Abstract
FHTR2163 is an antigen-binding fragment of a humanized immunoglobulin G1 monoclonal antibody directed against high-temperature requirement A serine peptidase 1 (HTRA1) that is being developed as a potential intravitreal (ITV) treatment for patients with geographic atrophy (GA), an advanced form of dry age-related macular degeneration. The nonclinical toxicology program was designed to assess the safety and tolerability of HTRA1 inhibition following ITV administration of FHTR2163 to support ITV administration in patients with GA. FHTR2163 was well tolerated in a single-dose ITV-administered 8-day toxicity study in cynomolgus monkeys following a 50 µL high (>700 mOsm/kg) osmolality formulation up to 12.5 mg/eye; however, 100 µL (2× 50 µL injections) of a high-osmolality formulation resulted in transient retinal detachment. Repeat-dose ITV administration every 2 weeks of FHTR2163 was well tolerated in 8- and 26-week studies with ITV injection of 100 µL (2× 50 μL) of iso-osmolar formulation up to 15 mg/eye, or 50 µL of the high-osmolality formulation up to 12.5 mg/eye. Observed transient and reversible ocular effects included inflammation and perivascular infiltrates, consistent with an immune response attributed to the administration of heterologous (humanized) protein. Overall, FHTR2163 was well tolerated, and the nonclinical package supported the continued clinical development of FHTR2163 in patients with GA.
Introduction
The high-temperature requirement A (HTRA) family of trypsin-like serine proteases are evolutionarily conserved in single and multicellular organisms, 1 and HTRAs are involved in numerous important cellular processes, including cell proliferation, migration, and fate. 2,3 In humans, there are 4 closely related human HTRA members, HTRA1, HTRA2, HTRA3, and HTRA4, which all share several common domains. 4 HTRA1 was shown to cleave a large number of substrates, many of which are extracellular matrix components. 4 -6 HTRA1 is implicated in various pathogenic and developmental processes, including osteoarthritis, Alzheimer's disease, neuronal maturation and development, tumor progression, and age-related macular degeneration (AMD). 5,7 -12 However, the precise biological role of HTRA1’s proteolytic activity, as it relates to the pathophysiology of AMD, remains elusive. 13
Early signs of AMD include the development of drusen, extracellular deposits between the retinal pigment epithelium (RPE) and Bruch’s membrane, and cause only minimal visual acuity impairment. As the disease advances and progresses into geographic atrophy (GA), a progressive loss of photoreceptor RPE and the choriocapillairis occurs, resulting in significant visual loss and blindness. 14 -16 The prevalence of GA increases exponentially with age and approximately quadruples per decade beyond 50 years of age. 17 There are currently no approved treatments for GA secondary to AMD, and a significant unmet medical need exists for treatment of this serious condition.
The
The pathogenesis of AMD is not well understood, but genetic and environmental factors appear to contribute to the disease process. In support of a role for HTRA1 in the pathophysiology and progression of AMD, transgenic mice overexpressing human HTRA1 in RPE cells recapitulated cardinal features associated with advanced AMD and polypoidal choroidal vasculopathy, including branching networks of choroidal vessels and severe degeneration of the elastic laminae. 22 At the molecular level, this phenotype appears to induce disruption of the Bruch’s membrane and dilation of choroidal vessels. 23 HTRA1 expressed by RPE 12,21,23 may contribute to drusen formation and inflammation in the extracellular space between the RPE and Bruch’s membrane, which are the hallmarks of early AMD. 22,24 By inhibiting HTRA1 protease activity, treatment with FHTR2163 may represent a novel therapeutic candidate option for the treatment of GA, which currently has no approved therapies.
This article describes the design, execution, results, and interpretation of a nonclinical toxicology program to support the development of FHTR2163, an antigen-binding fragment of a humanized immunoglobulin G1 (IgG1) monoclonal antibody directed against HTRA1, through phase II. It includes the process of species selection, information on the study design, and a description of the key findings. Notably, retinal detachments and hyper-reflective bands (HRBs) in the fovea and antidrug antibody (ADA)–related ocular inflammation were observed but were not considered directly related to FHTR2163 and not factored into the determination of the no-observed-effect-level (NOAEL).
Materials and Methods
Test Species
The International Council for Harmonisation Guideline S6, Preclinical Safety Evaluation of Biotechnology–derived Pharmaceuticals (commonly referred to as “ICH S6”) states that 2 pharmacologically relevant species, a rodent and non-rodent, should be used in general toxicology studies for the development of biologics. 25 FHTR2163 binds with comparable affinities to human, cynomolgus monkey, rabbit, and mouse HTRA1 orthologs and has demonstrable pharmacologic activity in these species (data not shown). However, rodents were not used for the toxicology evaluations because of (1) anatomical and physiological differences in the rodent eye that limits and complicates the repeat intravitreal (ITV) dosing procedure and (2) rapid clearance of therapeutic proteins after ITV injection, resulting in very low ocular exposures. 26,27 Rabbits were considered a choice for a second toxicology species. Although the rabbit eye is of adequate size for repeat-dose ITV injection, rabbits frequently develop fulminant ocular inflammation secondary to the repeat-dose ITV administration of humanized biologics. 28
The cynomolgus monkey eye most closely resembles the human eye anatomically (including the presence of a macula) and physiologically. 29 Additionally, cynomolgus monkey HTRA1 has 100% homology with the human HTRA1 catalytic domain. Furthermore, in vivo pharmacologic inhibition of HTRA1 enzymatic activity was observed following ITV injection of FHTR2163 into cynomolgus monkey eyes. 30 These data support the cynomolgus monkey as the only appropriate species due to physical anatomy of the eye, expression of the target epitope, functional engagement by FHTR2163, and favorable pharmacokinetic (PK) characteristics with respect to drug exposure and immunogenicity.
Naive male and female cynomolgus monkeys of Chinese origin (Covance Research Products, Inc) were used in the ITV-administered repeat-dose Good Laboratory Practice (GLP) studies and a single-dose non-GLP study (Table 1). Animals were group-housed in European guideline (ES 123) compliant pens and enriched with a bedding material under standard conditions, and all procedures complied with applicable animal welfare acts and were approved by the local institutional animal care and use committee.
List of Toxicity Studies Conducted With Bilateral Intravitreal Administration of FHTR2163 in Cynomolgus Monkeys.
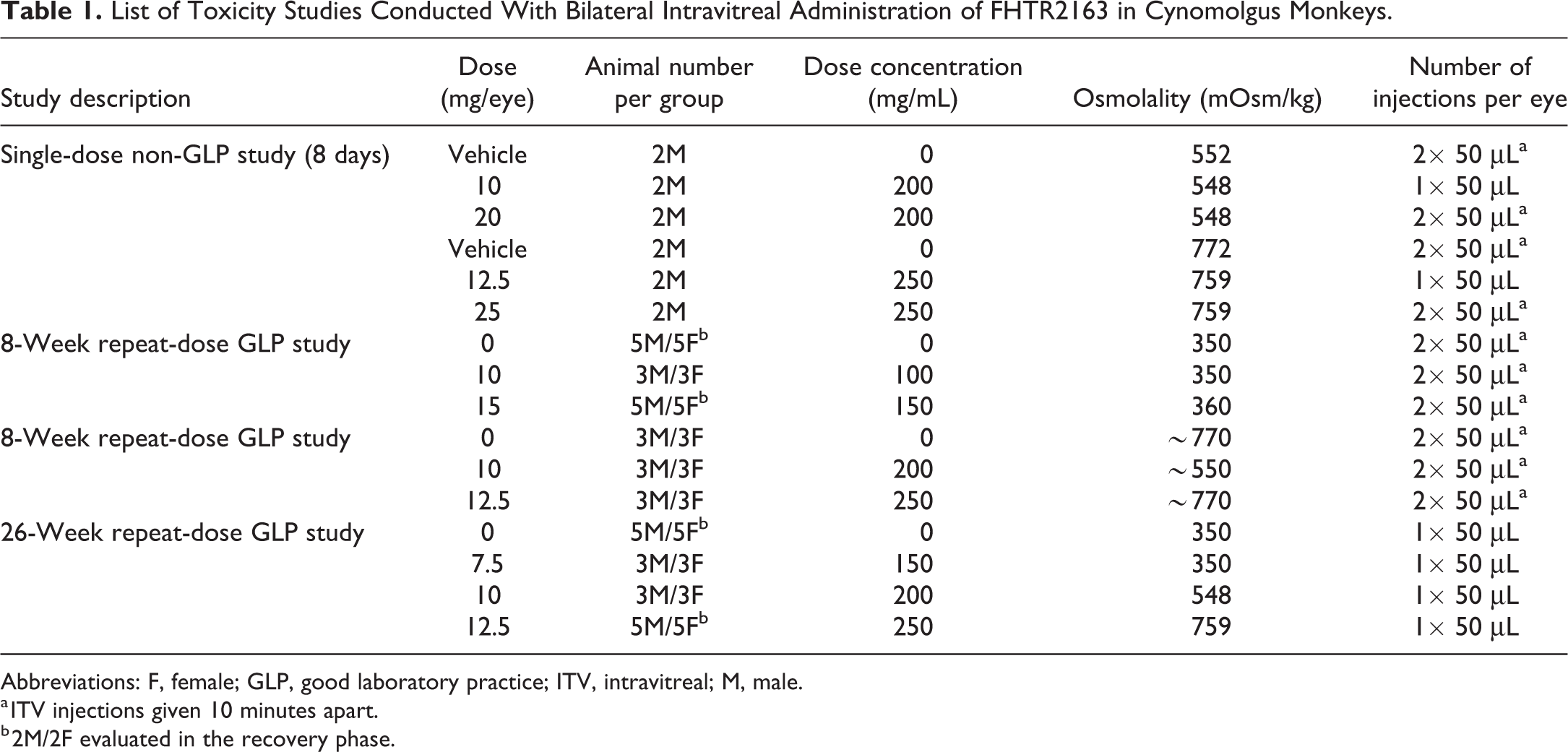
Abbreviations: F, female; GLP, good laboratory practice; ITV, intravitreal; M, male.
a ITV injections given 10 minutes apart.
b 2M/2F evaluated in the recovery phase.
Dose Administration
In each study, animals received bilateral ITV injections of vehicle control or FHTR2163 by a board-certified veterinary ophthalmologist. Before the ITV injections, animals were anesthetized with intramuscular injections of ketamine/dexmedetomidine. After aseptic preparation of the ocular surface, each eye received either a single 50 µL ITV injection or two 50 µL ITV injections administered 10 minutes apart, for a total volume of 100 μL/eye. Injections in the right eye were alternated, such that the first injection was administered at approximately the 7 o’clock position and the second injection was administered at approximately the 8 o’clock position. Injections in the left eye were alternated, such that the first injection was administered at approximately the 4 o’clock position and the second injection was administered at approximately the 5 o’clock position. A topical antibiotic (Tobrex) was instilled in each eye following dosing. A summary of the study dose groups in each study is provided in Table 1.
Non-GLP 8-day single-dose toxicity study
The ITV tolerability of high-osmolality FHTR2163 formulations was investigated in male cynomolgus monkeys (∼3-4 years of age). Two cohorts were used to investigate osmolality levels of ∼550 and ∼770 mOsm/kg, with 2 dose levels of FHTR2163 in each cohort (summarized in Table 1). For each osmolality cohort, 2 animals received bilateral ITV injections of vehicle, or mid or high dose of FHTR2163. Dose groups in cohort 1 (∼550 mOsm/kg) were 10 (mid) and 20 mg/eye (high), and dose groups in cohort 2 (∼770 mOsm/kg) were 12.5 (mid) and 25 mg/eye (high). Vehicle and high-dose group animals were administered two 50 µL ITV injections 10 minutes apart, and animals in the mid-dose group were administered a single 50 µL ITV injection.
GLP 8-week repeat-dose toxicity study with a 4-week recovery phase
Male and female cynomolgus monkeys (∼3-4 years of age) received repeat-dose bilateral ITV injections of vehicle or FHTR2163 (∼350 mOsm/kg) at dose levels of 10 or 15 mg/eye, given as two 50 µL ITV injections administered 10 minutes apart every 14 days, on days 1, 15, 29, 43, and 57. The vehicle and 15 mg/eye dose groups each had 5 animals/sex, whereas the 10 mg/eye had 3 animals/sex. On day 58, 3 animals/sex/group were necropsied and the remaining animals were followed for an additional 4-week recovery.
GLP 8-week repeat-dose toxicity study
Male and female cynomolgus monkeys (∼3-4 years of age) received repeat-dose bilateral ITV injections of high osmolality (>550 mOsm/kg) formulated vehicle or FHTR2163 at 10 or 12.5 mg/eye, given as a single 50 µL ITV injection every 14 days on days 1, 15, 29, 43, and 57. All animals were necropsied on day 58. The high-osmolality formulation was required to support high concentrations of FHTR2163 (≥250 mg/mL) and enable higher dose levels in a 50 µL ITV injection.
GLP chronic 26-week repeat-dose toxicity study with a 4-week recovery phase
Sexually mature male (≥5 years of age) and female (≥4 years of age) cynomolgus monkeys (3/sex/group) received repeat-dose bilateral ITV injections of vehicle or FHTR2163 (∼350 mOsm/kg) at dose levels of 7.5, 10, or 12.5 mg/eye, given as a single 50 µL ITV injection every 14 days on days 1, 15, 29, 43, 57, 71, 85, 99, 113, 127, 141, 155/156 (males/females), 169, and 183. The vehicle and 12.5 mg/eye dose groups each had 5 animals/sex, whereas the 7.5 and 10 mg/eye dose groups had 3 animals/sex. On day 184, 3 animals/sex/group were necropsied and the remaining animals were followed for an additional 4-week recovery.
Dosing frequency rationale for 26-week study
The objective was to cover a minimum concentration (
Study Procedures
Toxicity assessment
Comprehensive ophthalmic examinations (OEs) were conducted by a board-certified veterinary ophthalmologist and were performed at baseline and periodically throughout the studies. For repeat-dose administration studies, OEs were conducted predose on respective dosing days and 3 days postdose to assess acute ocular inflammation. Findings were graded using the modified McDonald-Shadduck scoring system. 33,34 Ocular imaging (ocular photography [OP]/fluorescein angiography [FA]) and intraocular pressure (IOP) were measured in conjunction with OEs. Spectral domain optical coherence tomography (SD-OCT) and electroretinography (ERG) evaluations were performed periodically during the dosing the phase of the repeat-dose studies and once at the end of the recovery periods. The SD-OCT and ERG evaluations were conducted by a vitreoretinal specialist and an electrophysiologist, respectively.
Assessment of toxicity was based on mortality, clinical signs, body weight, food consumption, OEs (slit-lamp biomicroscopy and indirect ophthalmoscopy), IOP, ERG, fundus OP, SD-OCT, and clinical and anatomical pathology evaluations of the eye. Additionally, in the GLP repeat-dose studies, safety pharmacology end points (neurological examinations and 8-lead electrocardiography [ECG]) were evaluated. Stage-aware microscopic evaluation of spermatogenesis in the testes was performed in the 26-week chronic study.
Toxicokinetic and ADA analysis
Blood was collected into tubes and allowed to clot at room temperature for at least 30 minutes. Blood samples were centrifuged for 10 to 15 minutes at approximately 1500 to 2000g, 2 °C to 8 °C within hour of collection, and the serum stored at 80 °C until analysis. Serum samples were analyzed using enzyme-linked immunosorbent assay–based methods for toxicokinetic (TK) and ADA. Toxicokinetic analysis included maximum observed concentration (
Results
Toxicokinetic and ADA Analysis
Sex differences in FHTR2163 exposure (serum mean
Group Mean Noncompartmental Toxicokinetic Parameter Estimates and Incidence of ADAs in Serum Following Repeat-Dose Intravitreal Administration of FHTR2163 in Monkeys.
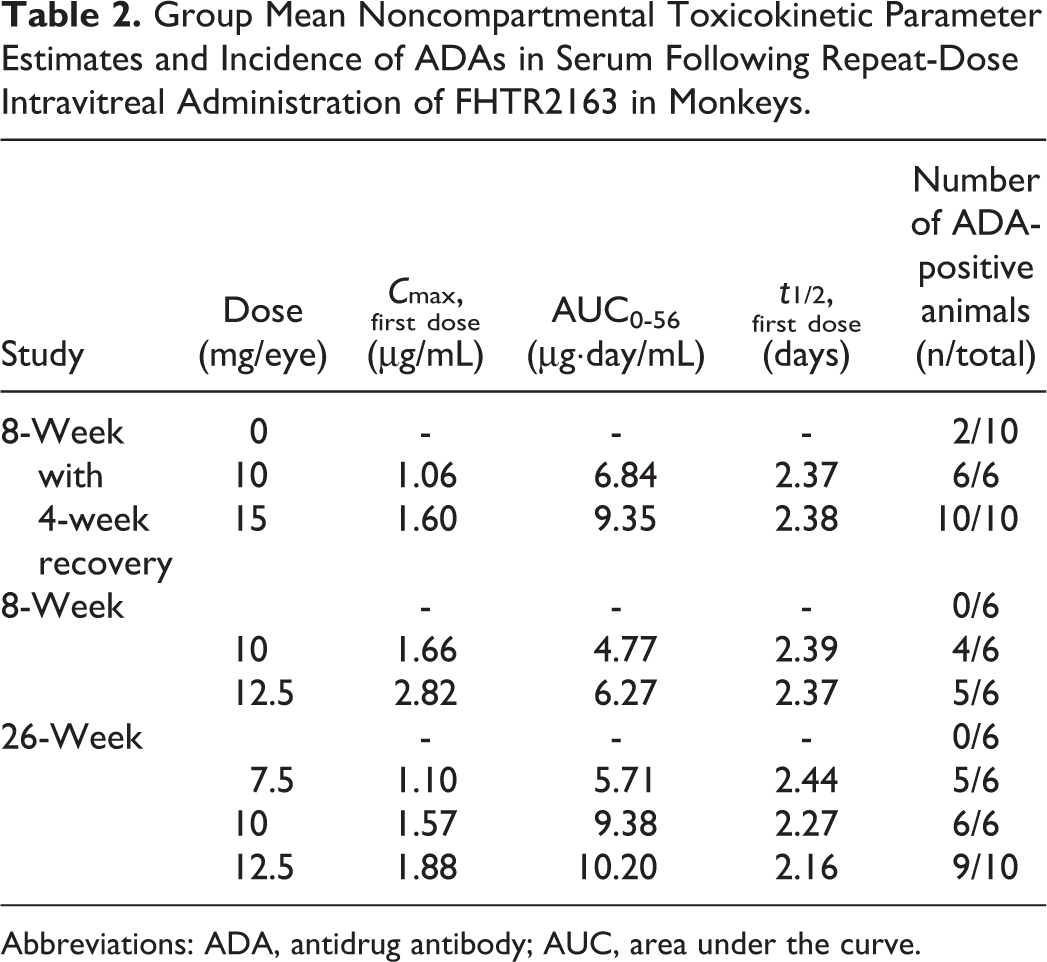
Abbreviations: ADA, antidrug antibody; AUC, area under the curve.
Animal Fate and Clinical Observations
There were no FHTR2163-related decreases in qualitative food consumption, body weight gain, IOP, OP, FA, and ERG. No systemic toxicity effects on the reproductive organs (including the reproductive tract, organ weights and histologic evaluation, and qualitative assessment of spermatogenic progression in males), safety pharmacology end points (respiratory rate, heart rate, body temperature, blood pressure, or ECGs) were observed in the GLP repeat-doses studies doses of ≤12.5 mg/eye for up to 26 weeks.
In the 26-week study, several females required veterinary anti-inflammatory therapy due to ocular inflammation. Treatment was prescribed by veterinary staff based on recommendations from the veterinary ophthalmologist, and dosing was suspended when considered necessary. One animal in 7.5 mg/eye dose group that developed severe uveitis was euthanized on day 46 after the fourth dose administration for animal welfare reasons. Additionally, ocular inflammation resulted in a pause in dosing of 2 animals, 1 animal from each 7.5 and 12.5 mg/eye dose groups. In all cases, the character and pattern of the inflammation was considered related to an immune-mediated response to FHTR2163, a humanized Fab. All other animals in the single- and repeat-dose studies survived until scheduled terminal and recovery necropsies.
Ophthalmic Examinations
In the single-dose and 8-week repeat-dose studies, trace to mild ocular inflammation was often observed following ITV administration of vehicle or FHTR2163 to cynomolgus monkeys as either a single 50 µL injection or two 50 µL injection and was characterized by identification of trace to mild aqueous cells on slit-lamp examination. Since this minimal-to-mild ocular inflammation also occurred in vehicle animals, this was considered procedure related. In the 26-week study, more significant ocular inflammation was observed in FHTR2163-administered eyes, characterized by identification of severe aqueous or vitreous cell infiltrates on slit-lamp examination, with perivascular cuffing of retinal vessels observed on indirect ophthalmoscopy fundus examination. Significant ocular inflammation was first observed on OE following the third ITV dose (day 31) and was observed in up to 25% of animals at some point in the study (Figure 1). The animal that was terminated early on day 46 developed severe ocular inflammation (4+) on beginning on day 31 with perivascular cuffing noted on day 37. Overall, the severity of the ocular inflammation was not dose-dependent and decreased in severity during the recovery phase and correlated with the presence of ADAs.
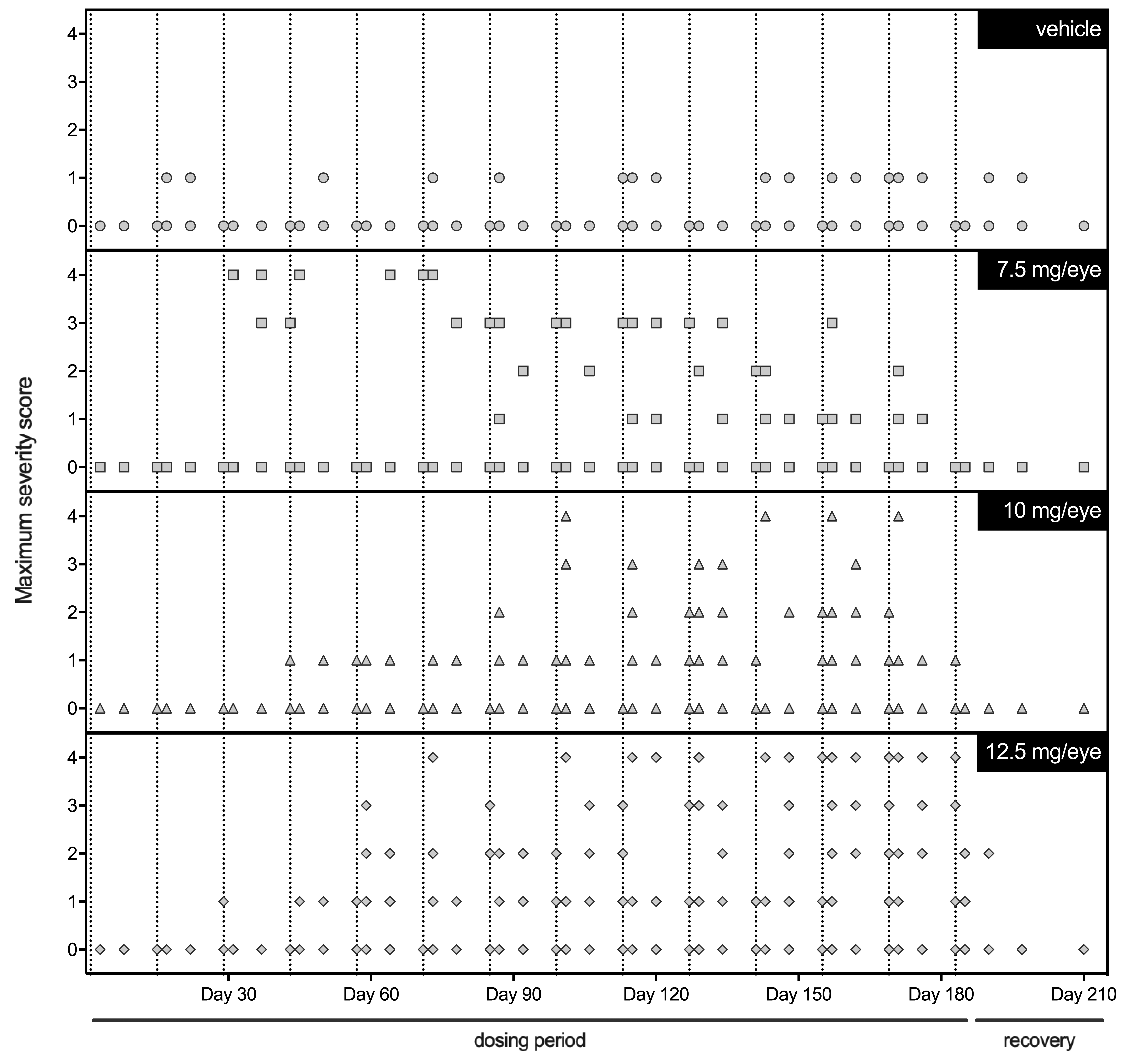
Ophthalmic examination (OE) results from cynomolgus monkeys of intravitreal-administered vehicle or FTHR2163 every 14 days for 26 weeks. Each symbol denotes an individual eye and corresponding score from the OE finding with the greatest severity in inflammation (eg, aqueous and vitreous cell). Vertical dashed lines represent days of FTHR2163 ITV administration.
Spectral Domain Optical Coherence Tomography
Hyper-reflective bands
The SD-OCT imaging identified HRBs in the fovea of some eyes in all the conducted studies (see Table 3 for the HRB incidence summary across all the conducted studies). HRBs were present in eyes from both vehicle and FHTR2163 treatment groups. In all treatment groups, the HRBs were located interior to the RPE and exterior to the inner segment/outer segment (IS/OS) and generally localized to the photoreceptor outer segment layer (see Figure 2A for a representative example of HRBs). The similarity in locations indicates that the HRBs are not FHTR2163 related. The HRBs were transient and not correlated with functional (ERG) or histopathologic abnormalities. The incidence of HRBs in vehicle-administered eyes was equal to or greater than FTHR2163-administered eyes and resolved by the end of the recovery phase.
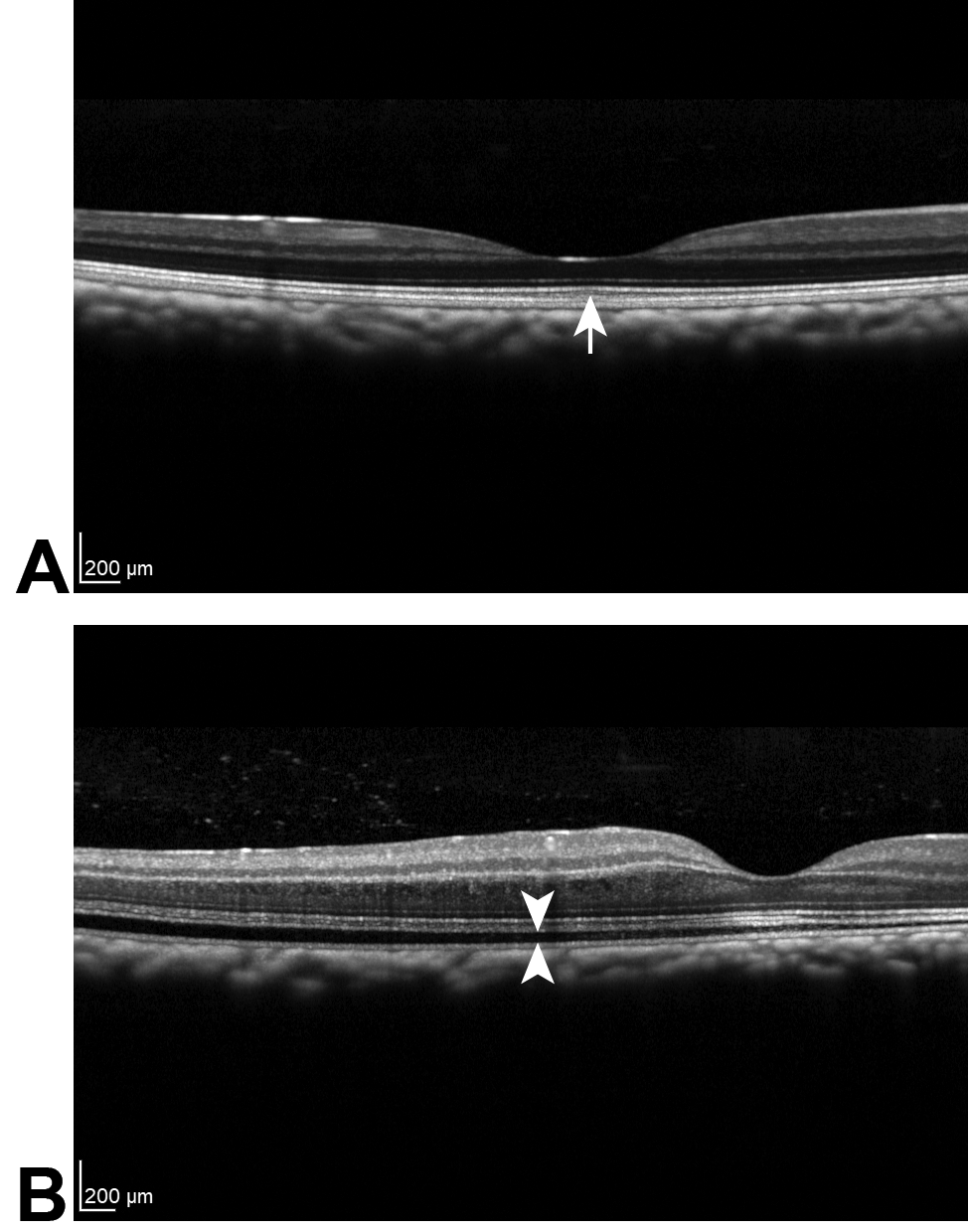
Example of spectral domain optical coherence tomography images showing a hyper-reflective band (A; denoted by the arrow) and retinal detachment (B; denoted by the space between the 2 arrowheads) in the photoreceptor outer segment layer.
Summary of the Incidence of HRBs Following Intravitreal Administration of FHTR2163 in Monkeys.
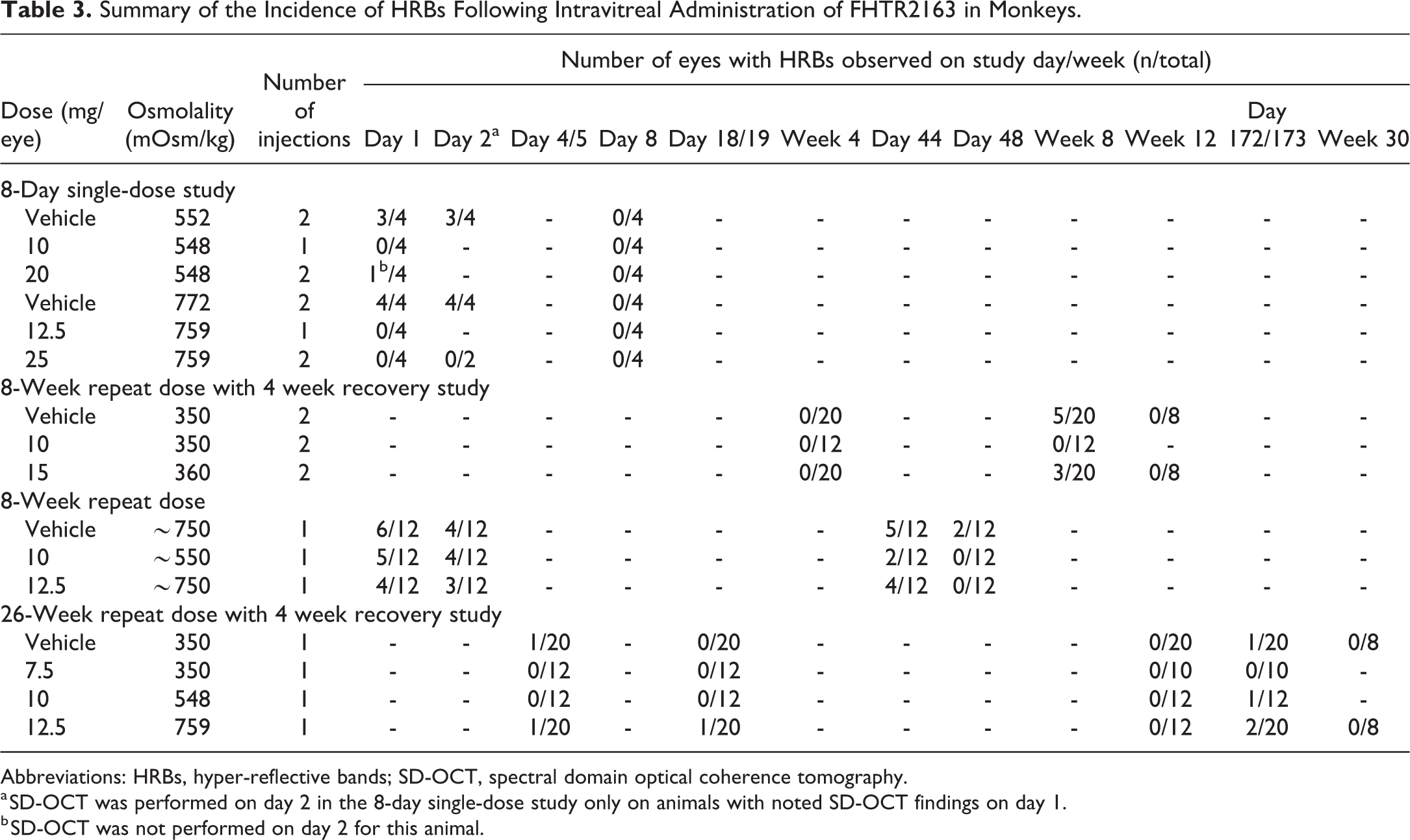
Abbreviations: HRBs, hyper-reflective bands; SD-OCT, spectral domain optical coherence tomography.
a SD-OCT was performed on day 2 in the 8-day single-dose study only on animals with noted SD-OCT findings on day 1.
b SD-OCT was not performed on day 2 for this animal.
Retinal detachment
Retinal detachments were noted on OCT examination in a few eyes of all the conducted studies (see Table 4 for the retinal detachment incidence summary across all the conducted studies), occurred acutely after ITV injection of both vehicle and FTHR2163, and resolved the following day. Detachments occurred predominantly in eyes administered vehicle or FTHR2163 in high-osmolality formulations (≥552 mOsm/kg; see Figure 2B for a representative example of retinal detachment). The frequency of retinal detachments was greater in eyes administered two 50 µL ITV injections of vehicle. Additionally, vehicle-administered eyes displayed evidence of greater significant retinal detachment, characterized by more extensive defects with a greater amount of fluid in the subretinal space than those in FHTR2163-injected eyes. In the single-dose study, effects were noted predominately in the macula of both the eyes of animals administered vehicle at 552 mOsm/kg. Effects noted outside the macula were noted in animals administered 772 mOsm/kg vehicle and tended to be in the temporal periphery in the right eyes and inferiorly in the left eyes. Although transient, the retinal detachments were considered adverse, but related to the ITV injection procedure and/or the high osmolality of the formulation, and were not directly due to the effects of FHTR2163.
Summary of the Incidence of Retinal Detachment Following Intravitreal Administration of FHTR2163 in Monkeys.
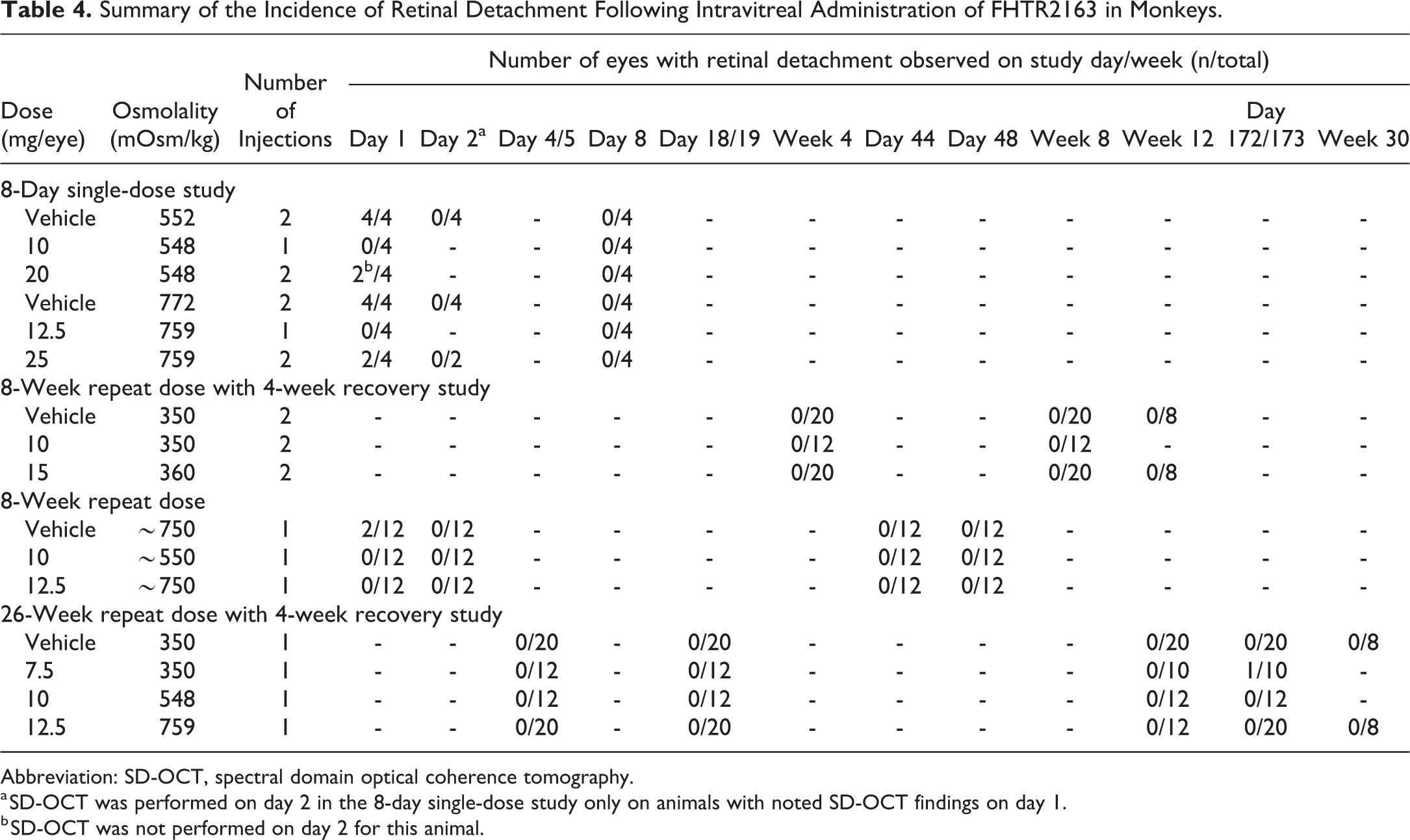
Abbreviation: SD-OCT, spectral domain optical coherence tomography.
a SD-OCT was performed on day 2 in the 8-day single-dose study only on animals with noted SD-OCT findings on day 1.
b SD-OCT was not performed on day 2 for this animal.
Clinical Pathology
Clinical pathology was evaluated in the 8- and 26-week repeat-dose GLP toxicity studies, with changes in evaluated end points limited to females in one of the 8-week studies. Minimal increases in total leukocyte (+28%;
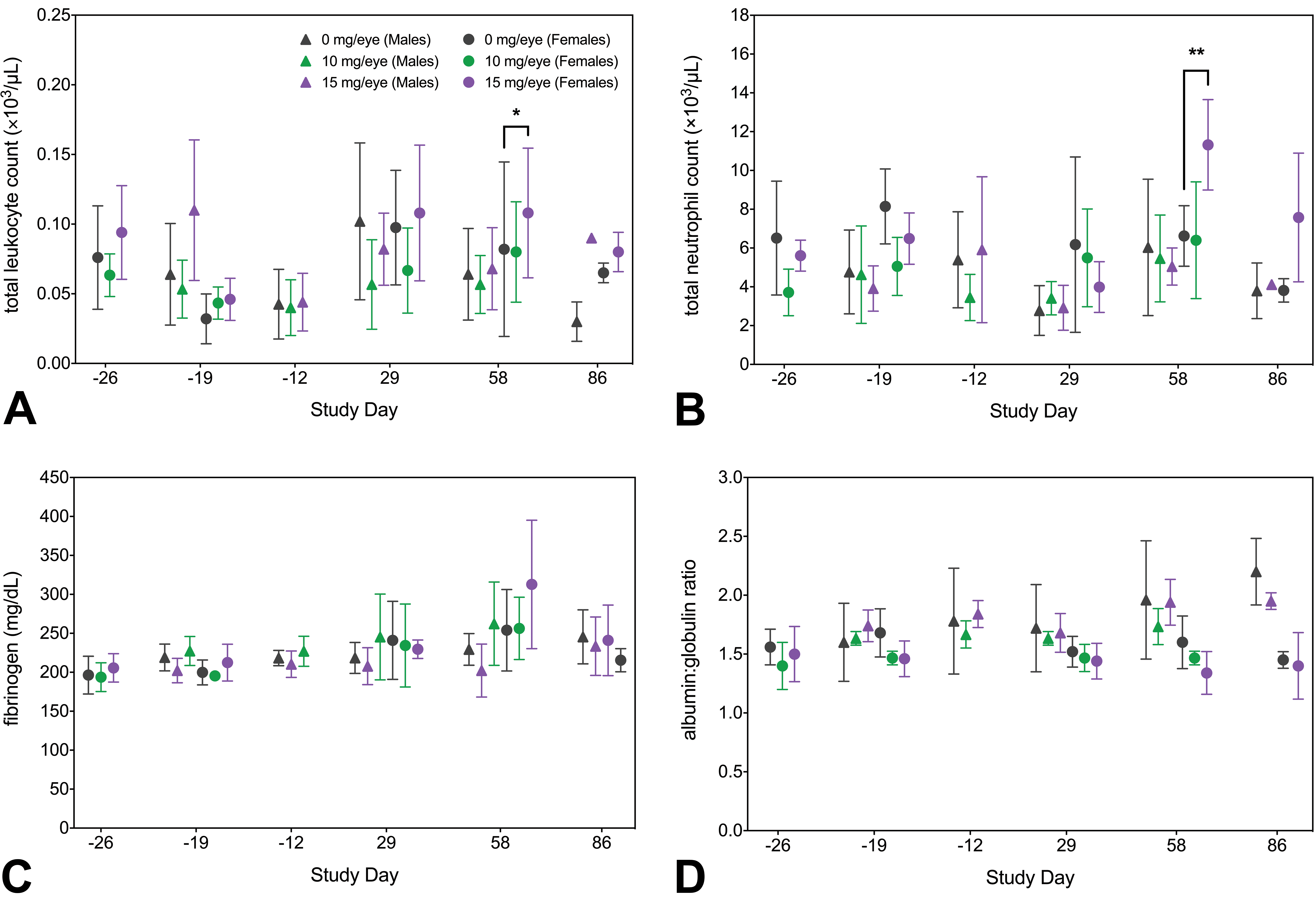
Clinical pathology data from the 8-week study with 4-week recovery period. Female animals administered 15 mg/eye FHTR2163 had minimally increased total leukocyte (+28%; A) and neutrophil (+23%; B) counts, minimally increased fibrinogen concentration (+23%; C), and minimally decreased albumin:globulin ratio (−19%; D) on study day 58. Statistical significance between treatment and vehicle controls of the same sex was calculated using 1-way analysis of variance with Dunnett correction for multiple testing (*
Anatomic Pathology
Macroscopic findings were not observed in any study. Microscopic findings in all conducted studies were limited to the eye. Severe ocular inflammation was identified on OE in 1 animal, administered 7.5 mg/eye FHTR2163, and resulted in early euthanasia on day 46 of the 26-week study.
Histologic findings did not exhibit dose dependency in incidence or severity and were limited to ocular inflammation and an increased incidence and severity of inflammatory cell infiltrates in the GLP repeat-dose studies. Inflammation was minimal to moderate, perivascular to vascular, and predominantly mononuclear. Retinal vessels were cuffed by variably dense infiltrates of lymphocytes and plasma cells, with occasional macrophages and rare neutrophils. More severe cases of ocular inflammation were observed only in the 26-week study. In these cases, inflammatory cells infiltrated, expanded and effaced the vessel walls, and extended into the adjacent retina, where they disrupted the ganglion cell and inner nuclear layers. Similar perivascular and vascular inflammation was often observed in the region of the optic disc. In some animals, inflammatory cells were also present in the vitreous. There was an increase in incidence and/or severity of minimal-to-moderate mononuclear cell infiltrates in the choroid, ciliary body, iris, and limbus/episclera in animals treated with FHTR2163; however, again, this lacked a relationship to dose. Ocular histopathology from the animal with the greatest severity of ocular inflammation that was euthanized on day 46 is shown in Figure 4. Recovery group animals administered 12.5 mg/eye with severe ocular inflammation resolved by the end of the 4-week recovery period. All animals with histologic evidence of ocular inflammation exhibited systemic ADAs to FHTR2163.
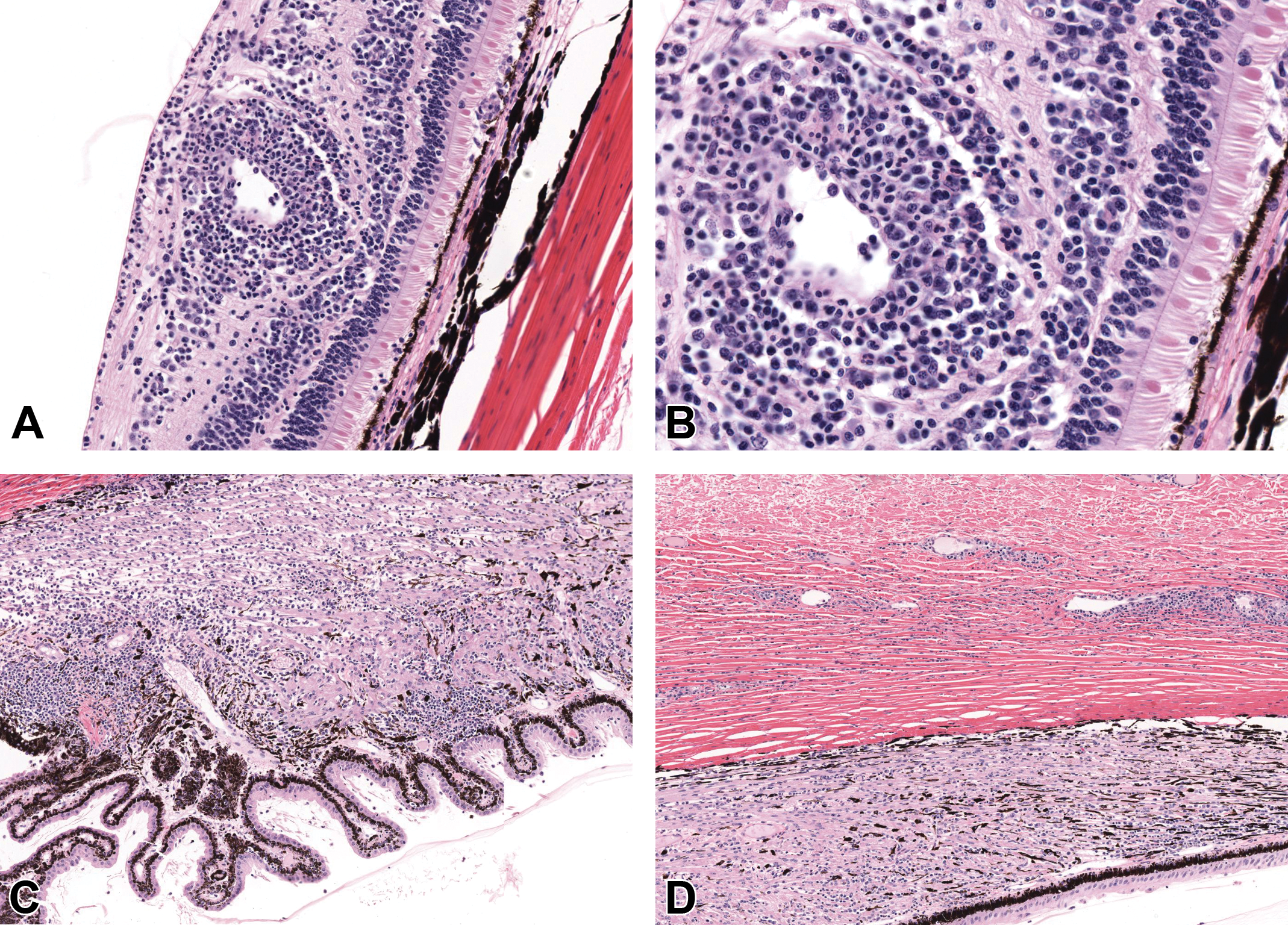
Ocular histopathology from an animal administered 7.5 mg/eye exhibiting the greatest severity of ocular inflammation. Ocular inflammation when observed was consistent with inflammation secondary to immunogenicity of the FTHR2163, a humanized Fab, in nonhuman primates. Diffusely within the retina, vessels were infiltrated and surrounded by prominent cuffs of inflammatory cells, composed predominantly of large numbers of lymphocytes and plasma cells (A, B). Diffusely the ciliary body was expanded by increased numbers of lymphocytes and plasma cells (C). Vessels within the sclera at the level of the pars plana were surrounded by small cuffs of lymphocytes and plasma cells (D). Hematoxylin and eosin: (A, C, D) ×50 magnification; (B) ×200 magnification.
Histologic evidence of vascular and perivascular inflammation in the retina and optic disc, particularly in more severe cases, was often correlated with the fundoscopic observation of perivascular sheathing on OE.
FHTR2163-related ocular inflammation and inflammatory cell infiltrates were consistent with an immune response to FHTR2163, rather than direct effects of FHTR2163, due to their character and localization, their correlation with the presence of systemic ADAs, and because their incidence and/or severity generally lacked a relationship to dose. No histologic evidence of inflammation was observed in the 8-day single-dose study.
Discussion
FHTR2163 is a Fab from a humanized IgG1 monoclonal antibody that selectively blocks the proteolytic activity of HTRA1. The safety profile of FHTR2163 was assessed in a single and multiple repeat-dose (up to 26 weeks in duration) ITV-administered toxicology studies with cynomolgus monkeys to support ITV clinical development in patients with GA.
Selecting the appropriate and relevant animal species for nonclinical toxicology studies is a critical consideration for all toxicology programs. The most important elements in species selection of ITV-administered therapeutics are the relevance for human risk assessment and the feasibility of conducting an ITV study. As such, species selection for ITV studies should be guided by pharmacological, physiological, and anatomical relevance to the human eye. 35 The ICH S6 guideline states that a rodent and non-rodent species should be used in general toxicology studies if both are pharmacologically relevant but should also be evaluated on a case-by-case basis. 25 FHTR2163 cross reacts with mouse Htra1 and is also pharmacologically active in the mouse. However, rodent (mouse and rat) eyes are generally inappropriate for studies requiring repeated ITV administration because of their small vitreal volume and the proportionally larger size of the lens, which result in difficulties administering sufficient levels of test article to obtain adequate exposures and a greater potential for iatrogenic trauma following repeated ITV injections. The cynomolgus monkey and rabbit are the most commonly species selected for the nonclinical development of ITV therapeutics, as the eye size and vitreal volume in these species are adequate for repeat ITV injection. Due to the extensive historical use of the rabbit for topical ocular studies with small molecule therapeutics, rabbits are often used as a lower order large animal species in lieu of rodents. However, rabbits should be used in the development of ITV therapeutics with caution—rabbits rapidly develop ADAs and severe ocular inflammation following ITV administration of biologics that often require early study termination and can significantly impact the ability to observe and interpret true, toxicologically relevant findings. 28 Furthermore, ICH S6 states that for the development of biologics, studies in 2 non-rodent species are not appropriate. Therefore, cynomolgus monkeys were selected as the only relevant species for nonclinical toxicity testing of FHTR2163.
Even in nonhuman primates, the nonclinical safety assessment of repeat administration of humanized biologics is often complicated by the onset of ADA-related ocular inflammation. 29,36 -41 Chronic ITV administration of FHTR2163 resulted in significant ocular inflammation in up to 25% of the animals on study. It was not dose dependent, decreased in severity during the recovery phase, and was consistent with the histologic character, location, and time course of ocular inflammation commonly observed with ITV repeat administration of biologics. The severity of the inflammation in the 26-week study required veterinary intervention in some animals. An animal administered 7.5 mg/eye developed severe ocular inflammation beginning on day 31 (following the third dose) and was euthanized on day 46 (following the fourth dose) for welfare reasons. In addition, ocular inflammation resulted in a pause in dosing of 2 animals, 1 animal from each 7.5 and 12.5 mg/eye dose groups. Although dosing in these 2 animals was able to resume once the inflammation subsided, it limited the number of FTHR2163 doses that these animals were able to receive. The overall incidence of ADAs against FHTR2163 in the GLP toxicity studies was low in vehicle-treated animals, with only 2 of 26 animals positive for ADAs. These ADA-positive animals had low ADA titers and exhibited minimal ocular inflammation. Conversely, the majority of animals administered FHTR2163 (45/50) were ADA positive, with higher titers relative to ADA-positive vehicle-treated animals. In general, the noted ocular inflammation in GLP toxicity studies correlated with the ADA status of the animals. Based on the clinical experience of Food and Drug Administration–approved ITV biologics, there is little translatability of ADA-related ocular inflammation from nonclinical toxicology studies to patients. 39 -47 Immunogenicity in nonclinical species is not predictive of clinical immunogenicity and as such is not used in the derivation of the NOAEL.
On SD-OCT imaging, a focal HRB was noted in the fovea of some eyes that were dosed with vehicle and FTHR2163 in all studies. The presence of HRBs was not associated with functional impairment (as assessed by ERG), was not correlated with histopathologic abnormalities, and was not considered adverse. They were not observed by the end of the recovery period. Furthermore, the incidence of HRBs in vehicle-administered eyes was equal to or greater than FTHR2163-administered eyes, suggesting that HRBs are procedure related and not directly due to FTHR2163. Although the etiology of HRBs is not well understood, they may be related to the disruption in the disk stacks of the OSs that occurs during ITV injections, and prior experience has demonstrated that HRBs are more frequently observed following 100 µL (via two 50 µL injections ∼10 minutes apart) ITV injection or trocar insertion. 48
Adverse retinal detachments were also observed by SD-OCT imaging, and similarly to HRBs, the incidence of detachments was greater in vehicle-administered eyes. Furthermore, the retinal detachments were transient and rapidly resolved within 24 hours. Eyes receiving 2 ITV injections of high-osmolality (552 or 772 mOsm/kg) vehicle in the single-dose study developed more pronounced retinal detachments that covered a greater area of the retina compared to those in FHTR2163-treated eyes, suggesting that the detachments are related to the ITV injection procedure and/or the high osmolality of the FTHR2163 formulation.
Local ITV route of administration maximizes drug exposure to the target ocular tissue and minimizes systemic exposure. However, because of the relatively small size of the cynomolgus monkey relative to the vitreal volume when compared with humans, bilateral dosing of biologics in cynomolgus monkey usually enables establishment of adequate systemic safety margins, without the need for a separate systemic study. Assessment of systemic toxicity in these studies was based on mortality, clinical signs, qualitative food consumption, body weight and body weight change, physical examinations, physiologic indices (blood pressure, heart rate, respiration rate, and body temperature), and clinical and anatomic pathology. Safety pharmacology parameters (neurological examinations and 8-lead ECG) were included in the repeat-dose studies, and stage-aware microscopic evaluation of spermatogenesis in the testes was included in the 26-week study. The systemic effects of ITV-administered FHTR2163 were limited to minimal changes in select clinical chemistry parameters in only a single study consistent with a mild inflammatory response and resolved by the end of the recovery period. Bilateral ITV administration of 12.5 mg/eye FTHR2163 (NOAEL) for 26 weeks resulted in low systemic exposures and provided
In summary, a comprehensive toxicology program, employing bilateral ITV dosing in a single nonclinical species (the cynomolgus monkey), provided a suitable characterization of the safety profile of FTHR2163 for the transition of the molecule into clinical development in patients with GA and continues to support the clinical development plan in phase II.
Footnotes
Acknowledgments
The authors thank the valuable support of Menno van Lookeren Campagne, Kelly Loyet, Yan Wu, Erich Strauss, and Roxanne Andaya (Genentech); Ewa Budzynski, Alok Sharma, Steven Sorden, and Tim Streit (Covance); Cindy Farman (Farman Pathology); and Carol Rasmussen, Jim VerHoeve, and T. Michael Nork (OSOD).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
