Abstract
Recent advances in human pluripotent stem cell (hPSC)-derived cell therapies and genome editing technologies such as CRISPR/Cas9 make regenerative medicines promising for curing diseases previously thought to be incurable. However, the possibility of off-target effects during genome editing and the nature of hPSCs, which can differentiate into any cell type and infinitely proliferate, inevitably raises concerns about tumorigenicity. Tumorigenicity acts as a major obstacle to the application of hPSC-derived and gene therapy products in clinical practice. Thus, regulatory authorities demand mandatory tumorigenicity testing as a key pre-clinical safety step for the products. In the tumorigenicity testing, regulatory guidelines request to include human cancer cell line injected positive control group (PC) animals, which must form tumors. As the validity of the whole test is determined by the tumor-forming rates (typically above 90%) of PC animals, establishing the stable tumorigenic condition of PC animals is critical for successful testing. We conducted several studies to establish the proper positive control conditions, including dose, administration routes, and the selection of cell lines, in compliance with Good Laboratory Practice (GLP) regulations and/or guidelines, which are essential for pre-clinical safety tests of therapeutic materials. We expect that our findings provide insights and practical information to create a successful tumorigenicity test and its guidelines.
Keywords
Introduction
Human pluripotent stem cells (hPSCs) can infinitely proliferate and differentiate into all three germ layer cells. 1 These properties make hPSCs promising for cell therapies, especially for degenerative diseases. However, the strong advantages of hPSCs could be a double-edged sword, as even a small amount of hPSCs may result in tumors.1-3 In addition to hPSC-derived cell products, gene therapies also raise concerns about tumorigenicity. Carriers such as retroviral vectors or genetic materials administered could cause genome disruption by retroviral integration, off-target effects, or homologous recombination by sequence similarity to the target genome. 4 Additionally, hPSC cell therapies and genome editing have been efficiently combined with ex vivo gene therapies. 5 Given the possible multi-factor etiology of tumorigenicity of gene therapies and hPSC-derived cell products, and an increasing number of such products in clinics, elucidation of tumorigenicity assessment methods and reach consensus worldwide is necessary. 6
Regulatory bodies worldwide, such as the European Medicines Agency (EMA),7-10 US Food and Drug Administration (FDA), 11 World Health Organization (WHO), 12 and the Korea Ministry of Food and Drug Safety (MFDS), 13 have introduced guidelines to support the development of therapeutic cell products intended for human use. There are concerns regarding the use of animal models to test the tumorigenicity of human-derived cells6,14; however, many regulatory bodies still require mandatory animal testing, given lack of a better alternative.11,13 For pre-clinical in vivo tumorigenicity tests, several factors must be considered, including animal model selection, proper controls, number of testing and control animals required to decide whether a tumorigenic event is unlikely to occur, limit of cells to be inoculated, study duration, site of administration. Currently, there is no global consensus regarding the above questions. 6
For tumorigenicity assessment, immunocompromised animals such as athymic nude mice or severely combined immunodeficient mice (SCID) are recommended. 12 As positive controls, human-derived cancer cell lines were used.12,15 The selection of a proper positive control is critical for a successful tumorigenicity test, as the validity of the test is determined by the tumorigenic ratio of positive controls in some guidelines. For example, WHO TRS 978 suggested that at least 9 of 10 positive reference animals should develop a tumor for a valid test. 12 Similarly, the MFDS guidelines recommend a tumor formation rate of >90% for positive controls. 13 Furthermore, the selection of a proper administration route for testing materials and positive control cells could be an issue. Positive control cells are often inoculated subcutaneously (SC) because of the ease of tumor detection. However, testing cell products is typically inoculated into animals at the clinical route intended for patients, given the importance of the tumor microenvironment. 11 In this regard, if the administration route of testing material is not identical to positive cancer cell lines (typically SC), this may cause problems in assessing the validity of the tests.
Nonclinical studies intended to support clinical trials should be conducted according to the Good Laboratory Practice (GLP) regulations or guidelines. GLP is a quality system designed to ensure that studies are performed through a validated and standardized protocol to produce reproducible, reliable, and traceable data sets.16-20 The GLP regulations and guidelines strongly recommend commitment to defined protocols throughout the study. Changing protocol and fixing deviation are allowed during the study, but the history of changes and the process should be clear and traceable. In this regard, well-defined control conditions are critical for conducting a successful GLP study. Otherwise, multiple pre-GLP studies demanding many animals, time, and money, are required.
Recently, we conducted several tumorigenicity tests in compliance with the MFDS, FDA GLP regulations and Organization for Economic Cooperation and Development (OECD) GLP guidelines.17,18,20 We also performed pre-GLP studies to optimize the experimental conditions for each study. These studies provided data on the tumorigenic ability of several human cancer cell lines (U-87MG, HCT116, and HeLa). These cell lines were administered through multiple routes, including intrathecal (IT), intracerebroventricular (ICV), Achilles tendon, subcutaneous (SC), and intravenous (IV) injections. Here, we share the historical data we gathered from those studies and describe the fixed conditions we are currently using as positive controls of tumorigenicity tests to conduct a successful tumorigenicity test.
Materials and Methods
Good Laboratory Practice Compliance
All studies starting with Gx were conducted following MFDS GLP regulations 17 and OECD GLP guidelines. 18 Study Gx166 followed additional FDA GLP regulations. 20 All studies starting with Nx were conducted using the same standards of Gx studies, however, no final quality assurance reports were issued.
Animal Welfare
Specific pathogen-free (SPF) BALB/c nude mice aged 4 to 8 weeks were purchased from Orient Bio Inc. (Gyeonggi, Korea) or Charles River Laboratories Japan Inc. (Yokohama, Japan). SPF NSG mice aged 5 weeks were purchased from Charles River Laboratories (USA). The mice were quarantined and acclimated for 6 to 19 days, depending on the study. During all studies, mice were maintained in a room at 23 ± 3°C, 30-70% relative humidity, 12 hours light/dark cycle, 150-300 lx, and 10-20 cycles per hour ventilation. The animals were housed in polycarbonate or polysulfone cages with appropriate animal densities (1-5 animals depending on cage size). Animals showing aggression or abnormal behavior during the study were housed individually. A standard mouse pellet diet (Lab Diet® #5053 PMI Nutrition International; gamma-ray irradiation) and filtered, ultraviolet light-irradiated municipal tap water were provided to the animals ad libitum. All studies were conducted at the Korea Institute of Toxicology (KIT) and approved by the Association for Assessment and Accreditation of Laboratory Animal Care International. All studies were reviewed and approved by the Institutional Animal Care and Use Committee of KIT.
Cell Culture
U-87MG (ATCC® HTB-14TM) cell line was purchased from ATCC and maintained in KIT. U-87MG cells were cultured in MEM media (Gibco) supplemented with 2.2 g/L of sodium bicarbonate, 2 mM L-glutamine, 100 units/mL of penicillin G sodium, 100 μg/mL of streptomycin sulfate, and 10% (v/v) fetal bovine serum (FBS) (Gibco) in a 37°C incubator with 5% CO2. HCT116 cell line was purchased from Korean Cell Line Bank (Seoul, Korea) and maintained in KIT. HCT116 cells were cultured in RPMI 1640 medium supplemented with 10% (v/v) FBS.
For the preparation of the positive control material, cells were amplified by subculturing until the desired cell numbers were established. On the day of administration, the cells were washed once with Dulbecco’s PBS (DPBS) and detached from culture dishes by trypsin solution (Gibco) treatment. After two more washes with DPBS to remove FBS in the cell suspension, appropriate media without FBS were added. Cell numbers were counted using a Vi-CellTM (Beckman Coulter) or a hematocytometer. Cells were used for administration only when the cell viability exceeded 80%. After dosing, a mycoplasma test was performed using an e-MycoTM VALiD Mycoplasma PCR detection kit (Lilif Diagnostics, Korea) according to the manufacturer’s protocol.
Clinical Observation and Body Weight Measurement
All animals were observed 1-2 times daily for mortality, moribundity, general appearance, behavioral changes and tumor tracking. Tumor size was calculated using the formula: Volume (mm3) = 1/2 × length (mm) × [width(mm)]. 2 Tumor width and length were measured externally using Vernier calipers once per week. Body weights were recorded at least once per week. All results and raw data were recorded using the Pristima System (Xybion).
Intrathecal Administration
The injection site was shaved and disinfected with 70% alcohol swabs before administration. Animals were anesthetized by isoflurane inhalation, and fixed with a U-frame stereotaxic apparatus (75-1808, Harvard Apparatus). After skin incision at the injection site, 15 μL per head U-87MG cells were intrathecally injected at 1 μL/min using a Hamilton syringe (80030-1701RN 10ul SYR 26s/2"/2, Hamilton). The syringe was maintained for an additional 5 min after administration to prevent backward flow. The incision site was sutured. Animals recovered from anesthesia on a heating pad.
Intracerebroventricular Administration
Following induction of anesthesia, the animals were placed in a stereotaxic frame. A 10 μL Hamilton syringe was attached to the micromanipulator of the stereotaxic device and filled with 6-10 μL of the injection solution. Approximately 1 cm midline incision was made in the scalp and the periosteal membranes were retracted from the skull with sterile cotton-tipped applicators. Bregma (the intersection of the midsagittal and coronal sutures) was identified, and the tip of the needle was positioned there. The needle was moved to the desired AP, ML position from the bregma. 21 The DV manipulator was used to drive the needle tip through the skull at this location. Once the lumen of the needle had passed through the surface of the skull, the needle was advanced to the desired DV location into the lateral ventricle. The U-87MG cells were injected using an infusion pump (70-4507, Harvard Apparatus), and the needle was allowed to remain in place for 1-5 min. The needle was slowly withdrawn from the skull, and the scalp was closed with sutures. The animals were allowed to recover from anesthesia in their home cage.
Achilles Tendon Administration
Animals were anesthetized by 2.5% isoflurane inhalation. Mouse hind leg fur was shaved and then disinfected with 70% ethanol or povidone-iodine swab. The upper side of the Achilles tendon was held with forceps, and the tendon region was incised to less than 10 mm using mosquito forcep. Twenty microliters of cells were injected at the center of the Achilles tendon using an insulin syringe, and the syringe was kept for an additional 10 seconds to ensure cell absorption. After narrowing the perforation site by pulling the ankle, the incision was sutured using 6-0 silk.
Macroscopic and Microscopic Examination
All animals that survived until the scheduled necropsy date of each study were euthanized by isoflurane inhalation. Necropsy examinations were performed on dead, accidentally dead, moribund sacrifice, and scheduled sacrificed animals during the treatment period, if not specified in each study. After exsanguination from the vena cava and aorta, all animals were examined for external abnormalities. The abdominal, thoracic, and cranial cavities were examined for abnormalities, and organs and tissues were removed for fixation using 10% (v/v) neutral buffered formalin. Lungs were fixed using formalin infusion via the trachea. Tissues were fixed for 48 hours and then transferred to 70% ethanol. Fixed tissues were embedded in paraffin wax, sectioned (4 μm), stained with hematoxylin and eosin (H&E), and examined under a microscope.
Statistical Analysis
Group body weight results are shown as mean ± standard deviation (SD). Statistical analysis of body weight was performed by comparing PC animals with the vehicle control group (VC) animals, unless otherwise noted, using the Pristima system or GraphPad Prism 8 (GraphPad Software). Homogeneous data were analyzed by ANOVA, and the significance of inter-group differences was calculated using Dunnett’s test. Heterogeneous data were analyzed using the Kruskal-Wallis test, and the significance of the differences between the PC and VC was evaluated using Dunn’s rank-sum test. P-values < 0.05 were considered significant.
Data Availability
The data generated in this study are available upon request from the corresponding author.
Results
U-87MG Cell Intrathecal and Intracerebroventricular Injection
IT injection of hPSC-derived cell products has been considered as a clinical route of administration for patients with spinal cord injury.22-24 In this regard, we attempted to establish a U-87MG cell IT injection into athymic nude mice as a positive control. We selected the U-87MG cell as the cell originating from glioma. 25 For study Nx019, 1 × 104 (T1), 5 × 104 (T2), and 1 × 105 U-87MG cells (T3) were administered through IT injection to 20 mice (10/10 male/female) per group. Including five animals that died on day 1, 11 animals died before day 5. Most mice that died during this period showed irregular respiration and bloody liquid ran from the nostril (data not shown). After these early deaths, the remaining animals started to die on day 19, and 46 animals died before day 56 (Supplementary Figure 1A). Three remaining animals were sacrificed on day 78. Among 49 animals who died after day 19, 44 animals developed nodules or masses around the medulla, cerebellum, olfactory tract, or hypothalamus (Supplementary Figure 1B). Interestingly, 11 animals that died before day 5 did not show any nodule or mass; instead, the animals showed dark red lung discoloration or edema (Supplementary Figure 1B). Previous research indicates that pressures on the central nervous system (CNS) including the medulla or spinal cord, can cause acute neurogenic pulmonary edema. 26 Our results imply that the massive death within a few days of treatment was related to the administration route, rather than U-87MG treatment.
The ICV injection route can be considered an alternative route to deliver drugs to the CNS.
27
We conducted three GLP studies (Gx016, Gx019, and Gx123) using U-87MG cells as a positive control, which were administered via ICV injection; 3 × 104 U-87MG cells were injected into 24 (Gx016, 12/12 male/female) or 26 (Gx019 and Gx123, 13/13 male/female for each study) mice. One weakness of ICV injection is that tumors are unlikely to be detected by simple observation. As the tumorigenicity of PC animals should be assessed by necropsy and histopathologic diagnosis, it is critical to estimate the date when the majority of animals form tumors. In this regard, we tracked clinical signs (i.e., body weight and survival rate) and compared the data with the final necropsy results. First, we checked the viability of the animals throughout the study (Figure 1A-C). In study Gx016, five (3 male/2 female) animals died between days 26 and 34, and scheduled necropsies were conducted on day 37 for all remaining animals. In two other studies, necropsies were conducted for moribund or dead animal(s). In these two studies, approximately half of the animals died around day 60 (Figure 1B and C). For study Gx019, 11 of 12 males died by day 127 (One male body was lost due to cannibalism) and 12 of 13 females died by day 95 (Figure 1B). For study Gx123, all males died by day 130, and 11 of 13 females died by day 176 (Figure 1C). For all three studies, VC animals did not show reduced viability compared to PC animals (Figure 1A-C, VC), suggesting that ICV injection did not affect the viability of animals. Compared to VC, several PC animals lost significantly more body weight (Supplementary Figure 2). In Gx016, both male and female mice displayed body weight loss on day 29 (Supplementary Figure 2A). In Gx019, the female group displayed body weight loss at day 43, while the male group did not lose significant body weight until day 50 (Supplementary Figure 2B). However, we noticed that the initial mean body weight (on day 1) of males in PC was higher than in VC. This may mask the body weight loss of the PC at later stages. In Gx123, male PCs showed body weight loss from day 50, but no body weight loss was detected among female PCs (Supplementary Figure 2C). This may be due to the drastic increase in death rate around day 50 in the female PCs in this study, as dead animals were not counted in the mean body weight calculations. U-87MG ICV injection. Survival rates of male (upper panel) and female (lower panel) nude mice after vehicle (VC) or 3×104 U-87MG cell (PC) ICV injection in study Gx016 (A), Gx019 (B), and Gx123 (C). 24 nude mice (12 male/12 female) were used for each group in Gx016. 26 (13 male/13 female) mice were used for each group in Gx019 and Gx123, respectively. (D) Representative H&E staining images of brain neoplasms of PC animals in Gx016. A VC animal brain image was used as a control. All scale bars represent 500 μm. (E) Tumor formation rates of PC animals in studies Gx016, Gx019, and Gx123. Tumor formation was determined by the observation during necropsy and the histopathologic staining of tissues. In study Gx019, one male body was lost by cannibalism during the study, thus excluded from the count. (F) Tumor detection dates of all tumor formed PC animals used in studies Gx016, Gx019, and Gx123. Abbreviations: PC, positive control group; VC, vehicle control group.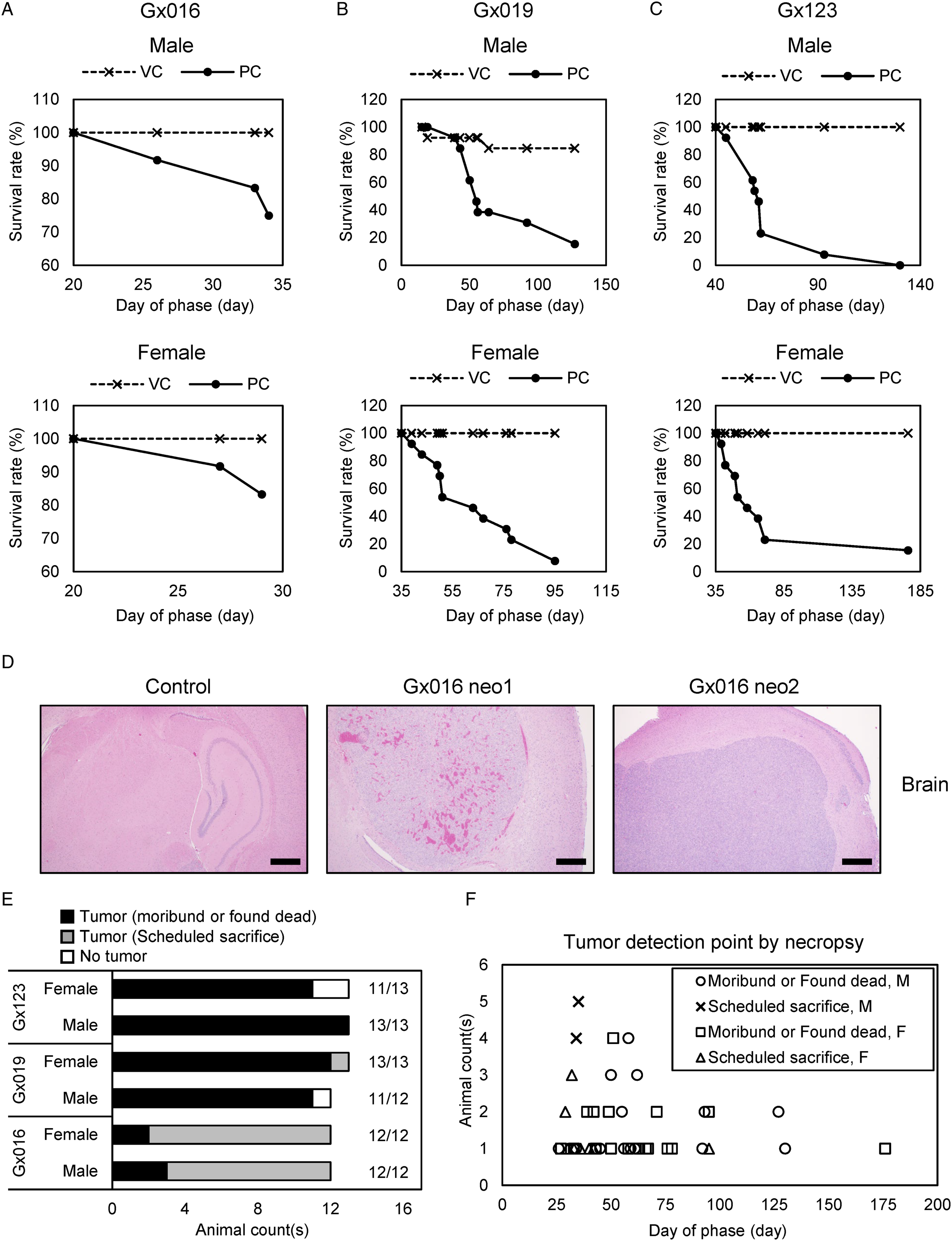
Necropsy revealed that 95% of males (35/37) and females (36/38) developed tumors around the injection site (Figure 1D and E). This rate is high enough to meet the 90% tumorigenicity requirement of the PC animal, as suggested by the WHO and MFDS.12,13 Interestingly, all 24 animals developed tumors within day 37 in Gx016 (scheduled sacrifice date) and both male and female mice showed reduced body weight on day 29 (Supplementary Figure 2A). In the other two studies, only one animal died before day 37. Gx019 PC females and Gx123 PC males showed body weight loss between days 43 and 50 (Supplementary Figure 2B and C), and a drastic reduction in survival rate occurred around this time (Figure 1B and C). Approximately 91% of males (32/35) and 97% of females (35/36) who formed tumors died before day 100 (Figure 1F). Collectively, our results indicate that ICV injection can be considered an administration route for cancer cells as well as testing material in animal studies. In addition, when the 3×104 U-87MG cells were injected, >90% tumorigenicity was achieved regardless of animal sex. Body weight loss is not definitive but can be considered an important clinical sign to decide the necropsy date, especially for males. A few days after the detection of mean body weight loss in male or female mice can be considered the earliest necropsy date possible, although the majority of animals survived. Alternatively, as all moribund or found dead animals in PC developed tumors, based on survival rate, a day about 50% to 90% animals died can be a more conservative option.
HCT116 Cell Achilles Tendon Injection
Tendons are dense fibrous connective tissues that connect muscle to the bone and require a long time to reacquire sufficient strength after injury due to their poor regenerative potential.28,29 In this regard, hPSC-derived cell products have been considered regenerative medication to cure tendon injuries and tendinopathies.30,31 We conducted a pioneering experiment (Nx053) using NOD/SCID/IL-2rγKO (NSG) mice 32 to establish Achilles tendon injection in three cancer cell lines (U-87MG, HCT116, and HeLa) to determine the tumorigenicity of each cell line. HeLa cells have been suggested as good positive control cells for SC injection, 12 and HCT116 cells, which originate from human colon cancer, are known to be highly tumorigenic when implanted into mice via SC injection. 33 For all three cell lines, 2 × 106 cells were injected via the Achilles tendon to five females per group. In this experiment, the HCT116 and U-87MG cell-treated groups lost significant body weight from day 29 for HCT116, from day 43 for U-87MG compared to HeLa treated group (Supplementary Figure 3A). All HCT116 and U-87MG treated animals developed visible masses around the injection site from day 35, while the HeLa treated group did not (Supplementary Figure 3B). Three animals from the U-87MG treated group died on day 43, so we conducted necropsies for all remaining U-87MG and HCT116 treated animals on day 46. The HeLa-treated animals were subjected to necropsies on day 92 which revealed that 2 of 5 mice developed masses around the injection site (Supplementary Figure 3C). This result indicates that the three different cancer cell lines show different tumorigenic abilities when administered into the Achilles tendon. HeLa cells demonstrated the poorest tumorigenic ability among the three cell lines tested. Both U-87MG and HCT116 cells showed 100% tumorigenicity and developed a visible mass around the injection site. The U-87MG injected group experienced the highest death rate. As HCT116 cells showed good tumorigenic ability and a good indication of body weight loss (from day 29) before visible mass formation (day 35) or animal death, we decided to use HCT116 cells as a positive control for further experiments using Achilles tendon injection.
Based on the study Nx053, we conducted another study Gx203 using HCT116 cells as a positive control. In this study, 1 × 106 HCT116 cells were administered via Achilles tendon injection to 24 (12/12 male/female) NSG mice. In this study, both male and female mice showed body weight loss from day 29 compared to the VC group (Figure 2A). One male died on day 43 in PC, and all remaining animals underwent necropsies on the same day. All 24 animals developed a mass around the injection site (Figure 2B). Interestingly, based on gross observation and histopathological diagnosis, we found tumor formation in several organs, including the kidney, lung, and liver, other than the injection site (Figure 2C and D, and Supplementary Figure 3D). Collectively, 1 × 106 or 2 × 106 HCT116 cell injection serves as an ideal positive control condition for Achilles tendon injection. In addition, HCT116 cells administered via the Achilles tendon can form tumors in multiple organs as well as at the injection site, implying that Achilles tendon-injected cells can reach multiple organs. HCT116 Achilles tendon injection. (A) Changes in mean body weight during the course of study Gx203. Vehicle (VC) or 1×106 HCT116 cells (PC) were injected to 12 male and 12 female NSG mice for each group through the Achilles tendon. Error bars indicate STD. (B) Tumor formation rates of study Gx203. Tumor formation was determined by tumors formed around the injection site, necropsy, and histopathological diagnosis. (C) Tumor-formed regions of male (upper) and female (lower panel) mice other than injection site. Each color represents the identity of each animal. (D) Representative H&E staining images of kidney, liver, and lung neoplasms of PC animals in Gx203. VC animal slide images were used as controls. All scale bars represent 500 μm. Abbreviations: PC, positive control group; VC, vehicle control group.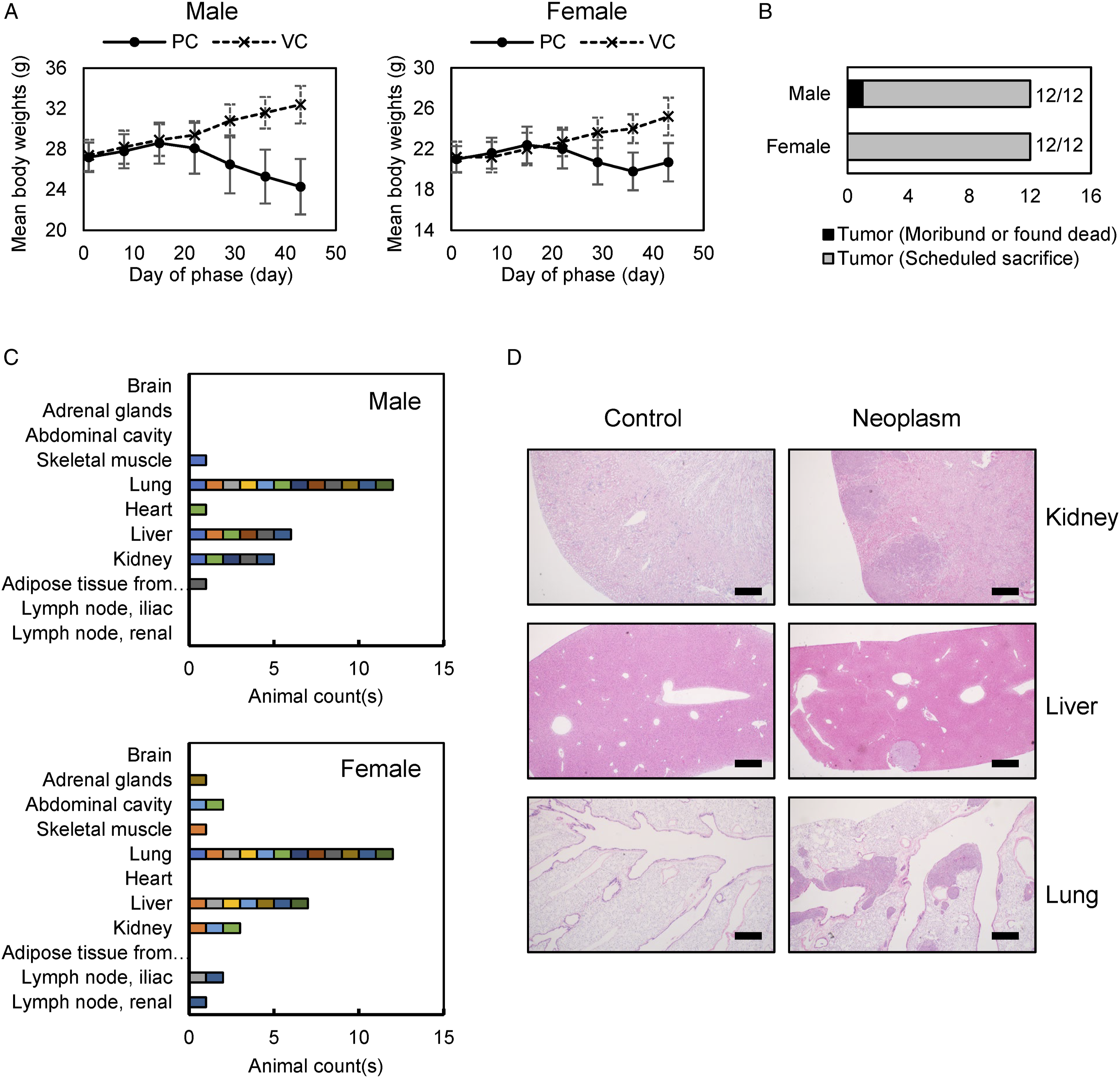
HCT116 Cell Subcutaneous Injection
SC injection is the administration route described in the WHO guidelines for positive control cells in tumorigenicity tests.
12
Tumor formation around the SC injection site can be easily detected by simple observation without necropsy. We conducted four studies (Gx139, Gx166, Gx185, and Gx191) following GLP regulations and guidelines. In those studies, 1×107 cells, as recommended by WHO guidelines,
12
were administered through SC injection to 24 (12/12 male/female) athymic nude mice per study. In all studies, significant mean body weight loss was detected in PC animals (Figure 3A and B, Supplementary Figure 4A and B). As expected, a visible mass was observed around the injection site (Figure 3C, Supplementary Figure 4C) for all animals (96/96) at a similar date when body weight losses were observed. Due to trackable mass formation, scheduled necropsies were conducted on day 36 for Gx139, day 47 for Gx166, and day 43 for Gx185. For Gx191, all animals were found dead or moribund between days 22 and 37 (Figure 3D). It is unclear why the animals in study Gx191 died sooner than other studies, but body weight loss and mass formation were detected earlier in Gx191 than in the other three studies. As one male body was lost during the study, necropsies were conducted on the remaining 95 tumor-formed animals (Figure 3E). Interestingly, 20 mice (21%) developed tumors in the lungs (Figure 3F). Tumor formation was not detected in organs other than the lungs. Collectively, we concluded that SC injection of 1×107 HCT116 cells is a reliable condition for tumorigenicity >90%. Mean body weight loss and visible mass detection serve as good signals to determine the proper necropsy date of PC. SC injection of HCT116 cells generally does not develop tumors in sites other than the injection site and lung. HCT116 subcutaneous injection. Mean body weights of 12 male (upper panel) and 12 female (lower panel) nude mice after vehicle (VC) or 1×106 HCT116 cell (PC) injection in study Gx139 (A) and Gx191 (B). Mean body weights were calculated until the survival rate of the PC animals was higher than 50%. Error bars indicate STD. (C) Tumor tracking results of Gx139 (upper panel) and Gx191 (lower panel). Tumors formed around the injection site were measured. Error bars indicate STD. (D) Necropsy conducting dates of all animals used in studies Gx139, Gx166, Gx185, and Gx191. For moribund or found dead animals, necropsies were conducted on the day of detection. For other animals, necropsies were conducted at the scheduled date. (E) Tumor formation rates of studies Gx139, Gx166, Gx185, and Gx191. Tumor formation was determined by tumor observation around the injection site, necropsy, and histopathologic diagnosis. (F) Representative H&E staining images of lung neoplasm of a PC animal in study Gx185. A lung slide image of a VC animal was used as a control. All scale bars represent 500 μm. Abbreviations: PC, positive control group; VC, vehicle control group.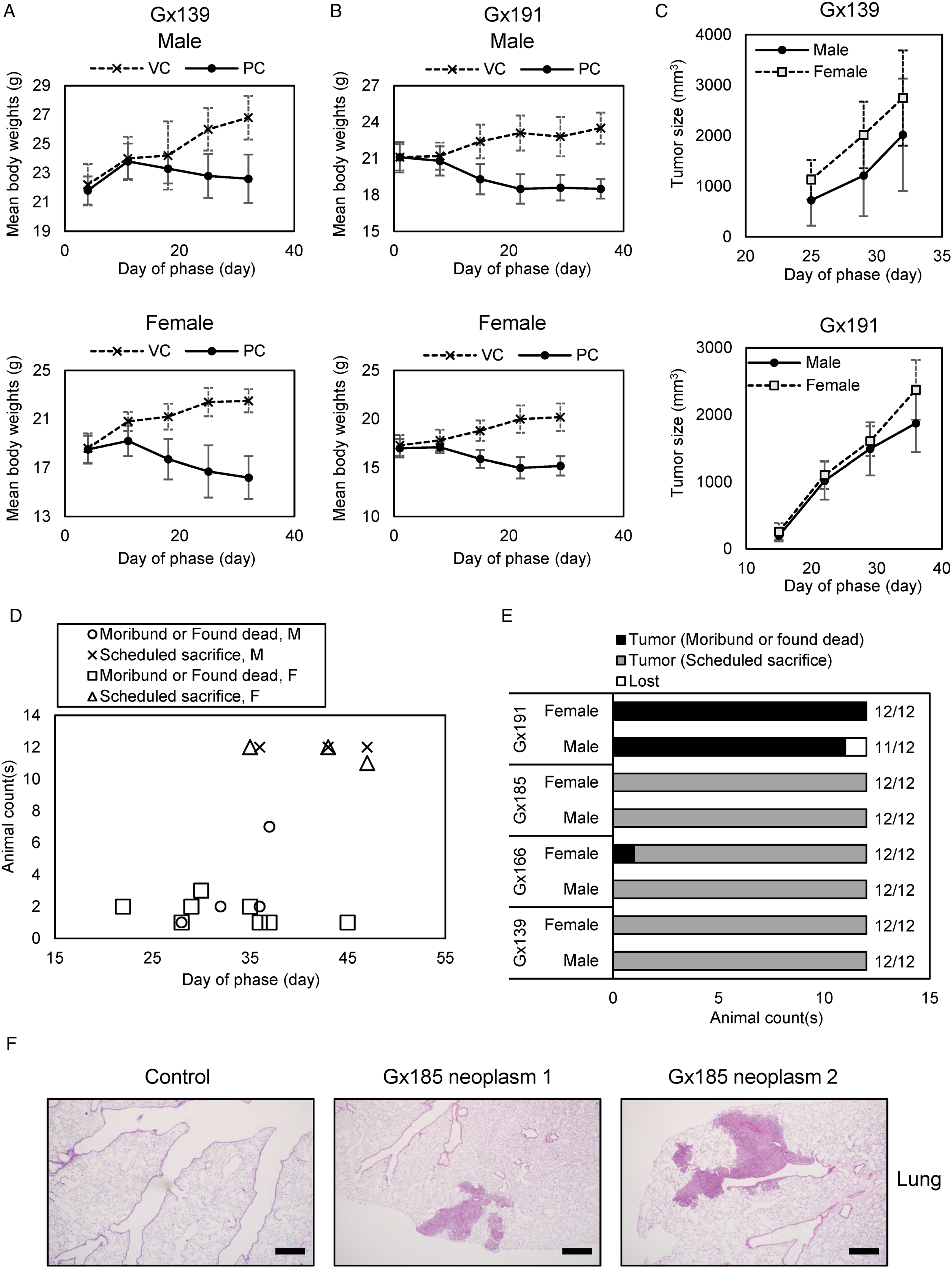
Establishment of HCT116 Intravenous Injection Dosing Amount
IV administration is a fast and direct way to deliver medication. The injected material directly enters the circulatory system and is thus quickly distributed. 34 In this regard, if the testing material has the potential to develop tumors and is considered for delivery through IV injection, tumorigenicity should be more rigorously assessed. We attempted to establish a reliable condition for tumorigenicity of positive control cell HCT116 delivered through IV injection. First, we conducted two pilot experiments (Nx042 and Nx061) to determine the appropriate HCT116 cell numbers that show sufficient tumorigenicity. For the two studies, tumorigenicity was assessed only by gross observation without histopathological diagnosis, as we intended to establish the condition that would permit detection of visible masses or nodules. In study Nx042, we injected 5 × 105 (G1), 1 × 106 (G2), and 2 × 106 (G3) cells into 10 nude mice (5/5 male/female) per group, and conducted necropsy on day 76. In study Nx061, 2×106 (G4) and 5×106 (G5) cells were injected into 10 nude mice (5 male/5 female) per group, and necropsies were conducted on day 87. In both studies, animal deaths were observed before the day of necropsy (Supplementary Figure 5A and B), and tumorigenicity was assessed by gross observation (Supplementary Figure 5C). Interestingly, while all male animals in G1 and G2 developed masses or nodules in multiple organs, female animals showed poor mass formation (60% for G1, 40% for G2). As we did not perform histopathological diagnosis, we could not exclude the possibility that we might miss tumor-formed tissues of females, as the viability of female animals was also significantly reduced during the study (Supplementary Figure 5A). However, this result indicates that IV injected HCT116 cells, especially in the relatively low number of cells (G1 and G2) may behave differently in males and females for unknown reasons. In G3 and G4, the same number of cells (2 × 106 cells) was used in different studies. In both studies, 2 × 106 injections showed 80% tumorigenicity, regardless of sex. When we injected more cells (5 × 106 cells in G5), no noticeable enhancement of mass formation was detected.
Even without performing histopathological diagnosis, we achieved 80% mass or nodule formation for both male and female mice when we injected 2×106 HCT116 cells intravenously. To confirm this, we conducted another study, Gx135: 2 × 106 HCT116 cells were injected intravenously into 34 athymic nude mice (17/17 male/female). Mice began to die from day 28, and 20 mice died by day 56, 32 mice (16/16 male/female) died by day 123 (Figure 4A). During the study, a statistically significant mean body weight loss in males was detectable from day 49 (Supplementary Figure 6A, left panel); however, the bodyweight of PC females did not differ significantly from VC females (Supplementary Figure 6A, right panel). All animals underwent necropsy, and tumorigenicity was assessed by gross observation and histopathological diagnosis. All moribund or found dead animals (32/34) developed tumors (Figure 4B). Two survived, which were sacrificed on day 182, did not develop a tumor. In this study, tumorigenicity was above 94% for both male and female mice, which met the 90% requirement of tumorigenicity of PC animals (Figure 4C). As the injected material is distributed by circulation when administered intravenously, tumors developed at multiple sites including the lung, brain, liver, heart, skeletal muscle, abdominal cavity, thoracic cavity, and adrenal gland (Figure 4D and E, Supplementary Figure 6B). Collectively, IV injection of HCT116 cells has the potential to achieve >90% tumorigenicity, although both macroscopic and microscopic observation after necropsy are required for tumorigenicity assessment. It is not clear if the mean body weight loss can be used as an indicator of tumor formation, especially for females. As HCT116 cells can generate tumors at multiple sites when injected intravenously, a more careful tumorigenicity assessment is required. HCT116 intravenous injection. (A) Survival rates of 17 male (left panel) and 17 female (right panel) nude mice after 2×106 HCT116 intravenous injection in study Gx135. (B) Tumor detection date of all tumor developed animals. Tumor formation was determined by necropsy and histopathological diagnosis. (C) Tumor formation rates of male and female mice in study Gx135. (D) Tumor formation sites of male (upper panel) and female (lower panel) mice. Each color represents the identity of each animal. (E) Representative H&E staining images of brain, heart, and liver neoplasms of positive control group animals in study Gx135. Vehicle control group animal slides were used as controls. All scale bars represent 500 μm.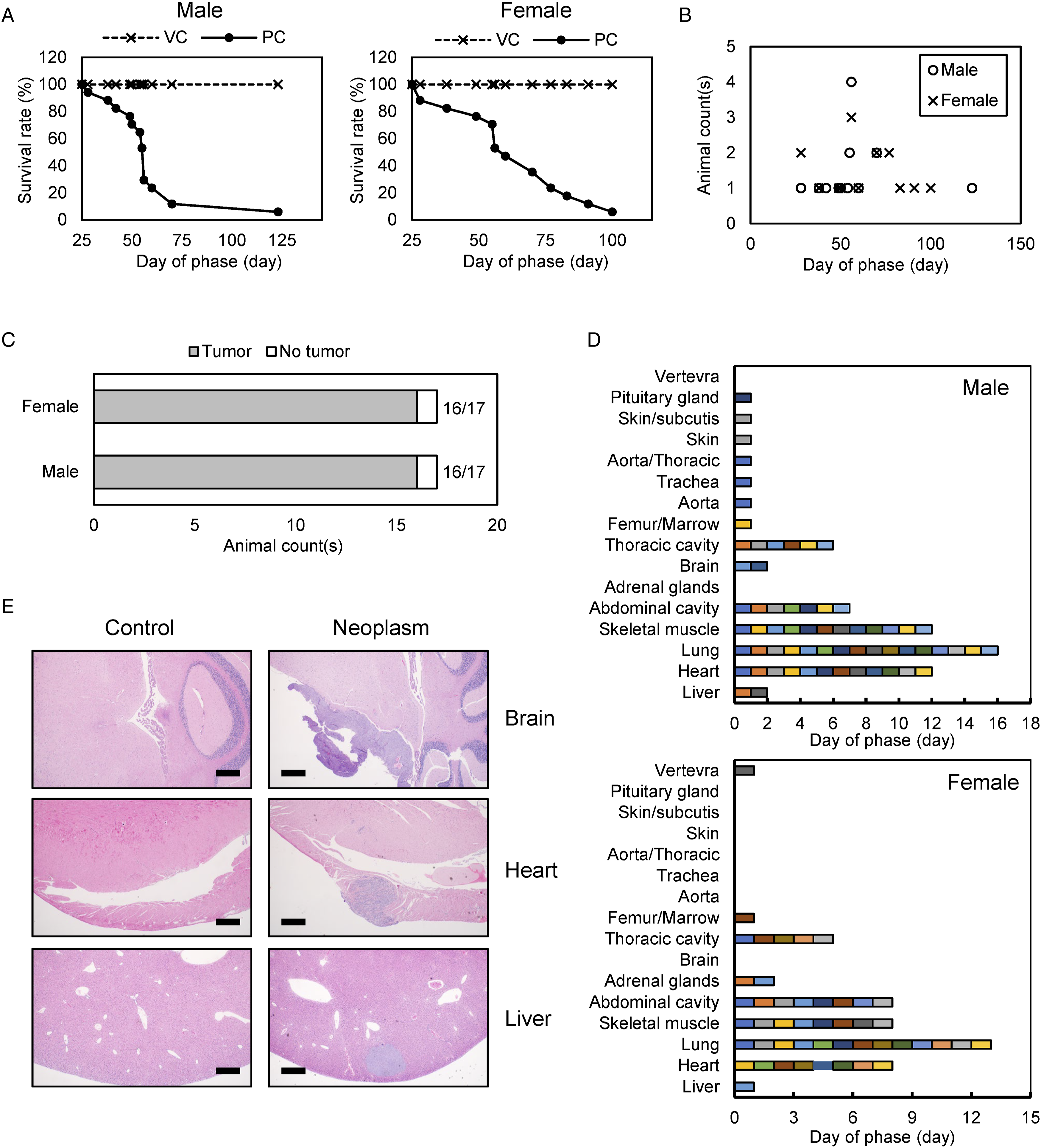
Discussion
Summarized Experimental Conditions and Results of Good Laboratory Practice Tumorigenicity Tests.
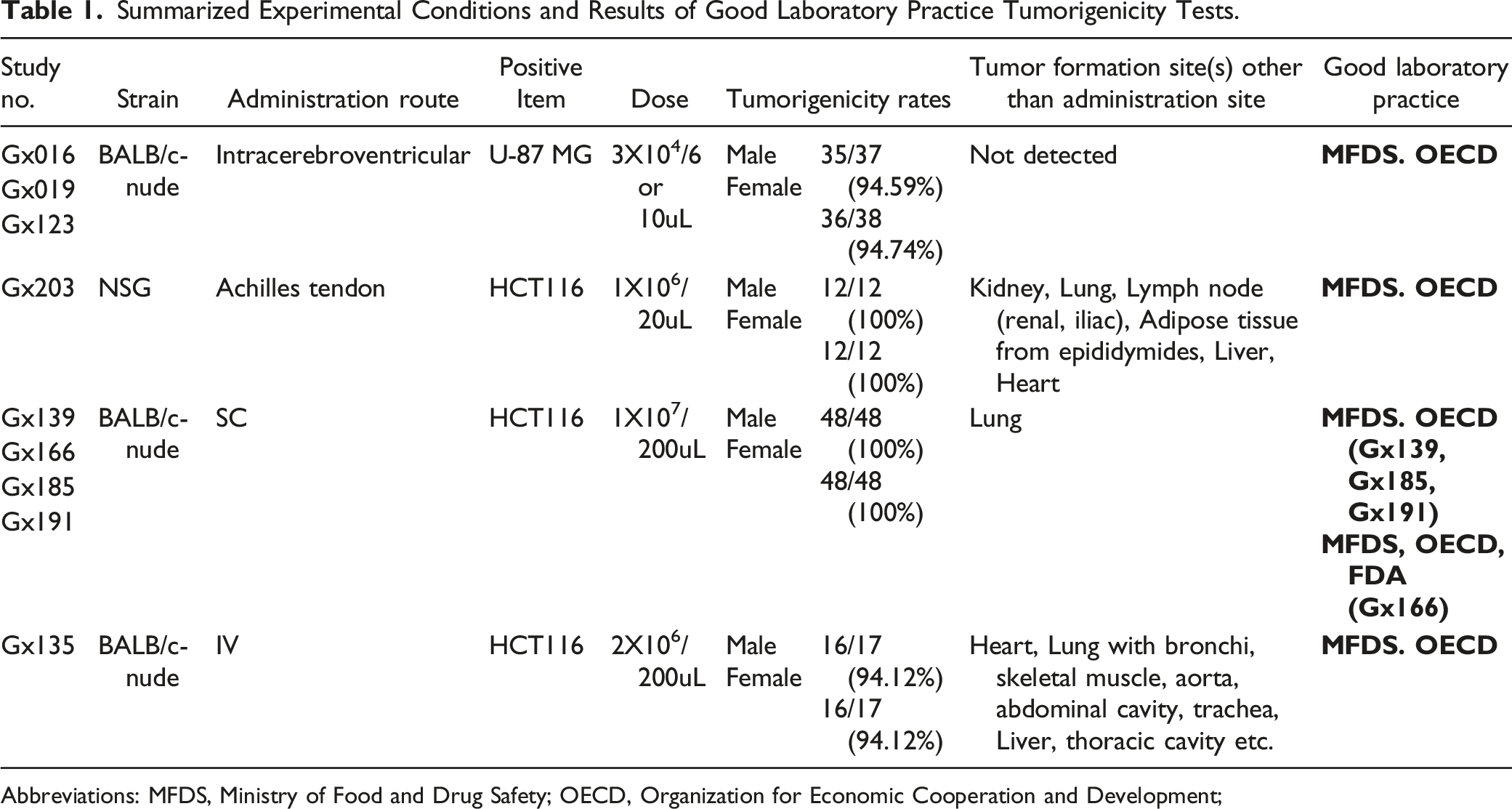
Abbreviations: MFDS, Ministry of Food and Drug Safety; OECD, Organization for Economic Cooperation and Development;
Overall, this study provides reliable tumorigenic conditions of HCT116 or U-87MG cancer cell lines which were xenografted onto immunodeficient mice via multiple routes including ICV, Achilles tendon, SC, or IV. We expect that this resource will help to develop successful tumorigenicity tests using proper positive controls, administration routes, and doses.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Supplemental Material
Supplemental material - Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing
Supplemental material for Tumorigenicity Assessment of Human Cancer Cell Lines Xenografted on Immunodeficient Mice as Positive Controls of Tumorigenicity Testing by Seunghee Oh, Eun-Young Gu, Ji-Seok Han, Byoung-Seok Lee, Kyoung-Sik Moon, Yong-Bum Kim, and Kang-Hyun Han in International Journal of Toxicology.
Footnotes
Acknowledgments
We thank ENCell Inc., SL BIGEN Inc., CORESTEM Inc., and AcesoStem Biostrategies for experimental supports during this project.
Author Contributions
Conception and design of the study: KH. Acquisition of data: JH, BL, KM, and YK. Analysis and interpretation of data: SO and EG. Drafting or revising the manuscript: SO, EG, and KH. All authors have approved the final article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by the Korea Institute of Toxicology of the Republic of Korea (No. 1711133843). These funding sources were not involved in any step of the study design; collection, analysis or interpretation of the data; or writing of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
