Abstract
Drug-induced vascular injury (DIVI) is commonly associated with phosphodiesterase (PDE) inhibitors. Despite histological characterization, qualified biomarkers for DIVI detection are lacking. We investigated whether a single administration of roflumilast (PDE-IV inhibitor) induces vascular damage and identified novel surrogate biomarkers of acute vascular injury. Pigs received postoperative 250, 375, or 500 μg of roflumilast or placebo/control. After 1.5 hr, coronary reactivity was determined by catheter-based administration of acetylcholine and sodium nitroprusside (SNP) in the coronary sinus. Immunohistochemical analysis of vessel integrity (von Willebrand factor [vWF]) and fibrin(ogen) deposition was performed in the coronary artery and aorta. Peripheral blood was collected for differential proteomics and microparticles analysis. Circulating interleukin (IL)-6 was analyzed. Roflumilast-treated animals displayed higher vasodilation to acetylcholine and SNP versus controls (
Introduction
Inhibitors of phosphodiesterase (PDE)-IV, by selectively interfering with the breakdown of cyclic adenosine monophosphate (cAMP), have shown to suppress proinflammatory cytokine production and vasodilate respiratory smooth muscle (Burnouf et al. 2000; Heaslip et al. 1994). These properties have made PDE-IV inhibitors effective drugs for treating pulmonary diseases with both inflammatory and constrictive components such as chronic obstructive pulmonary disease (COPD) and asthma (Giembycz and Field 2010). Yet, the development of these compounds has been complicated by dose-limiting gastrointestinal side effects (Losco et al. 2004) and the frequent development of drug-induced vascular injury (DIVI) in the mesenteric vasculature in preclinical toxicology studies (Losco, Evans, et al. 2004). Although the vascular lesions induced by PDE-IV inhibitors have been well described histopathologically, the mechanisms by which PDE-IV inhibitors lead to DIVI remain uncertain. Most importantly, it remains unknown whether preclinical DIVI is relevant to clinical applications because there are no sensitive and specific biomarkers for detecting and monitoring DIVI. Thus, there is a great interest in the detection of biomarkers capable of assessing the appearance of DIVI in order to promote and ensure effective and efficient drug development.
With this in mind, the objective of this study was to evaluate in swine, a model with human resemblance and of growing interest in predictive safety testing (Vilahur, Padro, and Badimon 2011), whether a single administration of roflumilast induces DIVI and to identify new potential predictive biomarkers of vascular injury.
Method
Animals
Twenty-four commercial cross-breeding female pigs (Landrace/Large-white; Pig Specific, S.A; Barcelona, Spain) weighing 40 to 43 kg were acclimated for 1 week in an environmentally controlled room with a 12-hr light/dark cycle before any experimental procedure. Animals were fed a regular chow and water was provided
The study protocol was approved by the institutional ethics committee (CSIC-ICCC), and all animal procedures were performed to conform the guidelines from Directive 2010/63/EU of the European Parliament on the protection of animals used for scientific purposes or the National Institute of Health (NIH) guidelines (NIH Publication No.85-23, revised 1996). In addition, we have followed the ARRIVE guidelines (Kilkenny et al. 2010).
Experimental Design
Pigs were randomly distributed in 4 groups (6 animals/group) to either receive a single oral dose of 250-, 375-, or 500-µg roflumilast or control. The dose of roflumilast was chosen based on its clinical use for the treatment of severe COPD (500 μg/daily; Calverley et al. 2009; Grootendorst et al. 2007). One and a half hour post dose animals were sedated and anesthetized, blood samples (whole blood, plasma, and serum) were collected for further analytical, flow cytometry, and proteomic studies. Thereafter, coronary reactivity (endothelium-dependent and -independent vasodilation) was evaluated
Vascular Reactivity Studies
One and a half hour after drug/placebo intake pigs were sedated with an intramuscular injection of tiletamine + zolazepam (7 mg/kg) + medetomidine (0.07 mg/kg), endotracheally intubated, and anesthesia was maintained with isofluorane (2%). Under aseptic conditions, an incision was made in the ventral portion of the neck to expose the carotid artery, and another incision was performed in the thorax to proceed with the opening of the chest, the removal of pericardium, and the exposure of the heart. Thereafter, an ultrasonic probe connected to a blood flow meter (Two Channel Perivascular Flow System; ADInstruments, Oxford, UK) was inserted in the midportion of the dissected left anterior descending coronary artery. A second flow probe was placed in the carotid artery in order to simultaneously measure flow changes in larger vessels for control purposes (peripheral assessment). After vessel diameter measurement and complete hemodynamic baseline measurements were performed, vascular reactivity was assessed by intracoronary delivery of vasoactive agents. To this end, animals underwent catheterization of the left main coronary artery. All drugs were diluted with physiologic 0.9% NaCl solution to a volume of 1 ml and were infused during a 30-sec period. There was at least a 15-min interval between completion of infusion of 1 drug and administration of the next. Endothelial-dependent vasodilation was assessed by the intracoronary infusion of acetylcholine (receptor-operated vasodilator; 10−8 to 10−6 M; #A6625, Sigma-Aldrich, Barcelona, Spain) whereas the endothelium-independent vasodilation (vascular smooth muscle-related) was assessed with a dose–response curve to sodium nitroprusside (SNP; 10−7 to 10−5 M; #PHR1423, Sigma-Aldrich, Barcelona, Spain). The doses of vasoactive substances, while producing the desired effects following intracoronary administration as previously reported (Vilahur et al. 2012; Saitoh et al. 1998), did not induce any systemic changes. Data are presented as the percentage change of coronary blood flow response measurements from baseline to maximal post-pharmacological agent infusion (i.e., percentage of relaxation). Femoral mean blood pressure and heart rate were continuously monitored by a blood pressure transducer and an electrocardiogram (ECG) throughout all the procedure.
Immunohistochemical Analysis of Arterial Beds
At sacrifice, both the thoracic aorta and the coronary artery of 12 animals (
Circulating Interleukin (IL)-6 Measurements
Cytokine IL-6 was analyzed at sacrifice using a commercially available enzyme-linked immunosorbent assay (ELISA; IL-6 porcine Quantikine ELISA assay #P6000B, R&D Systems, Abingdon, UK). According to the manufacturer, the minimum detection limit for IL-6 is 10 pg/ml.
Circulating Microparticle Isolation
The circulating microparticle fraction (cMP) was isolated from platelet-free plasma (PFP) by a 2-step high-speed centrifugation (Suades et al. 2013, 2014). Briefly, frozen PFP aliquots were thawed on melting ice for 1 hr and centrifuged (20,000×
Flow Cytometric Analysis of Circulating Microparticles
Three-color flow cytometric analysis was performed as previously described (Suades et al. 2013, 2014; Nieuwland et al. 2000). Briefly, washed cMP suspensions diluted in Annexin Binding Buffer (#556454; BD Pharmigen
Serum Proteomic Profiling Analysis
Serum samples were stored at −80°C until used. Protein concentration was measured with 2D-Quant kit (GE Healthcare) prior to proteomic analysis. Serum samples were separated by 2-dimensional electrophoresis (2-DE), and proteins were identified by matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF) as previously described (Cubedo et al. 2011; Cubedo, Padró, and Badimon 2014). Four animals of each group were analyzed.
Two-dimensional Gel Electrophoresis
For analytical (120 µg) and preparative (300 µg) gels, the urea/chaps soluble extracts were applied to 17-cm dry strips (pH 4–f7 linear range, #163-2008, BioRad, California). Gels were developed by fluorescent staining (Flamingo, #161-0492, BioRad). For each independent experiment, 2-DE from each group was processed in parallel to guarantee a maximum of comparability and each run was at least repeated twice. Differences in protein patterns were analyzed with the PD-Quest software (version 8.0, BioRad), using a single master including all gels of each independent experiment. Each spot was assigned a relative value corresponding to the single spot volume compared to the volume of all spots in the gel, following background extraction and normalization between gels.
Mass Spectrometry Analysis
Protein spots were excised from 2-DE gels, washed, dehydrated, dried, and enzymatic digested with sequence-grade modified porcine trypsin (#v5280, Promega, Madison, Wisconsin), as previously described, and analyzed by MALDI-TOF using an AutoFlex III Smartbeam MALDI-TOF/TOF (Bruker Daltonics, Bremen, Germany). Samples were applied to Prespotted AnchorChip plates (#255683, Bruker Daltonics) surrounding the calibrators provided on the plates. Spectra were acquired with flexControl on reflector mode, (mass range 850–4,000 m/z, reflector 1:21.06 kV; reflector 2:9.77 kV; ion source 1 voltage: 19 kV; ion source 2:16.5 kV; detection gain 2.37×) with an average of 3,500 shots at a frequency of 200 Hz. Each sample was processed with flexAnalysis (version 3.0, Bruker Daltonics) considering a signal-to-noise ratio over 3, applying statistical calibration and eliminating background peaks. For identification, peaks between 850 and 1,000 m/z were not considered (usually only matrix peaks are visible on this range). Processed spectra were sent to the interface BioTools (version 3.2, Bruker Daltonics) and software search engine MASCOT search on Swiss-Prot 57.15 database was done (Mass Tolerance 50 to 100, up to 2 miss cleavage, Global Modification: Carbamidomethyl (C), Variable Modification: Oxidation, Amschler 1995). Identification was carried out by peptide mass fingerprinting (PMF) where a mascot score higher than 56 was accepted as previously reported (Cubedo et al. 2011; Cubedo, Padró, and Badimon 2014). Identified proteins were then confirmed by peptide fragmentation working on the reflector mode (MS/MS).
In Silico Bioinformatic Analysis
The proteomic data were used to identify the canonical pathways and causal networks statistically upregulated using the ingenuity pathway analysis (IPA; Lipari, Benipal, and Kale-Pradhan 2013) technology (Ingenuity Systems, www.ingenuity.com).
Functional analysis of a network
The functional analysis of a network identified the biological functions and/or diseases that were most significant to the molecules in the network. The network molecules associated with biological functions and/or diseases in the Ingenuity Knowledge Base were considered for the analysis. Right-tailed Fisher’s exact test was used to calculate a
Canonical pathway analysis
Pathways analysis from the IPA library identified the canonical pathways that were most significant to the data set. The significance of the association between the data set and the canonical pathway was measured in 2 ways: (1) a ratio of the number of molecules from the data set that map the pathway divided by the total number of molecules that map the canonical pathway and (2) Fisher’s exact test to calculate a
Statistical Analysis
Normal distribution of the data was assessed by applying the Kolmogorov–Smirnov test. Statistical analysis of data that followed a normal distribution was performed through a one-way analysis of variance (ANOVA) followed by Fisher’s protected least significant difference post hoc analysis, and results are reported as media ± standard error of the mean (Heaslip et al. 1994). Non-normally distributed data were analyzed by a nonparametric statistical analysis, and results are reported as medians and interquartile range (IQR). All statistical tests conducted were 2-sided and
Results
Vascular Reactivity Studies
All animals exhibited similar size of coronary and carotid arteries at the beginning of the study (Figure 1A). Arterial pressure and heart rate (assessed at the femoral artery after intracoronary administration of both vasoactive agents) were comparable among all 4 animal groups (Figure 1B). Intracoronary infusion of acetylcholine induced a dose-dependent vasodilatory response in all animals as shown in Figure 1C (10−8 to 10−6 M;
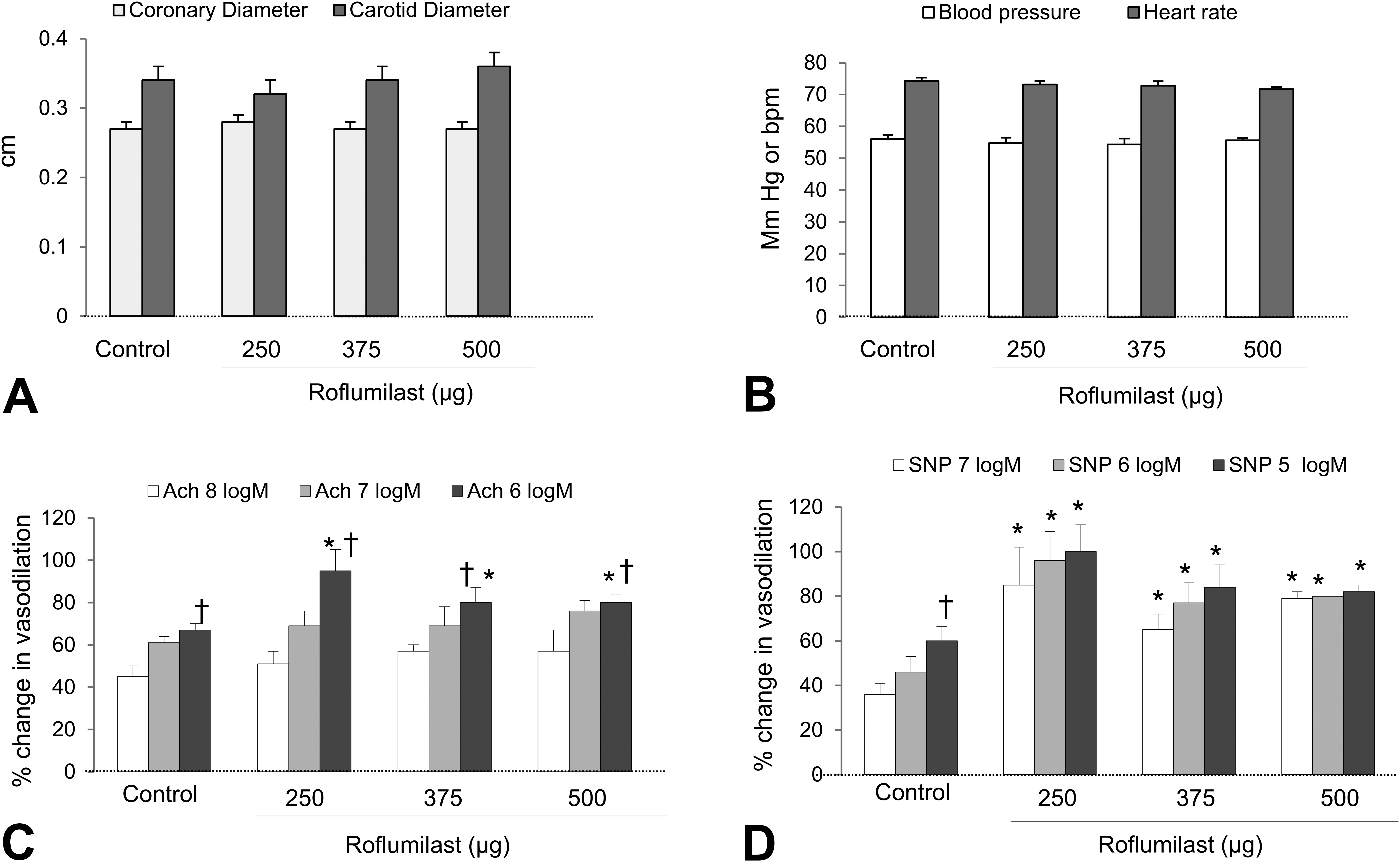
Vasoreactivity studies. Graphs showing coronary/carotid vessel diameters before flow probes implantation (A), hemodynamic parameters follow-up (B), relaxation response to acetylcholine (C), and sodium nitroprusside (SNP; D). Data are expressed as a percentage relaxation from baseline measurement.
Roflumilast Effects on the Vessel Wall
Immunohistochemical staining of vWF (Figure 2A) and fibrin (Figure 2B) was performed on the coronary artery (small vessel) and thoracic aorta (large vessel) of control and roflumilast-treated animals. Vessels of control animals showed a continuous and intense immunostaining for vWF along the endothelial layer (Figure 2A) whereas fibrin was barely detected (Figure 2B). A single dose of roflumilast 250 µg already induced a discontinued detection of endothelial vWF in both vessels (
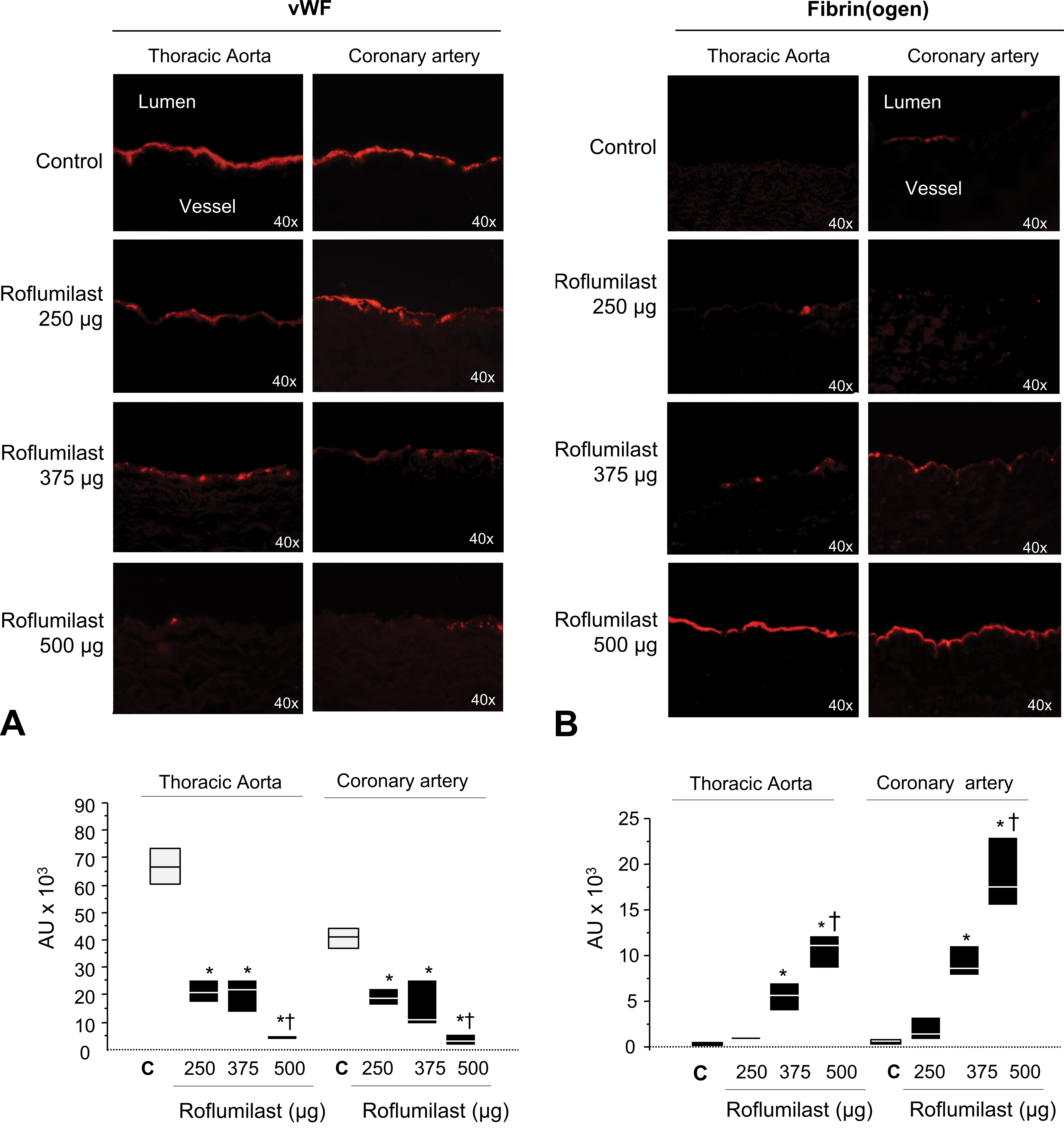
Assessment of vessel damage by immunohistochemistry. Immunohistochemical quantitation and representative images of von Willebrand factor (A) and fibrin(ogen) (B) deposition in the coronary and aortic vessels. *
Roflumilast Effect on IL-6 Release
No changes were observed in circulating IL-6 levels between control and roflumilast-treated animals at any tested dose indicating the absence of a systemic inflammatory response (Figure 3).
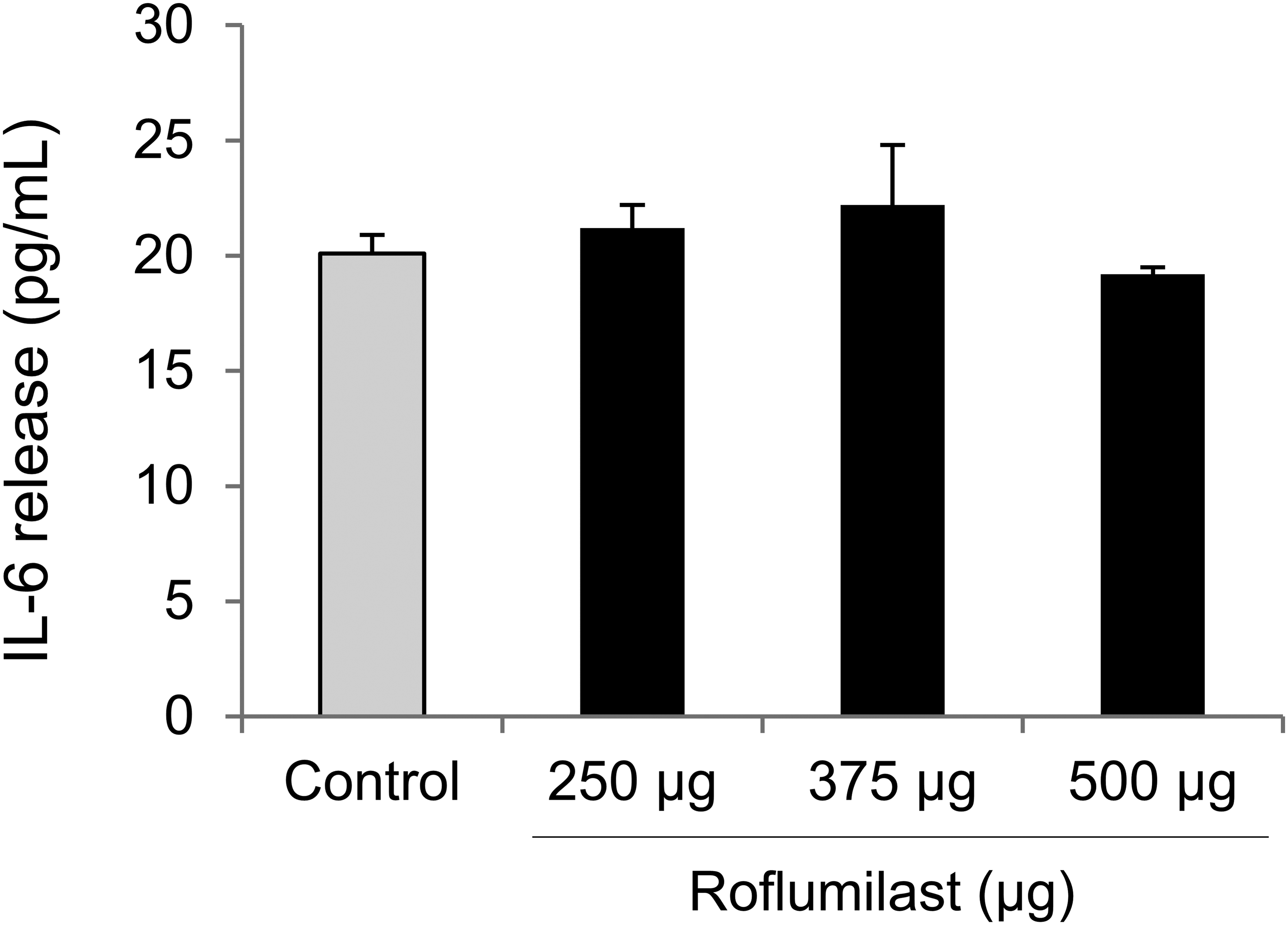
Interleukin (IL)-6 circulating levels.
Roflumilast Effect on cMPs Release
cMPs were identified at baseline and post-roflumilast administration by flow cytometry based on their light scattering properties (FSC and SSC) as events smaller than 1 μm. We have previously reported that the centrifugation conditions used for their isolation provide an optimal yield of microparticles (microvesicles; Suades et al. 2012). Cell and platelet contamination was negligible (less than 0.1%). cMP detection was comparable between control and roflumilast-treated animals at doses of 250 and 375 µg. However, roflumilast 500 µg was associated with a 2-fold increase in Annexin-V + MP (Figure 4A). Interestingly, analysis of surface markers for endothelial cells demonstrated a significant rise regarding the mean number of CD105+/CD45− endothelial-derived MPs in roflumilast-500-µg-treated animals as compared to controls (Figure 4B). There were no significant changes of CD61 + platelet-derived MPs.
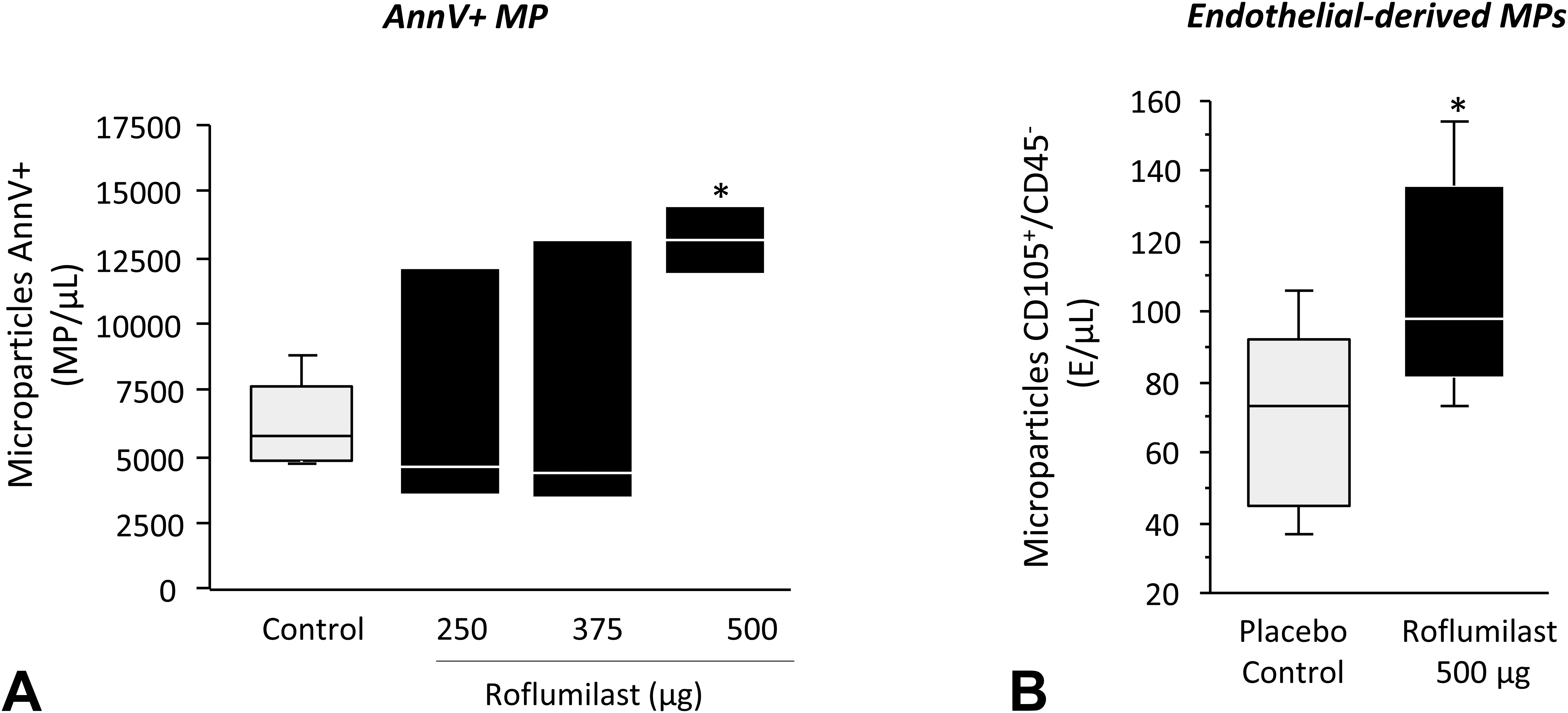
Analysis of circulating microparticles. (A) Annexin-V+ microparticle count per microliter of platelet-free plasma (PFP). (B) Total amount (per microliter of PFP) of endothelial microparticles. *
Roflumilast Effects on Swine Serum Proteome
After albumin and IgGs depletion, 475 protein spots were consistently detected in the 2-DE profile of serum samples on control and roflumilast-treated pigs (Figure 5A). There were significant changes in the serum proteome distribution profile after roflumilast treatment (Supplemental Table 1). The
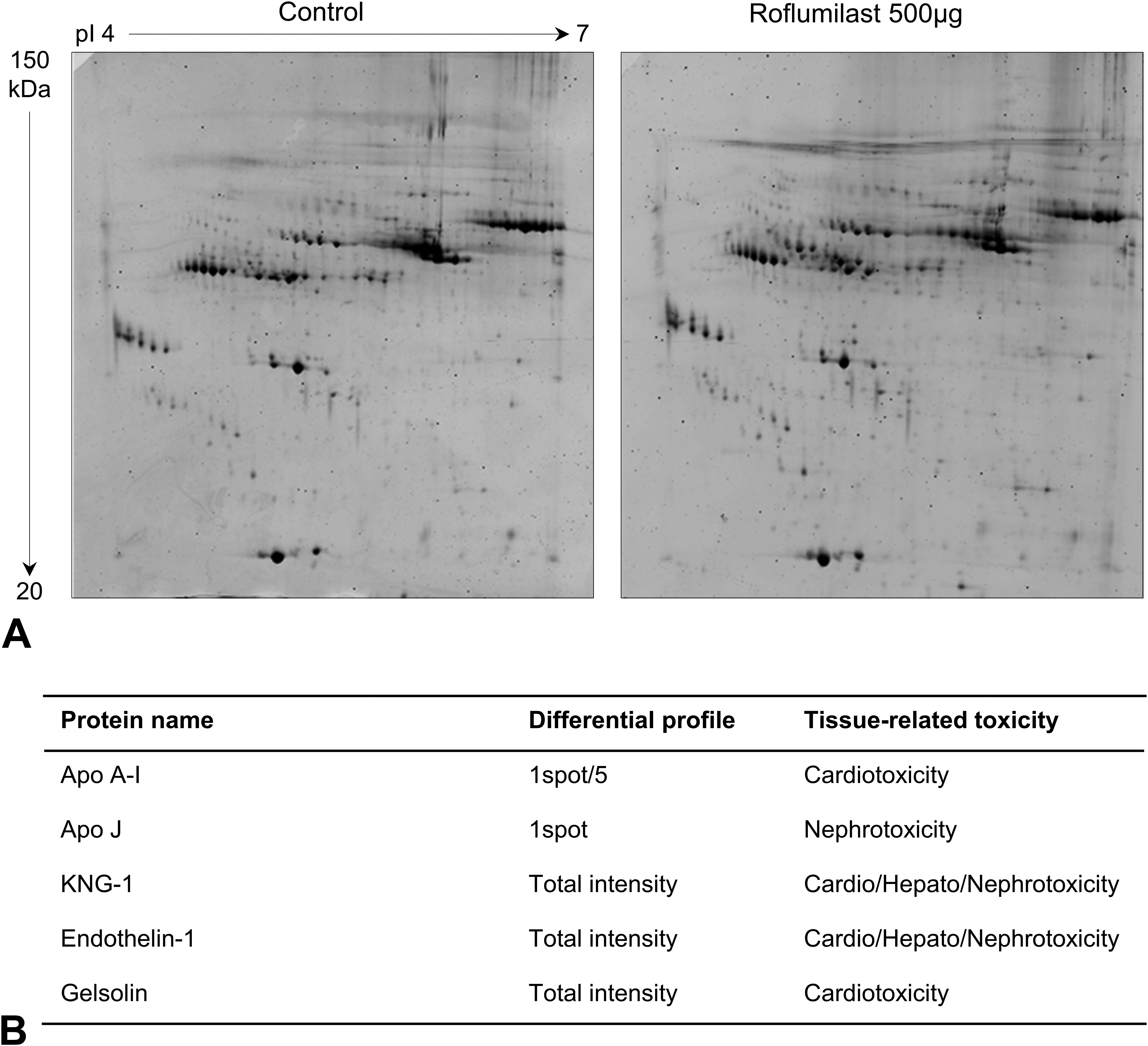
Differential proteomics. (A) Representative image of the 2-dimensional electrophoresis (2-DE) profile of albumin and IgGs depleted serum in both control animals and roflumilast 500 µg. (B) Table showing differentially expressed proteins between both groups and their specific tissue-related toxicity found in the ingenuity pathway analysis (IPA) analysis.
Within the differentially expressed proteins, 2 high-density lipoproteins (HDL)-related proteins were identified: apolipoprotein A-I (Apo A-I; SwissProt Number: P18648) and apolipoprotein J (Apo J; SwissProt Number: Q29549). Apo A-I was identified in both groups as a cluster of 5 spots with a molecular mass of 30 kDa and a Isoelectric point range between 5.1 and 5.7. The most basic spot showed a 2-fold decrease after roflumilast treatment (Figure 6A). Apo J was identified as a single spot of approximately 51 kDa and a Isoelectric point of 5.6 that was significantly decreased in the roflumilast group (
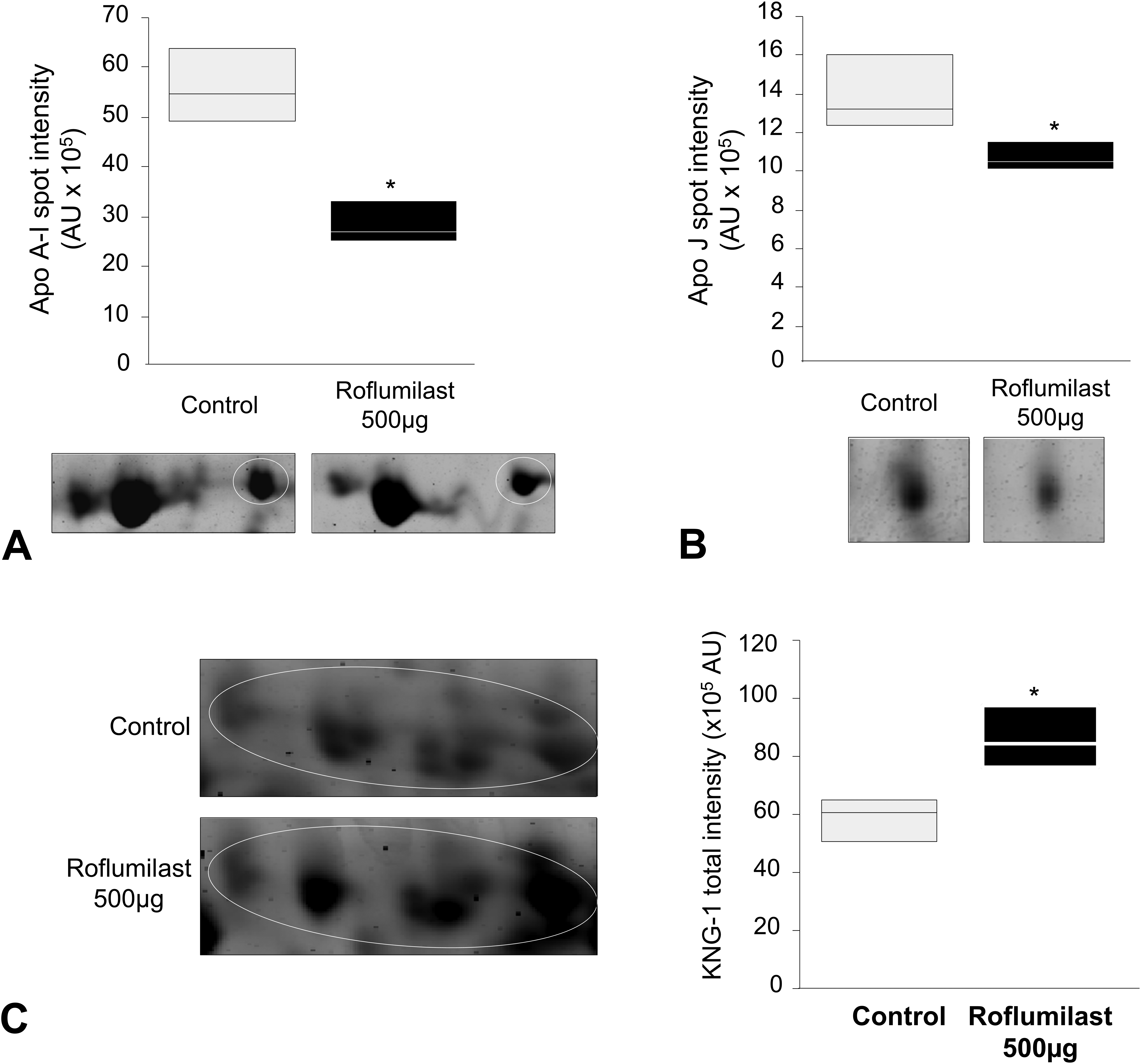
Analysis of protein changes I. Box plot diagram and representative image of the 2-dimensional electrophoresis (2-DE) profile showing the significant decrease in the intensity of (A) apolipoprotein (Apo) A-I and (B) Apo J spots in the roflumilast-500-µg-treated group. (C) Box plot diagram and representative image of the 2-dimensional electrophoresis (2-DE) profile showing the significant increase in kininogen-1 (KNG-1) total intensity in the roflumilast-500-µg-treated group. *
Kininogen-1 (KNG-1; Human SwissProt Number: P01042) was identified as a cluster of 8 spots with an apparent molecular mass of 70 kDa and a Isoelectric point of 6.0. Roflumilast-treated animals also showed a significant increase in total KNG-1 intensity (
Gelsolin (SwissProt Number: P20305) was identified as 4 spots, with an apparent molecular mass of 85 kDa and a Isoelectric point of 5.9, that were increased in the roflumilast group; the increase in the most basic forms was more evident (
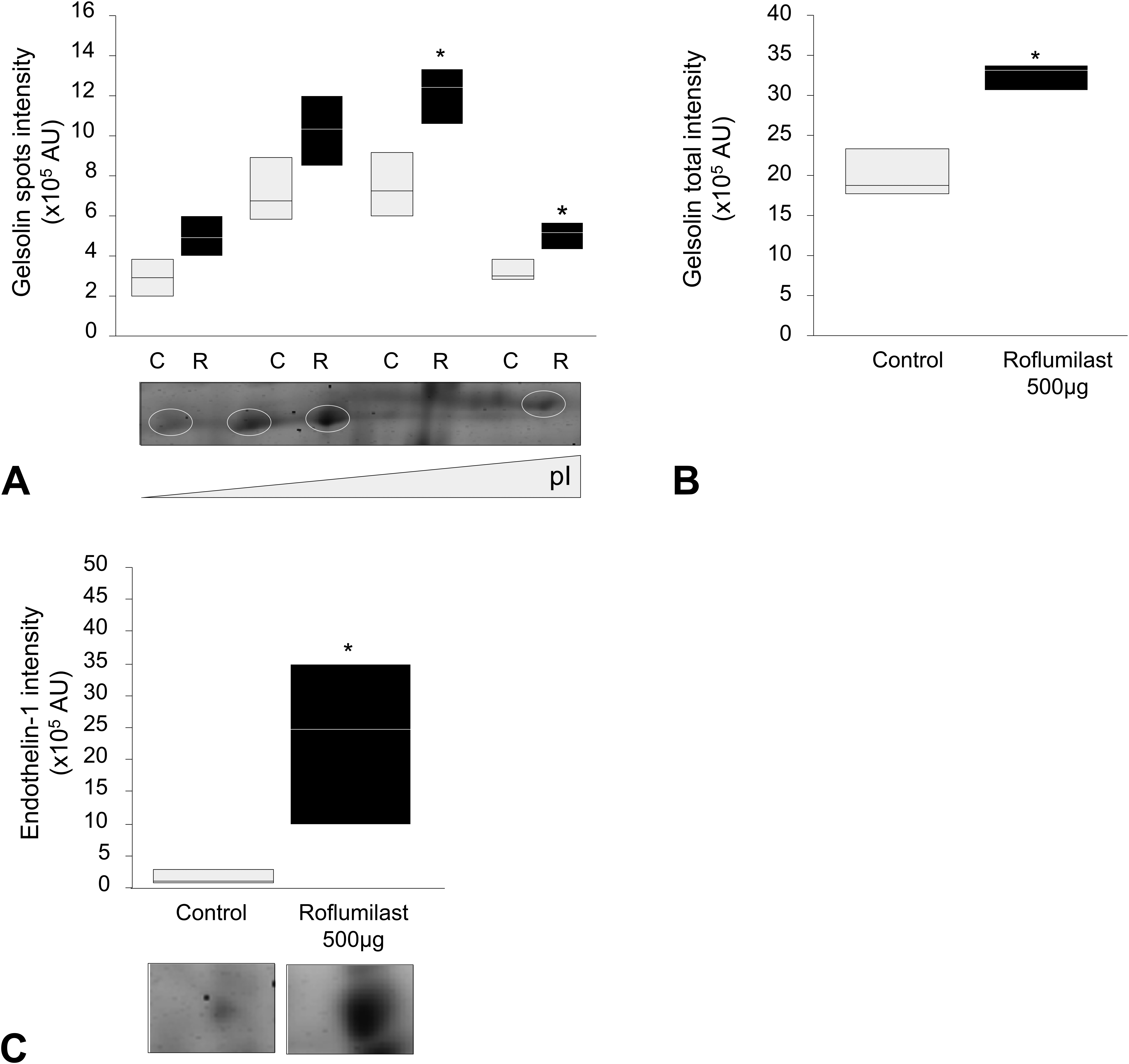
Analysis of protein changes II. Box plot diagram and representative image of the 2-dimensional electrophoresis (2-DE) profile showing the increase in (A) gelsolin spots and (B) its total intensity in the roflumilast-500-µg-treated group. (C) Box plot diagram and representative image of the 2-DE profile showing the significant increase in endothelin-1 spot in the roflumilast-500-µg-treated group. *
Discussion
The development of PDE-IV inhibitors has been hampered by the presence of DIVI in toxicological preclinical studies, and surrogate markers of DIVI are needed to detect vascular damage during toxicity testing. Vascular toxicity of PDE-IV inhibitors has been reported after prolonged dosing in animals and predominantly restricted to the mesentery of rodents and the coronary arteries of nonhuman primates (Losco, Evans, et al. 2004). We provide immunohistochemical characterization of acute dose-dependent injury in the vascular compartment (coronary and aortic beds) of swine after a single dose of roflumilast and provide compelling evidence of circulating surrogate markers—EMPs and different proteins (KNG-1, endothelin-1, gelsolin, Apo J, and ApoA-1)—as potential biomarkers for predicting early vascular drug toxicity.
First, we report that roflumilast elicits diffuse vascular injury in a dose-dependent manner within the first hours of drug administration. Furthermore, a single dose of 500 µg (dose used in the clinical setting for COPD treatment) is associated with endothelial cell loss, as evidenced by the absence of the endothelial marker vWF in both small (coronary artery) and large (thoracic aorta) vessels. Disruption of the endothelial integrity is also supported by the detection of a continuous thin layer of fibrin deposition. Taken together, these immunohistochemical findings sustain the presence of a dysfunctional/absent endothelium. Endothelial dysfunction has been associated with vascular constriction (Gutierrez et al. 2013). Yet, our
The mechanisms by which PDE-IV inhibitors induce vascular injury remain unknown. Studies in rats have postulated a direct pharmacological and/or chemical injury, changes in shear stress secondary to local vasodilation, and immunological and inflammatory reaction (Dagues et al. 2007; Larson et al. 1996). Regional coronary blood flow increase by several vasodilatory drugs, as reported herein, has shown to precede coronary vascular damage (Joseph 2000; Albassam, Smith, and Macallum 1998; Metz et al. 1991; Mesfin et al. 1989). However, we detect a significant rise in KNG-1. KNG-1 is an upstream mediator of the intrinsic coagulation pathway that triggers the kallikrein–kinin cascade, eventually liberating bradykinin, a potent vasoactive and pro-inflammatory peptide (Zhao et al. 2001). Hence, the kinin system is found in the interface between coagulation and activation of the immune system. In fact, we have recently associated KNG-1 activation with the detection of fibrin(ogen) fragments further supporting our observations (Cubedo et al. 2013). Yet, KNG-1 increase may also reflect an endothelial inflammatory response triggered by roflumilast, thereby contributing to perpetuate vascular injury. In fact, activation of the contact system of coagulation has been associated with the pathogenesis of autoimmune vasculitides (Kahn et al. 2002; Kahn and Karpman 2011). Proteomic analysis has also revealed an increase in endothelin-1 serum levels. This protein is a potent vasoconstrictor peptide released upon endothelial damage sustaining roflumilast-related DIVI (Kohan et al. 2011). As with KNG-1, we cannot overlook a possible pathogenic effect of endothelin-1 in vascular injury development since endothelin-1 has been also related to vasculitis (Iglarz and Clozel 2010, 2007). Gelsolin was also found to be differentially expressed in roflumilast-500-µg-treated animals. Gelsolin is an actin binding protein implicated in several cellular functions such as cell motility, dynamic rearrangement of the cytoskeletal architecture, and stimulation of programmed cell death (Kwiatkowski et al. 1989). Circulating gelsolin has been shown to be involved in the removal of actin released from damaged cells (Lee and Galbraith 1992). These properties make this protein a suitable biomarker for the detection of cell-to-cell disruption as occurs upon endothelial cell dislodgement. These findings expand on previous studies that reported increased levels of plasma gelsolin in amyloidosis and rhabdomyolysis (Lofberg et al. 1998). Our serum proteomic profiling analysis also reveals decreases in protein spots corresponding to 2 important HDL-related components, Apo A-I and Apo J (Jenne et al. 1991). It is well known that the vasoprotective properties associated with HDL particles occur mainly through increasing endothelial-related nitric oxide production (Besler et al. 2011). Therefore, the detected decrease in Apo A-I and Apo J, likely suggest a reduced protective effects of HDL on vascular cell damage. These apolipoproteins have also been related to vasculitis (Huang et al. 2010; Yu et al. 2010), with a direct implication of Apo J in preventing complement activation in specific types of vasculitides (Dauchel et al. 1993).
Finally, by flow cytometry analysis, we demonstrate a significant rise in both total and endothelial MPs (annexin V+ and CD105+ (endoglin)/CD45−) in roflumilast-administered animals. Though a consensus protocol to measure MPs does not yet exist, flow cytometry–based detection of MP has been shown to be a reliable and reproducible method (Piccin, Murphy, and Smith 2007). Endothelial MPs are increased in many vascular disorders (e.g., hypertension and diabetes mellitus) and autoimmune diseases, which are characterized by vascular damage and thrombosis (Koga et al. 2005). More recently, endothelial MPs have been found augmented in nondrug induced vasculitides (antineutrophil cytoplasmic antibody [ANCA], Kawasaki disease, Behcet’s disease, etc.; Brogan et al. 2004; Erdbruegger et al. 2008). Here, we provide evidence that endothelial MPs also serve as an early marker of endothelial damage upon drug treatment expanding their use in drug development.
Conclusions
We provide new candidate biomarkers that reflect the onset of acute vascular injury after administration of a clinically relevant dose of an approved PDE-IV inhibitor (roflumilast).
We cannot exclude that the herein presented proteins, besides serving as DIVI biomarkers, may also be effectors or mediators in triggering or exacerbating the vascular damage caused by the pharmacological compound. Nevertheless, they do reflect early DIVI. Translational validation studies in human trials are required to eventually qualify them as predictive biomarkers of DIVI.
Footnotes
Acknowledgments
P. Catalina, M. A. Canovas, S. Florit, and M. A. Velasco’s support with animal handling and care and for the proper conduct of the experimental and molecular work is gratefully and highly recognized. We thank Fundacion Jesus Serra, Barcelona, for their continuous support. G.V. is a “Ramon y Cajal” scientist funded by Spanish Ministry of Science and Innovation (MICINN).
Authors’ Note
Gemma Vilahur and Judit Cubedo contributed equally to the work.
Author Contribution
G. Vilahur contributed to design, analysis, and interpretation, and drafted the manuscript. J. Cubedo contributed to design, analysis, and interpretation, and drafted the manuscript. T. Padro contributed to analysis, interpretation, and drafted the manuscript. L. Casani contributed to acquisition, interpretation, and drafted the manuscript. O. Juan-Babot contributed to acquisition, interpretation, and drafted the manuscript. J. Crespo contributed to acquisition, analysis, and interpretation, and drafted the manuscript. K. Bendjama contributed to design and drafted the manuscript. M. Lawton contributed to design and drafted the manuscript. L. Badimon contributed to conception and design, and critically revised the manuscript.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was funded by the IMI–JU under SAFE-T (project Grant Agreement Nº 115003, IMI-JU), PNS-SAF2013-42962-R (to LB), PNS-SAF-2012-40208 (to GV) from the Spanish Ministry of Science and Innovation; Red de Investigación Cardiovascular (RIC) and TERCEL from Instituto Salud Carlos III (to LB).
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
