7× FDM at Cmin, levels that were sufficient to cause substantial DGAT1 inhibition during the dose interval and were noted to be more significant at higher levels of sebaceous gland atrophy (Figure 4). There were no notable sex differences, the only exception being the increased incidence in skin lesions observed in-life, which was higher in males than in females. The histopathological phenotype observed following substantial DGAT1 inhibition for 6 months closely resembled that of DGAT1 knockout mice. Figure 5 shows skin photomicrographs of male wild-type and DGAT1(−/−) mice as well as male mice treated with vehicle or AZD7687 for 6 months.
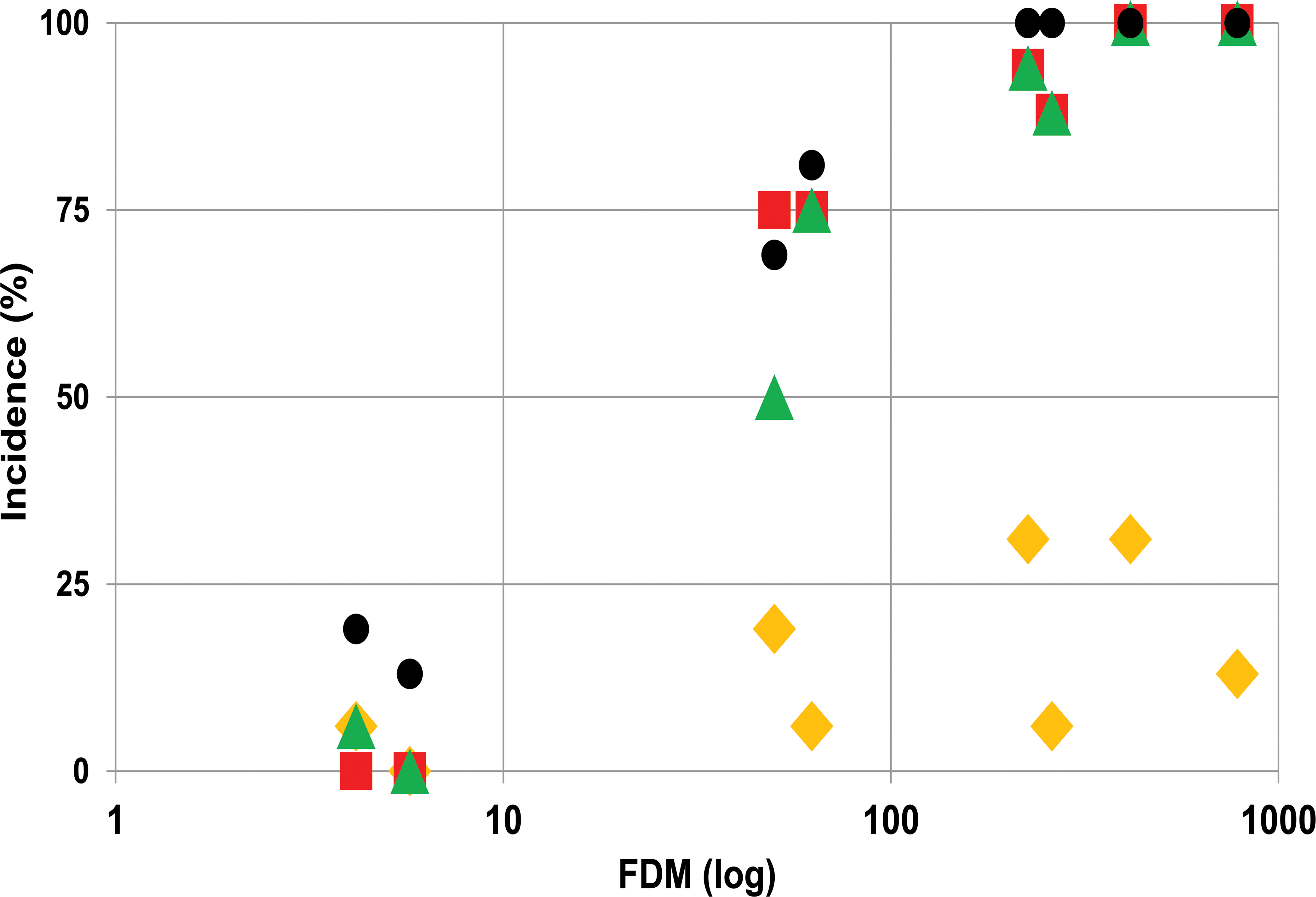
Effects of different degrees of DGAT1 inhibition, expressed as FDM, on sebaceous gland atrophy (red squares), epidermal hyperplasia/hyperkeratosis (green triangles), and in-life observations of hair loss (black circles) and skin lesions (yellow diamonds) in mice dosed with AZD7687 for 6 months. Data are group means for males and females (n ≥ 10/sex/group for histopathology data and n = 3/sex/group for exposure data). DGAT1 = diacylglycerol O-acyltransferase 1; FDM = free drug concentration in blood plasma required to inhibit the microsomal DGAT1 enzyme by 50%.
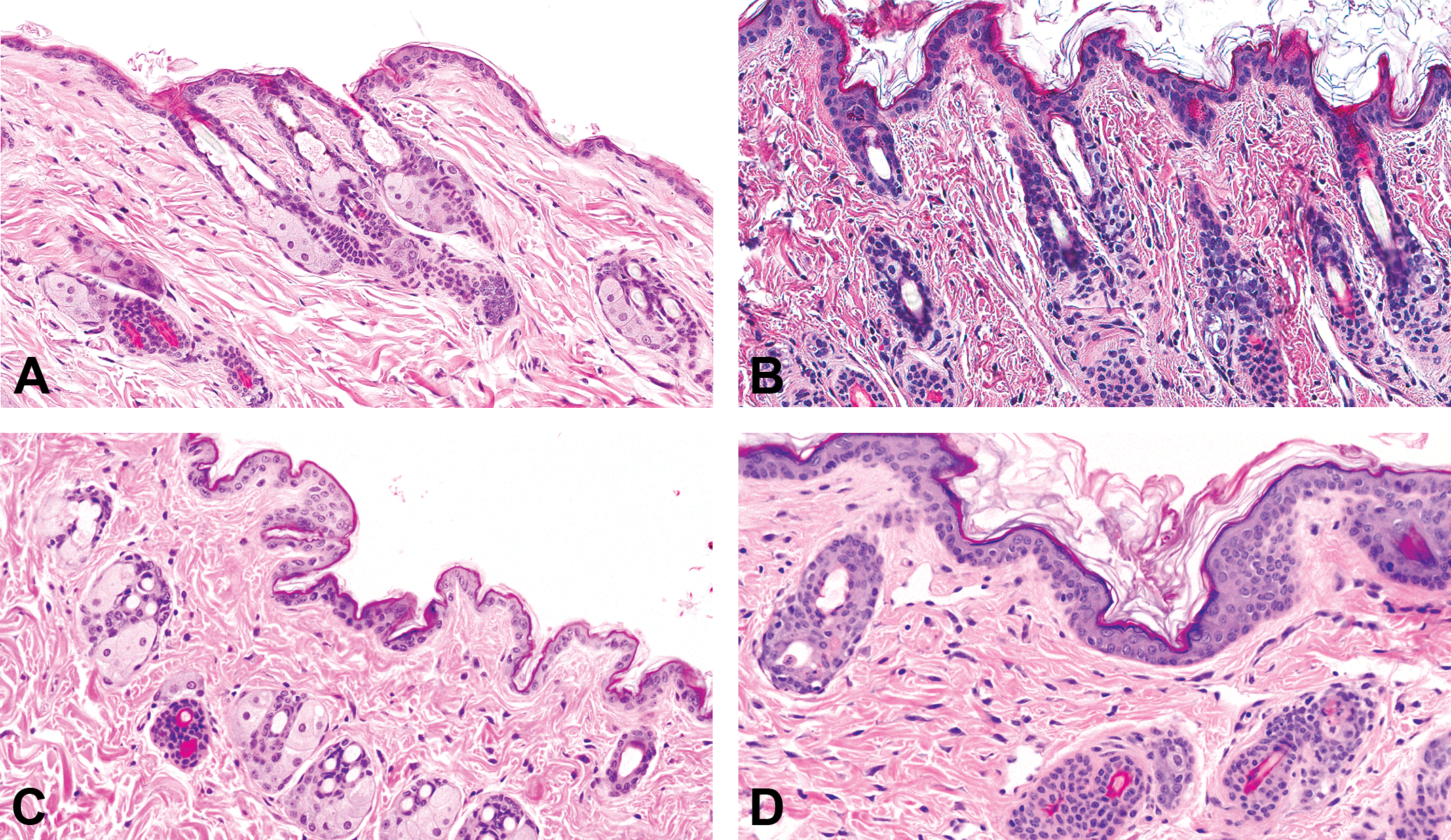
Photomicrographs of skin from (A) a male control mouse, (B) a male mouse dosed with 300 mg/kg/day AZD7687 for 6 months, (C) a male DGAT wild-type mouse, and (D) a male homozygous DGAT1 knock out (−/−) mouse. Note the normal appearance of sebum-filled sebaceous glands in (A) and (C), and the substantial atrophy of sebaceous glands with much reduced sebum and mild hyperkeratosis in (B) and (D). (H&E; original objective lens magnifications ×20.) DGAT1 = diacylglycerol O-acyltransferase 1.
Effects of DGAT1 Inhibition in the Dog
The DGAT1 inhibitor AZD7687 was administered to dogs, orally, once daily for 7 and twice daily for 28 or 180 days at doses predicted to cause minimal to marked DGAT1 inhibition over the dose interval. Toxicokinetic analysis revealed that Cmax was above the dog microsomal DGAT1 enzyme IC50 at all dose levels, ranging from 10× IC50 in the 4-mg/kg/day dose group in the 3-month study to 292× IC50 in the 1,000-mg/kg/day dose group in the same study. For all dose levels and durations, Cmin was above the DGAT1 IC50 for the duration of the dose interval, ranging from just 1× IC50 in the 4-mg/kg/day dose group in the 3-month study to 54× IC50 in the 1,000-mg/kg/day dose group in the 7-day study.
AZD7687 administration was well tolerated in all studies. As in the mouse, drug-induced sebaceous gland atrophy in the skin was detected following histopathological examination (Figure 6) and a clear dose–response relationship was noted. Thus, group mean exposures of up to 12× FDM at Cmax and 2× at Cmin during the dose interval did not result in sebaceous gland atrophy. However, minimal sebaceous gland atrophy occurred at mean exposures of 32× FDM at Cmax and 7× FDM at Cmin, indicating that this level of continuous DGAT1 enzyme inhibition was required for this morphological change to occur. At higher exposures, minimal to mild sebaceous gland atrophy was noted at 48 to 193× FDM at Cmax and 7 to 54× FDM at Cmin and mild to moderate sebaceous gland atrophy was noted at an exposure above 165× FDM at Cmax and 36× FDM at Cmin. The relationship between FDM (Cmax and Cmin) and severity of sebaceous gland atrophy is shown in Figure 7.
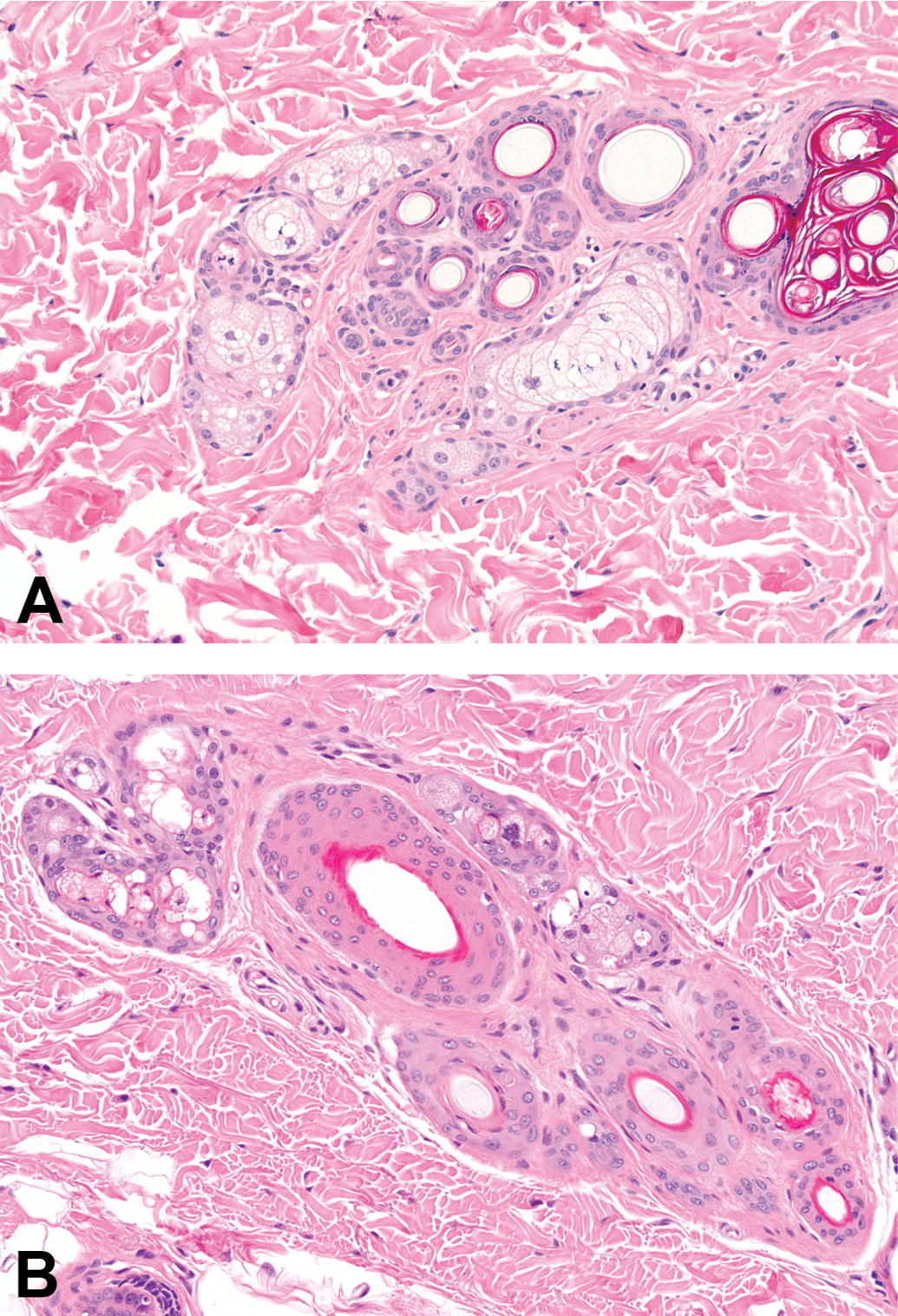
Photomicrographs of skin of (A) a control dog and (B) a dog dosed with 100-mg/kg/day AZD7687 for 6 months. Note the prominent sebum-filled sebaceous glands in (A), and the atrophic sebaceous glands that contain little sebum in (B). (H&E; original objective lens magnifications ×20.)
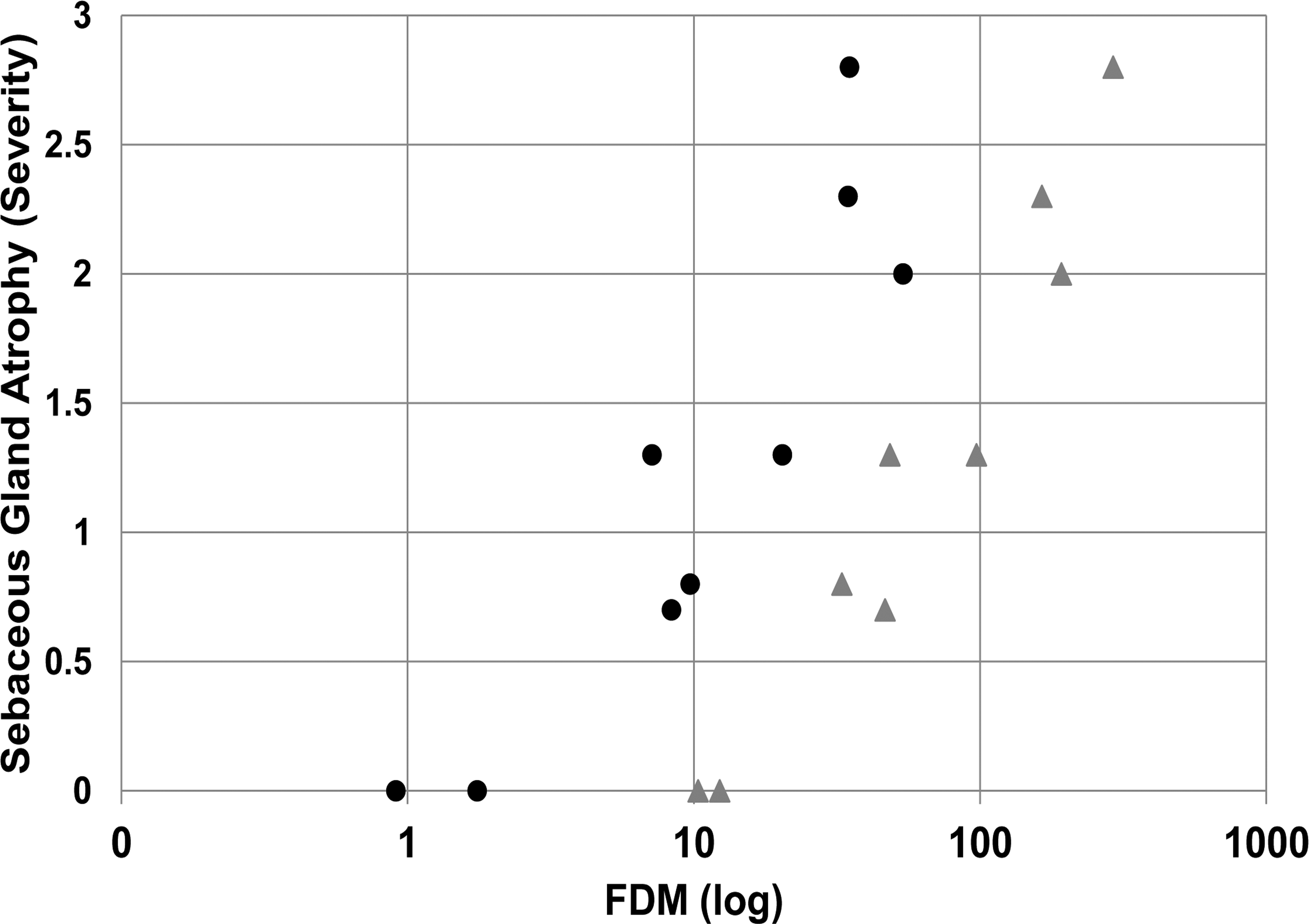
Effects of different degrees of AZD7687-mediated DGAT1 inhibition, expressed as FDM (log), on sebaceous gland atrophy at Cmax (gray triangles) and Cmin (black circles) in dogs. A dose-related increase in sebaceous gland atrophy is demonstrated. The score for severity of sebaceous gland atrophy is 0 (absent), 1 (minimal), 2 (mild), and 3 (moderate). Data are combined group means for males and females (n = 4/sex/group for histopathology data and exposure data) and are taken from the 14-day and 6-month studies. Cmax = maximum plasma drug concentration; Cmin = minimum plasma drug concentration at dose interval; DGAT1 = diacylglycerol O-acyltransferase 1; FDM = free drug concentration in blood plasma required to inhibit the microsomal DGAT1 enzyme by 50%.
A clear relationship to duration of exposure and severity of sebaceous gland atrophy was also evident. Although sebaceous gland atrophy was absent in dogs dosed for up to 7 days, the group mean severity of this finding ranged from minimal to mild after 14 days of dosing with an increase to minimal to moderate mean severity after 3 months (90 days) of dosing and further increase to moderate level in the highest dose group after 6 months (180 days; Figure 8). Following a 4-week drug-free period, the observed sebaceous gland atrophy noted after 3 months of dosing had resolved, indicating complete reversibility of this finding. Reversibility was not assessed following 6 months of dosing.
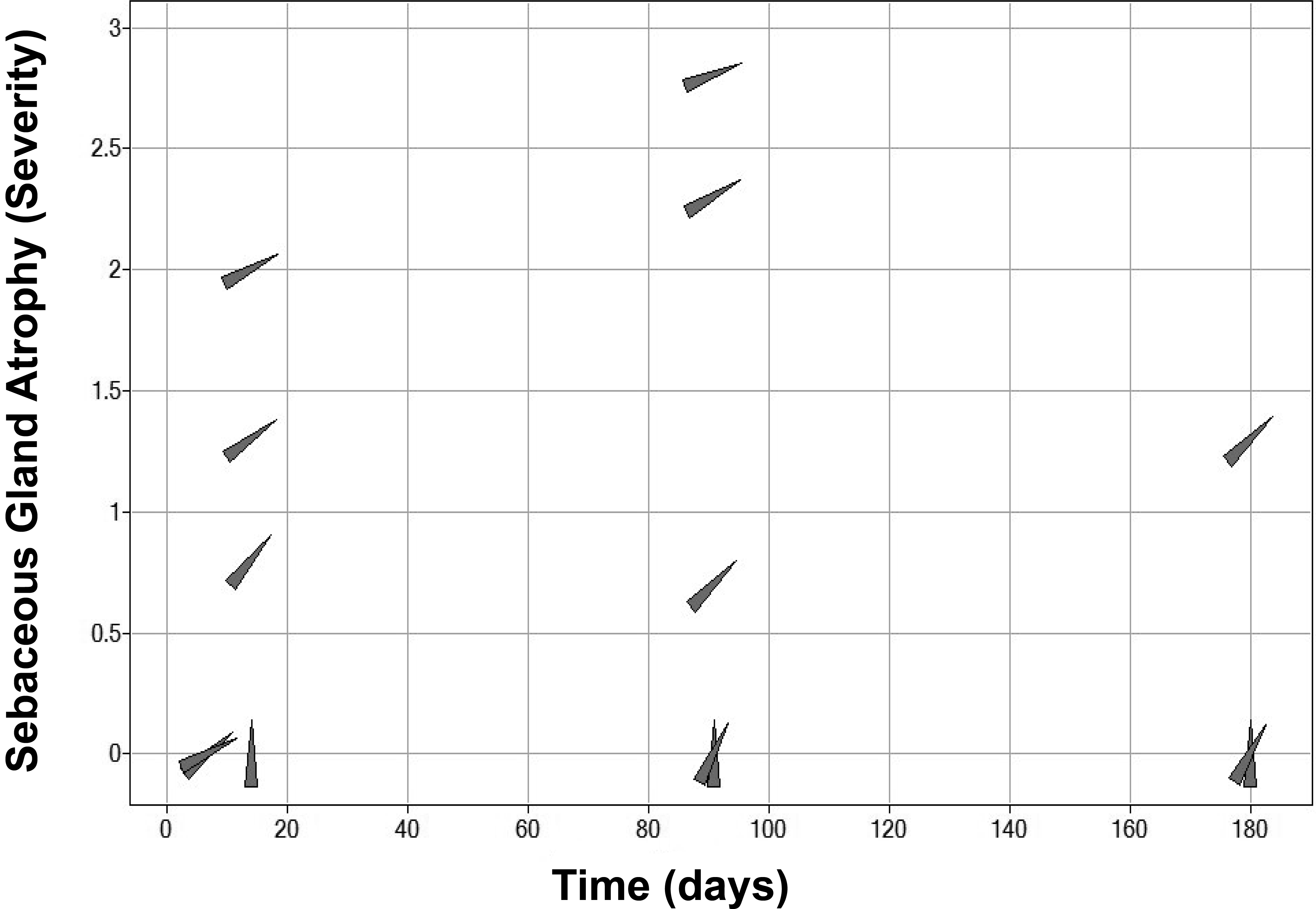
Effects of duration of AZD7687 dosing and different degrees of DGAT1 inhibition, expressed as FDM, on sebaceous gland atrophy in dogs. A time- and dose-related increase in sebaceous gland atrophy is demonstrated. Data points (gray triangles) are rotated to the right by increasing level of DGAT1 inhibition. The score for severity of sebaceous gland atrophy is 0 (absent), 1 (minimal), 2 (mild), and 3 (moderate). Data are combined group means for males and females (n = 4/sex/group for histopathology data and exposure data at Cmax). Cmax = maximum plasma drug concentration; DGAT1 = diacylglycerol O-acyltransferase 1; FDM = free drug concentration in blood plasma required to inhibit the microsomal DGAT1 enzyme by 50%.
In addition to sebaceous gland atrophy, minimal hyperkeratosis of the epithelium covering the infundibula of hair follicles was observed in the dog following exposure to AZD7687 for 6 months. However, the additional skin changes observed after 6 months of treatment in the mouse (i.e., occasional hair follicle atrophy, as well as in-life observation of hair loss and skin lesions) were not observed in the dog.
Discussion
Following administration of AZD7687, sebaceous gland atrophy was noted at Cmax/Cmin levels of at least 22×/10× and 32×/7× FDM, but was absent at 6×/1.5× and 12×/2× times the FDM in the mouse and the dog, respectively. This is indicative of the requirement for substantial levels of DGAT1 inhibition, sustained throughout the dosing interval, for induction of sebaceous gland atrophy. Furthermore, with regard to duration of exposure in both species, inhibition of DGAT1 by AZD7687 for more than 7 days was required for microscopical detection of sebaceous gland atrophy, demonstrating that not only substantial but also prolonged inhibition of DGAT1 was a key requirement.
In the mouse, but not in the dog, inhibition of DGAT1 for 6 months also resulted in significant epidermal hyperplasia, occasional hair follicle atrophy, as well as in-life observation of hair loss and skin lesions. This phenotype, which was only seen at high levels of DGAT1 inhibition, was similar to that seen in the DGAT1 (−/−) mouse that exhibit dry fur and hair loss and which is associated with atrophic sebaceous glands and fur lipid abnormalities, especially the absence of wax esters (Chen et al. 2002; Yen et al. 2005). Fur and sebaceous gland abnormalities were most apparent in postpubertal male DGAT1 (−/−) mice, suggesting a potential hormone-based sex difference (Chen et al. 2002). Our data did not indicate a gender difference in sebaceous gland atrophy, epidermal hyperplasia or hyperkeratosis, or in-life hair loss following administration of AZD7687. However, the increased incidence of in-life skin lesions was more pronounced in male animals and may indicate some role of androgens in mediating the effects of DGAT1 inhibition on the skin, although it cannot be excluded that fighting, which is commonly observed in group housed males, was a significant influence.
In the search for a mechanism by which DGAT1 deficiency in DGAT (−/−) results in skin abnormalities, Shih et al. (2009) demonstrated that retinol deprivation prevents alopecia in DGAT (−/−) mice, leading to the hypothesis that DGAT1 functions as an ARAT in murine skin and that inhibition of DGAT1 may result in an increase in retinoic acid levels and subsequent retinoid toxicity. Hair loss is a consistent finding during retinoid toxicity (Everts 2012). The potential for pharmacological inhibition of DGAT1 by prolonged dosing with AZD7687 in the mouse affecting the skin by the same mechanism is worthy of consideration. However, although retinoid homeostasis is an important factor in normal dog and human skin development and retinoids are important agents in the treatment of acne (Smith and Thiboutot 2008; Everts 2012), there is currently no understanding of whether or not DGAT1 inhibition can affect retinoid homeostasis and subsequent hair development or maintenance in the dog or in human. Our observations in the mouse suggest that DGAT1-mediated sebum production plays an important role in the skin and in hair development and their maintenance, although the exact function of the sebaceous gland and the effects of quantitative and qualitative differences in sebum on hair development in dogs or indeed in humans are not clear.
In the dog, we observed sebaceous gland atrophy and minimal hyperkeratosis of the epithelium only, but not the more severe phenotype that included hair loss, dry fur, and skin lesions observed in the mouse, which implies that a species difference in the role of DGAT1 in fur maintenance of the mouse and the dog may exist. Experimentally derived information that could account for any differences in the role of DGAT1 in mouse and dog skin is sparse. In both species, a high content of type II diester waxes is present, although the proportion of unsaturated diesters is higher in the mouse while the proportion of saturated branched diesters is higher in the dog (Nikkari 1974). It remains unclear as to whether or not this qualitative disparity could account for the observed species differences. Comparisons of the skin surface lipids of humans with those of animals reveal that only in humans are skin lipids predominantly (˜60%) triglycerides and their breakdown products (i.e., mono- and diglycerides) as well as free fatty acids, with wax esters and squalene making up most of the remainder (Nicolaides, Fu, and Rice 1968). By contrast, neither mice nor dogs have significant amounts of triglycerides or free fatty acids in their skin surface lipid: the main products of surface lipids on furry animals are a wide variety of mono- and diester waxes. Interestingly, the pig, like human, has a sparse pilosebaceous population and most of its skin lipid classes are similar to those obtained from man (Nicolaides, Fu, and Rice 1968). Importantly, humans are the only species with a generalized eccrine sweating system, which may play a significant role in providing skin with emulsifying glycerides and limit the importance of the role of the sebaceous unit in maintaining skin integrity and function.
Although pharmacological inhibition of DGAT1 in the mouse indicates a potential for alopecia, this is not supported by our data in the dog, and given the differences between human and animal skin surface lipids and the role of the eccrine sweating system in man, the effects of prolonged DGAT1 inhibition on the integrity of the human skin and scalp may be less significant.
However, a change in lipid composition or in reduction of skin surface lipids in man has been implicated in the pathogenesis of atopic dermatitis (Sator, Schmidt, and Hönigsmann 2003); and thus, it will be important to monitor for effects of DGAT1 inhibition on the skin in clinical trials with AZD7687. Interestingly, although sebaceous gland, sebaceous gland duct, or hair follicle inflammation is a common finding in human asebia (Al-Zaid et al. 2011), a significant increase in inflammation was not seen in the skin of mice and dogs treated with AZD7687—folliculitis involving the sebaceous glands was very rare in the studies, and postinflammation scarring was not observed.
In conclusion, our data indicate that not all species show the same degree of adverse skin changes as a consequence of DGAT1 inhibition and that these adverse changes occur only at sustained and substantial levels of DGAT1 inhibition or in DGAT1 deficient mice. Additionally, the high level of inhibition responsible for the adverse phenotype in the mouse gives some confidence that a significant margin to the desired primary pharmacology (i.e., loss of body weight and/or body fat) will permit progression of DGAT1 inhibitors through clinical development. To date, no effects of AZD7687 administration on the skin of healthy volunteers have been noted in clinical trials of short duration (Denison et al. 2013). It is noteworthy that our data are also supportive of the use of pharmacological inhibition of DGAT1 in the treatment of hyperseborrheic conditions such as acne, where a DGAT1 inhibitor–mediated atrophy of the sebaceous gland may result in a clinically relevant reduction in sebum production.
Footnotes
Acknowledgments
The authors would like to acknowledge all of their colleagues within AstraZeneca who have contributed to this research. The DGAT1 (−/−) mice were kindly generated and supplied by John Wiseman at the RAD transgenic unit at AstraZeneca Mölndal.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
