Abstract
Islet-like clusters derived from human embryonic stem cells (hESC) hold the potential to cure type 1 diabetes mellitus. Differentiation protocols of islet-like clusters lead to the generation of minor fractions of nonendocrine cells, which are mainly from endodermal and mesodermal lineages, and the risk of implanting these is unclear. In the present study, the histogenesis and the tumorigenicity of nonendocrine cells were investigated in vivo. Immunodeficient mice were implanted under the kidney capsule with islet-like clusters which were derived from differentiation of cells batches with either an intermediate or poor cell purity and followed for 8 or 26 weeks. Using immunohistochemistry and other techniques, it was found that the intermediate differentiated cell implants had limited numbers of small duct-like cysts and nonpancreatic tissue resembling gastrointestinal and retinal pigmented epithelium. In contrast, highly proliferative cystic teratomas were found at a high incidence at the implant site after 8 weeks, only in the animals implanted with the poorly differentiated cells. These findings indicate that the risk for teratoma formation and the amount of nonpancreatic tissue can be minimized by careful in-process characterization of the cells and thus highlights the importance of high purity at transplantation and a thorough ex-vivo characterization during cell product development.
Keywords
Introduction
Type 1 diabetes is a chronic and increasingly prevalent autoimmune disease caused by a progressive loss of insulin-producing β cells following an autoimmune reaction triggered by a combination of poorly defined genetic and environmental factors. 1 The loss of insulin-producing β cells leads to hyperglycemia and if not treated, to a fatal state of ketoacidosis. Although lifesaving for type 1 diabetics, treatment with exogenous insulin may not be sufficient to obtain full glycemic control and lack hereof may lead to late-stage complications including kidney failure, blindness, stroke, and limb amputations. 2 Recent research has also identified other morbidities associated with long-term, unregulated diabetes including cognitive impairment and increased susceptibility to infectious agents including severe acute respiratory syndrome coronavirus 2. 3,4
Proof of concept clinical studies using insulin-producing human islets from deceased donors have been performed successfully. This cell replacement therapy has resulted in remission of diabetes for up to several years and a reduction in hypoglycemic events. 5 Unfortunately, this approach is limited by variable donor tissue quality and limited tissue availability due to lack of donors; therefore, in vitro-produced human embryonic stem cell (hESC)-derived islets are a scalable alternative. 6
Considerable progress has been made since these early reports on reproducible and high-quality differentiation protocols. 6,7 Many investigators in stem cell research focus on protocol improvements used to generate the endocrine cells with enhanced efficacy in treating diabetes. 8,9 However, the function and histogenesis of the nonendocrine cell populations generated during in vitro differentiation lack characterization. It is not clear if these nonendocrine cells are passive bystanders or pose a potential safety risk, including the formation of teratomas. Regulatory guidance on the assessment of hESC-derived therapies is limited although there is a general expectation of fully characterizing the phenotype of all implanted cells. 10,11 The aim of this study was to characterize the nonendocrine cells within the grafted hESC-derived islets to gain insights into their histogenesis and tumorigenicity. We accomplished this by comparing the nonendocrine cell populations present after transplantation of intermediate differentiated and poorly differentiated cell batches, both implanted under the kidney capsule of immunodeficient SCID-Beige mice.
Materials and Methods
Human Subjects Statement
Human pancreas tissue control samples used in immunohistochemistry (IHC) and in situ hybridization (ISH) were procured from the National Hospital of Denmark (Rigshospitalet) in Copenhagen, Denmark, and were of surgical origin only. Sampling was in all cases conducted with bioethical approval (H-3-2010-076) from the hospital’s institutional review board and Institutional Ethics Committee IRB/IEC with informed consent provided and received from all donor(s).
Human Embryonic Stem Cell–Derived Islet Differentiation
The Cellartis Human ES Cell Line 121 (SA121, Takara, cat: Y00020) was used as a source of hESC. This line is derived at Takara Bio Europe AB and follows all applicable laws in Sweden and European Union and is approved by the regional ethical authority. Informed donor consent has been given for research purposes. The cells were expanded using the Cellartis DEF-CS 500 Culture System (Cat. No. Y30010) before starting differentiation. Once expanded, the embryonic stem cells were differentiated using a multistage protocol, similar to published protocols. 8 The protocol takes the cells through definitive endoderm (DE) specified by expression of SOX17, pancreatic endoderm defined by coexpression of PDX1/NKX6.1, Endocrine progenitor stage without marker definition and a final β-cell stage with β cells defined as NKX6.1/C-peptide positive cells. The differentiation protocol used a research grade cell line with research grade components only meant for research purposes. Therefore, we used the term research grade hESC-derived islet implants. The differentiated cells were a kind gift from the department of stem cell biology, Novo Nordisk A/S.
Flow Cytometry
Cells were characterized by flow cytometry to determine protein expression of markers relevant to islets differentiated from hESC (Supplemental Table S1 for markers and protocol). Clusters were converted to single cells by dissociation with TrypLE-Select (Gibco#12563-011). Dissociation was stopped with RPMI (Gibco #61870) with 12% KnockOut Serum Replacement (Gibco#10828-028), and cells were washed twice in phosphate-buffered saline containing calcium and magnesium (PBS +/+) before fixation with 10% formalin. After fixation, cells were stored in PBS without calcium and magnesium (−/−) with 1% bovine serum albumin (BSA) at 4 °C until analysis. Before analysis, cells were filtered using a 40 μM filter. Cells were permeabilized in PBS+/+ with 5% donkey serum and 0.2% Triton X100 before incubation with primary or directly conjugated antibodies (See Supplemental Table S1). After staining, the cells were washed twice in PBS−/− with 1% BSA and analyzed using an LSRFortessa instrument from BD Bioscience. Results were analyzed using FlowJo and cells were gated according to nonstained and single stained controls.
Single-Cell RNA Sequencing (SC RNA-Seq)
Detection of RNA expression in the differentiated cell batches was performed with SC RNA-Seq. To obtain single cells, clusters at the β-cell stage were dissociated into single cells the same day as the cells were transplanted using Accutase dissociation (Stem cell technologies #7920). Cells were cryopreserved in stem cell banker (AMS Biotechnology #11890) and stored in liquid nitrogen until analysis. Gene expression profiles for single cells were obtained for around 3000 cells per protocol. Single cells were captured using droplet-based Chromium Next GEM single-cell 3′ kits (10× Genomics, PN-1000120), according to the manufacturer’s recommendations. The single cells were encapsulated with barcoded beads, followed by cell lysis and reverse transcription in droplets. Reverse transcription was performed with polyT primers containing cell-specific barcodes, unique molecular identifiers, and adaptor sequences. Samples were sequenced on Illumina NextSeq550 for 75 cycles. Cell ranger was used for alignment and transcript counting of libraries, and the R package “Seurat” version 3.0, for cell-type identification. We removed genes that were not found in at least 3 cells and removed cells that had less than 200 transcripts of those genes. The gene expression profile of each cell was analyzed and compared to all cells sequenced and clustered together with cells that had a similar gene expression profile independent of the protocol used to generate the cells.
Animals
All live animal procedures employed were reviewed and approved by the Novo Nordisk Ethical Review Committee and were compliant with all applicable Danish animal welfare rules via the Animal Experiment Inspectorate (Dyreforsøgstilsynet). Animals included in this study were immunodeficient Fox Chase SCID-Beige (CB17.Cg-PrkdcscidLystbg-J/Crl), male mice, sourced from Charles River Laboratories (Germany) at 4 to 6 weeks of age. Once acclimatized, animals underwent hESC-derived islet implantation under the left kidney capsule (procedure described in detail in Supplemental Methods). Animals were kept on a 12-hour light/dark cycle and fed an Altromin diet ad libitum. Animals were visually inspected daily for signs of pain or clinical signs of distress and weighed once a week. Animal termination was done under isoflurane anesthesia followed by cervical dislocation prior to necropsy. For more detailed information regarding surgical procedures, see Supplemental Methods.
Study Design
The study involved 2 different hESC-derived islet batches, one intermediate differentiated, that went through an endocrine enrichment procedure, 12 and one poorly differentiated, with no endocrine enrichment procedure, both derived using a suboptimal research grade protocol (Table 1). The poorly differentiated batch functioned as a positive control for tumor formation. Group 1 (n = 10) and group 2 (n = 68) were transplanted with intermediate differentiated islets and followed until necropsy at 8- or 26-week posttransplantation, respectively. Group 3 (n = 10) and group 4 (n = 30) were transplanted with poorly differentiated islets and followed until necropsy at 8 or 10 weeks posttransplantation, respectively. Group 4 was planned to be followed until study termination at 26 weeks but had to be killed early (week 10) due to large cystic tumors from the implant effacing adjacent abdominal organs. Group 5 (n = 18) was a sham control group that was killed 26 weeks posttransplantation. For more detailed information regarding study design, see Supplemental Methods.
Study Design.
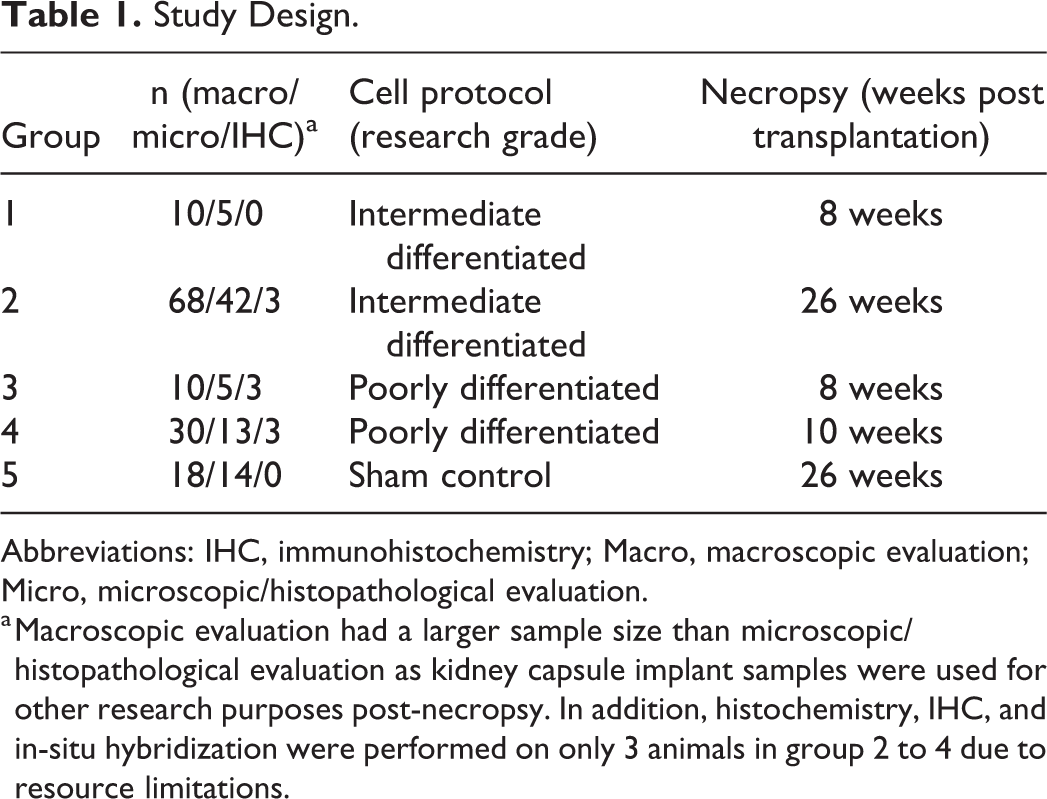
Abbreviations: IHC, immunohistochemistry; Macro, macroscopic evaluation; Micro, microscopic/histopathological evaluation.
a Macroscopic evaluation had a larger sample size than microscopic/histopathological evaluation as kidney capsule implant samples were used for other research purposes post-necropsy. In addition, histochemistry, IHC, and in-situ hybridization were performed on only 3 animals in group 2 to 4 due to resource limitations.
Necropsy and Tissue Processing
All animals underwent a gross examination according to standard veterinary necropsy procedures of rodents. 13 All organs in the abdominal and thoracic cavity were assessed for gross changes. Liver, kidneys including implant, spleen, heart, lungs, and any abnormalities were collected and fixed in 10% neutral buffered formalin for histological processing and histopathologic evaluation.
Gross Pathological Evaluation
Gross assessment was made on all kidney capsule implant areas, to assess size and additional findings including pigmentation in the implant. Grades were assigned as minimal, mild, moderate, marked, or severe based on an increasing extent and/or complexity of change unless otherwise specified in the results. 14
Histology of hESC-Derived Islet Implants
Trimming was performed by placing the kidney in a stainless steel matrix typically used for coronal mouse brain dissection and making 4 cross sections distributed equally along the length of the kidney, resulting in 2 sections from the kidney poles and 2 around the pelvis. Tissues were processed for permanent paraffin embedding on a Leica ASP 300 tissue processor (Leica Microsystems) and embedded in paraffin. Paraffin blocks were cut at 4 µm on a rotating microtome (Leica Microsystems) and placed on adhesive glass slides (Fisher Scientific). Semiquantitative scoring of hematoxylin and eosin (H&E) slides was performed. The implant containing hESC-derived islets under the left kidney capsule was morphologically assessed and thus human cells were evaluated in mouse tissue. Complexity and extent for each finding were graded using a semiquantitative 5-point severity score (minimal, mild, moderate, marked, severe). 14
Histochemistry, IHC, and ISH
Based on the morphology of H&E, 3 animals from one intermediate differentiated group necropsied 26 weeks posttransplantation and 3 animals from each of the poorly differentiated groups killed at 8- and 10-week posttransplantation were chosen for further characterization using histochemistry, IHC, and ISH. These animals were representatively chosen to reflect variation in morphology and the extent of the findings. We only characterized one intermediate differentiated group due to similar morphology between the 2 groups. The animals were also selected based on planned necropsy (no autolysis) and no systemic lesions (eg, thymic lymphoma). For all tissue staining protocols utilized (ie, histochemistry, IHC, and ISH), adjacent tissue sections from the same individual animals were stained. We used 3 histochemical stains, namely H&E (Sigma-Aldrich), Fontana Masson’s (Abcam), and Periodic acid–Schiff (PAS, Merck; see Supplemental Methods for protocols).
The IHC protocols consisted of both manual chromogen and immunofluorescence staining and the use of the Discovery ULTRA automated stainer from Ventana Medical System. The following primary antibodies were used: cytokeratin 19 (CK19, Abcam), carbonic anhydrase II (CA2, Atlas Antibodies), caudal type homeobox 2 (CDX2, Thermo Scientific), cystic fibrosis transmembrane conductance regulator (CFTR, Atlas Antibodies), Retinal pigment epithelium-specific 65 kDa protein (RPE65, Abcam), KU80 (Cell Signalling), SRY-box 2 (SOX2, R&D Systems), Ki-67 (Acris), Vimentin (Abcam), Nestin (Merck; Supplemental Methods and Supplemental Table S2 for protocols).
Chromogen-based IHC detection and visualization were done by secondary antibody-conjugated horseradish peroxidase and 3,3-diaminobenzidine and counterstained with hematoxylin and for immunofluorescence protocols, we used secondary antibodies conjugated with Cy2, Cy3, and Cy5 counterstained with 4′,6-diamidino-2-phenylindole.
Fully automated ISH was performed using the Ventana RNAscope Discovery ULTRA system in combination with the RNAscope 2.5 VS detection kit (mRNA RED, #760-234, Advanced Cell Diagnostics), according to manufacturer’s instructions (#322250-USM-ULT, Advanced Cell Diagnostics). The following probes were used: Human CFTR (ACD Bio) and Human Bestrophin-1 (ACD Bio) (Supplemental Methods and Supplemental Table S2 for protocols), and the slides were counterstained with hematoxylin.
Scanning, Viewing, and Imaging of Histological Specimens
Glass slides with tissues that were histochemical stained (eg, H&E) or immunostained with chromogens (IHC) were scanned at 20× using the Leica Aperio AT2 slide scanner and viewed with Aperio Imagescope v. 12.4.0.5043 (Leica Biosystems). Glass slides with tissues that were immunofluorescence stained were scanned at 10× using the Olympus VS120 and viewed with Olympus Olyvia 3.1 (Olympus).
Results
In Vitro Characterization of 2 hESC-Derived Islet Batches With Different Levels of Nonendocrine Impurities
This investigation on the histogenesis and potential safety risks of nonendocrine subpopulations, which form during cell differentiation, employed 2 research-grade hESC-derived islet batches. Both batches were generated using similar research stage differentiation protocols with research-grade cells and raw materials and gave rise to more than 50% of β-like cells, defined by NKX6.1 and c-peptide coexpression (Table 2). Despite giving rise to a similar percentage of β-like cells, the 2 batches showed very different differentiation efficacy at each substage, as listed under impurities in Table 3. The batch referred to as poorly differentiated was included as a positive control for tumor formation. This batch had a high number of undifferentiated cells already at the DE stage (9% defined by OCT 3/4 expression) and also a large proportion of cells coexpressing pluripotency markers (SOX2, NANOG, and POU5F1, Figure 1A) at the β-cell stage prior to transplantation. In addition, in this batch, more than 60% of the cells did not become pancreatic endoderm, as defined by lack of PDX1 expression (Table 3). Furthermore, to maximize the nonendocrine cell populations, this batch was not taken through cryopreservation and reaggregation, a process known to efficiently decrease the amount of nonendocrine cells. 9,12 The batch referred to as intermediate differentiated was included to identify a threshold for the amount of proliferative and nonendocrine subpopulations which would not give rise to tumors but would still allow the investigation of nonpancreatic tissue formation. This batch still contained nonendocrine cells at each stage, but the percentages were significantly lower compared to the poorly differentiated batch, for example, 2.5% nonendocrine cells, based on the cells being negative for the endocrine marker chromogranin A versus 39.7% in the poorly differentiated batch in the SC RNA-Seq data (Table 3). In addition, based on flow cytometry, the proliferation was much higher in the poorly differentiated batch with 5.1% Ki-67 positive cells versus 0.2% in the intermediate differentiated batch, and there was no coexpression of 3 pluripotency markers (Figure 1B).
hESC Differentiation in-Process Characterization of Purities.
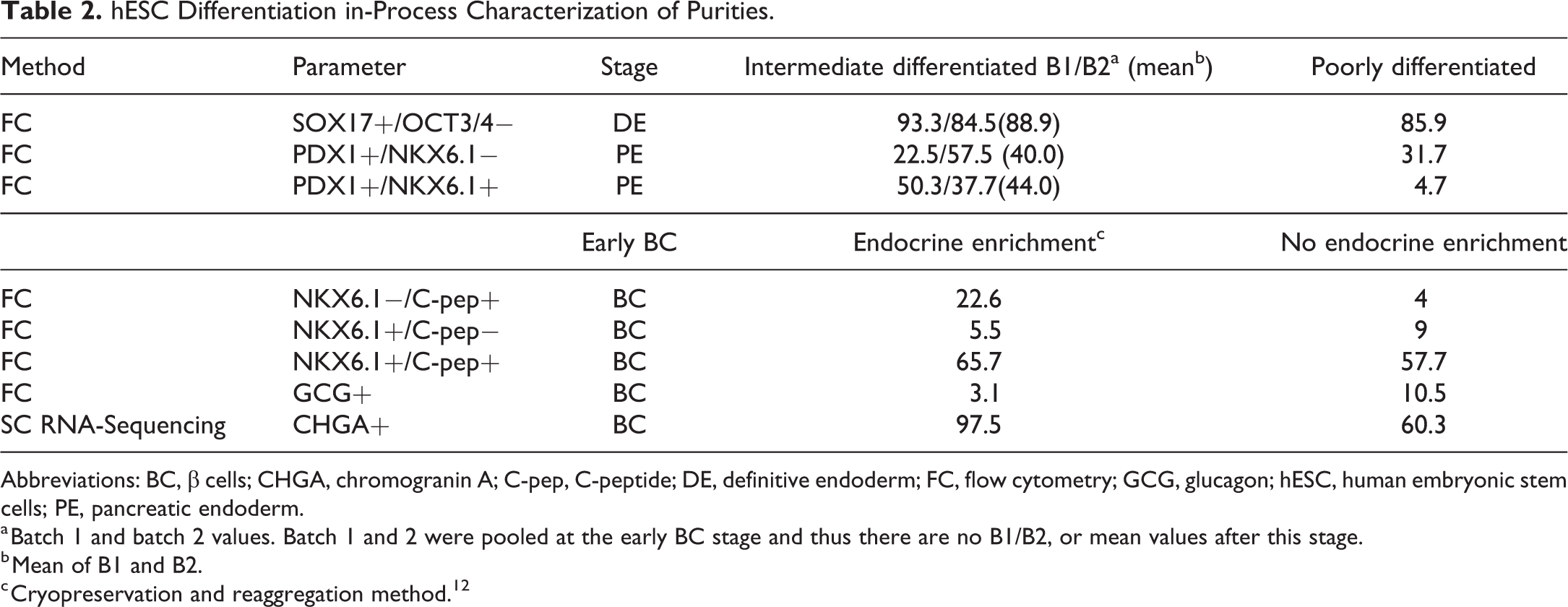
Abbreviations: BC, β cells; CHGA, chromogranin A; C-pep, C-peptide; DE, definitive endoderm; FC, flow cytometry; GCG, glucagon; hESC, human embryonic stem cells; PE, pancreatic endoderm.
a Batch 1 and batch 2 values. Batch 1 and 2 were pooled at the early BC stage and thus there are no B1/B2, or mean values after this stage.
b Mean of B1 and B2.
c Cryopreservation and reaggregation method. 12
hESC Differentiation in-Process Characterization of Impurities.
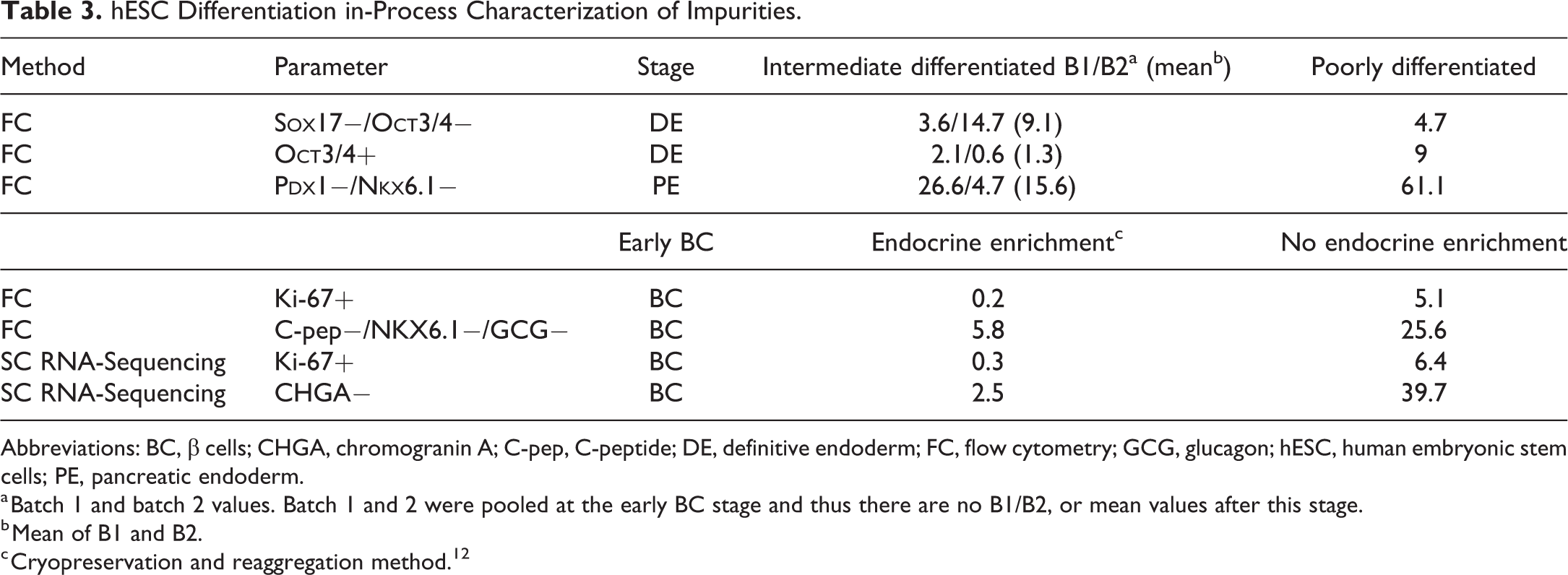
Abbreviations: BC, β cells; CHGA, chromogranin A; C-pep, C-peptide; DE, definitive endoderm; FC, flow cytometry; GCG, glucagon; hESC, human embryonic stem cells; PE, pancreatic endoderm.
a Batch 1 and batch 2 values. Batch 1 and 2 were pooled at the early BC stage and thus there are no B1/B2, or mean values after this stage.
b Mean of B1 and B2.
c Cryopreservation and reaggregation method. 12
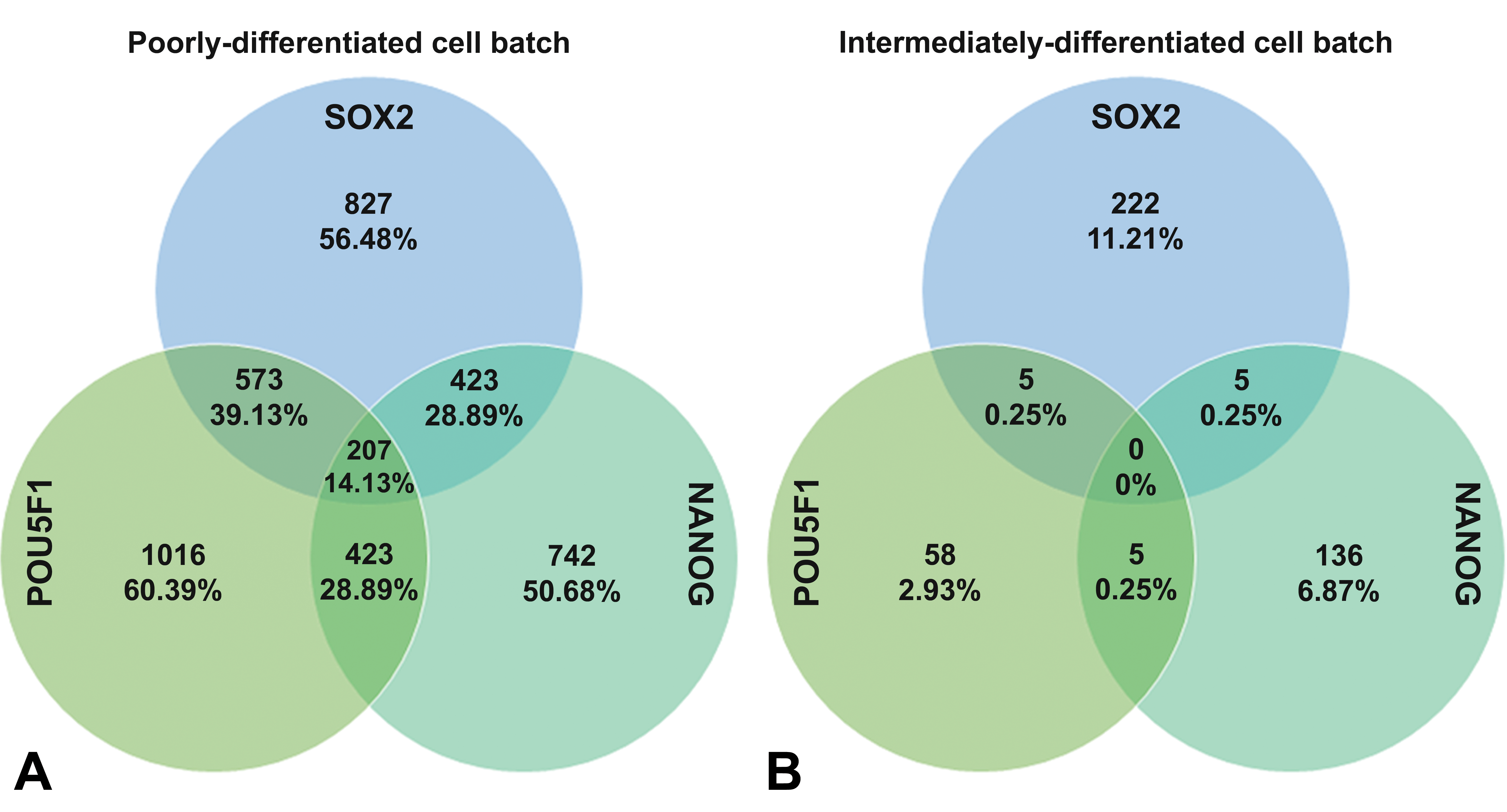
Venn diagram of 3 common pluripotency markers and their expression measured by single-cell RNA sequencing. (A) In the poorly differentiated human embryonic stem cell (hESC)-derived islet cell batch, there were 14,1% of the analyzed cells coexpressing all 3 pluripotency markers, which we consider a strong signal for pluripotency. Furthermore, 28.9% to 39.1% of the cells coexpress 2 pluripotency markers. (B) In the intermediate differentiated hESC-derived islet cell batch sample, there were no cells coexpressing all 3 pluripotency markers, and the maximum proportion that coexpressed 2 markers were 0.25%. Percentages are based on total cells analyzed for each batch.
Poorly Differentiated hESC-Derived Islets Caused Teratomas and Affected Survivability in SCID-Beige Mice
The intermediate and poorly differentiated hESC-derived islets were implanted under the kidney capsule of immunodeficient SCID-Beige mice. To gain insights on severity and time to incidence, a group of animals transplanted with each of these batches was killed 8 weeks postimplantation. At this point, large cystic tumors were found at the implant site in 90% of animals implanted with the poorly differentiated batch in contrast to only rare benign cysts in the intermediate differentiated batch (Table 4, Figure 2A and B). Metastasis of tumors beyond the implant site was not observed. These tumors led to the early termination of all animals transplanted with the poorly differentiated batch of cells 10 weeks after transplantation, and an overall tumor incidence of 93% was confirmed in this group during necropsy. These groups had tumors exclusively at the implant site, while there was no evidence of invasion or metastasis. The presence of large cystic tumors and the high incidence rate at 8 and 10 weeks showed that the implant environment did support tumor formation and affected the survivability due to early necropsy for animal welfare reasons (Supplemental Figure S1). Further histological examination of the tumors from the animals transplanted with poorly differentiated cells showed that the tumors were mucinous and expansile and consisted of variable amounts of cartilage, mucosal epithelium, and neuroectoderm-like cells forming pseudo palisades and primitive neural tubes (Figure 2C and D). Staining with vimentin, CK19, and Nestin (Figure 2E and F) confirmed the presence of tissue with mesoderm, endoderm, and neuroectoderm origin characteristic of teratomas. The incidence, confirmed histologically, was also high (80% vs 77% at 8 and 10 weeks, respectively; Table 5).
Summary and Incidence of Macroscopic Findings, Implant Site.a
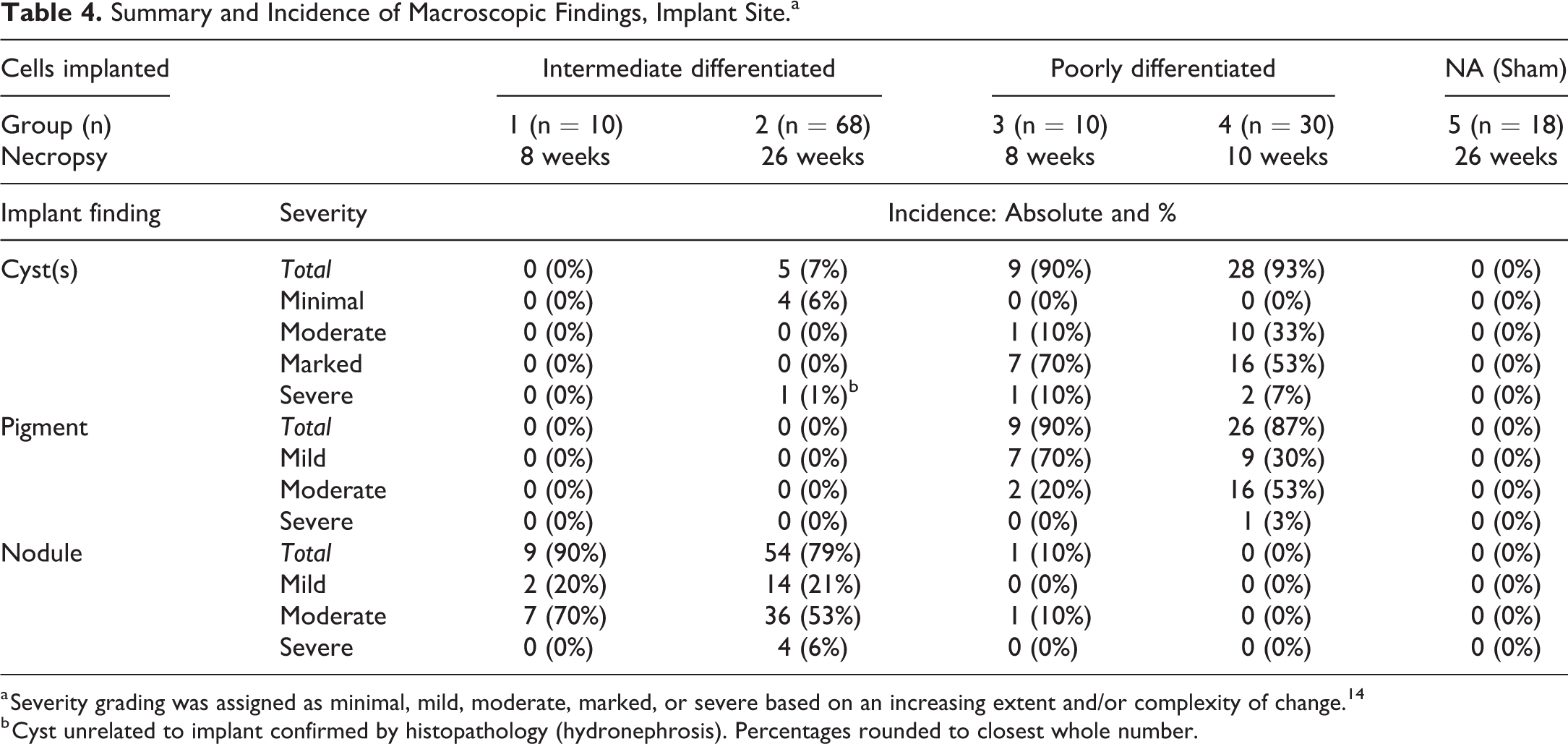
a Severity grading was assigned as minimal, mild, moderate, marked, or severe based on an increasing extent and/or complexity of change. 14
b Cyst unrelated to implant confirmed by histopathology (hydronephrosis). Percentages rounded to closest whole number.
Summary and Incidence of Histopathological Findings, Implant Site.a
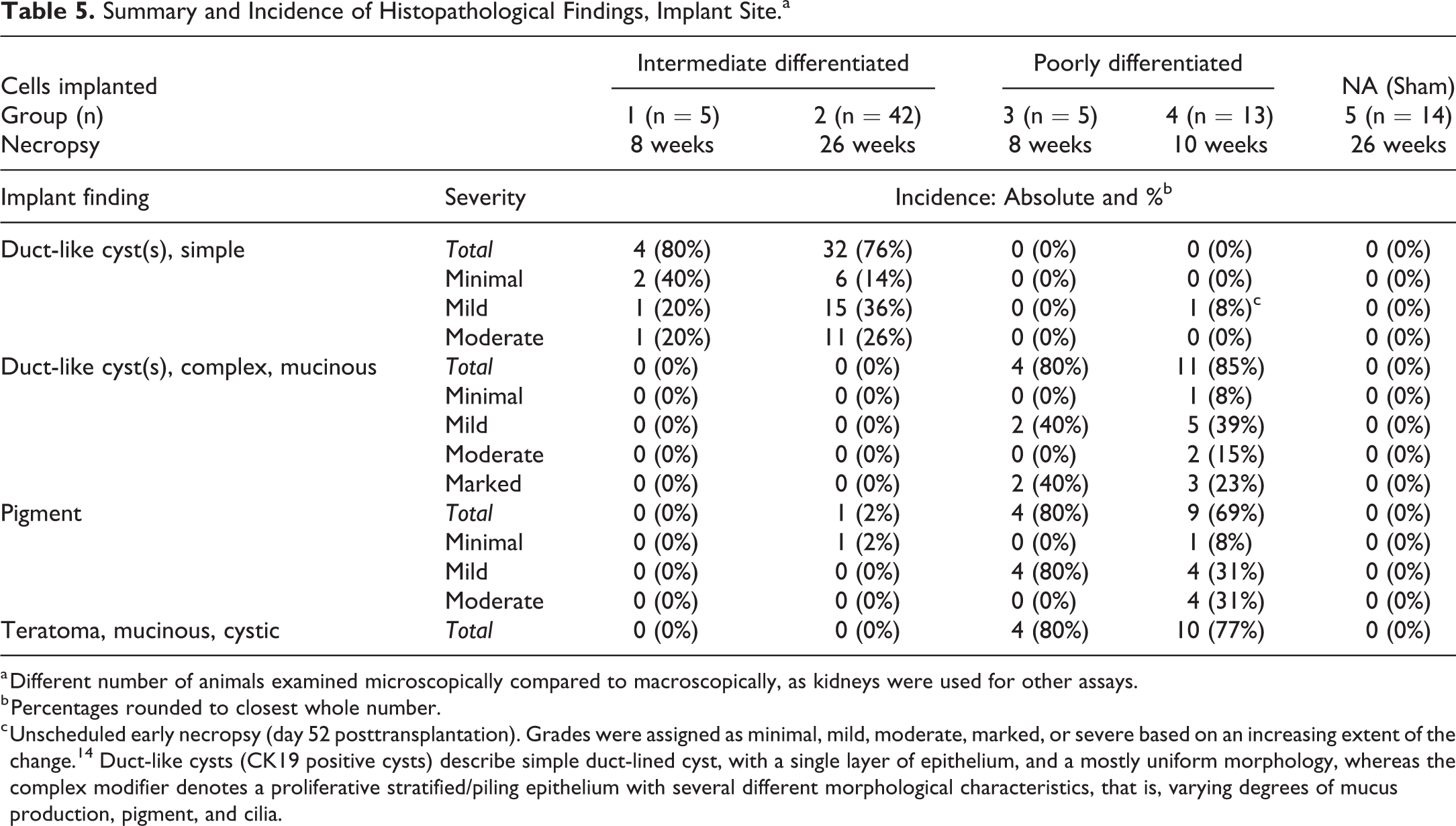
a Different number of animals examined microscopically compared to macroscopically, as kidneys were used for other assays.
b Percentages rounded to closest whole number.
c Unscheduled early necropsy (day 52 posttransplantation). Grades were assigned as minimal, mild, moderate, marked, or severe based on an increasing extent of the change. 14 Duct-like cysts (CK19 positive cysts) describe simple duct-lined cyst, with a single layer of epithelium, and a mostly uniform morphology, whereas the complex modifier denotes a proliferative stratified/piling epithelium with several different morphological characteristics, that is, varying degrees of mucus production, pigment, and cilia.
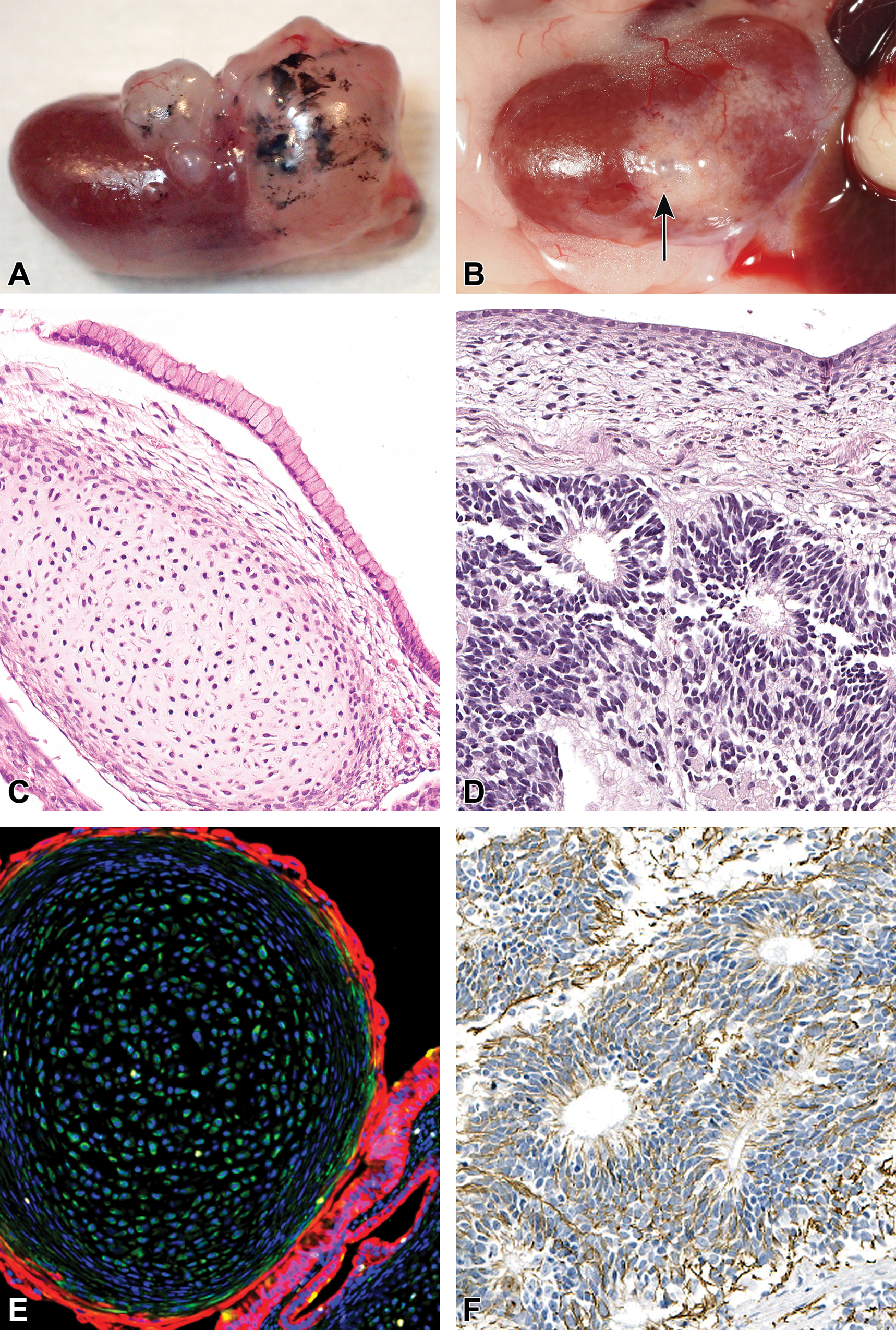
Representative photo of implant & photomicrographs of teratoma characterization in hESC-derived islet implants. A and C-F represents poorly differentiated hESC-derived islet implants and B represent an implant from the intermediate differentiated cell batch. (A) Photo of poorly differentiated hESC-derived islets implanted under the capsule of the kidney 10-weeks post transplantation gave rise to large, expansile cystic tumors with variable amounts of black pigment. (B) Photo of intermediate differentiated hESC-derived islets implanted under the capsule of the kidney, 26-weeks post transplantation. Small cyst present (arrow). (C) H&E stain showing endodermal (mucous secreting ductal epithelium) and mesodermal tissue (cartilage). (D) H&E stain showing neuro-ectoderm like cells forming pseudopalisades and primitive neural tubes. (E) Immunofluorescence stain with ductal, endodermal cytokeratin-19 positive (red) cells and mesenchymal, vimentin positive (green) cells. (F) Nestin positive cells (Brown) characteristic of neuronal tissue. hESC, human embryonic stem cell.
In contrast to the poorly differentiated implants, no tumors originating from the implants were found in the groups transplanted with the intermediate differentiated batch and 60% of the animals implanted with the intermediate differentiated cells, survived until the scheduled terminal necropsy at 26 weeks, which was not significantly different than the sham group survival (Figure S1). The early mortalities in the intermediate differentiated implanted groups were primarily associated with (thymic) lymphoma, a common background finding described in similar SCID mice strains, which were unrelated to the cell xenograft implants. 15,16 The incidence of lymphoma was higher in the sham group (34% vs 44%, respectively; Supplemental Table S3).
Duct-Like Cysts Developed in All Groups But Only With Distinct, High Growth Potential in Poorly Differentiated hESC-Derived Islet Implants
Macroscopically, large and expansile cysts were evident with a high incidence (>90%) and with variable amounts of black pigment in the poorly differentiated hESC-derived islet implants (Figure 2A, Table 4). In contrast, in the intermediate differentiated hESC-derived islet implants (Figure 2B, Table 4), cysts were macroscopically small and only developed rarely in the long-term implanted animals. We did not observe black pigment in the intermediate differentiated batch macroscopically. Hence, intermediate differentiated hESC-derived islet implants demonstrated a much lower tendency to develop macroscopic cysts and did not contain black pigment. When cysts were not present in the implant, the graft was visible as a gray nodule under the kidney capsule.
We evaluated the histological morphology of all implant groups and specifically the cysts with H&E and PAS staining. Microscopically, the cysts had a ductal, nonendocrine morphology that was present in all implant groups (Table 5, Figure 3A-D). These cysts were previously described in an hESC-derived islet implant study as duct-like cysts. 17 Duct-like cysts were morphologically consistent with endoderm-lineage tissue and characterized by CK19 IHC expression consistent with ductal epithelium-forming cystic structures. Importantly, the duct-like cysts showed a distinct complexity and proliferative potential between intermediate and poorly differentiated hESC-derived islets implanted groups (Figure 3A-D and Table 5). Also, there was a very small proportion of tissue with an endocrine morphology present in poorly differentiated compared to the intermediate differentiated (Figure 3C and D).
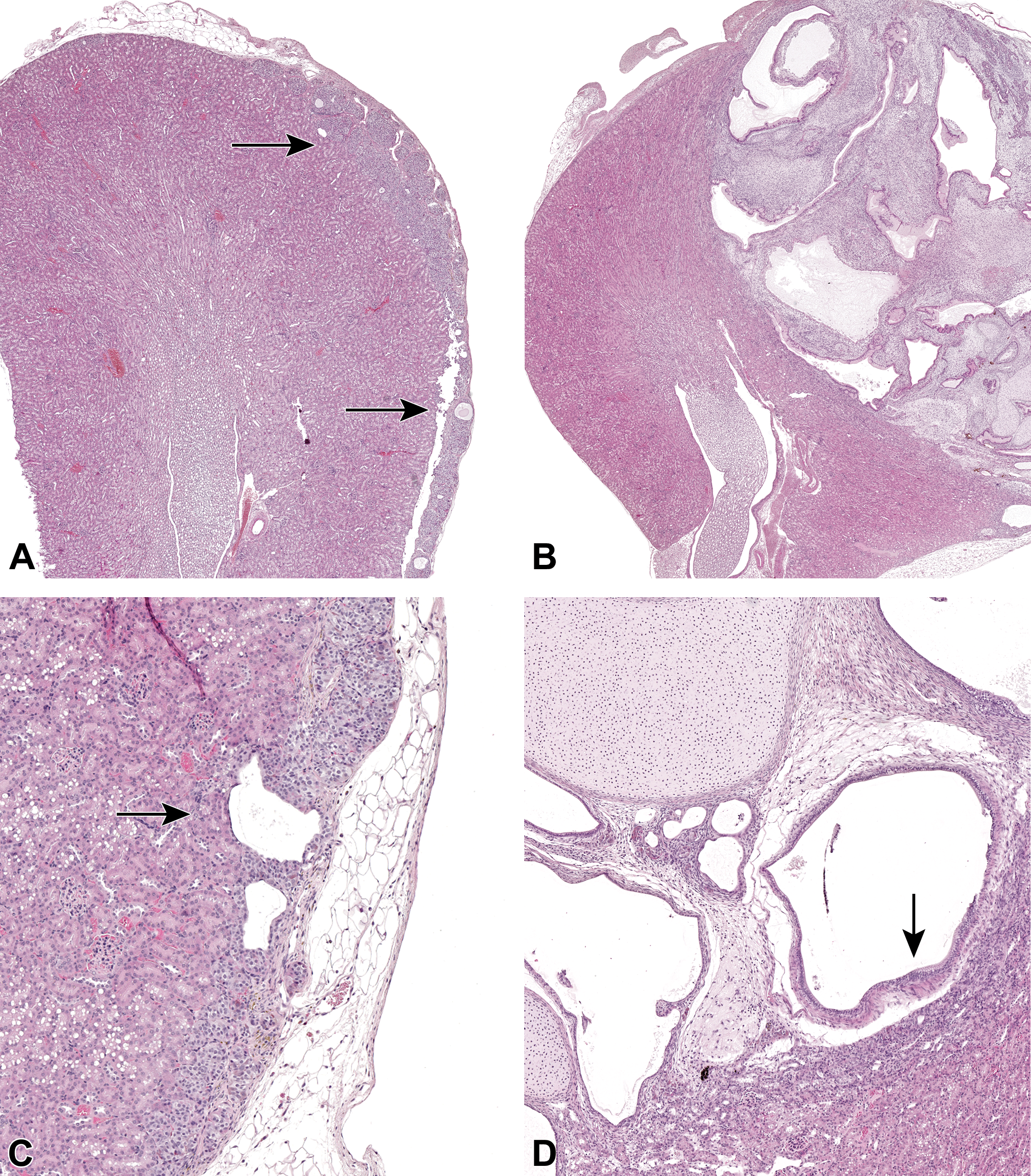
Representative photomicrograph showing subgross and microscopic morphology of the implants. (A, C, E) Intermediate differentiated cell implants. (A) At a subgross level, the implant lies smoothly under the kidney capsule (arrow). (C) The implant is small with simple duct-like cysts (arrow) and most of the implant has an endocrine tissue morphology. (E) Periodic acid–Schiff (PAS) stain shows some mucus containing epithelial cells. (B, D, F) Poorly differentiated cell implants. (B) Large, expansile cystic tumor have effaced part of the kidney. (D) The implants consist of large, complex duct-like cysts, with multilayered epithelium (arrow) with different epithelial types and variable ciliation and no endocrine tissue. (F) Periodic acid–Schiff (PAS) stain confirms a mucinous phenotype of the tall columnar epithelium. A, B, C, D stained with hematoxylin and eosin (H&E).
The duct-like cysts were characterized and classified as simple or complex, based on tissue complexity. In the poorly differentiated hESC-derived islet implants, the duct-like cysts were large, proliferative and with an increased tissue complexity. This complexity included an increased variation in cell types consistent with multiple germ layers (eg, both endoderm and mesoderm) and thus were described as complex duct-like cysts (Figure 3B, D, and F). The incidence of the complex duct-like cysts was 80% at 8 weeks posttransplantation and 85% at 10 weeks posttransplantation (Table 5). In many of these animals, the complex duct-like cysts were partially lined by black-pigmented epithelium or had a pseudostratified to stratified, cuboidal to columnar, variably ciliated, and mucus-secreting epithelium (Figure 3B, D and F, and Figure 4B and D). The high proliferative rate of the complex, duct-like cysts was confirmed with an immunofluorescence stain for Ki-67 and CK19, and potential mesenchymal tissue was identified with vimentin staining (Figure 4F).
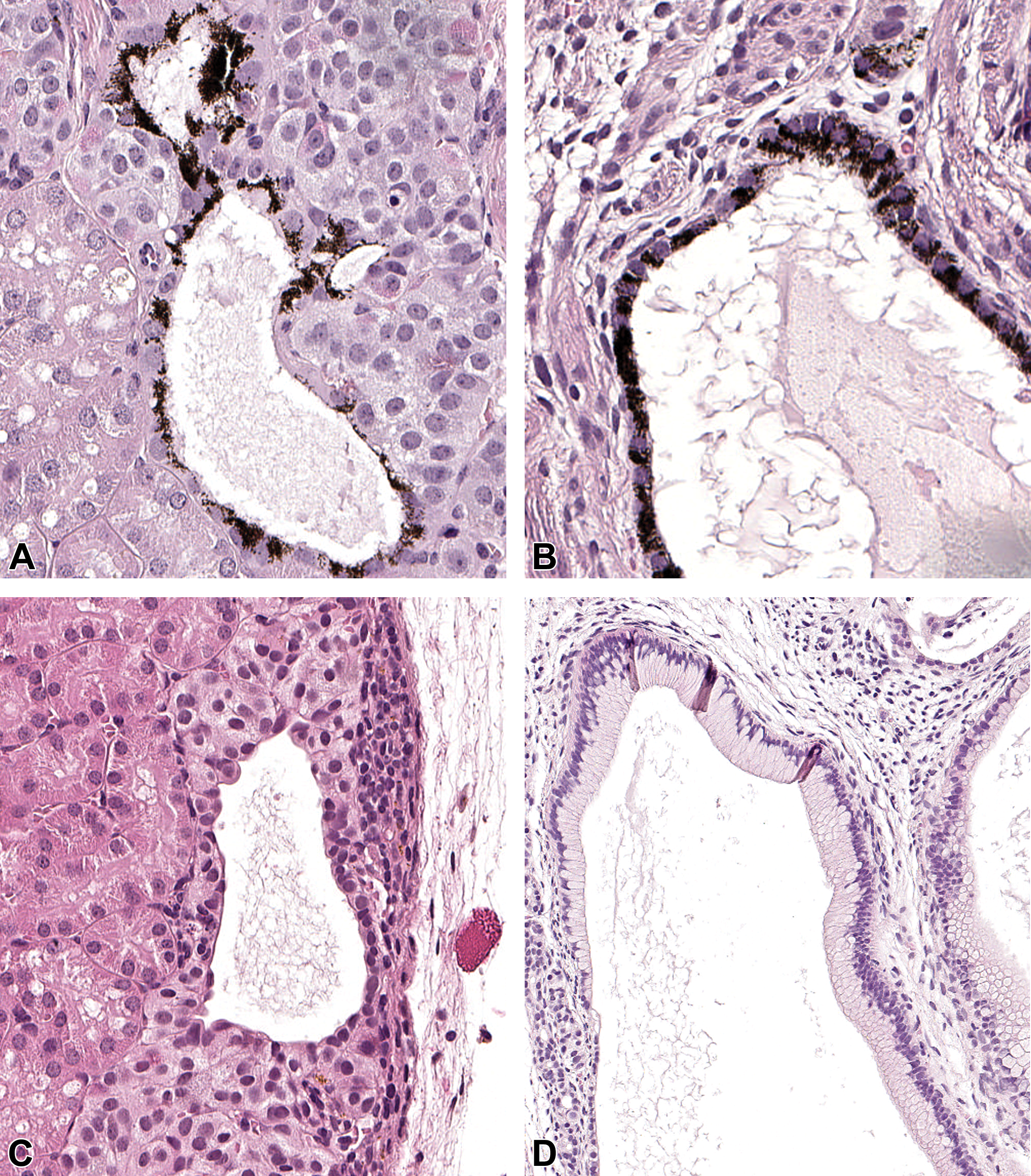
Representative photomicrographs and initial characterization of duct-like cysts. (A, C, E) Intermediate differentiated cell implants. (A) Pigment-laden epithelial cells in duct-like cyst seen in one animal 26 weeks posttransplantation. (C) Uniform, simple duct-like cyst with squamous and variable cuboidal epithelium. (E) Cytokeratin-19 (CK19, red) positive epithelium confirming a ductal phenotype, and very rarely positive-Ki67 (yellow) and vimentin (green) cells, demonstrating minimal proliferation and negligible amounts of mesodermal cells. (B, D, F) Poorly differentiated cell implants. (B) Pigment-laden epithelial cells in duct-like cysts occurred with high incidence. (D) Increased pleomorphism, varying epithelial types, and piling of nuclei in complex duct-like cysts. (F) Ductal epithelial phenotype (cytokeratin-19). Mesodermal tissue is evident shown with vimentin (green, magnified in insert). Many dividing cells are present (yellow, Ki-67). (A, B, C, D) Hematoxylin and eosin (H&E) stain. (E, F) Cytokeratin-19 (red), Ki-67 (yellow), and vimentin (green) immunofluorescence.
We observed only simple duct-like cysts in the intermediate differentiated hESC-derived islet implants, but with a high incidence (>75%) at both 8 weeks and 26 weeks posttransplantation (Table 5). A simple duct-like cyst denotes a small, uniform, nonproliferative duct-lined cyst with a single layer of epithelium (Figure 4C). Rarely, duct-like cysts secreted mucus and in one animal a duct-like cyst was lined by black-pigmented epithelium 26 weeks posttransplantation (Figure 3E and Figure 4A, Table 5). Implants from the intermediate differentiated hESC-derived islets were minimally proliferative and did not contain tissue of mesoderm or ectoderm origin (Figure 4E) (except for the single cyst in one animal noted above with retinal pigmented epithelium-like cells). Thus, it was concluded from the in vitro and ex vivo characterization data that nonendocrine tissue was the origin of the duct-like cysts in the intermediate differentiated hESC-derived islet implants at both 8 and 26 weeks posttransplantation.
Presented results following in vivo transplantation were in line with the in vitro data prior to transplantation (flow cytometry and single-cell RNA-sequencing) that showed a much higher proportion of Ki-67 positive cells (5.1% or 6.4% in flow cytometry and RNA-seq analysis, respectively; Table 3) versus 0.3% in the intermediate differentiated batch (RNA-seq analysis). Also, the large proportion of nonendocrine cells (39.7% CHGA negative, Table 3) in the poorly differentiated batches versus 2.5% nonendocrine cells in the intermediate differentiated groups were, as mentioned, reflected by much less endocrine tissue histologically.
Duct-Like Cysts Contained Nonpancreatic Cells
Because hESC-derived islets are differentiated through endoderm stages and pancreatic lineages, duct-like cysts are assumed to be of pancreatic origin although thorough in situ characterization of these cysts, to our knowledge, has not been done. 17 The initial characterization of the duct-like cysts was to confirm their human origin by using the human nuclei marker KU80 (Figure 5A and B). Next, we evaluated the pancreatic phenotype of the duct-like cyst with the human pancreatic duct and gallbladder-specific IHC marker, CFTR. 18 Furthermore, we evaluated the presence of CA2, an enzyme involved in bicarbonate synthesis, which is secreted by pancreatic ductal epithelium, but also by other duct epithelia (eg, respiratory and gastrointestinal tract). Cystic fibrosis transmembrane conductance regulator was expressed regionally in duct-like cysts in both poorly and intermediate differentiated hESC-derived islet implants (Figure 5C and D). This suggested a regional pancreatic duct or gallbladder epithelium identity. Carbonic anhydrase II showed a similar staining pattern although mostly cuboidal and columnar epithelia were positive (Figure 5E and F). Whereas this suggested that some of the duct-like cysts were of pancreatic phenotype, only parts of the epithelium were positive for CFTR and thus this prompted further investigation. Since part of the gastrointestinal tract also arise from the primitive gut tube posterior to the pancreatic buds, we hypothesized that some of the duct-like structures could be of gastrointestinal origin. To investigate this, we used CDX2 as a marker to distinguish a gastrointestinal tract phenotype (eg, small and large intestine). The ductal epithelium in intermediate differentiated hESC-derived islet implant groups contained a few cells that were CDX2 positive at 26 weeks posttransplantation (Figure 6A). In contrast, the ductal epithelium in the poorly differentiated hESC-derived islet implant groups contained whole segments that were positive for CDX2 at this same time point (Figure 6B). We showed that a high CDX2 expression posttransplantation was associated with low efficiency of PDX1 induction during pancreatic endoderm differentiation, where the PDX1 expression in the poorly differentiated and intermediate differentiated groups was approximately 36% and 84%, respectively (sum of PDX1 expression, Table 2).
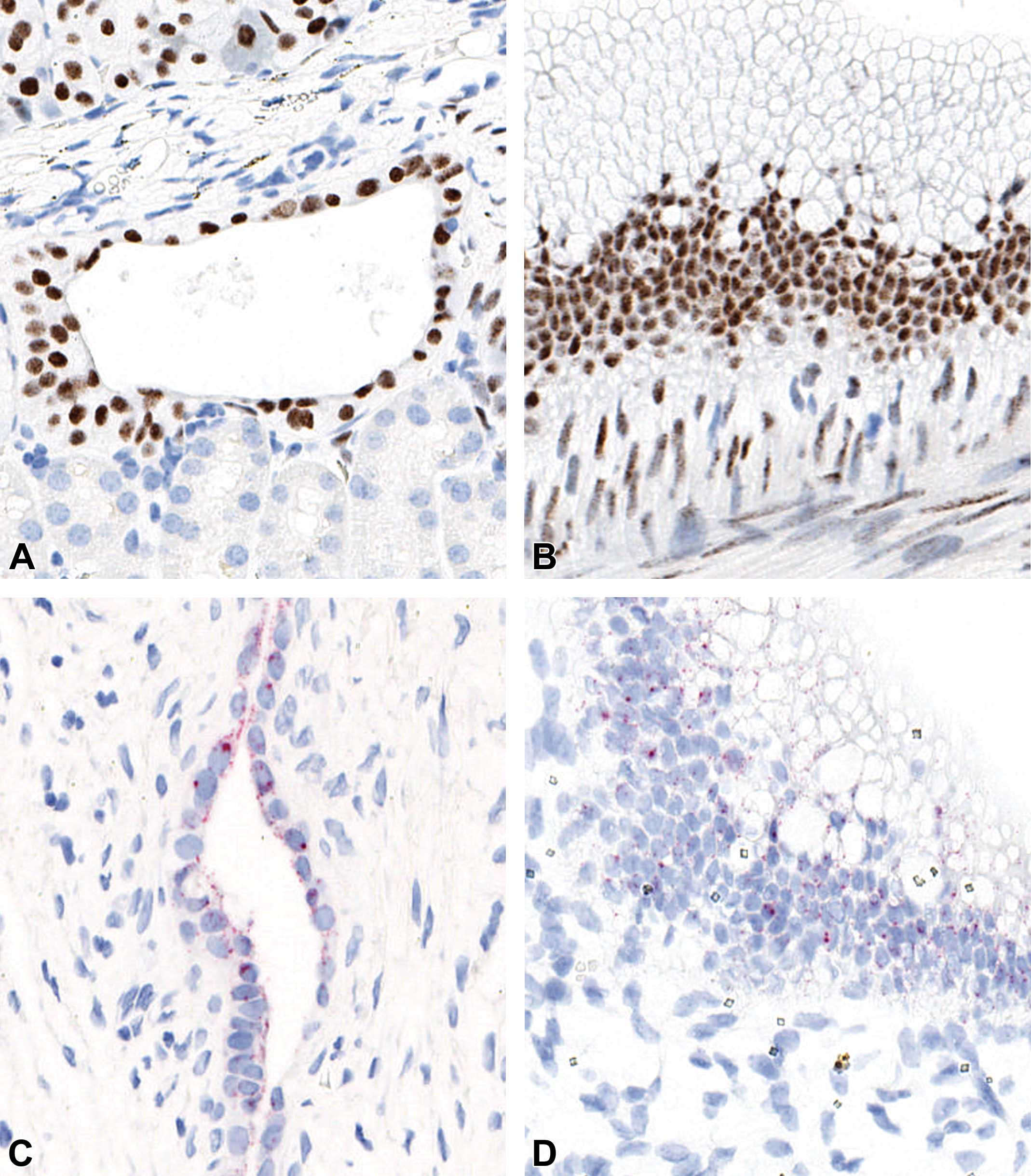
Representative photomicrographs using ISHa and IHCb to confirm human cell nuclei and pancreatic duct phenotype. (A, C, E) Intermediate differentiated cell implants. (A) Implant is of human origin staining positive for KU80. (C) A variable expression of a pancreatic duct marker (CFTR). (E) A bicarbonate-secreting (CA2) phenotype in some regions of the duct-like cysts. (B, D, F) Poorly differentiated cell implants. (B) Implant is of human origin staining positive for KU80. (D) A variable expression of a pancreatic duct marker (CFTR). (F) A bicarbonate-secreting (CA2) phenotype in some regions of the duct-like cysts in both batches. aIn-situ hybridization: C, D. bImmunohistochemistry: A, B, E, F. CA2 indicates carbonic anhydrase 2; CFTR, cystic fibrosis transmembrane conductance regulator.
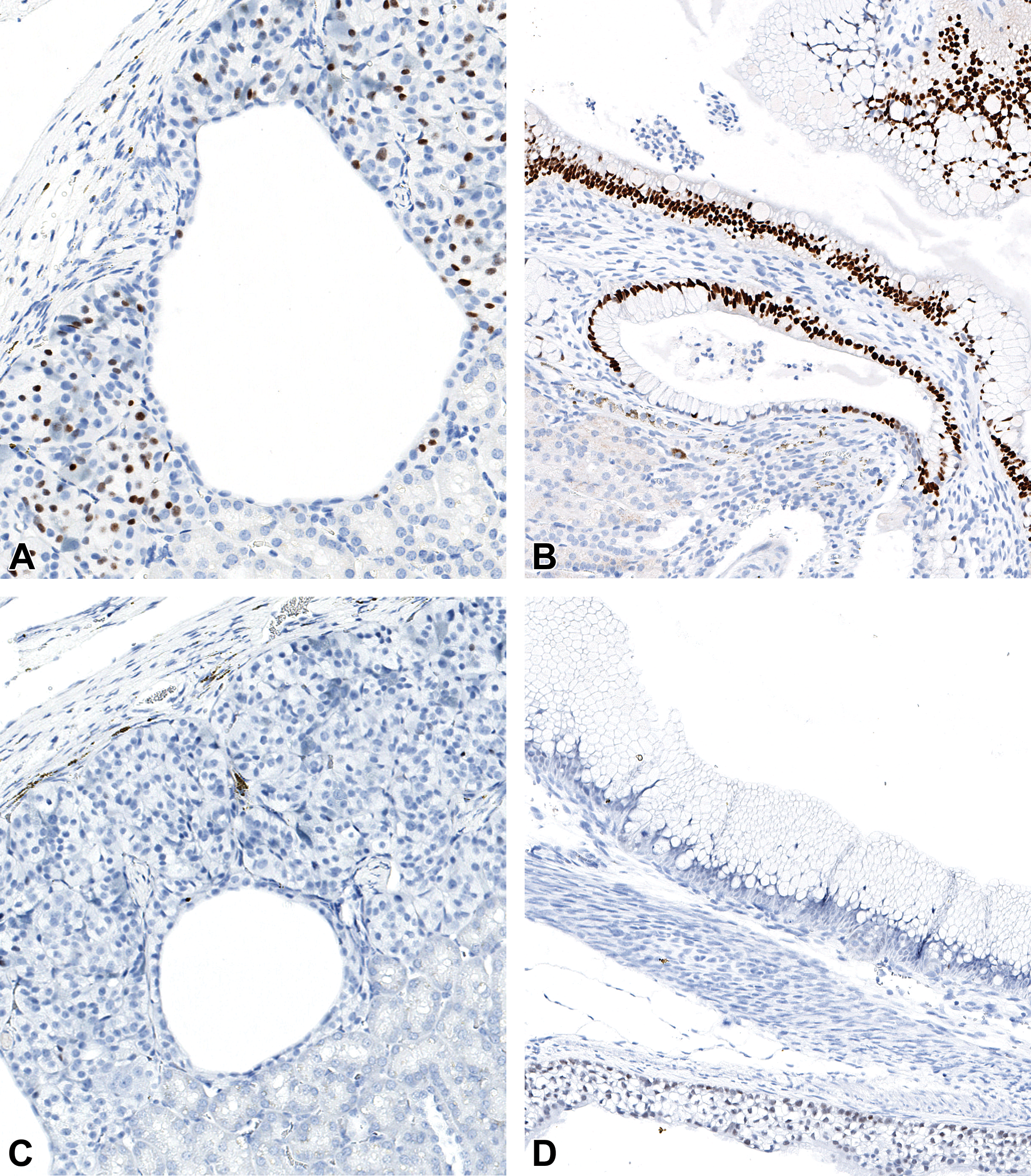
Representative photomicrographs of selected developmental gut markers in duct-like cysts using IHCa. (A and C) Intermediate differentiated cell implants. (A) Rare positive CDX2+ cells suggesting an intestinal phenotype. (B) The epithelium is regionally positive for CDX2. (B and D) Poorly differentiated cell implants. (C) Negative SOX2 stain suggesting an absence of anterior foregut origin. (D) The epithelium is regionally positive for SOX2. aImmunohistochemistry. CDX2, caudal homeobox type 2; SOX2, SRY-Box 2.
The primitive gut tube near the pancreatic buds is the section of the anterior foregut which gives rise to the respiratory tract. We thus hypothesized that the epithelium could also originate from anterior foregut and used SOX2 as a marker to evaluate this question. The intermediate differentiated hESC-derived islet implants with simple duct-like cysts did not contain SOX2 IHC staining at 26 weeks posttransplantation (Figure 6C). However, in the poorly differentiated hESC-derived islet implants, segments of complex duct-like cysts were SOX2 positive (Figure 6D). This highlighted a difference in phenotype between intermediate and poorly differentiated hESC-derived islet implants and suggested an anterior foregut cell phenotype that translated to either the respiratory tract or cranial gastrointestinal tract, that is, esophagus and stomach. 18
Lastly, we characterized the pigmented epithelial cells lining segments of duct-like cysts by histochemistry, IHC, and ISH (Figure 7). This cell population was mainly seen in poorly differentiated hESC-derived islet implants at both 8 and 26 weeks but was also present in a single cyst in one animal in the intermediate differentiated implant 26 weeks after transplantation. This pigment was characterized by Fontana Masson’s stain showing black cytoplasmic granules that, in combination with the H&E morphology, was interpreted to be melanin (Figure 7A and B). We then tested whether the cells had a retinal pigment epithelium phenotype with an antibody against RPE65 and an in-situ hybridization probe for Bestrophin 1. Both markers stained the pigmented epithelium (Figure 7C-F), which strongly suggested a retinal pigment epithelium-like phenotype of these cells which was a finding in hESC-derived islet implants, not reported previously.
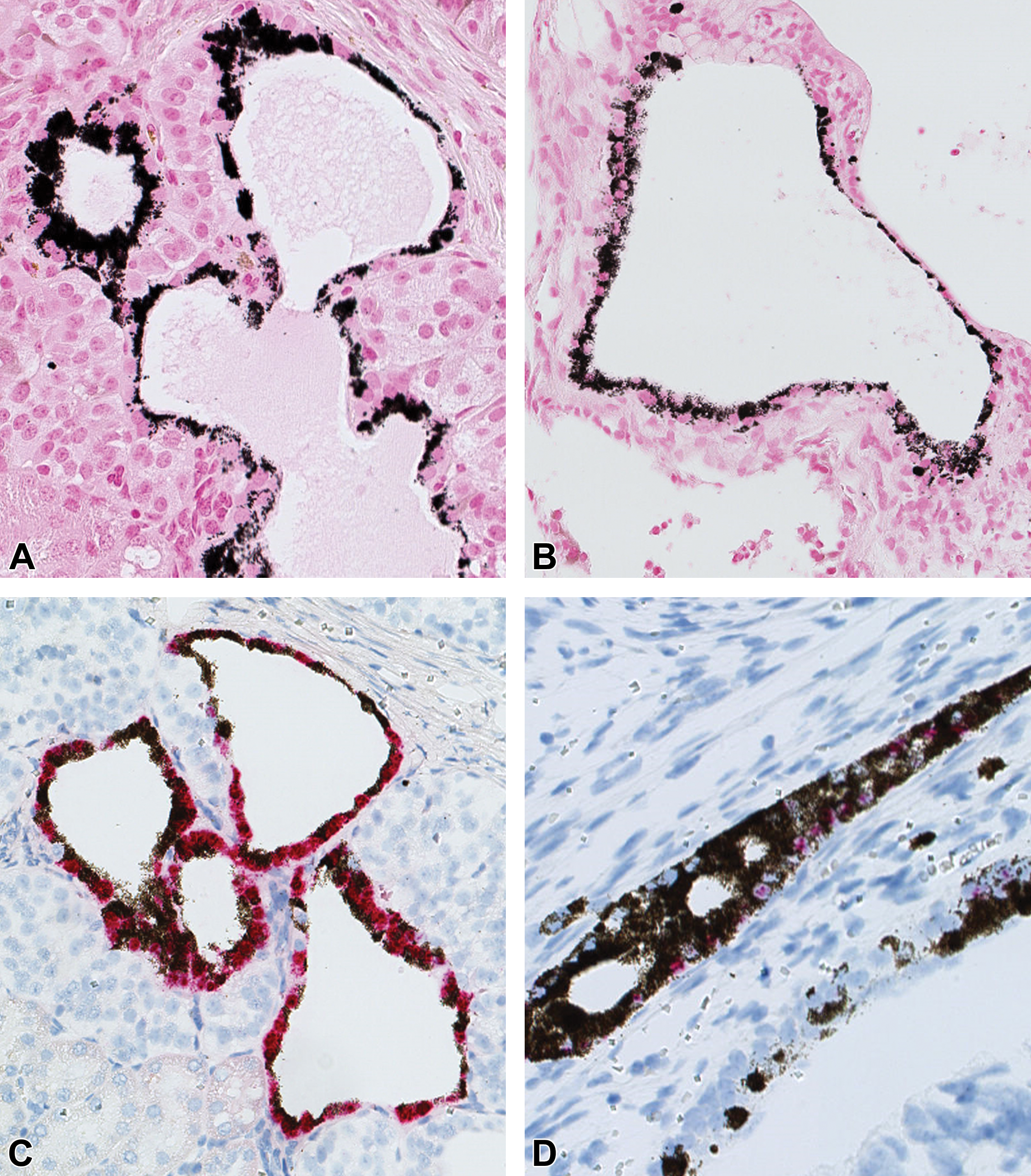
Representative photomicrographs and characterization of pigment-laden ductal epithelium using histochemistry, IHCa, and ISHb. (A, C, E) Intermediate differentiated cell implants. (A) Fontana Masson’s stain suggesting melanin pigment. (C, E) Bestrophin-1 (ISH) and RPE-65 (IHC) positive stain strongly suggesting a retinal pigment epithelium-like phenotype. This occurred in one animal 26 weeks posttransplantation in these groups. (B, D, F) Poorly differentiated cell implants. (B) Fontana Masson’s stain suggesting melanin pigment. (D, F) Bestrophin-1 (ISH) and RPE-65 (IHC) positive stain strongly suggesting a retinal pigment epithelium-like phenotype. This occurred with a high incidence in these groups. aImmunohistochemistry. bIn-situ hybridization. RPE-65, retinal pigment epithelium-specific 65 kDa protein5.
Discussion
Cell-based therapies for type 1 diabetes have a curative potential and are therefore an extremely appealing treatment option. 6 Most publications on this topic focus on the differentiation of functional β cells but partly ignore the codevelopment of nonendocrine cells. Such cells cannot be completely avoided in the differentiation process and it is thus important to characterize these and potentially identify a quantitative threshold where their presence does not create an adverse impact on the host. Therefore, in the present study, we assessed the nonendocrine cells in transplanted hESC-derived islets up to 26 weeks posttransplantation, with a focus on the histogenesis and tumorigenicity of this rarely studied cell population in research-grade hESC-derived islet implants.
The identification of teratomas, which were characterized as expansile masses containing all 3 embryological layers (endoderm, mesoderm, and ectoderm), occurred at a high incidence in animals transplanted with a poorly differentiated batch. This underlines the need for proper safety evaluation of cell therapies involving hESC-derived cells. The lack of tumors in the intermediate differentiated groups however demonstrates that the risk of tumorigenicity can be minimized by proper in-process and pretransplantation characterization. We found that the teratoma-forming cell batch, that is, poorly differentiated hESC-derived islets had a much higher percentage of OCT3/4 positive cells (9.0% vs 1.3%, Table 3) already at the DE stage, as well as a much lower induction of PDX1 (36.4% vs 84.0%, Table 2 [sum of PDX1 expressing cells]), which eventually was also reflected in the proportion of nonendocrine (CHGA−, NKX6.1/C-pep/Glu triple-negative) and proliferative cells (Ki-67) prior to transplantation. Although other investigations have demonstrated tumors in hESC-derived islet implants, 8,9,17 this is the first report to our knowledge that explores the associations between in vitro and ex vivo results for hESC-derived islet implants. Furthermore, this characterization demonstrates that in-process and pretransplantation cell biology data can predict tumor incidence and the identity of the posttransplant impurities, as exemplified by the presence of SOX2- and CDX2-positive cells.
It is generally assumed that teratomas arise from a heterogeneous pool of progenitor cells that originate from residual pluripotent cells in the final product. 19,20 Hence, in the development of cell therapies for the treatment of various diseases, much effort is put into the development of assays that can detect these pluripotent cells with high sensitivity such as reverse transcription–polymerase chain reaction. 21
In this study, we did not perform this type of qualified high-sensitivity pluripotency detection prior to transplantation. Alternatively, we performed SC RNA-Seq prior to transplantation that showed coexpression of the 3 commonly used pluripotency markers (SOX2, NANOG, and POU5F1/OCT4) in the poorly differentiated batch suggesting that the teratomas developed from residual pluripotent stem cells (Figure 1A). Also, a large percentage of the cells analyzed in this batch expressed at least 1 to 2 of the pluripotency markers. In contrast, we did not find any cells coexpressing these 3 markers in the intermediate differentiated batch (Figure 1B), and a low percentage expressed any of the markers, indicating that, in this study, single-cell RNA-sequencing was adequately sensitive for predicting the risk of teratomas caused by residual pluripotent stem cells. Whereas the cellular origin of teratomas might not be essential in the safety evaluation, analyses are essential to ensure they do not arise post graft hESC-derived graft implantation. Looking at the single-cell RNA-Seq data, a clear definition of residual pluripotent cells becomes needed. If pluripotency is defined by coexpression of only 2 markers, then 5 pluripotent cells (0.25% of total analyzed cells) were also present in the intermediate differentiated batch which, as shown, did not cause any teratomas. Two very different conclusions may be drawn from such results, either that 5 pluripotent cells were insufficient to cause teratomas or that the coexpression of 2 of the 3 specified markers are not indicative of a pluripotent cell. This scientific challenge becomes even more difficult to assess when looking at bulk data such as polymerase chain reaction–based methods where coexpression resolution does not exist.
Although we did not observe tumors in the animals transplanted with intermediate differentiated cells, we do report that duct-like cysts may form in hESC-derived islet implants and that these have a heterogeneous phenotype. Cysts have been shown previously by others but have not been phenotypically characterized to the same extent as presented in this study. 17,22,23 These studies report a low proliferation and a benign phenotype compatible with our findings. Most importantly on the translational relevance of these data, we did not find indications of adversity impacting the life span of these animals in the intermediate differentiated islet implants during the 26-week study period.
Cells resembling epithelium seen in the gastrointestinal, respiratory tract, pancreas, and retina were present based on antibody marker panel expression. 18 This confirmed the hypothesis put forward by Pepper et al that not all epithelial cells in the cysts are derived from pancreatic progenitors, but some have a nonpancreatic histogenesis. It is plausible that this heterogeneity in cell origin is a general challenge in the field which is supported by differentiation data from other groups, that used similar differentiation protocols and found, for example, CDX2- and SOX2-positive cells at certain stages during in vitro differentiation. 9,24 Furthermore, this hypothesis is supported as the PDX1-positive pancreatic endoderm, in embryologic development, arises from a CDX2-positive posterior foregut, and thus failure in repressing CDX2 in pancreatic patterning leads to a CDX2 gastrointestinal fate. 25 Similarly, SOX2 patterns the anterior foregut in embryologic development just cranial to the pancreatic bud, but SOX2 is also a stem cell and glial cell marker. 26
The presence of retinal pigmented epithelium in hESC-derived islet implants has not been reported previously, although retinal-like tissue in teratomas has been reported in a few case studies. 27,28 With the absence of teratomas in the intermediate differentiated groups but where retinal pigmented epithelium developed in one animal after 26 weeks, we need an alternative explanation for the finding. The retina is derived from the optic neuroepithelium which develops from the anterior neuroectoderm, 29 whereas hESC-derived islet protocols are optimized for endoderm-lineage pancreatic development and thus a different germ layer. Therefore, the finding is likely caused by incomplete differentiation at the endoderm stage where a subset of cells alternatively committed to an ectoderm fate and subsequently were then irreversibly differentiated in that direction. Interestingly, there is considerable overlap between neuronal cells (neuroectoderm) and β cells both in regard to important developmental transcription factors and physiology which may explain why certain cells commit to a neuroectodermal fate during in vitro differentiation. 25,29- 31 This duct-like cyst with retinal pigmented epithelium in this single animal with intermediate differentiated cells did not affect the outcome of the animal and the cyst was small, uniform, and nonproliferative and therefore considered a nonadverse finding. In addition, we considered whether metaplasia or dedifferentiation upon transplantation could have caused the heterogeneity in the duct-like cysts. However, our in vitro characterizations of the cells (Tables 2 and 3) do not support this hypothesis and thus we found that the most plausible explanation for these nonendocrine structures is impurities in the original transplantation batches.
The marker panels used in this study were based on the Human Protein Atlas resource that contains validated protein expression data from multiple human tissues, including the pancreas. 32 One limitation that is acknowledged is that several markers are expressed in different tissues and are associated with different functions and therefore establishing the cell phenotype and histogenesis is a challenge. This is especially true in the ductal system which is common anatomical components of many organs and where many phenotypic markers are expressed. 11,32 Such challenges are exemplified, as mentioned, by SOX2 being both a marker for anterior foregut, stem cells, and glial cells. Additionally, the markers used to characterize a pancreatic duct histogenesis, such as CFTR and CA2 in this study, are reported in the literature as pancreatic duct markers 33,34 and are seen in several other tissues, for example, ducts in the respiratory system. 35,36 Also, it may not be possible to find an in vivo equivalent to all cell types generated during in vitro differentiation and the marker expression may therefore be difficult to relate to in vivo cell types. It is recognized that the sample size for the immunohistochemical analysis was small (n = 3 for the long-term transplanted intermediate differentiated group and n = 3 for each poorly differentiated groups) and that increased sample size could potentially have identified other phenotypes and revealed variations and outliers which would not be identified with this low sample size. Despite these limitations, we demonstrate that nonpancreatic tissue develops in duct-like cysts from hESC-derived islet implants, which have not been reported previously.
This study serves as a valuable resource for future studies in the hESC-derived islet field, demonstrating that nonendocrine cell populations before and after transplantation will benefit from a more thorough characterization than previously performed. We provide evidence on the value of evaluating common nonendocrine phenotypes seen after transplantation of hESC-derived islets. Additionally, we demonstrate value in associating in process in vitro data with ex vivo characterization data and the importance of in-process quality controls. Future research that assesses the histogenesis and cell phenotype of nonendocrine cells in even greater detail is warranted and may benefit the field by providing important phenotypic information to be used in the differentiation process and safety assessment of the implants.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211036395 - Characterization of the Nonendocrine Cell Populations in Human Embryonic Stem Cell–Derived (hESC) Islet-Like Clusters Posttransplantation
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211036395 for Characterization of the Nonendocrine Cell Populations in Human Embryonic Stem Cell–Derived (hESC) Islet-Like Clusters Posttransplantation by Nikolai K. Jensen, Camilla Ingvorsen, Dorthe R. Petersen, Maria J. Pereira, Tess T. H. Lu, Thomas J. Alsted, Jeannette S. Kirkegaard and Kevin A. Keane in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-docx-2-tpx-10.1177_01926233211036395 - Characterization of the Nonendocrine Cell Populations in Human Embryonic Stem Cell–Derived (hESC) Islet-Like Clusters Posttransplantation
Supplemental Material, sj-docx-2-tpx-10.1177_01926233211036395 for Characterization of the Nonendocrine Cell Populations in Human Embryonic Stem Cell–Derived (hESC) Islet-Like Clusters Posttransplantation by Nikolai K. Jensen, Camilla Ingvorsen, Dorthe R. Petersen, Maria J. Pereira, Tess T. H. Lu, Thomas J. Alsted, Jeannette S. Kirkegaard and Kevin A. Keane in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-tif-1-tpx-10.1177_01926233211036395 - Characterization of the Nonendocrine Cell Populations in Human Embryonic Stem Cell–Derived (hESC) Islet-Like Clusters Posttransplantation
Supplemental Material, sj-tif-1-tpx-10.1177_01926233211036395 for Characterization of the Nonendocrine Cell Populations in Human Embryonic Stem Cell–Derived (hESC) Islet-Like Clusters Posttransplantation by Nikolai K. Jensen, Camilla Ingvorsen, Dorthe R. Petersen, Maria J. Pereira, Tess T. H. Lu, Thomas J. Alsted, Jeannette S. Kirkegaard and Kevin A. Keane in Toxicologic Pathology
Footnotes
Acknowledgments
The authors would like to acknowledge and thank the following colleagues: Mie Berndorff, Nicolai Hansen, Caroline B. Kvist, Hanne N. Søndregaard, and Ann-Charlott Kemp for conducting the surgical and in-life animal work. Jonas Ahnfelt-Rønne and Rikke K. Kirk for immunohistochemistry and In-situ Hybridization protocols and expertise. Susanne J. Rasmussen and Pia G. Mortensen for excellent histopathological laboratory work. Lina Thorén for performing and providing flow cytometry data and Elham Jaberi for additional SC RNA-Seq data generation. The authors thank Helene Jacobsen, Mattias Hansson, and Namiyo Miyashita for their scientific and editorial input to the manuscript. This work was submitted by the lead author (Nikolai K. Jensen) in part for the completion of a Master of Science Degree from University of Copenhagen, awarded in March 2020. The authors would like to thank Professor Poul Hyttel of the University of Copenhagen for serving as the academic supervisor for this project.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: N.J., C.I., D.P., M.P., T.A., J.K., and K.K. are employed by Novo Nordisk A/S and holds shares in the company.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by Novo Nordisk A/S, Bagsvaerd, Denmark.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
