Abstract
AZD7969 is a potent inhibitor of glycogen synthase kinase 3 (GSK3β), which is a multifunctional serine/threonine kinase that negatively regulates the Wnt/β-catenin signaling pathway. Treatment of rats and dogs with AZD7969 for periods of up to 4 weeks resulted in a number of changes, the most significant of which was a dose-dependent, and treatment-related, increase in proliferation in a number of tissues that was thought to arise from derepression of Wnt/β-catenin signaling in the stem cell compartment. Phenotypically, this resulted in hyperplasia that either maintained normal tissue architecture in the gastrointestinal tract, liver, kidney, and adrenals or effaced normal tissue architecture within the bones, incisor teeth, and femorotibial joint. In addition to these changes, we noted a treatment-related increase in iron loading in the liver and proximal small intestines. This off-target effect was robust, potent, and occurred in both dogs and rats suggesting that AZD7969 might be a useful tool compound to study iron storage disorders in the laboratory.
Introduction
GSK3 is a widespread multifunctional serine/threonine kinase that exists in 2 isoforms, α and β (Stambolic and Woodgett 1994), with 98% sequence homology (Kannoji et al. 2008). It was initially identified in rabbit skeletal muscle as an enzyme that phosphorylated and inactivated its target substrate, glycogen synthase (Embi, Rylatt, and Cohen 1980; Frame, Cohen, and Biondi 2001), but it is now known to also regulate a number of other cellular processes such as tau protein (Eldar-Finkelman 2002) and the Wnt/β-catenin pathway (Jope and Johnson 2004).
GSK3β is an unusual serine/threonine kinase in that it is active under resting conditions and inactivated upon cell stimulation. It exists as a multimeric complex of adenomatosis polyposis coli (APC), axin, and casein kinase 1 (CK1)—the “destruction complex”—where one of its primary functions under resting conditions is to phosphorylate β-catenin (CTNNB1 catenin [cadherin-associated protein] β 1), leading to its ubiquitination and proteasomal degradation (Rubinfeld et al. 1996; Chen, Ding, and McCormick 2000). Upon cell stimulation, Wnt glycoprotein ligand activates the canonical β-catenin pathway by signaling through Frizzled and Low-density lipoprotein receptor–related protein 5 (LRP5) and 6 (Cadigan and Liu 2006), which inhibits the destruction complex allowing β-catenin to stabilize in the cytoplasm and translocate to the nucleus. Accumulation of β-catenin in the nucleus leads to binding to T cell factor/lymphocyte enhancing factor (Tcf/Lef) and transactivation of target genes such as c-Myc (He et al. 1998), cyclin D1 (Tetsu and McCormick 1999), and PPARdelta (He et al. 1999).
The role of GSK3β in tumorigenesis is complex and often paradoxical. It was initially thought to act as a tumor suppressor in an analogous manner to that of the “gatekeeper” gene APC where inactivation of APC (present in 80% of sporadic cases) through mutation ultimately results in colorectal cancer (Kinzler and Vogelstein 1996; Wang et al. 2011). To this effect, loss of tumor suppressor function through inactivation of GSK3β has been found in 30% of human cancers (Kang et al. 2008), particularly cancers of epithelial origin, such as skin, breast, and oral cancers (Mishra 2010). However, it is now becoming clear that GSK3β may also act as an oncogene in other types of cancer such as bone (Tang et al. 2012), liver, stomach, colorectal, and kidney tumors (Mishra 2010). The mechanism of this context-dependent role is still poorly understood, but it is thought that in some cell types, GSK3β acts as a positive regulator of NF-κB to promote cell proliferation and survival (Hoeflich et al. 2000; Tang et al. 2012).
In addition to its role in cancer, the Wnt/β-catenin pathway regulates stem cell fate determination in both development and the adult animal. In the adult, Wnt/β-catenin signaling serves to regulate adult tissue renewal in stem cell niches such as the intestinal crypt, hair follicle, and bone marrow (Clevers 2006). Experimental modulation of the Wnt/β-catenin signaling axis in these compartments leads to profound changes in tissue development and renewal such as intestinal crypt loss (Kuhnert et al. 2004), hair loss (Huelsken et al. 2001), and changes in bone mass (Kato et al. 2002). In the nervous system, GSK3 inhibition increases Notch intracellular domain and Gli proteins (Kim and Snider 2011) and has been shown to promote proliferation of neural progenitor cells in the presence of bFGF and EGF (Esfandiari et al. 2012). It is not surprising therefore that GSK3β inhibitors have attracted considerable attention. This has not gone unnoticed by the pharmaceutical industry, as they potentially offer a novel target to treat a number of diseases such as type 2 diabetes, obesity, Alzheimer’s disease, mood disorders, osteoporosis, atherosclerosis, and certain types of cancer (Kannoji et al. 2008). We therefore generated AZD7969 (AR79), a novel small molecule inhibitor of GSK3β which inhibited GSKα/β enzymes with equivalent potency (IC50 3–5 nM), increased β-catenin stabilization, and stimulated bone mineralization in vitro (Gilmour et al. 2013). Daily oral administration of AZD7969 increased bone mass following 1 to 2 weeks of dosing in rats (Gilmour et al. 2013). These pharmacological responses were commensurate with modulation of GSK3β and similar to effects seen with other small molecule GSK3 inhibitors, for example, AZD2858 (Marsell et al. 2012), lithium chloride (Clement-Lacroix et al. 2005), and LY603281-31-8 (Kulkarni et al. 2006). Additional pharmacological screening for off-target effects with AZD7969 has shown inhibition at several different kinases (Table 1) with equipotent inhibition of HIPK2, activity at PKD1, PIM-3, and DYRK-1A. In a range of G-protein-coupled receptor and enzyme assays, AZD7969 was >40× selective for 5-HT1A/B (Table 1).
Potency determination for AZD7969 in kinase, GPCR, and enzyme assays.
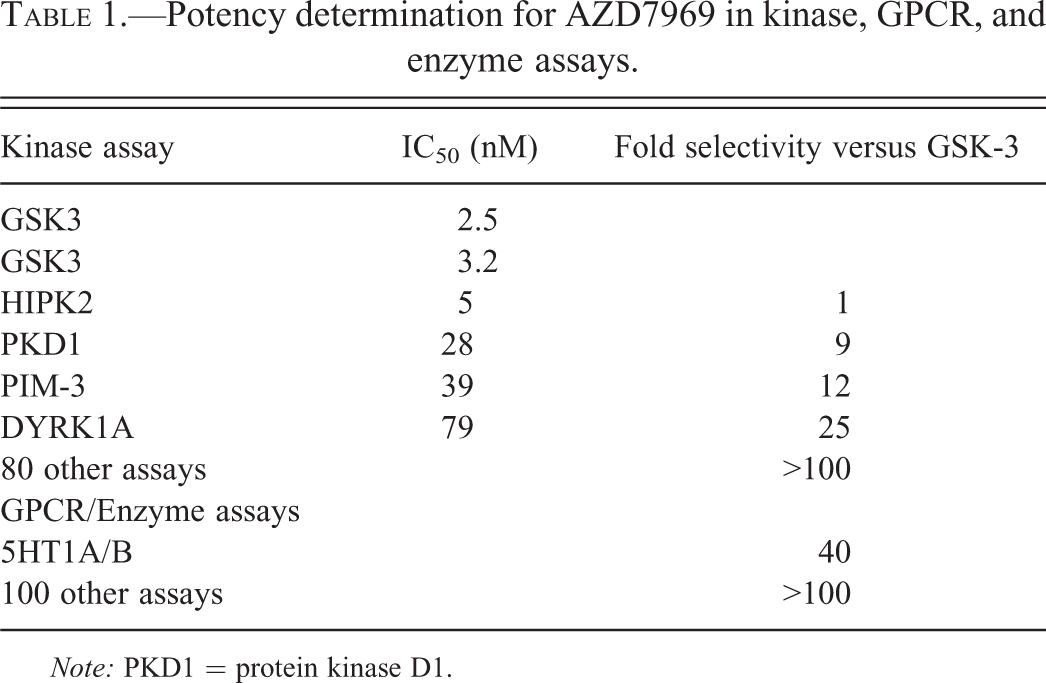
Note: PKD1 = protein kinase D1.
In this report, we describe the finding resulting from daily oral administration of AZD7969 to rats and dogs in 3-week and 1-month pivotal safety studies. We discuss the range of changes induced by treatment with AZD7969 and describe how they might be reconciled with the known functions of GSK3β inhibition within the stem cell compartment. We also describe an unexpected off-target effect of AZD7969 treatment that resulted in iron accumulation within the liver and proximal small intestine that may act as a useful model to investigate other types of iron storage disease.
Materials and Methods
Test Material
For the in vivo studies, test compound was supplied by the formulation and analytical support group, AstraZeneca, Alderley Park. For the 3-week studies, AZD7969 was formulated as a solution in 0.5% hydroxypropyl methylcellulose/0.1% polysorbate 80/phosphate-buffered saline. For the 4-week studies, AZD7969 was formulated as a solution in 0.5% w/v HPMC and 0.1% w/v polysorbate 80 in 0.01 M phosphate buffer. For all studies, compound or vehicle was administered once daily by oral gavage.
Animals and Husbandry
All the in vivo studies were conducted in accordance with the standards of animal care and ethics described in “Guidance on the Operation of the Animals (Scientific Procedures) Act 1986 (HC321)” issued by the U.K. Home Office. They were conducted so that any clinical expression of toxicity remained within a moderate severity limit as described in guidelines agreed with the U.K. Home Office inspector.
Male and female Wistar Hannover rats (HsdHan:WIST) aged 10 weeks were obtained from Harlan Laboratories UK Ltd (Blackthorn, Bicester, Oxfordshire, OX25 1TP) and were group housed (3–5 per cage) in transparent solid-bottom plastic cages and acclimatized for at least 2 weeks before the first dose. The animals had free access to pelleted RM1 (E) special quality service (SQC) diet and to water from the site drinking water supply.
Beagle dogs, between 12.5 and 15.5 months of age, were obtained from the Dog Breeding Unit, AstraZeneca, Alderley Park. They were single penned and acclimatized for at least 4 weeks before the first dose. Water from the site drinking water supply was freely available and a set quantity of a dry pelleted dog diet (Dog-D3 [E] SQC) was provided daily in the afternoon, access being allowed for a minimum of 2 hr. The dogs were not fed on the day of scheduled necropsy.
Temperature and relative humidity ranges were maintained at 19°C to 23°C and 40% to 70%, respectively, for the rats, whereas for the dogs, temperature was set at 21°C and was controlled within limits of 15°C and 24°C. The animal rooms and exercise areas were illuminated by artificial light from fluorescent tubes on a 12 hr light–dark cycle.
Study Design
Rat 3-week dose range finding (DRF) study
This study comprised a 5-day repeat phase study (data not shown) to assess tolerability and determine the dose levels for the 21-day repeat dose study. For the 3-week repeat dose phase, 4 groups of Wistar Hannover rats (HsdHan:WIST), each consisting of 3 males and 3 females, were treated with AZD7969 at 0, 10, 30, and 70 mg/kg/day. At 70 mg/kg/day, 3 animals (F22, M36, and M35) were euthanized for welfare reasons on days 12, 14, and 15, respectively, due to weight loss. All remaining animals dosed at 70 mg/kg/day were terminated on day 15 (main study animals) or on day 16 (satellite animals) after their last toxicokinetic bleed. As a consequence of findings at 70 mg/kg/day, an additional group of animals was included in this study at a reduced dose of 50 mg/kg/day for 21 days.
Rat 1-month study
Four groups of Wistar Hannover rats (HsdHan:WIST), each consisting of 10 males and 10 females, were treated with AZD7969 at 0, 10, 30, and 50 mg/kg/day for 28 or 29 days (with the exception of 3 males at the high dose which were euthanized early for welfare reasons after either 22 days [M62] or 26 days [M61 and M63] of dosing) due to weight loss. Additional groups (5 males and 5 females) were included to assess recovery after a treatment-free period of 4 weeks. Two recovery females (F96 and F97) were euthanized early for welfare reasons during the recovery period (day 33) due to weight loss.
Dog 3-week study
This study comprised an initial maximum tolerated dose (MTD) phase (data not shown) that was used to establish tolerability and dose levels for the main repeat phase 21-day study. For the 3-week repeat dose phase, 2 groups of Beagle dogs, each consisting of one male and one female were treated with AZD7969 at 5 and 10 mg/kg/day.
Dog 1-month study
Four groups of Beagle dogs, each consisting of 3 males and 3 females, were treated with AZD7969 at 0, 1, 3, and 10 mg/kg/day for 28, 29, or 30 days. An additional high-dose group (3 males and 3 females) was included to assess recovery at 4 weeks after cessation of dosing.
For both dogs and rats, on the scheduled day of necropsy, animals were sacrificed and necropsied according to standard operating procedures. Body weight, organ weights, and macroscopic abnormalities were recorded. Tissues were fixed and processed to H&E slides and pathology data were recorded in Pathdata V6.2d. Group mean organ weight data where appropriate was analyzed using the PathData weighing program, which applied Dunnett’s test based on pooled variance and Dunn’s test. For further investigatory work, sections of liver from both the rat and dog studies were stained with Perls’ Prussian Blue. Additional sections of gastrointestinal (GI) tract and heart from the 1-month rat study and heart from the 1-month dog study were also stained with Perls’ Prussian Blue.
Clinical Signs and Measurements
Clinical signs were recorded once or twice daily, and a physical examination was performed at least once weekly for the 1-month studies. Food consumption was measured over weekly intervals for each cage of rats or daily for dogs. Body weight was recorded during the pretest period and either daily (21-day studies) or twice weekly (1-month studies) until necropsy. For the 1-month studies, an ophthalmoscopic examination was performed prestudy and toward the end of the repeat dose phase and recovery period. Blood samples for toxicokinetics, clinical hematology, and plasma chemistry were collected from the tail vein or terminally from the vena cava (rat studies) or the jugular vein or another peripheral vein (dog studies) both predose and at the end of the dosing period and recovery period. Urine samples were collected once pretest and toward the end of the repeated dose phase and recovery period. Toxicokinetic samples were taken prior to dosing and then at various time points within the first 24-hr postdose at each dose level on day 1 and on the final day of the study as well as 24 hr after the final dose. Finally, for the dog studies, an electrocardiogram was recorded twice prestudy and then prior to dosing and at approximately 1 hr and 3 hr after dosing on day 1 and toward the end of the dosing periods.
Results
Tolerability and Clinical Signs
Rat studies
In the repeat phase of the DRF study, there was no consistent gender difference in the exposure. At steady state, C max and AUC( 0-last) increased more than in proportion to dose in the dose range 10 to 50 mg/kg/day, but at 70 mg/kg/day on day 15, exposure decreased to approximately the exposure observed at 30 mg/kg/day. Mean t max increased with increasing dose, occurring at 3, 6, and 12 hr after administration at doses of 10, 30, and 50 mg/kg/day, respectively. Treatment with AZD7969 at 70 mg/kg/day was not tolerated, and all remaining animals were sacrificed early on day 15. Treatment at 50 mg/kg/day with AZD7969 resulted in a 32% to 51% decrease in body weight gain, which was accompanied by reduced food intake of up to 33% (Table 2). Termination of the high-dose group and the adverse effect on body weight gain/food consumption were considered to be due to the clinically observed sign of abnormal growth of the incisor teeth (which was confirmed to be dental dysplasia at histological examination). Other clinical signs consisted of increased salivation, ploughing (a sign of abdominal discomfort where the animal pushes through the bedding material head first), and alopecia (primarily ventral abdomen, dorsum, and head).
Body weight changes and food consumption.
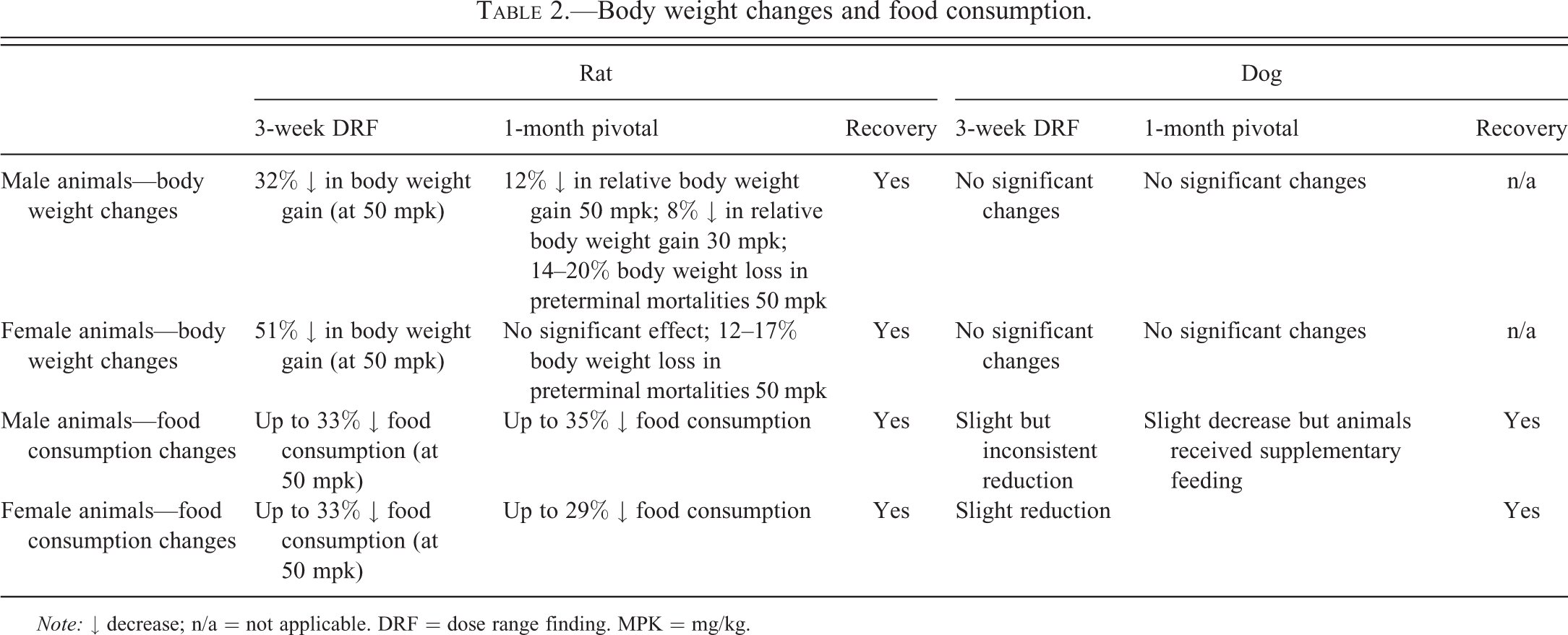
Note: ↓ decrease; n/a = not applicable. DRF = dose range finding. MPK = mg/kg.
In the 1-month study, there was no consistent gender difference in the exposure and, at steady state, exposure increased more than in proportion to dose. Treatment with AZD7969 resulted in a significant treatment-related decrease in food consumption and body weight in male animals dosed at 30 to 50 mg/kg/day (Table 2). This change had partially recovered by the end of the treatment-free period but was still significantly reduced compared to the concurrent control group. As a result of body weight loss and adverse clinical signs, 3 animals (M61, M62, and M63) treated with 50 mg/kg/day were sacrificed for welfare reasons (14–20% body weight loss) on days 21–26 of study. A further 2 female animals (F96 and F97) treated with 50 mg/kg/day were sacrificed for welfare reasons (12–17% body weight loss) on day 33 of study. The cause of morbidity for all animals was established as incisor tooth dental dysplasia, which was considered to be the dose limiting toxicity. In surviving animals, the most consistent clinical signs were increased salivation, patchy hair loss from around the muzzle, ploughing, and abnormal growth of incisor teeth and red discharge from the eyes. The observation of tooth abnormalities continued into the recovery period.
Dog studies
During the repeat dose phase of the 3-week DRF study, treatment with AZD7969 at 10 mg/kg/day was associated with vomiting once or twice a day. As a result of this, it was not possible to determine the relationship between dose and exposure. Clinical signs consisted of increased salivation in both the male and female dog together with subdued behavior and soft/fluid faeces in the female dog. No significant effects were noted in body weight.
In the 1-month study, at steady state, there was a more than dose proportional increase in exposure with dose. Treatment was well tolerated but induced clinical signs of vomiting, increased salivation, soft/fluid feces, ataxia, trembling, and a slight decrease in motor activity.
Clinical Pathology
Rat studies
In both the 3-week and 1-month study, a number of relatively minor changes were noted during clinical pathology assessment (summarized in Table 3). Hematological changes consisted of decreases in red cell indices (hemoglobin [Hgb], hematocrit [HCT], and red blood cells [RBC]), increases in reticulocytes and red cell distribution width (RDW) and increases in white blood cell (WBC) indices of up to 2-fold. This was accompanied by small changes in clinical chemistry, the most significant of which were slight to moderate increases in urea and creatinine, calcium, and phosphate. In the 1-month study only, minor changes were also seen in urine analysis in male and female animals treated with 50 mg/kg/day characterized by 2 to 2.5 fold increases in urinary N-acetyl-β-D-glucosaminidase (NAG)/creatinine and 3-fold increases in glucose/creatinine values. All the changes noted in the 1-month study showed complete recovery after the end of the 1-month treatment-free period except for the decreases in red cell parameters (Hb, HCT, and RBC), which showed partial recovery by the end of the treatment-free period (summarized in Table 3).
Summary of clinical pathology changes in 3-week and 1-month rat and dog studies.
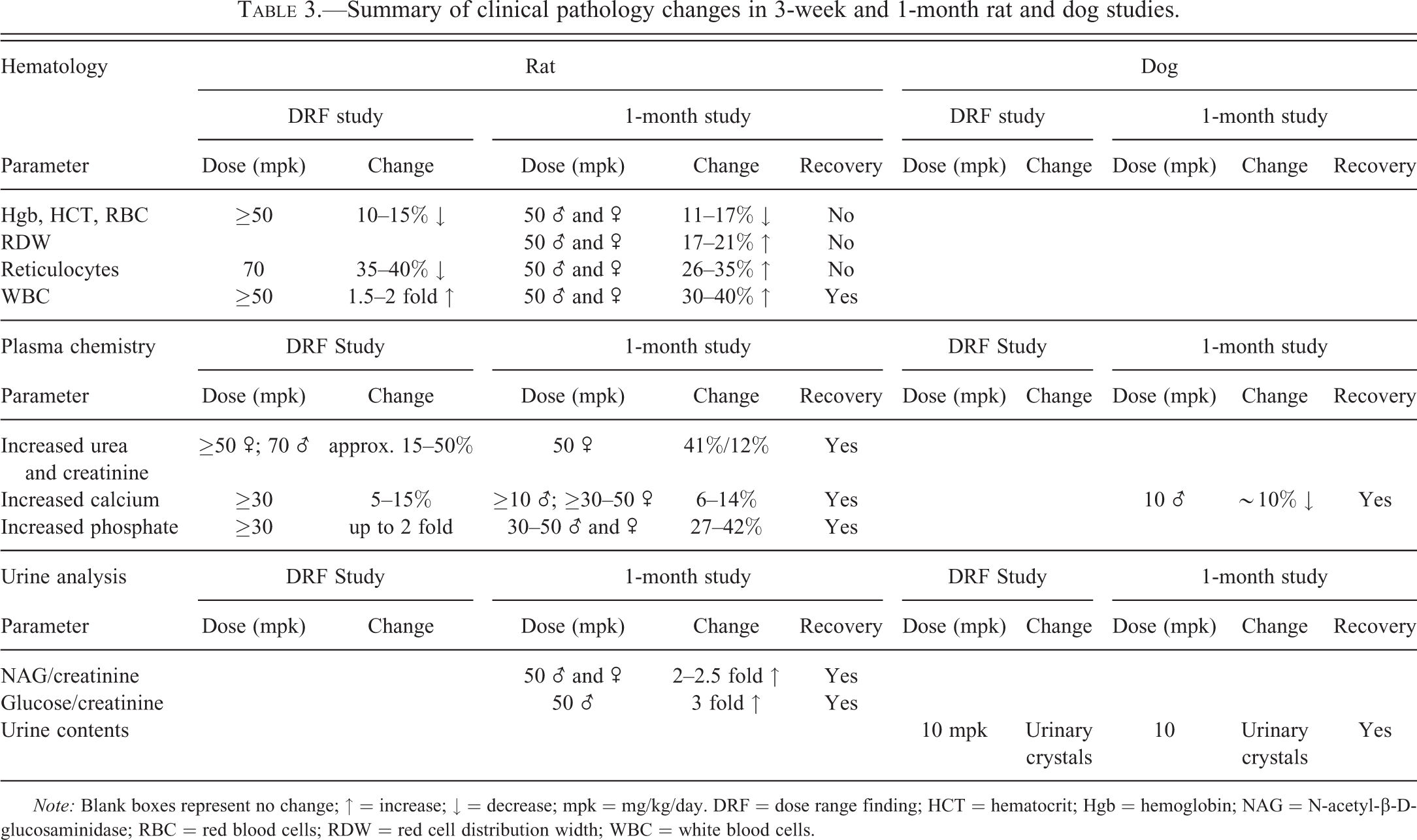


Note: Blank boxes represent no change; ↑ = increase; ↓ = decrease; mpk = mg/kg/day. DRF = dose range finding; HCT = hematocrit; Hgb = hemoglobin; NAG = N-acetyl-β-D-glucosaminidase; RBC = red blood cells; RDW = red cell distribution width; WBC = white blood cells.
Dog studies
No significant changes were noted in hematological assessment, and only minor changes were noted in all other clinical pathology assessments (summarized in Table 3), the most significant of which were mild increases in total calcium in male dogs noted in the 1-month study and the presence of urinary crystals in the bladder of dogs treated with 10 mg/kg/day in both the 3-week and 1-month studies. All changes noted in the 1-month study showed complete recovery by the end of the treatment-free period.
Necropsy Findings
Rat studies
In both the 3-week and 1-month study, treatment-related macroscopic findings were noted in the lower incisors (malformation/elongated) and small intestines (thickening). During the in-life phase of the study, alopecia or patchy hair loss of the skin was also noted. A number of other less significant changes were noted and are summarized in Table 4. All the changes noted in the 1-month study had recovered at the end of the treatment-free period, except incisor tooth malformation/elongation that persisted throughout the treatment-free period.
Macroscopic changes noted in 3-week and pivotal 1-month rat study.
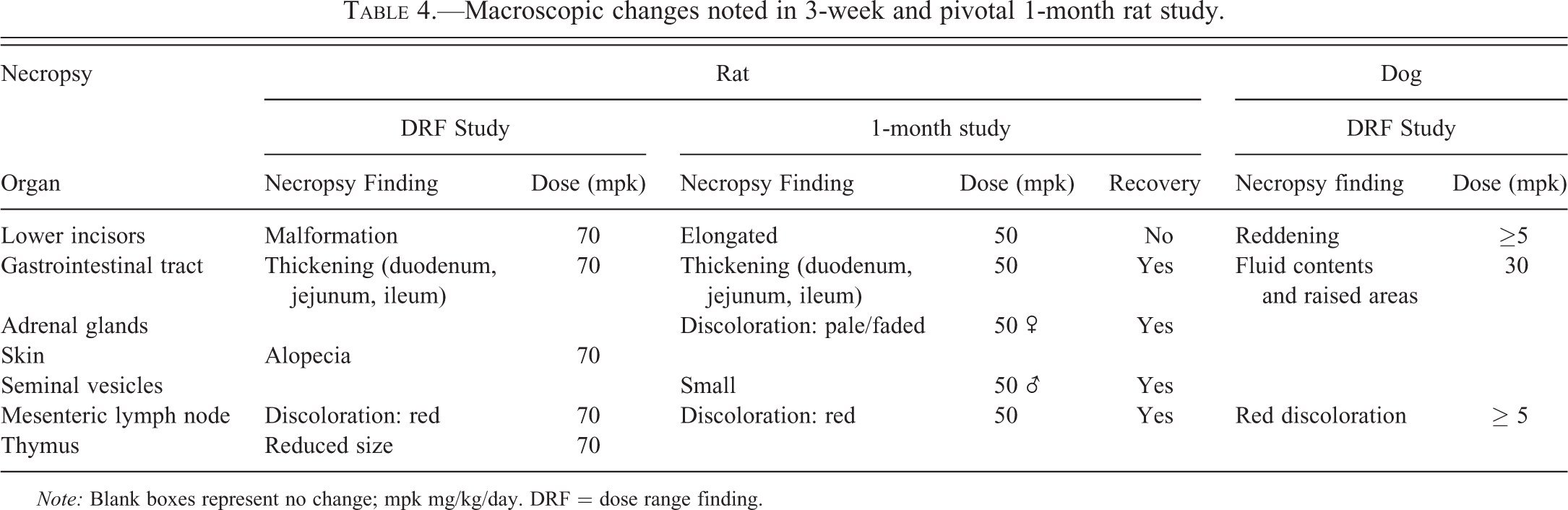
Note: Blank boxes represent no change; mpk mg/kg/day. DRF = dose range finding.
Dog studies
In the 3-week-DRF study, reddening and fluid contents with raised areas (likely correlates with inflammation) were noted in the gastrointestinal tract as well as red discoloration of the mesenteric lymph node. In the 1-month study, no treatment-related macroscopic changes were noted.
Organ Weights
Rat studies
Significant treatment-related increases in organ weights were noted in the liver, kidneys, adrenal glands, spleen, and heart. Significant decreases were noted in the prostate gland with a nonsignificant trend toward decreased weight in the thymus (Table 5). These changes tended to be dose-related and of greatest magnitude in animals treated with 50 mg/kg/day. The adrenal and prostate gland had fully recovered by the end of the treatment-free period. Partial recovery, indicated by a decrease in the magnitude of the organ weight change, was also noted in the liver, kidneys, spleen, and heart, whereas the thymus showed no evidence of recovery. In this organ, a larger (and statistically significant) decrease in organ weight was noted in male animals at the end of the treatment-free period compared to recovery control animals.
Summary of organ weight changes in 1-month rat and dog studies.

Note: Blank boxes represent no change; ↑ = increase; ↓ = decrease; mpk = mg/kg/day.
Dog studies
Due to the small group size, organ weights were not analyzed for statistical significance. However, treatment-related changes in absolute and relative (to body weight) organ weights that correlated with microscopic observations were noted in the liver (decreased), thymus (increased), and testes (decreased) in the 1-month study. By the end of the treatment-free period, the liver organ weight change had recovered whereas the testes and thymus were still altered compared to concurrent controls.
Major Histological Findings
A number of treatment and dose-related findings were noted in the dog and rat (summarized in Table 6). Several of these toxicities were common to the dog and rat and could be broadly characterized into proliferative changes (increased mitotic figures or hypercellularity), degenerative changes (single cell necrosis, hypocellularity, and atrophy), and miscellaneous changes (secondary to inanition, stress, or of unknown aetiology).
Summary of histopathology in 3-week and 1-month rat and dog studies.
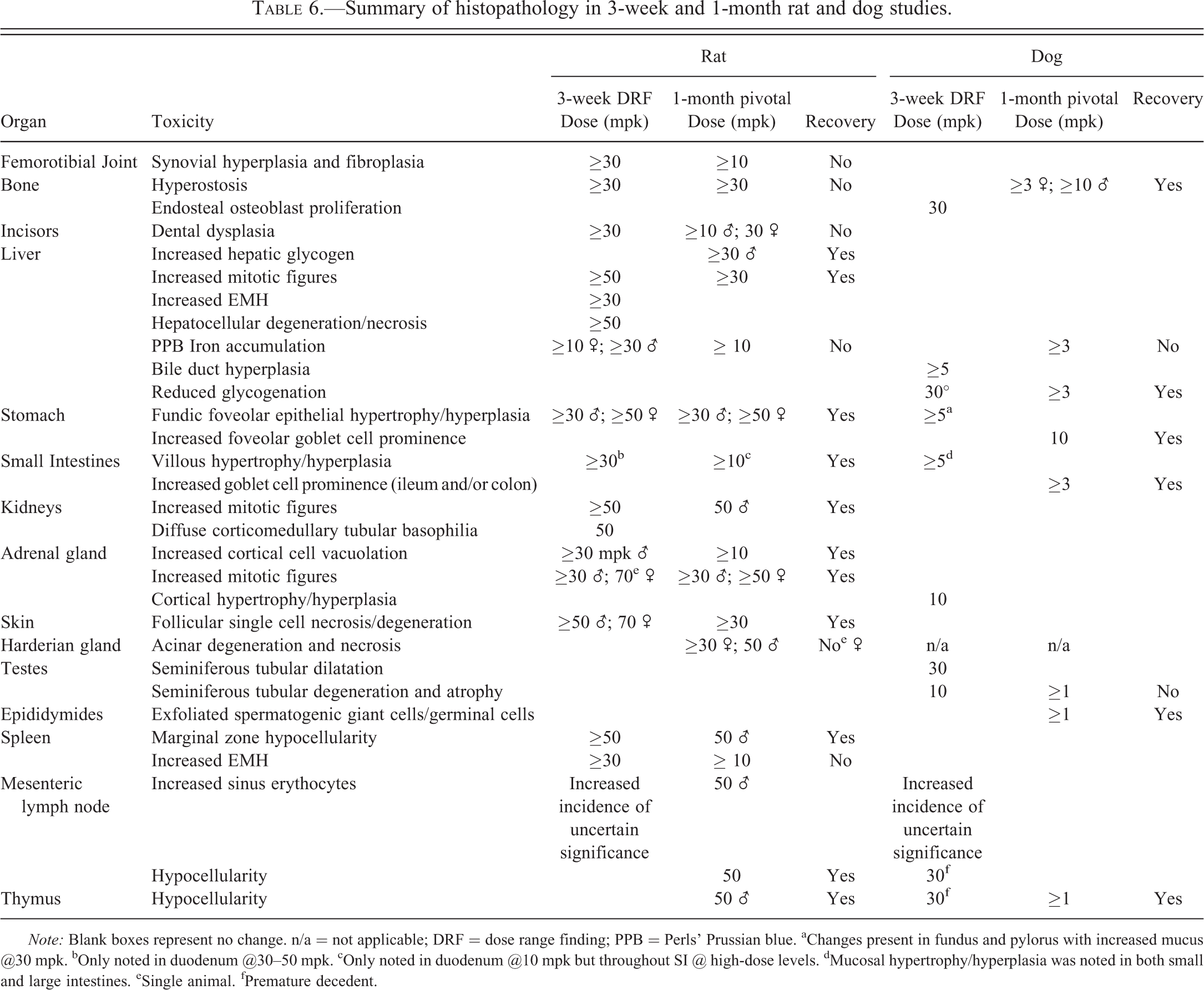
Note: Blank boxes represent no change. n/a = not applicable; DRF = dose range finding; PPB = Perls’ Prussian blue. aChanges present in fundus and pylorus with increased mucus @30 mpk. bOnly noted in duodenum @30–50 mpk. cOnly noted in duodenum @10 mpk but throughout SI @ high-dose levels. dMucosal hypertrophy/hyperplasia was noted in both small and large intestines. eSingle animal. fPremature decedent.
Proliferative Changes
Femorotibial joint, sternum, and incisor teeth
In the femorotibial joint, bones, and incisor teeth, treatment with AZD7969 resulted in a number of changes in both the rat and dog that were proliferative in nature. These changes had generally not fully reversed by the end of the treatment-free period.
In the femorotibial joint of the rat, treatment with AZD7969 resulted in synovial hyperplasia and fibroplasia. These changes were characterized by disorganized proliferation of the synovium and fibroblasts associated with the joint capsule and quadriceps femoris tendon, together with areas of prominent and sometimes mature blood vessels indicating prior angiogenesis (Figure 1A). Inflammation was not a feature of the hyperplastic change. In addition, in a proportion of animals, these changes were also associated with a periosteal reaction characterized by degeneration, necrosis, and hemorrhage.
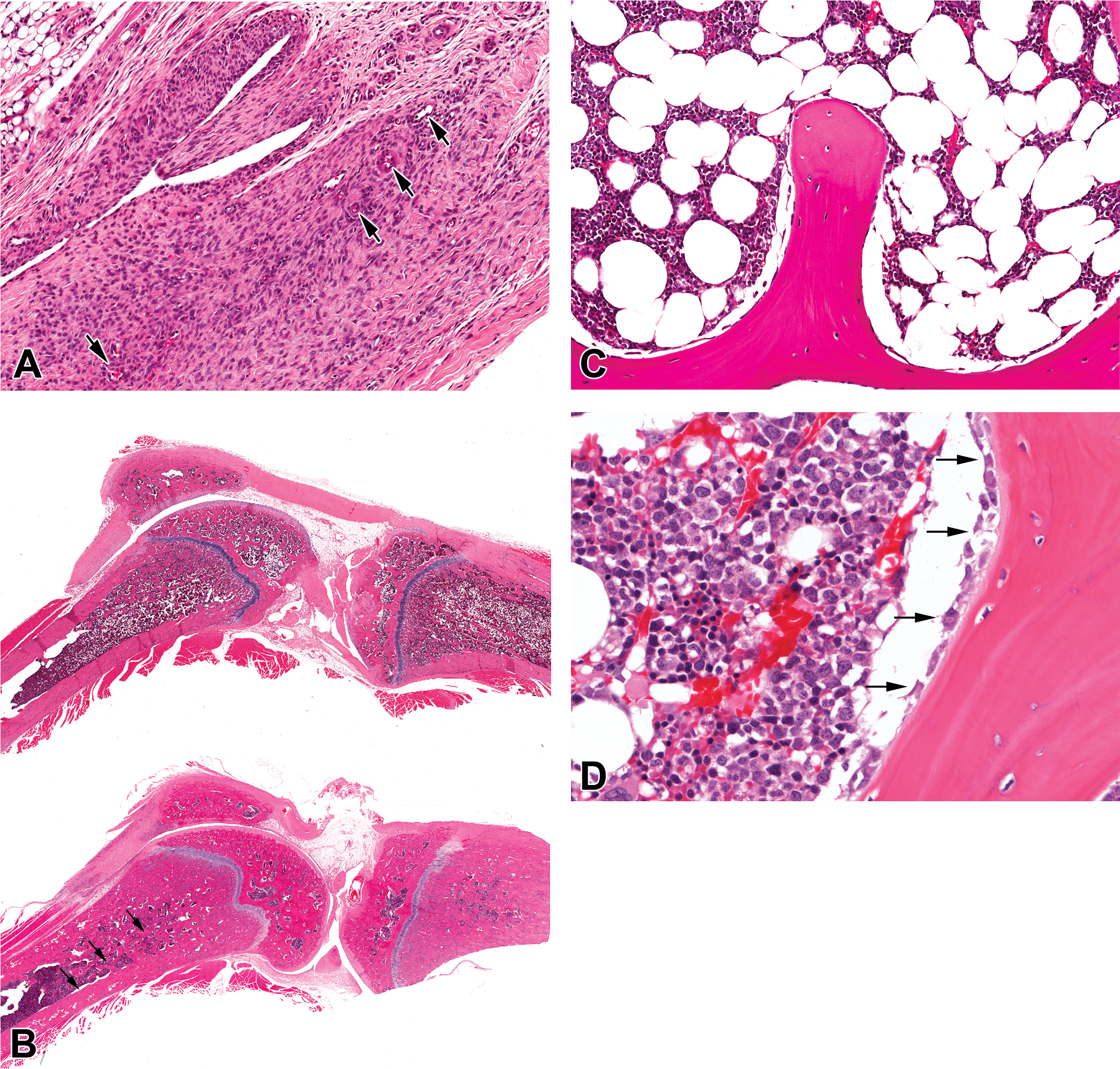
(A) Femorotibial Joint H&E higher power view of synovial membrane and quadriceps femoris tendon from a rat treated with AZD7969 for 1 month showing hyperplasia of fibrous and epitheliod cells together with prominent blood vessels (angiogenesis; arrows). (B) H&E Aperio whole slide scan of femorotibial joint from a control (upper panel) and a rat treated with 50-mg/kg/day AZD7969 for 1 month (lower panel) showing increased bone mass within the epiphysis and metaphysis together with an area of fibrodysplasia within the quadriceps femoris tendon (arrows). (C) Sternum 20× from an untreated dog showing inactive looking osteoblasts and (D) H&E high power view of sternum from a dog treated with AZD7969 for 3 weeks at 10 mg/kg/day showing increased numbers of activated osteoblasts (arrows) attached to endosteum of trabecular bone.
In the bones of both the rat and dog, treatment with AZD7969 resulted in a minimal (dog) to marked (rat) increase in bone mass (hyperostosis). In the rat, after 1 month of treatment, this change presented subgrossly as a loss of normal epiphyseal and metaphyseal architecture and was characterized by marked thickening of trabecular bone within the metaphysis and epiphysis of the femorotibial joint and sternum (Figure 1B). In the most severe cases, bone marrow space was significantly effaced. In the dog that showed less severe changes, hyperostosis was characterized by minimal cortical and trabecular hyperostosis within the sternum. In the dog 3-week study, precursor lesions were seen, characterized by minimal endosteal osteoblast proliferation (Figure 1D).
Remarkably similar morphological changes were also noted in the pulp cavity of the rat incisor, which showed changes disorganized proliferation of odontoblasts resulting in dental dysplasia. After 1 month of treatment, this change was characterized by severe changes within the odontogenic epithelium characterized by proliferation with inappropriate inward migration of odontoblasts into the pulp cavity resulting in effacement of the pulp cavity space (Figure 2B). Microscopically, this presented as hypercellularity with ectopic deposition of abnormal eosinophilic material (presumptive dentine) within the pulp cavity accompanied by large numbers of odontoblasts attached to and surrounding spicules of dentine (epithelial “rimming”; Figure 2C). These changes correlated with the macroscopic finding of malformation and elongation of the incisors and the clinical observation of abnormal tooth growth. In rats treated with 50 mg/kg/day, variable amounts of pulp necrosis was also observed. This latter change was thought to be caused by the in-life procedure of teeth trimming, as these changes were not present in the treatment groups in which this procedure had not been performed.
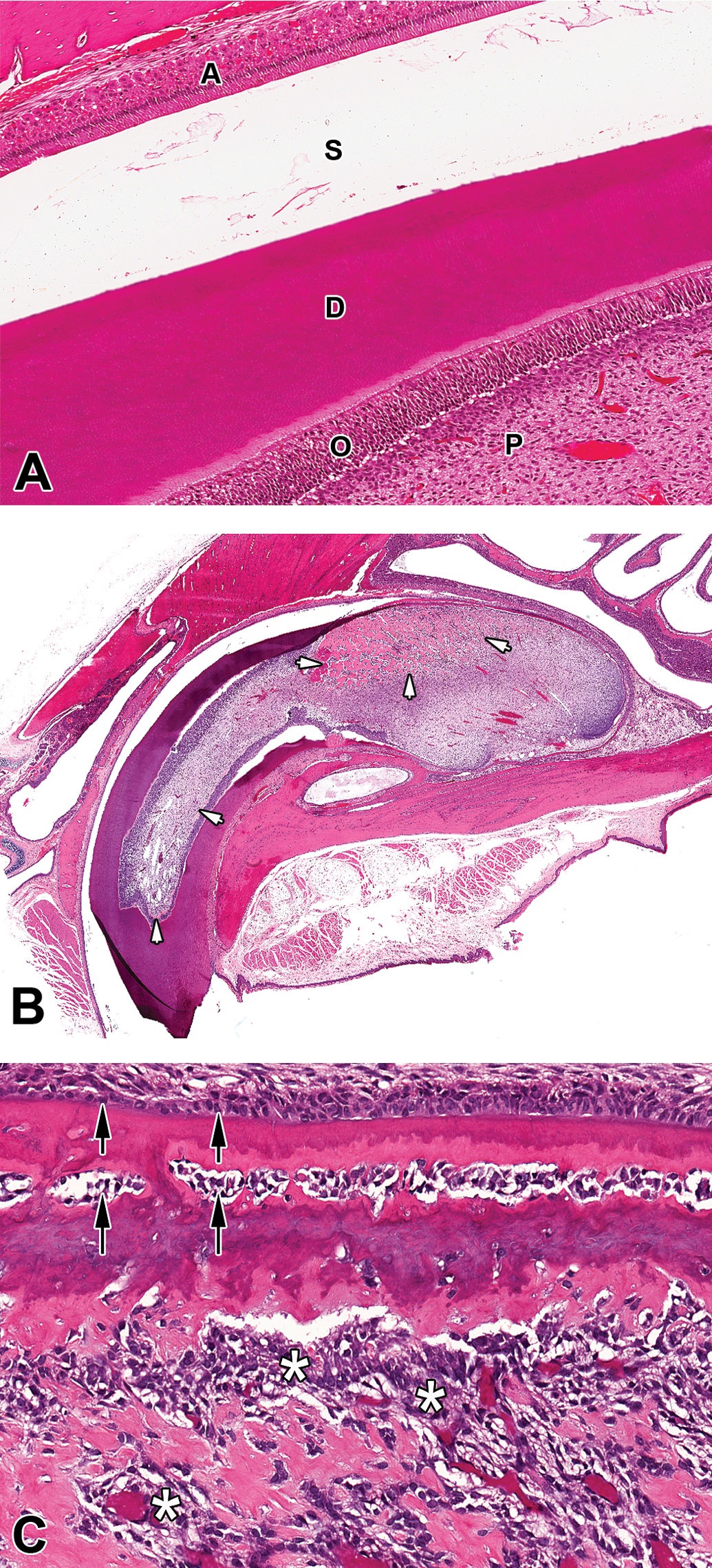
(A) Incisor tooth H&E high power image from an untreated rat showing normal layers of tooth (A = ameloblasts, S = enamel space, D = dentine, O = odontoblasts, P = pulp cavity). (B) H+E Aperio whole slide scan of upper incisor from a rat treated with AZD7969 for 1 month at 50 mg/kg/day showing abnormal growth and disorganization of odontogenic epithelium and dentine. The pulp cavity is enlarged and hypercellular with prominent areas of eosinophilic hyaline material (arrows) that appears morphologically similar to dentine. (C) H&E showing a higher power view of disorganized odontogenic epithelium with abnormal dentine, odontogenic epithelial hyperplasia and migration of odontoblasts into the pulp cavity (asterisks) with cuffing around areas of abnormal dentine-like material (arrowheads).
Liver and Gastrointestinal System
In the liver and throughout the gastrointestinal system, treatment with AZD7969 in both the rat and dog resulted in hyperplasia that maintained normal tissue architecture. There was no evidence of prior tissue damage or inflammation to indicate regeneration (except in the intestines of one dog killed prematurely). In the liver of the rat in both the 3-week and 1-month studies, proliferation was characterized by a reversible increase in the numbers of hepatocyte mitotic figures, whereas in the dog, increased size and numbers of bile ducts were noted in the 3-week study.
Treatment with AZD7969 also resulted in a nonreversible increase in hepatocellular iron in the liver of both rats and dogs (Table 7). This change was first noticed in the rat studies, which presented as a minimal increase in brown pigment in periportal hepatocytes during routine H&E examination. Staining with Perls’ Prussian Blue (PPB; Figure 3A; Hall et al. 2013) revealed a treatment and dose-related increase in hepatocellular iron which showed no no observed effect level (NOEL). This change was characterized by a predominantly periportal increase in fine pericanalicular intracytoplasmic inclusions that spread toward the centrilobular region with increasing dose of AZD7969 (Hall et al. 2013). In addition, increased iron was noted in the proximal small intestines (duodenum). PPB staining was localized predominantly within lamina propria macrophages toward the tip of the villi, but occasionally, fine intracellular granules were visible within villous enterocytes (Figure 3B). In the dog, H&E-stained sections showed no histologically visible brown pigment within hepatocytes; however, PPB staining revealed a treatment-related increase in hepatocellular iron in the 4-week study that appeared morphologically identical to that seen in the rat studies.
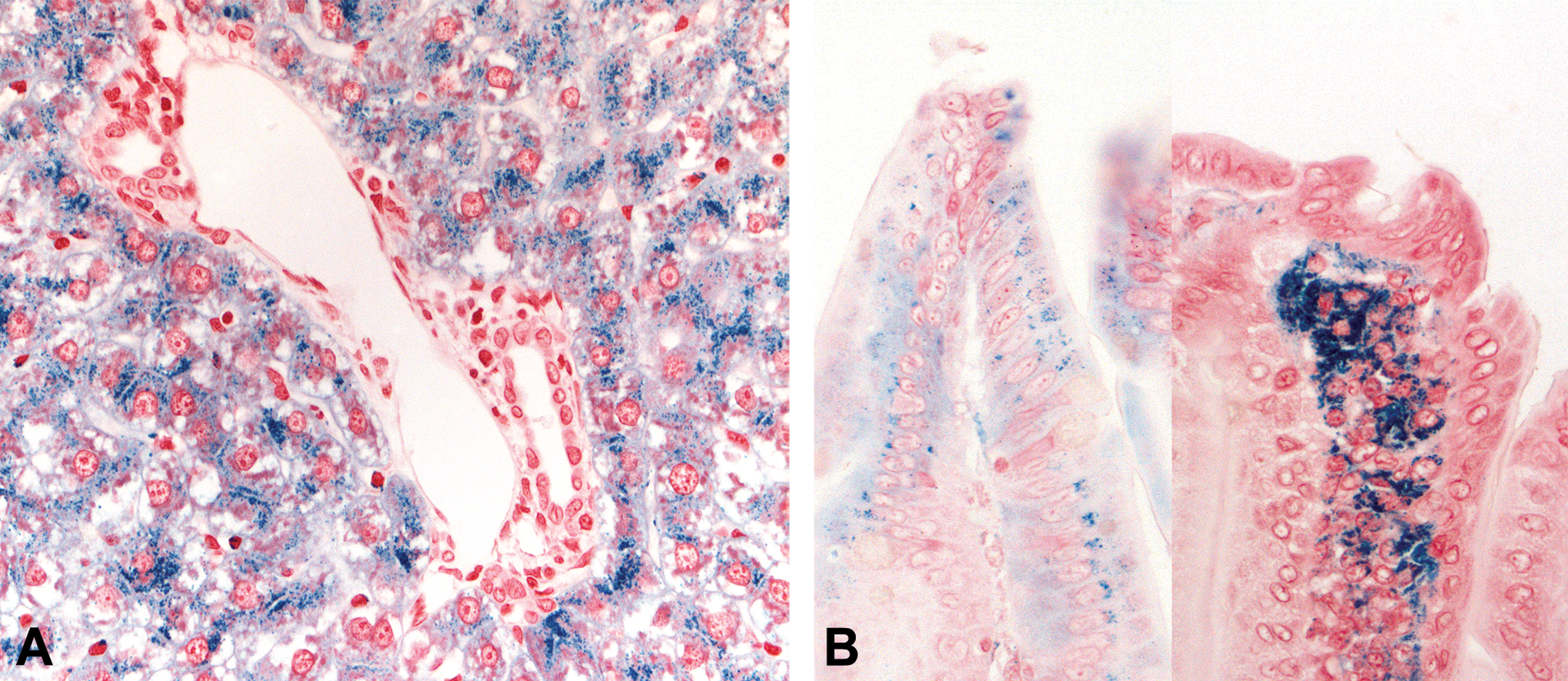
(A) Liver Prussian Blue 40× high power image from a rat treated with AZD7969 for 1 month at 50 mg/kg/day showing accumulation of pericanalicular Perls’ Prussian blue (PPB) positive iron in the periportal region of the liver. No significant Perls’ staining was noted in controls. (B) Duodenum Perls’ Prussian Blue High power image of duodenal villi from rats treated with AZD7969 for 1 month at 50 mg/kg/day showing absorption of PPB positive staining material (left panel) in gut enterocytes and accumulation of PPB stained material (right panel) in lamina propria. No significant Perls’ staining was noted in control sections.
PPB staining in 3-week and 1-month studies.
Note: Blank boxes represent no change; n/a = not applicable; DRF = dose range finding; PPB = Perls’ Prussian blue; mpk = mg/kg.
In addition to these changes, increased hepatocellular glycogen vacuolation was noted in the 1-month rat study, whereas in the 3-week rat study, increased extramedullary hematopoiesis (EMH) and increased single cell necrosis were noted. The increases in mitotic index, glycogen, EMH, and iron correlated with the increase in organ weight noted at necropsy. The increase in liver organ weight failed to recover after the 4-week treatment-free period.
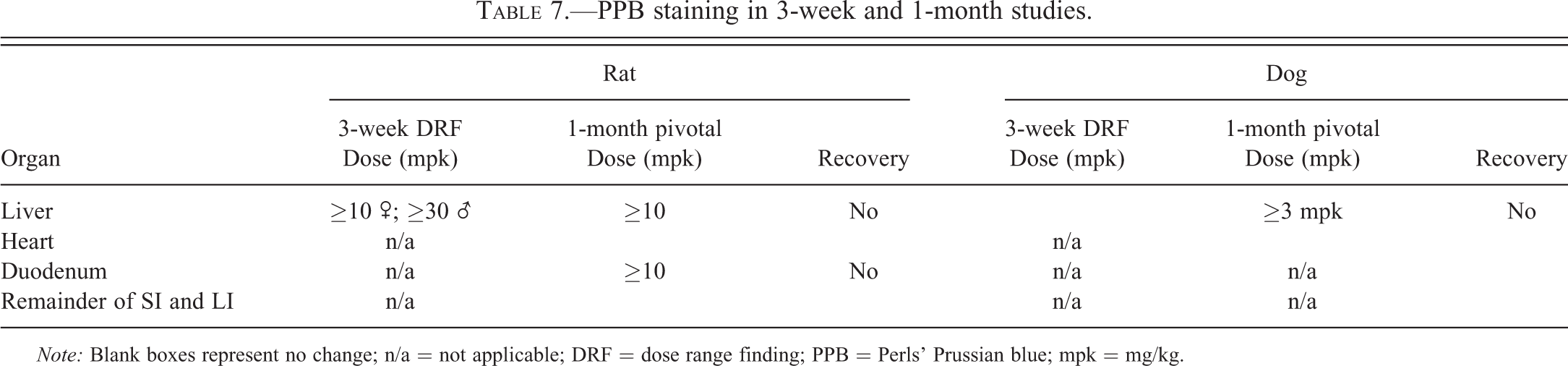
Changes in the GI tract of both the dog and rat were characterized by diffuse mucosal epithelial hypertrophy/hyperplasia of the fundic and pyloric (dog only) stomach foveolar epithelium and small and large (dog only) intestinal mucosa. Within the intestines, this appeared subgrossly as an enlargement of the intestinal villi/mucosa (Figure 4A) which was characterized by marked intestinal epithelial cell hypertrophy and prominent/enlarged crypts (Figure 4B). In the rat, this change correlated with the macroscopic finding of “thickened” noted throughout the small intestines in both the 3-week and 1-month studies.
(A) Duodenum H&E low power images of duodenum from a control rat (left panel) and a rat treated with AZD7969 for 1 month at 50 mg/kg/day (right panel) showing diffuse hypertrophy/hyperplasia of the mucosa. Intestinal crypts are prominent and villi are enlarged. Inset—Aperio whole slide scan showing areas where 4× images from control (upper) and treated (lower) images were taken. (B) H&E higher power view of a control (left panel) and treated rat (right panel) showing detail of villi. Enterocytes are hypertrophic but show regular morphology.
Kidneys and Adrenals
In the rat, reversible proliferative changes were noted in the kidney renal cortical tubules and adrenal cortex in both the 3-week and 1-month studies. As was the case in the liver, proliferation consisted of increased numbers of mitotic figures that resulted in hyperplasia with no architectural changes. Again, no prior tissue injury or inflammatory changes were present. In the kidneys from the 3-week DRF study, this change was accompanied by a diffuse corticomedullary tubular basophilia indicative of a more diffuse proliferative change. In the 1-month study, the proliferative changes were accompanied by a correlating increase in organ weight. Histological changes had reversed by the end of the treatment-free period, but organ weights were still significantly increased. Mildly increased urea and creatinine were also noted in the 1-month study, but it is unclear whether this change is related to the increase in cortical tubular proliferation.
In the adrenals, increased mitotic figures (noted at the zona fasiculara/glomerulosa boundary) was accompanied by a diffuse increase in cortical cell vacuolation which correlated in the 1-month study with the gross observation of discoloration (pale/faded). The increase in mitotic figures and cortical cell vacuolation were again accompanied in male animals by a correlating and reversible increase in adrenal weight.
Degenerative Changes
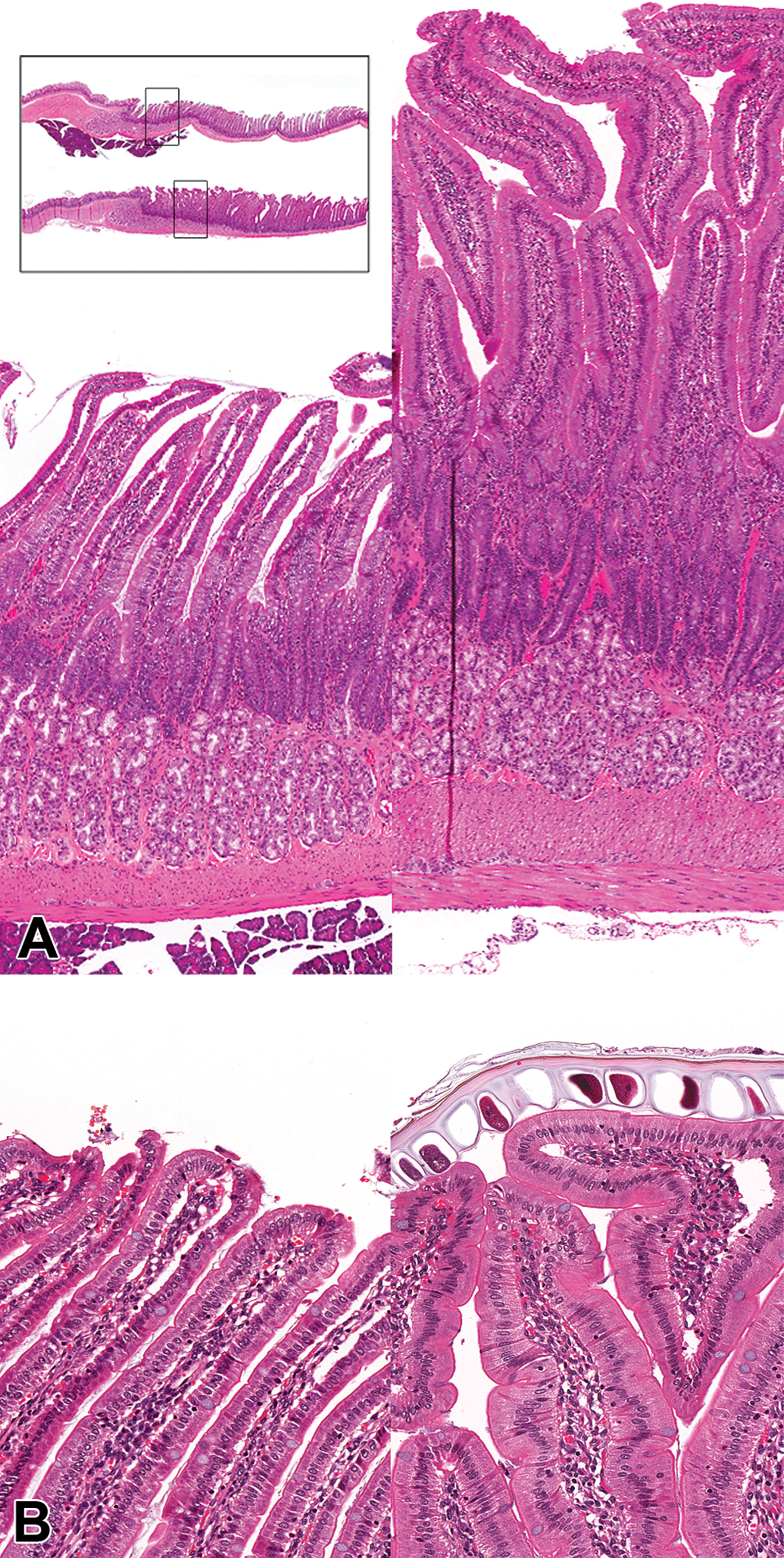
Degenerative changes were noted in the skin and Harderian gland of the rat and the testis of the dog. In the skin, single cell necrosis (apoptosis) of the follicular hair bulb/shaft was seen in both the 3-week and 1-month rat studies. In the most severely affected animals from the 3-week study, abnormal growth and orientation of the hair follicle was observed (Figure 5B) accompanied by degenerative vacuolar changes within the hair bulb and hair shaft with abnormal production of hair (Figure 5C). These changes correlated with alopecia that was noted in various areas of the body at necropsy in the 3-week-DRF study. In addition, patchy hair loss around the muzzle was noted during the in-life phase of the 1-month study which may correlate with this change, but could also be due to rubbing/irritation secondary to dental dysplasia. In the Harderian gland, single cell necrosis was seen in the 1-month study which resulted in acinar degeneration with compensatory regeneration. This change had almost completely recovered by the end of the treatment-free period except for the presence of moderate degeneration and single cell necrosis noted in a single female animal.
(A) Skin H&E low power and higher power view (inset) from an untreated rat showing normal arrangement and structure of hair follicles. (B) H&E low power image of skin from a rat treated with 70 mg/kg/day of AZD7969 showing degeneration of the hair follicle with vacuolation of the hair bulb/shaft and abnormal orientation of some hair follicles (arrowheads). (C) H&E showing detail of the hair bulb from another rat treated with 70 mg/kg/day of AZD7969 showing single cell necrosis (apoptosis).
In the dog only, treatment with AZD7969 resulted in seminiferous tubular degeneration/atrophy of the testis in both the 3-week and 1-month studies. This change was accompanied in the 1-month study by exfoliation of spermatogenic giant cells and germ cells into the lumen of the epididymides. Seminiferous tubular degeneration correlated with the decrease in organ weight noted at necropsy. Neither seminiferous tubular degeneration nor the organ weight decrease had recovered by the end of the treatment-free period (although exfoliation of germ cells into the epididymidis did recover). Finally, in dogs treated with 30 mg/kg/day in the 3-week study, seminiferous tubular dilatation was also noted.
Miscellaneous Changes
Lymphohematopoietic System
In the rat, increased EMH and hypocellularity of the marginal zone were seen in the spleen in both the 3-week and 1-month studies. Increased EMH correlated with the increase in organ weight noted at necropsy and the changes in RBC indices noted during clinical pathology assessment. Marginal zone hypocellularity showed complete recovery by the end of the treatment-free period whereas increases in EMH and changes in RBC indices, as well as increased organ weight, did not return to normal.
This was accompanied in the mesenteric lymph node by reduced cellularity in a small number of male animals dosed at 50 mg/kg/day and an increased incidence and severity of (agonal) hemorrhage into the medullary sinus. This latter change correlated with the macroscopic observation of “discoloration, red.” In the thymus, decreased cellularity was noted in the rat 1-month pivotal study, which correlated with the decrease in organ weight noted at necropsy in rats treated with 70 mg/kg/day. Histologically, this change was not present at the end of the treatment-free period, although organ weights were still decreased in male animals.
In the dog, hypocellularity was also noted in the thymus in the 1-month study. This change did not correlate with the increase in organ weight noted in this study. At the end of the treatment-free period, a slight reduction in the amount of atrophy was noted in the treated animals compared to concurrent controls.
Discussion
Treatment of rats and dogs with AZD7969 at dosages up to MTD resulted in a variety of treatment-related effects, many of which could be attributed to inhibition of GSK3β and downstream effects on β-catenin/Wnt signaling. Tolerability and clinical signs were, in general, dose-dependent and correlated well with histopathological assessment. Treatment-related changes could loosely be clustered into proliferative changes induced by GSK3β inhibition, and degenerative changes that resulted from a variety of mechanisms, the most likely of which are inanition (at or near MTD), stress, and off-target pharmacology.
In the rat, the most significant clinical effects resulting from treatment with AZD7969 were alopecia and decreases in body weight and food consumption. Alopecia correlated with the histological change of single cell necrosis within the hair follicle. Decreases in body weight and food consumption were considered to be secondary to the abnormal growth of the incisor teeth. This change was confirmed histologically as dental dysplasia and was considered the dose limiting toxicity.
In the dog, the most significant clinical signs were alopecia and soft/fluid feces that are common clinical signs seen in animals dosed at or near the MTD. Consequently, these adverse events are often attributed to locally high concentrations of compound producing an irritant effect on the gastric and intestinal mucosa. However, in this study, a generalized hyperplasia of the gastrointestinal tract was also noted, and so it is interesting to speculate whether this change may have altered intestinal transit and contributed to the clinical observations of soft/fluid feces and vomiting noted in this study. In the rat, the most interesting clinical pathology changes consisted of decreases in red cell parameters, increases in plasma urea and creatinine, and urinary NAG/creatinine and glucose/creatinine ratios. The hematological changes were consistent with a slight regenerative anemia secondary to increased demand due to lowered red cell mass. This change correlated with an increase in EMH noted in the spleen in the 3-week rat DRF study. The cause of this effect is not entirely clear, but a rare cause of anemia in man has been attributed to hyperostosis (Sreehari, Naik, and Eapen 2011). In this clinical syndrome, increases in bone mass result in effacement of bone marrow space and, consequently, a decrease in red cell production leading to a secondary regenerative effort in extrahemopoietic tissues. In our studies, this idea is supported by the observation that hyperostosis was particularly marked in the rat, whereas in the dog, where hyperostosis was only minimal, no significant hematological changes were seen. This change also correlates with the increases in serum calcium and phosphate, which were also more severe in the rat compared to the dog suggesting increased bone turnover (data not shown) in this species.
The changes noted in clinical chemistry may reflect damage to or loss of function within proximal tubular cells (Kavukcu, Soylu, and Turkmen 2002). However, elevations in these parameters occurred in the absence of cortical tubular basophilia or any other histological sign of cortical tubular damage (inflammation, necrosis, etc.) and are therefore unlikely to correlate with, or be the cause of, the increase in mitotic figures noted in the kidneys. This latter change is an unusual toxicologic change that is discussed subsequently.
During the development of AZD7969 and subsequent safety testing in the rodent and dog 3-week and 1-month toxicity studies, we noted a number of unexpected changes that were characterized by gross increases in organ size, increases in organ weight, and diffuse histological increases in proliferation, in normal adult tissues. In the GI tract, liver, kidney, and adrenal glands, proliferation maintained normal tissue architecture, whereas in the bones, incisor teeth, and femorotibial joint, proliferation resulted in reduced function or effacement of normal tissue space with significant changes in tissue architecture. These changes were particularly unusual in that they occurred in the absence of any precipitating cause or prior requirement for proliferation. However, their consistency with the known published effects of Wnt/β-catenin signal perturbation (discussed subsequently) together with the known specificity of AZD7969 leads us to conclude that these changes most probably resulted from pharmacological inhibition of GSK3β in the stem cell compartment.
The Wnt/β-catenin signaling pathway plays a fundamental role in the maintenance of adult stem cells by promoting self-renewal and cell survival. Inhibition of GSK3β by AZD7969 would be expected to increase Wnt/β-catenin signaling through decreased targeting of β-catenin for ubiquitin-mediated proteasomal degradation (Rubinfeld et al. 1996; Chen, Ding, and McCormick 2000). Although changes in β-catenin within the stem cell compartment were not directly assessed in these toxicity studies, previous studies in rat and human stem cells have shown that AZD7969 increased β-catenin stabilization and altered stem cell fate (Gilmour et al. 2013). In addition, other GSK3β inhibitors have been shown to alter stem cell responses in vivo (Gambardella et al. 2011), suggesting that AZD7969 may affect stem cell function via GSK3β inhibition in these preclinical toxicity studies. In the gastrointestinal system, treatment with AZD7969 presented grossly as a thickening of the intestinal tract, which reflected a diffuse hypertrophy/hyperplasia of the stomach fundic foveolar epithelium and small intestinal mucosa in both the rat and dog. Histological examination revealed significant hypertrophy of intestinal epithelial cells and an increased prominence/enlargement of the crypts indicating expansion of the stem cell niche. These data correlate with the known function of GSK3β in the intestines since the Wnt/β-catenin signaling axis is critical for the establishment and maintenance of the crypt stem cell niche. Transgenic mice lacking Tcf4 (transcription factor 4) do not develop intestinal crypts (Korinek et al. 1998; Clevers 2006) whereas conditional expression of the Wnt signaling inhibitor, Dkk-1 (Dickkopf-related protein 1), in adult mice results in reversible loss of crypts (Kuhnert et al. 2004). Our results are therefore consistent with these effects and suggest that treatment with AZD7969 derepresses stem cell Wnt/β-catenin signaling to promote crypt proliferation. Although these findings are thought to be linked to the effects of AZD7969 on GSK3 and the Wnt/β-catenin signaling pathway, it is possible that GSK3 interacts with other downstream transcription factors such as Notch and DISC (disrupted in schizophrenia 1), as it does in the nervous system to alter stem cell fate (Kim and Snider 2011). In the skeletal system, a generalized increase in bone mass was observed in both the rat and dog. Early changes were characterized by osteoblast hyperplasia and synthesis of trabecular bone. These changes are known to be accompanied by minimal increases in osteoclasts (Kugimiya et al. 2007), which is indicative of increased bone turnover and correlates with the moderate increase in serum phosphate and calcium seen in both the 3-week and 1-month rat studies. Later changes observed in the rat 1-month study consisted of increases in mature bone mass characterized primarily by hyperplasia of osteoblasts and thickening of epiphyseal and metasphyseal bony trabeculae. Again these results are consistent with inhibition of GSK3β in the stem cell niche since canonical Wnt/ββ-catenin signaling is strongly implicated in bone homeostasis (Clement-Lacroix et al. 2005; Kulkarni et al. 2006; Marsell et al. 2012). Pluripotent mesenchymal stem cells within the bone marrow give rise to osteoblastic, chondrogenic, adipogenic, and myogenic lineages; and Wnt/β-catenin signaling is thought to regulate osteoblast lineage commitment (Si et al. 2006). In experiments where Wnt/β-catenin signaling is disrupted due to a defect in LRP5, abnormalities characterized by low bone mass are seen both in man (osteoporosis-pseudoglioma syndrome; Gong et al. 2001) and in mice (abnormal osteoblast proliferation and maturation; Kato et al. 2002). Conversely, experiments in which there are gain-of-function mutations in LRP5 result in bone abnormalities characterized by increased bone mass with increased numbers of active osteoblasts (Little et al. 2002; Hofbauer, Maisch, and Schaefer 2002). Similarly, mice that are heterozygous GSK3β deficient also show increased bone formation (Kugimiya et al. 2007).
The changes noted in the rodent incisor appeared morphologically to be remarkably similar to those noted in the skeleton. Here, disorganization and hyperplasia of the odontogenic epithelium were noted, which resulted in deposition of abnormal dentine-like material within the pulp cavity (morphologically similar to hyperostosis in the bone). Many causes for dental changes in the growing rodent incisor have been documented including angiogenic inhibition (Hall, Westwood, and Wadsworth 2006; Patyna et al. 2008; Fletcher et al. 2010) and antitumor agents that target proliferating cells (Satoh et al. 2001). However, the changes induced by AZD7969 appeared to be unique and distinct from those induced by other types of compounds since the principal change appeared to be a proliferative rather than a degenerative change of the odontoblastic epithelium. In addition, the similarity of changes to those noted in the skeleton suggested to us that this change again is due to enhanced Wnt/β-catenin signaling in the odontogenic stem cell niche due to pharmacological inhibition of GSK3β by AZD7969. It also seems likely to us that the increased proliferation noted elsewhere in these studies ie femorotibial joint, liver, kidneys, and adrenals are also likely to be mediated via the same mechanism, namely increased Wnt/β-catenin signaling in the stem cell niche thorough inhibition of GSK3β.
Slight decreases in red cell mass were noted in the hematopoietic system, which are consistent with loss of marrow space due to hyperostosis. This mechanism is also consistent with the known signaling effects of Wnt/β-catenin within the marrow compartment. While in vitro data suggest that Wnt/β-catenin expression is important in maintaining hemopoietic stem cell self-renewal (Van Den Berg et al. 1998), in vivo results show that conditional deletion of β-catenin in transgenic mice has no significant impact upon bone marrow progenitor self-renewal (Cobas et al. 2004). Our results are consistent with those of Cobas et al. (2004) and suggest that Wnt/β-catenin signaling in the adult does not play a significant role in adult bone marrow hematopoietic stem cell renewal.
Treatment with AZD7969 resulted in single cell necrosis in the hair follicle, Harderian gland, and dog testis. In the murine testis, constitutive β-catenin/Wnt signaling in Sertoli cells results in infertility due to defective germ cell differentiation and apoptosis (Tanwar et al. 2010), whereas administration of lithium carbonate (a well-known inhibitor of GSK3b; Galli et al. 2012) to rats resulted in degeneration of spermatogenic cells and vacuolization of sertoli cells cytoplasm in the testis (Thakur et al. 2003). Taken together, these reports suggest that the loss of germ cells and generalized tubular atrophy noted in the dog testis is due to derepression of β-catenin/Wnt signaling by AZD7969. In the skin, however, these data are more difficult to reconcile with the generally growth promoting effects of β-catenin/Wnt signaling in stem cells, especially given that inhibition of GSK3β by valproic acid has been shown to activate the Wnt/β-catenin pathway and promote hair regrowth in vitro and in vivo in mice (Lee et al. 2012). It is, therefore, likely that this effect is mediated through competing off-target activity of AZD7969. Given the in vitro pharmacology profile for AZD7969, 2 possible candidates might account for this effect, protein kinase D1 (PKD 1) and dual specificity tyrosine-regulated kinase 1 (DYRK1), as both of these kinases are known to be expressed in mouse skin (Ernest Dodd et al. 2005; Rahmani et al. 1998). PKD 1 is expressed in proliferating keratinocytes (and a number of other cell types) and is thought to be pro-proliferative and/or anti-differentiative (Ernest Dodd et al. 2005) whereas DYRK1 has sequence homology to cyclin-dependent protein kinase (cdk) kinases (Rahmani et al. 1998) that may regulate cell proliferation and apoptosis (Yoshida 2008). Inhibition of either of these proteins might potentially disrupt the cell cycle control within the hair follicle resulting in promotion of apoptosis. The roles of GSK3β, PKD 1, and DYRK1 (as well as the other off-target hits) in the Harderian gland are, as far as the authors are aware, unknown.
In the 1-month rat study, treatment with AZD7969 resulted in increased hepatic glycogen vacuolation in male animals treated with 30 and 50 mg/kg/day despite an 8% to 12% loss in relative body weight gain in these animals compared to concurrent controls. This result was therefore considered significant and consistent with the expected effect of GSK3β inhibition in the liver. In the steady state condition, GSK3β is constitutively active and, in the resting cell, represses glycogen synthase (GS) activity, a key enzyme regulating the conversion of glucose to glycogen, resulting in the accumulation and storage of glycogen in the liver (Embi, Rylatt, and Cohen 1980; Frame, Cohen, and Biondi 2001). Derepression of GS is normally achieved through insulin which inactivates GSK3β and promotes dephosphorylation of GS via protein phosphatase 1 (Patel, Doble, and Woodgett 2004). In the liver, GSK3β inhibitors have been shown to promote increased GS and glucose uptake (Lochhead et al. 2001; Nikoulina et al. 2002; Cline et al. 2002). Our results are therefore consistent with these data and support the mechanistic rationale that GSK3β inhibitors might act as a therapeutic target for the treatment of type 2 diabetes (Patel, Doble, and Woodgett 2004).
Treatment with AZD7969 and staining with PPB revealed increased hepatic iron accumulation in both the rat and dog (Hall et al. 2013). This change was accompanied by increased iron deposition (PPB staining) in the duodenal mucosa indicating increased uptake from the diet (primary iron overloading). In order to quantify the amount of iron accumulation, and determine the source of the iron loading in the liver, as well as understand the possible risk of iron deposition in other organs such as the heart, sections of liver, intestines, and heart were cut and stained with PPB. The results of the liver investigation have been reported separately (Hall et al. 2013) and summarized in Table 7. No increase in PPB staining was visible in the heart, distal small intestines, or large intestines. However, significantly increased PPB staining was present in the proximal small intestines (duodenum), indicating that the source of the iron loading in the liver was most likely derived from increased absorption of dietary iron from within the proximal small intestines.
Genetic hemochromatosis in humans is a clinical syndrome that is characterized by iron overloading as a consequence of deregulated absorption of iron at the level of the proximal small intestine (Mclaren et al. 1984). Iron is usually first deposited in the periportal region of the liver and as the disease progresses, it spreads centripetally toward the central vein in the same pattern as that noted in our experiments with increasing exposure to AZD7969. This inborn error of metabolism usually results from an inherited defect in a small number of genes that regulate the master iron regulating hormone of the body—hepcidin (Nemeth and Ganz 2009). Although the long-term toxic consequences of iron overloading characteristic of genetic hemochromatosis in man were not investigated in this study, treatment with AZD7969 did result in a potent and dose-responsive iron loading disorder (Hall et al. 2013) that might prove to be a useful tool compound to study these types of diseases in the laboratory. However, one limitation to this model might be a lack of understanding around the mechanism through which this effect is mediated. We therefore offer 2 possible explanations. Since GSK3β has not been reported to regulate iron metabolism, we suggest that this effect is due to off-target pharmacology. Other internal studies using GSK3β inhibitors from different structural series support this notion (personal observation). AZD7969 structural activity relationships suggest that hepcidin may be an off-target candidate, but we have not pursued this possibility. In addition, HIPK2 (homeodomain-interacting protein kinase 2) may represent an off-target candidate, since it is known that under conditions of genotoxic stress, activation of HIPK2 results in decreased ferritin H transcription (the major intracellular iron storage gene) via activation of ATF1 (activating transcription factor 1; Hailemariam et al. 2010). Inhibition of HIPK2 might therefore lead to increased ferritin H transcription and increased storage of iron bound to ferritin. However, AZD7969 and its metabolic products are not directly genotoxic (data not shown) and therefore it is uncertain whether under these conditions HIPK2 repression would markedly affect iron storage.
Summary
Taken together, we have presented data that show good concordance between the Han Wistar rat and the Beagle dog in both 3-week and 1-month toxicity studies. Our data are also consistent with the known effects of GSK3β inhibition within the adult stem cell compartment resulting in hyperplasia that was either organized and nontissue effacing, or disorganized and tissue effacing. We also show that treatment with AZD7969 increases hepatocyte glycogen accumulation consistent with the expected effects of inhibition of GSK3β in the liver. Finally, we demonstrate that treatment with AZD7969 results in iron overloading in both rats and dogs that might prove to be a useful tool compound for studying other types of iron loading disorders.
Our data also support the mechanistic rationale for use of GSK3β inhibitors to treat bone disorders, such as pathological bone loss secondary to myeloma, and disorders of glucose/glycogen metabolism (type 2 diabetes). GSK3β inhibitors have also been proposed as potential new drugs to treat a number of human cancers. However, our data suggest that inhibition of GSK3β induces proliferative effects in a wide range of normal organs and may therefore promote tumor growth, or the proliferation of precancerous lesions, in a subset of human cancers.
Footnotes
Acknowledgments
The authors would like to acknowledge the original study pathologists Jayne Harris, Jen Barnes, and Russell Westwood. We would also like to acknowledge John Foster for helpful comments in proofreading the manuscript.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
