Abstract
Drug-induced vascular injury (DIVI) is a common preclinical toxicity usually characterized by hemorrhage, vascular endothelial and smooth muscle damage, and inflammation. DIVI findings can cause delays or termination of drug candidates due to low safety margins. The situation is complicated by the absence of sensitive, noninvasive biomarkers for monitoring vascular injury and the uncertain relevance to humans. The Safer And Faster Evidence-based Translation (SAFE-T) consortium is a public–private partnership funded within the European Commission’s Innovative Medicines Initiative (IMI) aiming to accelerate drug development by qualifying biomarkers for drug-induced organ injuries, including DIVI. The group is using patients with vascular diseases that have key histomorphologic features (endothelial damage, smooth muscle damage, and inflammation) in common with those observed in DIVI, and has selected candidate biomarkers associated with these features. Studied populations include healthy volunteers, patients with spontaneous vasculitides and other vascular disorders. Initial results from studies with healthy volunteers and patients with vasculitides show that a panel of biomarkers can successfully discriminate the population groups. The SAFE-T group plans to seek endorsement from health authorities (European Medicines Agency and Food and Drug Administration) to qualify the biomarkers for use in regulatory decision-making processes.
Introduction
The development of new medicines is associated with a high level of attrition (Arrowsmith 2011; Pammolli, Magazzini, and Riccaboni 2011). It is well known that less than 20% of all drug candidates entering clinical testing will complete clinical development and be submitted for regulatory approval (Kaitin and DiMasi 2011). In fact, the overall success rate for developing a drug is currently commonly estimated to be 1 in 5,000 to 10,000. Drug-induced vascular injury (DIVI) in preclinical animal toxicology studies (referred to as DIVI from here on) is one example of a finding that can cause considerable delays in the drug development process. In many cases, promising candidate drugs are terminated because in the absence of specific, sensitive biomarkers, the occurrence of this lesion cannot be monitored or conclusively ruled out in patients. It is estimated that on average, approximately 2.5% of the typical pharmaceutical company’s preclinical portfolio is affected by DIVI-related safety concerns, leading to significant delays or project termination (Predictive Safety Testing Consortium [PSTC] unpublished survey 2010). Historically, these delays were most commonly associated with compounds that caused vascular injury by altering vascular tone.
DIVI due to change in vascular tone is a toxicity that develops acutely (hours to days) in response to drug administration and often progresses to vascular inflammation (Kerns et al. 2005). It is induced by many structurally diverse compounds that come from numerous chemical and pharmacological classes, including dopamine agonists (Yuhas et al. 1985), phosphodiesterase inhibitors (Isaacs, Joseph, and Betton 1989), adenosine agonists (Albassam, Smith, and Macallum 1998), and potassium channel openers (Herman et al. 1989). Most early reports in the literature described DIVI caused by systemically vasoactive compounds (i.e., those that cause changes in heart rate and systemic blood pressure) but more recently DIVI has been reported/found with compounds that do not alter systemic hemodynamics but may do so in local vascular beds (Louden et al. 2000). While heart rate and blood pressure can be used as biomarkers of vascular injury for those compounds causing systemic hemodynamic changes, this is not true for compounds with only localized vasoactivity or with a different mode of action. The absence of noninvasive biomarkers that can be used to monitor potential vascular injury in humans has increased drug attrition and thus had a profound impact on drug development. For this reason, there has been a search for circulating biomarkers that can detect the onset, progression, and reversibility of DIVI.
The SAFE-T consortium is a unique public–private partnership between the European Commission and the pharmaceutical industry (as represented by the European Federation of Pharmaceutical Industries and Associations [EFPIA]). It operates under the framework of the European Union (EU) Innovative Medicines Initiative–Joint Undertaking (IMI-JU), which is partly funding the research activities under the project. The consortium proposes to accelerate drug development by qualifying biomarkers for drug-induced organ injuries in clinical studies (Matheis et al. 2011). New translational safety biomarkers will ideally allow the identification and management of the adverse effects of drugs throughout drug development, helping to reduce the risks involved with developing medicines while also improving the safety management of patients. One subgroup of the SAFE-T consortium, named Workpackage 4 (WP4), aims to address the question of human relevance of DIVI by qualifying a set of translational biomarkers for the monitoring of vascular injury in clinical studies.
A significant challenge for the qualification of translatable DIVI biomarkers is the uncertain relevance of DIVI to humans. With the exception of certain vasopressors (e.g., dopamine), the drugs that cause DIVI are not known to cause vascular injury in humans. Indeed, a significant number of drugs that produced DIVI in animal toxicity testing (Joseph 2000; National Toxicology Program 1998) have long-established clinical safety records. While sporadic occurrences of clinical drug–induced vasculitis (CDIV) are reported for a large number of drugs, including antimicrobials, antirheumatics, antithyroid drugs, vaccines, and many others, these occurrences manifest as a vasculitis syndrome that cannot always be differentiated from idiopathic vasculitides (Radic, Kaliterna, and Radic 2012).
The poor translation of DIVI to CDIV for most drugs is likely related to the differences in its pathophysiology in animals and humans. DIVI is seen most frequently with small molecules that have the capacity to modulate vascular tone (vasoconstrictors and vasodilators). There is a smaller group of other compounds that are thought to cause injury through other mechanisms at different anatomic sites (e.g., cytostatic agents and large molecules), but histomorphologic changes are generally similar to vascular active compounds (Greaves 2000; Kerns et al. 2005; Louden and Morgan 2001). While the onset of DIVI following administration of many small-molecule drugs is generally acute (hours to days following drug exposure; Kerns et al. 2005), and thought to be due to changes in wall tension and local blood flow, the onset of CDIV occurs typically weeks to months after exposure. In addition, the classes of drugs causing vascular injury in humans and animals are generally different. Also in contrast to DIVI, the skin is the predominant recognized target of humans CDIV (hypersensitivity vasculitis), while in more severe cases multiple organ systems may be involved. CVID is usually diagnosed based on macroscopic lesions or organ dysfunction, while DIVI is generally diagnosed by histopathology without clinical symptoms (Doyle and Cuellar 2003; Wiik 2008). Mechanistically, CDIV is considered to have an autoimmune or immune complex origin, compared to DIVI caused by most small molecules (Doyle and Cuellar 2003; Wiik 2008) which is believed to have a predominantly hemodynamic-mediated injury in animals in many cases. Some protein and nucleotide–based therapeutics can cause vascular injury in animal models due to formation of immune complexes, complement activation, or other immune system–mediated mechanisms.
The comparison between DIVI and CDIV highlights the challenge in identifying relevant human conditions for the clinical qualification of translatable vascular injury biomarkers. This is in contrast to the situation for drug-induced liver and kidney injury, where exposure in humans and animals to the same drug can lead to similar histopathological findings and can also lead to changes in the same biomarkers. For example, various forms of keratin-18 and High Mobility Group Box-1 are circulating indicators of hepatocellular injury in clinical acetaminophen hepatotoxicity and in animal models (Antoine et al. 2012; Antoine et al. 2009). To overcome the lack of directly relevant clinical models for DIVI, the SAFE-T WP4 group has proposed testing candidate biomarkers in surrogate populations of humans with vascular disease or injury, including the human vasculitides and other relevant disorders. The key assumption underlying this translational strategy is that the human vasculitides are associated with histopathological alterations similar to those observed in DIVI, as will be described subsequently in this article.
To ensure that the biomarkers associated with vascular injury are translatable across species, SAFE-T WP4 is collaborating with the PSTC’s Vascular Injury Working Group (VIWG), which is also using a morphologic-based, mechanism-independent approach to qualifying biomarkers of DIVI but is focusing on animal models. More details on the VIWG biomarker strategy can be found in the companion article by Mikaelian et al. (2014). The interaction between PSTC and SAFE-T involves the sharing of analytical and statistical methods, the development of complementary lists of candidate biomarkers, the collaborative review of study outcomes, and a unified strategy for seeking regulatory support for biomarker qualification. In this article, we describe the strategy developed by the SAFE-T WP4 group to translate clinically relevant biomarkers while overcoming the unique challenge that DIVI in most animal models is currently not detected in humans.
Comparative Histopathology
The lack of a direct correlate of DIVI in humans poses a unique challenge for the qualification of translational DIVI biomarkers. In several decades of developing drugs that cause vascular injury in laboratory animals, the existence of equivalent lesions has not been proved in man. The best examples for this conundrum is the vasodilator Minoxidil: while vascular lesions in animals are well described (Hanton et al. 2008; Joseph 2000; Mesfin et al. 1989), similar lesions could not be identified in a large number of autopsies of patients who had received the drug (Sobota 1989; Sobota et al. 1980). In addition, the mechanisms by which small molecules cause DIVI are incompletely understood. Therefore, the possibility of a simple translational experiment using one or a few compounds that produce vascular lesions both in man and animals does not exist.
Despite many differences between vascular injury in animals and humans, including the extent of involvement of the vascular compartments and their location, there are considerable similarities in the morphologic characteristics of DIVI and CDIV that can be leveraged for the selection of biomarkers and surrogate patient populations (Zhang, Hanig, and De Felice 2012). These similarities include the early-stage events of endothelial and smooth muscle cell damage and inflammation, and the later-stage events of vascular wall hyperplasia and fibrosis. More specifically, the morphologic similarities provide a basis for the detection of biomarker profiles that reflect the pathophysiologic status of the various vascular compartments. We therefore hypothesize that similar histopathology between DIVI and vascular injury/disease in humans will lead to overlapping biomarker signatures. As detailed subsequently, the primary features of histopathologic change in humans and animals (endothelial change, smooth muscle damage, and inflammation) serve as the connecting link between the preclinical and clinical conditions, and as a logical anchor against which to investigate biomarkers for qualification in DIVI independent of species differences in mechanisms of injury.
In the early stage of vascular injury, degenerative and/or apoptotic/necrotic changes can be observed in the smooth muscle layer and endothelium of blood vessels both in laboratory animals and man. Necrosis of smooth muscle cells in the tunica media (illustrated in Figure 1A–D) is a characteristic change at this stage and is frequently seen in humans and various preclinical species (Bourdois et al. 1982; Kerns, Arena, and Morgan 1989; Losco et al. 2004; Louden and Morgan 2001; Ohmachi et al. 1998b). Destruction of smooth muscle cells is expected to release structural proteins (e.g., transgelin, α-actin, and smoothelin) that can serve as biomarkers of injury. Secretion of such proteins into circulation would depend on impaired endothelial barrier function, something that generally occurs along with smooth muscle damage in vascular injury.
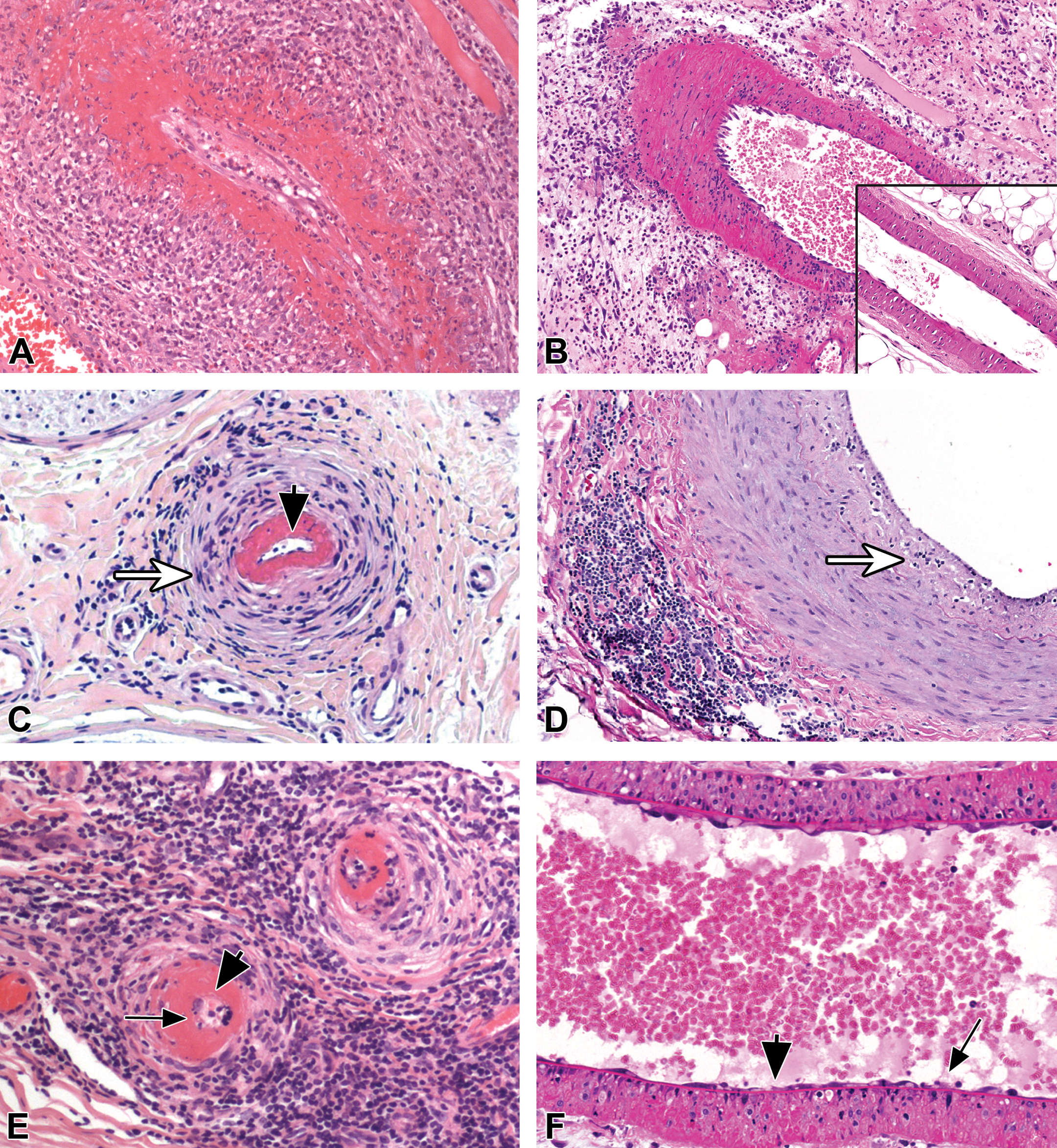
(A) Characteristic smooth muscle cell damage (necrosis) accompanied by mixed perivascular and medial inflammation; arteriole, skeletal muscle from a patient with classical polyarteritis nodosa; H&E stain. (B) Similar to the human example in A, prominent smooth muscle damage (necrosis) is present, accompanied by edema and mixed inflammation; arteriole, mesentery from a Sprague-Dawley rat 48 hr after administration of fenoldopam (100 mg/kg s.c.). H&E stain. Inset: normal mesenteric arteriole, rat. (C) Characteristic perivascular fibrosis (arrow outline) accompanied by endothelial damage (apoptosis/necrosis-arrowhead), smooth muscle damage (necrosis), and lymphocytic inflammation, arteriole, peripheral nerve from a patient with mixed cryoglobulinemia vasculitis; H&E stain. (D) Characteristic intimal fibrosis (arrow outline) and few intimal mononuclear inflammatory cells in a chronic incidental lesion; main coronary artery from a cynomolgus monkey; H&E stain. Similar lesions can be seen in laboratory animals following administration of vasotoxic substances. (E) Endothelial damage (loss-arrowhead; necrosis/apoptosis-arrow), and marked smooth muscle damage (necrosis), perivascular lymphocytic inflammation, and fibrosis; arteriole, peripheral nerve from a patient with mixed cryoglobulinemia vasculitis; H&E stain. (F) Endothelial damage (loss-arrowhead; apoptosis/necrosis-arrow), smooth muscle damage (necrosis), and perivascular edema and mixed inflammation; mesentery from a Sprague-Dawley rat 48 hr after administration of fenoldopam (100 mg/kg s.c.); H&E stain.
Endothelial cell activation, with adhesion of inflammatory cells and endothelial cell damage, is also an early event in DIVI that would be expected to be associated with a different set of biomarkers. Detection of circulating levels of soluble adhesion molecules such as ICAMs and VCAMs can serve as biomarkers of activation. Endothelial cell apoptosis and necrosis (illustrated in Figure 1C, E, and F) have been described as early events in DIVI (Bregman et al. 1987; Fujino, Kim, and Ito 2007; Joseph 2000; Mikaelian et al. 2010) and are known to occur in humans after treatment with oncology drugs (Soultati et al. 2012). In longer term preclinical studies with continued dosing and in human systemic vasculitides, early and late stage changes can coexist in the same tissue.
Following damage to the vessel wall, vascular integrity is disrupted leading to perivascular edema (Figure 1B) and hemorrhage, as well as inflammatory cell infiltrates in the vessel wall and perivascular space (Figure 1A, B, D, and E). Inflammation causes the release of different biomarkers, including inflammatory mediators like nitric oxide (NO), cytokines, cellular enzymes, and acute phase proteins. The composition of the inflammatory infiltrate varies similarly widely both in DIVI and CVID; however, neutrophils, macrophages, and lymphocytes are typically involved (Albassam et al. 1999; Boor, Nelson, and Chieco 1980; Fujino, Kim, and Ito 2007; Joseph, Rees, and Dayan 1996; Kerns, Arena, Macia, et al. 1989; Ohmachi et al. 1998a, 1998b). Further separation and biomarker identification for the various inflammatory lesion subtypes is considered impractical.
At a later stage, hypertrophy and hyperplasia of endothelial and vascular smooth muscle cells, as well as proliferation mainly of fibroblasts and myocytes in the vessel wall and surrounding tissues, can be observed (Figure 1A–C, and E; Boor, Nelson, and Chieco 1980; Bregman et al. 1987; Greaves 1998; Joris and Majno 1981; Miyauchi et al. 1993; Ohmachi et al. 1998b). Proliferative changes are associated with expression of specific genes and may also result in the release of specific biomarkers into the blood. Biomarkers related to the proliferation of these cell groups include cytokines such as transforming growth factor β (TGF-β) and vascular growth factors such as vascular endothelium growth factor (VEGF).
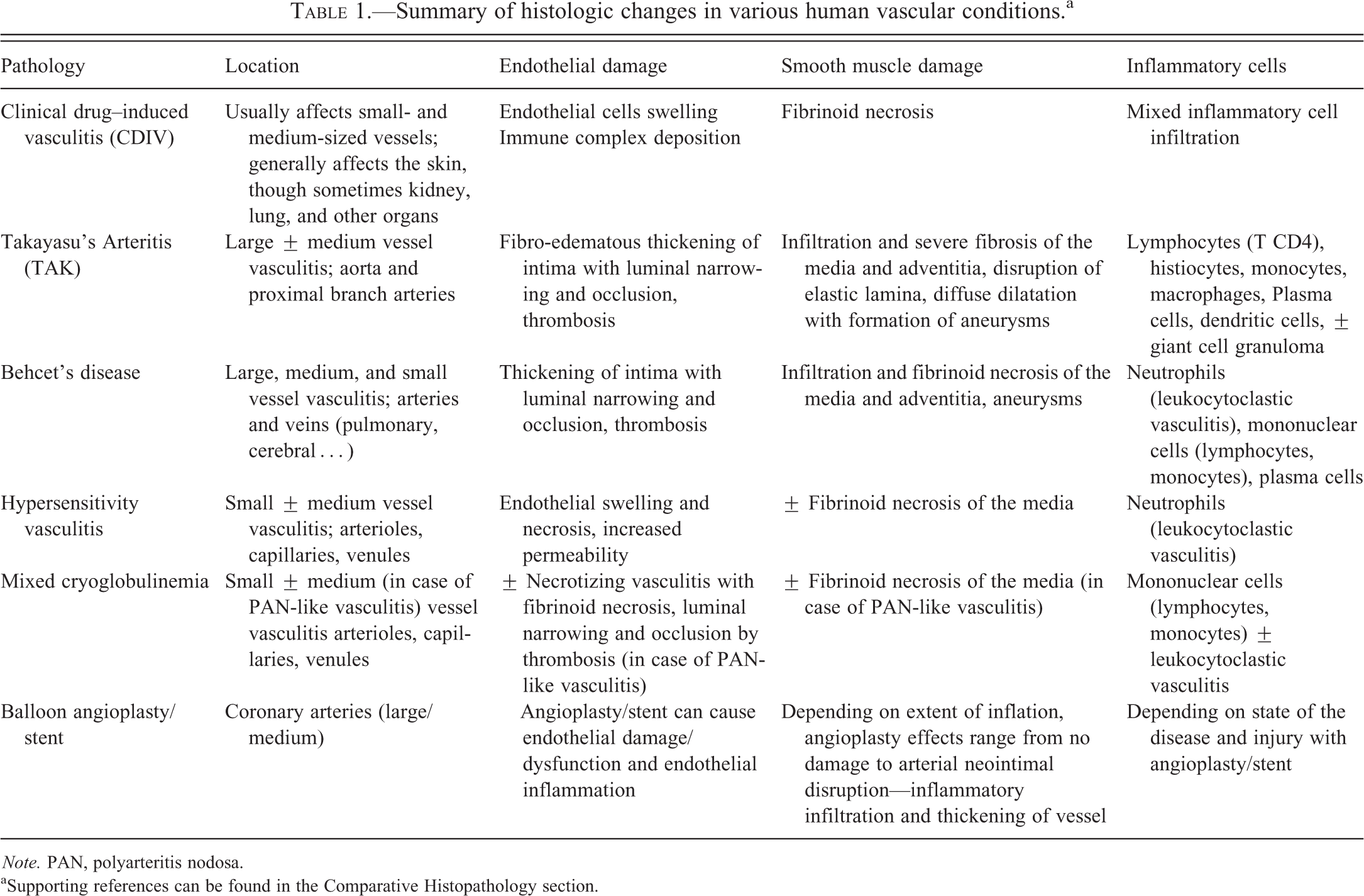
Not all features, however, are shared between DIVI in animals and CDIV. Morphologic details that are characteristic for certain types of human vasculitides, such as the occurrence of giant cells in the inflammatory infiltrate (in giant cell arteritis and Takayasu’s disease; Saadoun et al. 2012) or predominantly lymphocytic infiltrates (in mixed cryoglobulinemia; Cacoub et al. 2001; Terrier, Semoun, et al. 2011), are very rare in DIVI. Specific vascular beds are predominantly affected by various compound classes in DIVI and by human vasculitides. For example, vasodilators affect mainly small to medium size arteries (Joseph 2000; Kerns et al. 2005; Louden and Morgan 2001); in comparison, distinct diseases in humans affect large arteries (Takayasu’s disease, giant cell arteritis), or small to medium size arteries, capillaries, and venules (hypersensitivity vasculitis, mixed cryoglobulinemia; Jennette and Falk 1997). In addition, different organs are affected: while, for example, vasodilators generally cause damage to the vascular bed in the mesentery and splanchnic organs in rats (Kerns, Arena, and Morgan 1989; Yuhas et al. 1985), and the coronary arteries in dogs (Albassam et al. 1999; Albassam et al. 2001; Clemo et al. 2003; Greaves 1998; Isaacs, Joseph, and Betton 1989), the skin, kidneys, and other visceral organs are mainly affected in human vasculitis (Terrier, Izzedine, et al. 2011). It can be hypothesized that biomarker profiles may be influenced by these differences. Future analyses will show whether such differences can be captured using our biomarker panels. Histopathologic changes to the different vascular compartments associated with spontaneous or drug-induced human diseases are summarized in Table 1.
Summary of histologic changes in various human vascular conditions.a
Translation Using Surrogate Patient Populations
The disease populations for biomarker qualification were selected to cover the histopathological features found in DIVI as described in the previous section. The selected patient populations include diseases involving vessels of different types and sizes, different vascular compartments, and various types of infiltrating inflammatory cells. Patients with CDIV would be preferred for qualification studies, but we concluded that the limited access to this population made it impractical to include. Instead, we chose patients presenting with large-size vessel vasculitides (Takayasu’s arteritis, Behçet’s disease), medium-size vascular disease (patients undergoing balloon/stent angioplasty; see subsequently), or small-size vessel (arterioles, capillaries, and venules) vasculitides (hypersensitivity vasculitis, mixed cryoglobulinemia). Such selection criteria also permitted us to include diseases affecting predominantly different parts of the artery wall, such as the endothelium (hypersensitivity vasculitis, mixed cryoglobulinemia, Behçet’s disease), the media (patients undergoing balloon/stent angioplasty), or the adventitia (Takayasu’s arteritis). It also covered different types of vessels such as arteries (Takayasu’s arteritis, patients undergoing balloon/stent angioplasty) and veins (Behçet’s disease). For the vasculitis patients, inclusion criteria were based on “standard” definitions of disease as recently described by Jennette et al. (2013). A synopsis of the protocol for a prospective, longitudinal study to assess novel biomarkers of vascular injury in patients diagnosed with systemic vasculitis is also included as Supplemental Table 1.
When possible, biomarkers will be assessed in the same patient and at different time points including during remission (nonactive phase) and during a relapse (active phase). Patients in active phase generally present acute histopathologic alterations of the vessel, whereas vessels from patients in the nonactive phase do not exhibit these injuries. Relapse is usually diagnosed based on a combination of clinical, biological, and sometimes imaging tests. Additional information about scoring of remission versus relapse can be found in Supplemental Table 2. Patients in nonactive phase should make a useful control group for the specific association of a biomarker signal with vascular histologic damage rather than inflammation. Histologic characteristics of the various patient populations are summarized in Table 1. Figure 2 shows the overlapping between histopathologic features and tissue distribution of DIVI findings and clinical vascular disorders as the basis of selecting our patient populations.

Diagrammatic representation of overlapping blood vessel involvement and histopathologic manifestation in preclinical DIVI and surrogate populations.
The selection of patients undergoing balloon angioplasty was based on the goal of including a population with acute injury to the vessel wall and endothelium. Following a screening visit, a sample of blood will be collected to establish a baseline biomarker level. Patients will then undergo balloon angioplasty and another collection of blood will be performed within 24 hr and 3 months after the procedure. A synopsis of the protocol for the study on the assessment of safety biomarkers for acute vascular injury in patients undergoing balloon angioplasty is included as Supplemental Table 3. Several studies have shown the activation of genes and release of biomarkers within 24 hr of angioplasty (Bonello et al. 2006; Sardella et al. 2006). For example, IL-1β and IL-6 levels were significantly increased in the coronary sinus of patients 20 min after angioplasty (Sardella et al. 2006). An additional observational study in patients undergoing angiography is being performed to determine the association of biomarkers with atherosclerosis. Patients will undergo angiographic examination, and assessment of arterial stenosis and biomarker range will be calculated for these specific populations.
Anti-neutrophil cytoplasmic antibodies (ANCA)-associated vasculitis (Wegener’s granulomatosis and microscopic polyangiitis) is not a focus of the project because of a large study already published on promising biomarkers in these diseases (Monach et al. 2011; Monach et al. 2013). Other pathologies were excluded based on the challenges of clinical studies (e.g., rare pediatric diseases such as Kawasaki’s disease and Henoch-Schonlein purpura). Despite its many similarities to CDIV, hypersensitivity vasculitis was also not included in the initial qualification studies because, like CDIV, this disease is also rare and blood samples are hard to obtain.
Selection of Biomarker Candidates
Although many circulating biomarkers have been reported to be associated with vascular injury, their utility for drug development studies is unclear because data on preclinical–clinical translation and sensitivity and specificity are limited. After an extensive literature review, the WP4 group initially selected approximately 80 biomarker candidates that were likely to be associated with the three main histomorphologic features involved in DIVI: damage to vascular endothelium, damage to smooth muscle, and inflammation. This list was then filtered on the basis of various criteria that were used to score the biomarker candidates, including the existence of published data in preclinical and clinical settings, parameters related to the feasibility of appropriate sampling or of large-scale biomarker measurement, and intellectual property status. This approach resulted in the biomarkers that are listed in Table 2.
Biomarker candidates to be qualified in the SAFE-T project.a

bMesoscale Discovery.
Cytokines and other biomarkers of inflammation, such as C-reactive protein (CRP) and interleukin 6 (IL6), have been reported to be elevated in patients with vasculitis as well as during DIVI. However, these biomarkers are not specific to the vasculature and therefore cannot be used in isolation. In addition to inflammatory factors, endothelial-related biomarkers and specific smooth muscle proteins constitute other promising sources of biomarkers of vascular injury. By combining biomarkers of inflammation along with those reflective of damage to different vascular compartments, the consortium expects to qualify a panel of translational biomarkers allowing sensitive and specific detection and monitoring of DIVI. Additional details are presented in the section that follows and in Figure 3.
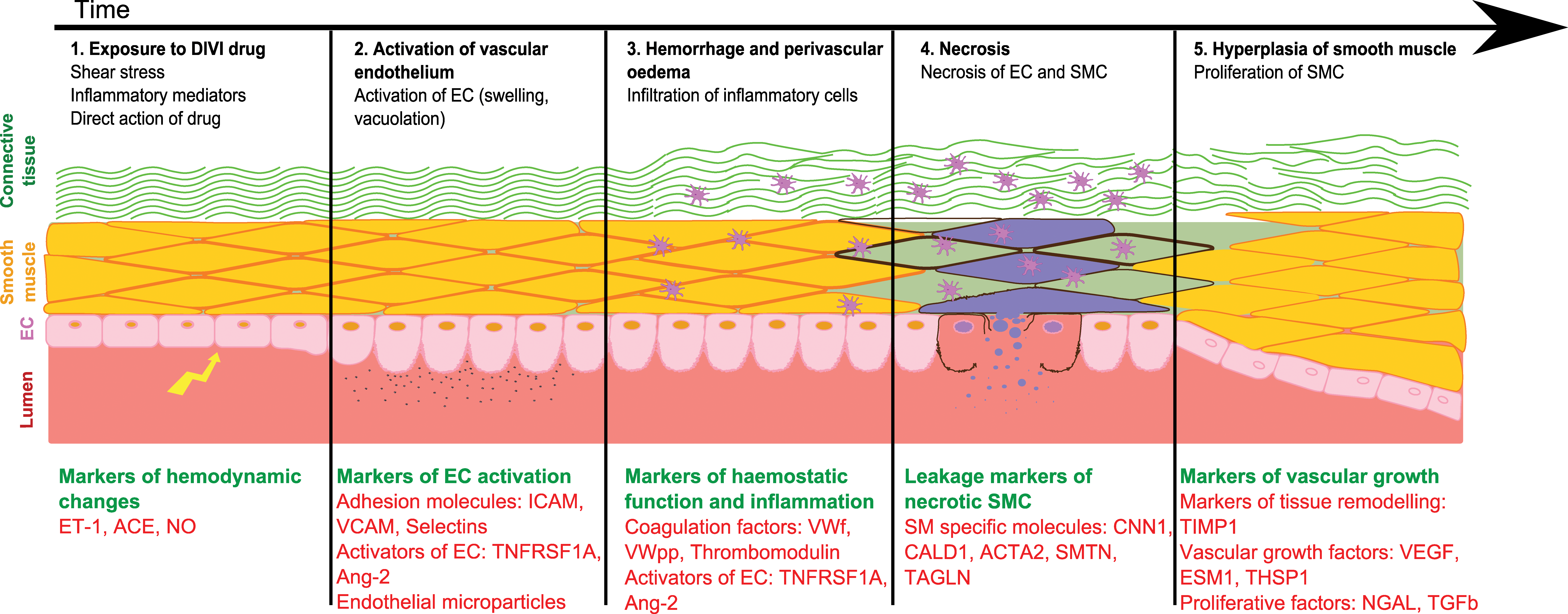
Relationship between pathophysiology of vascular injury and candidate biomarkers. While the multiple steps depicted in this diagram represent an idealized progression of injury, microscopic evidence shows that stages 2 to 4 often exist in parallel, and some or all of the described features may be present depending on specific compounds and their mechanisms of injury.
Changes in the expression of adhesion molecules such as increases in ICAM-1, VCAM, soluble E-selectin, and P-selectin are early biomarkers of endothelial cell (EC) activation. Higher levels of adhesion molecules have been associated with various clinical entities as well as with DIVI. Adhesion molecules have been shown to be expressed in response to shear stress and to activation of tumor necrosis factor (TNF) pathways. TNFα-related biomarkers such as the tumor necrosis factor receptor superfamily 1 A (TNFRSF1A) or angiopoietin 2 (Monach et al. 2012), a sensitizing factor of the endothelium to TNF signaling, have been shown to increase in clinical vasculitides and are included in our studies. However, in the case of DIVI, it is unclear whether activation is due to shear stress or secondary to inflammation. Biomarkers related to vascular function such as the angiotensin converting enzyme 1 (ACE-1), endothelin 1 (ET-1), the indirect measurement of NO through serum nitrites, and circulating levels of Caveolin-1 (Cav-1) may be expected to reflect hemodynamic changes and provide prodromal signals of the development of DIVI. Changes in biomarkers such as von Willebrand Factor (VWF), von Willebrand Factor propeptide (VWFpp), and thrombomodulin are indicative of functional changes in ECs and may have potential predictive value (Brott et al. 2005).
In some cases, the acute phase of DIVI can include the development of necrotic lesions in the vascular smooth muscle. In this stage, one would expect to see leakage of tissue-specific molecules into the circulation, enabling the detection of the necrotic damage. Several smooth muscle-specific proteins—H-caldesmon (CALD1), H1-calponin (CNN1), smooth muscle alpha actin (ACTA2), smoothelin (SMTN), and transgelin (TAGLN)—were selected as specific biomarkers of the smooth muscle damage. These proteins are involved in the structural organization and regulation of the contractile complex and have the potential to serve as a sensitive and specific signal of smooth muscle necrosis both in DIVI and in human vascular disease. This concept is supported by elevated serum levels of smooth muscle troponin-like protein levels in patients with aortic aneurysm and aortic dissection (Suzuki, Distante, and Eagle 2010).
There are numerous reports of increased levels of inflammatory factors in both DIVI (Weaver et al. 2010; Zhang et al. 2002; Zhang et al. 2008) and in clinical vasculitides (Kekow et al. 1993; Monach et al. 2011). Inflammatory changes would be reflected by secretion of factors such as monokine induced by gamma interferon (MIG), interferon gamma induced protein 10 (IP10), stromal cell-derived factor 1 (SDF-1), IL6, CRP, macrophage inflammatory protein 1α (MIP-1α), interleukin 8 (IL8), monocyte chemotactic protein 1 (MCP-1), and macrophage inflammatory protein 3β (MIP-3β) that promote the recruitment of inflammatory cells in the vascular wall and in the perivascular space. It is reasonable to assume that these factors play a central role in the pathophysiology of the disease, although it is unclear whether the activation of the cytokine network leads to the development of the vascular disease (as is believed to be the case with phosphodiesterase 4 [PDE4] inhibitors in rodents) or whether the inflammatory response is secondary to the initial damage. In this project, we anticipate that inflammatory biomarkers will serve as sensitive parameters for the detection of the onset of the disease and provide useful insights for the elucidation of the pathophysiologic mechanisms leading to the development of vasculitis.
The more chronic DIVI lesions are characterized by hyperplasia of the smooth muscle cells and fibroblastic proliferation and neovascularization. Intimal and luminal fibrotic processes are expected to be reflected by changes of neutrophil gelatinase–associated lipocalin (NGAL) through stabilization of matrix metalloprotease 9, or TGF-β levels (Bu et al. 2006; Suwanabol, Kent, and Liu 2011). Increases in endothelial specific molecule-1 (ESM-1) and VEGF levels have been correlated with the level of neovascularization of tumors (Roudnicky et al. 2013) and may be useful in detecting the perivascular neovascularization. At this stage, vessels also display chronic changes and significant fibrosis. Fibrosis is a physiologic healing process characterized by remodeling of the tissue through catalysis of existing matrix and collagen deposition. Candidate biomarkers selected to monitor this stage include proteins involved in this remodeling process, such as tissue inhibitor of metalloproteinase 1, (TIMP-1), thrombospondin 1 (THSP1), endocan (ESM-1), and VEGF.
In the setting of a drug development toxicological study, it would be useful to be able to detect changes that have occurred at an early time point in the study and have led to chronic lesions. Therefore, we have selected biomarkers that may be reflective of such changes. Patients with spontaneous forms of vasculitis will initially present with acute lesions characterized by inflammatory infiltrates and fibrinoid necrosis (Jennette et al. 2013). As these lesions progress and heal, they show increasingly vascular proliferation in and around the affected muscular vessels, marked fibrous proliferation, and neovascularization. In case of a flare-up, both the acute and the chronic lesions may coexist in the same individual. Samples derived from vasculitis patients are therefore expected to enable the discovery of biomarkers of acute as well as chronic changes.
While this project focuses primarily on the qualification of circulating protein biomarkers, other types of biomarkers, including ultrasound imaging, microRNAs, and microparticles, are also under consideration. For example, several microRNAs have been shown to be involved in vascular damage and disease (Urbich, Kuehbacher, and Dimmeler 2008; Wang et al. 2008). In addition, there are numerous reports of endothelial and other microparticles as biomarkers of vascular damage (Chironi et al. 2009; Jimenez et al. 2005). More details on the potential of microRNAs and endothelial microparticles can be found in the accompanying article by Mikaelian et al. (2014). These biomarkers were not initially included as part of the project because many of the archived samples available to SAFE-T were not collected in such a way to optimize their analysis.
Development and Validation of DIVI Biomarker Assays
The lack of appropriate measurement method with sufficient throughput and level of validation is often a bottleneck in projects aiming at the qualification of biomarkers. While various platforms became available for the early discovery and association of a biomarker with a given disease or physiologic phenomenon, it often remains problematic to measure a biomarker across different studies, projects, and companies. A significant effort is sustained across the consortium to develop methods that would be validated and made available industry-wide.
The consortium has focused on the MesoScale Discovery (MSD) multiplexed immunoassay format, in addition to the standard sandwich ELISA. The assays on the different formats were available from third-party vendors or developed on the basis of monoclonal antibodies developed by partners within the SAFE-T consortium. At the confirmatory stage, a decision will be taken on which platform to progress the assay based on the performance of the biomarker and the possibilities offered by vendors to ensure continued availability of such assay. Other platforms to be considered include the Luminex X-Map® or the Singulex Erenna® platforms.
Regulatory guidelines set forth by the Food and Drug Administration (FDA) for biomarker qualification require thorough validation. As described in the FDA’s “Guidance for Industry: Qualification of Biomarker Development Tools,” the applicant is required to submit technical data related to the measurement device and its analytical performance. The Clinical and Laboratory Standards Institute (CLSI 2005) has also released detailed guidelines for the validation of diagnostic assays, and similar guidelines are enforced in Europe, as described in the International Organization for Standardization (ISO) 17025 (Majewski and Bernards 2011). The SAFE-T consortium has established common standard operating procedures for the validation of assays and the measurement of samples and bioanalytical activity will be performed in a quality-controlled environment. The consortium will perform the validation of assays based on a “fit-for-purpose” approach as previously described (Lee et al. 2006; Lee, Figeys, and Vasilescu 2007; Lee and Hall 2009) by proceeding in discrete stages of validation of increasing stringency. The assays will be characterized in 2 steps: first, an initial technical validation during the exploratory phase followed by an extensive validation during the confirmatory phase. Assay performance will be assessed by estimating assay dynamic range (limit of detection, lower and upper limit of quantification), precision (intra-assay precision, intermediate precision), linearity, recovery, short-term stability, and freeze and thaw cycle stability in the matrices of interest (serum, lithium-heparin plasma, and EDTA plasma).
Another common bottleneck in biomarker projects is the challenge of measuring the same set of biomarkers in different species. Off-the-shelf, species-specific reagents available from commercial vendors can sometimes circumvent this issue. However, this approach cannot be used extensively, given the limited number of biomarkers for which reagents work both in human and in preclinical species. Several approaches are being used by the SAFE-T group to develop assays that detect biomarkers in multiple species. For instance, during the development of antibodies, the need for a cross-species reagent is taken into account when designing the immunogenic material, and sequences with a high level of similarity across rat, nonhuman primate, and human are preferred. Also, once monoclonal antibodies have been obtained, method development and selection of antibodies are performed using human and animal controls.
Clinical Qualification Study Plan
The SAFE-T project originally adopted a 2-step clinical qualification plan consisting of an initial exploratory phase followed by a confirmatory phase (Matheis et al. 2011). Generally, the exploratory phase studies will explore reference population variation, biomarker variation in patients with disease, biomarker variation in other organ diseases, and biomarker variation in drug-induced organ injury. Biomarkers will be compared and then selected for the next stage of qualification in the confirmatory phase studies based on evidence showing minimal biological variation, limited response in diseases affecting other organs, and evidence of some response in disease that is preferably pathophysiologically similar to drug-induced organ injury.
In an adaptation of this original qualification plan, the WP4 group included an initial feasibility phase to assess the biomarkers’ variability and ability to detect differences between the disease patients and healthy donors using a limited number of samples, called a “Stage-gate cohort” selected from a subgroup of vasculitis patients (including both flare and remission) and healthy volunteers. As discussed further in the Preliminary Results section, analysis of the stage-gate data using a panel of 20 biomarkers demonstrates that a multivariate statistical approach can be used to distinguish patients with disease from healthy volunteers, supporting the value of a panel of biomarkers.
All of the biomarkers from the feasibility phase of the project have progressed into the subsequent exploratory phase, where the intra-subject variation of the biomarker over time and the sensitivity of the biomarker to detect vascular damage associated with vasculitides and other vascular conditions, such as balloon angioplasty, will be established. Data from the studies in the exploratory phase, which were recently completed (but will not be reported here), are currently under analysis and will allow biomarker sensitivity to be established for the identification of biomarker combinations that can be progressed into the third stage of the project, called the confirmatory phase, where the most promising biomarkers will be progressed into larger confirmatory studies.
In the confirmatory phase of the project, the objective is to assess the performance of the biomarkers in larger patient populations by establishing normal ranges for the biomarkers and collecting data on the specificity/sensitivity of the biomarker for vascular injury. These studies will be conducted by observing the biomarker signals in control populations affected by common morbidities, disorders that are pathophysiologically comparable to vasculitides in that they show a strong inflammatory or autoimmune component. These pathologies may involve diabetes, acute infections, hypertension, systemic lupus erythematosus, rheumatoid arthritis, or chronic obstructive pulmonary disease (COPD). Another group of interest for the consortium will be disorders in which patients are likely to be exposed to classes of drugs that are associated with DIVI, such as COPD patients who may be treated with PDE4 inhibitors. In addition, it is possible that reference levels of candidate biomarkers in healthy subjects may be biased by the presence of subclinical atherosclerotic lesions. While it will not be within the scope of this project to perform an in-depth analysis of the effect of atherosclerosis on biomarker levels, an initial correlation of biomarker levels and atherothrombotic disorders will be completed in a study with coronary angiography patients. Biomarker assays retained at this stage will be validated more extensively in accordance, whenever possible, with the CLSI (2005) guidelines.
Recruitment of patients for all three phases of the project is being performed according to representative study protocols included as the Supplemental Tables that were described in the Surrogate Patient Populations section. To facilitate statistical analysis of biomarker data, detailed patient information is being collected using an electronic data capture system (OpenClinica) from all subjects, including medical histories, demographics (e.g., age, gender), and standard clinical chemistry values.
As stated earlier, a challenge for the qualification of biomarkers of DIVI is that drugs known to cause vascular injury in animal toxicity testing (e.g., theophylline and minoxidil) have long-established records of safety following clinical use. However, it is unknown whether the apparent absence of vascular damage in humans dosed with these drugs is due to the lack of species translation (i.e., DIVI manifestation), to a sufficiently high safety margin, or to the lack of an appropriate monitoring method in humans. Because there are currently no specific, sensitive biomarkers that can detect localized, low-level vascular injury, the occurrence in humans following treatment with these drugs cannot be excluded. This raises the question of how one would confirm the utility of biomarkers that may have been initially discovered and evaluated only in preclinical animal models but that have not been evaluated in relevant human populations. Before such biomarkers can be used in clinical trials with compounds that produced DIVI in preclinical studies, it must be demonstrated that these biomarkers are in fact responsive to vascular injury in humans; this is the rationale for selection of surrogate patient populations that was described previously. Once the biomarkers have been shown to be able to detect vascular injury in humans, we propose testing the biomarkers in healthy volunteers exposed to a drug known to cause DIVI in preclinical models. Several drugs that are currently approved for use in humans, including minoxidil and roflumilast, could be used in such studies. Negative results would suggest that DIVI seen in rodents (and presumably other species) is not relevant to man. (Alternatively, it cannot be completely ruled out that there may be DIVI lesions in humans that cannot be detected because the selected biomarkers are not sensitive enough). If, on the other hand, the biomarkers produce a signal indicative of injury, then this would support a conclusion that DIVI is a toxicity finding that is clinically relevant and can be monitored with the used biomarkers.
Finally, while the exploratory and confirmatory clinical studies are designed to demonstrate that the biomarkers are associated with vascular damage in humans, it is important to show that the biomarkers can also detect DIVI in animals. Therefore, “reverse translation” will be performed by testing the biomarkers in preclinical studies. Here, the SAFE-T group is collaborating with the PSTC’s VIWG as discussed in the Introduction. The preclinical studies focus on investigations in rats because of the prevalence of DIVI in this species. As mentioned, the relationship between changes to specific morphologic endpoints with changes in biomarkers specific for vascular compartments and/or inflammatory component is the basis of these studies. In some cases, biomarkers shown to detect vascular injury in animals will be tested with clinical samples (i.e., “forward translation”). In contrast, reverse translation is the process of selecting candidate biomarkers on the basis of their confirmed association with clinical conditions with vascular involvement and then testing them in preclinical animal models of DIVI.
Data Analysis
When determining the performance of a novel biomarker, a comparison is typically made to the conventional gold standard for the disease or toxicity being investigated. However, as discussed previously, the lack of a gold standard for the assessment of CDIV requires a different approach to studies for characterization of biomarker candidates and for the qualification of vascular biomarkers. While the comparator for assessing DIVI biomarker performance in animal studies is histopathology, biopsies are not feasible for the exploratory and confirmatory clinical studies. Instead, we propose using the current diagnostic criteria from multiple surrogate populations with human vascular injuries as the basis for determining biomarker performance in exploratory and confirmatory studies. The performance of a biomarker versus the diagnostic criteria of an array of vascular diseases will give insight on the vascular specificity and mechanistic profile of the biomarker.
Several descriptive, exploratory, and predictive statistical methodologies are being used in the various stages of the project. These methods may be expanded depending on the nature of the data obtained especially in the exploratory studies. Given the involvement of multiple histological features associated with vascular injury, we expect that one or more panels, comprised of biomarkers of endothelial and smooth muscle injury and inflammation, will be necessary to detect DIVI. Therefore, several multivariate statistical approaches are being proposed.
During the exploratory phase, multivariate approaches will be used to recognize patterns of biomarkers that may be reflective of particular subpopulations and assess whether this unsupervised classification results in differentiation of subgroups of interest for the project, for instance, if particular histologic features such as the vessel bed affected relates to clustering on biomarker values. Principal components analysis will allow reduction of the number of biomarkers and will serve as a basis for the development of multivariate models. Also this method will help identify the most significant covariates across the populations. Multivariate models will be constructed using machine learning derived classifiers using various algorithms (e.g., random forest, support vector machine, and Bayesian classifiers). The performance of these classifiers will be assessed on the test population using a leave-one-out method and using cross-validation approach. Also the effect of covariates such as age, gender, or other covariate identified earlier will be evaluated. The performance of the models will be validated prospectively during the confirmatory phase using the receiver operating characteristic (ROC) curve with logistic regression. The performance of individual biomarkers and biomarker panels will be measured using the area under the ROC curve (AUROC). A good biomarker should have an AUROC value over 0.8 (Lasko et al. 2005).
Preliminary Results
Patients with conditions involving inflammatory infiltrates, activation, and injury to the vascular endothelium and fibrinoid necrosis of the media were included, along with 50 age- and sex-matched healthy volunteers. The patients included were diagnosed with cryoglobulinemic vasculitis, giant cell arteritis, ANCA-associated vasculitis, Tayasu’s arteritis, Behçet’s disease, or vasculitis associated with Sjögren’s syndrome. Forty-six samples were available from patients in the acute phase, while 40 samples from patients in the remission phase (samples were available in both disease stage from 39 patients). Twenty soluble proteins were measured using immunoassays in serum samples collected in healthy subjects and in patients during the acute vasculitis (flare) and the resolution phases of disease (remission). The panel of biomarkers included inflammatory factors as well as endothelial proteins: CRP, E-Selectin, GROa, IL-6, IL-8, IP-10, I-TAC, NGAL, MCP-1, MIG, MIP-1, P-Selectin, SAA, sICAM-1, sICAM-3, sVCAM-1, thrombomodulin, TIMP-1, TNFRSF1A, and VEGF. Univariate analysis showed that no single biomarker measurement provided a sufficient predictive power with an AUROC above 0.8 (data not shown). In contrast, multivariate analysis based on Naïve Bayes, support vector machine,
To visually demonstrate the predictive potential of this set of markers in predicting vasculitis category, Figure 4 shows the scores from the first two components of a Partial Least Squares (PLS) classification model. Briefly, the PLS methodology identifies linear combinations of the markers that simultaneously reduce dimension and correlate with the vasculitis categorization (Khun and Johnson 2013). Note that because the figure shows only the first two components, visualization of the biomarkers’ ability to separate vasculitis categories would improve with the inclusion of additional components in the model.
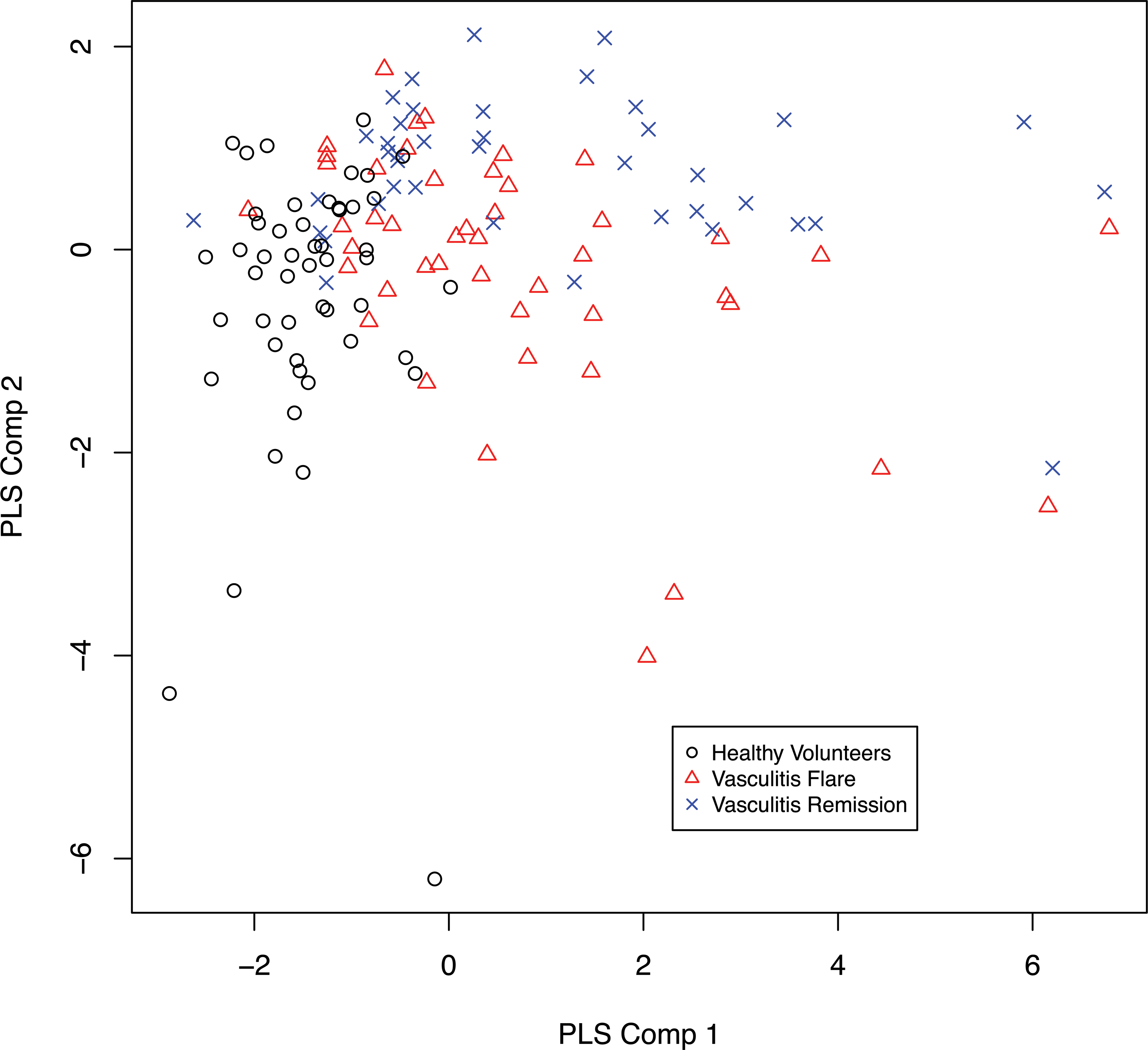
Multivariate analysis of 20 biomarkers from the stage-gate cohort of healthy volunteers (
Conclusion
The DIVI Workpackage 4 (DIVI WP4) of the SAFE-T consortium is addressing the current lack of sensitive and specific clinical assays to monitor DIVI in humans by testing candidate biomarkers in surrogate populations of human subjects with vascular disease or injury. Validation of biomarkers as a monitoring tool of potential vascular injury in clinical practice could provide the basis for clinical risk management strategies designed to overcome safety concerns arising from the identification of DIVI in preclinical toxicology studies. By combining biomarkers of inflammation along with those reflective of damage to different vascular compartments, WP4 expects to qualify a panel of translational biomarkers allowing sensitive and specific detection and monitoring of vascular injury in humans. If successful, this work will support the safe clinical development of important new medicines.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The research described was partially funded by the European Commission in the frame the IMI-JU program.
